Perturbations in Osteogenic Cell Fate Following Exposure to Constituents Present in Tobacco: A Combinatorial Study
Abstract
1. Introduction
2. Methods
2.1. Cell Passaging and Maintenance
2.2. Osteogenic Differentiation of Human Embryonic Stem Cells
2.3. Chemical Exposure
2.4. Detection of Calcium
2.5. MTT Assay
2.6. Toxicity Forecaster Data Mining
2.7. ToxPI Pie Chart Generation
2.8. Statistical Analysis
3. Results
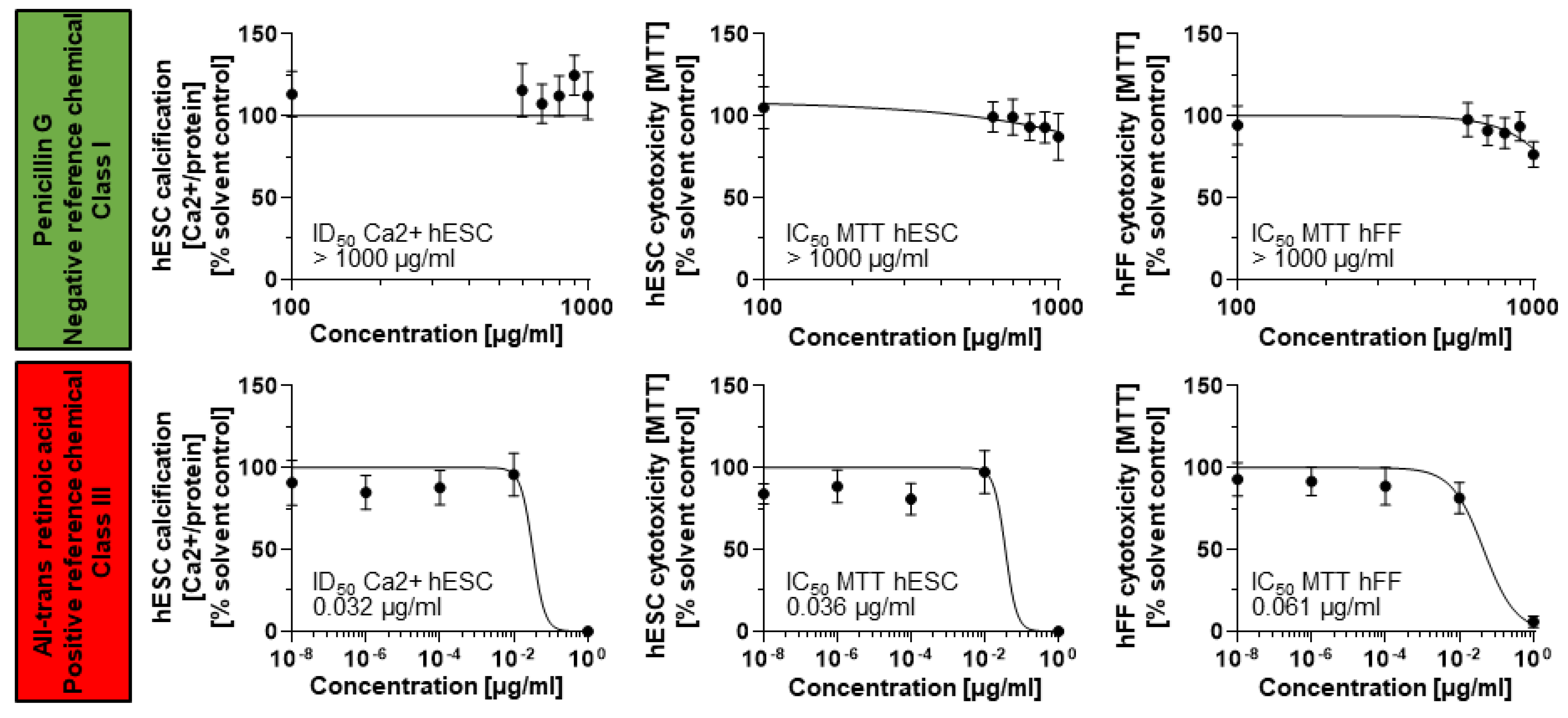
3.1. Dose–Response Curves Revealed Skeletal Embryotoxicity for ToxPI Positive Chemicals
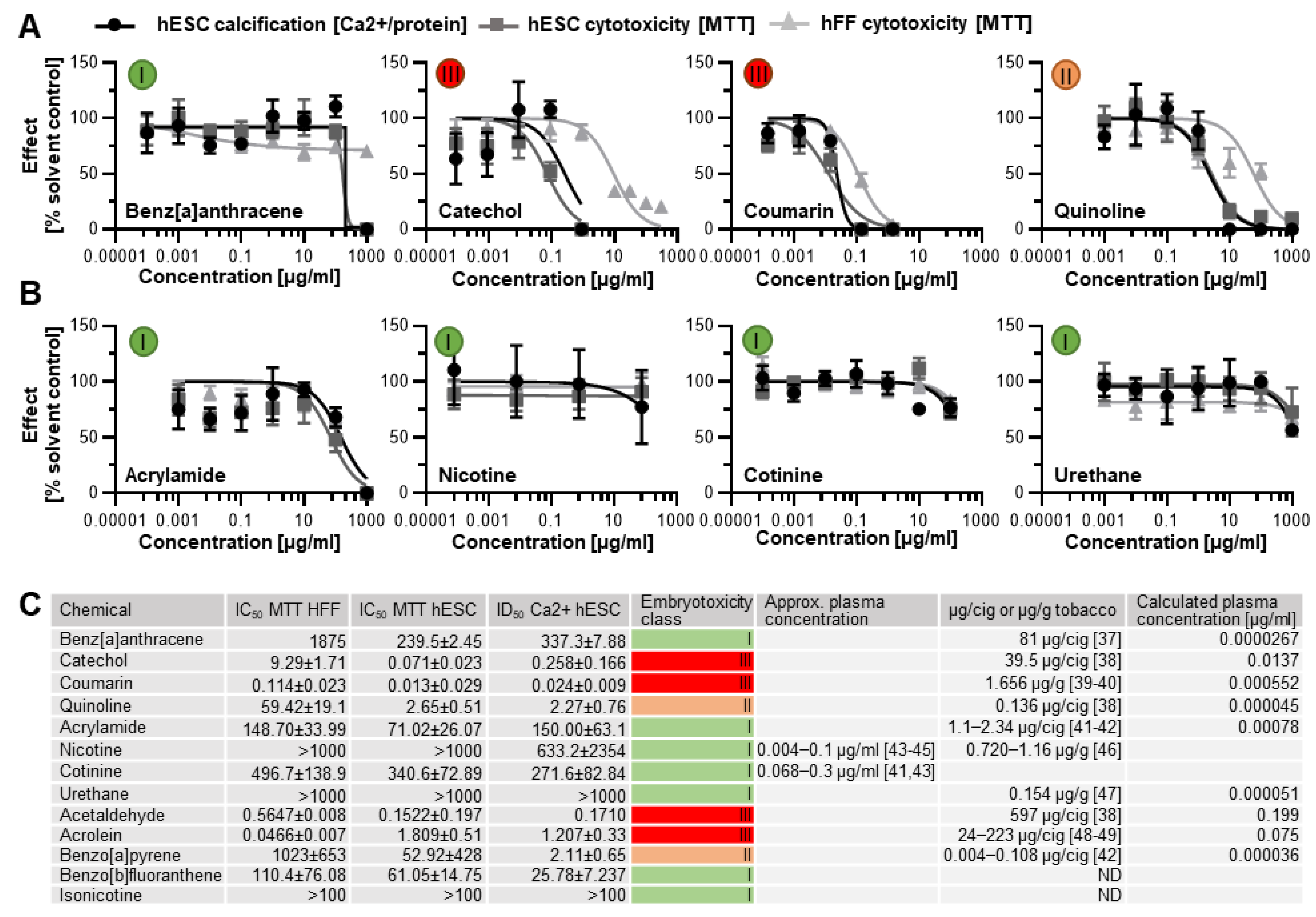
3.2. Human In Vitro Data for ToxPI Negative Chemicals
3.3. Screening of Additional Tobacco Constituents
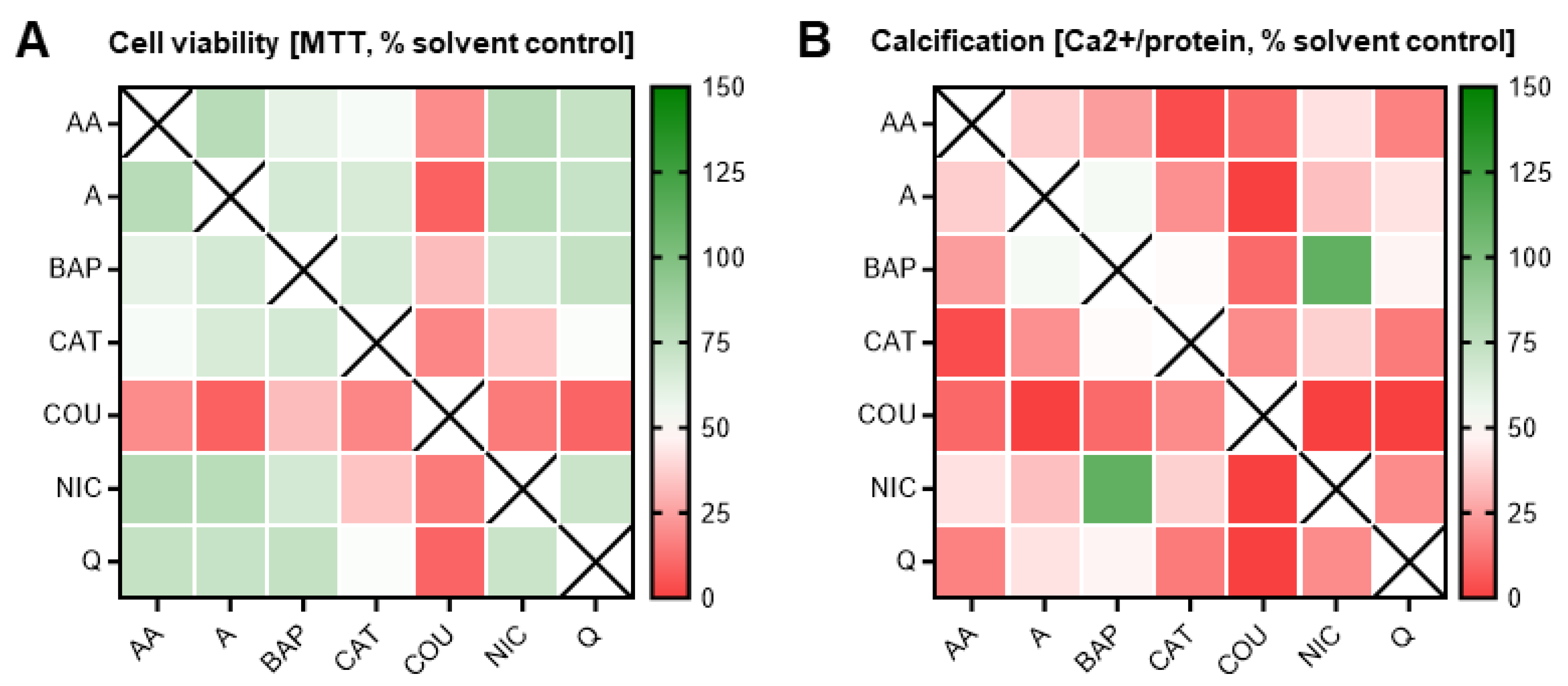
3.4. Double Combinatorial Exposures Elicit Apparent Synergistic or Antagonistic Effects
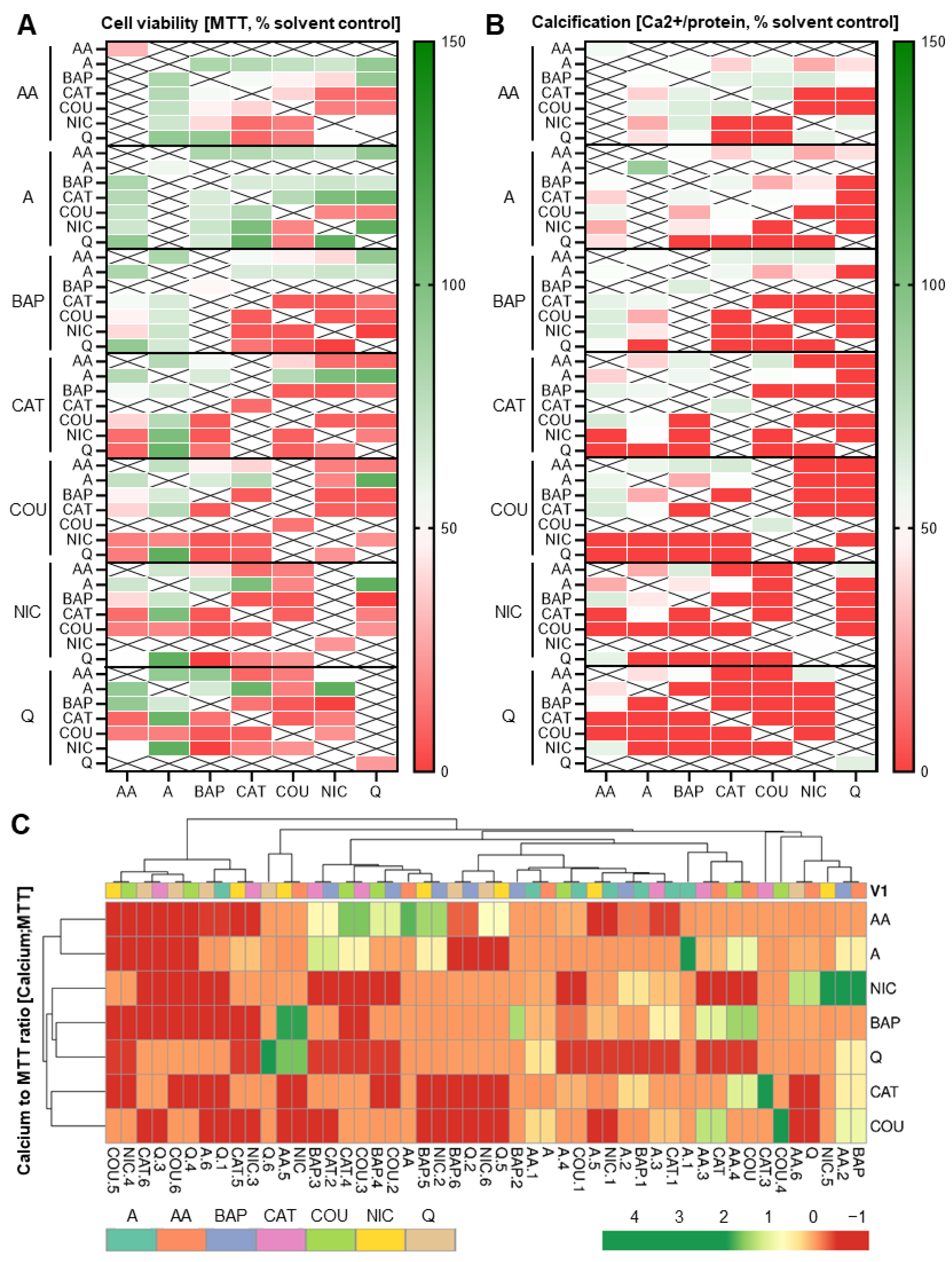
3.5. Addition of a Third Constituent Elicited Different Effects from Two-Constituent Combinations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Prüss-Ustün, A.; Wolf, J.; Corvalàn, C.; Bos, R.; Neira, M. Preventing Disease through Healthy Environments: A Global Assessment of the Environmental Burden of Disease; World Health Organization: Geneva, Switzerland, 2016; ISBN 9789241565196. [Google Scholar]
- National Clearinghouse for Smoking and Health, United States; Public Health Service; Office of the Surgeon General. The Health Consequences of Smoking, A Public Health Service Review. 1967. Available online: http://resource.nlm.nih.gov/101584932X157 (accessed on 19 September 2022).
- National Clearinghouse for Smoking and Health, United States; Public Health Service; Office of the Surgeon General. The Health Consequences of Smoking: 1969 Supplement to the 1967 Public Health Service Review. 1969. Available online: http://resource.nlm.nih.gov/101584932X174 (accessed on 19 September 2022).
- Office of the Surgeon General (US); Office on Smoking and Health (US). The Health Consequences of Smoking: A Report of the Surgeon General; Centers for Disease Control and Prevention (US): Atlanta, GA, USA, 2004. [Google Scholar]
- Musk, A.W.; de Klerk, N.H. History of tobacco and health. Respirology 2003, 8, 286–290. [Google Scholar] [CrossRef]
- English, D.R.; Holman, C.D.J.; Milne, E.; Winter, M.G.; Hulse, G.; Codde, J.P.; Bower, C.I.; Corti, B.; De Klerk, N.; Knuiman, M.W.; et al. The Quantification of Drug Caused Morbidity and Mortality in Australia, 1995th ed.; Commonwealth Department of Human Services and Health: Canberra, AU, USA, 1995. [Google Scholar]
- Ward, K.D.; Klesges, R.C. A meta-analysis of the effects of cigarette smoking on bone mineral density. Calcif. Tissue Int. 2001, 68, 259–270. [Google Scholar] [CrossRef]
- Smoking and Bone Health. Retrieved from National Institute of Arthritis and Musculoskeletal and Skin Diseases. NIH Publication No. 18–7883. May 2016. Available online: https://www.bones.nih.gov/sites/bones/files/pdfs/smokingbonehealth-508–12–18.pdf (accessed on 19 September 2022).
- Curtin, S.C.; Matthews, T.J. Smoking Prevalence and Cessation Before and During Pregnancy: Data from the Birth Certificate, 2014. Natl. Vital. Stat. Rep. 2016, 65, 1–14. [Google Scholar]
- Wisborg, K.; Kesmodel, U.; Henriksen, T.B.; Olsen, S.F.; Secher, N.J. Exposure to tobacco smoke in utero and the risk of stillbirth and death in the first year of life. Am. J. Epidemiol. 2001, 154, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Werler, M.M. Teratogen update: Smoking and reproductive outcomes. Teratology 1997, 55, 382–388. [Google Scholar] [CrossRef]
- Malik, S.; Cleves, M.A.; Honein, M.A.; Romitti, P.A.; Botto, L.D.; Yang, S.; Hobbs, C.A.; National Birth Defects Prevention Study. Maternal smoking and congenital heart defects. Pediatrics 2008, 121, e810–e816. [Google Scholar] [CrossRef] [PubMed]
- Macdonald-Wallis, C.; Tobias, J.H.; Davey Smith, G.; Lawlor, D.A. Parental smoking during pregnancy and offspring bone mass at age 10 years: Findings from a prospective birth cohort. Osteoporos. Int. 2011, 22, 1809–1819. [Google Scholar] [CrossRef]
- Jones, G.; Riley, M.; Dwyer, T. Maternal smoking during pregnancy, growth, and bone mass in prepubertal children. J. Bone Miner. Res. 1999, 14, 146–151. [Google Scholar] [CrossRef]
- Godfrey, K.; Walker-Bone, K.; Robinson, S.; Taylor, P.; Shore, S.; Wheeler, T.; Cooper, C. Neonatal bone mass: Influence of parental birthweight, maternal smoking, body composition, and activity during pregnancy. J. Bone Miner. Res. 2001, 16, 1694–1703. [Google Scholar] [CrossRef]
- Harvey, N.C.; Javaid, M.K.; Arden, N.K.; Poole, J.R.; Crozier, S.R.; Robinson, S.M.; Inskip, H.M.; Godfrey, K.M.; Dennison, E.M.; Cooper, C. SWS Study Team Maternal predictors of neonatal bone size and geometry: The Southampton Women’s Survey. J. Dev. Orig. Health Dis. 2010, 1, 35–41. [Google Scholar] [CrossRef]
- Parviainen, R.; Auvinen, J.; Pokka, T.; Serlo, W.; Sinikumpu, J.J. Maternal smoking during pregnancy is associated with childhood bone fractures in offspring—A birth-cohort study of 6718 children. Bone 2017, 101, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Karmach, O.; Madrid, J.V.; Dasgupta, S.; Volz, D.C.; zur Nieden, N.I. Embryonic Exposure to Cigarette Smoke Extract Impedes Skeletal Development and Evokes Craniofacial Defects in Zebrafish. Int. J. Mol. Sci. 2022, 23, 9904. [Google Scholar] [CrossRef] [PubMed]
- Hatsukami, D.K.; Carroll, D.M. Tobacco harm reduction: Past history, current controversies and a proposed approach for the future. Prev. Med. 2020, 140, 106099. [Google Scholar] [CrossRef] [PubMed]
- FDA News Release. FDA Grants First-Ever Modified Risk Orders to Eight Smokeless Tobacco Products. FDA Concludes Completely Switching from Cigarettes to These Authorized Products Lowers Certain Health Risks. Available online: https://www.fda.gov/news-events/press-announcements/fda-grants-first-ever-modified-risk-orders-eight-smokeless-tobacco-products (accessed on 19 September 2022).
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique; Universities Federation for Animal Welfare: Wheathampstead, UK, 1959; as reprinted 1992. [Google Scholar]
- Tannenbaum, J.; Bennett, B.T. Russell and Burch’s 3Rs then and now: The need for clarity in definition and purpose. J. Am. Assoc. Lab. Anim. Sci. 2015, 54, 120–132. [Google Scholar] [PubMed]
- Spielmann, H.; Pohl, I.; Döring, B.; Liebsch, M.; Moldenhauer, F. The embryonic stem cell test (EST), an in vitro embryotoxicity test using two permanent mouse cell lines: 3T3 fibroblasts and embryonic stem cells. Vitr. Toxicol. 1997, 10, 119–127. [Google Scholar]
- Genschow, E.; Scholz, G.; Brown, N.; Piersma, A.; Brady, M.; Clemann, N.; Huuskonen, H.; Paillard, F.; Bremer, S.; Becker, K.; et al. Development of prediction models for three in vitro embryotoxicity tests in an ECVAM validation study. Vitr. Mol. Toxicol. 2000, 13, 51–66. [Google Scholar]
- zur Nieden, N.I.; Kempka, G.; Ahr, H.J. Multiple endpoint embryonic stem cell test—A possible approach to test for the teratogenic potential of compounds. Toxicol. Appl. Pharmacol. 2004, 194, 257–269. [Google Scholar] [CrossRef]
- zur Nieden, N.I.; Davis, L.A.; Rancourt, D.E. Comparing three novel endpoints for developmental osteotoxicity in the embryonic stem cell test. Toxicol. Appl. Pharmacol. 2010, 247, 91–97. [Google Scholar] [CrossRef]
- zur Nieden, N.I.; Davis, L.A.; Rancourt, D.E. Monolayer cultivation of osteoprogenitors shortens duration of the embryonic stem cell test while reliably predicting developmental osteotoxicity. Toxicology 2010, 277, 66–73. [Google Scholar] [CrossRef]
- Walker, L.; Baumgartner, L.; Ast, J.; Keller, K.C.; Trettner, S.; zur Nieden, N.I. Non-human primate and rodent embryonic stem cells are differentially sensitive to teratogens. Tox. Rep. 2015, 2, 165–174. [Google Scholar] [CrossRef]
- Martinez, I.K.C.; Sparks, N.R.L.; Madrid, J.V.; Talbot, P.; zur Nieden, N.I. Exposure to cigarette smoke impedes human osteoblast differentiation independently of nicotine. Nicotine Tob. Res. 2022, 24, 1921–1926. [Google Scholar] [CrossRef]
- Martinez, I.K.C.; Madrid, J.V.; Sparks, N.R.L.; Affeldt, H., III; Vera-Colon, M.V.; Bhanu, B.; zur Nieden, N.I. Video-based kinetic analysis of calcification in live osteogenic human embryonic stem cell cultures reveals the developmentally toxic effect of Snus tobacco extract. Toxicol. Appl. Pharmacol. 2019, 363, 111–121. [Google Scholar] [CrossRef]
- Reif, D.M.; Martin, M.T.; Tan, S.W.; Houck, K.A.; Judson, R.S.; Richard, A.M.; Knudsen, T.B.; Dix, D.J.; Kavlock, R.J. Endocrine profiling and prioritization of environmental chemicals using ToxCast data. Environ. Health Perspect. 2010, 118, 1714–1720. [Google Scholar] [CrossRef] [PubMed]
- Sparks, N.R.L.; Martinez, I.K.C.; Soto, C.H.; zur Nieden, N.I. Low osteogenic yield in human pluripotent stem cells associates with differential methylation of neural crest associated promoters. Stem Cells 2018, 36, 349–362. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.A.; Dienelt, A.; zur Nieden, N.I. Absorption-based assays for the analysis of osteogenic and chondrogenic yield. Methods Mol. Biol. 2011, 690, 255–272. [Google Scholar] [CrossRef] [PubMed]
- zur Nieden, N.I.; Baumgartner, L. Assessing developmental osteotoxicity of chlorides in the embryonic stem cell test. Reprod. Toxicol. 2010, 30, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.M.; Sparks, N.R.L.; Puig-Sanvicens, V.; Rodrigues, B.; zur Nieden, N.I. An Evaluation of Human Induced Pluripotent Stem Cells to Test for Cardiac Developmental Toxicity. Int. J. Mol. Sci. 2021, 22, 8114. [Google Scholar] [CrossRef]
- Metsalu, T.; Vilo, J. ClustVis: A web tool for visualizing clustering of multivariate data using Principal Component Analysis and heatmap. Nucleic. Acids Res. 2015, 43, W566–W570. [Google Scholar] [CrossRef]
- Hoffmann, D.; Sanghvi, L.D.; Wynder, E.L. Comparative chemical analysis of Indian bidi and american cigarette smoke. Int. J. Cancer 1974, 14, 49–53. [Google Scholar] [CrossRef]
- Jaccard, G.; Djoko, D.T.; Korneliou, A.; Stabbert, R.; Belushkin, M.; Esposito, M. Mainstream smoke constituents and in vitro toxicity comparative analysis of 3R4F and 1R6F reference cigarettes. Toxicol. Rep. 2019, 6, 222–231. [Google Scholar] [CrossRef]
- Christakopoulos, A.; Feldhusen, K.; Norin, H.; Palmqvist, A.; Wahlberg, I. Determination of natural levels of coumarin in different types of tobacco using a mass fragmentographic method. J. Agric. Food Chem. 1992, 40, 1358–1361. [Google Scholar] [CrossRef]
- McAdam, K.; Enos, T.; Goss, C.; Kimpton, H.; Faizi, A.; Edwards, S.; Wright, C.; Porter, A.; Rodu, B. Analysis of coumarin and angelica lactones in smokeless tobacco products. Chem. Cent. J. 2018, 12, 142. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, X.; Shi, B.; Yang, Z.; Luo, Y.; Xu, T.; Liu, D.; Jiang, C.; Du, G.; Lu, N.; et al. Investigation of exposure biomarkers in human plasma following differing levels of tobacco-specific N-nitrosamines and nicotine in cigarette smoke. Environ. Res. 2022, 214 Pt 2, 113811. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.J.; Perfetti, T.A.; Rumple, M.A.; Rodgman, A.; Doolittle, D.J. “IARC group 2A Carcinogens” reported in cigarette mainstream smoke. Food Chem. Toxicol. 2000, 38, 371–383. [Google Scholar] [CrossRef] [PubMed]
- Benowitz, N.L.; Hansson, A.; Jacob, P., 3rd. Cardiovascular effects of nasal and transdermal nicotine and cigarette smoking. Hypertension 2002, 39, 1107–1112. [Google Scholar] [CrossRef]
- Russell, M.A.; Feyerabend, C. Blood and urinary nicotine in nonsmokers. Lancet 1975, 1, 179–181. [Google Scholar] [CrossRef] [PubMed]
- Dhar, P. Measuring tobacco smoke exposure: Quantifying nicotine/ cotinine concentration in biological samples by colorimetry, chromatography and immunoassay methods. J. Pharm. Biomed. Anal. 2004, 35, 155–168. [Google Scholar] [CrossRef]
- Andersson, G.; Vala, E.K.; Curvall, M. The influence of cigarette consumption and smoking machine yields of tar and nicotine on the nicotine uptake and oral mucosal lesions in smokers. J. Oral. Pathol. Med. 1997, 26, 117–123. [Google Scholar] [CrossRef]
- McAdam, K.; Vas, C.; Kimpton, H.; Faizi, A.; Liu, C.; Porter, A.; Synnerdahl, T.; Karlsson, P.; Rodu, B. Ethyl carbamate in Swedish and American smokeless tobacco products and some factors affecting its concentration. Chem. Cent. J. 2018, 12, 86. [Google Scholar] [CrossRef]
- Talhout, R.; Opperhuizen, A.; van Amsterdam, J.G. Sugars as tobacco ingredient: Effects on mainstream smoke composition. Food Chem. Toxicol. 2006, 44, 1789–1798. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Acrolein, Crotonaldehyde, and Arecoline: IARC Monographs on the Identification of Carcinogenic Hazards to Humans; International Agency for Research on Cancer: Lyon, France, 2021; Volume 128. [Google Scholar]
- Giuliani, N.; Girasole, G.; Vescovi, P.P.; Passeri, G.; Pedrazzoni, M. Ethanol and acetaldehyde inhibit the formation of early osteoblast progenitors in murine and human bone marrow cultures. Alcohol. Clin. Exp. Res. 1999, 23, 381–385. [Google Scholar] [CrossRef]
- Reimers, M.J.; Flockton, A.R.; Tanguay, R.L. Ethanol- and acetaldehyde-mediated developmental toxicity in zebrafish. Neurotoxicol. Teratol. 2004, 26, 769–781. [Google Scholar] [CrossRef]
- Lee, L.L.; Lee, J.S.; Waldman, S.D.; Casper, R.F.; Grynpas, M.D. Polycyclic aromatic hydrocarbons present in cigarette smoke cause bone loss in an ovariectomized rat model. Bone 2002, 30, 917–923. [Google Scholar] [CrossRef]
- Kung, M.H.; Yukata, K.; O’Keefe, R.J.; Zuscik, M.J. Aryl hydrocarbon receptor-mediated impairment of chondrogenesis and fracture healing by cigarette smoke and benzo(a)pyrene. J. Cell Physiol. 2012, 227, 1062–1070. [Google Scholar] [CrossRef]
- Garland, M.A.; Geier, M.C.; Bugel, S.M.; Shankar, P.; Dunham, C.L.; Brown, J.M.; Tilton, S.C.; Tanguay, R.L. Aryl hydrocarbon receptor mediates larval zebrafish fin duplication following exposure to benzofluoranthenes. Toxicol. Sci. 2020, 176, 46–64. [Google Scholar] [CrossRef]
- Le Bihanic, F.; Sommard, V.; de Perrine, L.; Pichon, A.; Grasset, J.; Berrada, S.; Budzinski, H.; Cousin, X.; Morin, B.; Cachot, J. Environmental concentrations of benz[a]anthracene induce developmental defects and DNA damage and impair photomotor response in Japanese medaka larvae. Ecotoxicol. Environ. Saf. 2015, 113, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Levy, S.; Papadopoulo, D.; Nocentini, S.; Chamaillard, L.; Beesau, O.; Hubert-Habart, M.; Markovits, P. Transformation of hamster embryo cells by 7-methyl benz(a)anthracene in vitro: Tumorigenicity, chromosome pattern and spheroid formation. Eur. J. Cancer 1976, 12, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Warshawsky, D.; Livingston, G.K.; Fonouni-Fard, M.; LaDow, K. Induction of micronuclei and sister chromatid exchanges by polycyclic and N-heterocyclic aromatic hydrocarbons in cultured human lymphocytes. Environ. Mol. Mutagen. 1995, 26, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Wells, P.G.; Winn, L.M. Biochemical toxicology of chemical teratogenesis. Crit. Rev. Biochem. Mol. Biol. 1996, 31, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Sparks, N.R.L.; zur Nieden, N.I. Pluripotent Stem Cells as Tools to Assess Developmental Toxicity: Diversity Instead of Consolidation. In Handbook of Nanotoxicology, Nanomedicine and Stem Cell Use in Toxicology; Sahu, S.C., Casciano, D.A., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2014; pp. 299–308. [Google Scholar] [CrossRef]
- Kühnert, A.; Vogs, C.; Seiwert, B.; Aulhorn, S.; Altenburger, R.; Hollert, H.; Küster, E.; Busch, W. Biotransformation in the zebrafish embryo -temporal gene transcription changes of cytochrome P450 enzymes and internal exposure dynamics of the AhR binding xenobiotic benz[a]anthracene. Environ. Pollut. 2017, 230, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Buening, M.K.; Wislocki, P.G.; Levin, W.; Yagi, H.; Thakker, D.R.; Akagi, H.; Koreeda, M.; Jerina, D.M.; Conney, A.H. Tumorigenicity of the optical enantiomers of the diastereomeric benzo[a]pyrene 7,8-diol-9,10-epoxides in newborn mice: Exceptional activity of (+)-7beta,8alpha-dihydroxy-9alpha,10alpha-epoxy-7,8,9,10-tetrahydrobenzo[a]pyrene. Proc. Natl. Acad. Sci. USA 1978, 75, 5358–5361. [Google Scholar] [CrossRef]
- Gelboin, H.V. Benzo[alpha]pyrene metabolism, activation and carcinogenesis: Role and regulation of mixed-function oxidases and related enzymes. Physiol. Rev. 1980, 60, 1107–1166. [Google Scholar] [CrossRef]
- Shum, S.; Jensen, N.M.; Nebert, D.W. The murine Ah locus: In utero toxicity and teratogenesis associated with genetic differences in benzo[a]pyrene metabolism. Teratology 1979, 20, 365–376. [Google Scholar] [CrossRef]
- Pelkonen, O.; Kaltiala, E.H.; Larmi, T.K.; Kärki, N.T. Cytochrome P-450-linked monooxygenase system and drug-induced spectral interactions in human liver microsomes. Chem. Biol. Interact. 1974, 9, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Fort, D.J.; Stover, E.L.; Propst, T.; Hull, M.A.; Bantle, J.A. Evaluation of the developmental toxicities of coumarin, 4-hydroxycoumarin, and 7-hydroxycoumarin using FETAX. Drug Chem. Toxicol. 1998, 21, 15–26. [Google Scholar] [CrossRef]
- Weigt, S.; Huebler, N.; Strecker, R.; Braunbeck, T.; Broschard, T.H. Developmental effects of coumarin and the anticoagulant coumarin derivative warfarin on zebrafish (Danio rerio) embryos. Reprod. Toxicol. 2012, 33, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.R.; Schultz, T.W.; Dumont, J.N. Toxic and teratogenic effects of selected aromatic amines on embryos of the amphibian Xenopus laevis. Arch. Environ. Contam. Toxicol. 1981, 10, 371–391. [Google Scholar] [CrossRef] [PubMed]
- Black, J.A.; Birge, W.J.; Westerman, A.G.; Francis, P.C. Comparative aquatic toxicology of aromatic hydrocarbons. Fundam. Appl. Toxicol. 1983, 3, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Taysse, L.; Troutaud, D.; Khan, N.; Deschaux, P. Structure-activity relationship of phenolic compounds (phenol, pyrocatechol and hydroquinone) on natural lymphocytotoxicity of carp (Cyprinus carpio). Toxicology 1995, 98, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, T.; Hayashi, N.; Maizumi, H.; Huff, J.; Barrett, J.C. Benzene-, catechol-, hydroquinone- and phenol-induced cell transformation, gene mutations, chromosome aberrations, aneuploidy, sister chromatid exchanges and unscheduled DNA synthesis in Syrian hamster embryo cells. Mutat. Res. 1997, 373, 113–123. [Google Scholar] [CrossRef]
- McCue, J.M.; Lazis, S.; Cohen, J.J.; Modiano, J.F.; Freed, B.M. Hydroquinone and catechol interfere with T cell cycle entry and progression through the G1 phase. Mol. Immunol. 2003, 39, 995–1001. [Google Scholar] [CrossRef]
- Goodale, B.C.; Tilton, S.C.; Corvi, M.M.; Wilson, G.R.; Janszen, D.B.; Anderson, K.A.; Waters, K.M.; Tanguay, R.L. Structurally distinct polycyclic aromatic hydrocarbons induce differential transcriptional responses in developing zebrafish. Toxicol. Appl. Pharmacol. 2013, 272, 656–670. [Google Scholar] [CrossRef]
- Draths, K.M.; Frost, J.W. Conversion of D-glucose into catechol: The not-so-common pathway of aromatic biosynthesis. J. Am. Chem. Soc. 1991, 113, 9361–9363. [Google Scholar] [CrossRef]
- Del Signore, A.; Romeo, F.; Giaccio, M. Content of phenolic substances in basidiomycetes. Mycol. Res. 1997, 101, 552–556. [Google Scholar] [CrossRef]
- Choi, H.S.; Kim, J.; Kim, S.L.; Deng, H.Y.; Lee, D.; Kim, C.S.; Yun, B.S.; Lee, D.S. Catechol derived from aronia juice through lactic acid bacteria fermentation inhibits breast cancer stem cell formation via modulation Stat3/IL-6 signaling pathway. Mol. Carcinog. 2018, 57, 1467–1479. [Google Scholar] [CrossRef] [PubMed]
- Bansal, P.; Bhanjana, G.; Prabhakar, N.; Dhau, J.S.; Chaudhary, G.R. Electrochemical sensor based on ZrO2 NPs/Au electrode sensing layer for monitoring hydrazine and catechol in real water samples. J. Mol. Liq. 2017, 248, 651–657. [Google Scholar] [CrossRef]
- Chapman, D.E.; Namkung, M.J.; Juchau, M.R. Benzene and benzene metabolites as embryotoxic agents: Effects on cultured rat embryos. Toxicol. Appl. Pharmacol. 1994, 128, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Barclay, L.R.C.; Edwards, C.E.; Vinqvist, M.R. Media effects on antioxidant activities of phenols and catechols. J. Am. Chem. Soc. 1999, 121, 6226–6231. [Google Scholar] [CrossRef]
- Tejero, N.; Gonzalez-García, A.; Gonzalez-Lafont, A.; Lluch, J.M. Tunneling in green tea: Understanding the antioxidant activity of catechol-containing compounds. A variational transition-state theory study. J. Am. Chem. Soc. 2007, 129, 5846–5854. [Google Scholar] [CrossRef] [PubMed]
- Nachar, A.; Eid, H.M.; Vinqvist-Tymchuk, M.; Vuong, T.; Kalt, W.; Matar, C.; Haddad, P.S. Phenolic compounds isolated from fermented blueberry juice decrease hepatocellular glucose output and enhance muscle glucose uptake in cultured murine and human cells. BMC Complement. Altern. Med. 2017, 17, 138. [Google Scholar] [CrossRef]
- Zhao, Y.; Fang, C.; Jin, C.; Bao, Z.; Yang, G.; Jin, Y. Catechin from green tea had the potential to decrease the chlorpyrifos induced oxidative stress in larval zebrafish (Danio rerio). Pestic. Biochem. Physiol. 2022, 182, 105028. [Google Scholar] [CrossRef]
- Knezevic, S.; Ghafoor, A.; Mehri, S.; Barazi, A.; Dziura, M.; Trant, J.F.; Dieni, C.A. Catechin and other catechol-containing secondary metabolites: Bacterial biotransformation and regulation of carbohydrate metabolism. PharmaNutrition 2021, 17, 100273. [Google Scholar] [CrossRef]
- Alafiatayo, A.A.; Lai, K.S.; Syahida, A.; Mahmood, M.; Shaharuddin, N.A. Phytochemical evaluation, embryotoxicity, and teratogenic effects of curcuma longa extract on zebrafish (Danio rerio). Evid.-Based Complement. Alternat. Med. 2019, 2019, 3807207. [Google Scholar] [CrossRef]
- Sparks, N.R.L.; Walker, L.M.; Sera, S.R.; Madrid, J.V.; Hanna, M.; Dominguez, E.C.; zur Nieden, N.I. Sidestream smoke extracts from harm-reduction and conventional Camel cigarettes inhibit osteogenic differentiation via oxidative stress and differential activation of intrinsic apoptotic pathways. Antioxidants 2022, 11, 2474. [Google Scholar] [CrossRef] [PubMed]
- Lindley, A.A.; Becker, S.; Gray, R.H.; Herman, A.A. Effect of continuing or stopping smoking during pregnancy on infant birth weight, crown-heel length, head circumference, ponderal index, and brain:body weight ratio. Am. J. Epidemiol. 2000, 152, 219–225. [Google Scholar] [CrossRef]
- Bruin, J.E.; Gerstein, H.C.; Holloway, A.C. Long-term consequences of fetal and neonatal nicotine exposure: A critical review. Toxicol. Sci. 2010, 116, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Ross, E.J.; Graham, D.L.; Money, K.M.; Stanwood, G.D. Developmental consequences of fetal exposure to drugs: What we know and what we still must learn. Neuropsychopharmacology 2015, 40, 61–87. [Google Scholar] [CrossRef] [PubMed]
- Holbrook, B.D. The effects of nicotine on human fetal development. Birth Defects Res. C Embryo Today 2016, 108, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Wessler, I.; Kirkpatrick, C.J. Acetylcholine beyond neurons: The non-neuronal cholinergic system in humans. Br. J. Pharmacol. 2008, 154, 1558–1571. [Google Scholar] [CrossRef]
- Borrego-Soto, G.; Eberhart, J.K. Embryonic Nicotine Exposure Disrupts Adult Social Behavior and Craniofacial Development in Zebrafish. Toxics 2022, 10, 612. [Google Scholar] [CrossRef]
- Lake, B.G. Coumarin metabolism, toxicity and carcinogenicity: Relevance for human risk assessment. Food Chem. Toxicol. 1999, 37, 423–453. [Google Scholar] [CrossRef] [PubMed]
- Feron, V.J.; Kruysse, A.; Woutersen, R.A. Respiratory tract tumours in hamsters exposed to acetaldehyde vapour alone or simultaneously to benzo(a)pyrene or diethylnitrosamine. Eur. J. Cancer Clin. Oncol. 1982, 18, 13–31. [Google Scholar] [CrossRef] [PubMed]
- Barnes, S.L.; Singletary, K.W.; Frey, R. Ethanol and acetaldehyde enhance benzo[a]pyrene-DNA adduct formation in human mammary epithelial cells. Carcinogenesis 2000, 21, 2123–2128. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Biswal, S.; Maxwell, T.; Rangasamy, T.; Kehrer, J.P. Modulation of benzo[a]pyrene-induced p53 DNA activity by acrolein. Carcinogenesis 2003, 24, 1401–1406. [Google Scholar] [CrossRef][Green Version]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madrid, J.V.; Vera-Colón, M.K.M.; zur Nieden, N.I. Perturbations in Osteogenic Cell Fate Following Exposure to Constituents Present in Tobacco: A Combinatorial Study. Toxics 2023, 11, 998. https://doi.org/10.3390/toxics11120998
Madrid JV, Vera-Colón MKM, zur Nieden NI. Perturbations in Osteogenic Cell Fate Following Exposure to Constituents Present in Tobacco: A Combinatorial Study. Toxics. 2023; 11(12):998. https://doi.org/10.3390/toxics11120998
Chicago/Turabian StyleMadrid, Joseph V., Madeline K. M. Vera-Colón, and Nicole I. zur Nieden. 2023. "Perturbations in Osteogenic Cell Fate Following Exposure to Constituents Present in Tobacco: A Combinatorial Study" Toxics 11, no. 12: 998. https://doi.org/10.3390/toxics11120998
APA StyleMadrid, J. V., Vera-Colón, M. K. M., & zur Nieden, N. I. (2023). Perturbations in Osteogenic Cell Fate Following Exposure to Constituents Present in Tobacco: A Combinatorial Study. Toxics, 11(12), 998. https://doi.org/10.3390/toxics11120998






