The Dioscorea Genus (Yam)—An Appraisal of Nutritional and Therapeutic Potentials
Abstract
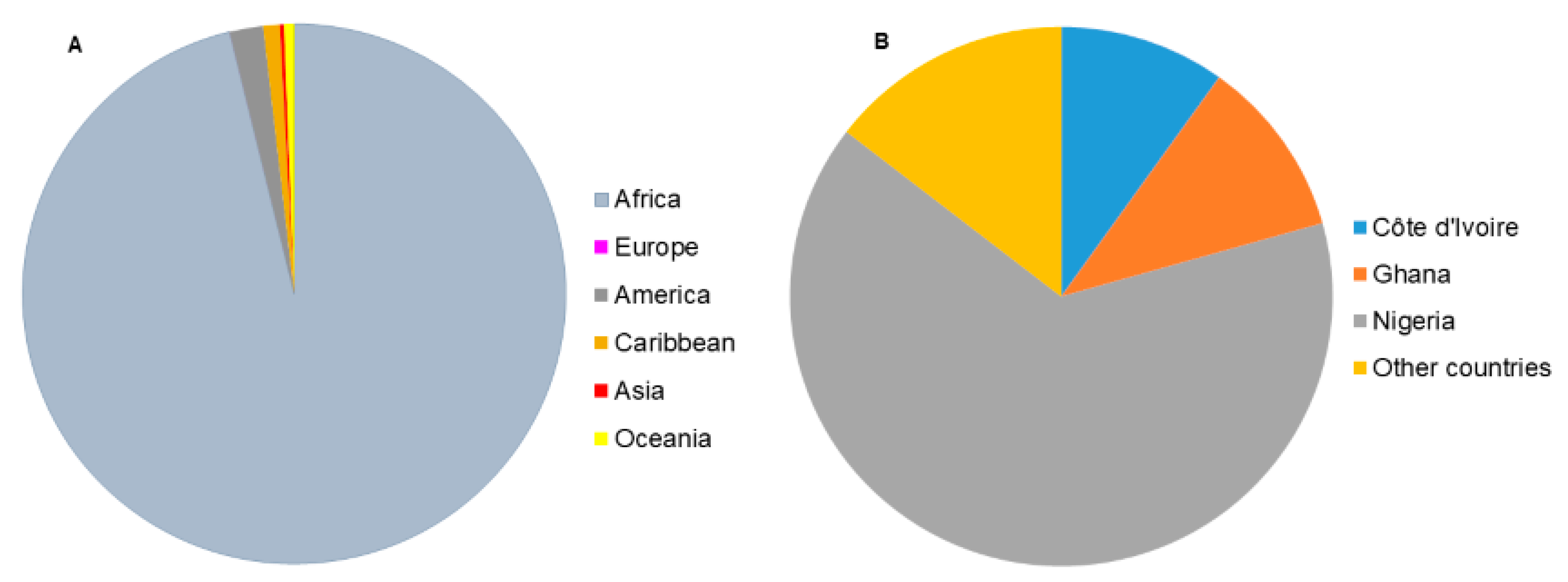
:1. Introduction
2. Yam Nutritional Value
2.1. Yam as a Source of Dietary Energy
2.2. Yam as a Source of Minerals
3. Bioactive Compounds in Yam
3.1. Steroidal Saponin
3.2. Dioscorin
3.3. Alkaloids
3.4. Flavonoids
3.5. Phenols and Phenolic Acids
3.6. Other Bioactive Compounds
4. Therapeutic Potentials of Yams
4.1. Antimicrobial Potential of Yam
4.2. Antioxidant Activities of Yam
4.3. Anti-Inflammatory Activity of Yam
4.4. Anticancer Activity of Yam
4.5. Anti-Diabetic Activity of Yam
4.6. Anti-Obesity and -Hypercholesterolemic Activities of Yam
4.7. Yam as an Agent for Degenerative Disease Management
4.8. Yam as an Agent for the Management of Menopausal Symptoms
4.9. Yam as Pharmaceutical Excipient
5. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alexander, J.; Coursey, D.G. The Origins of Yam Cultivation; Ucko, P.J., Dimbleby, G.H., Eds.; Gerald Duckworth & Co.: London, UK, 1969. [Google Scholar]
- Karnick, C.R. Dioscorea (YAMS)—The food of the slaves, with potentials for newer drugs: A review. Q. J. Crude Drug Res. 1969, 9, 1372–1391. [Google Scholar] [CrossRef]
- Singh, S.C. Some wild plants of food value in Nepal. J. Tribhuban Univ. Kath 1960, 4, 50–56. [Google Scholar]
- Coursey, D.; Yams, G. An account of the Nature, Origins, Cultivation and Utilisation of the Useful Members of the Dioscoreaceae; Rhind, D., Ed.; Longmans, Green and Co LTD: London, UK, 1967. [Google Scholar]
- Scarcelli, N.; Chaïr, H.; Causse, S.; Vesta, R.; Couvreur, T.L.; Vigouroux, Y. Crop wild relative conservation: Wild yams are not that wild. Boil. Conserv. 2017, 210, 325–333. [Google Scholar] [CrossRef]
- Govaerts, R.; Wilkin, P.; Saunders, R.M.K. World Check-list of the Dioscoreales: Yams and their Allies; Royal Botanic Gardens: Kew, UK, 2007. [Google Scholar]
- Dutta, B. Food and medicinal values of certain species of Dioscorea with special reference to Assam. J. Pharmacogn. Phytochem. 2015, 3, 15–18. [Google Scholar]
- Trimanto, T.; Hapsari, L. Diversity and utilization of Dioscorea spp. tuber as alternative food source in Nganjuk Regency, East Java. Agrivita 2015, 37, 97–107. [Google Scholar] [CrossRef]
- Lev, L.S.; Shriver, A.L. A trend analysis of yam production, area, yield, and trade (1961–1996). In Ligname, Plante Seculaire et Culture Davenir; Actes du Seminaire International CIRAD-INRA-ORSTOM-CORAF; CIRAD: Montpellier, France, 1998; pp. 8–10. [Google Scholar]
- Dansi, A.; Dantsey-Barry, H.; Dossou-Aminon, I.; N’Kpenu, E.K.; Agré, A.P.; Sunu, Y.D.; Kombaté, K.; Loko, Y.L.; Dansi, M.; Assogba, P.; et al. Varietal diversity and genetic erosion of cultivated yams (Dioscorea cayenensis Poir - D. rotundata Lam complex and D. alata L.) in Togo. Int. J. Biodivers. Conserv. 2013, 5, 223–239. [Google Scholar] [CrossRef]
- FAOSTAT—Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 9 April 2020).
- Mignouna, H.D.; Abang, M.M.; Asiedu, R. Genomics of yams, a common source of food and medicine in the tropics. In Genomics of Tropical Crop Plants. Plant Genetics and Genomics: Crops and Models; Moore, P.H., Ming, R., Eds.; Springer: New York, NY, USA, 2008; pp. 549–570. [Google Scholar]
- Lebot, V. Tropical Root and Tuber Crops: Cassava, Sweet Potato, Yams and Aroids, 2nd ed.; CABI: Wallingford, CT, USA, 2019. [Google Scholar]
- Obidiegwu, J.E.; Akpabio, E.M. The geography of yam cultivation in southern Nigeria: Exploring its social meanings and cultural functions. J. Ethn. Foods 2017, 4, 28–35. [Google Scholar] [CrossRef]
- Zannou, A.; Ahanchédé, A.; Struik, P.C.; Richards, P.; Zoundjihékpon, J.; Tossou, R.; Vodouhè, S. Yam and cowpea diversity management by farmers in the Guinea-Sudan transition zone of Benin. NJAS Wagening J. Life Sci. 2004, 52, 393–420. [Google Scholar] [CrossRef] [Green Version]
- Lásztity, R.; Hidvégi, M.; Bata, Á. Saponins in food. Food Rev. Int. 1998, 14, 371–390. [Google Scholar] [CrossRef]
- IITA. Yam (Dioscorea Species). Available online: https://www.iita.org/cropsnew/dioscoria/ (accessed on 6 April 2020).
- Thompson, A.K. Chinese yam. In Fruit and Vegetables: Harvesting, Handling and Storage; John Wiley & Sons: Hoboken, NJ, USA, 2014; p. 1429. [Google Scholar]
- Davidson, A. Yam. In Oxford Companion to Food; Oxford University Press: Oxford, UK, 2016; p. 883. [Google Scholar]
- Guaadaoui, A.; Benaicha, S.; Elmajdoub, N.; Bellaoui, M.; Hamal, A. What is a bioactive compound? A combined definition for a preliminary consensus. Int. J. Food Sci. Nutr. 2014, 3, 17–179. [Google Scholar] [CrossRef]
- Okwu, D.E.; Ndu, C.U. Evaluation of the phytonutrients, mineral and vitamin contents of some varieties of yam.pdf. Int. J. Mol. Med. Adv. Sci. 2006, 2, 199–203. [Google Scholar]
- Zhang, L.; Ng, T.B.; Lam, J.K.W.; Wang, S.W.; Lao, L.; Zhang, K.Y.Z.; Zhang, S.C.W. Research and Development of proteins and peptides with therapeutic potential from yam tubers. Curr. Protein Pept. Sci. 2019, 20, 277–284. [Google Scholar] [CrossRef]
- Kumar, S.; Das, G.; Shin, H.S.; Patra, J.K. Dioscorea spp (A Wild Edible Tuber): A study on its ethnopharmacological potential and traditional use by the local people of similipal biosphere reserve, India. Front. Pharmacol. 2017, 8, 52. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Mahanti, P.; Singh, N.R.; Rath, S.K.; Jena, P.K.; Patra, J.K. Antioxidant activity, antibacterial potential and characterization of active fraction of Dioscorea pentaphylla L. tuber extract collected from Similipal Biosphere Reserve, Odisha, India. Brazilian J. Pharm. Sci. 2017, 53. [Google Scholar] [CrossRef]
- Nashriyah, M.; Salmah, T.; NurAtiqah, M.Y.; Siti Nor Indah, O.; Muhamad Azhar, A.W.; Munirah, S.; Nornasuha, Y.; Abdul Manaf, A. Ethnobotany and distribution of Dioscorea hispida Dennst (Dioscoreaceae) in Besut, Marang and Setiu Districts of Terengganu, Peninsular Malaysia. World Acad. Sci. Eng. Technol. 2012, 72, 240–243. [Google Scholar]
- De Pasquale, A. Pharmacognosy: The oldest modern science. J. Ethnopharmacol. 1984, 11, 1–16. [Google Scholar] [CrossRef]
- Nyaboga, E.; Tripathi, J.N.; Manoharan, R.; Tripathi, L. Agrobacterium-mediated genetic transformation of yam (Dioscorea rotundata): An important tool for functional study of genes and crop improvement. Front. Plant Sci. 2014, 5, 463. [Google Scholar] [CrossRef] [Green Version]
- Bevan, C.W.L.; Broadbent, J.L.; Hirst, J. A convulsant alkaloid of Dioscorea dumetorum. Nature 1956, 177, 935. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, M.R.; Kawabata, J. Organic acid, phenolic content and antioxidant activity of wild yam (Dioscorea spp.) tubers of Nepal. Food Chem. 2004, 88, 163–168. [Google Scholar] [CrossRef]
- Reddy, D.S. Ethnobotanical study of body coolants used by tribals of Nallamalais in Telangana, India. Int. J. Adv. Res. 2015, 3, 411–415. [Google Scholar]
- Kumar, S.; Mahanti, P.; Sk, R.; Jk, P. Qualitative phytochemical analysis and antibacterial activity of Dioscorea alata L.: A nutraceutical tuber crops of rural odisha. J. Altern. Med. Res. 2017, 3, 122. [Google Scholar]
- Saleha, C.; Saidi, N.; Murniana, S.; Rasnovi, S.; Iqbalsyah, T.M. Nutritional composition of Dioscorea hispida from different locations around Leuser ecosystem area. J. Nat. 2018, 18, 1–6. [Google Scholar] [CrossRef]
- Shim, W.; Oh, U. Histamine-induced itch and its relationship with pain. Mol. Pain 2008, 4, 1744–8069. [Google Scholar] [CrossRef] [Green Version]
- Hur, G.Y.; Park, H.J.; Kim, H.A.; Ye, Y.M.; Park, H.S. Identification of Dioscorea batatas (Sanyak) allergen as an inhalant and oral allergen. J. Korean Med. Sci. 2008, 23, 72–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Z.G.; Jiang, W.; Nitin, M.; Bao, X.Q.; Chen, S.L.; Tao, Z.M. Characterizing diversity based on nutritional and bioactive compositions of yam germplasm (Dioscorea spp.) commonly cultivated in China. J. Food Drug Anal. 2016, 24, 367–375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bekele, A.; Bekele, E. Proximate and mineral composition variability in Ethiopian yam (Dioscorea spp.). J. Food Nutr. Sci. 2018, 6, 12–17. [Google Scholar] [CrossRef] [Green Version]
- Chu, E.P.; Figueiredo-Ribeiro, R.C.L. Native and exotic species of Dioscorea used as food in Brazil. Econ. Bot. 1991, 45, 467–479. [Google Scholar] [CrossRef]
- Mohan, V.R.; Kalidass, C. Nutritional and antinutritional evaluation of some unconventional wild edible plants. Trop. Subtrop. Agroecosystems 2010, 12, 495–506. [Google Scholar]
- Shanthakumari, S.; Mohan, V.R.; de Britto, J. Nutritional evaluation and elimination of toxic principles in wild yam (Dioscorea spp.). Trop. Subtrop. Agroecosystems 2008, 8, 319–325. [Google Scholar]
- USDA-United State Department og Agriculture (USDA) Food Compositional Database. 2018. Available online: https://fdc.nal.usda.gov/fdc-app.html#/ (accessed on 5 May 2020).
- Ayeleso, B.; Ramachela, K.; Mukwevho, E. A review of therapeutic potentials of sweet potato: Pharmacological activities and influence of the cultivar. Trop. J. Pharm. Res. 2016, 15, 2751–2761. [Google Scholar] [CrossRef] [Green Version]
- Polycarp, D.; Afoakwa, E.O.; Budu, A.S.; Otoo, E. Characterization of chemical composition and anti-nutrition factors in seven species within the Ghanaian yam (Dioscorea) germplasm. Int. Food Res. J. 2012, 19, 985–992. [Google Scholar]
- Sanful, R.E.; Oduro, I.; Ellis, W.O. Proximate and functional properties of five local varieties of aerial yam (Dioscorea bulbifera) in Ghana. Middle East J. Sci. Res. 2013, 14, 947–951. [Google Scholar]
- FAO. Continental Programme on Post-Harvest Losses (PHL) Reduction, Rapid Country Needs Assessment, Ghana; No. Rapport No: 09/016 FAO-AfDB GHA); Food and Agriculture Organisation of the United Nations, Rome, 2011. Available online: http://www.fao.org/3/a-au870e.pdf (accessed on 16 May 2020).
- Zhang, Z.; Gao, W.; Wang, R.; Huang, L. Changes in main nutrients and medicinal composition of Chinese yam (Dioscorea opposita) tubers during storage. J. Food Sci. Technol. 2014, 51, 2535–2543. [Google Scholar] [CrossRef] [PubMed]
- Estiasih, T.; Apriliyanti, M.W.; Afriliana, A.; Kusnadi, J. Physicochemical and bioactives characteristics of purple and yellow water yam (Dioscorea alata) tubers. Int. J. PharmTech Res. 2013, 5, 1691–1701. [Google Scholar]
- Jiang, Q.; Gao, W.; Li, X.; Xia, Y.; Wang, H.; Wu, S.; Huang, L.; Liu, C.; Xiao, P. Characterizations of starches isolated from five different Dioscorea L. species. Food Hydrocoll. 2012, 29, 35–41. [Google Scholar] [CrossRef]
- Muzac-Tucker, I.; Asemota, H.N.; Ahmad, M.H. Biochemical composition and storage of Jamaican yams (Dioscorea sp.). J. Sci. Food Agric. 1993, 62, 219–224. [Google Scholar] [CrossRef]
- Ogbo, A.F.; Agu, K. Proximate composition, physiological changes during storage, and shelf life of some Nigerian varieties of yams (Dioscorea species). J. Sci. Res. Rep. 2014, 3, 553–562. [Google Scholar] [CrossRef]
- Ojo, A.; Odedeji, J.O.; Oladapo, S.A.; Olosunde, O.O. Comparative studies on the chemical compositions of cereal (wheat and corn) and tubers (water yam and bitter yam) flours. Int. J. Innov. Sci. Eng. Technol. 2017, 4, 369–385. [Google Scholar]
- Shajeela, P.S.; Mohan, V.R.; Jesudas, L.L.; Soris, P.T. Nutritional and antinutritional evaluation of wild yam (Dioscorea spp.). Trop. Subtrop. Agroecosystems 2011, 14, 723–730. [Google Scholar]
- Tortoe, C.; Dowuona, S.; Akonor, P.T.; Dziedzoave, N.T. Examining the physicochemical, functional and rheological properties in flours of farmers’ 7 key yam (Dioscorea spp.) varieties in Ghana to enhance yam production. Cogent Food Agric. 2017, 3, 1371564. [Google Scholar] [CrossRef]
- Udensi, E.A.; Oselebe, H.O.; Iweala, O.O. Investigation of chemical composition and functional properties of water yam (Dioscorea alata): Effect of varietal differences. Pakistan J. Nutr. 2008, 7, 342–344. [Google Scholar] [CrossRef] [Green Version]
- Wanasundera, J.P.D.; Ravindran, G. Nutritional assessment of yam (Dioscorea alata) tubers. Plant Foods Hum. Nutr. 1994, 46, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Baah, F.D.; Maziya-Dixon, B.; Asiedu, R.; Oduro, I.; Ellis, W.O. Nutritional and biochemical composition of D. alata (Dioscorea spp.) tubers. J. FoodAgric. Environ. 2009, 7, 373–378. [Google Scholar]
- Chaudhury, S.; Rahaman, C.H.; Singh, H.; Chaudhuri, K.; Pillai, B.; Seal, T. Dioscorea alata: A potent wild edible plant consumed by the Lodha Tribal community of West Bengal, India. J. Pharmacogn. Phytochem. 2018, 7, 654–663. [Google Scholar]
- Dufie, W.-M.F.; Oduro, I.; Ellis, W.O.; Asiedu, R.; Maziya-Dixon, B. Potential health benefits of water yam (Dioscorea alata). Food Funct. 2013, 4, 1496–1501. [Google Scholar] [CrossRef]
- Ezeabara, C.A.; Anona, R.O. Comparative analyses of phytochemical and nutritional compositions of four species of Dioscorea. Acta Sci. Nutr. Health 2018, 2, 90–94. [Google Scholar]
- Ezeocha, C.V.; Ojimelukwe, C.P. The impact of cooking on the proximate composition and anti-nutritional factors of water yam (Dioscorea alata). J. Stored Prod. Postharvest Res. 2012, 3, 172–176. [Google Scholar] [CrossRef]
- Ukom, A.N.; Ojimelukwe, P.C.; Ezeama, C.F.; Ortiz, D.O.; Aragon, I. Phenolic content and antioxidant activity of some under-utilized Nigerian yam (Dioscorea spp.) and cocoyam (Xanthosomamaffa (scoth)) tubers. IOSR J. Environ. Sci. Toxicol. Food Technol. 2014, 8, 104–111. [Google Scholar] [CrossRef]
- Ukom, A.N.; Ojimelukwe, P.C.; Emetole, J.M. Physicochemical and functional properties of starch from under-utilized yam (Dioscorea spp.) and cocoyam (Xanthosoma maffa (Scoth)) tubers of Southeast Nigeria. Starch Staerke 2016, 68, 410–415. [Google Scholar] [CrossRef]
- Ifeanacho, M.O.; Ikewuchi, C.C.; Ikewuchi, J.C. Proximate and antinutrient profiles of tubers of Dioscorea bulbifera from Rivers State, Nigeria: Nutritional and health implications of consumption. Elixir Food Sci. 2017, 109, 47847–47850. [Google Scholar]
- Kayode, R.M.O.; Buhari, O.J.; Otutu, L.O.; Ajibola, T.B.; Oyeyinka, S.A.; Opaleke, D.O.; Akeem, S.A. Physicochemical properties of processed aerial yam (Dioscorea bulbifera) and sensory properties of paste (amala) prepared with cassava flour. J. Agric. Sci. 2017, 12, 84. [Google Scholar] [CrossRef] [Green Version]
- Ayo, J.A.; Ojo, M.; Obike, J. Proximate composition, functional and phytochemical properties of pre-heated aerial yam flour. Res. J. Food Sci. Nutr. 2018, 3, 1–8. [Google Scholar] [CrossRef]
- Bhandari, M.R.; Kasai, T.; Kawabata, J. Nutritional evaluation of wild yam (Dioscorea spp.) tubers of Nepal. Food Chem. 2003, 82, 619–623. [Google Scholar] [CrossRef]
- Ezeocha, V.C.; Nwogha, J.S.; Ohuoba, A.N.; Chukwu, L.I. Evaluation of poultry manure application rates on the nutrient composition of Dioscorea bulbifera (Aerial yam). Niger. Food J. Off. 2014, 32, 92–96. [Google Scholar] [CrossRef] [Green Version]
- Adepoju, O.T.; Boyejo, O.; Adeniji, P.O. Effects of processing methods on nutrient and antinutrient composition of yellow yam (Dioscorea cayenensis) products. Food Chem. 2018, 238, 160–165. [Google Scholar] [CrossRef]
- Afoakwa, E.O.; Sefa-Dedeh, S. Chemical composition and quality changes occurring in Dioscorea dumetorum pax tubers after harvest. Food Chem. 2001, 75, 85–91. [Google Scholar] [CrossRef]
- Onwuamanam, C.I.; Iwuoha, C.I.; Onuegbu, N.C.; Ogueke, C.C.; Nwosu, J.N. Quality characteristics of processed flours from trifoliate yam (Dioscorea dumetorum) as influenced by steeping and boiling in varying concentration of trona solution over time. Am. J. Food Technol. 2013, 8, 162–172. [Google Scholar] [CrossRef] [Green Version]
- Egbuonu, A.; Nzewi, D. Influence of Blanching on Some Nutrient and Anti-nutrient Compositions of Bitter Yam (Dioscorea dumetorum). Br. J. Appl. Sci. Technol. 2016, 16, 1–8. [Google Scholar] [CrossRef]
- Oyeyinka, S.A.; Adeleke, O.F.; Dauda, A.O.; Abiodun, O.A.; Kayode, R.M.O.; Adejuyitan, J.A. Flour composition and physicochemical properties of white and yellow bitter yam (Dioscorea dumetorum) starches. Ind. Crops Prod. 2018, 120, 135–139. [Google Scholar] [CrossRef]
- Adeyeye, S.A.O.; Jacob, O.O. Quality assessment and acceptability of pounded yam from different varieties of yam. Nat. Sci. 2014, 12, 115–119. [Google Scholar]
- Sharlina, M.S.E.; Yaacob, W.A.; Mat, A.; Fazry, S.; Joe, S.; Abdullah, S.; Noordin, A.; Kumaran, M. Physicochemical Properties of Starch from Dioscorea pyrifolia tubers. Food Chem. 2017, 220, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Soto, L.J.M.; González, J.V.; Nicanor, A.B.; Cruz, L.G.; Fernández, J.Y. Chemical characterisation and nutritional evaluation of mountain yam (Dioscorea remotiflora Kunth) tubers. Adv. Biores. 2014, 5, 153–160. [Google Scholar] [CrossRef]
- Alamu, E.O.; Maziya-Dixon, B.; Okonkwo, C.C.; Asiedu, R. Physicochemical and bioactive properties of selected white yam (Dioscorea rotundata) varieties adapted to riverine areas of Nigeria. Afr. J. Food Sci. 2014, 8, 402–409. [Google Scholar] [CrossRef]
- Ahmadu, U.; Agbomeji, O.; Yahya, M.; Odeku, A. Physicochemical and material properties of starches from three cultivars of Dioscorea rotundata. Agric. Nat. Resour. 2018, 52, 79–83. [Google Scholar] [CrossRef]
- Mulualem, T.; Mekbib, F.; Hussein, S.; Gebre, E. Analysis of biochemical composition of yams (Dioscorea spp.) landraces from Southwest Ethiopia. Agrotechnology 2018, 7, 1–8. [Google Scholar] [CrossRef]
- Alinnor, I.J.; Akalezi, C.O. Proximate and mineral compositions of Dioscorea rotundata (white yam) and Colocasi esculenta (white cocoyam). Pak. J. Nutr. 2010, 9, 998–1001. [Google Scholar]
- Lawal, O.O.; Agiang, M.A.; Eteng, M.U. Proximate and anti-nutrient composition of white Guinea yam (Dioscorea rotundata) diets consumed in Ibarapa, South West region of Nigeria. J. Nat. Prod. Plant Res. 2012, 2, 256–260. [Google Scholar]
- Washaya, S.; Mupangwa, J.F.; Muranda, E. Nutritional value and utilization of Yams (Dioscorea steriscus) by residents of Bindura town high density suburbs, Zimbabwe. Adv. Agric. 2016, 2016, 5934738. [Google Scholar] [CrossRef] [Green Version]
- Pérez, E.; Gibert, O.; Rolland-Sabaté, A.; Jiménez, Y.; Sánchez, T.; Giraldo, A.; Pontoire, B.; Guilois, S.; Lahon, M.C.; Reynes, M.; et al. Physicochemical, functional, and macromolecular properties of waxy yam starches discovered from “mapuey” (Dioscorea trifida) Genotypes in the Venezuelan Amazon. J. Agric. Food Chem. 2011, 59, 263–273. [Google Scholar] [CrossRef]
- Gutiérrez, T.J.; Pérez, E.; Guzmán, R.; Tapia, M.S.; Famá, L. Physicochemical and Functional Properties of Native and Modified by Crosslinking, Dark-Cush-Cush Yam (Dioscorea Trifida) and Cassava (Manihot Esculenta) Starch. J. Polym. Biopolym. Phys. Chem. 2014, 2, 1–5. [Google Scholar] [CrossRef]
- Afiukwa, C.A.; Ogah, O.; Ugwu, O.P.C.; Oguguo, J.O.; Ali, F.U.; Ossai, E.C. Nutritional and antinutritional characterization of two wild yam species from Abakaliki, Southeast Nigeria. Res. J. Pharm. Biol. Chem. Sci. 2013, 4, 840–848. [Google Scholar]
- Zhu, F. Isolation, composition, structure, properties, modifications, and uses of yam starch. Compr. Rev. Food Sci. Food Saf. 2015, 14, 357–386. [Google Scholar] [CrossRef]
- Afoakwa, E.O.; Polycarp, D.; Budu, A.S.; Mensah-brown, H.; Otoo, E. Variability in biochemical composition and cell wall constituents among seven varieties in Ghanaian yam (Dioscorea sp.) germplasm. Afr. J. Food Agric. Nutr. Dev. 2013, 13, 8106–8127. [Google Scholar]
- Wireko-Manu, F.D.; Oduro, I.; Ellis, W.O.; Asiedu, R.; Maziya-Dixon, B. Food quality changes in water yam (Dioscorea alata) during growth and storage. Asian J. Agric. Food Sci. 2013, 01, 66–72. [Google Scholar]
- Van Eck, H.J. Genetics of morphological and tuber traits. In Potato Biology and Biotechnology. Advances and Perspectives; Vreugdenhil, D., Bradshaw, J., Gebhardt, C., Govers, F., Mackerron, D.K.L., Taylor, M.A., Ross, H.A., Eds.; Elsevier Science B.V.: Amsterdam, The Netherlands, 2007; pp. 91–115. ISBN 978-0-444-51018-1. [Google Scholar]
- Wang, L.; Liu, X.; Wang, J. Structural properties of chemically modified Chinese yam starches and their films. Int. J. Food Prop. 2017, 20, 1239–1250. [Google Scholar] [CrossRef]
- El Seoud, O.A.; Nawaz, H.; Arêas, E.P.G. Chemistry and applications of polysaccharide solutions in strong electrolytes/dipolar aprotic solvents: An overview. Molecules 2013, 18, 1270–1313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otegbayo, B.; Oguniyan, D.; Akinwumi, O. Physicochemical and functional characterization of yam starch for potential industrial applications. Starch Staerke 2014, 66, 235–250. [Google Scholar] [CrossRef]
- Tattiyakul, J.; Naksriarporn, T.; Pradipasena, P.; Miyawaki, O. Effect of moisture on hydrothermal modification of yam Dioscorea hispida Dennst starch. Starch Staerke 2006, 58, 170–176. [Google Scholar] [CrossRef]
- Huang, C.C.; Lin, M.C.; Wang, C.C.R. Changes in morphological, thermal and pasting properties of yam (Dioscorea alata) starch during growth. Carbohydr. Polym. 2006, 64, 524–531. [Google Scholar] [CrossRef]
- Magallanes-Cruz, P.A.; Flores-Silva, P.C.; Bello-Perez, L.A. Starch structure influences its digestibility: A review. J. Food Sci. 2017, 82, 2016–2023. [Google Scholar] [CrossRef] [Green Version]
- Dhingra, D.; Michael, M.; Rajput, H.; Patil, R.T. Dietary fibre in foods: A review. J. Food Sci. Technol. 2012, 49, 255–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Satija, A.; Hu, F.B. Cardiovascular benefits of dietary fiber. Curr. Atheroscler. Rep. 2012, 14, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-Zapata, E.; Viuda-Martos, M.; Fernández-López, J.; Pérez-Alvarez, J.A. Resistant starch as functional ingredient. In Polysaccharides; Ramawat, K.G., Mérillon, J.-M., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 1911–1931. ISBN 978-3-319-16298-0. [Google Scholar]
- Abara, A.E.; Tawo, E.N.; Obi-Abang, M.E.; Obochi, G.O. Dietary fibre components of four common Nigerian Dioscorea species. Pakistan J. Nutr. 2011, 10, 383–387. [Google Scholar] [CrossRef]
- Otegbayo, B.O.; Oguniyan, D.J.; Olunlade, B.A.; Oroniran, O.O.; Atobatele, O.E. Characterizing genotypic variation in biochemical composition, anti-nutritional and mineral bioavailability of some Nigerian yam (Dioscorea spp.) land races. J. Food Sci. Technol. 2018, 55, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Arinathan, V.; Mohan, V.R.; Maruthupandian, A. Nutritional and anti-nutritional attributes of some under–utilized tubers. Trop. Subtrop. Agroecosystems 2009, 10, 273–278. [Google Scholar]
- Ofuya, Z.M.; Akhidue, V. The role of pulses in human nutrition: A review. J. Appl. Sci. Environ. Manag. 2005, 9, 99–104. [Google Scholar] [CrossRef] [Green Version]
- FAO. Roots, Tubers, Plantains and Bananas in Human Nutrition. Available online: http://www.fao.org/3/T0207E00.htm#Contents (accessed on 21 February 2020).
- Chandrasekara, A.; Josheph Kumar, T. Roots and tuber crops as functional foods: A review on phytochemical constituents and their potential health benefits. Int. J. Food Sci. 2016, 2016, 3631647. [Google Scholar] [CrossRef] [Green Version]
- Doss, A.; Tresina, P.S.; Mohan, V.R. Amino acid composition of wild yam (Dioscorea spp.). Food Res. 2019, 3, 617–621. [Google Scholar] [CrossRef]
- Jayakody, L.; Hoover, R.; Liu, Q.; Donner, E. Studies on tuber starches. II. Molecular structure, composition and physicochemical properties of yam (Dioscorea sp.) starches grown in Sri Lanka. Carbohydr. Polym. 2007, 69, 148–163. [Google Scholar] [CrossRef]
- Calder, P.C.; Deckelbaum, R.J. Dietary lipids: More than just a source of calories. Curr. Opin. Clin. Nutr. Metab. Care 1999, 2, 105–107. [Google Scholar] [CrossRef] [PubMed]
- Mondy, N.I.; Mueller, T.O. Potato discoloration in relation to anatomy and lipid composition. J. Food Sci. 1977, 42, 14–18. [Google Scholar] [CrossRef]
- Lewu, M.N.; Adebola, P.; Afolayan, A.J. Comparative assessment of the nutritional value of commercially available cocoyam and potato tuber in South Africa. J. Food Qual. 2010, 33, 461–476. [Google Scholar] [CrossRef]
- Leonel, M.; do Carmo, E.L.; Fernandes, A.M.; Soratto, R.P.; Eburneo, J.A.M.; Garcia, E.L.; Santos, T.P.R. Dos Chemical composition of potato tubers: The effect of cultivars and growth conditions. J. Food Sci. Technol. 2017, 54, 2372–2378. [Google Scholar] [CrossRef] [Green Version]
- Somendrika, M.A.D.; Wickramasinghe, I.; Wansapala, M.A.J.; Peiris, S. Nutritional composition of cassava cultivar “CARI-555”. Pak. J. Nutr. 2017, 16, 482–487. [Google Scholar] [CrossRef]
- Khaw, K.-T.; Barrett-Connor, E. Dietary potassium and stroke-associated mortality. N. Engl. J. Med. 1987, 316, 235–240. [Google Scholar] [CrossRef]
- Neela, S.; Fanta, S.W. Review on nutritional composition of orange—Fleshed sweet potato and its role in management of vitamin A deficiency. Food Sci. Nutr. 2019, 7, 1920–1945. [Google Scholar] [CrossRef] [Green Version]
- Beto, J.A. The role of calcium in human aging. Clin. Nutr. Res. 2015, 4, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Wosje, K.S.; Specker, B.L. Role of calcium in bone health during childhood. Nutr. Rev. 2000, 58, 253–268. [Google Scholar] [CrossRef]
- Karp, H.J.; Vaihia, K.P.; Karkkainen, M.U.M.; Niemisto, M.J.; Lamberg-Allardt, C.J.E. Acute effects of different phosphorus sources on calcium and bone metabolism in young women: A whole-foods approach. Calcif. Tissue Int. 2007, 80, 251–258. [Google Scholar] [CrossRef]
- Saris, N.L.; Mervaala, E.; Karppanen, H.; Khawaja, J.A.; Lewenstam, A. Magnesium: An update on physiological, clinical and analytical aspects. Clin. Chim. Acta 2000, 294, 1–26. [Google Scholar] [CrossRef]
- Sales, C.H.; Pedrosa, L.D.F.C. Magnesium and diabetes mellitus: Their relation. Clin. Nutr. 2006, 25, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Bashiri, A.; Burstein, E.; Sheiner, E.; Mazor, M. Anemia during pregnancy and treatment with intravenous iron: Review of the literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 2003, 110, 2–7. [Google Scholar] [CrossRef]
- Bernhoft, A. A brief review on bioactive compounds in plants. In Bioactive Compounds in Plants—Benefits and Risks for Man and Animals; Bernhoft, A., Ed.; Det Norske Videnskaps-Akademi: Oslo, Norway, 2010; pp. 13–14. ISBN 9788270995837. [Google Scholar]
- Correia, R.T.P.; Borges, K.C.; Medeiros, M.F.; Genovese, M.I. Bioactive compounds and phenolic-linked functionality of powdered tropical fruit residues. Food Sci. Technol. Int. 2012, 18, 539–547. [Google Scholar] [CrossRef]
- Price, E.J.; Bhattacharjee, R.; Lopez-Montes, A.; Fraser, P.D. Metabolite profiling of yam (Dioscorea spp.) accessions for use in crop improvement programmes. Metabolomics 2017, 13, 144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, L.; Ren, S.; Xu, F.; Ma, Z.; Liu, X.; Wang, L. Recent advances in the pharmacological activities of dioscin. Hindawi BioMed Res. Int. 2019, 2019, 5763602. [Google Scholar] [CrossRef] [Green Version]
- Padhan, B.; Biswas, M.; Panda, D. Nutritional, anti-nutritional and physico-functional properties of wild edible yam (Dioscorea spp.) tubers from Koraput, India. Food Biosci. 2020, 34, 100527. [Google Scholar] [CrossRef]
- Adeosun, O.M.; Arotupin, D.J.; Toba, O.A.; Adewole, A. Antibacterial activities and phytochemical properties of extracts of Dioscorea bulbifera Linn (Air Potatoe) tubers and peels against some pathogenic bacteria. J. Phytopharm. 2016, 5, 20–26. [Google Scholar]
- Poornima, G.N.; Ravishankar, R.V. Evaluation of phytonutrients and vitamin contents in a wild yam, Dioscorea belophylla (Prain) Haines. Afr. J. Biotechnol. 2009, 8, 971–973. [Google Scholar] [CrossRef]
- Kwon, Y.K.; Jie, E.Y.; Sartie, A.; Kim, D.J.; Liu, J.R.; Min, B.W.; Kim, S.W. Rapid metabolic discrimination and prediction of dioscin content from African yam tubers using Fourier transform-infrared spectroscopy combined with multivariate analysis. Food Chem. 2015, 166, 389–396. [Google Scholar] [CrossRef]
- Sonibare, M.A.; Abegunde, R.B. In vitro antimicrobial and antioxidant analysis of Dioscorea dumetorum (Kunth) Pax and Dioscorea hirtiflora (Linn.) and their bioactive metabolites from Nigeria. J. Appl. Biosci. 2012, 51, 3583–3590. [Google Scholar]
- Price, E.J.; Wilkin, P.; Sarasan, V.; Fraser, P.D. Metabolite profiling of Dioscorea (yam) species reveals underutilised biodiversity and renewable sources for high-value compounds. Sci. Rep. 2016, 6, 29136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wadkar, K.A.; Magdum, C.S.; Patil, S.S.; Naikwade, N.S. Anti-diabetic potential and Indian medicinal plants. J. Herb. Med. Toxicol. 2014, 2, 45–50. [Google Scholar]
- Chiu, C.-S.; Deng, J.-S.; Chang, H.-Y.; Chen, Y.-C.; Lee, M.-M.; Hou, W.-C.; Lee, C.-Y.; Huang, S.-S.; Huang, G.-J. Antioxidant and anti-inflammatory properties of Taiwanese yam (Dioscorea japonica Thunb. var. pseudojaponica (Hayata) Yamam.) and its reference compounds. Food Chem. 2013, 141, 1087–1096. [Google Scholar] [CrossRef]
- Dong, M.; Feng, X.-Z.; Wu, L.-J.; Wang, B.-X.; Ikejima, T. Two New Steroidal Saponins from the Rhizomes of Dioscorea panthaica and their Cytotoxic Activity. Planta Med 2001, 67, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, X.; Mao, X.; Huang, H.; Wang, T.; Qu, Z.; Miao, J.; Gao, W. Effects of drying processes on starch-related physicochemical properties, bioactive components and antioxidant properties of yam flours. Food Chem. 2017, 224, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Tabopda, T.K.; Mitaine-Offer, A.C.; Tanaka, C.; Miyamoto, T.; Mirjolet, J.F.; Duchamp, O.; Ngadjui, B.T.; Lacaille-Dubois, M.A. Steroidal saponins from dioscorea preussii. Fitoterapia 2014, 97, 198–203. [Google Scholar] [CrossRef]
- Escobar-Sánchez, M.L.; Sánchez-Sánchez, L.; Sandoval-Ramírez, J. Steroidal saponins and cell death in cancer. In Cell Death; Ntuli, T., Ed.; InTechOpen: London, UK, 2016; pp. 331–351. [Google Scholar] [CrossRef] [Green Version]
- Ali, Z.; Smillie, T.J.; Khan, I.A. Cholestane steroid glycosides from the root of Dioscorea villosa (wild yam). Carbohydr. Res. 2013, 370, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.C.; Lin, J.T.; Liu, S.C.; Yang, D.J. A spirostanol glycoside from wild yam (Diosccorea villosa) extract and its cytostatic activity on three cancer cells. J. Food Drug Anal. 2007, 15, 310–315. [Google Scholar]
- Yin, J.; Kouda, K.; Tezuka, Y.; Tran, Q.L.; Miyahara, T.; Chen, Y.; Kadota, S. Steroidal glycosides from the rhizomes of Dioscorea spongiosa. J. Nat. Prod. 2003, 66, 646–650. [Google Scholar] [CrossRef]
- Raju, J.; Rao, C. V Diosgenin, a steroid saponin constituent of yams and fenugreek: Emerging evidence for applications in medicine. In Bioactive Compounds in Phytomedicine; Rasooli, I., Ed.; IntechOpen: London, UK, 2012; pp. 125–142. [Google Scholar] [CrossRef] [Green Version]
- Cai, B.; Zhang, Y.; Wang, Z.; Xu, D.; Jia, Y.; Guan, Y.; Liao, A.; Liu, G.; Chun, C.; Li, J. Review article therapeutic potential of diosgenin and its major derivatives against neurological diseases: Recent advances. Oxid. Med. Cell. Longev. 2020, 2020, 3153082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balandrin, M.F.; Klocke, J.A.; Wurtele, E.S.; Bollinger, W.H. Natural plant chemicals: Sources of industrial and medicinal materials. Science 1985, 228, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Dong, C.; Huang, C.; Zhu, Y. Enhanced production of diosgenin from Dioscorea zingiberensis in mixed culture solid state fermentation with Trichoderma reesei and Aspergillus fumigatus. Biotechnol. Biotechnol. Equip. 2015, 29, 773–778. [Google Scholar] [CrossRef] [Green Version]
- Lubbe, A.; Verpoorte, R. Cultivation of medicinal and aromatic plants for specialty industrial materials. Ind. Crops Prod. 2011, 34, 785–801. [Google Scholar] [CrossRef]
- Chen, Y.; Tang, Y.; Yu, S.; Han, Y.; Kou, J.; Liu, B.; Yu, B. Advances in the pharmacological activities and mechanisms of diosgenin. Chin. J. Nat. Med. 2015, 13, 578–587. [Google Scholar] [CrossRef]
- Jesus, M.; Martins, A.P.J.; Gallardo, E.; Silvestre, S. Diosgenin: Recent Highlights on Pharmacology and Analytical Methodology. J. Anal. Methods Chem. 2016, 2016, 4156293. [Google Scholar] [CrossRef]
- Tada, Y.; Kanda, N.; Haratake, A.; Tobiishi, M.; Uchiwa, H.; Watanabe, S. Novel effects of diosgenin on skin aging. Steroids 2009, 74, 504–511. [Google Scholar] [CrossRef]
- Yi, T.; Fan, L.L.; Chen, H.-L.; Zhu, G.Y.; Suen, H.M.; Tang, Y.-N.; Zhu, L.; Chu, C.; Zhao, Z.-Z.; Chen, H.-B. Comparative analysis of diosgenin in Dioscorea species and related medicinal plants by UPLC-DAD-MS. BMC Biochem. 2014, 15, 9. [Google Scholar] [CrossRef] [Green Version]
- Kaimal, A.; Kemper, K.J. Wild Yam (Dioscoreaceae). In The Longwood Herbal Task Force and Center for Holistic Pediatric Education and Research; Syosset Public Library: Syossset, NY, USA, 1999. [Google Scholar] [CrossRef]
- Xiang, L.; JianZhong, M.; YunDong, S. Research progress and prospects of Dioscorea and diosgenin. Chem. Ind. For. Prod. 2010, 30, 107–112. [Google Scholar]
- de Lourdes Contreras-Pacheco, M.; Santacruz-Ruvalcaba, F.; García-Fajardo, J.A.; de Jesús Sánchez G, J.; Ruíz L, M.A.; Estarrón-Espinosa, M.; Castro-Castro, A. Diosgenin quantification, characterisation and chemical composition in a tuber collection of Dioscorea spp. in the state of Jalisco, Mexico. Int. J. Food Sci. Technol. 2013, 48, 2111–2118. [Google Scholar] [CrossRef]
- Huai, Z.P.; Ding, Z.Z.; He, S.A.; Sheng, C.G. Research on correlations between climatic factors and diosgenin content in Dioscorea zingiberensis Wright. Yao Xue Xue Bao 1989, 24, 702–706. [Google Scholar] [PubMed]
- Sautour, M.; Mitaine-Offer, A.C.; Lacaille-Dubois, M.A. The Dioscorea genus: A review of bioactive steroid saponins. J. Nat. Med. 2007, 61, 91–101. [Google Scholar] [CrossRef]
- Hou, W.C.; Chen, H.J.; Lin, Y.H. Dioscorins from different Dioscorea species all exhibit both carbonic anhydrase and trypsin inhibitor activities. Bot. Bull. Acad. Sin. 2000, 41, 191–196. [Google Scholar] [CrossRef]
- Conlan, S.; Griffiths, L.A.; Turner, M.; Fido, R.; Tatham, A.; Ainsworth, C.; Shewry, P. Characterisation of the yam tuber storage protein dioscorin. J. Plant Physiol. 1998, 153, 25–31. [Google Scholar] [CrossRef]
- Conlan, R.S.; Griffiths, L.A.; Napier, J.A.; Shewry, P.R.; Mantell, S.; Ainsworth, C. Isolation and characterisation of cDNA clones representing the genes encoding the major tuber storage protein (dioscorin) of yam (Dioscorea cayenensis Lam.). Plant Mol. Biol. 1995, 28, 369–380. [Google Scholar] [CrossRef]
- Liao, Y.H.; Tseng, C.Y.; Chen, W. Structural characterization of dioscorin, the major tuber protein of yams, by near infrared Raman spectroscopy. J. Phys. Conf. Ser. 2006, 28, 119–122. [Google Scholar] [CrossRef]
- Liao, Y.H.; Wang, C.H.; Tseng, C.Y.; Chen, H.L.; Lin, L.L.; Chen, W. Compositional and conformational analysis of yam proteins by near infrared Fourier transform Raman spectroscopy. J. Agric. Food Chem. 2004, 52, 8190–8196. [Google Scholar] [CrossRef]
- Hou, W.C.; Liu, J.S.; Chen, H.J.; Chen, T.E.; Chang, C.F.; Lin, Y.H. Dioscorin, the major tuber storage protein of yam (Dioscorea batatas Decne) with carbonic anhydrase and trypsin inhibitor activities. J. Agric. Food Chem. 1999, 47, 2168–2172. [Google Scholar] [CrossRef]
- Hou, W.C.; Chen, H.J.; Lin, Y.H. Dioscorins, the major tuber storage proteins of yam (Dioscorea batatas Decne), with dehydroascorbate reductase and monodehydroascorbate reductase activities. Plant Sci. 1999, 149, 151–156. [Google Scholar] [CrossRef]
- Gaidamashvili, M.; Ohizumi, Y.; Iijima, S.; Takayama, T.; Ogawa, T.; Muramoto, K. Characterization of the yam tuber storage proteins from Dioscorea batatas exhibiting unique lectin activities. J. Biol. Chem. 2004, 279, 26028–26035. [Google Scholar] [CrossRef] [Green Version]
- Hou, W.C.; Lee, M.H.; Chen, H.J.; Liang, W.L.; Han, C.H.; Liu, Y.W.; Lin, Y.H. Antioxidant activities of dioscorin, the storage protein of yam (Dioscorea batatas Decne) tuber. J. Agric. Food Chem. 2001, 49, 4956–4960. [Google Scholar] [CrossRef]
- Liu, Y.H.; Liang, H.J.; Cheng, H.C.; Liu, Y.W.; Hou, W.C. Comparisons of in vitro antioxidant activities of storage proteins in tuber of two Dioscorea species. Bot. Stud. 2006, 47, 231–237. [Google Scholar]
- Hsu, F.L.; Lin, Y.H.; Lee, M.H.; Lin, C.L.; Hou, W.C. Both dioscorin, the tuber storage protein of yam (Dioscorea alata cv. Tainong No. 1), and its peptic hydrolysates exhibited angiotensin converting enzyme inhibitory activities. J. Agric. Food Chem. 2002, 50, 6109–6113. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.L.; Hsu, Y.H.; Lee, P.Y.; Hou, W.C.; Hung, L.C.; Lin, C.H.; Chen, C.M.; Huang, Y.J. Dioscorin isolated from Dioscorea alata activates TLR4-signaling pathways and induces cytokine expression in macrophages. Biochem. Biophys. Res. Commun. 2006, 339, 137–144. [Google Scholar] [CrossRef]
- Moreira, R.; Pereira, D.M.; Valentão, P.; Andrade, P.B. Pyrrolizidine alkaloids: Chemistry, pharmacology, toxicology and food safety. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cushnie, T.P.T.; Cushnie, B.; Lamb, A.J. Alkaloids: An overview of their antibacterial, antibiotic-enhancing and antivirulence activities. Int. J. Antimicrob. Agents 2014, 44, 377–386. [Google Scholar] [CrossRef]
- Kurek, J. Introductory chapter: Alkaloids—Their importance in nature and for human life. In Alkaloids—Their Importance in Nature and for Human Life; Kurek, J., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Astonishing diversity of natural surfactants: 7. Biologically active hemi- and monoterpenoid glycosides. Lipids 2006, 41, 1–27. [Google Scholar] [CrossRef]
- Vlietinck, A.; De Bruyne, T.; Vanden Berghe, D. Plant substances as antiviral agents. Curr. Org. Chem. 1997, 1, 307–344. [Google Scholar] [CrossRef] [Green Version]
- Adebowale, A.R.A.; Wahab, A.B.; Sobukola, P.O.; Obadina, A.O.; Kajihausa, E.O.; Adegunwa, O.M.; Sanni, O.L.; Tomlins, K. The antinutritional and vitamin composition of high-quality yam flour as affected by yam specie, pretreatment, and drying method. Food Sci. Nutr. 2018, 6, 1985–1990. [Google Scholar] [CrossRef]
- Senanayake, S.; Ranaweera, K.; Bamunuarachchi, A.; Gunaratne, A. Proximate analysis and phytochemical and mineral constituents in four cultivars of yams and tuber crops in Sri Lanka. Trop. Agric. Res. Ext. 2013, 15, 32. [Google Scholar] [CrossRef]
- Broadbent, J.L.; Schnieden, H. A comparison of some pharmacological properties of dioscorine and dioscine. Br. J. Pharmacol. Chemother. 1958, 13, 213–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banaag, A.; Honda, H.; Shono, T. Effects of alkaloids from yam, Dioscorea hispida Schlussel, on feeding and development of larvae of the diamondback moth, Plutella xylostella (Lepidoptera: Yponomeutidae). Appl. Entomol. Zool. 1997, 32, 119–126. [Google Scholar] [CrossRef] [Green Version]
- Havsteen, B. Flavonoids, a class of natural products of high pharmacological potency. Biochem. Pharmacol. 1983, 32, 1141–1148. [Google Scholar] [CrossRef]
- Brown, J.P. A review of the genetic effects of naturally occurring flavonoids, anthraquinones and related compounds. Mutat. Res. 1980, 75, 243–277. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [Green Version]
- Sangeetha, S.K.S.; Umamaheswari, S.; Reddy, M.U.; Narayana, K.S. Flavonoids: Therapeutic potential of natural pharmacological agents. Int. J. Pharm. Sci. Res. 2016, 7, 3924–3930. [Google Scholar] [CrossRef]
- Padhan, B.; Nayak, J.K.; Panda, D. Natural antioxidant potential of selected underutilized wild yams (Dioscorea spp.) for health benefit. J. Food Sci. Technol. 2020, 57, 2370–2376. [Google Scholar] [CrossRef]
- Sobiesiak, M. Chemical structure of phenols and its consequence for sorption processes. In Phenolic Compounds—Natural Sources, Importance and Applications; Soto-Hernandez, M., Palma-Tenango, M., Garcia-Mateos, R., Eds.; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef] [Green Version]
- Kougan, G.B.; Tabopda, T.; Kuete, V.; Verpoorte, R. Simple phenols, phenolic acids, and related esters from the medicinal plants of Africa. In Medicinal Plant Research in Africa; Kuete, V., Ed.; Elsevier: Oxford, UK, 2013; pp. 225–249. ISBN 978-0-12-405927-6. [Google Scholar]
- Yang, C.S.; Landau, J.M.; Huang, M.-T.; Newmark, H.L. Inhibition of carcinogenesis by dietary polyphenolic compounds. Annu. Rev. Nutr. 2001, 21, 381–406. [Google Scholar] [CrossRef] [Green Version]
- Pereira, D.M.; Valentão, P.; Pereira, J.A.; Andrade, P.B. Phenolics: From chemistry to biology. Molecules 2009, 14, 2202–2211. [Google Scholar] [CrossRef]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for analysis of plant phenolic compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef]
- Zhao, C.; Zhou, B.; Miao, J.; Li, X.; Jing, S.; Zhang, D.; Wang, J.Y.; Li, X.; Huang, L.; Gao, W. Multicomponent analysis and activities for evaluation of Dioscorea oppositifolia and Dioscorea hamiltonii. Food Agric. Immunol. 2019, 30, 1148–1161. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Tian, H.; Zhan, P.; Du, F.; Zong, A.; Xu, T. Isolation and identification of phenolic compounds in Chinese purple yam and evaluation of antioxidant activity. LWT Food Sci. Technol. 2018, 96, 161–165. [Google Scholar] [CrossRef]
- Ozo, O.N.; Caygill, J.C.; Coursey, D.G. Phenolics of five yam (Dioscorea) species. Phytochemistry 1984, 23, 329–331. [Google Scholar] [CrossRef]
- Graham-Acquaah, S.; Ayernor, G.S.; Bediako-Amoa, B.; Saalia, F.K.; Afoakwa, E.O. Spatial distribution of total phenolic content, enzymatic activities and browning in white yam (Dioscorea rotundata) tubers. J. Food Sci. Technol. 2014, 51, 2833–2838. [Google Scholar] [CrossRef] [PubMed]
- Soibam, H.; Ayam, V.S.; Mitra, S. Evaluation of the nutritional and antinutritional components of twelve Evaluation of the nutritional and antinutritional components of twelve Dioscorea cultivars. Pharma Innov. J. 2017, 6, 465–469. [Google Scholar]
- Chen, H.; Liu, J.; Cui, K.; Lu, Q.; Wang, C.; Wu, H.; Yang, Z.; Ding, W.; Shao, S.; Wang, H.; et al. Molecular mechanisms of tannin accumulation in Rhus galls and genes involved in plant-insect interactions. Sci. Adv. 2018, 8, 9841. [Google Scholar] [CrossRef]
- Chung, K.-T.; Wong, T.Y.; Wei, C.-I.; Huang, Y.-W.; Lin, Y. Tannins and Human Health: A Review. Crit. Rev. Food Sci. Nutr. 1998, 38, 421–464. [Google Scholar] [CrossRef] [PubMed]
- Smeriglio, A.; Barreca, D.; Bellocco, E.; Trombetta, D. Proanthocyanidins and hydrolysable tannins: Occurrence, dietary intake and pharmacological effects. Br. J. Pharmacol. 2017, 174, 1244–1262. [Google Scholar] [CrossRef] [Green Version]
- Kumar, V.; Sinha, A.K.; Makkar, H.P.S.; Becker, K. Dietary roles of phytate and phytase in human nutrition: A review. Food Chem. 2010, 120, 945–959. [Google Scholar] [CrossRef]
- Singh, M.; Krikorian, A.D. Inhibition of trypsin activity in vitro by phytate. J. Agric. Food Chem. 1982, 30, 799–800. [Google Scholar] [CrossRef]
- Noonan, S.C.; Hons, G.P.S.; Nutr, N.Z.R. Oxalate content of foods and its effect on humans. Asia Pac. J. Clin. Nutr. 1999, 8, 64–74. [Google Scholar] [PubMed]
- Widiastuti, V.; Ernawati, E.; Fatmadewi, V.; Anindyajati, S.; Faradina, S.N. Analysis of cyanide content on yams using spectrophotometry methods. Indones. J. Chem. Environ. 2017, 1, 7–14. [Google Scholar]
- Akin-Idowu, P.E.; Odunola, O.A.; Asiedu, R.; Maziya-Dixon, B.; Uwaifo, A.O. Variation in nutrient and antinutrient contents of tubers from yellow yam (Dioscorea cayenensis) genotypes grown at two locations. J. Food Agric. Environ. 2008, 6, 95–100. [Google Scholar]
- Gurib-Fakim, A. Medicinal plants: Traditions of yesterday and drugs of tomorrow. Mol. Asp. Med. 2006, 27, 1–93. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Gurjar, S.; Pandey, D.K. Medicinal aspect of saponins shows their wide range of pharmacological/biological activities. Pharmacologyonline 2010, 2, 579–584. [Google Scholar]
- Pinzón-Rico, Y.A.; Raz, L. Commercialization of Andean wild yam species (Dioscorea L.) for medicinal use in Bogotá, DC, Colombia. Econ. Bot. 2017, 71, 45–57. [Google Scholar]
- Riley, C.K.; Wheatley, A.O.; Asemota, H.N. Isolation and characterization of starches from eight Dioscorea alata cultivars grown in Jamaica. Afr. J. Biotechnol. 2006, 5, 1528–1536. [Google Scholar] [CrossRef]
- Lalfakzuala, R.; Lalramnghinglova, H.; Kayang, H. Ethnobotanical usages of plants in western Mizoram. Indian J. Tradit. Knowl. 2007, 6, 486–493. [Google Scholar]
- Jadhav, V.D.; Mahadkar, S.D.; Valvi, S.R. Documentation and ethnobotanical survey of wild edible plants from Kolhapur district. Recent Res. Sci. Technol. 2011, 3, 58–63. [Google Scholar]
- Sakthidevi, G.; Mohan, V.R. Total phenolic, flavonoid contents and in vitro antioxidant activity of Dioscorea alata l. tuber. J. Pharm. Sci. Res. 2013, 5, 115–119. [Google Scholar]
- Dey, P.; Chowdhuri, S.R.; Sarkar, M.P.; Chaudhuri, T.K. Evaluation of anti-inflammatory activity and standardisation of hydro-methanol extract of underground tuber of Dioscorea alata. Pharm. Biol. 2016, 54, 1474–1482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, S.; Dash, S.K.; Padhy, S.N. Ethno-medicinal Informations from Orissa State, India, A Review. J. Hum. Ecol. 2003, 14, 165–227. [Google Scholar] [CrossRef]
- Samanta, A.K.; Biswas, K.K. Climbing plants with special reference to their medicinal importance from Midnapore Town and its adjoining areas. J. Econ. Taxon. Bot. 2009, 33, 180–188. [Google Scholar]
- Aiyeloja, A.A.; Bello, O.A. Ethnobotanical potentials of common herbs in Nigeria: A case study of Enugu state. Educ. Res. Rev. 2006, 1, 16–22. [Google Scholar]
- Michel, J.; Duarte, R.E.; Yao, P.; Bolton, J.L.; Huang, Y.; Armando, C.; Veliz, M.; Soejarto, D.D.; Mahady, G.B. Medical potential of plants used by the Q’eqchi Maya of Livingston, Guatemala for the treatment of women’s health complaints. J. Ethnopharmacol. 2007, 114, 92–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srivastava, R.C.; Community, N. Traditional knowledge of Nyishi (Daffla) tribe of Arunachal Pradesh. Indian J. Tradit. Knowl. 2010, 9, 26–37. [Google Scholar]
- Neha, B.; Arun, K.; Preeti, K. Choudary Alka Evaluation of therapeutic potential of Dioscorea bulbifera tubers on learning and memory impairment in high fat diet (HFD) and ICV streptozotocin (STZ)—Induced experimental dementia in mice. Glob. J. Res. Med. Plants Indig. Med. 2013, 2, 808–823. [Google Scholar]
- Ghosh, S.; More, P.; Nitnavare, R.; Jagtap, S.; Chippalkatti, R.; Derle, A.; Kitture, R.; Asok, A.; Kale, S.; Singh, S.; et al. Antidiabetic and antioxidant properties of copper nanoparticles synthesized by medicinal plant Dioscorea bulbifera. J. Nanomed. Nanotechnol. 2015, S6, 007. [Google Scholar] [CrossRef] [Green Version]
- Murray, R.D.H.; Jorge, Z.D.; Khan, N.H.; Shahjahan, M.; Quaisuddin, M. Diosbulbin D and 8-epidiosbulbin E acetate, norclerodane diterpenoids from Dioscorea bulbifera tubers. Phytochemistry 1984, 23, 623–625. [Google Scholar] [CrossRef]
- Gao, H.Y.; Shui, A.L.; Chen, Y.H.; Zhang, Z.Y.; Wu, L.J. The chemical compositions of Dioscorea bulbifera. J. Shenyang Pharm. Univ. 2003, 20, 178–180. [Google Scholar]
- Tapondjou, A.L.; Jenett-siems, K.; Böttger, S.; Melzig, M.F. Steroidal saponins from the flowers of Dioscorea bulbifera var. sativa. Phytochemistry 2013, 95, 341–350. [Google Scholar] [CrossRef]
- Tang, Z.; Zhou, Y.; Zeng, Y.; Zang, S.; He, P.; Fang, Y. Capillary electrophoresis of the active ingredients of Dioscorea bulbifera L. and its medicinal preparations. Chromatographia 2006, 63, 617–622. [Google Scholar] [CrossRef]
- Abhyankar, R.K.; Upadhyay, R. Ethnomedicinal studies of tubers of Hoshangabad M. P. Bull. Environ. Pharmacol. Life Sci. 2011, 1, 57–59. [Google Scholar]
- Punjani, B.L. Some less known ethnomedicinal plants used by Maldaris of Sasan Gir forest in Junagardh district, Gujarat. Ethnobotany 2007, 19, 116–119. [Google Scholar]
- Singh, N.; Pangtey, Y.P.S.; Khatoon, S.; Rawat, A.K.S. Some ethnomedicinal plants of Ranikhet region, Uttaranchal. J. Econ. Taxon. Bot. 2009, 33, 198–204. [Google Scholar]
- Swarnkar, S.; Katewa, S.S. Ethnobotanical observation on tuberous plants from tribal area of Rajasthan (India). Ethnobot. Leafl. 2008, 12, 647–666. [Google Scholar]
- Kamble, S.Y.; Patil, S.R.; Sawant, P.S.; Sawant, S.; Pawar, S.G.; Singh, E.A. Studies on plant used in traditional medicine by Bhilla tribe of Maharashtra. Indian J. Tradit. Knowl. 2010, 9, 591–598. [Google Scholar]
- Mishra, S.; Swain, S.; Chaudhary, S.S.; Ray, T. Wild edible tubers (Dioscorea spp.) and their contribution to the food security of tribes of Jaypore tract, Orissa, India. Plant Genet. Resour. 2008, 156, 63–67. [Google Scholar]
- Sahu, S.C.; Dhal, N.K.; Mohanty, R.C. Potential medicinal plants used by the Tribal of Deogarh District, Orissa, India. Stud. Ethno Med. 2010, 4, 53–61. [Google Scholar] [CrossRef]
- Radha, B.; Singh, R.D.; Tiwari, J.K.; Tiwari, P.; Gairola, A. Wild edible plant resources of the lohba range of kedarnath forest division (KFD), Garhwal Himalaya, India. Int. Res. J. Biol. Sci. 2013, 2, 65–73. [Google Scholar]
- Ghosh, S.; Derle, A.; Ahire, M.; More, P.; Jagtap, S.; Phadatare, S.D.; Patil, A.B.; Jabgunde, A.M.; Sharma, G.K.; Shinde, V.S.; et al. Phytochemical analysis and free radical scavenging activity of medicinal plants Gnidia glauca and Dioscorea bulbifera. PLoS ONE 2013, 8, e82529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kosalge, S.B.; Fursule, R.A. Investigation of anthelmintic potential of some plants claimed by tribals of satpuda hills. Int. J. PharmTech Res. 2009, 1, 68–72. [Google Scholar]
- Rahmatullah, M.; Noman, A.; Hossan, S.M.; Harun-Or-Rashid, M.; Rahman, T.; Chowdhury, M.H.; Jahan, R. A survey of medicinal plants in two areas of Dinajpur District, Bangladesh Including Plants which can be Used as functional foods. Am. J. Sustain. Agric. 2009, 3, 862–876. [Google Scholar]
- Girach, R.D.; Shaik, A.A.; Singh, S.S.; Ahmad, M. The medicinal flora of Similipahar forests, Orissa State, India. J. Ethnopharmacol. 1999, 65, 165–172. [Google Scholar] [CrossRef]
- Hu, K.; Yao, X. The cytotoxicity of protoneodioscin (NSC-698789), a furostanol saponin from the rhizomes of Dioscorea collettii var. hypoglauca, against human cancer cells in vitro. Phytomedicine 2002, 9, 560–565. [Google Scholar] [CrossRef]
- Dweck, A.C. The Wild Yam—A review. Pers. Care Mag. 2002, 3, 7–9. [Google Scholar]
- Kumari, P.; Joshi, G.C.; Tewari, L.M. Indigenous uses of threatened ethnomedicinal plants used to cure different diseases by ethnic peoples of Almora district of Western Himalaya. Int. J. Ayurvedic Herb. Med. 2012, 2, 661–678. [Google Scholar]
- Dangwal, L.R.; Chauhan, A.S. Dioscorea deltoidea wall. ex Griseb. A highly threatened Himalayan medicinal plant: An overview. Int. J. Pharma Bio Sci. 2015, 6, 452–460. [Google Scholar]
- Jain, S.K. Medicinal Plants, 2nd ed.; National Book Trust: New Delhi, India, 1975. [Google Scholar]
- Etim, N.-A.A.; Thompson, D.; Onyenweaku, C.E. Measuring efficiency of yam (Dioscorea spp.) production among resource poor farmers in rural Nigeria. J. Agric. Food Sci. 2013, 1, 42–47. [Google Scholar]
- Iwu, M.M.; Okunji, C.O.; Ohiaeri, G.O.; Akah, P.; Corley, D.; Tempesta, M.S. Hypoglycaemic activity of dioscoretine from tubers of Dioscorea dumetorum in normal and alloxan diabetic rabbits. Planta Med. 1990, 56, 264–267. [Google Scholar] [CrossRef]
- Malviya, N.; Jain, S.; Malviya, S. Antidiabetic potential of medicinal plants. Acta Pol. Pharm. Drug Res. 2010, 67, 113–118. [Google Scholar]
- Edison, S.; Unnikrishnan, M.; Vimala, B.; Pillai, S.V.; Sheela, M.N.; Sreekumari, M.T.; Abraham, K. Biodiversity of Tropical Tuber Crops in India; National Biodiversity Authority: Chennai, India, 2006. [Google Scholar]
- Sharma, L.N.; Bastakoti, R. Ethnobotany of Dioscorea L. with emphasis on food value in Chepang communities in Dhading district, central Nepal. Bot. Orient. J. Plant Sci. 2009, 6, 12–17. [Google Scholar] [CrossRef] [Green Version]
- Meena, K.L.; Yadav, B.L. Some ethnomedicinal plants used by the Garasia tribe of district Sirohi, Rajasthan. Indian J. Tradit. Knowl. 2011, 10, 354–357. [Google Scholar]
- Moon, E.; Lee, S.O.; Kang, T.H.; Kim, H.J.; Choi, S.Z.; Son, M.W.; Kim, S.Y. Dioscorea extract (DA-9801) modulates markers of peripheral neuropathy in type 2 diabetic db/db mice. Biomol. Ther. 2014, 22, 445–452. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Wang, C.; Chang, C.; Wang, T. Effects of Taiwanese yam (Dioscorea japonica Thunb var. pseudojaponica Yamamoto) on upper gut function and lipid metabolism in balb/c ice. Basic Nutr. Investig. 2003, 19, 646–651. [Google Scholar] [CrossRef]
- Itharat, A.; Houghton, P.J.; Eno-Amooquaye, E.; Burke, P.J.; Sampson, J.H.; Raman, A. In vitro cytotoxic activity of Thai medicinal plants used traditionally to treat cancer. J. Ethnopharmacol. 2004, 90, 33–38. [Google Scholar] [CrossRef]
- Xu, L.; Wang, W. Chinese Materia Medica: Combinations and Applications; Donica Publishing Ltd.: Hertfordshire, UK, 2002. [Google Scholar]
- Tang, Y.N.; Zhang, H.; Li, J.; Yi, T. Pharmacognostic identification of original plants of rhizoma Dioscoreae nipponica and rhizoma Dioscoreae panthaicae. West China J. Pharm. Sci. 2008, 23, 444–447. [Google Scholar]
- Feng, J.; Tang, Y.; Ji, H.; Xiao, Z.; Zhu, L.; Yi, T. Biotransformation of Dioscorea nipponica by rat intestinal microflora and cardioprotective effects of diosgenin. Oxid. Med. Cell. Longev. 2017. [CrossRef]
- Lu, J.; Wong, R.N.S.; Zhang, L.; Wong, R.Y.L.; Ng, T.B.; Lee, K.F.; Zhang, Y.B.; Lao, L.X.; Liu, J.Y.; Sze, S.C.W. Comparative analysis of proteins with stimulating activity on ovarian estradiol biosynthesis from four different Dioscorea species in vitro using both phenotypic and target-based approaches: Implication for treating menopause. Appl. Biochem. Biotechnol. 2016, 180, 79–93. [Google Scholar] [CrossRef]
- Felix, R.; Kumar, N.N.; Raj, T.L.S. Pharmacognostical study of Dioscorea oppositifolia L. Ethnobot. Leafl. 2009, 13, 77–82. [Google Scholar]
- Sheikh, N.; Kumar, Y.; Pfoze, L. Phytochemical screening to validate the ethnobotanical importance of root tubers of Dioscorea species of Meghalaya, North East India Nilofer. J. Med. Plants Stud. Phytochem. 2013, 1, 62–69. [Google Scholar]
- Neelima, M.; Prasad, G.P.; Sudarsanam, G.; Pratap, G.P.; Jyothi, B. Ethnobotanical studies in Rapur forest division of Nellore district in Andhra Pradesh. Life Sci. Leafl. 2011, 11, 333–345. [Google Scholar]
- Zhao, X.-L.; Ma, X.-J.; Jing, W.-G.; Zhang, Q.-W.; Wang, Z.-M.; Liu, A. A new steroidal saponin from Dioscorea panthaica. J. Asian Nat. Prod. Res. 2011, 13, 659–664. [Google Scholar] [CrossRef]
- Rahman, M.A.; Uddin, S.B.; Wilcock, C.C. Medicinal plants used by Chakma tribe in hill tracts districts of Bangladesh. Indian J. Tradit. Knowl. 2007, 6, 508–517. [Google Scholar]
- Choudhary, K.; Singh, M.; Pillai, U. Ethnobotanical survey of rajasthan—An update. Am. J. Bot. 2008, 1, 38–45. [Google Scholar]
- Jiang, Y.; Jiang, D.; Wang, P.; Wu, K.; Wang, X.; Ai, W.; Yu, Z. Effect of sulfur-fumigated Rhizoma Dioscoreae on organ morphology and function in rats. Biomed. Res. 2018, 29, 421–425. [Google Scholar] [CrossRef] [Green Version]
- Pandey, A.K.; Rout, S.D. Ethnobotanical use of plants of Similipal biosphere reserve (Orissa). Ethnobotany 2006, 18, 102–106. [Google Scholar]
- Kumar, S.; Satapathy, M.K. Medicinal plants in an urban environment; plants in an urban environment; herbaceous medicinal flora from the campus of Regional Institute of Education, Bhubaneswar, Odisha. Int. J. Pharm. Life Sci. 2011, 2, 1206–1210. [Google Scholar]
- Zhang, X.; Xue, X.; Zhao, J.; Qian, C.; Guo, Z.; Ito, Y.; Sun, W. Diosgenin attenuates the brain injury induced by transient focal cerebral ischemia-reperfusion in rats. Steroids 2016, 113, 103–112. [Google Scholar] [CrossRef]
- Zhao, W.L.; He, J.Y.; Xi, J.Q. A second discussion on the position of a new species mianbixie. China J. Chin. Mater. Med. 1994, 19, 199–200. [Google Scholar]
- Mander, M.; Mander, J.; Crouch, N.; McKean, S.; Nichols, G. Catchment Action: Growing and Knowing Muthi Plants; ShareNet Booklet, Institute of Natural Resources Scottsville: Pietermaritzburg, South Africa, 1995. [Google Scholar]
- Kala, C.P. Ethnomedicinal botany of the apatani in the Eastern Himalayan region of India. J. Ethnobiol. Ethnomed. 2005, 1, 11. [Google Scholar] [CrossRef] [Green Version]
- Braun, L.; Cohen, M. Herbs and Natural Supplements—An Evidence-Base, 2nd ed.; Elsevier: London, UK, 2007. [Google Scholar]
- Soffa, V.M. Alternatives to hormone replacement for menopause. Altern. Ther. Health Med. 1996, 2, 34–39. [Google Scholar] [PubMed]
- Carroll, D.G. Nonhormonal therapies for hot flashes in menopause. Am. Fam. Physician 2006, 73, 457–464. [Google Scholar] [PubMed]
- Rout, S.D.; Panda, S.K. Ethnomedicinal plant resources of Mayurbhanj district, Orissa. Indian J. Tradit. Knowl. 2010, 9, 68–72. [Google Scholar]
- Zhu, J.; Guo, X.; Fu, S.; Zhang, X.; Liang, X. Characterization of steroidal saponins in crude extracts from Dioscorea zingiberensis C.H. Wright by ultra-performance liquid chromatography/electrospray ionization quadrupole time-of-flight tandem mass spectrometry. J. Pharm. Biomed. Anal. 2010, 53, 462–474. [Google Scholar] [CrossRef] [PubMed]
- Begum, A.T.; Anbazhakan, S. Evaluation of antibacterial activity of the mucilage of Dioscorea esculenta (Lour.) Burkill. Int. J. Mod. Biol. Med. 2013, 4, 140–146. [Google Scholar]
- Kuete, V.; Betrandteponno, R.; Mbaveng, A.T.; Tapondjou, L.A.; Meyer, J.J.M.; Barboni, L.; Lall, N. Antibacterial activities of the extracts, fractions and compounds from Dioscorea bulbifera. Complement. Altern. Med. 2012, 12, 228. [Google Scholar] [CrossRef] [Green Version]
- Xu, L.; Zhou, L.; Zhao, J.; Li, J.; Li, X.; Wang, J. Fungal endophytes from Dioscorea zingiberensis rhizomes and their antibacterial activity. Lett. Appl. Microbiol. 2008, 46, 68–72. [Google Scholar] [CrossRef]
- Lubag, A.J.M.; Laurena, A.C.; Tecson-Mendoza, E.M. Antioxidants of Purple and White Greater Yam (Dioscorea alata L.) Varieties from the Philippines. Philipp. J. Sci. 2008, 137, 61–67. [Google Scholar]
- Chunthorng-Orn, J.; Panthong, S.; Itharat, A. Antimicrobial, antioxidant activities and total phenolic content of Thai medicinal plants used to treat HIV patients. J. Med. Assoc. Thail. 2012, 95, S154–S158. [Google Scholar]
- Murugan, M.; Mohan, V.R. In vitro antioxidant studies of Dioscorea esculenta (Lour). Burkill. Asian Pac. J. Trop. Biomed. 2012, 2, S1620–S1624. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, N.; Nagashima, T. Antioxidative activity of water extracts from the yam (Dioscorea opposita Thunb.) tuber mucilage tororo. Eur. J. Lipid Sci. Technol. 2006, 108, 526–531. [Google Scholar] [CrossRef]
- Theerasin, S.; Baker, A.T. Analysis and identification of phenolic compounds in Dioscorea hispida Dennst. Asian J. Food Agro Ind. 2009, 2, 547–560. [Google Scholar]
- Olayemi, J.O.; Ajaiyeoba, E.O. Anti-inflammatory studies of yam (Dioscorea esculenta) extract on wistar rats. Afr. J. Biotechnol. 2007, 6, 1913–1915. [Google Scholar]
- Park, J.M.; Kim, Y.J.; Kim, J.S.; Han, Y.M.; Kangwan, N.; Hahm, K.B.; Kim, T.S.; Kwon, O.; Kim, E.H. Anti-inflammatory and carbonic anhydrase restoring actions of yam powder (Dioscorea spp) contribute to the prevention of cysteamine-induced duodenal ulcer in a rat model. Nutr. Res. 2013, 33, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Mollica, J.Q.; Cara, D.C.; Auriol, M.D.; Oliveira, V.B.; Cesar, I.C.; Brandoa, M.G.L. Anti-inflammatory activity of American yam Dioscorea trifida L.f. in food allergy induced by ovalbumin in mice. J. Funct. Foods 2013, 5, 1975–1984. [Google Scholar] [CrossRef] [Green Version]
- Itharat, A.; Plubrukarn, A.; Kongsaeree, P.; Bui, T.; Keawpradub, N.; Houghton, P.J. Dioscorealides and dioscoreanone, novel cytotoxic naphthofuranoxepins, and 1,4-phenanthraquinone from Dioscorea membranacea Pierre. Org. Lett. 2003, 5, 2879–2882. [Google Scholar] [CrossRef]
- Itharat, A.; Ooraikul, B. Research on Thai medicinal plants for cancer treatment. In Advances in Medicinal Plant Research; Acharya, S.N., James, E.T., Eds.; Signpost: Kerala, India, 2007; Volume 37/661, pp. 287–317. ISBN 8177362550. [Google Scholar]
- Itharat, A.; Thongdeeying, P.; Ruangnoo, S. Isolation and characterization of a new cytotoxic dihydrophenanthrene from Dioscorea membranacea rhizomes and its activity against five human cancer cell lines. J. Ethnopharmacol. 2014, 156, 130–134. [Google Scholar] [CrossRef]
- Thongdeeying, P.; Itharat, A.; Umehara, K.; Ruangnoo, S. A novel steroid and cytotoxic constituents from Dioscorea membranacea Pierre against hepatocellular carcinoma and cholangiocarcinoma cells. J. Ethnopharmacol. 2016, 194, 91–97. [Google Scholar] [CrossRef]
- Wang, J.-M.; Ji, L.-L.; Branford-White, C.J.; Wang, Z.-Y.; Shen, K.-K.; Liu, H.; Wang, Z.-T. Antitumor activity of Dioscorea bulbifera L. rhizome in vivo. Fitoterapia 2012, 83, 388–394. [Google Scholar] [CrossRef]
- Chan, Y.S.; Ng, T.B. A Lectin with Highly Potent Inhibitory Activity toward Breast Cancer Cells from Edible Tubers of Dioscorea opposita cv. Nagaimo. PLoS ONE 2013, 8, e54212. [Google Scholar] [CrossRef] [PubMed]
- Kaskiw, M.J.; Tassotto, M.L.; Th’ng, J.; Jiang, Z.H. Synthesis and cytotoxic activity of diosgenyl saponin analogues. Bioorganic Med. Chem. 2008, 16, 3209–3217. [Google Scholar] [CrossRef] [PubMed]
- Kaskiw, M.J.; Tassotto, M.L.; Mok, M.; Tokar, S.L.; Pycko, R.; Th’ng, J.; Jiang, Z.H. Structural analogues of diosgenyl saponins: Synthesis and anticancer activity. Bioorganic Med. Chem. 2009, 17, 7670–7679. [Google Scholar] [CrossRef] [PubMed]
- Tong, Q.Y.; Qing, Y.; Shu, D.; He, Y.; Zhao, Y.L.; Li, Y.; Wang, Z.L.; Zhang, S.Y.; Xing, Z.H.; Xu, C.; et al. Deltonin, a steroidal saponin, inhibits colon cancer cell growth in vitro and tumor growth in vivo via induction of apoptosis and antiangiogenesis. Cell. Physiol. Biochem. 2011, 27, 233–242. [Google Scholar] [CrossRef]
- Zhang, S.; He, Y.; Tong, Q.; Chen, Q.; Wu, X.; Huang, W. Deltonin induces apoptosis in MDA-MB-231 human breast cancer cells via reactive oxygen species-mediated mitochondrial dysfunction and ERK/AKT signaling pathways. Mol. Med. Rep. 2013, 7, 1038–1044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shu, D.; Qing, Y.; Tong, Q.; He, Y.; Xing, Z.; Zhao, Y.; Li, Y.; Wei, Y.; Huang, W.; Wu, X. Deltonin isolated from Dioscorea zingiberensis inhibits cancer cell growth through inducing mitochondrial apoptosis and suppressing akt and mitogen activated protein kinase signals. Biol. Pharm. Bull. 2011, 34, 1231–1239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, K.; Dong, A.; Yao, X.; Kobayashi, H.; Iwasaki, S.; Anti-neoplastic agents, I. Three spirostanol glycosides from rhizomes of Dioscorea collettii var. hypoglauca. Planta Med. 1996, 62, 573–575. [Google Scholar] [CrossRef] [PubMed]
- Guohua, Z.; Zhixiao, L.; Zongdao, C. Structural analysis and antitumor activity of RDPS-I polysaccharide from Chinese yam. Yao Xue Xue Bao 2003, 38, 37–41. [Google Scholar]
- He, Z.; Tian, Y.; Zhang, X.; Bing, B.; Zhang, L.; Wang, H.; Zhao, W. Anti-tumour and immunomodulating activities of diosgenin, a naturally occurring steroidal saponin. Nat. Prod. Res. 2012, 26, 2243–2246. [Google Scholar] [CrossRef]
- Raju, J.; Mehta, R. Cancer chemopreventive and therapeutic effects of diosgenin, a food saponin. Nutr. Cancer 2008, 61, 27–35. [Google Scholar] [CrossRef]
- Mukesh, R.; Namita, P. Medicinal Plants with Antidiabetic Potential-A Review. Am. J. Agric. Environ. Sci. 2013, 13, 81–94. [Google Scholar] [CrossRef]
- Ahmed, Z.; Chishti, M.Z.; Johri, R.K.; Bhagat, A.; Gupta, K.K.; Ram, G. Antihyperglycemic and antidyslipidemic activity of aqueous extract of Dioscorea bulbifera tubers. Diabetol. Croat. 2009, 38, 63–72. [Google Scholar]
- Maithili, V.; Dhanabal, S.; Mahendran, S.; Vadivelan, R. Antidiabetic activity of ethanolic extract of tubers of Dioscorea alata in alloxan induced diabetic rats. Indian J. Pharmacol. 2011, 43, 455. [Google Scholar] [PubMed] [Green Version]
- de Salgado Rêgo, T.; da Silva Ash, L.; Pessoa, L.; da Silva Feijó, M.B.; Leite, J.; de Sousa dos Santos, A.; Soares da Costa, C.A.; Boaventura, G.T. The intake of Yam (Dioscorea bulbifera Linn) attenuated the hyperglycemia and the bone fragility in female diabetic rats. Nutr. Hosp. 2014, 29, 370–375. [Google Scholar] [CrossRef]
- Gao, X.; Li, B.; Jiang, H.; Liu, F.; Xu, D.; Liu, Z. Dioscorea opposita reverses dexamethasone induced insulin resistance. Fitoterapia 2007, 78, 12–15. [Google Scholar] [CrossRef]
- Choi, S.Z.; Son, M.W. A novel botanical drug for the treatment of diabetic neuropathy. Arch. Pharm. Res. 2011, 34, 865–867. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Kim, H.; Ryu, J.; Lee, J.; Cho, Y.; Son, M.; Kim, S. A novel botanical drug (DA-9801) for the treatment of diabetic neuropathy. Planta Med. 2011, 77, PL17. [Google Scholar] [CrossRef]
- Song, I.-S.; Kong, T.Y.; Jeong, H.-U.; Kim, E.N.; Kwon, S.-S.; Kang, H.E.; Choi, S.-Z.; Son, M.; Lee, H.S. Evaluation of the transporter-mediated herb-drug interaction potential of DA-9801, a standardized Dioscorea extract for diabetic neuropathy, in human in vitro and rat in vivo. BMC Complement. Altern. Med. 2014, 14, 251. [Google Scholar] [CrossRef] [Green Version]
- Jin, H.Y.; Kim, S.H.; Yu, H.M.; Baek, H.S.; Park, T.S. Therapeutic potential of Dioscorea extract (DA-9801) in comparison with alpha lipoic acid on the peripheral nerves in experimental diabetes. J. Diabetes Res. 2013, 2013, 631218. [Google Scholar] [CrossRef]
- Sato, K.; Fujita, S.; Iemitsu, M. Acute administration of diosgenin or Dioscorea improves hyperglycemia with increases muscular steroidogenesis in STZ-induced type 1 diabetic rats. J. Steroid Biochem. Mol. Biol. 2014, 143, 152–159. [Google Scholar] [CrossRef] [Green Version]
- Omoruyi, F.O. Jamaican bitter yam sapogenin: Potential mechanisms of action in diabetes. Plant Foods Hum. Nutr. 2008, 63, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Jeong, E.J.; Jegal, J.; Ahn, J.; Kim, J.; Yang, M.H. Anti-obesity effect of Dioscorea oppositifolia extract in high-fat diet- induced obese mice and its chemical characterization. Biol. Pharm. Bull. 2016, 39, 409–414. [Google Scholar] [CrossRef] [Green Version]
- Kwon, C.; Sohn, H.Y.; Kim, S.H.; Kim, J.H.; Son, K.H.; Lee, J.S.; Lim, J.K.; Kim, J. Anti-obesity effect of Dioscorea nipponica Makino with lipase-inhibitory activity in rodents. Biosci. Biotechnol. Biochem. 2003, 67, 1451–1456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dzomba, P.; Musekiwa, C. Anti-obesity and antioxidant activity of dietary flavonoids from Dioscorea steriscus tubers. J. Coast. Life Med. 2014, 2, 465–470. [Google Scholar] [CrossRef]
- McKoy, M.-L.; Thomas, P.-G.; Asemota, H.; Omoruyi, F.; Simon, O. Effects of Jamaican bitter yam (Dioscorea polygonoides) and diosgenin on blood and fecal cholesterol in rats. J. Med. Food 2014, 17, 1183–1188. [Google Scholar] [CrossRef]
- Chen, H.-L.; Wang, C.-H.; Chang, C.-T.; Wang, T.-C. Effects of Taiwanese yam (Dioscorea alata L. cv. Tainung No. 2) on the mucosal hydrolase activities and lipid metabolism in Balb/c mice. Nutr. Res. 2003, 23, 791–801. [Google Scholar] [CrossRef]
- Yeh, Y.; Lee, Y.; Hwang, D. Yam (Dioscorea alata) inhibits hypertriglyceridemia and liver enlargement in rats with hypercholesterol diet. J. Chin. Med. 2007, 18, 65–74. [Google Scholar]
- Chiu, C.-S.; Deng, J.-S.; Hsieh, M.-T.; Fan, M.-J.; Lee, M.-M.; Chueh, F.-S.; Han, C.-K.; Lin, Y.-C.; Peng, W.-H. Yam (Dioscorea pseudojaponica Yamamoto) ameliorates cognition deficit and attenuates oxidative damage in senescent mice induced by D-galactose. Am. J. Chin. Med. 2009, 37, 889–902. [Google Scholar] [CrossRef]
- Tohda, C.; Urano, T.; Umezaki, M.; Nemere, I.; Kuboyama, T. Diosgenin is an exogenous activator of 1,25D 3-MARRS/Pdia3/ERp57 and improves Alzheimer’s disease pathologies in 5XFAD mice. Sci. Rep. 2012, 2, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Tohda, C.; Lee, Y.A.; Goto, Y.; Nemere, I. Diosgenin-induced cognitive enhancement in normal mice is mediated by 1,25D 3-MARRS. Sci. Rep. 2013, 3, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Tohda, C.; Yang, X.; Matsui, M.; Inada, Y.; Kadomoto, E.; Nakada, S.; Watari, H.; Shibahara, N. Diosgenin-rich yam extract enhances cognitive function: A placebo-controlled, randomized, double-blind, crossover study of healthy adults. Nutrients 2017, 9, 1160. [Google Scholar] [CrossRef]
- Peng, K.-Y.; Horng, L.-Y.; Sung, H.-C.; Huang, H.-C.; Wu, R.T. Antiosteoporotic activity of Dioscorea alata L. cv. phyto through driving mesenchymal stem cells differentiation for bone formation. Evid. Based Complement. Altern. Med. 2011, 2011, 712892. [Google Scholar] [CrossRef] [Green Version]
- Han, N.; Xu, J.; Xu, F.; Liu, Z.; Yin, J. The in vivo effects of a fraction from Dioscorea spongiosa on glucocorticoid-induced osteoporosis. J. Ethnopharmacol. 2016, 185, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Tulin, E.E.; Ecleo, Z.T. Cytokine-Mimetic properties of some Philippine food and medicinal plants. J. Med. Food 2007, 10, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.L.; Hong, L.T.; Lee, J.K.; Huang, C.J. The bone-protective effect of a Taiwanese yam (Dioscorea alata L. cv. Tainung No. 2) in ovariectomised female BALB/C mice. J. Sci. Food Agric. 2009, 89, 517–522. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, N.; Tanoue, Y.; Kai, N.; Nagashima, T. Antioxidant and antihypertensive activities of autolysate and enzymatic hydrolysates from yam Dioscorea opposita Thunb.) ichyoimo tubers. J. Food Agric. Environ. 2007, 5, 64–68. [Google Scholar]
- Amat, N.; Amat, R.; Abdureyim, S.; Hoxur, P.; Osman, Z.; Mamut, D.; Kijjoa, A. Aqueous extract of Dioscorea opposita thunb. normalizes the hypertension in 2K1C hypertensive rats. BMC Complement. Altern. Med. 2014, 14, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Ma, T.; Zheng, Y.; Lv, S.; Li, Y.; Liu, S. Diosgenin inhibits IL-1β-induced expression of inflammatory mediators in human osteoarthritis chondrocytes. Int. J. Clin. Exp. Pathol. 2015, 8, 4830–4836. [Google Scholar]
- Burger, H.; Woods, N.F.; Dennerstein, L.; Alexander, J.L.; Kotz, K.; Richardson, G. Nomenclature and endocrinology of menopause and perimenopause. Expert Rev. Neurother. 2007, 7, S35–S43. [Google Scholar] [CrossRef]
- Shuster, L.T.; Rhodes, D.J.; Gostout, B.S.; Grossardt, B.R.; Rocca, W.A. Premature menopause or early menopause: Long-term health consequences. Maturitas 2010, 65, 161–166. [Google Scholar] [CrossRef] [Green Version]
- Lacey, J.V.; Mink, P.J.; Lubin, J.H.; Sherman, M.E.; Troisi, R.; Hartge, P.; Schatzkin, A.; Schairer, C. Menopausal hormone replacement therapy and risk of ovarian cancer. JAMA 2002, 288, 334–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossouw, J.E.; Anderson, G.L.; Prentice, R.L.; LaCroix, A.Z.; Kooperberg, C.; Stefanick, M.L.; Jackson, R.D.; Beresford, S.A.A.; Howard, B.V.; Johnson, K.C.; et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women. JAMA 2002, 288, 321–333. [Google Scholar]
- Stuart, D. Dangerous Garden: The Quest for Plants to Change Our Lives 2004; Havard University Press: Cambridge, MA, USA, 2004. [Google Scholar]
- Komesaroff, P.A.; Black, C.V.S.; Cable, V.; Sudhir, K. Effects of wild yam extract on menopausal symptoms, lipids and sex hormones in healthy menopausal women. Climacteric 2001, 4, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-C.; Kuo, H.-C.; Chang, S.-Y.; Wu, T.-C.; Huang, K.-E. The assessment of efficacy of Diascorea alata for menopausal symptom treatment in Taiwanese women. Climacteric 2011, 14, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.-H.; Liu, L.-Y.; Chung, C.-J.; Jou, H.-J.; Wang, T.-A. Estrogenic effect of yam ingestion in healthy postmenopausal women. J. Am. Coll. Nutr. 2005, 24, 235–243. [Google Scholar] [CrossRef]
- Li, W. The clinical study of treating peripheral menopause syndrome (PMS) with the decoction of Bu-Shen-An-Geng. In Chinese Gynecology; Shandong University of Traditional Chinese Medicine: Jinan, China, 2006. [Google Scholar]
- Hu, K.; Yao, X.S.; Dong, A.J.; Kobayashi, H.; Iwasaki, S.; Jing, Y.K. A new pregnane glycoside from Dioscorea collettii var. hypoglauca. J. Nat. Prod. 1999, 62, 299–301. [Google Scholar] [CrossRef]
- Ososki, A.L.; Lohr, P.; Reiff, M.; Balick, M.J.; Kronenberg, F.; Fugh-Berman, A.; O’Connor, B. Ethnobotanical literature survey of medicinal plants in the Dominican Republic used for women’s health conditions. J. Ethnopharmacol. 2002, 79, 285–298. [Google Scholar] [CrossRef]
- Higdon, K.; Scott, A.; Tucci, M.; Benghuzzi, H.; Tsao, A.; Puckett, A.; Cason, Z.; Hughes, J. The use of estrogen, DHEA, and diosgenin in a sustained delivery setting as a novel treatment approach for osteoporosis in the ovariectomized adult rat model. Biomed. Sci. Instrum. 2001, 37, 281–286. [Google Scholar]
- Zuluaga, M.F.; Baena, Y.; Mora, C.E.; D’León, L.F.P. Physicochemical characterization and application of yam (Dioscorea cayenensis-rotundata) starch as a pharmaceutical excipient. Starch Staerke 2007, 59, 307–317. [Google Scholar] [CrossRef]
- Nasipuri, R. Evaluation of yam starch as tablet binder and disintegrant. Niger. J. Pharm. 1979, 10, 182–187. [Google Scholar]
- Riley, C.K.; Adebayo, S.A.; Wheatley, A.O.; Asemota, H.N. Surface properties of yam (Dioscorea sp.) starch powders and potential for use as binders and disintegrants in drug formulations. Powder Technol. 2008, 185, 280–285. [Google Scholar] [CrossRef]
- Odeku, O.A.; Picker-Freyer, K.M. Analysis of the material and tablet formation properties of four Dioscorea starches. Starch Staerke 2007, 59, 430–444. [Google Scholar] [CrossRef]
- Okunlola, A.; Odeku, O.A. Evaluation of starches obtained from four Dioscorea species as binding agent in chloroquine phosphate tablet formulations. Saudi Pharm. J. 2011, 19, 95–105. [Google Scholar] [CrossRef] [Green Version]
- Rates, S.M.K. Plants as source of drugs. Toxicon 2001, 39, 603–613. [Google Scholar] [CrossRef]
- Wagner, H. Guidelines for quality control of phytomedicines. In Congress on Medicinal and Aromatic Plants for Human Welfare; ICMPA/ISHS/SAIPOA: Mendoza, Argentina, 1997; Volume 2, pp. 1–4. [Google Scholar]
- Ou-Yang, S.-H.; Jiang, T.; Zhu, L.; Yi, T. Dioscorea nipponica Makino: A systematic review on its ethnobotany, phytochemical and pharmacological profiles. Chem. Cent. J. 2018, 12, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Sheng, F.; He, Z.; Chen, K. Research progress of the extraction, separation, detection method and pharmacological action of the diosdenin. Lishizhen Med. Mater. Med. Res. 2013, 24, 914–917. [Google Scholar]
- Yang, H.; Yin, H.; Wang, X.; Li, Z.; Shen, Y.-P.; Jia, X. In situ pressurized biphase acid hydrolysis, a promising approach to produce bioactive diosgenin from the tubers of Dioscorea zingiberensis. Pharmacogn. Mag. 2015, 11, 636. [Google Scholar] [CrossRef] [Green Version]
- Shi, W.; Zhang, C.; Zhao, D.; Wang, L.; Li, P.; Li, H. Discovery of hepatotoxic equivalent combinatorial markers from Dioscorea bulbifera tuber by fingerprint-toxicity relationship modeling. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Wang, J.; Ji, L.; Liu, H.; Wang, Z. Study of the hepatotoxicity induced by Dioscorea bulbifera L. rhizome in mice. Biosci. Trends 2010, 4, 79–85. [Google Scholar] [PubMed]
- Wang, J.; Liang, Q.; Ji, L.; Liu, H.; Wang, C.; Wang, Z. Gender-related difference in liver injury induced by Dioscorea bulbifera L. rhizome in mice. Hum. Exp. Toxicol. 2011, 30, 1333–1341. [Google Scholar] [CrossRef]
- Georges, F.; Ray, H. Genome editing of crops: A renewed opportunity for food security. GM Crop. Food 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rojas-Vásquez, R.; Gatica-Arias, A. Use of genome editing technologies for genetic improvement of crops of tropical origin. Plant Cell. Tissue Organ Cult. 2020, 140, 215–244. [Google Scholar] [CrossRef]
- Zhou, W.; Li, B.; Li, L.; Ma, W.; Liu, Y.; Feng, S.; Wang, Z. Genome survey sequencing of Dioscorea zingiberensis. Genome 2018, 61, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Bredeson, J.V.; Lyons, J.B.; Shu, S.; Ogunleye, I.; Bhattacharjee, R.; Obidiegwu, J.; Rokhsar, D.S. The genome sequence of D. alata.. Assembled Genome Sequence. in press. Available online: https://phytozome-next.jgi.doe.gov/info/Dalata_v2_1. (accessed on 07 August 2020).
- Siadjeu, C.; Pucker, B.; Viehöver, P.; Albach, D.C.; Weisshaar, B. High contiguity de novo genome sequence assembly of Trifoliate yam (Dioscorea dumetorum) using long read sequencing. Genes 2020, 11, 274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sugihara, Y.; Darkwa, K.; Yaegashi, H.; Natsume, S.; Shimizu, M.; Abe, A.; Hirabuchi, A.; Ito, K.; Oikawa, K.; Tamiru-Oli, M.; et al. Genome analyses reveal the hybrid origin of the staple food crop white Guinea yam. bioRxiv 2020, 53, 1689–1699. [Google Scholar] [CrossRef]
- Laveaga, G.S. Jungle Laboratories: Mexican Peasants, National Projects, and the Making of the Pill; Duke University Press: Durham, NC, USA, 2009. [Google Scholar]
| Species | Proximate Composition (Percentage, %) | ||||||
|---|---|---|---|---|---|---|---|
| Moisture | Crude Protein | Crude Fat | Crude Fiber | Ash | Starch | Reference | |
| D. alata | 64.9–87.8 | 0.6–18.7 | 0.23–5.28 | 0.75–11.0 | 0.69–8.81 | 15.6–84.3 | [35,36,37,39,42,46,47,48,49,50,51,52,53,54,55,56,57,58,59] |
| D. abyssinica | NR | 3.13–5.37 | 0.31–1.22 | 1.94–4.91 | 2.31–3.58 | NR | [36] |
| D. bulbifera | 61.6–92.5 | 0.89–15.8 | 0.30–8.13 | 0.61–18.2 | 0.05–8.15 | 12.5–62.7 | [36,37,39,42,43,47,51,58,60,61,62,63,64,65,66] |
| D. cayenensis | 62.2–89.4 | 2.62–6.63 | 0.27–7.86 | 0.17–3.26 | 0.63–5.48 | 80.75 | [36,37,42,48,58,60,67] |
| D. delicata | 92.7 | 0.41 | NR | 4.87 | NR | 0.54 | [37] |
| D. deltoidea | 80.2 | 1.6 | 0.2 | 1.5 | 0.6 | NR | [65] |
| D. dodecaneura | 68.4 | 1.50 | NR | NR | NR | 18.46 | [37] |
| D. dumetorum | 64.3–90.2 | 0.19–10.3 | 0.37–3.65 | 0.82–5.65 | 2.17–7.79 | 17.0–63.34 | [37,42,49,50,60,61,68,69,70,71] |
| D. esculenta | 50.65–86.67 | 5.60 –10.50 | 0.08–2.58 | 1.23–7.82 | 0.25–8.50 | 17.25 | [37,39,42,48,51,72] |
| D. fordii | NR | 9.8–10.2 | NR | 0.92–1.14 | NR | 75.7–77.1 | [35] |
| D. hamiltonii | 78.73 | 4.37 | 10.2 | 4.15 | 8.70 | NR | [38] |
| D. hispida | 15.8–37.8 | 1.13–6.20 | 1.99–9.36 | NR | 0.29–1.24 | 11.5 | [32] |
| D. laxiflora | 82.0 | 0.26 | NR | 2.34 | NR | 8.92 | [37] |
| D. nipponica | NR | NR | NR | NR | NR | 35.4 | [47] |
| D. olfersiana | 84.6 | 0.42 | NR | 9.53 | NR | 0.54 | [37] |
| D. oppositifolia | 78.5–92.1 | 7.00–13.54 | 4.40–7.42 | 4.92–8.47 | 2.60–6.38 | NR | [38,39,51] |
| D. pentaphylla | 90.1–93.1 | 6.48–9.18 | 4.01–6.24 | 5.14–7.24 | 3.36–4.64 | NR | [38,39,51] |
| D. persimilis | NR | 7.70–8.20 | NR | 0.88–0.92 | NR | 68.2–72.2 | [35] |
| D. piperifolia | 55.4–74.8 | 2.27–4.38 | NR | NR | NR | 18.2–26.1 | [37] |
| D. polystachya | NR | 6.30–12.2 | NR | 0.99–1.50 | NR | 60.7–72.5 | [35,47] |
| D. praehensilis | 64.1 | 3.64–5.38 | 0.26–7.83 | 1.41–3.21 | 2.13–4.90 | NR | [36,42] |
| D. pyrifolia | NR | 1.34 | NR | NR | 0.88 | NR | [73] |
| D. remotiflora | 78.18 | 1.91 | 0.47 | 1.22 | 0.85 | NR | [74] |
| D. rotundata | 54.5–75.2 | 0.09–8.28 | 0.09–3.39 | 0.41–4.33 | 1.03–4.92 | 22.0–80.8 | [36,37,42,48,49,52,58,72,75,76,77,78,79] |
| D. sanpaulesis | 69.2 | 0.77 | NR | 10.3 | NR | 2.62 | [37] |
| D. sinuata | 75.6 | 2.32 | NR | NR | NR | 8.00 | [37] |
| D. spicata | 81.5–89.3 | 6.38–8.20 | 3.26–4.78 | 4.67–6.31 | 5.18–5.20 | NR | [38,51] |
| D. steriscus | 72.5 | 0.83 | NR | 16.8 | 2.06 | 9.02 | [80] |
| D. subhastata | 89.0 | 0.59 | NR | 0.95 | NR | 3.69 | [37] |
| D. tomentosa | 84.5–93.7 | 5.25–9.54 | 2.86–6.84 | 3.21–4.38 | 2.48–6.53 | NR | [38,39,51] |
| D. trifida | 69.4–81.3 | 0.38–6.79 | 0.03–0.30 | NR | 0.2–3.37 | 7.94–64.0 | [37,48,81,82] |
| D. triphylla | 76.9 | 2.3 | 0.2 | 0.6 | 0.6 | NR | [65] |
| D. versicolor | 80.1 | 1.7 | 0.2 | 1.1 | 0.5 | NR | [65] |
| D. villosa | 76.4 | 2.21 | 6.01 | 3.50 | 3.13 | NR | [83] |
| D. wallichi | 71.1–76.4 | 10.5–10.8 | 1.18–3.34 | 7.48–9.23 | 6.36–8.42 | NR | [39,51] |
| Species | No of Varieties | Minerals (mg/100g) | Reference | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| K | Na | P | Ca | Mg | Cu | Fe | Mn | Zn | |||
| D. abyssinica1 | 13 | NR | NR | 5.1–56.5 | 31.02–118.8 | NR | NR | 20.3–69.7 | NR | 0.48–0.77 | [36] |
| D. alata1 | 7 | 1157–2016 | 52–82.7 | 117–194 | 62.5–78.0 | 64.0–74.6 | 6.4–6.9 | 9.9–10.9 | 3.1–4.3 | 3.4–4.3 | [54] |
| D. alata1 | 7 | 240–400 | 190–380 | 100–340 | 20.04–80.2 | 24.31–97.2 | NR | NR | NR | NR | [53] |
| D. alata1 | 9 | NR | NR | NR | 31.64–45.3 | 32.68–47.8 | 0.42–0.48 | 0.83–2.2 | NR | 0.82–2.6 | [35] |
| D. alata1 | 20 | 1055-2010 | 8.30–13.1 | NR | 26.0–53.5 | 39.0–59.5 | NR | NR | NR | 1.01–1.8 | [57] |
| D. alata3 | 1 | 476.8 ± 0.1 | 68.9 ± 0.02 | 163.7 ± 0.10 | 285.8 ± 0.02 | 116.3 ± 0.69 | NR | 2.48 ± 0.02 | NR | 2.12 ± 0.00 | [58] |
| D. alata3 | 2 | 622.5–742.5 | 62.5–95.0 | 219.0–239 | 6.50–16.50 | 40.0–41.5 | 0.10–0.15 | 1.50–2.00 | 2.15–2.20 | 6.65–6.80 | [42] |
| D. alata1 | 16 | 1055–2010 | 8.4–13.1 | 87.8–190.0 | 26.0–41.0 | 39.0–58.0 | 1.23–1.57 | NR | 0.48–2.21 | 1.01–1.41 | [55] |
| D. alata1 | 4 | NR | NR | 26.59–49.12 | 11.24–120.0 | NR | NR | 17.75–51.1 | NR | 0.38–1.18 | [36] |
| D. alata1 | 1 | 3932.9 ± 0.16 | 75.4 ± 0.02 | NR | 3032.1 ± 0.25 | 120.7 ± 0.005 | 1.216 ± 0.001 | 124.3 ± 0.004 | 1.33 ± 0.001 | 5.7 ± 0.001 | [56] |
| D. alata1 | 1 | 5.25 ± 2.12 | 0.35 ± 0.0 | NR | 0.22 ± 0.99 | 0.65 ± 0.71 | NR | 0.75 ± 0.73 | NR | NR | [50] |
| D. alata1 | 1 | 786.3 ± 0.14 | 44.56 ± 0.3 | 140.14 ± 0.14 | 448.36 ± 0.11 | 656.31 ± 0.07 | 11.20 ± 0.14 | 24.30 ± 0.19 | 6.36 ± 0.21 | 2.26 ± 0.01 | [51] |
| D. buibifera1 | 12 | NR | NR | 8.72–55.26 | 15.74–121.3 | NR | NR | 20.26–90.9 | NR | 0.4–8.33 | [36] |
| D. buibifera1 | 1 | NR | NR | 0.521 | 1410.0 | 250 | NR | NR | NR | NR | [66] |
| D. buibifera1 | 1 | 1554.4 ± 0.36 | 78.24 ± 0.07 | 154.42 ± 0.53 | 338.15 ± 0.09 | 396.20 ± 1.07 | 2.14 ± 0.04 | 19.20 ± 0.20 | 9.40 ± 0.14 | 1.48 ± 0.03 | [51] |
| D. buibifera3 | 1 | 525.8 ± 1.41 | 87.8 ± 0.10 | 159.5 ± 0.04 | 378.5 ± 0.10 | 128.7 ± 0.04 | NR | 3.14 ± 0.02 | NR | 2.79 ± 0.01 | [58] |
| D. buibifera2 | 1 | 560 ± 49 | 17.8 ± 9.8 | 61.61 ± 0.8 | 29.3 ± 4.8 | 25.9 ± 9.2 | 0.21 ± 0.03 | 2.92 ± 0.3 | 0.35 ± 0.03 | 0.53 ± 0.06 | [65] |
| D. buibifera3 | 2 | 1250–1475 | 92.5–102.5 | 223.5–224.5 | 103–116.5 | 76.5–83.5 | 0.20 | 6.00–6.50 | 1.30–1.35 | 6.10–6.35 | [42] |
| D. cayenensis1 | 2 | NR | NR | 19.15–26.12 | 6.3–27.6 | NR | NR | 17.2–27.95 | NR | 0.74–0.75 | [36] |
| D. cayenensis2 | 1 | 262.3 ± 0.25 | 8.53 ± 0.05 | 19.5 ± 0.10 | 22.53 ± 0.13 | 61.53 ± 0.25 | NR | 0.79 ± 0.02 | NR | 0.39 ± 0.01 | [67] |
| D. cayenensis3 | 1 | 523.8 ± 0.04 | 76.8 ± 0.03 | 167.8 ± 0.02 | 345.8 ± 0.01 | 120.2 ± 0.55 | NR | 2.50 ± 0.08 | NR | 2.18 ± 0.02 | [58] |
| D. cayenensis3 | 2 | 700–825 | 62.5–70.0 | 164.5–190.5 | 74.5–80.0 | 57.5–38.0 | 0.20 | 5.0–5.5 | 1.2–1.25 | 5.45–5.85 | [42] |
| D. deltoidea2 | 1 | 340 ± 51 | 9.12 ± 1.6 | 33.1 ± 0.6 | 46.9 ± 6.2 | 22.8 ± 7.1 | 0.10 ± 0.0 | 1.85 ± 1.0 | 0.31 ± 0.02 | 0.22 ± 0.04 | [65] |
| D. dumetorum1 | 1 | 7.03 ± 0.78 | 0.41 ± 0.14 | NR | 0.81 ± 0.21 | 0.95 ± 0.71 | NR | 0.07 ± 0.14 | NR | NR | [50] |
| D. dumetorum3 | 2 | 670–772 | 72.5–77.5 | 269–286 | 27.5–29.5 | 61.5 | 0.10 | 2.0–2.50 | 2.50–2.65 | 5.80 | [42] |
| D. dumetorum3 | 2 | 0.03 | 0.02 | NR | 0.19–0.21 | 0.65–0.72 | 1.36–1.48 | 0.13–0.16 | 0.34–0.38 | 0.03–0.18 | [71] |
| D. dumetorum3 | NR | NR | 151 | 57.8 | NR | NR | 8.89 | NR | NR | [68] | |
| D. esculenta3 | 2 | 765–795 | 87.5–92.5 | 273.5–294.5 | 20.5–27.0 | 67.5–73.0 | 0.10 | 2.0 | 2.70–2.95 | 6.20–7.80 | [42] |
| D. esculenta1 | 1 | 1594.3 ± 1.34 | 86.40 ± 0.14 | 138.10 ± 0.14 | 314.01 ± 0.33 | 436.06 ± 0.54 | 3.40 ± 0.01 | 11.48 ± 0.11 | 5.46 ± 0.11 | 1.76 ± 0.04 | [51] |
| D. fordii1 | 3 | NR | NR | NR | 28.56–30.05 | 34.58–35.63 | 0.45–0.51 | 1.82–2.02 | NR | 1.79–1.85 | [35] |
| D. oppositifolia1 | 1 | 1431 ± 1.56 | 102.2 ± 0.54 | 78.2 ± 0.08 | 680.6 ± 0.82 | 432.5 ± 1.11 | 2.74 ± 0.03 | 22.0 ± 0.08 | 6.34 ± 0.01 | 3.24 ± 0.08 | [38] |
| D. oppositifolia1 | 2 | 1534–1624 | 124–168.2 | 114.1–124.1 | 294.2–646.2 | 540.1–634.1 | 7.62–14.56 | 32.16–40.76 | 7.42–9.04 | 1.56–6.26 | [51] |
| D. pentaphylla1 | 1 | 1322 ± 2.40 | 95.2 ± 0.12 | 96.1 ± 0.06 | 632.1 ± 0.22 | 380.0 ± 0.74 | 12.60 ± 0.14 | 103.48 ± 0.94 | 1.32 ± 0.01 | 3.10 ± 0.01 | [38] |
| D. pentaphylla1 | 1 | 1441.0 ± 0.98 | 96.20 ± 0.63 | 158.18 ± 0.21 | 444.24 ± 0.09 | 532.12 ± 0.56 | 13.26 ± 0.05 | 66.32 ± 0.14 | 3.46 ± 0.21 | 3.42 ± 0.01 | [51] |
| D. persimilis1 | 3 | NR | NR | NR | 46.55–47.64 | 46.70–47.42 | 0.382–0.423 | 1.73–1.93 | NR | 1.32–1.45 | [35] |
| D. polystachya1 | 10 | NR | NR | NR | 39.73–55.82 | 33.26–54.47 | 0.35–0.54 | 1.43–2.58 | NR | 0.99–2.27 | [35] |
| D. praehensilis1 | 5 | NR | NR | 20.9–39.0 | 13.1–118.2 | NR | NR | 18.36–76.4 | NR | 0.4–1.09 | [36] |
| D. praehensilis3 | 1 | 1000 ± 21.2 | 80.0 ± 7.07 | 200.5 ± 0.71 | 79.5 ± 3.54 | 43.5 ± 0.71 | 0.40 ± 0.14 | 9.0 ± 0.0 | 0.95 ± 0.07 | 5.4 ± 0.57 | [42] |
| D. remotiflora2 | 1 | 4891 ± 25 | 79 ± 6 | 720 ± 20 | 242 ± 14 | 250 ± 10 | 3.3 ± 0.2 | 12.4 ± 0.5 | 4.1 ± 0.2 | 7.1 ± 0.3 | [74] |
| D. rotundata3 | 2 | 475–900 | 70.0–87.5 | 158–211.5 | 91.5–103.3 | 35.5–53.0 | 0.20–0.25 | 5.0–6.75 | 1.15–1.80 | 6.30–6.80 | [42] |
| D. rotundata1 | 6 | NR | NR | 26.96–40.21 | 22.77–114.4 | NR | NR | 17.75–78.3 | NR | 0.35–1.02 | [36] |
| D. rotundata1 | 1 | 1591 | 10.4 | NR | 31.0 | 51.0 | NR | NR | NR | 1.23 | [57] |
| D. rotundata3 | 3 | 9.00–71.00 | NR | 22.00–35.00 | 2.00–4.00 | 11.00 | NR | 1.00 | NR | 1.00 | [76] |
| D. rotundata3 | 1 | 530.7 ± 0.10 | 80.75 ± 0.14 | 168.7 ± 0.01 | 278.8 ± 0.15 | 125.7 ± 0.08 | NR | 2.88 ± 0.02 | NR | 2.34 ± 0.00 | [58] |
| D. rotundata3 | 1 | 209.13 ± 0.03 | 185.2 ± 0.05 | 54.00 ± 0.04 | 132.02 ± 0.04 | 45.90 ± 0.02 | 10.06 ± 0.05 | 81.85 ± 0.01 | NR | 5.46 ± 0.02 | [78] |
| D. spicata1 | 1 | 1255 ± 0.48 | 52.2 ± 0.11 | 86.1 ± 0.11 | 172.0 ± 0.21 | 112.4 ± 0.32 | 0.78 ± 0.21 | 22.36 ± 0.38 | 0.98 ± 0.14 | 4.18 ± 0.13 | [38] |
| D. spicata1 | 1 | 1136 ± 0.74 | 66.34 ± 0.54 | 166.30 ± 0.27 | 234.10 ± 0.58 | 324.16 ± 0.24 | 7.41 ± 0.11 | 24.10 ± 0.26 | 6.70 ± 0.14 | 2.56 ± 0.04 | [51] |
| D. tomentosa1 | 1 | 1354 ± 1.34 | 32.2 ± 0.18 | 96.1 ± 0.04 | 272.1 ± 1.01 | 120.4 ± 0.08 | 1.34 ± 0.01 | 24.56 ± 0.04 | 1.32 ± 0.04 | 5.20 ± 0.03 | [38] |
| D. tomentosa1 | 1 | 1245.6 ± 1.14 | 46.14 ± 0.30 | 104.06 ± 0.09 | 266.36 ± 0.16 | 321.04 ± 0.14 | 2.46 ± 0.14 | 28.50 ± 0.07 | 2.10 ± 0.11 | 5.40 ± 0.02 | [51] |
| D. trifida1 | 3 | 830–1350 | NR | 50.0–120.0 | 40.0 | 40.0–50.0 | 0.67–1.19 | NR | NR | 0.62–1.79 | [81] |
| D. triphylla2 | 1 | 317 ± 32 | 4.15 ± 0.7 | 56.6 ± 0.1 | 39.7 ± 8.1 | 27.3 ± 5.6 | 0.18 ± 0.05 | 1.00 ± 0.05 | 0.25 ± 0.07 | 0.39 ± 0.1 | [65] |
| D. versicolor2 | 1 | 250 ± 4 | 4.91 ± 2.5 | 40.8 ± 0.2 | 14.3 ± 1.8 | 18.3 ± 3.8 | 0.18 ± 0.02 | 0.39 ± 0.1 | 0.14 ± 0.0 | 0.3 ± 0.06 | [65] |
| D. villosa3 | 1 | 145.33 ± 1.15 | 5.40 ± 0.10 | 43.82 ± 0.49 | 28.06 ± 4.01 | 9.47 ± 0.23 | NR | NR | 0.032 ± 0.0 | 0.26 ± 0.0 | [83] |
| D. wallichi1 | 1 | 1361.7 ± 1.01 | 63.01 ± 0.27 | 106.40 ± 0.11 | 748.31 ± 0.32 | 578.06 ± 0.19 | 2.46 ± 0.08 | 20.14 ± 0.04 | 3.31 ± 0.05 | 6.66 ± 0.01 | [51] |
| Species | Phytochemicals | Reference |
|---|---|---|
| D. alata | Phenolics, phenol, flavonoid, flavonol, phytates/phytic acid, saponin, oxalates, alkaloid, tannins, allantoin, dioscin, diosgenin, dioscorin, hydrogen cyanide | [35,42,46,48,51,54,58,59,99,123] |
| D. bulbifera | Carotenoid, phenolics, phenol, polyphenol, flavonoid, terpenoid, saponin, steroid, alkaloid, tannins, phytates/phytic acid, oxalates, hydrogen cyanide | [29,42,51,56,58,62,63,64,99,123,124] |
| D. belophylla | Saponins, alkaloids, flavonoids, tannins and phenols | [125] |
| D. cayenensis | Phenolics, phenol, saponin, alkaloid, tannins, phytates/phytic acid, oxalates, dioscin | [29,42,48,58,67,99,126] |
| D. deltoida | Polyphenol | [29] |
| D. dumetorum | Phenols, flavonoid, alkaloid, tannins, phytates/phytic acid, oxalates, dioscorine | [42,70,99,127] |
| D. esculenta | Phenolics, tannins, phytates/phytic acid, oxalates, hydrogen cyanide | [42,48,51] |
| D. fordii | Allantoin, dioscin | [35] |
| D. glabra | Phenol, flavonoid | [123] |
| D. hamiltonii | Phenol, flavonoid | [123] |
| D. hirtiflora | Phenol, flavonoid | [127,128] |
| D. hirsute | Dioscorine | [128] |
| D. hispida | Dioscorine, phenol, flavonoid | [123] |
| D. japonica | Phenols, flavonoilds, glycans | [129,130] |
| D. mangenotiana | dioscin | [126] |
| D. oppositifolia | Phenolics, phenol, flavonoid, tannins, oxalates, hydrogen cyanide | [38,51,123] |
| D. panthaica | Saponins | [131] |
| D. persimilis | Allantoin, dioscin | [35] |
| D. pentaphylla | Phenolics, tannins, oxalates, hydrogen cyanide, phenol, flavonoid | [38,51,123] |
| D. polystachya | Flavones, polyphenols, allantoin, dioscin | [35,132] |
| D. praehensalis | Tannins, phytates/phytic acid, oxalates, | [42] |
| D. preussii | Saponins | [133] |
| D. pubera | Phenol, flavonoid | [123] |
| D. rotundata | Phenolics, phenol, tannins, phytates/phytic acid, oxalates, saponin, alkaloid, hydrocyanatem dioscin | [42,48,58,75,77,79,99,126] |
| D. sansibarensis | Dioscorine | [128] |
| D. spicata | Phenolics, tannins, oxalates, hydrogen cyanide | [38,51] |
| D. tomentosa | Phenolics, tannins, oxalates, hydrogen cyanide | [38,51] |
| D. trifida | Phenolics | [48] |
| D. triphylla | Polyphenol | [29] |
| D. versicolor | Polyphenol | [29] |
| D. villosa | Flavonoid, phenol, saponin, alkaloid, tannins, Phytates/ Phytic acid, oxalates | [84] |
| D. wallichi | Phenolics, tannins, oxalates, hydrogen cyanide, phenol, flavonoid | [51,123] |
| Species | Source of Extract | Biological Properties/Administration | Reference |
|---|---|---|---|
| D. alata | Tuber/bulb | Cure piles, gonorrhea and leprosy, anti-inflammatory, purgative, diuretic, anti-rheumatic properties; prevent cancer, reduce blood sugar, diabetes | [200,201] 2, [202,203] 1 |
| Tuber | Antihelminthic properties | [204,205] 2 | |
| Leaf | Fever | [206] 2 | |
| D. bartletti | Rhizome | Stagnation of blood, anemia | [207] 2 |
| D. belophylla | Tuber | Treatment of fever, dysentery, headache and malaria | [208] 2 |
| D. bulbifera | Tuber | Treatment of dementia | [209] 1 |
| Treatment of diabetes | [210] 1 | ||
| Leprosy and tumors | [211,212] 1 | ||
| Microbial infections and pig cysticercosis | [213] 1 | ||
| Antispasmodic, analgesic, aphrodisiac, diuretic and rejuvenative tonic | [214] 1 | ||
| Effects on liver and heart, reduces carbuncles, lung abscesses, breast lumps, goiter | [212] 1 | ||
| Abdominal pain | [215,216] 2 | ||
| Cough | [217] 2 | ||
| Oral contraceptive. | [218,219] 2 | ||
| Raw tuber consumed as an appetizer | [220] 2 | ||
| Rheumatism | [221] 2 | ||
| Aphrodisiac | [222] 2 | ||
| Aerial bulb | Oxidative stress induced pathological disorders | [210,223] 1 | |
| Anthelmintic treatment | [224] 1 | ||
| Leaf | Treatment of Elephantiasis | [225] 2 | |
| Leaf paste fights dermatological diseases | [226] 2 | ||
| Stem | Fresh stem shoots are used on hair to fight dandruff | [7] 2 | |
| D. bellophylla | Tuber | Lowers blood cholesterol and reduces heart attack | [7] 2 |
| D. cayenensis | Tuber | Anti-diarrheal | [206] 2 |
| D. collettii | Rhizome | Cervical carcinoma, urinary bladder carcinoma, renal tumor | [227] 1 |
| D. deltoidea | Tuber | Digestive disorders, sore throat, diarrhea, abdominal pains, wounds, burns, anemia | [228,229,230] 2 |
| Anti-rheumatic and treatment of ophthalmic conditions | [231] 2 | ||
| Antihelmintic treatment | [229] 2 | ||
| Birth control, oral contraceptive | [217] 2 | ||
| Antihelminthic | [204,232] 2 | ||
| D. dumetorum | Tuber | Treatment of diabetes | [233] 1 |
| Control hyperlipidemia, hypercholesterolemia and hyperketonemia | [234] 2 | ||
| Jaundice treatment | [235] 2 | ||
| D. esculenta | Tuber | Inflammations, nervous disorders and respiratory infections | [235] 2 |
| Dysentery and pain relief | [7] 2 | ||
| D. hamittonii | Tuber | Stomach ache and appetizer | [235] 2 |
| Management of diarrhea | [236] 2 | ||
| Piles | [220] 2 | ||
| D. hirtiflora | Tuber | Gonorrhea treatment | [127] 1 |
| D. hispida | Leaf/root/tuber | Treatment of mole, insect bites and insomnia | [225] 2 |
| Tuber | Treatment of vomiting, indigestion and serves as a purgative when consumed fresh | [7] 2 | |
| Treatment of wounds and injuries | [219] 2 | ||
| Ophthalmic ointment | [237] 2 | ||
| D. japonica | Rhizome | Diarrhea and dysentery due to spleen deficiency, fatigue, wasting and thirsting, seminal emission, vaginal discharge and frequent urination | [238] 1 |
| Inflammation, asthma, rheumatoid arthritis | [239] 1 | ||
| coughing and wheezing | [239] 1 | ||
| D. membranacea | Rhizome | Cancer | [240] 1 |
| D. nipponica | Rhizome | Dissipation of lumps and goiter, clears heat, relieves toxicity, cools the blood, stops bleeding and coughing, calms sneezing, poisonous snake bites, bleeding due to blood-heat and whooping cough | [241] 2 |
| Anti-rheumatic, analgesic, aids blood circulation, anti-diuretic, aids digestion | [242,243] 1 | ||
| D. oppositifolia | Rhizomes/tuber | Relief of menopausal syndromes, rejuvenation of early mothers | [220,244] 2 |
| Leaf/flower/tuber | Antiseptic for ulcer; the roots are chewed to cure toothache and aphtha | [245,246] 2 | |
| Tuber | Increasing fertility in men | [222,236,247] 2 | |
| Constituent in epileptic and nasal relief formula | [219] 2 | ||
| D. panthaica | Rhizome | Gastric diseases, bone injuries, rheumatic arthritis | [122] 2 |
| Cardiovascular diseases | [248] 1 | ||
| D. pentaphylla | Leaf/vine | Treatment of paralysis | [225] 2 |
| Tuber/flower/young shoot | Rheumatism | [7,249] 2 | |
| Tuber | Pain relief and reduce swelling | [219] 2 | |
| Stomach disorders | [7,250] 2 | ||
| D. polystachya | Rhizome | Consumptive cough and dysentery, aid for digestion and gastric motility and for restraining nocturnal emissions | [251] 1 |
| D. prazeri | Tuber | Antihelminthic | [204] 2 |
| D. pubera | Tuberous rhizome/bulb | Cure colic pain | [246,252] 2 |
| Tuber | Weakness | [253] 2 | |
| D. septembola | Rhizome | Rheumatism, urethra, renal infection | [254] 1 |
| D. spongiosa | Rhizome | Rheumatism, urethral, renal infections | [255] 2 |
| D. sylvatica | Tuber | Decoction used to treat cuts, wounds and sores | [256] 2 |
| D. trinervia | Tuber | Chronic diarrhea, asthma and diabetes | [7] 2 |
| D. vexans | Tuber | Anti-fertility | [257] 2 |
| D. villosa | Rhizome | Rheumatism | [135] 1, [258] 2 |
| Menstrual complaints, perimenopausal symptoms | [259,260] 2 | ||
| D. wallichii | Tuber | Flatulence and stomach pain | [7,204,261] 2 |
| De-appetizer | [222] 2 | ||
| D. zingiberensis | Rhizome | Cough, anthrax, rheumatic heart disease, rheum, arthritis, tumefaction, sprain | [262] 2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obidiegwu, J.E.; Lyons, J.B.; Chilaka, C.A. The Dioscorea Genus (Yam)—An Appraisal of Nutritional and Therapeutic Potentials. Foods 2020, 9, 1304. https://doi.org/10.3390/foods9091304
Obidiegwu JE, Lyons JB, Chilaka CA. The Dioscorea Genus (Yam)—An Appraisal of Nutritional and Therapeutic Potentials. Foods. 2020; 9(9):1304. https://doi.org/10.3390/foods9091304
Chicago/Turabian StyleObidiegwu, Jude E., Jessica B. Lyons, and Cynthia A. Chilaka. 2020. "The Dioscorea Genus (Yam)—An Appraisal of Nutritional and Therapeutic Potentials" Foods 9, no. 9: 1304. https://doi.org/10.3390/foods9091304
APA StyleObidiegwu, J. E., Lyons, J. B., & Chilaka, C. A. (2020). The Dioscorea Genus (Yam)—An Appraisal of Nutritional and Therapeutic Potentials. Foods, 9(9), 1304. https://doi.org/10.3390/foods9091304





