Advances on the Valorisation and Functionalization of By-Products and Wastes from Cereal-Based Processing Industry
Abstract
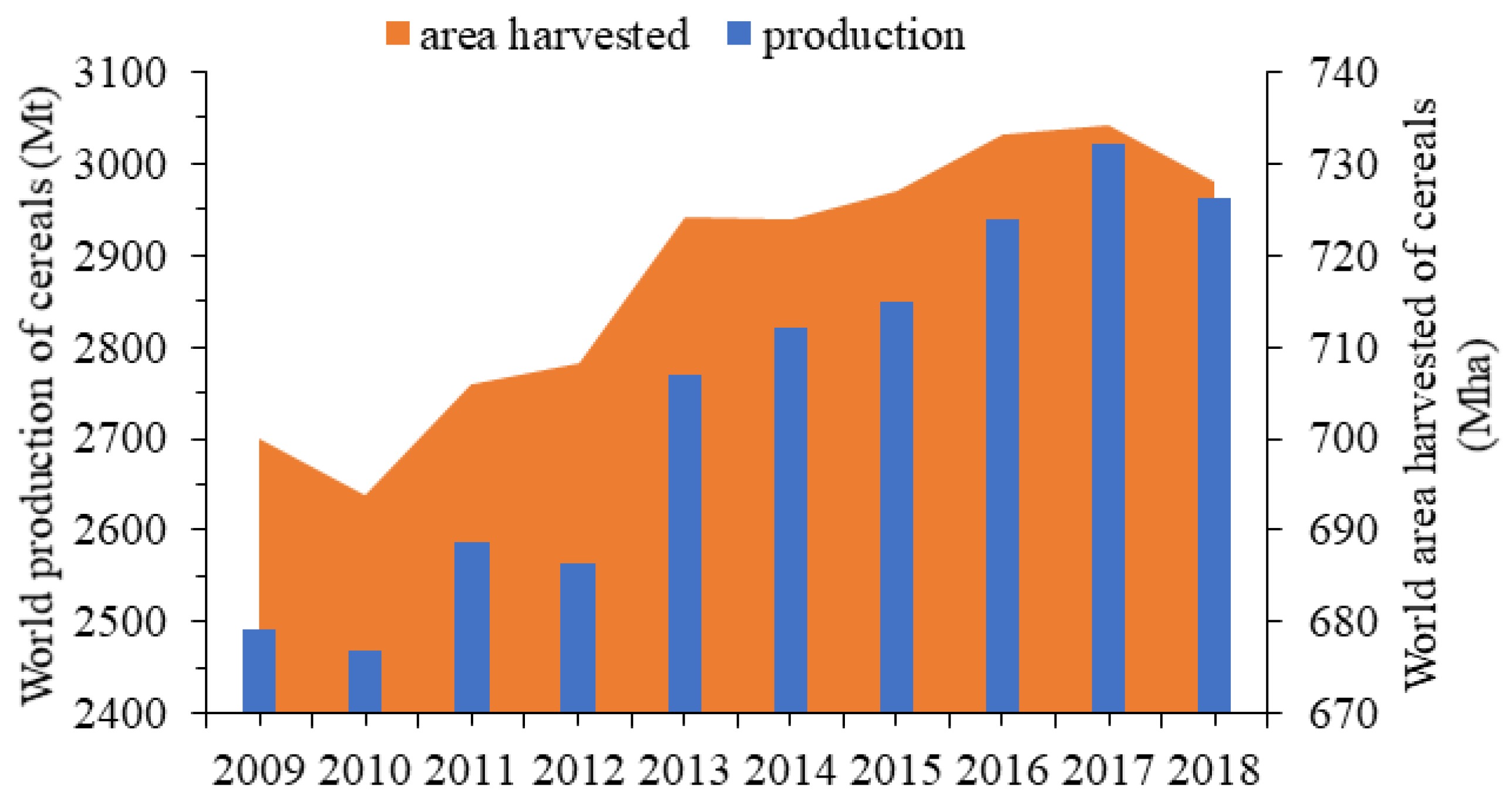
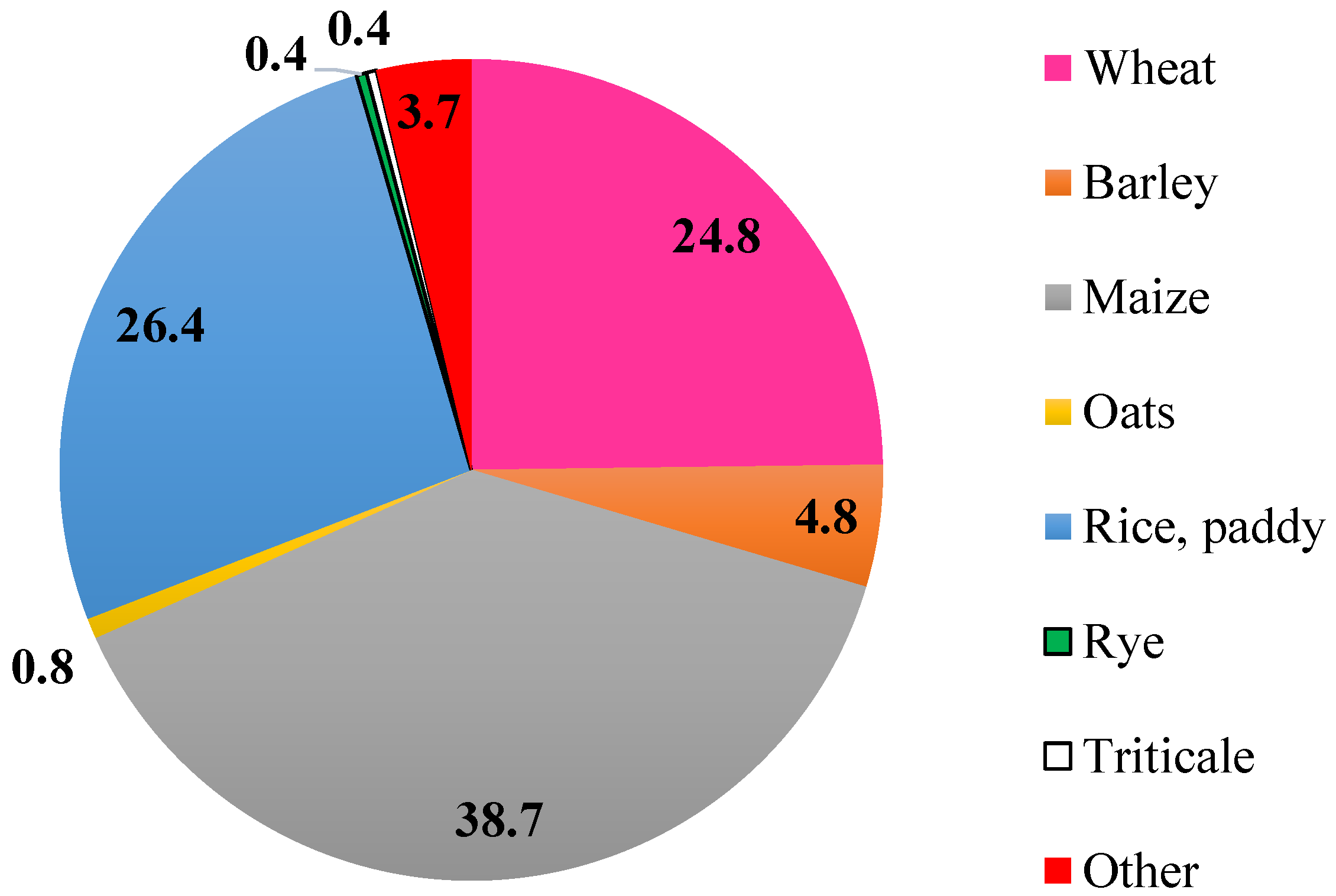
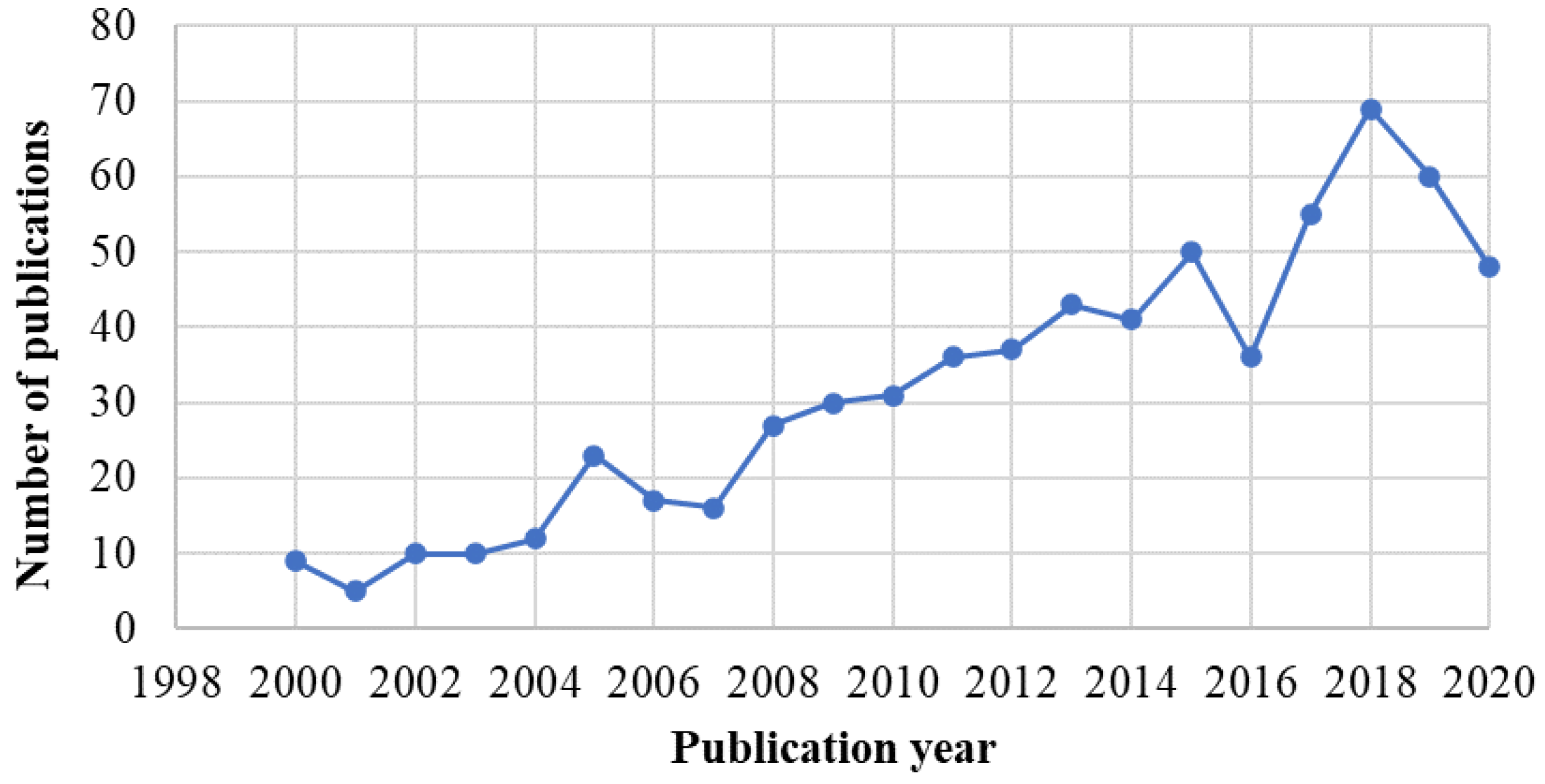
:1. Introduction
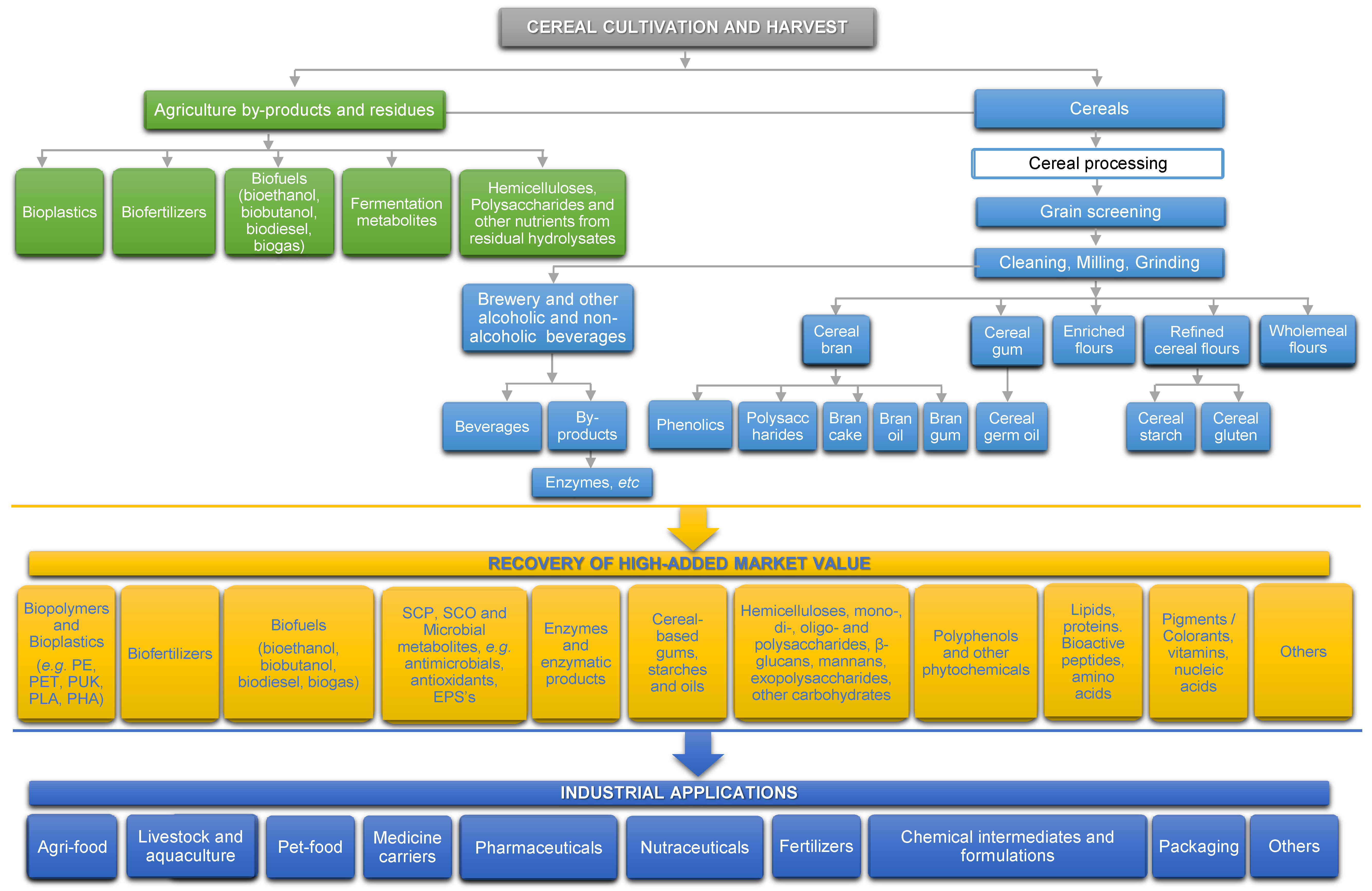
2. Processing of Cereals and the Derived By-Products
3. Dietary Fibres Extracted from Cereal Processing By-Products
4. Phytochemicals Extracted from Cereal Processing By-Products
5. Proteins Extracted from Cereal Processing By-Products
6. Biopolymer-Based Packaging from Cereal By-Products
7. Valorisation of Cereal By-Products through New Product Development (NPD)
8. Environmental and Economic Sustainability Outlook
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Araf | α-L-arabinofuranosyl |
| A/X | Arabinose to xylose ratio |
| AXOS | Arabinoxylan-oligosaccharides |
| AXs | Arabinoxylans |
| BLIS | Bacteriocin-like inhibitory substances |
| bio-PE | Bio-based polyethylene |
| bio-PET | Bio-based polyethylene terephthalate |
| bio-PUR | Bio-based polyurethane |
| BMI | Body mass index |
| C | Carbon |
| CO2 | Carbon dioxide |
| CVD | Cardiovascular disease |
| DP | Degree of polymerization |
| DAG | Diacylglycerols |
| DDGS | Dried distiller’s grain with solubles |
| d.b. | Dry basis |
| EFSA | European Food Safety Authority |
| EU | European Union |
| EPS | Exopolysaccharides |
| FA | Fatty acid |
| FA | Ferulic acid |
| FAO | Food and Agriculture Organization |
| FDA | Food and Drug Administration |
| GABA | γ-aminobutyric acid |
| GI | Glycaemic index |
| H2O2 | Hydrogen peroxide |
| LAB | Lactic acid bacteria |
| LCA | Life cycle assessment |
| LDL | Low density lipoproteins |
| MAG | Monoglycerides |
| MUFA | Monounsaturated fatty acids |
| NPD | New Product Development |
| O2 | Molecular oxygen |
| N | Nitrogen |
| O | Oxygen |
| PHA | Polyhydroxyalkanoates |
| PLA | Polylactic acid |
| AX | Polysaccharide arabinoxylan |
| PUFA | Polyunsaturated fatty acids |
| PEF | Product environmental footprint |
| S&T | Scientific-technological |
| SCO | Single cell oils |
| SCP | Single cell proteins |
| SSF | Solid-state fermentation |
| SE | Sterol esters |
| SWE | Subcritical water extraction |
| SC-CO2 | Supercritical carbon dioxide extraction |
| TAG | Triglycerides |
| UAE | Ultrasonic(/Ultrasound)-assisted extraction |
| UN | United Nations |
| WEAX | Water-extractable arabinoxylans |
| WUAX | Water-unextractable arabinoxylans |
| WHO | World Health Organization |
References
- Pleissner, D.; Qi, Q.; Gao, C.; Rivero, C.P.; Webb, C.; Lin, C.S.K.; Venus, J. Valorization of organic residues for the production of added value chemicals: A contribution to the bio-based economy. Biochem. Eng. J. 2016, 116, 3–16. [Google Scholar] [CrossRef]
- The World Counts Comes. 2020. Available online: https://www.theworldcounts.com/challenges/consumption/foods-and-beverages/food-waste-facts (accessed on 20 July 2020).
- United Nations. Transforming Our World: The 2030 Agenda for Sustainable Development. General Assembly Resolution A/RES/70/1; Division for Sustainable Development, United Nations Department of Economic and Social Affairs; United Nations: New York, NY, USA, 2015; Available online: https://www.un.org/en/development/desa/population/migration/generalassembly/docs/globalcompact/A_RES_70_1_E.pdf (accessed on 20 July 2020).
- European Union (EU). Farm to Fork Strategy—For a Fair, Healthy and Environmentally-Friendly Food System (2020). PDF Report. Available online: https://ec.europa.eu/food/farm2fork_en (accessed on 20 July 2020).
- EU. European Green Deal—Striving to be the First Climate-Neutral Continent. 2020. Available online: https://ec.europa.eu/info/strategy/priorities-2019-2024/european-green-deal_en (accessed on 20 July 2020).
- EU. Directive 2008/98/EC of the European Parliament and of the Council of 19 November 2008 on waste and repealing certain Directives. Off. J. Eur. Union 2008, 312, 3–30. [Google Scholar]
- EU. European Union (EU). Directive 2008/98/EC on Waste (Waste Framework Directive). 2020. Available online: https://ec.europa.eu/environment/waste/framework/ (accessed on 20 July 2020).
- Roth, M.; Jekle, M.; Becker, T. Opportunities for upcycling cereal byproducts with special focus on Distiller’s grains. Trends Food Sci. Technol. 2019, 91, 282–293. [Google Scholar] [CrossRef]
- FAOSTAT. FAO Statistics Division. 2020. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 2 June 2020).
- Belitz, H.D.; Grosch, W. Food Chemistry, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 1999. [Google Scholar]
- Kalo, P.J.; Ollilainen, V.; Rocha, J.M.; Malcata, F.X. Identification of molecular species of simple lipids by normal phase liquid chromatography–positive electrospray tandem mass spectrometry, and application of developed methods in comprehensive analysis of low erucic acid rapeseed oil lipids. Int. J. Mass Spectrom. 2006, 254, 106–121. [Google Scholar] [CrossRef]
- Rocha, J.M.; Kalo, P.J.; Malcata, F.X. Neutral lipids in non-starch lipid and starch lipid extracts from Portuguese sourdough bread. Eur. J. Lipid Sci. Technol. 2010, 112, 1138–1149. [Google Scholar] [CrossRef]
- Rocha, J.M.; Kalo, P.J.; Malcata, F.X. Neutral Lipids in Free, Bound, and Starch Lipid Extracts of Flours, Sourdough, and Portuguese Sourdough Bread Determined by NP-HPLC-ELSD. Cereal Chem. 2011, 88, 400–408. [Google Scholar] [CrossRef]
- Rocha, J.M.; Kalo, P.J.; Malcata, F.X. Composition of neutral lipid classes and content of fatty acids throughout sourdough breadmaking. Eur. J. Lipid Sci. Technol. 2012, 114, 294–305. [Google Scholar] [CrossRef]
- Rocha, J.M.; Kalo, P.J.; Malcata, F.X. Fatty Acid Composition of Non-Starch and Starch Neutral Lipid Extracts of Portuguese Sourdough Bread. J. Am. Oil Chem. Soc. 2012, 89, 2025–2045. [Google Scholar] [CrossRef]
- Rocha, J.M.; Kalo, P.J.; Ollilainen, V.; Malcata, F.X. Separation and identification of neutral cereal lipids by normal phase high-performance liquid chromatography, using evaporative light-scattering and electrospray mass spectrometry for detection. J. Chromatogr. A 2010, 1217, 3013–3025. [Google Scholar] [CrossRef]
- Marques, G.; Rencoret, J.; Gutiérrez, A.; del Río, J.C. Lipophilic compounds from maize fiber and rice husk residues—An abundant and inexpensive source of valuable phytochemicals. Ind. Crop. Prod. 2020, 146. [Google Scholar] [CrossRef]
- Narducci, V.; Finotti, E.; Galli, V.; Carcea, M. Lipids and fatty acids in Italian durum wheat (Triticum Durum Desf.) cultivars. Foods 2019, 8, 223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pastor, K.; Ilić, M.; Vujić, D.; Jovanović, D.; Ačanski, M. Characterization of Fatty Acids in Cereals and Oilseeds from the Republic of Serbia by Gas Chromatography–Mass Spectrometry (GC/MS) with Chemometrics. Anal. Lett. 2020, 53, 1177–1189. [Google Scholar] [CrossRef]
- Kouřimská, L.; Sabolová, M.; Horčička, P.; Rys, S.; Božik, M. Lipid content, fatty acid profile, and nutritional value of new oat cultivars. J. Cereal Sci. 2018, 84, 44–48. [Google Scholar] [CrossRef]
- Chatzifragkou, A.; Charalampopoulos, D. 3—Distiller’s dried grains with solubles (DDGS) and intermediate products as starting materials in biorefinery strategies. In Sustainable Recovery and Reutilization of Cereal Processing By-Products; Galanakis, C.M., Ed.; Woodhead Publishing: Cambridge, UK, 2018; pp. 63–86. [Google Scholar] [CrossRef]
- Papageorgiou, M.; Skendi, A. 1—Introduction to cereal processing and by-products. In Sustainable Recovery and Reutilization of Cereal Processing By-Products; Galanakis, C.M., Ed.; Woodhead Publishing: Cambridge, UK, 2018; pp. 1–25. [Google Scholar] [CrossRef]
- Skendi, A.; Harasym, J.; Galanakis, C.M. 7—Recovery of high added-value compounds from brewing and distillate processing by-products. In Sustainable Recovery and Reutilization of Cereal Processing By-Products; Galanakis, C.M., Ed.; Woodhead Publishing: Cambridge, UK, 2018; pp. 189–225. [Google Scholar] [CrossRef]
- Ma, S.; Wang, C.; Li, L.; Wang, X. Effects of particle size on the quality attributes of wheat flour made by the milling process. Cereal Chem. 2020, 97, 172–182. [Google Scholar] [CrossRef]
- Létang, C.; Samson, M.F.; Lasserre, T.M.; Chaurand, M.; Abécassis, J. Production of Starch with Very Low Protein Content from Soft and Hard Wheat Flours by Jet Milling and Air Classification. Cereal Chem. 2002, 79, 535–543. [Google Scholar] [CrossRef]
- Barrett, E.M.; Foster, S.I.; Beck, E.J. Whole grain and high-fibre grain foods: How do knowledge, perceptions and attitudes affect food choice? Appetite 2020, 149. [Google Scholar] [CrossRef] [PubMed]
- Raddatz, G.C.; de Souza da Fonseca, B.; Poletto, G.; Jacob-Lopes, E.; Cichoski, A.J.; Muller, E.I.; Flores, E.M.M.; de Bona da Silva, C.; Ragagnin de Menezes, C. Influence of the prebiotics hi-maize, inulin and rice bran on the viability of pectin microparticles containing Lactobacillus acidophilus LA-5 obtained by internal gelation/emulsification. Powder Technol. 2020, 362, 409–415. [Google Scholar] [CrossRef]
- Rocha, J.M. Microencapsulation of probiotic bacteria. Ecronicon Microbiol. 2016, 3, 480–481. [Google Scholar]
- Demirci, T.; Aktaş, K.; Sözeri, D.; Öztürk, H.İ.; Akın, N. Rice bran improve probiotic viability in yoghurt and provide added antioxidative benefits. J. Funct. Foods 2017, 36, 396–403. [Google Scholar] [CrossRef]
- Kim, S.-M.; Lim, S.-T. Enhanced antioxidant activity of rice bran extract by carbohydrase treatment. J. Cereal Sci. 2016, 68, 116–121. [Google Scholar] [CrossRef]
- Mingyai, S.; Kettawan, A.; Srikaeo, K.; Singanusong, R. Physicochemical and Antioxidant Properties of Rice Bran Oils Produced from Colored Rice Using Different Extraction Methods. J. Oleo Sci. 2017, 66, 565–572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sierra, S.; Lara–Villoslada, F.; Olivares, M.; Jiménez, J.; Boza, J.; Xaus, J. Increased immune responsein mice consuming rice bran oil. Eur. J. Nutr. 2005, 44, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Mahesh, M.S.; Thakur, S.S. Rice gluten meal, an agro-industrial by-product, supports performance attributes in lactating Murrah buffaloes (Bubalus bubalis). J. Clean. Prod. 2018, 177, 655–664. [Google Scholar] [CrossRef]
- Paraskevopoulou, A.; Anagnostara, I.; Bezati, G.; Rizou, T.; Pavlidou, E.; Vouvoudi, E.; Kiosseoglou, V. Water extraction residue from maize milling by-product as a potential functional ingredient for the enrichment with fibre of cakes. LWT 2020, 129. [Google Scholar] [CrossRef]
- Saulnier, L.; Vigouroux, J.; Thibault, J.-F. Isolation and partial characterization of feruloylated oligosaccharides from maize bran. Carbohydr. Res. 1995, 272, 241–253. [Google Scholar] [CrossRef]
- Carvajal-Millan, E.; Rascón-Chu, A.; Márquez-Escalante, J.A.; Micard, V.; León, N.P.d.; Gardea, A. Maize bran gum: Extraction, characterization and functional properties. Carbohydr. Polym. 2007, 69, 280–285. [Google Scholar] [CrossRef]
- Yan, W.; Zhang, B.; Yadav, M.P.; Feng, L.; Yan, J.; Jia, X.; Yin, L. Corn fiber gum-soybean protein isolate double network hydrogel as oral delivery vehicles for thermosensitive bioactive compounds. Food Hydrocoll. 2020, 107, 105865. [Google Scholar] [CrossRef]
- Marquez-Escalante, J.A.; Carvajal-Millan, E. Feruloylated arabinoxylans from Maize Distiller’s dried grains with solubles: Effect of feruloyl esterase on their macromolecular characteristics, gelling, and antioxidant properties. Sustainability 2019, 11. [Google Scholar] [CrossRef] [Green Version]
- Kalscheur, K.F.; Garcia, A.D.; Schingoethe, D.J.; Diaz Royón, F.; Hippen, A.R. Chapter 7. Feeding biofuel co-products to dairy cattle. In Biofuel Co-products as Livestock Feed: Opportunities and Challenges; Makkar, H., Ed.; Food and Agriculture Organization of the United Nation (FAO): Rome, Italy, 2012; pp. 115–154. [Google Scholar]
- FAO. Cereals and Cereal Products. Chapter 11. Fodder Crops and Products. In Definition and Classification of Commodities. 1994. Available online: http://www.fao.org/es/faodef/fdef01e.htm#:~:text=CEREALS%20are%20generally%20of%20the,grazingare%20classified%20as%20fodder%20crops.&text=The%20FAO%20definitions%20cover%2017,is%20a%20component%20of%20maize (accessed on 15 June 2020).
- Rocha, C.; Gonalves, M.P.; Teixeira, J.A. Immobilization of trypsin on spent grains for whey protein hydrolysis. Process Biochem. 2011, 46, 505–511. [Google Scholar] [CrossRef] [Green Version]
- Girelli, A.M.; Astolfi, M.L.; Scuto, F.R. Agro-industrial wastes as potential carriers for enzyme immobilization: A review. Chemosphere 2020, 244, 125368. [Google Scholar] [CrossRef]
- Pauline, M.; Roger, P.; Sophie Natacha Nina, N.E.; Arielle, T.; Eugene, E.E.; Robert, N. Physico-chemical and nutritional characterization of cereals brans enriched breads. Sci. Afr. 2020, 7. [Google Scholar] [CrossRef]
- Koletta, P.; Irakli, M.; Papageorgiou, M.; Skendi, A. Physicochemical and technological properties of highly enriched wheat breads with wholegrain non wheat flours. J. Cereal Sci. 2014, 60, 561–568. [Google Scholar] [CrossRef]
- Özkaya, B.; Baumgartner, B.; Özkaya, H. Effects of concentrated and dephytinized wheat bran and rice bran addition on bread properties. J. Texture Stud. 2018, 49, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Hussein, A.M.S.; Ibrahim, G.E. Effects of various brans on quality and volatile compounds of bread. Foods Raw Mater. 2019, 7. [Google Scholar] [CrossRef]
- Lin, S.; Gao, J.; Jin, X.; Wang, Y.; Dong, Z.; Ying, J.; Zhou, W. Whole-wheat flour particle size influences dough properties, bread structure and in vitro starch digestibility. Food Funct. 2020, 11, 3610–3620. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Sun, X.; Raddatz, J.; Chen, G. Effects of microfluidization on microstructure and physicochemical properties of corn bran. J. Cereal Sci. 2013, 58, 355–361. [Google Scholar] [CrossRef]
- Wang, T.; Zhu, Y.; Sun, X.; Raddatz, J.; Zhou, Z.; Chen, G. Effect of microfluidisation on antioxidant properties of corn bran. Food Chem. 2014, 152, 37–45. [Google Scholar] [CrossRef]
- Ortiz de Erive, M.; Wang, T.; He, F.; Chen, G. Development of high-fiber wheat bread using microfluidized corn bran. Food Chem. 2020, 310, 125921. [Google Scholar] [CrossRef]
- Du, B.; Meenu, M.; Xu, B. Insights into Improvement of Physiochemical and Biological Properties of Dietary Fibers from Different Sources via Micron Technology. Food Rev. Int. 2020, 36, 367–383. [Google Scholar] [CrossRef]
- Sarker, N.C.; Ray, P.; Pfau, C.; Kalavacharla, V.; Hossain, K.; Quadir, M. Development of Functional Nanomaterials from Wheat Bran Derived Arabinoxylan for Nucleic Acid Delivery. J. Agric. Food Chem. 2020, 68, 4367–4373. [Google Scholar] [CrossRef]
- Zokaityte, E.; Cernauskas, D.; Klupsaite, D.; Lele, V.; Starkute, V.; Zavistanaviciute, P.; Ruzauskas, M.; Gruzauskas, R.; Juodeikiene, G.; Rocha, J.M.; et al. Bioconversion of milk permeate with selected lactic acid bacteria strains and apple by-products into beverages with antimicrobial properties and enriched with galactooligosaccharides. Microorganisms 2020, 8, 1182. [Google Scholar] [CrossRef] [PubMed]
- Bartkiene, E.; Lele, V.; Ruzauskas, M.; Domig, K.J.; Starkute, V.; Zavistanaviciute, P.; Bartkevics, V.; Pugajeva, I.; Klupsaite, D.; Juodeikiene, G.; et al. Lactic Acid Bacteria Isolation from Spontaneous Sourdough and Their Characterization Including Antimicrobial and Antifungal Properties Evaluation. Microorganisms 2019, 8. [Google Scholar] [CrossRef] [Green Version]
- Low, D.Y.; Pluschke, A.M.; Gerrits, W.J.J.; Zhang, D.; Shelat, K.J.; Gidley, M.J.; Williams, B.A. Cereal dietary fibres influence retention time of digesta solid and liquid phases along the gastrointestinal tract. Food Hydrocoll. 2020, 104. [Google Scholar] [CrossRef]
- FDA. Food and Drug Administration, Revision of the Nutrition and Supplement Facts Label. Fed. Regist. 2016, 81, 33741–33999. [Google Scholar]
- EU. Regulation (EU) No 1169/2011 of the European parliament and of the Council on the provision of food information to consumers. Off. J. Eur. Union 2011, L 304, 18–63. [Google Scholar]
- EU. Regulation (EC) No 1924/2006 of the European Parliament and of the Council on nutrition and health claims made on foods. Off. J. Eur. Union 2006, L 404, 9–24. [Google Scholar]
- EFSA. European Food Safety Authority, Scientific Opinion on Dietary Reference Values for carbohydrates and dietary fibre. EFSA Panel on Dietetic Products, Nutrition, and Allergies. EFSA J. 2010, 8, 1462. [Google Scholar]
- Barrett, E.M.; Amoutzopoulos, B.; Batterham, M.J.; Ray, S.; Beck, E.J. Whole grain intake compared with cereal fibre intake in association to CVD risk factors: A cross-sectional analysis of the National Diet and Nutrition Survey (UK). Public Health Nutr. 2020, 23, 1392–1403. [Google Scholar] [CrossRef]
- Barrett, E.M.; Batterham, M.J.; Beck, E.J. Whole grain and cereal fibre intake in the Australian Health Survey: Associations to CVD risk factors. Public Health Nutr. 2020, 23, 1404–1413. [Google Scholar] [CrossRef]
- Della Pepa, G.; Vetrani, C.; Vitale, M.; Riccardi, G. Wholegrain Intake and Risk of Type 2 Diabetes: Evidence from Epidemiological and Intervention Studies. Nutrients 2018, 10, 1288. [Google Scholar] [CrossRef] [Green Version]
- Marcotuli, I.; Colasuonno, P.; Hsieh, Y.S.Y.; Fincher, G.B.; Gadaleta, A. Non-starch polysaccharides in durum wheat: A review. Int. J. Mol. Sci. 2020, 21, 2933. [Google Scholar] [CrossRef]
- Cruz-Requena, M.; Escobedo-García, S.; Salas-Tovar, J.A.; Mora-Cura, Y.; Chávez-González, M.L.; Castillo-Reyes, F.; Flores-Gallegos, A.C.; Rodríguez-Herrera, R. Chapter 1—Definitions and Regulatory Perspectives of Dietary Fibers. In Dietary Fiber: Properties, Recovery, and Applications; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 1–25. [Google Scholar] [CrossRef]
- Skendi, A.; Biliaderis, C.G.; Lazaridou, A.; Izydorczyk, M.S. Structure and rheological properties of water soluble β-glucans from oat cultivars of Avena sativa and Avena bysantina. J. Cereal Sci. 2003, 38, 15–31. [Google Scholar] [CrossRef]
- Cui, W.; Wood, P.J. Relationships between structural features, molecular weight and rheological properties of cereal β-D-glucans. In Hydrocolloids; Nishinari, K., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2000; pp. 159–168. [Google Scholar] [CrossRef]
- Zheng, G.H.; Rossnagel, B.G.; Tyler, R.T.; Bhatty, R.S. Distribution of β-Glucan in the Grain of Hull-less Barley. Cereal Chem. 2000, 77, 140–144. [Google Scholar] [CrossRef]
- Miller, S.S.; Fulcher, R.G. Distribution of (1→3) (1→4)-β-D-glucan in kernels of oats and barley using micro spectrofluorometry. Cereal Chem. 1994, 71, 64–68. [Google Scholar]
- Lazaridou, A.; Biliaderis, C.G. Molecular aspects of cereal β-glucan functionality: Physical properties, technological applications and physiological effects. J. Cereal Sci. 2007, 46, 101–118. [Google Scholar] [CrossRef]
- Mikkelsen, M.S.; Jespersen, B.M.; Larsen, F.H.; Blennow, A.; Engelsen, S.B. Molecular structure of large-scale extracted β-glucan from barley and oat: Identification of a significantly changed block structure in a high β-glucan barley mutant. Food Chem. 2013, 136, 130–138. [Google Scholar] [CrossRef]
- Vaikousi, H.; Biliaderis, C.G.; Izydorczyk, M.S. Solution flow behavior and gelling properties of water-soluble barley (1→3,1→4)-β-glucans varying in molecular size. J. Cereal Sci. 2004, 39, 119–137. [Google Scholar] [CrossRef]
- Abdel-Haleem, A.M.H.; Agwa, A.M.; Mahgoub, S.A.; Shehata, W.M. Characterization of β-glucan gum for food applications as influenced by genotypic variations in three hulless barley varieties. J. Food Sci. 2020, 85, 1689–1698. [Google Scholar] [CrossRef]
- Wolever, T.M.S.; Tosh, S.M.; Spruill, S.E.; Jenkins, A.L.; Ezatagha, A.; Duss, R.; Johnson, J.; Chu, Y.; Steinert, R.E. Increasing oat β-glucan viscosity in a breakfast meal slows gastric emptying and reduces glycemic and insulinemic responses but has no effect on appetite, food intake, or plasma ghrelin and PYY responses in healthy humans: A randomized, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2020, 111, 319–328. [Google Scholar] [CrossRef]
- Maheshwari, G.; Sowrirajan, S.; Joseph, B. β-Glucan, a dietary fiber in effective prevention of lifestyle diseases—An insight. Bioact. Carbohydr. Diet. Fibre 2019, 19. [Google Scholar] [CrossRef]
- Kuley, E.; Özyurt, G.; Özogul, I.; Boga, M.; Akyol, I.; Rocha, J.M.; Özogul, F. The Role of Selected Lactic Acid Bacteria on Organic Acid Accumulation during Wet and Spray-Dried Fish-based Silages. Contributions to the Winning Combination of Microbial Food Safety and Environmental Sustainability. Microorganisms 2020, 8, 172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Păcularu-Burada, B.; Georgescu, L.A.; Vasile, M.A.; Rocha, J.M.; Bahrim, G.-E. Selection of Wild Lactic Acid Bacteria Strains as Promoters of Postbiotics in Gluten-Free Sourdoughs. Microorganisms 2020, 8, 643. [Google Scholar] [CrossRef] [PubMed]
- Phillips, G.O.; Williams, P.A. Handbook of Hydrocolloids; Woodhead Publishing Limited: Oxford, UK, 2009. [Google Scholar]
- Cyran, M.R. Dietary Fiber Arabinoxylans in Processed Rye: Milling- and Breadmaking-Induced Changes. Milling- and Breadmaking-Induced Changes. In Processing and Impact on Active Components in Food; Academic Press: Cambridge, MA, USA, 2015; pp. 319–328. [Google Scholar] [CrossRef]
- Rosicka-Kaczmarek, J.; Komisarczyk, A.; Nebesny, E.; Makowski, B. The influence of arabinoxylans on the quality of grain industry products. Eur. Food Res. Technol. 2016, 242, 295–303. [Google Scholar] [CrossRef]
- Fadel, A.; Plunkett, A.; Ashworth, J.; Mahmoud, A.M.; Ranneh, Y.; El Mohtadi, M.; Li, W. The effect of extrusion screw-speed on the water extractability and molecular weight distribution of arabinoxylans from defatted rice bran. J. Food Sci. Technol. 2018, 55, 1201–1206. [Google Scholar] [CrossRef] [PubMed]
- Skendi, A.; Biliaderis, C.G.; Izydorczyk, M.S.; Zervou, M.; Zoumpoulakis, P. Structural variation and rheological properties of water-extractable arabinoxylans from six Greek wheat cultivars. Food Chem. 2011, 126, 526–536. [Google Scholar] [CrossRef]
- Marcotuli, I.; Hsieh, Y.S.Y.; Lahnstein, J.; Yap, K.; Burton, R.A.; Blanco, A.; Fincher, G.B.; Gadaleta, A. Structural Variation and Content of Arabinoxylans in Endosperm and Bran of Durum Wheat (Triticum turgidum L.). J. Agric. Food Chem. 2016, 64, 2883–2892. [Google Scholar] [CrossRef] [PubMed]
- Skendi, A.; Biliaderis, C.G. Gelation of wheat arabinoxylans in the presence of Cu+2 and in aqueous mixtures with cereal β-glucans. Food Chem. 2016, 203, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.J.; Inglett, G.E.; Liu, S.X. Utilisation of corn (Zea mays) bran and corn fiber in the production of food components. J. Sci. Food Agric. 2010, 90, 915–924. [Google Scholar] [CrossRef]
- Malunga, L.N.; Beta, T. Antioxidant capacity of water-extractable arabinoxylan from commercial barley, wheat, and wheat fractions. Cereal Chem. 2015, 92, 29–36. [Google Scholar] [CrossRef]
- Demuth, T.; Betschart, J.; Nyström, L. Structural modifications to water-soluble wheat bran arabinoxylan through milling and extrusion. Carbohydr. Polym. 2020, 240. [Google Scholar] [CrossRef]
- Daou, C.; Zhang, H. Study on functional properties of physically modified dietary fibres derived from defatted rice bran. J. Agric. Sci. 2012, 4, 85–97. [Google Scholar] [CrossRef]
- Rashid, S.; Rakha, A.; Anjum, F.M.; Ahmed, W.; Sohail, M. Effects of extrusion cooking on the dietary fibre content and Water Solubility Index of wheat bran extrudates. Int. J. Food Sci. Technol. 2015, 50, 1533–1537. [Google Scholar] [CrossRef]
- Fadel, A.; Ashworth, J.; Plunkett, A.; Mahmoud, A.M.; Ranneh, Y.; Li, W. Improving the extractability of arabinoxylans and the molecular weight of wheat endosperm using extrusion processing. J. Cereal Sci. 2018, 84, 55–61. [Google Scholar] [CrossRef]
- Jeon, S.-J.; Singkhornart, S.; Ryu, G.-H. The Effect of Extrusion Conditions on Water-extractable Arabinoxylans from Corn Fiber. Prev Nutr Food Sci 2014, 19, 124–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roye, C.; Henrion, M.; Chanvrier, H.; De Roeck, K.; De Bondt, Y.; Liberloo, I.; King, R.; Courtin, C.M. Extrusion-Cooking Modifies Physicochemical and Nutrition-Related Properties of Wheat Bran. Foods 2020, 9, 738. [Google Scholar] [CrossRef] [PubMed]
- Broekaert, W.F.; Courtin, C.M.; Verbeke, K.; van de Wiele, T.; Verstraete, W.; Delcour, J.A. Prebiotic and other health-related effects of cereal-derived arabinoxylans, arabinoxylan-oligosaccharides, and xylooligosaccharides. Crit. Rev. Food Sci. Nutr. 2011, 51, 178–194. [Google Scholar] [CrossRef]
- De Paepe, K.; Verspreet, J.; Rezaei, M.N.; Martinez, S.H.; Meysman, F.; Van De Walle, D.; Dewettinck, K.; Courtin, C.M.; Van De Wiele, T. Modification of wheat bran particle size and tissue composition affects colonisation and metabolism by human faecal microbiota. Food Funct. 2019, 10, 379–396. [Google Scholar] [CrossRef]
- Vangsøe, C.T.; Sørensen, J.F.; Bach Knudsen, K.E. Aleurone cells are the primary contributor to arabinoxylan oligosaccharide production from wheat bran after treatment with cell wall-degrading enzymes. Int. J. Food Sci. Technol. 2019, 54, 2847–2853. [Google Scholar] [CrossRef]
- Saeed, F.; Pasha, I.; Anjum, F.M.; Sultan, J.I.; Arshad, M. Arabinoxylan and Arabinogalactan Content in Different Spring Wheats. Int. J. Food Prop. 2014, 17, 713–721. [Google Scholar] [CrossRef]
- Baiano, A. Recovery of Biomolecules from Food Wastes—A Review. Molecules 2014, 19, 14821–14842. [Google Scholar] [CrossRef] [Green Version]
- Fava, F.; Totaro, G.; Diels, L.; Reis, M.; Duarte, J.; Carioca, O.B.; Poggi-Varaldo, H.M.; Ferreira, B.S. Biowaste biorefinery in Europe: Opportunities and research & development needs. New Biotechnol. 2015, 32, 100–108. [Google Scholar] [CrossRef]
- Matassa, S.; Boon, N.; Pikaar, I.; Verstraete, W. Microbial protein: Future sustainable food supply route with low environmental footprint. Microb. Biotechnol. 2016, 9, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Najafpour, G.D. Single-Cell Protein. In Biochemical Engineering and Biotechnology; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Thomas, L.; Larroche, C.; Pandey, A. Current developments in solid-state fermentation. Biochem. Eng. J. 2013, 81, 146–161. [Google Scholar] [CrossRef]
- Williams, I.D.; Schneider, F.; Syversen, F. The “food waste challenge” can be solved. Waste Manag. 2015, 41, 1–2. [Google Scholar] [CrossRef]
- Decimo, M.; Quattrini, M.; Ricci, G.; Fortina, M.G.; Brasca, M.; Silvetti, T.; Manini, F.; Erba, D.; Criscuoli, F.; Casiraghi, M.C. Evaluation of microbial consortia and chemical changes in spontaneous maize bran fermentation. Amb Express 2017, 7. [Google Scholar] [CrossRef]
- Messia, M.C.; Reale, A.; Maiuro, L.; Candigliota, T.; Sorrentino, E.; Marconi, E. Effects of pre-fermented wheat bran on dough and bread characteristics. J. Cereal Sci. 2016, 69, 138–144. [Google Scholar] [CrossRef]
- Zhao, H.M.; Guo, X.N.; Zhu, K.X. Impact of solid state fermentation on nutritional, physical and flavor properties of wheat bran. Food Chem. 2017, 217, 28–36. [Google Scholar] [CrossRef]
- Jiang, Y.; Bai, X.; Lang, S.; Zhao, Y.; Liu, C.; Yu, L. Optimization of ultrasonic-microwave assisted alkali extraction of arabinoxylan from the corn bran using response surface methodology. Int. J. Biol. Macromol. 2019, 128, 452–458. [Google Scholar] [CrossRef]
- Li, L.; Ma, S.; Fan, L.; Zhang, C.; Pu, X.; Zheng, X.; Wang, X. The influence of ultrasonic modification on arabinoxylans properties obtained from wheat bran. Int. J. Food Sci. Technol. 2016, 51, 2338–2344. [Google Scholar] [CrossRef]
- Li, Y.; Yang, C. Synthesis and properties of feruloyl corn bran arabinoxylan esters. Int. J. Cosmet. Sci. 2016, 38, 238–245. [Google Scholar] [CrossRef]
- Truong, K.T.P.; Rumpagaporn, P. Oligosaccharides preparation from rice bran arabinoxylan by two different commercial endoxylanase enzymes. J. Nutr. Sci. Vitaminol. 2019, 65, S171–S174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, R.; Yang, J.; Jang, J.M.; Liu, J.; Zhang, Y.; Liu, L.; Yuan, H. Efficient ferulic acid and xylo-oligosaccharides production by a novel multi-modular bifunctional xylanase/feruloyl esterase using agricultural residues as substrates. Bioresour. Technol. 2020, 297. [Google Scholar] [CrossRef] [PubMed]
- Yuwang, P.; Sulaeva, I.; Hell, J.; Henniges, U.; Böhmdorfer, S.; Rosenau, T.; Chitsomboon, B.; Tongta, S. Phenolic compounds and antioxidant properties of arabinoxylan hydrolysates from defatted rice bran. J. Sci. Food Agric. 2018, 98, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Moza, J.; Gujral, H.S. Influence of non-starchy polysaccharides on barley milling behavior and evaluating bioactive composition of milled fractions. Food Chem. 2017, 218, 137–143. [Google Scholar] [CrossRef]
- Bijalwan, V.; Ali, U.; Kesarwani, A.K.; Yadav, K.; Mazumder, K. Hydroxycinnamic acid bound arabinoxylans from millet brans-structural features and antioxidant activity. Int. J. Biol. Macromol. 2016, 88, 296–305. [Google Scholar] [CrossRef]
- Ma, F.; Lee, Y.Y.; Baik, B.K. Bran characteristics influencing quality attributes of whole wheat Chinese steamed bread. J. Cereal Sci. 2018, 79, 431–439. [Google Scholar] [CrossRef]
- Mense, A.L.; Shi, Y.C. Dissolution of Wheat Bran by NaOH/Urea Solutions and Structure of Soluble Materials. ACS Sustain. Chem. Eng. 2018, 6, 4264–4271. [Google Scholar] [CrossRef]
- Zhang, Z.; Smith, C.; Li, W.; Ashworth, J. Characterization of Nitric Oxide Modulatory Activities of Alkaline-Extracted and Enzymatic-Modified Arabinoxylans from Corn Bran in Cultured Human Monocytes. J. Agric. Food Chem. 2016, 64, 8128–8137. [Google Scholar] [CrossRef]
- Anderson, C.; Simsek, S. A novel combination of methods for the extraction and purification of arabinoxylan from byproducts of the cereal industry. J. Food Meas. Charact. 2019, 13, 1049–1057. [Google Scholar] [CrossRef]
- Bender, D.; Schmatz, M.; Novalin, S.; Nemeth, R.; Chrysanthopoulou, F.; Tömösközi, S.; Török, K.; Schoenlechner, R.; D’Amico, S. Chemical and rheological characterization of arabinoxylan isolates from rye bran. Chem. Biol. Technol. Agric. 2017, 4. [Google Scholar] [CrossRef]
- Herrera-Balandrano, D.D.; Báez-González, J.G.; Carvajal-Millán, E.; Méndez-Zamora, G.; Urías-Orona, V.; Amaya-Guerra, C.A.; Niño-Medina, G. Feruloylated arabinoxylans from nixtamalized maize bran byproduct: A functional ingredient in frankfurter sausages. Molecules 2019, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rudjito, R.C.; Ruthes, A.C.; Jiménez-Quero, A.; Vilaplana, F. Feruloylated arabinoxylans from wheat bran: Optimization of extraction process and validation at pilot scale. ACS Sustain. Chem. Eng. 2019, 7, 13167–13177. [Google Scholar] [CrossRef]
- Andersson, A.A.M.; Andersson, R.; Jonsäll, A.; Andersson, J.; Fredriksson, H. Effect of Different Extrusion Parameters on Dietary Fiber in Wheat Bran and Rye Bran. J. Food Sci. 2017, 82, 1344–1350. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, L.; Chen, Z.; Li, Y.; Li, J. Facile and green preparation of diverse arabinoxylan hydrogels from wheat bran by combining subcritical water and enzymatic crosslinking. Carbohydr. Polym. 2020, 241. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Wang, L.; Wang, R.; Luo, X.; Li, Y.; Li, J.; Li, Y.; Chen, Z. Ultrasound-assisted extraction from defatted oat (Avena sativa L.) bran to simultaneously enhance phenolic compounds and β-glucan contents: Compositional and kinetic studies. J. Food Eng. 2018, 222, 1–10. [Google Scholar] [CrossRef]
- Giordano, D.; Locatelli, M.; Travaglia, F.; Bordiga, M.; Reyneri, A.; Coïsson, J.D.; Blandino, M. Bioactive compound and antioxidant activity distribution in roller-milled and pearled fractions of conventional and pigmented wheat varieties. Food Chem. 2017, 233, 483–491. [Google Scholar] [CrossRef]
- Kim, E.H.; Lee, Y.J.; Jang, G.Y.; Kim, M.Y.; Yoon, N.; Ji, Y.M.; Lee, M.J.; Lee, J.; Jeong, H.S. Functional components of different varieties of barley powder with varying degrees of milling. Korean J. Food Sci. Technol. 2016, 48, 256–261. [Google Scholar] [CrossRef]
- Šimić, G.; Horvat, D.; Lalić, A.; Komlenić, D.K.; Abičić, I.; Zdunić, Z. Distribution of β-glucan, phenolic acids, and proteins as functional phytonutrients of hull-less barley grain. Foods 2019, 8. [Google Scholar] [CrossRef] [Green Version]
- Wiege, I.; Sluková, M.; Vaculová, K.; Pančíková, B.; Wiege, B. Characterization of milling fractions from new sources of barley for use in food industry. Starch-Stärke 2016, 68, 321–328. [Google Scholar] [CrossRef]
- Jung, T.D.; Shin, G.H.; Kim, J.M.; Choi, S.I.; Lee, J.H.; Lee, S.J.; Park, S.J.; Woo, K.S.; Oh, S.K.; Lee, O.H. Comparative analysis of γ-oryzanol, β-glucan, total phenolic content and antioxidant activity in fermented rice bran of different varieties. Nutrients 2017, 9, 571. [Google Scholar] [CrossRef] [Green Version]
- Karimi, R.; Azizi, M.H.; Sahari, M.A.; Kazem, A.E. In vitro fermentation profile of soluble dietary fibers obtained by different enzymatic extractions from barley bran. Bioact. Carbohydr. Diet. Fibre 2020, 21. [Google Scholar] [CrossRef]
- Karimi, R.; Azizi, M.H.; Xu, Q. Effect of different enzymatic extractions on molecular weight distribution, rheological and microstructural properties of barley bran β-glucan. Int. J. Biol. Macromol. 2019, 126, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Karimi, R.; Azizi, M.H.; Xu, Q.; Sahari, M.A.; Hamidi, Z. Enzymatic removal of starch and protein during the extraction of dietary fiber from barley bran. J. Cereal Sci. 2018, 83, 259–265. [Google Scholar] [CrossRef]
- Brahma, S.; Weier, S.A.; Rose, D.J. Moisture content during extrusion of oats impacts the initial fermentation metabolites and probiotic bacteria during extended fermentation by human fecal microbiota. Food Res. Int. 2017, 97, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Comino, P.; Collins, H.; Lahnstein, J.; Gidley, M.J. Effects of diverse food processing conditions on the structure and solubility of wheat, barley and rye endosperm dietary fibre. J. Food Eng. 2016, 169, 228–237. [Google Scholar] [CrossRef] [Green Version]
- Honců, I.; Sluková, M.; Vaculová, K.; Sedláčková, I.; Wiege, B.; Fehling, E. The effects of extrusion on the content and properties of dietary fibre components in various barley cultivars. J. Cereal Sci. 2016, 68, 132–139. [Google Scholar] [CrossRef]
- Sayanjali, S.; Ying, D.; Sanguansri, L.; Buckow, R.; Augustin, M.A.; Gras, S.L. The effect of extrusion on the functional properties of oat fibre. LWT 2017, 84, 106–113. [Google Scholar] [CrossRef]
- Kodama, S.; Shoda, T.; Machmudah, S.; Wahyudiono; Kanda, H.; Goto, M. Extraction of β–glucan by hydrothermal liquidization of barley grain in a semi-batch reactor. Sep. Sci. Technol. 2016, 51, 278–289. [Google Scholar] [CrossRef]
- Yoo, H.U.; Ko, M.J.; Chung, M.S. Hydrolysis of beta-glucan in oat flour during subcritical-water extraction. Food Chem. 2020, 308. [Google Scholar] [CrossRef]
- Wang, W.; Guo, J.; Zhang, J.; Peng, J.; Liu, T.; Xin, Z. Isolation, identification and antioxidant activity of bound phenolic compounds present in rice bran. Food Chem. 2015, 171, 40–49. [Google Scholar] [CrossRef]
- Wang, L.; Weller, C.L. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci. Technol. 2006, 17, 300–312. [Google Scholar] [CrossRef]
- Spinelli, S.; Conte, A.; Lecce, L.; Padalino, L.; Del Nobile, M.A. Supercritical carbon dioxide extraction of brewer’s spent grain. J. Supercrit. Fluids 2016, 107, 69–74. [Google Scholar] [CrossRef]
- Benito-Román, O.; Varona, S.; Sanz, M.T.; Beltrán, S. Valorization of rice bran: Modified supercritical CO2 extraction of bioactive compounds. J. Ind. Eng. Chem. 2019, 80, 273–282. [Google Scholar] [CrossRef]
- Huang, H.; Wang, Z.; Aalim, H.; Limwachiranon, J.; Li, L.; Duan, Z.; Ren, G.; Luo, Z. Green recovery of phenolic compounds from rice byproduct (rice bran) using glycerol based on viscosity, conductivity and density. Int. J. Food Sci. Technol. 2019, 54, 1363–1371. [Google Scholar] [CrossRef]
- López-Perea, P.; Guzmán-Ortiz, F.A.; Román-Gutiérrez, A.D.; Castro-Rosas, J.; Gómez-Aldapa, C.A.; Rodríguez-Marín, M.L.; Falfán-Cortés, R.N.; González-Olivares, L.G.; Torruco-Uco, J.G. Bioactive compounds and antioxidant activity of wheat bran and barley husk in the extracts with different polarity. Int. J. Food Prop. 2019, 22, 646–658. [Google Scholar] [CrossRef]
- Călinoiu, L.F.; Vodnar, D.C. Thermal processing for the release of phenolic compounds from wheat and oat bran. Biomolecules 2020, 10. [Google Scholar] [CrossRef] [Green Version]
- Pazo-Cepeda, V.; Benito-Román, Ó.; Navarrete, A.; Alonso, E. Valorization of wheat bran: Ferulic acid recovery using pressurized aqueous ethanol solutions. Waste Biomass Valoriz. 2019. [Google Scholar] [CrossRef]
- Peanparkdee, M.; Yamauchi, R.; Iwamoto, S. Characterization of antioxidants extracted from Thai riceberry bran using ultrasonic-assisted and conventional solvent extraction methods. Food Bioprocess Technol. 2018, 11, 713–722. [Google Scholar] [CrossRef]
- Das, A.B.; Goud, V.V.; Das, C. Extraction of phenolic compounds and anthocyanin from black and purple rice bran (Oryza sativa L.) using ultrasound: A comparative analysis and phytochemical profiling. Ind. Crop. Prod. 2017, 95, 332–341. [Google Scholar] [CrossRef]
- Luo, X.; Cui, J.; Zhang, H.; Duan, Y.; Zhang, D.; Cai, M.; Chen, G. Ultrasound assisted extraction of polyphenolic compounds from red sorghum (Sorghum bicolor L.) bran and their biological activities and polyphenolic compositions. Ind. Crop. Prod. 2018, 112, 296–304. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, M.; Liu, X.; Zhong, K.; Tong, L.; Zhou, X.; Zhou, S. Effect of steam explosion-assisted extraction on phenolic acid profiles and antioxidant properties of wheat bran. J. Sci. Food Agric. 2016, 96, 3484–3491. [Google Scholar] [CrossRef] [PubMed]
- Pourali, O.; Asghari, F.S.; Yoshida, H. Production of phenolic compounds from rice bran biomass under subcritical water conditions. Chem. Eng. J. 2010, 160, 259–266. [Google Scholar] [CrossRef]
- Ramos-Enríquez, J.R.; Ramírez-Wong, B.; Robles-Sánchez, R.M.; Robles-Zepeda, R.E.; González-Aguilar, G.A.; Gutiérrez-Dorado, R. Effect of extrusion conditions and the optimization of phenolic compound content and antioxidant activity of wheat bran using Response Surface Methodology. Plant Foods Hum. Nutr. 2018, 73, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Li, Y.; Bai, Y.; Yuan, C.; Wu, C.; Hu, Y. Intelligent gelatin/oxidized chitin nanocrystals nanocomposite films containing black rice bran anthocyanins for fish freshness monitorings. Int. J. Biol. Macromol. 2020, 155, 1296–1306. [Google Scholar] [CrossRef]
- Wu, C.; Sun, J.; Zheng, P.; Kang, X.; Chen, M.; Li, Y.; Ge, Y.; Hu, Y.; Pang, J. Preparation of an intelligent film based on chitosan/oxidized chitin nanocrystals incorporating black rice bran anthocyanins for seafood spoilage monitoring. Carbohydr. Polym. 2019, 222. [Google Scholar] [CrossRef]
- Apprich, S.; Tirpanalan, T.; Hell, J.; Reisinger, M.; Böhmdorfer, S.; Siebenhandl-Ehn, S.; Novalin, S.; Kneifel, W. Wheat bran-based biorefinery 2: Valorization of products. LWT—Food Sci. Technol. 2014, 56, 222–231. [Google Scholar] [CrossRef]
- Serna Saldivar, S.O. CEREALS | Dietary Importance. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Caballero, B., Ed.; Academic Press: Oxford, UK, 2003; pp. 1027–1033. [Google Scholar] [CrossRef]
- Balandran-Quintana, R.R. Recovery of proteins from cereal processing by-products. In Sustainable Recovery and Reutilisation of Cereal Processing By-products; Galanakis, C.M., Ed.; Woodhead Publishing: Cambridge, UK, 2018; pp. 125–158. [Google Scholar]
- Cheetangdee, N.; Benjakul, S. Antioxidant activities of rice bran protein hydrolysates in bulk oil and oil-in-water emulsion. J. Sci. Food Agric. 2015, 95, 1461–1468. [Google Scholar] [CrossRef]
- Satterlee, L.D. Utilization of proteins from biomass byproducts. Ind. Eng. Chem. Prod. Res. Dev. 1984, 23, 278–283. [Google Scholar] [CrossRef]
- Arte, E.; Huang, X.; Nordlund, E.; Katina, K. Biochemical characterization and technofunctional properties of bioprocessed wheat bran protein isolates. Food Chem. 2019, 289, 103–111. [Google Scholar] [CrossRef] [Green Version]
- Lorite, G.S.; Rocha, J.M.; Miilumäki, N.; Saavalainen, P.; Selkälä, T.; Morales-Cid, G.; Gonçalves, M.P.; Pongrácz, E.; Rocha, C.M.R.; Toth, G. Evaluation of physicochemical/microbial properties and life cycle assessment (LCA) of PLA-based nanocomposite active packaging. LWT—Food Sci. Technol. 2016, 75, 305–315. [Google Scholar] [CrossRef] [Green Version]
- Zinoviadou, K.G.; Gougouli, M.; Biliaderis, C.G. Innovative Biobased Materials for Packaging Sustainability. In Innovation Strategies in the Food Industry: Tools for Implementation; Academic Press: Cambridge, MA, USA, 2016; pp. 167–189. [Google Scholar] [CrossRef]
- Jonoobi, M.; Oladi, R.; Davoudpour, Y.; Oksman, K.; Dufresne, A.; Hamzeh, Y.; Davoodi, R. Different preparation methods and properties of nanostructured cellulose from various natural resources and residues: A review. Cellulose 2015, 22, 935–969. [Google Scholar] [CrossRef]
- Nikkilä, M. Cereal Waste Valorization through Development of Functional Key Fibres to Innovate in Fibre Packaging Materials. 2020. Available online: http://cordis.europa.eu/project/rcn/110025_en.html (accessed on 30 June 2020).
- Fritsch, C.; Staebler, A.; Happel, A.; Márquez, M.A.C.; Aguiló-Aguayo, I.; Abadias, M.; Gallur, M.; Cigognini, I.M.; Montanari, A.; López, M.J.; et al. Processing, valorization and application of bio-waste derived compounds from potato, tomato, olive and cereals: A review. Sustainability 2017, 9. [Google Scholar] [CrossRef] [Green Version]
- Peelman, N.; Ragaert, P.; De Meulenaer, B.; Adons, D.; Peeters, R.; Cardon, L.; Van Impe, F.; Devlieghere, F. Application of bioplastics for food packaging. Trends Food Sci. Technol. 2013, 32, 128–141. [Google Scholar] [CrossRef] [Green Version]
- Jamshidian, M.; Tehrany, E.A.; Imran, M.; Jacquot, M.; Desobry, S. Poly-Lactic Acid: Production, applications, nanocomposites, and release studies. Compr. Rev. Food Sci. Food Saf. 2010, 9, 552–571. [Google Scholar] [CrossRef]
- Djukić-Vuković, A.; Mladenović, D.; Radosavljević, M.; Kocić-Tanackov, S.; Pejin, J.; Mojović, L. Wastes from bioethanol and beer productions as substrates for l(+) lactic acid production—A comparative study. Waste Manag. 2016, 48, 478–482. [Google Scholar] [CrossRef]
- Djukić-Vuković, A.P.; Mojović, L.V.; Semenčenko, V.V.; Radosavljević, M.M.; Pejin, J.D.; Kocić-Tanackov, S.D. Effective valorisation of distillery stillage by integrated production of lactic acid and high quality feed. Food Res. Int. 2015, 73, 75–80. [Google Scholar] [CrossRef]
- Bilo, F.; Pandini, S.; Sartore, L.; Depero, L.E.; Gargiulo, G.; Bonassi, A.; Federici, S.; Bontempi, E. A sustainable bioplastic obtained from rice straw. J. Clean. Prod. 2018, 200, 357–368. [Google Scholar] [CrossRef]
- Souza Filho, P.F.; Zamani, A.; Ferreira, J.A. Valorization of wheat byproducts for the co-production of packaging material and enzymes. Energies 2020, 13. [Google Scholar] [CrossRef] [Green Version]
- Adebiyi, A.P.; Adebiyi, A.O.; Jin, D.H.; Ogawa, T.; Muramoto, K. Rice bran protein-based edible films. Int. J. Food Sci. Technol. 2008, 43, 476–483. [Google Scholar] [CrossRef]
- Gnanasambandam, R.; Hettiarachchy, N.S.; Coleman, M. Mechanical and barrier properties of rice bran films. J. Food Sci. 1997, 62, 395–398. [Google Scholar] [CrossRef]
- Schmidt, C.G.; Cerqueira, M.A.; Vicente, A.A.; Teixeira, J.A.; Furlong, E.B. Rice bran protein-based films enriched by phenolic extract of fermented rice bran and montmorillonite clay. CyTA—J. Food 2015, 13, 204–212. [Google Scholar] [CrossRef] [Green Version]
- Wang, N.; Saleh, A.S.M.; Gao, Y.; Wang, P.; Duan, Y.; Xiao, Z. Effect of protein aggregates on properties and structure of rice bran protein-based film at different pH. J. Food Sci. Technol. 2019, 56, 5116–5127. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.J.; Cerqueira, M.A.; Ruiz, H.A.; Fougnies, C.; Richel, A.; Vicente, A.A.; Teixeira, J.A.; Aguedo, M. Use of wheat bran arabinoxylans in chitosan-based films: Effect on physicochemical properties. Ind. Crop. Prod. 2015, 66, 305. [Google Scholar] [CrossRef] [Green Version]
- Anderson, C.; Simsek, S. Mechanical profiles and topographical properties of films made from alkaline extracted arabinoxylans from wheat bran, maize bran, or dried distillers grain. Food Hydrocoll. 2018. [Google Scholar] [CrossRef]
- Anderson, C.; Simsek, S. How do arabinoxylan films interact withwater and soil? Foods 2019, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aboukhalaf, A.; El Amraoui, B.; Tabatou, M.; Rocha, J.M.; Belahsen, R. Screening of the antimicrobial activity of some extracts of edible wild plants in Morocco. J. Funct. Food Health Dis. 2020, 6, 265–273. [Google Scholar] [CrossRef]
- Ajmal, M.; Butt, M.S.; Sharif, K.; Nasir, M.; Nadeem, M.T. Preparation of fiber and mineral enriched pan bread by using defatted rice bran. Int. J. Food Prop. 2006, 9, 623–636. [Google Scholar] [CrossRef]
- Sharif, M.K.; Butt, M.S.; Anjum, F.M.; Nawaz, H. Preparation of fiber and mineral enriched defatted rice bran supplemented cookies. Pak. J. Nutr. 2009, 8, 571–577. [Google Scholar] [CrossRef] [Green Version]
- Makowska, A.; Polcyn, A.; Chudy, S.; Michniewicz, J. Application of oat, wheat and rye bran to modify nutritional properties, physical and sensory characteristics of extruded corn snacks. Acta Sci. Pol. Technol. Aliment. 2015, 14, 375–386. [Google Scholar] [CrossRef] [Green Version]
- Tuncel, N.B.; Yilmaz, N.; Kocabiyik, H.; Uygur, A. The effect of infrared stabilized rice bran substitution on physicochemical and sensory properties of pan breads: Part, I. J. Cereal Sci. 2014, 59, 155–161. [Google Scholar] [CrossRef]
- Levent, H.; Koyuncu, M.; Bilgiçli, N.; Adıgüzel, E.; Dedeoğlu, M. Improvement of chemical properties of noodle and pasta using dephytinized cereal brans. LWT 2020, 128. [Google Scholar] [CrossRef]
- Pontonio, E.; Dingeo, C.; Gobbetti, M.; Rizzello, C.G. Maize milling by-products: From food wastes to functional ingredients through lactic acid bacteria fermentation. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santala, O.; Kiran, A.; Sozer, N.; Poutanen, K.; Nordlund, E. Enzymatic modification and particle size reduction of wheat bran improves the mechanical properties and structure of bran-enriched expanded extrudates. J. Cereal Sci. 2014, 60, 448–456. [Google Scholar] [CrossRef]
- Aktas-Akyildiz, E.; Masatcioglu, M.T.; Köksel, H. Effect of extrusion treatment on enzymatic hydrolysis of wheat bran. J. Cereal Sci. 2020, 93, 102941. [Google Scholar] [CrossRef]
- Wen, Y.; Niu, M.; Zhang, B.; Zhao, S.; Xiong, S. Structural characteristics and functional properties of rice bran dietary fiber modified by enzymatic and enzyme-micronization treatments. LWT 2017, 75, 344–351. [Google Scholar] [CrossRef]
- Călinoiu, L.F.; Cătoi, A.F.; Vodnar, D.C. Solid-State Yeast Fermented Wheat and Oat Bran as A Route for Delivery of Antioxidants. Antioxidants 2019, 8. [Google Scholar] [CrossRef] [Green Version]
- Irakli, M.; Mygdalia, A.; Chatzopoulou, P.; Katsantonis, D. Impact of the combination of sourdough fermentation and hop extract addition on baking properties, antioxidant capacity and phenolics bioaccessibility of rice bran-enhanced bread. Food Chem. 2019, 285, 231–239. [Google Scholar] [CrossRef]
- Bolarinwa, I.F.; Hanis-Syazwani, M.G.; Muhammad, K. Optimisation of important processing conditions for rice bran sourdough fermentation using Lactobacillus plantarum. Foods Raw Mater. 2019, 7, 131–142. [Google Scholar] [CrossRef]
- Kaur, R.; Riar, C.S. Sensory, rheological and chemical characteristics during storage of set type full fat yoghurt fortified with barley β-glucan. J. Food Sci. Technol. 2020, 57, 41–51. [Google Scholar] [CrossRef]
- Karp, S.; Wyrwisz, J.; Kurek, M.A. Comparative analysis of the physical properties of o/w emulsions stabilised by cereal β-glucan and other stabilisers. Int. J. Biol. Macromol. 2019, 132, 236–243. [Google Scholar] [CrossRef]
- Lazaridou, A.; Duta, D.; Papageorgiou, M.; Belc, N.; Biliaderis, C.G. Effects of hydrocolloids on dough rheology and bread quality parameters in gluten-free formulations. J. Food Eng. 2007, 79, 1033–1047. [Google Scholar] [CrossRef]
- Skendi, A.; Papageorgiou, M.; Biliaderis, C.G. Effect of barley β-glucan molecular size and level on wheat dough rheological properties. J. Food Eng. 2009, 91, 594–601. [Google Scholar] [CrossRef]
- Skendi, A.; Biliaderis, C.G.; Papageorgiou, M.; Izydorczyk, M.S. Effects of two barley β-glucan isolates on wheat flour dough and bread properties. Food Chem. 2010, 119, 1159–1167. [Google Scholar] [CrossRef]
- Skendi, A.; Papageorgiou, M.; Biliaderis, C.G. Influence of water and barley β-glucan addition on wheat dough viscoelasticity. Food Res. Int. 2010, 43, 57–65. [Google Scholar] [CrossRef]
- Ames, N.; Storsley, J.; Tosh, S. Effects of processing on physicochemical properties and efficacy of β-glucan from oat and barley. Cereal Foods World 2015, 60, 4–8. [Google Scholar] [CrossRef]
- Kaur, R.; Sharma, M.; Ji, D.; Xu, M.; Agyei, D. Structural features, modification, and functionalities of beta-glucan. Fibers 2020, 8. [Google Scholar] [CrossRef] [Green Version]
- Supawong, S.; Thawornchinsombut, S.; Iuchi, Y.; Park, J.W. Rice bran hydrolysates reduce browning reaction and function as endogenous antioxidant enzymes in wild type-mouse embryo fibroblast (WT-MEF) cells. In Proceedings of the Institute of Food Technologists (IFT) conference 2017, Las Vegas, NV, USA, 25–28 June 2017. [Google Scholar]
- Kartikawati, M.; Purnomo, H. Improving meatball quality using different varieties of rice bran as natural antioxidant. Food Res. 2019, 3, 79–85. [Google Scholar] [CrossRef]
- Wolfer, T.L.; Acevedo, N.C.; Prusa, K.J.; Sebranek, J.G.; Tarte, R. Replacement of pork fat in frankfurter-type sausages by soybean oil oleogels structured with rice bran wax. Meat Sci. 2018, 145, 352–362. [Google Scholar] [CrossRef]
- Al-Mansour, A.; Chow, C.L.; Feo, L.; Penna, R.; Lau, D. Green concrete: By-products utilization and advanced approaches. Sustainability 2019, 11, 5145. [Google Scholar] [CrossRef] [Green Version]
| Compound | Techniques | References |
|---|---|---|
| Arabinoxylans | Fermentation | [102,103,104] |
| Ultrasonic/microwave | [105,106] | |
| Enzymatic treatment | [107,108,109,110] | |
| Milling fractions | [111,112,113] | |
| Alkaline treatment | [114,115,116,117] | |
| Nixtamalization | [118] | |
| Starch removal | [119] | |
| Extrusion | [80,86,89,120] | |
| Subcritical water treatment | [119,121] | |
| β-glucans | Ultrasound | [122] |
| Roller-milling/pearling | [123] | |
| Milling | [124,125,126] | |
| Fermentation | [127,128] | |
| Enzymatic treatment | [129,130] | |
| Extrusion | [131,132,133,134] | |
| Subcritical water treatment | [119,135,136] |
| Extraction Method | Conditions | Cereal By-Product | Reference |
|---|---|---|---|
| Supercritical carbon dioxide | Pressure (15–35 MPa), temperature (40–60 °C) and CO2 + ethanol (0–60% ethanol concentration, v/v) | Brewer’s spent grain | [139] |
| Temperature (40 and 60 °C), pressure (30 and 40 MPa) and ethanol (0, 5 and 10%) | Rice bran | [140] | |
| Green method using glycerol | Temperature (40–70 °C), concentration of glycerol (10–70%, v/v) and liquid-to-solid ratio (10–40 mL g−1) | Rice bran | [141] |
| Solvent extraction | Absolute methanol, 80% methanol, absolute ethanol, 80% ethanol, and 50% acetone | Wheat bran and barley husk | [142] |
| Thermal processing coupled with ultrasound extraction | Temperature 80 °C for 10 min | Wheat and oat bran | [143] |
| Pressurized solutions | Concentrations of ethanol in water (0–80%), temperature (130–160 °C) and extraction times (20–60 min) | Wheat bran | [144] |
| Ultrasonic-assisted extraction (UAE) | Ethanol and limonene, temperature 51 and 60 °C respectively | Rice berry bran | [145] |
| Temperature (30–60 °C), pH (2–4), solvent concentration 920–60%), time (10–60 min) | Black and purple rice bran | [146] | |
| Time (10–30 min), ethanol concentration (30–70%), solvent to solids (mL g−1) | Red sorghum bran | [147] | |
| Temperature (20–90 °C), time (1–25 min) | Defatted oat (Avena sativa L.) bran | [122] | |
| Steam explosion-assisted extraction | Temperature 215 °C for 120 s | Wheat bran | [148] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skendi, A.; Zinoviadou, K.G.; Papageorgiou, M.; Rocha, J.M. Advances on the Valorisation and Functionalization of By-Products and Wastes from Cereal-Based Processing Industry. Foods 2020, 9, 1243. https://doi.org/10.3390/foods9091243
Skendi A, Zinoviadou KG, Papageorgiou M, Rocha JM. Advances on the Valorisation and Functionalization of By-Products and Wastes from Cereal-Based Processing Industry. Foods. 2020; 9(9):1243. https://doi.org/10.3390/foods9091243
Chicago/Turabian StyleSkendi, Adriana, Kyriaki G. Zinoviadou, Maria Papageorgiou, and João M. Rocha. 2020. "Advances on the Valorisation and Functionalization of By-Products and Wastes from Cereal-Based Processing Industry" Foods 9, no. 9: 1243. https://doi.org/10.3390/foods9091243
APA StyleSkendi, A., Zinoviadou, K. G., Papageorgiou, M., & Rocha, J. M. (2020). Advances on the Valorisation and Functionalization of By-Products and Wastes from Cereal-Based Processing Industry. Foods, 9(9), 1243. https://doi.org/10.3390/foods9091243








