Remediating Garden Soils: EDTA-Soil Washing and Safe Vegetable Production in Raised Bed Gardens
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil
2.2. Preliminary Cultivar Selection Experiment
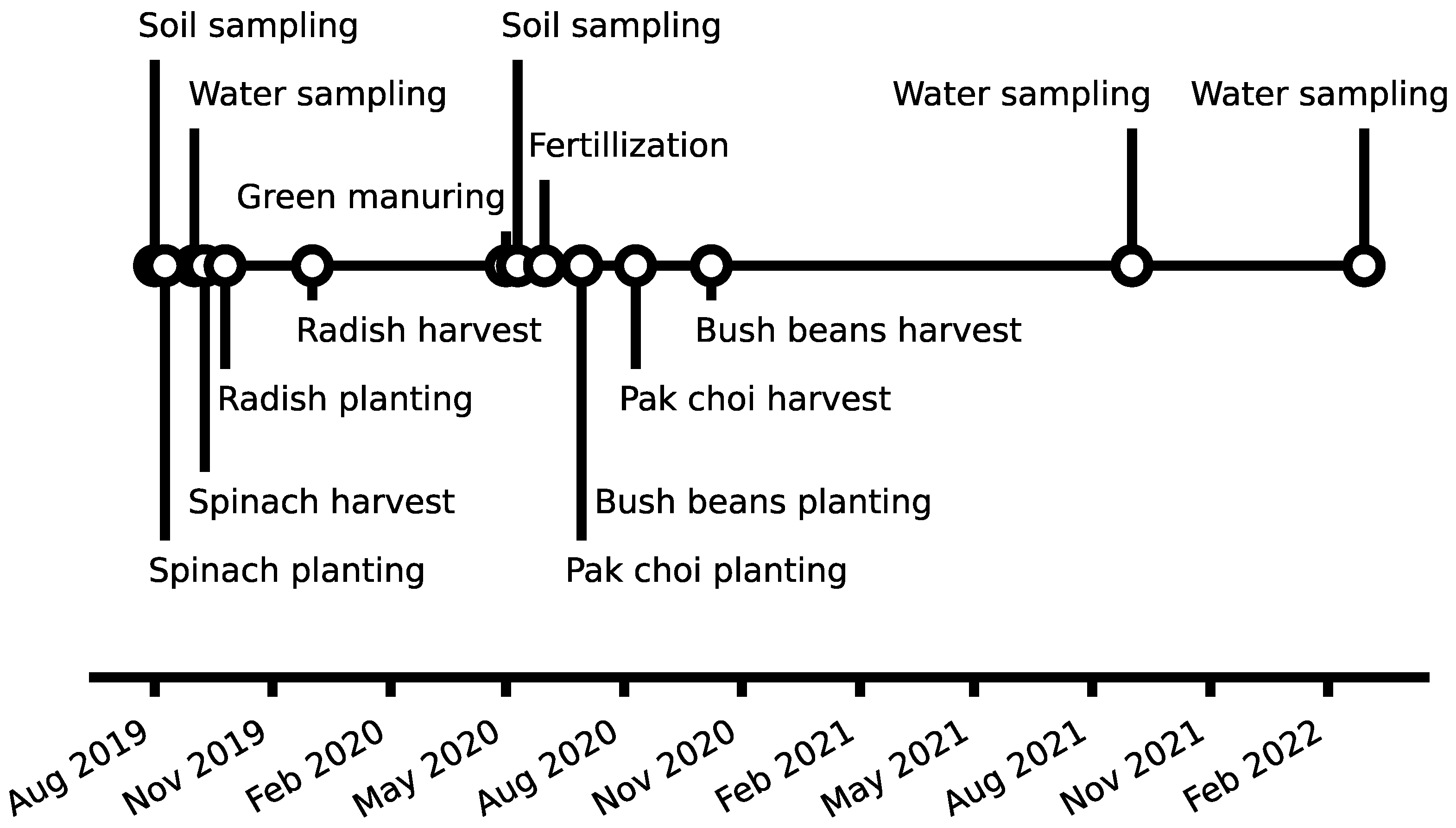
2.3. Raised Bed Experiment
2.4. Soil and Plant Analysis
2.5. Statistical Analysis
3. Results
3.1. Cultivar Selection
3.2. Soil Properties
3.2.1. Basic Properties
3.2.2. Heavy Metal Behavior
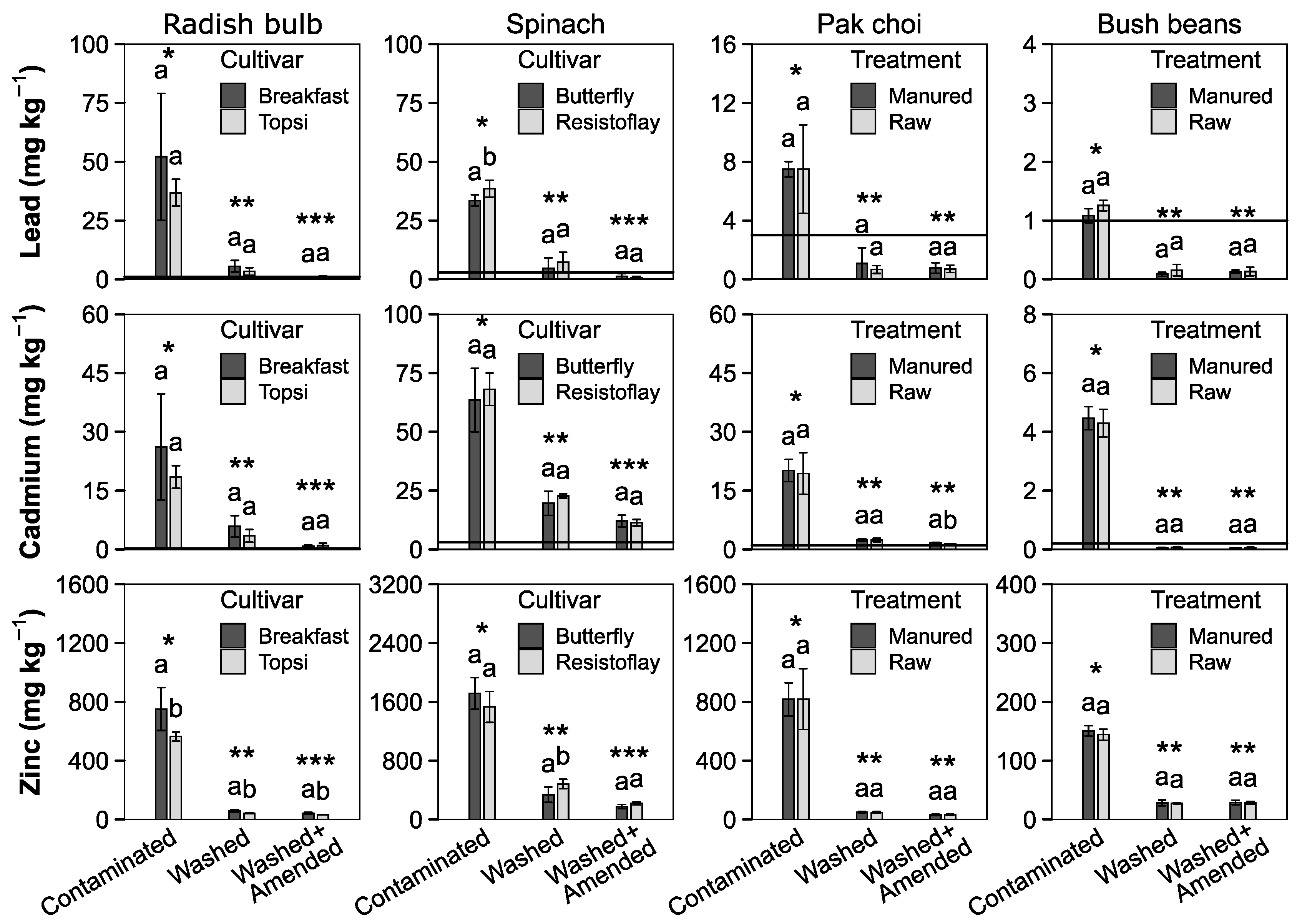
3.3. Plant Yield
4. Discussion
4.1. Impact of Soil Washing on the Soil Fertility
4.2. Impact of Soil Washing on Plant Biomass Production
4.3. Behavior of EDTA and Heavy Metals in the Soil System
4.4. Heavy Metal Uptake into Plants
4.5. EDTA and Heavy Metals in the Soil Leachate
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eigenbrod, C.; Gruda, N. Urban Vegetable for Food Security in Cities. A Review. Agron. Sustain. Dev. 2015, 35, 483–498. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC). Climate Change and Land: An IPCC Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems. In Approved Summary for Policymakers (SPM); IPCC: Geneva, Switzerland, 2019; Volume 8. [Google Scholar]
- Via, M. The Malnutrition of Obesity: Micronutrient Deficiencies That Promote Diabetes. ISRN Endocrinol. 2012, 2012, 103472. [Google Scholar] [CrossRef] [PubMed]
- Opitz, I.; Berges, R.; Piorr, A.; Krikser, T. Contributing to Food Security in Urban Areas: Differences between Urban Agriculture and Peri-Urban Agriculture in the Global North. Agric. Hum. Values 2016, 33, 341–358. [Google Scholar] [CrossRef]
- Siegner, A.; Sowerwine, J.; Acey, C. Does Urban Agriculture Improve Food Security? Examining the Nexus of Food Access and Distribution of Urban Produced Foods in the United States: A Systematic Review. Sustainability 2018, 10, 2988. [Google Scholar] [CrossRef]
- Brown, K.H.; Jameton, A.L. Public Health Implications of Urban Agriculture. J. Public Health Policy 2000, 21, 20–39. [Google Scholar] [CrossRef]
- Wakefield, S.; Yeudall, F.; Taron, C.; Reynolds, J.; Skinner, A. Growing Urban Health: Community Gardening in South-East Toronto. Health Promot. Int. 2007, 22, 92–101. [Google Scholar] [CrossRef]
- Commission, E.-E. Green Infrastructure (GI)—Enhancing Europe’s Natural Capital; European Commission: Brussels, Belgium, 2013.
- Kaiser, M.L.; Williams, M.L.; Basta, N.; Hand, M.; Huber, S. When Vacant Lots Become Urban Gardens: Characterizing the Perceived and Actual Food Safety Concerns of Urban Agriculture in Ohio. J. Food Prot. 2015, 78, 2070–2080. [Google Scholar] [CrossRef]
- De Silva, S.; Ball, A.S.; Huynh, T.; Reichman, S.M. Metal Accumulation in Roadside Soil in Melbourne, Australia: Effect of Road Age, Traffic Density and Vehicular Speed. Environ. Pollut. 2016, 208, 102–109. [Google Scholar] [CrossRef]
- Ziss, E.; Friesl-Hanl, W.; Götzinger, S.; Noller, C.; Puschenreiter, M.; Watzinger, A.; Hood-Nowotny, R. Exploring the Potential Risk of Heavy Metal Pollution of Edible Cultivated Plants in Urban Gardening Contexts Using a Citizen Science Approach in the Project “Heavy Metal City-Zen”. Sustainability 2021, 13, 8626. [Google Scholar] [CrossRef]
- Wahsha, M.; Bini, C.; Argese, E.; Minello, F.; Fontana, S.; Wahsheh, H. Heavy Metals Accumulation in Willows Growing on Spolic Technosols from the Abandoned Imperina Valley Mine in Italy. J. Geochem. Explor. 2012, 123, 19–24. [Google Scholar] [CrossRef]
- Kasperowski, E. Schwermetalle in Böden Im Raum Arnoldstein; Umweltbundesamt, Bundesministerium für Umwelt, Jugend u. Familie: Vienna, Austria, 1993; ISBN 3-85457-108-9. [Google Scholar]
- Finzgar, N.; Jez, E.; Voglar, D.; Lestan, D. Spatial Distribution of Metal Contamination before and after Remediation in the Meza Valley, Slovenia. Geoderma 2014, 217, 135–143. [Google Scholar] [CrossRef]
- Jelusic, M.; Lestan, D. Effect of EDTA Washing of Metal Polluted Garden Soils. Part I: Toxicity Hazards and Impact on Soil Properties. Sci. Total Environ. 2014, 475, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Adriano, D.C.; Wenzel, W.W.; Vangronsveld, J.; Bolan, N.S. Role of Assisted Natural Remediation in Environmental Cleanup. Geoderma 2004, 122, 121–142. [Google Scholar] [CrossRef]
- Hou, X.; Parent, M.; Savard, M.M.; Tassé, N.; Bégin, C.; Marion, J. Lead Concentrations and Isotope Ratios in the Exchangeable Fraction: Tracing Soil Contamination near a Copper Smelter. Geochem. Explor. Environ. Anal. 2006, 6, 229–236. [Google Scholar] [CrossRef]
- McFarland, M.J.; Hauer, M.E.; Reuben, A. Half of US Population Exposed to Adverse Lead Levels in Early Childhood. Proc. Natl. Acad. Sci. USA 2022, 119, e2118631119. [Google Scholar] [CrossRef] [PubMed]
- Needleman, H.L.; Gunnoe, C.; Leviton, A.; Reed, R.; Peresie, H.; Maher, C.; Barrett, P. Deficits in Psychologic and Classroom Performance of Children with Elevated Dentine Lead Levels. New Engl. J. Med. 1979, 300, 689–695. [Google Scholar] [CrossRef]
- Ettler, V. Soil Contamination near Non-Ferrous Metal Smelters: A Review. Appl. Geochem. 2016, 64, 56–74. [Google Scholar] [CrossRef]
- Qu, C.-S.; Ma, Z.-W.; Yang, J.; Liu, Y.; Bi, J.; Huang, L. Human Exposure Pathways of Heavy Metals in a Lead-Zinc Mining Area, Jiangsu Province, China. PLoS ONE 2012, 7, e46793. [Google Scholar] [CrossRef]
- Lwin, C.S.; Seo, B.-H.; Kim, H.-U.; Owens, G.; Kim, K.-R. Application of Soil Amendments to Contaminated Soils for Heavy Metal Immobilization and Improved Soil Quality—A Critical Review. Soil Sci. Plant Nutr. 2018, 64, 156–167. [Google Scholar] [CrossRef]
- Lestan, D. Novel Chelant-Based Washing Method for Soil Contaminated with Pb and Other Metals: A Pilot-Scale Study. Land Degrad. Dev. 2017, 28, 2585–2595. [Google Scholar] [CrossRef]
- Brown, G.A.; Elliott, H.A. Influence of Electrolytes on EDTA Extraction of Pb from Polluted Soil. Water Air Soil Pollut. 1992, 62, 157–165. [Google Scholar] [CrossRef]
- Neale, C.N.; Bricka, R.Y.; Chao, A.C. Evaluating Acids and Chelating Agents for Removing Heavy Metals from Contaminated Soils. Environ. Prog. 1997, 16, 274–280. [Google Scholar] [CrossRef]
- Im, J.; Yang, K.; Jho, E.H.; Nam, K. Effect of Different Soil Washing Solutions on Bioavailability of Residual Arsenic in Soils and Soil Properties. Chemosphere 2015, 138, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Reed, B.E.; Carriere, P.C.; Moore, R. Flushing of a Pb(II) Contaminated Soil Using HCl, EDTA, and CaCl2. J. Environ. Eng. 1996, 122, 48–50. [Google Scholar] [CrossRef]
- Zupanc, V.; Kastelec, D.; Lestan, D.; Grcman, H. Soil Physical Characteristics after EDTA Washing and Amendment with Inorganic and Organic Additives. Environ. Pollut. 2014, 186, 56–62. [Google Scholar] [CrossRef]
- Gluhar, S.; Jez, E.; Lestan, D. The Use of Zero-Valent Fe for Curbing Toxic Emissions after EDTA-Based Washing of Pb, Zn and Cd Contaminated Calcareous and Acidic Soil. Chemosphere 2019, 215, 482–489. [Google Scholar] [CrossRef]
- Beiyuan, J.; Lau, A.Y.T.; Tsang, D.C.W.; Zhang, W.; Kao, C.-M.; Baek, K.; Ok, Y.S.; Li, X.-D. Chelant-Enhanced Washing of CCA-Contaminated Soil: Coupled with Selective Dissolution or Soil Stabilization. Sci. Total Environ. 2018, 612, 1463–1472. [Google Scholar] [CrossRef]
- Hazrati, S.; Farahbakhsh, M.; Heydarpoor, G.; Besalatpour, A.A. Mitigation in Availability and Toxicity of Multi-Metal Contaminated Soil by Combining Soil Washing and Organic Amendments Stabilization. Ecotoxicol. Environ. Saf. 2020, 201, 110807. [Google Scholar] [CrossRef]
- Chen, Y.; Jiang, H.; Li, Y.; Liu, Y.; Chen, Y.; Chen, L.; Luo, X.; Tang, P.; Yan, H.; Zhao, M.; et al. A Critical Review on EDTA Washing in Soil Remediation for Potentially Toxic Elements (PTEs) Pollutants. Rev. Environ. Sci. Bio/Technol. 2022, 21, 399–423. [Google Scholar] [CrossRef]
- Jiang, J.; Yang, M.; Gao, Y.; Wang, J.; Li, D.; Li, T. Removal of Toxic Metals from Vanadium-Contaminated Soils Using a Washing Method: Reagent Selection and Parameter Optimization. Chemosphere 2017, 180, 295–301. [Google Scholar] [CrossRef]
- Zou, Z.; Qiu, R.; Zhang, W.; Dong, H.; Zhao, Z.; Zhang, T.; Wei, X.; Cai, X. The Study of Operating Variables in Soil Washing with EDTA. Environ. Pollut. 2009, 157, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Voglar, D.; Lestan, D. Pilot-Scale Washing of Pb, Zn and Cd Contaminated Soil Using EDTA and Process Water Recycling. Chemosphere 2013, 91, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Gluhar, S.; Kaurin, A.; Lestan, D. Soil Washing with Biodegradable Chelating Agents and EDTA: Technological Feasibility, Remediation Efficiency and Environmental Sustainability. Chemosphere 2020, 257, 127226. [Google Scholar] [CrossRef] [PubMed]
- Kaurin, A.; Cernilogar, Z.; Lestan, D. Revitalisation of Metal-Contaminated, EDTA-Washed Soil by Addition of Unpolluted Soil, Compost and Biochar: Effects on Soil Enzyme Activity, Microbial Community Composition and Abundance. Chemosphere 2018, 193, 726–736. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, S.; Zhong, Q.; Xu, X.; Li, T.; Jia, Y.; Zhang, Y.; Peijnenburg, W.J.G.M.; Vijver, M.G. Effect of Soil Washing with Biodegradable Chelators on the Toxicity of Residual Metals and Soil Biological Properties. Sci. Total Environ. 2018, 625, 1021–1029. [Google Scholar] [CrossRef]
- Hasegawa, H.; Mamun, M.A.A.; Tsukagoshi, Y.; Ishii, K.; Sawai, H.; Begum, Z.A.; Asami, M.S.; Maki, T.; Rahman, I.M.M. Chelator-Assisted Washing for the Extraction of Lead, Copper, and Zinc from Contaminated Soils: A Remediation Approach. App. Geochem. 2019, 109, 104397. [Google Scholar] [CrossRef]
- Kaurin, A.; Gluhar, S.; Tilikj, N.; Lestan, D. Soil Washing with Biodegradable Chelating Agents and EDTA: Effect on Soil Properties and Plant Growth. Chemosphere 2020, 260, 127673. [Google Scholar] [CrossRef]
- Noller, C.; Friesl-Hanl, W.; Hood-Nowotny, R.; Puschenreiter, M.; Watzinger, A. Effect of Chelant-Based Soil Washing and Post-Treatment on Pb, Cd, and Zn Bioavailability and Plant Uptake. Water Air Soil Pollut. 2021, 232, 405. [Google Scholar] [CrossRef]
- Gong, Y.; Zhao, D.; Wang, Q. An Overview of Field-Scale Studies on Remediation of Soil Contaminated with Heavy Metals and Metalloids: Technical Progress over the Last Decade. Water Res. 2018, 147, 440–460. [Google Scholar] [CrossRef]
- Gluhar, S.; Kaurin, A.; Finžgar, N.; Gerl, M.; Kastelec, D.; Lestan, D. Demonstrational Gardens with EDTA-Washed Soil. Part I: Remediation Efficiency, Effect on Soil Properties and Toxicity Hazards. Sci. Total Environ. 2021, 792, 149060. [Google Scholar] [CrossRef]
- Kaurin, A.; Gluhar, S.; Maček, I.; Kastelec, D.; Lestan, D. Demonstrational Gardens with EDTA-Washed Soil. Part II: Soil Quality Assessment Using Biological Indicators. Sci. Total Environ. 2021, 792, 148522. [Google Scholar] [CrossRef] [PubMed]
- Gluhar, S.; Kaurin, A.; Vodnik, D.; Kastelec, D.; Zupanc, V.; Lestan, D. Demonstration Gardens with EDTA-Washed Soil. Part III: Plant Growth, Soil Physical Properties and Production of Safe Vegetables. Sci. Total Environ. 2021, 792, 148521. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.L.; Wu, T.Y.; Lim, P.N.; Shak, K.P.Y. The Use of Vermicompost in Organic Farming: Overview, Effects on Soil and Economics. J. Sci. Food Agric. 2015, 95, 1143–1156. [Google Scholar] [CrossRef]
- Fischer, D.; Glaser, B. Synergisms between Compost and Biochar for Sustainable Soil Amelioration. In Management of Organic Waste; InTech: Rijeka, Croatia, 2012; Volume 1, ISBN 978-953-307-925-7. [Google Scholar]
- Friesl-Hanl, W.; Platzer, K.; Horak, O.; Gerzabek, M.H. Immobilising of Cd, Pb, and Zn Contaminated Arable Soils Close to a Former Pb/Zn Smelter: A Field Study in Austria over 5 Years. Environ. Geochem. Health 2009, 31, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Lestan, D. Washing of Contaminated Soils. United States US9108233B2, 18 August 2015. [Google Scholar]
- Schmidt, H.P.; Bucheli, T.; Kammann, C.; Glaser, B.; Abiven, S.; Leifeld, J. European Biochar Certificate-Guidelines for a Sustainable Production of Biochar; European Biochar Certificat (EBC): Arbaz, Switzerland, 2016. [Google Scholar] [CrossRef]
- Kalra, Y.P.; Maynard, D.G. Methods Manual for Forest Soil and Plant Analysis; Northern Forestry Centre: Edmonton, AB, Canada, 1991; Volume 319.
- Aboukila, E.; Abdelaty, E. Assessment of Saturated Soil Paste Salinity from 1:2.5 and 1:5 Soil-Water Extracts for Coarse Textured Soils. Alex. Sci. Exch. J. 2017, 38, 722–732. [Google Scholar] [CrossRef]
- Burt, R. (Ed.) Soil Survey Laboratory Methods Manual, 4th ed.; Soil Survey Investigations Report; Natural Resources Conservation Service: Washington, DC, USA, 2004.
- ÖNORM L 1084:2016 07 01; Chemische Bodenuntersuchungen—Bestimmung von Carbonat unter Berücksichtigung von Luftdruck und Temperatur. Austrian Standards: Vienna, Austria, 2016.
- Buckee, G.K. Determination of Total Nitrogen in Barley, Malt and Beer by Kjeldahl Procedures and the Dumas Combustion Methodcollaborative Trial. J. Inst. Brew. 1994, 100, 57–64. [Google Scholar] [CrossRef]
- DIN V 19730:1993-02; Bodenbeschaffenheit-Ammoniumnitratextraktion Zur Bestimmung Mobiler Spurenelemente in Mineralböden. Beuth Verlag: Berlin, Germany, 1993.
- Pueyo, M.; López-Sánchez, J.F.; Rauret, G. Assessment of CaCl2, NaNO3 and NH4NO3 Extraction Procedures for the Study of Cd, Cu, Pb and Zn Extractability in Contaminated Soils. Anal. Chim. Acta 2004, 504, 217–226. [Google Scholar] [CrossRef]
- Mehlich, A. Mehlich 3 Soil Test Extractant: A Modification of Mehlich 2 Extractant. Commun. Soil Sci. Plant Anal. 1984, 15, 1409–1416. [Google Scholar] [CrossRef]
- Zhang, M.-K.; Liu, Z.-Y.; Wang, H. Use of Single Extraction Methods to Predict Bioavailability of Heavy Metals in Polluted Soils to Rice. Commun. Soil Sci. Plant Anal. 2010, 41, 820–831. [Google Scholar] [CrossRef]
- ÖNORM L 1085: 2013 11 15; Chemical Analyses of Soils—Method for the Extraction of Elements with a Mixture of Nitric-Acid and Perchloric-Acid. Austrian Standards: Vienna, Austria, 2013.
- Zavala, Y.J.; Duxbury, J.M. Arsenic in Rice: I. Estimating Normal Levels of Total Arsenic in Rice Grain. Environ. Sci. Technol. 2008, 42, 3856–3860. [Google Scholar] [CrossRef]
- Wang, J.; Yu, J.; Kong, X.Z.; Hou, L. Spectrophotometric Determination of EDTA in Aqueous Solution through Ferroin Formation Using Sodium Sulfite as the Reducer. Chemosphere 2013, 91, 351–357. [Google Scholar] [CrossRef] [PubMed]
- ÖNORM L 1061-2: 2019 03 01; Physical Analysis of Soils—Determination of Particle Size Distribution in Mineral Soils Used for Agriculture and Forestry—Part 2: Fine Soil. Austrian Standards: Vienna, Austria, 2019.
- DIN 19683-16:2015-12; Bodenbeschaffenheit—Physikalische Laboruntersuchungen—Teil_16: Bestimmung Der Aggregatstabilität Nach Dem Siebtauchverfahren. Beuth Verlag: Berlin, Germany,, 2015. [CrossRef]
- ÖNORM EN 13041: 2011 12 15; Bodenverbesserungsmittel und Kultursubstrate—Bestimmung der Physikalischen Eigenschaften—Rohdichte (trocken), Luftkapazität, Wasserkapazität, Schrumpfungswert und Gesamtporenvolumen. Austrian Standards: Vienna, Austria, 2011.
- Allaire, J. RStudio: Integrated Development Environment for R. Boston MA 2012, 537, 538. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. 2013. Available online: http://www.R-project.org/ (accessed on 26 October 2022).
- Mallarino, A.P.; Sawyer, J.E.; Barnhart, S.K. A General Guide for Crop Nutrient and Limestone Recommendations in Iowa; Iowa State University: Ames, IA, USA, 2013. [Google Scholar]
- Jahn, R.; Blume, H.P.; Asio, V.B.; Spaargaren, O.; Schad, P. Guidelines for Soil Description; FAO: Rome, Italy, 2006. [Google Scholar]
- Keren, R. Water-Drop Kinetic Energy Effect on Water Infiltration in Calcium and Magnesium Soils. Soil Sci. Soc. Am. J. 1989, 53, 1624–1628. [Google Scholar] [CrossRef]
- Levy, G.J.; Van Der Watt, H.V.H. Effect of Exchangeable Potassium on the Hydraulic Conductivity and Infiltration Rate of Some South African Soils. Soil Sci. 1990, 149, 69–77. [Google Scholar] [CrossRef]
- Miller, W.P.; Newman, K.D.; Frenkel, H. Flocculation Concentration and Sodium/Calcium Exchange of Kaolinitic Soil Clays. Soil Sci. Soc. Am. J. 1990, 54, 346–351. [Google Scholar] [CrossRef]
- Guo, X.; Yang, Y.; Ji, L.; Zhang, G.; He, Q.; Wei, Z.; Qian, T.; Wu, Q. Revitalization of Mixed Chelator–Washed Soil by Adding of Inorganic and Organic Amendments. Water Air Soil Pollut. 2019, 230, 112. [Google Scholar] [CrossRef]
- Muneer, M.; Oades, J.M. The Role of Ca-Organic Interactions in Soil Aggregate Stability. I. Laboratory Studies with Glucose 14C, CaCO3 and CaSO4.2.H2O. Soil Res. 1989, 27, 389–399. [Google Scholar] [CrossRef]
- Rowley, M.C.; Grand, S.; Verrecchia, É.P. Calcium-Mediated Stabilisation of Soil Organic Carbon. Biogeochemistry 2018, 137, 27–49. [Google Scholar] [CrossRef]
- Regelink, I.C.; Stoof, C.R.; Rousseva, S.; Weng, L.; Lair, G.J.; Kram, P.; Nikolaidis, N.P.; Kercheva, M.; Banwart, S.; Comans, R.N.J. Linkages between Aggregate Formation, Porosity and Soil Chemical Properties. Geoderma 2015, 247–248, 24–37. [Google Scholar] [CrossRef]
- Tisdall, J.M.; Oades, J.M. Organic Matter and Water-Stable Aggregates in Soils. Eur. J. Soil Sci. 1982, 33, 141–163. [Google Scholar] [CrossRef]
- Mench, M.; Vangronsveld, J.; Clijsters, H.; Lepp, N.W.; Edwards, R. In Situ Metal Immobilization and Phytostabilization of Contaminated Soils. In Phytoremediation of Contaminated Soil and Water; CRC Press: Boca Raton, FL, USA, 2000; ISBN 978-0-367-80314-8. [Google Scholar]
- Brallier, S.; Harrison, R.B.; Henry, C.L.; Dongsen, X. Liming Effects on Availability of Cd, Cu, Ni and Zn in a Soil Amended with Sewage Sludge 16 Years Previously. Water Air Soil Pollut. 1996, 86, 195–206. [Google Scholar] [CrossRef]
- Acosta, J.A.; Jansen, B.; Kalbitz, K.; Faz, A.; Martínez-Martínez, S. Salinity Increases Mobility of Heavy Metals in Soils. Chemosphere 2011, 85, 1318–1324. [Google Scholar] [CrossRef]
- McBride, M.B. Reactions Controlling Heavy Metal Solubility in Soils. In Advances in Soil Science; Stewart, B.A., Ed.; Advances in Soil Science; Springer: New York, NY, USA, 1989; Volume 10, pp. 1–56. ISBN 978-1-4613-8847-0. [Google Scholar]
- Jez, E.; Bravo, C.; Lestan, D.; Gluhar, S.; Martin-Neto, L.; De Nobili, M.; Contin, M. Changes in Organic Matter Composition Caused by EDTA Washing of Two Soils Contaminated with Toxic Metals. Environ. Sci. Pollut. Res. 2021, 28, 65687–65699. [Google Scholar] [CrossRef]
- Hosseini, S.S.; Lakzian, A.; Halajnia, A.; Hammami, H. The Effect of Olive Husk Extract Compared to the Edta on Pb Availability and Some Chemical and Biological Properties in a Pb-Contaminated Soil. Int. J. Phytoremediation 2018, 20, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Tahmasbian, I.; Safari Sinegani, A.A.; Nguyen, T.T.N.; Che, R.; Phan, T.D.; Hosseini Bai, S. Application of Manures to Mitigate the Harmful Effects of Electrokinetic Remediation of Heavy Metals on Soil Microbial Properties in Polluted Soils. Environ. Sci. Pollut. Res. 2017, 24, 26485–26496. [Google Scholar] [CrossRef] [PubMed]
- Shammas, N. Interactions of Temperature, PH, and Biomass on the Nitrification Process. Water Pollut. Control Fed. 1986, 58, 52–59. [Google Scholar]
- Balesdent, J.; Chenu, C.; Balabane, M. Relationship of Soil Organic Matter Dynamics to Physical Protection and Tillage. Soil Tillage Res. 2000, 53, 215–230. [Google Scholar] [CrossRef]
- Mühlbachová, G. Microbial Biomass Dynamics after Addition of EDTA into Heavy Metal Contaminated Soils. Plant Soil Environ. 2009, 55, 544–550. [Google Scholar] [CrossRef]
- Kaurin, A.; Lestan, D. Multi-Substrate Induced Microbial Respiration, Nitrification Potential and Enzyme Activities in Metal-Polluted, EDTA-Washed Soils. Environ. Pollut. 2018, 243, 238–245. [Google Scholar] [CrossRef]
- Maček, I.; Šibanc, N.; Kavšček, M.; Lestan, D. Diversity of Arbuscular Mycorrhizal Fungi in Metal Polluted and EDTA Washed Garden Soils before and after Soil Revitalization with Commercial and Indigenous Fungal Inoculum. Ecol. Eng. 2016, 95, 330–339. [Google Scholar] [CrossRef]
- Penn, C.J.; Camberato, J.J. A Critical Review on Soil Chemical Processes That Control How Soil PH Affects Phosphorus Availability to Plants. Agriculture 2019, 9, 120. [Google Scholar] [CrossRef]
- Liu, C.-C.; Lin, Y.-C. Reclamation of Copper-Contaminated Soil Using EDTA or Citric Acid Coupled with Dissolved Organic Matter Solution Extracted from Distillery Sludge. Environ. Pollut. 2013, 178, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Klik, B.; Gusiatin, Z.M.; Kulikowska, D. Quality of Heavy Metal-Contaminated Soil before and after Column Flushing with Washing Agents Derived from Municipal Sewage Sludge. Sci. Rep. 2021, 11, 15773. [Google Scholar] [CrossRef] [PubMed]
- Kotuby-Amacher, J.; Koenig, R.; Kitchen, B. Salinity and Plant Tolerance. In Electronic Publication AG-SO-03; Utah State University Extension: Logan, UT, USA, 2000. [Google Scholar]
- Geebelen, W.; Vangronsveld, J.; Adriano, D.C.; Van Poucke, L.C.; Clijsters, H. Effects of Pb-EDTA and EDTA on Oxidative Stress Reactions and Mineral Uptake in Phaseolus Vulgaris. Physiol. Plant. 2002, 115, 377–384. [Google Scholar] [CrossRef]
- Sunda, W.G.; William, G. The Relationship between Cupric Ion Activity and the Toxicity of Copper to Phytoplankton. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 1975. [Google Scholar]
- Neugschwandtner, R.W.; Tlustoš, P.; Komárek, M.; Száková, J. Phytoextraction of Pb and Cd from a Contaminated Agricultural Soil Using Different EDTA Application Regimes: Laboratory versus Field Scale Measures of Efficiency. Geoderma 2008, 144, 446–454. [Google Scholar] [CrossRef]
- Grčman, H.; Velikonja-Bolta, Š.; Vodnik, D.; Kos, B.; Leštan, D. EDTA Enhanced Heavy Metal Phytoextraction: Metal Accumulation, Leaching and Toxicity. Plant Soil 2001, 235, 105–114. [Google Scholar] [CrossRef]
- Currie, J.A. The Importance of Aeration in Providing the Right Conditions for Plant Growth. J. Sci. Food Agric. 1962, 13, 380–385. [Google Scholar] [CrossRef]
- Gliński, J.; Stępniewski, W. Soil Aeration and Its Role for Plants; CRC Press: Boca Raton, FL, USA, 2017; ISBN 978-1-351-07668-5. [Google Scholar]
- Passioura, J.B. Soil Structure and Plant Growth. Soil Res. 1991, 29, 717–728. [Google Scholar] [CrossRef]
- Tejada, M.; Gonzalez, J.L.; García-Martínez, A.M.; Parrado, J. Application of a Green Manure and Green Manure Composted with Beet Vinasse on Soil Restoration: Effects on Soil Properties. Bioresour. Technol. 2008, 99, 4949–4957. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, J. Recovery of EDTA from Soil-Washing Wastewater with Calcium-Hydroxide-Enhanced Sulfide Precipitation. Chemosphere 2019, 237, 124286. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, B.; Deng, T.; Li, F. Reclamation of EDTA by Sodium Tetraethylenepentamine-Multi Dithiocarbamate after Soil Washing Process with EDTA. Environ. Earth Sci. 2017, 76, 311. [Google Scholar] [CrossRef]
- Krishnan, K.A.; Sheela, A.; Anirudhan, T.S. Kinetic and Equilibrium Modeling of Liquid-Phase Adsorption of Lead and Lead Chelates on Activated Carbons. J. Chem. Technol. Biotechnol. 2003, 78, 642–653. [Google Scholar] [CrossRef]
- Nowack, B. Environmental Chemistry of Aminopolycarboxylate Chelating Agents. Environ. Sci. Technol. 2002, 36, 4009–4016. [Google Scholar] [CrossRef] [PubMed]
- Begum, Z.A.; Rahman, I.M.; Tate, Y.; Egawa, Y.; Maki, T.; Hasegawa, H. Formation and Stability of Binary Complexes of Divalent Ecotoxic Ions (Ni, Cu, Zn, Cd, Pb) with Biodegradable Aminopolycarboxylate Chelants (Dl-2-(2-Carboxymethyl) Nitrilotriacetic Acid, GLDA, and 3-Hydroxy-2,2′-Iminodisuccinic Acid, HIDS) in Aqueous Solutions. J. Solut. Chem. 2012, 41, 1713–1728. [Google Scholar] [CrossRef]
- Kubier, A.; Wilkin, R.T.; Pichler, T. Cadmium in Soils and Groundwater: A Review. Appl. Geochem. 2019, 108, 1–16. [Google Scholar] [CrossRef]
- Tessier, A.; Campbell, P.G.; Bisson, M. Sequential Extraction Procedure for the Speciation of Particulate Trace Metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- Adriano, D.C. Trace Elements in Terrestrial Environments: Biogeochemistry, Bioavailability, and Risks of Metals, 2nd ed.; Springer: New York, NY, USA, 2001; ISBN 978-0-387-98678-4. [Google Scholar]
- Trivedi, P.; Axe, L. Long-Term Fate of Metal Contaminants in Soils and Sediments: Role of Intraparticle Diffusion in Hydrous Metal Oxides. In Natural Attenuation of Trace Element Availability in Soils; Society of Environmental Toxicity and Chemistry: Pensacola, FL, USA, 2006; pp. 57–71. [Google Scholar] [CrossRef]
- Noubactep, C. An Analysis of the Evolution of Reactive Species in Fe0/H2O Systems. J. Hazard. Mater. 2009, 168, 1626–1631. [Google Scholar] [CrossRef]
- Ren, Z.; Sivry, Y.; Dai, J.; Tharaud, M.; Cordier, L.; Zelano, I.; Benedetti, M.F. Exploring Cd, Cu, Pb, and Zn Dynamic Speciation in Mining and Smelting-Contaminated Soils with Stable Isotopic Exchange Kinetics. Appl. Geochem. 2016, 64, 157–163. [Google Scholar] [CrossRef]
- Castaldi, P.; Santona, L.; Melis, P. Heavy Metal Immobilization by Chemical Amendments in a Polluted Soil and Influence on White Lupin Growth. Chemosphere 2005, 60, 365–371. [Google Scholar] [CrossRef]
- Karer, J.; Zehetner, F.; Dunst, G.; Fessl, J.; Wagner, M.; Puschenreiter, M.; Stapkēviča, M.; Friesl-Hanl, W.; Soja, G. Immobilisation of Metals in a Contaminated Soil with Biochar-Compost Mixtures and Inorganic Additives: 2-Year Greenhouse and Field Experiments. Environ. Sci. Pollut. Res. 2018, 25, 2506–2516. [Google Scholar] [CrossRef]
- Quintela-Sabarís, C.; Marchand, L.; Kidd, P.S.; Friesl-Hanl, W.; Puschenreiter, M.; Kumpiene, J.; Müller, I.; Neu, S.; Janssen, J.; Vangronsveld, J.; et al. Assessing Phytotoxicity of Trace Element-Contaminated Soils Phytomanaged with Gentle Remediation Options at Ten European Field Trials. Sci. Total Environ. 2017, 599–600, 1388–1398. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Liu, X.; Luo, L.; Zhou, Y.; Wei, J.; Chen, A.; Tang, L.; Wu, H.; Deng, Y.; Zhang, F.; et al. Remediation of Cu, Pb, Zn and Cd-Contaminated Agricultural Soil Using a Combined Red Mud and Compost Amendment. Int. Biodeterior. Biodegrad. 2017, 118, 73–81. [Google Scholar] [CrossRef]
- Smeyers-Verbeke, J.; De Graeve, M.; Francois, M.; De Jaegeref, R.; Massart, D.L. Cd Uptake by Intact Wheat Plants. Plant Cell Environ. 1978, 1, 291–296. [Google Scholar] [CrossRef]
- Kabata-Pendias, A. Trace Elements in Soils and Plants, 4th ed.; CRC Press: Boca Raton, FL, USA, 2010; ISBN 978-1-4200-9370-4. [Google Scholar]
- Smolders, E.; Wagner, S.; Prohaska, T.; Irrgeher, J.; Santner, J. Sub-Millimeter Distribution of Labile Trace Element Fluxes in the Rhizosphere Explains Differential Effects of Soil Liming on Cadmium and Zinc Uptake in Maize. Sci. Total Environ. 2020, 738, 140311. [Google Scholar] [CrossRef] [PubMed]
- Babich, H.; Stotzky, G. Effects of Cadmium on the Biota: Influence of Environmental Factors. In Advances in Applied Microbiology; Perlman, D., Ed.; Academic Press: Cambridge, MA, USA, 1978; Volume 23, pp. 55–117. [Google Scholar]
- Naidu, R.; Kookana, R.S.; Sumner, M.E.; Harter, R.D.; Tiller, K.G. Cadmium Sorption and Transport in Variable Charge Soils: A Review. J. Environ. Qual. 1997, 26, 602–617. [Google Scholar] [CrossRef]
- Loganathan, P.; Vigneswaran, S.; Kandasamy, J.; Naidu, R. Cadmium Sorption and Desorption in Soils: A Review. Crit. Rev. Environ. Sci. Technol. 2012, 42, 489–533. [Google Scholar] [CrossRef]
- ÖNORM S 2088-2: 2014 09 01; Contaminated Sites—Part 2: Application-Specific Evaluation of Soil Pollution of Old Sites and Old Waste Dumps. Austrian Standards International: Wien, Austria. Available online: https://shop.austrian-standards.at/action/en/public/details/526295/OENORM_S_2088-2_2014_09_01;jsessionid=06C8C2D679658182A1E89E1F92F2BF1B (accessed on 6 July 2020).
- Jarrell, W.M.; Beverly, R.B. The Dilution Effect in Plant Nutrition Studies. In Advances in Agronomy; Brady, N.C., Ed.; Academic Press: Cambridge, MA, USA, 1981; Volume 34, pp. 197–224. [Google Scholar]
- Chien, S.H.; Menon, R.G. Dilution Effect of Plant Biomass on Plant Cadmium Concentration as Induced by Application of Phosphate Fertilizers. In Fertilizers and Environment: Proceedings of the International Symposium “Fertilizers and Environment”, Salamanca, Spain, 26–29 September 1994; Rodriguez-Barrueco, C., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 1996; pp. 437–442. ISBN 978-94-009-1586-2. [Google Scholar]
- Saha, J.K.; Coumar, M.V. Alteration of Contamination Threat Due to Dilution Effect on Metal Concentration in Maize–Wheat Biomass on Sludge Amended Clayey Soil. Environ. Monit. Assess. 2022, 194, 273. [Google Scholar] [CrossRef]
- Singh, S.; Zacharias, M.; Kalpana, S.; Mishra, S. Heavy Metals Accumulation and Distribution Pattern in Different Vegetable Crops. JECE 2012, 4, 75–81. [Google Scholar] [CrossRef]
- Herren, T.; Feller, U. Transfer of Zinc from Xylem to Phloem in the Peduncle of Wheat. J. Plant Nutr. 1994, 17, 1587–1598. [Google Scholar] [CrossRef]
- Zeb, A.; Liu, W.; Lian, Y.; Zheng, Z.; Meng, L.; Chen, C.; Song, X. Selection and Breeding of Pollution-Safe Cultivars (PSCs)—An Eco-Friendly Technology for Safe Utilization of Heavy Metal(Loid) Contaminated Soils. Environ. Technol. Innov. 2022, 25, 102142. [Google Scholar] [CrossRef]
- Oviedo, C.; Rodríguez, J. EDTA: The Chelating Agent under Environmental Scrutiny. Quím. Nova 2003, 26, 901–905. [Google Scholar] [CrossRef]
- Tsang, D.C.W.; Olds, W.E.; Weber, P. Residual Leachability of CCA-Contaminated Soil after Treatment with Biodegradable Chelating Agents and Lignite-Derived Humic Substances. J. Soils Sediments 2013, 13, 895–905. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality, 3rd ed.; World Health Organization: Geneva, Switzerland, 2008; ISBN 978-92-4-154761-1. [Google Scholar]
- Wei, M.; Chen, J.; Wang, X. Removal of Arsenic and Cadmium with Sequential Soil Washing Techniques Using Na2EDTA, Oxalic and Phosphoric Acid: Optimization Conditions, Removal Effectiveness and Ecological Risks. Chemosphere 2016, 156, 252–261. [Google Scholar] [CrossRef] [PubMed]
- Deng, T.; Zhang, B.; Li, F.; Jin, L. Sediment Washing by EDTA and Its Reclamation by Sodium Polyamidoamine-Multi Dithiocarbamate. Chemosphere 2017, 168, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Delil, A.D.; Köleli, N. The Removal of Pb and Cd from Heavily Contaminated Soil in Kayseri, Turkey by a Combined Process of Soil Washing and Electrodeposition. Soil Sediment Contam. Int. J. 2018, 27, 469–484. [Google Scholar] [CrossRef]


| Contaminated | Washed | Washed + Amended | Contaminated | Washed | Washed + Amended | |
|---|---|---|---|---|---|---|
| 2019 | 2020 | |||||
| pH (water) | 5.86 ± 0.16 c | 7.14 ± 0.01 b | 7.34 ± 0.01 a | 5.46 ± 0.04 c | 7.13 ± 0.03 b | 7.20 ± 0.02 a * |
| ECe mS cm−1 | 2.25 ± 0.23 b | 8.03 ± 0.05 a | 7.75 ± 1.12 a | 2.90 ± 0.83 c | 11.6 ± 0.68 b * | 12.8 ± 0.87 a * |
| CECeff (cmolc kg−1) | 10.4 ± 0.17 a | 10.7 ± 0.07 a | 11.8 ± 0.13 b | 10.3 ± 0.09 a | 10.4 ± 0.26 a | 11.5 ± 0.10 b |
| CN ratio | 9.5 c | 10.1 c | 16.6 a | 8.8 b | 8.5 c | 14.3 a |
| SOC (%) | 2.86 ± 0.11 b | 2.93 ± 0.27 b | 5.31 ± 0.58 a | 3.18 ± 0.14 b | 2.76 ± 0.07 c | 5.44 ± 0.7 a |
| Ntot (%) | 0.30 ± 0.01 a | 0.29 ± 0.03 a | 0.32 ± 0.03 a | 0.36 ± 0.01 a * | 0.29 ± 0.02 b | 0.38 ± 0.01 a |
| P (mg kg−1) | 50.8 ± 1.62 b | 27.4 ± 0.58 c | 58.0 ± 1.76 a | 50.6 ± 1.39 a | 18.5 ± 2.90 c * | 37.1 ± 4.19 b * |
| K (mg kg−1) | 75.9 ± 3.73 c | 110 ± 2.50 b | 511 ± 23.02 a | 73.4 ± 5.36 c | 89.1 ± 4.66 b * | 456 ± 46.8 a |
| NH4-N (mg kg−1) | 8.64 ± 1.57 a | 10.7 ± 0.32 a | 7.83 ± 1.38 a | 18.3 ± 3.27 a * | 11.6 ± 0.33 b * | 9.88 ± 1.56 c |
| NO3-N (mg kg−1) | 75.9 ± 30.8 a | 52.9 ± 6.7 a | 60.7 ± 11.1 a | 103 ± 45 a | 92.0 ± 22.3 a * | 154 ± 42.0 a * |
| EDTA (mg kg−1) | n.d. | 42.2 ± 1.57 a | 37.4 ± 5.17 a | n.d. | 15.7 ± 3.56 a * | 17.6 ± 4.61 a * |
| Ca (mg kg−1) | 883 ± 69.8 b | 3360 ± 92.2 a | 3624 ± 201 a | 713 ± 77.0 c | 3380 ± 82.2 b | 3950 ± 175 a |
| Fe (mg kg−1) | 190 ± 14.1 b | 583 ± 11.2 a | 567 ± 10.9 a | 113 ± 2.94 b * | 384 ± 14.1 a * | 377 ± 5.28 a * |
| Mg (mg kg−1) | 118 ± 4.75 b | 72.8 ± 2.31 c | 146 ± 6.77 a | 149 ± 23.6 b | 133 ± 6.52 b * | 168 ± 16.3 a |
| Mn (mg kg−1) | 32.6 ± 1.34 c | 78.9 ± 1.27 a | 68.2 ± 2.66 b | 33.9 ± 1.17 a | 30.7 ± 2.76 b * | 36.3 ± 2.22 a * |
| Na (mg kg−1) | 62.2 ± 8.22 a | 656 ± 14.1 b | 552 ± 33.1 c | - | - | - |
| S (mg kg−1) | 154 ± 25.9 c | 1627 ± 44.1 a | 1440 ± 86.3 b | 24.9 ± 2.09 b * | 1100 ± 52 a * | 1120 ± 76.7 a * |
| Contaminated | Washed | Washed + Amended | |
|---|---|---|---|
| Sand (%) | 38.2 | 32.8 | 40.6 |
| Silt (%) | 47.2 | 49.9 | 43.4 |
| Clay (%) | 14.6 | 17.3 | 16.0 |
| SAS 1 (%) | 79.5 ± 0.83 a | 84.4 ± 0.4 b | 83.3 ± 0.53 b |
| Bulk density (g cm−3) | 1.11 ± 0.02 b | 1.17 ± 0.03 c | 1.06 ± 0.03 a |
| Total porosity (%) | 55.2 ± 2.30 b | 46.2 ± 1.62 a | 49.9 ± 4.30 a |
| Field capacity (%) | 38.5 ± 9.8 b | 32.6 ± 3.1 a | 31.5 ± 3.2 a |
| Contaminated | Washed | Washed + Amended | Contaminated | Washed | Washed + Amended | |
|---|---|---|---|---|---|---|
| Total metals (mg kg−1) | 2019 | 2020 | ||||
| Pb | 795 ± 22.0 a | 189 ± 13.6 b | 201 ± 3.0 b | - | - | - |
| Cd | 4.47 ± 0.24 a | 2.36 ± 0.09 b | 2.14 ± 0.17 b | - | - | - |
| Zn | 484 ± 10.5 a | 410 ± 7.05 b | 373 ± 9.67 c | - | - | - |
| NH4NO3 (mg kg−1) | ||||||
| Pb | 3.54 ± 0.15 a | 1.37 ± 0.02 b | 0.90 ± 0.07 c | 8.27 ± 1.00 a * | 0.87 ± 0.05 b * | 0.48 ± 0.11 c * |
| Cd | 0.60 ± 0.0 a | 0.22 ± 0.03 b | 0.19 ± 0.03 b | 1.03 ± 0.063 a * | 0.27 ± 0.014 b | 0.24 ± 0.021 b |
| Zn | 25.3 ± 0.72 a | 0.68 ± 0.03 c | 0.96 ± 0.02 b | 45.8 ± 3.64 a * | 0.28 ± 0.037 b * | 0.23 ± 0.024 b * |
| Mehlich3 (mg kg−1) | ||||||
| Pb | 351 ± 10.5 a | 55.3 ± 1.80 b | 53.9 ± 1.61 b | 321 ± 5.52 a | 47.2 ± 3.64 b * | 46.19 ± 2.88 b * |
| Cd | 3.42 ± 0.03 a | 1.09 ± 0.05 b | 1.05 ± 0.03 b | 3.22 ± 0.21 a | 0.88 ± 0.03 b * | 0.87 ± 0.05 b |
| Zn | 112 ± 2.37 a | 21.6 ± 0.37 b | 20.9 ± 0.66 b | 103 ± 2.16 a * | 21.0 ± 0.65 b | 19.9 ± 0.5 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noller, C.; Friesl-Hanl, W.; Hood-Nowotny, R.; Watzinger, A. Remediating Garden Soils: EDTA-Soil Washing and Safe Vegetable Production in Raised Bed Gardens. Toxics 2022, 10, 652. https://doi.org/10.3390/toxics10110652
Noller C, Friesl-Hanl W, Hood-Nowotny R, Watzinger A. Remediating Garden Soils: EDTA-Soil Washing and Safe Vegetable Production in Raised Bed Gardens. Toxics. 2022; 10(11):652. https://doi.org/10.3390/toxics10110652
Chicago/Turabian StyleNoller, Christoph, Wolfgang Friesl-Hanl, Rebecca Hood-Nowotny, and Andrea Watzinger. 2022. "Remediating Garden Soils: EDTA-Soil Washing and Safe Vegetable Production in Raised Bed Gardens" Toxics 10, no. 11: 652. https://doi.org/10.3390/toxics10110652
APA StyleNoller, C., Friesl-Hanl, W., Hood-Nowotny, R., & Watzinger, A. (2022). Remediating Garden Soils: EDTA-Soil Washing and Safe Vegetable Production in Raised Bed Gardens. Toxics, 10(11), 652. https://doi.org/10.3390/toxics10110652








