Is There Scope for a Novel Mycelium Category of Proteins alongside Animals and Plants?
Abstract
1. Introduction
2. Methods
- (1)
- Did not focus on human, adult populations;
- (2)
- Were not a systematic review or meta-analysis publication;
- (3)
- Focused on one specific global population/region;
- (4)
- Did not clearly specify the protein of focus;
- (5)
- Had research outcomes unrelated to health.
3. Animal-Derived Proteins
4. Plant-Derived Proteins
5. Mycelium/Fungal-Derived Proteins
5.1. Mushrooms
5.2. Mycoprotein
5.3. Others
6. Discussion and Future Directions
| Carbon (kg CO2e kg−1) | Land (ha kg−1) | Green Water (L kg−1) | Blue Water (L kg−1) | Grey Water (L kg−1) | |
|---|---|---|---|---|---|
| Animal-Based | |||||
| Beef, general | 16.2 | 0.0068 | 13,921 | 752 | 1016 |
| Beef, mixed | 26.7 | 0.0035 | 15,500 | 250 | 4000 |
| Beef, grazed | 121 | 0.0049 | 16,500 | 300 | 5000 |
| Pork | 8.29 | 0.0012 | 5070 | 416 | 509 |
| Plant-Based | |||||
| Soy | 1.02 | 0.0014 | 1855 | 240 | 573 |
| Fungal | |||||
| Mycoprotein | 0.8 | 0.00018 | 539 | 35 | 202 |
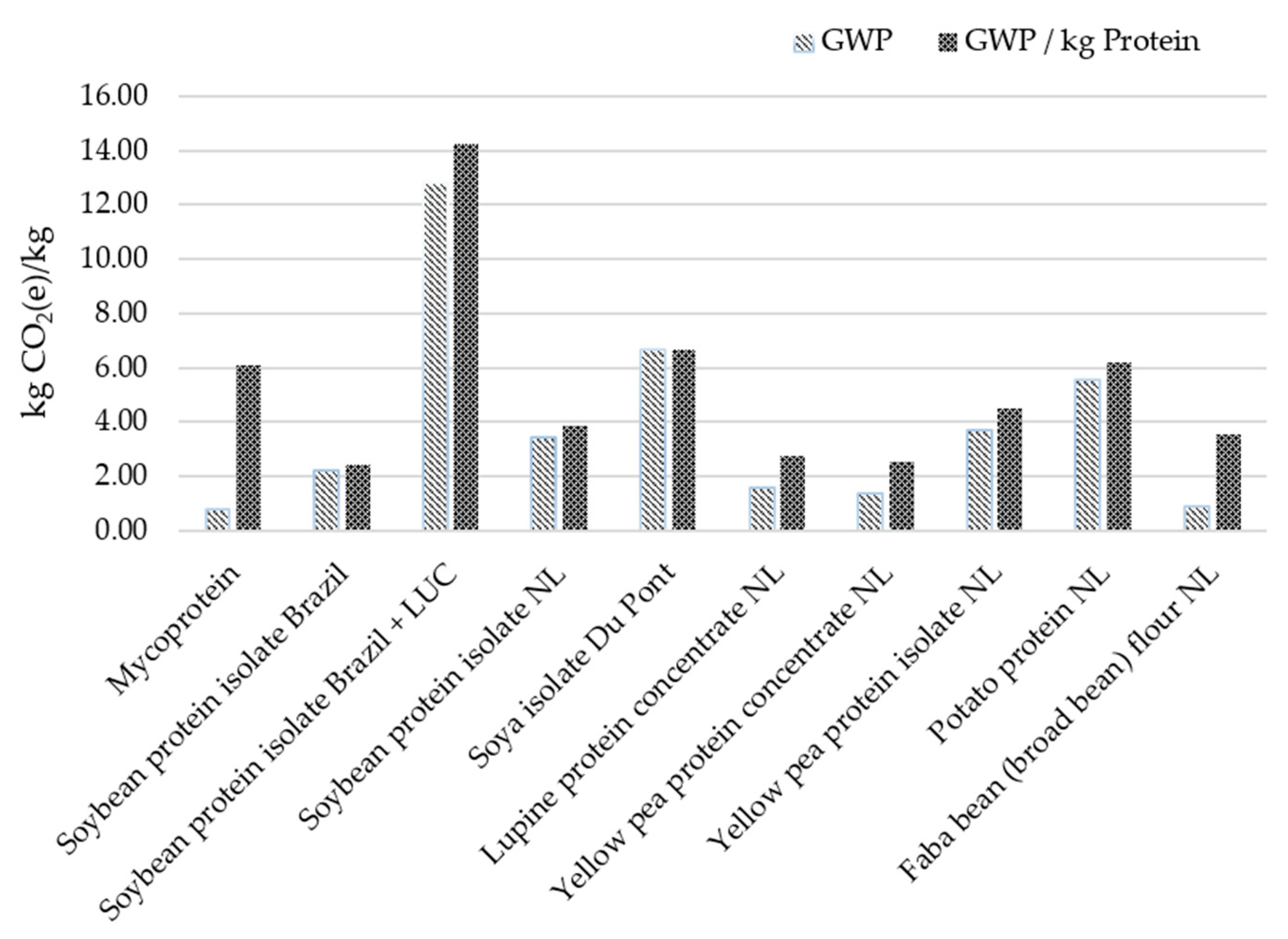
7. Conclusions
Funding
Conflicts of Interest
References
- Scovronick, N.; Budolfson, M.B.; Dennig, F.; Fleurbaey, M.; Siebert, A.; Socolow, R.H.; Spears, D.; Wagner, F. Impact of population growth and population ethics on climate change mitigation policy. Proc. Natl. Acad Sci. USA 2017, 114, 12338–12343. [Google Scholar] [CrossRef]
- Willett, W.; Rockstrom, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A.; et al. Food in the Anthropocene: The EAT-Lancet Commission on healthy diets from sustainable food systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]
- Herforth, A.; Arimond, M.; Alvarez-Sanchez, C.; Coates, J.; Christianson, K.; Muehlhoff, E. A Global Review of Food-Based Dietary Guidelines. Adv. Nutr. 2019, 10, 590–605. [Google Scholar] [CrossRef]
- Jo Feeney, M.; Miller, A.M.; Roupas, P. Mushrooms-Biologically Distinct and Nutritionally Unique: Exploring a “Third Food Kingdom”. Nutr. Today 2014, 49, 301–307. [Google Scholar] [CrossRef]
- Bayer, E. The Mycelium Revolution Is upon Us. 2019. Available online: https://blogs.scientificamerican.com/observations/the-mycelium-revolution-is-upon-us/ (accessed on 18 August 2020).
- Giovannetti, M.E.A. At the Root of the Wood Wide Web. Plant. Signal. Behav. 2006, 1, 1–5. [Google Scholar] [CrossRef]
- Coelho, M.O.C.; Monteyne, A.; Dunlop, M.V.; Harris, H.C.; Morrison, D.J.; Stephens, F.B.; Wall, B.T. Mycoprotein as a possible alternative source of dietary protein to support muscle and metabolic health. Nutr. Res. Rev. 2019, 78, 486–497. [Google Scholar] [CrossRef]
- Derbyshire, E.J.; Ayoob, K.T. Mycoprotein Nutritional and Health Properties. Nutr. Today Clin. Nutr. 2019, 54, 1–9. [Google Scholar]
- Finnigan, T. Mycoprotein: Origins, production and properties. In Handbook of Food Proteins; Phillips, G.O., William, P.A., Eds.; Woodhead Publishing: London, UK, 2011; pp. 335–352. [Google Scholar]
- The Carbon Trust. The Carbon Trust. Product Carbon Footprint and Reduction Certification Letter CERT-12751. Unpublished work. 2019. [Google Scholar]
- Ismail, I.; Hwang, Y.H.; Joo, S.T. Meat analog as future food: A review. J. Anim. Sci. Technol. 2020, 62, 111–120. [Google Scholar] [CrossRef]
- Malav, O.P.; Talukder, S.; Gokulakrishnan, P.; Chand, S. Meat analog: A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 1241–1245. [Google Scholar] [CrossRef]
- Kumar, P.; Chatli, M.K.; Mehta, N.; Singh, P.; Malav, O.P.; Verma, A.K. Meat analogues: Health promising sustainable meat substitutes. Crit. Rev. Food Sci. Nutr. 2017, 57, 923–932. [Google Scholar] [CrossRef]
- Guasch-Ferre, M.; Satija, A.; Blondin, S.A.; Janiszewski, M.; Emlen, E.; O’Connor, L.E.; Campbell, W.W.; Hu, F.B.; Willett, W.C.; Stampfer, M.J. Meta-Analysis of Randomized Controlled Trials of Red Meat Consumption in Comparison With Various Comparison Diets on Cardiovascular Risk Factors. Circulation 2019, 139, 1828–1845. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Li, Y.; Wang, C.; Mao, Z.; Zhou, W.; Zhang, L.; Yang, X.; Cui, S.; Li, L. Dietary Protein Consumption and the Risk of Type 2 Diabetes: ADose-Response Meta-Analysis of Prospective Studies. Nutrients 2019, 11, 2783. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Hyeon, J.; Lee, S.A.; Kwon, S.O.; Lee, H.; Keum, N.; Lee, J.K.; Park, S.M. Role of Total, Red, Processed, and White Meat Consumption in Stroke Incidence and Mortality: A Systematic Review and Meta-Analysis of Prospective Cohort Studies. J. Am. Heart Assoc. 2017, 6. [Google Scholar] [CrossRef]
- Tian, S.; Xu, Q.; Jiang, R.; Han, T.; Sun, C.; Na, L. Dietary Protein Consumption and the Risk of Type 2 Diabetes: A Systematic Review and Meta-Analysis of Cohort Studies. Nutrients 2017, 9, 982. [Google Scholar] [CrossRef]
- Naghshi, S.; Sadeghi, O.; Willett, W.C.; Esmaillzadeh, A. Dietary intake of total, animal, and plant proteins and risk of all cause, cardiovascular, and cancer mortality: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2020, 370, m2412. [Google Scholar] [CrossRef]
- Zhao, H.; Song, A.; Zheng, C.; Wang, M.; Song, G. Effects of plant protein and animal protein on lipid profile, body weight and body mass index on patients with hypercholesterolemia: A systematic review and meta-analysis. Acta Diabetol. 2020. [Google Scholar] [CrossRef]
- Blanco Mejia, S.; Messina, M.; Li, S.S.; Viguiliouk, E.; Chiavaroli, L.; Khan, T.A.; Srichaikul, K.; Mirrahimi, A.; Sievenpiper, J.L.; Kris-Etherton, P.; et al. A Meta-Analysis of 46 Studies Identified by the FDA Demonstrates that Soy Protein Decreases Circulating LDL and Total Cholesterol Concentrations in Adults. J. Nutr. 2019, 149, 968–981. [Google Scholar] [CrossRef]
- Messina, M.; Lynch, H.; Dickinson, J.M.; Reed, K.E. No Difference between the Effects of Supplementing With Soy Protein Versus Animal Protein on Gains in Muscle Mass and Strength in Response to Resistance Exercise. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 674–685. [Google Scholar] [CrossRef]
- Shams-White, M.M.; Chung, M.; Fu, Z.; Insogna, K.L.; Karlsen, M.C.; LeBoff, M.S.; Shapses, S.A.; Sackey, J.; Shi, J.; Wallace, T.C.; et al. Animal versus plant protein and adult bone health: A systematic review and meta-analysis from the National Osteoporosis Foundation. PLoS ONE 2018, 13, e0192459. [Google Scholar] [CrossRef]
- Kou, T.; Wang, Q.; Cai, J.; Song, J.; Du, B.; Zhao, K.; Ma, Y.; Geng, B.; Zhang, Y.; Han, X.; et al. Effect of soybean protein on blood pressure in postmenopausal women: A meta-analysis of randomized controlled trials. Food Funct. 2017, 8, 2663–2671. [Google Scholar] [CrossRef]
- Li, S.S.; Blanco Mejia, S.; Lytvyn, L.; Stewart, S.E.; Viguiliouk, E.; Ha, V.; de Souza, R.J.; Leiter, L.A.; Kendall, C.W.C.; Jenkins, D.J.A.; et al. Effect of Plant Protein on Blood Lipids: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Am. Heart Assoc. 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Eichelmann, F.; Schwingshackl, L.; Fedirko, V.; Aleksandrova, K. Effect of plant-based diets on obesity-related inflammatory profiles: A systematic review and meta-analysis of intervention trials. Obes. Rev. 2016, 17, 1067–1079. [Google Scholar] [CrossRef] [PubMed]
- Viguiliouk, E.; Stewart, S.E.; Jayalath, V.H.; Ng, A.P.; Mirrahimi, A.; de Souza, R.J.; Hanley, A.J.; Bazinet, R.P.; Blanco Mejia, S.; Leiter, L.A.; et al. Effect of Replacing Animal Protein with Plant Protein on Glycemic Control in Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2015, 7, 9804–9824. [Google Scholar] [CrossRef]
- Cherta-Murillo, A.; Lett, A.M.; Frampton, J.; Chambers, E.S.; Finnigan, T.J.A.; Frost, G.S. Effects of mycoprotein on glycaemic control and energy intake in humans: A systematic review. Br. J. Nutr. 2020, 123, 1321–1332. [Google Scholar] [CrossRef]
- Dicks, L.; Ellinger, S. Effect of the Intake of Oyster Mushrooms (Pleurotus ostreatus) on Cardiometabolic Parameters-A Systematic Review of Clinical Trials. Nutrients 2020, 12, 1134. [Google Scholar] [CrossRef]
- Cashman, K.D.; Kiely, M.; Seamans, K.M.; Urbain, P. Effect of Ultraviolet Light-Exposed Mushrooms on Vitamin D Status: Liquid Chromatography-Tandem Mass Spectrometry Reanalysis of Biobanked Sera from a Randomized Controlled Trial and a Systematic Review plus Meta-Analysis. J. Nutr. 2016, 146, 565–575. [Google Scholar] [CrossRef]
- Fritz, H.; Kennedy, D.A.; Ishii, M.; Fergusson, D.; Fernandes, R.; Cooley, K.; Seely, D. Polysaccharide K and Coriolus versicolor extracts for lung cancer: A systematic review. Integr. Cancer Ther. 2015, 14, 201–211. [Google Scholar] [CrossRef]
- González, N.; Marques, M.; Nadal José, M.; Domingo, L. Meat consumption: Which are the current global risks? A review of recent (2010–2020) evidences. Food Res. Int. 2020, 137, 109341. [Google Scholar] [CrossRef]
- Basu, S. The transitional dynamics of caloric ecosystems: Changes in the food supply around the world. Crit. Public Health 2015, 25, 248–264. [Google Scholar] [CrossRef]
- Wolk, A. Potential health hazards of eating red meat. J. Intern. Med. 2017, 281, 106–122. [Google Scholar] [CrossRef]
- Domingo, J.L.; Nadal, M. Carcinogenicity of consumption of red meat and processed meat: A review of scientific news since the IARC decision. Food Chem. Toxicol. 2017, 105, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Flachowsky, G.; Meyer, U.; Sudekum, K.H. Land Use for Edible Protein of Animal Origin—A Review. Animals 2017, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Laskowski, W.; Gorska-Warsewicz, H.; Kulykovets, O. Meat, Meat Products and Seafood as Sources of Energy and Nutrients in the Average Polish Diet. Nutrients 2018, 10, 1412. [Google Scholar] [CrossRef] [PubMed]
- Derbyshire, E. Associations between Red Meat Intakes and the Micronutrient Intake and Status of UK Females: A Secondary Analysis of the UK National Diet and Nutrition Survey. Nutrients 2017, 9, 768. [Google Scholar] [CrossRef]
- Cuparencu, C.; Pratico, G.; Hemeryck, L.Y.; Sri Harsha, P.S.C.; Noerman, S.; Rombouts, C.; Xi, M.; Vanhaecke, L.; Hanhineva, K.; Brennan, L.; et al. Biomarkers of meat and seafood intake: An extensive literature review. Genes Nutr. 2019, 14, 35. [Google Scholar] [CrossRef]
- DEFRA/FSA. Meat Products: Sell Them Legally in England. 2017. Available online: https://www.gov.uk/guidance/meat-products-sell-them-legally-in-england (accessed on 18 August 2020).
- WHO. Q&A on the Carcinogenicity of the Consumption of Red Meat and Processed Meat. 2015. Available online: http://www.cancer.ie/content/qa-carcinogenicityconsumption-red-meat-and-processed-meat#sthash (accessed on 18 August 2020).
- O’Connor, L.E.; Gifford, C.L.; Woerner, D.R.; Sharp, J.L.; Belk, K.E.; Campbell, W.W. Dietary Meat Categories and Descriptions in Chronic Disease Research Are Substantively Different within and between Experimental and Observational Studies: A Systematic Review and Landscape Analysis. Adv. Nutr. 2020, 11, 41–51. [Google Scholar] [CrossRef]
- Wen, X.; Sun, S.; Li, L.; He, Q.; Tsai, F.S. Avian Influenza-Factors Affecting Consumers’ Purchase Intentions toward Poultry Products. Int. J. Environ. Res. Public Health 2019, 16, 4139. [Google Scholar] [CrossRef]
- Fung, T.T.; Hu, F.B. Plant-based diets: What should be on the plate? Am. J. Clin. Nutr. 2003, 78, 357–358. [Google Scholar] [CrossRef]
- EAT. Healthy Diets for Sustainable Food Systems: Food Planet Health; EAT-Lancet Commission: Stockholm, Sweden, 2019. [Google Scholar]
- Cifelli, C.J.; Houchins, J.A.; Demmer, E.; Fulgoni, V.L. Increasing Plant Based Foods or Dairy Foods Differentially Affects Nutrient Intakes: Dietary Scenarios Using NHANES 2007–2010. Nutrients 2016, 8, 422. [Google Scholar] [CrossRef]
- Chardigny, J.; Walrand, S. Plant protein for food: Opportunities and bottlenecks. Oilseeds Fats Crops Lipids 2016, 1–6. [Google Scholar] [CrossRef]
- Pihlanto, A.; Mattila, P.; Makinen, S.; Pajari, A.M. Bioactivities of alternative protein sources and their potential health benefits. Food Funct. 2017, 8, 3443–3458. [Google Scholar] [CrossRef] [PubMed]
- Michail, N. Germany Backs Proposal for Legal Definition of Vegetarian and Vegan Food. 2016. Available online: https://www.foodnavigator.com/Article/2016/06/01/Germany-backs-proposal-for-legal-definition-of-vegetarian-and-vegan-food (accessed on 18 August 2020).
- Michail, N. EU to Set Legal Definition of Vegetarian and Vegan Food. 2017. Available online: https://www.foodnavigator.com/Article/2017/11/03/EU-to-set-legal-definition-of-vegetarian-and-vegan-food (accessed on 18 August 2020).
- Carlile, M.J.; Watkinson, S.J. The fungi as a major group of organisms. In The Fungi; Academic Press: London, UK, 2015. [Google Scholar]
- Naranjo-Ortiz, M.A.; Gabaldon, T. Fungal evolution: Diversity, taxonomy and phylogeny of the Fungi. Biol. Rev. Camb. Philos Soc. 2019, 94, 2101–2137. [Google Scholar] [CrossRef] [PubMed]
- Denny, A. Mycoprotein and health. Br. Nutr. Found. Nutr. Bull. 2008, 33, 298–310. [Google Scholar] [CrossRef]
- FAOSTAT. Mushrooms and Truffles; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013; Available online: http://data.un.org/Data.aspx?d=FAO&f=itemCode:449 (accessed on 18 August 2020).
- Feeney, M.J.; Dwyer, J.; Hasler-Lewis, C.M.; Milner, J.A.; Noakes, M.; Rowe, S.; Wach, M.; Beelman, R.B.; Caldwell, J.; Cantorna, M.T.; et al. Mushrooms and Health Summit proceedings. J. Nutr. 2014, 144, 1128S–1136S. [Google Scholar] [CrossRef]
- Finnigan, T.J.A.; Wall, B.T.; Wilde, P.J.; Stephens, F.B.; Taylor, S.L.; Freedman, M.R. Mycoprotein: The Future of Nutritious Nonmeat Protein, a Symposium Review. Curr. Dev. Nutr. 2019, 3, nzz021. [Google Scholar] [CrossRef]
- Grocer. The Grocer. Britain’s Biggest Brands 2020: The Top 100. 2020. Available online: https://www.thegrocer.co.uk/britains-biggest-brands/britains-biggest-brands-2020-the-top-100/603257.article (accessed on 18 August 2020).
- De Mattos-Shipley, K.M.; Ford, K.L.; Alberti, F.; Banks, A.M.; Bailey, A.M.; Foster, G.D. The good, the bad and the tasty: The many roles of mushrooms. Stud. Mycol. 2016, 85, 125–157. [Google Scholar] [CrossRef]
- Valverde, M.E.; Hernandez-Perez, T.; Paredes-Lopez, O. Edible mushrooms: Improving human health and promoting quality life. Int. J. Microbiol. 2015, 2015, 376387. [Google Scholar] [CrossRef]
- Regana, H.; Lamb, A.; Wilkinson, D. The missing mushrooms: Searching for fungi in ancient human dietary analysis. J. Archaeol. Sci. 2016, 75, 139–143. [Google Scholar] [CrossRef]
- Rathore, H.; Prasad, S.; Sharma, S. Mushroom nutraceuticals for improved nutrition and better human health: A review. PharmaNutrition 2017, 5, 35–46. [Google Scholar] [CrossRef]
- Cardwell, G.; Bornman, J.F.; James, A.P.; Black, L.J. A Review of Mushrooms as a Potential Source of Dietary Vitamin D. Nutrients 2018, 10, 1498. [Google Scholar] [CrossRef]
- PHE. Public Health England. McCance and Widdowson’s Composition of Foods Integrated Dataset. 2019. Available online: https://www.gov.uk/government/publications/composition-of-foods-integrated-dataset-cofid (accessed on 18 August 2020).
- Shin, A.; Kim, J.; Lim, S.Y.; Kim, G.; Sung, M.K.; Lee, E.S.; Ro, J. Dietary mushroom intake and the risk of breast cancer based on hormone receptor status. Nutr. Cancer 2010, 62, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zou, L.; Chen, W.; Zhu, B.; Shen, N.; Ke, J.; Lou, J.; Song, R.; Zhong, R.; Miao, X. Dietary mushroom intake may reduce the risk of breast cancer: Evidence from a meta-analysis of observational studies. PLoS ONE 2014, 9, e93437. [Google Scholar] [CrossRef] [PubMed]
- Ba, D.; Ssentongo, P.; Beelman, R.B.; Gao, X.; Richie, J.P. Mushroom Consumption Is Associated with Low Risk of Cancer: A Systematic Review and Meta-Analysis of Observation Studies. Curr. Dev. Nutr. 2020, 4, 307. [Google Scholar] [CrossRef]
- Ganesan, K.; Xu, B. Anti-Obesity Effects of Medicinal and Edible Mushrooms. Molecules 2018, 23, 2880. [Google Scholar] [CrossRef] [PubMed]
- Grotto, D.; Camargo, I.F.; Kodaira, K.; Mazzei, L.G.; Castro, J.; Vieira, R.A.L.; Bergamaschi, C.C.; Lopes, L.C. Effect of mushrooms on obesity in animal models: Study protocol for a systematic review and meta-analysis. Syst. Rev. 2019, 8, 288. [Google Scholar] [CrossRef]
- Friedman, M. Mushroom Polysaccharides: Chemistry and Antiobesity, Antidiabetes, Anticancer, and Antibiotic Properties in Cells, Rodents, and Humans. Foods 2016, 5, 80. [Google Scholar] [CrossRef]
- Hess, J.; Wang, Q.; Gould, T.; Slavin, J. Impact of Agaricus bisporus Mushroom Consumption on Gut Health Markers in Healthy Adults. Nutrients 2018, 10, 1402. [Google Scholar] [CrossRef]
- EC. Efsa, regulation (ec) no 1924/2006 of the european parliament and of the council of 20 December 2006 on nutrition and health claims made on foods. Off. J. Eur. Union 2006, 1994, 2006. [Google Scholar]
- European Comminsion. Commission Directive 2008/100/EC of 28 October 2008 amending Council Directive 90/496/EEC on nutrition labelling for foodstuffs as regards recommended daily allowances, energy conversion factors and definitions. Off. J. Eur. Union 2008, 285, 9. [Google Scholar]
- Derbyshire, E. Could we be Overlooking a Potential Choline Crisis in the United Kingdom. BMJ Nutr. Prev. Health 2019, 1–4. [Google Scholar] [CrossRef]
- Edwards, D.G.; Cummings, J.H. The protein quality of mycoprotein. Proc. Nutr. Soc. 2010, 69. [Google Scholar] [CrossRef]
- Dunlop, M.V.; Kilroe, S.P.; Bowtell, J.L.; Finnigan, T.J.A.; Salmon, D.L.; Wall, B.T. Mycoprotein represents a bioavailable and insulinotropic non-animal-derived dietary protein source: A dose-response study. Br. J. Nutr. 2017, 118, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Monteyne, A.J.; Coelho, M.O.; Porter, C.; Abdelrahman, D.R.; Jameson, T.S.; Jackman, S.R.; Blackwell, J.R.; Finnigan, T.J.; Stephens, F.B.; Dirks, M.L.; et al. Mycoprotein ingestion stimulates protein synthesis rates to a greater extent than milk protein in rested and exercised skeletal muscle of healthy young men. Am. J. Clin. Nutr. 2020, 112, 318–333. [Google Scholar] [CrossRef]
- van Vliet, S.; Shy, E.L.; Abou Sawan, S.; Beals, J.W.; West, D.W.; Skinner, S.K.; Ulanov, A.V.; Li, Z.; Paluska, S.A.; Parsons, C.M.; et al. Consumption of whole eggs promotes greater stimulation of postexercise muscle protein synthesis than consumption of isonitrogenous amounts of egg whites in young men. Am. J. Clin. Nutr. 2017, 106, 1401–1412. [Google Scholar] [CrossRef]
- Bottin, J.H.; Swann, J.R.; Cropp, E.; Chambers, E.S.; Ford, H.E.; Ghatei, M.A.; Frost, G.S. Mycoprotein reduces energy intake and postprandial insulin release without altering glucagon-like peptide-1 and peptide tyrosine-tyrosine concentrations in healthy overweight and obese adults: A randomised-controlled trial. Br. J. Nutr. 2016, 116, 360–374. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, W.H.; Walton, J.; Leeds, A.R. Acute effects of mycoprotein on subsequent energy intake and appetite variables. Am. J. Clin. Nutr. 1993, 58, 507–512. [Google Scholar] [CrossRef]
- Williamson, D.A.; Geiselman, P.J.; Lovejoy, J.; Greenway, F.; Volaufova, J.; Martin, C.K.; Arnett, C.; Ortego, L. Effects of consuming mycoprotein, tofu or chicken upon subsequent eating behaviour, hunger and safety. Appetite 2006, 46, 41–48. [Google Scholar] [CrossRef]
- Burley, V.; Paul, A.; Blundell, J.E. Influence of a high-fibre food (myco-protein) on appetite: Effects on satiation (within meals) and satiety (following meals). Eur. J. Clin. Nutr. 1993, 47, 409–418. [Google Scholar]
- Ruxton, C.; McMilan, B. The impact of mycoprotein on blood cholesterol levels: A pilot study. Br. Food J. 2010, 112, 109. [Google Scholar] [CrossRef]
- Turnbull, W.H.; Leeds, A.R.; Edwards, G.D. Effect of mycoprotein on blood lipids. Am. J. Clin. Nutr. 1990, 52, 646–650. [Google Scholar] [CrossRef]
- Turnbull, W.H.; Leeds, A.R.; Edwards, D.G. Mycoprotein reduces blood lipids in free-living subjects. Am. J. Clin. Nutr. 1992, 55, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T. Effect of mycoprotein on serum lipids and apolipoproteins in normolipidemic and hypercholesterolemic subjects. Atherosclerosis 1994, 109, 76. [Google Scholar] [CrossRef]
- Nakamura, H.; Ishikawa, T.; Akanuma, M.; Nishiwaki, M.; Yamashita, T.; Tomiyasu, K.; Yoshida, H.; Nishio, E.; Hosoai, K.; Shiga, H.; et al. Effect of mycoprotein intake on serum lipids of healthy subjects. Prog. Med. 1994, 14, 1972–1976. [Google Scholar]
- Homma, Y.; Nakamura, H.; Kumagai, Y.; Ryuzo, A.; Saito, Y.; Ishikawa, T.; Takada, K.; Yamagami, H.; Kikuchi, H.; Inadera, H. Effects of eight week ingestion of mycoprotein on plasma levels of lipids and Apo (Lipo) proteins. Prog. Med. 1995, 15, 183–195. [Google Scholar]
- Coelho, M.O.C. Daily mycoprotein consumption for one week does not affect insulin sensitivity or glycaemic control but strongly modulates the plasma lipoma. Br. J. Nutr. 2020, submitted. [Google Scholar]
- Bottin, J.H.; Cropp, E.; Finnigan, T.J.A.; Hogban, A. Mycoprotein reduces energy intake and improves insulin sensitivity compared to chicken. Obes. Facts 2012, 5, 55–79. [Google Scholar]
- Turnbull, W.H.; Ward, T. Mycoprotein reduces glycemia and insulinemia when taken with an oral-glucose-tolerance test. Am. J. Clin. Nutr. 1995, 61, 135–140. [Google Scholar] [CrossRef]
- Harris, H.C.; Edwards, C.A.; Morrison, D.J. Short Chain Fatty Acid Production from Mycoprotein and Mycoprotein Fibre in an In Vitro Fermentation Model. Nutrients 2019, 11, 800. [Google Scholar] [CrossRef]
- Kalra, R.; Conlan, X.A.; Goel, M. Fungi as a Potential Source of Pigments: Harnessing Filamentous Fungi. Front. Chem. 2020, 8, 369. [Google Scholar] [CrossRef]
- Nangul, A.; Bhatia, R. Microorganisms: A marvellous source of single cell proteins. J. Microbiol. Biotechnol. Food Sci. 2013, 3, 15–18. [Google Scholar]
- Souza Filho, P.F.; Nair, R.B.; Andersson, D.; Lennartsson, P.R.; Taherzadeh, M.J. Vegan-mycoprotein concentrate from pea-processing industry byproduct using edible filamentous fungi. Fungal. Biol. Biotechnol. 2018, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Cena, H.; Calder, P.C. Defining a Healthy Diet: Evidence for the Role of Contemporary Dietary Patterns in Health and Disease. Nutrients 2020, 12, 334. [Google Scholar] [CrossRef] [PubMed]
- AGDoH. The Australian Dietary Guidelines; Australian Government, Department of Health: Canberra, Australia, 2019.
- Derbyshire, E. Protein Guidance—Is It Time for an Update? Dietetics Today 2020, 22–23. [Google Scholar]
- Kalaras, M.D.; Richie, J.P.; Calcagnotto, A.; Beelman, R.B. Mushrooms: A rich source of the antioxidants ergothioneine and glutathione. Food Chem. 2017, 233, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Martel, J.; Ojcius, D.M.; Ko, Y.F.; Chang, C.J.; Young, J.D. Antiaging effects of bioactive molecules isolated from plants and fungi. Med. Res. Rev. 2019, 39, 1515–1552. [Google Scholar] [CrossRef] [PubMed]
- Chin, V.K.; Yong, V.C.; Chong, P.P.; Amin Nordin, S.; Basir, R.; Abdullah, M. Mycobiome in the Gut: A Multiperspective Review. Mediat. Inflamm. 2020, 2020, 9560684. [Google Scholar] [CrossRef]
- Fasolin, L.H.; Pereira, R.N.; Pinheiro, A.C.; Martins, J.T.; Andrade, C.C.P.; Ramos, O.L.; Vicente, A.A. Emergent food proteins—Towards sustainability, health and innovation. Food Res. Int. 2019, 125, 108586. [Google Scholar] [CrossRef]
- Derbyshire, E.J. Flexitarian Diets and Health: A Review of the Evidence-Based Literature. Front. Nutr. 2016, 3, 55. [Google Scholar] [CrossRef]
- Saint-Eve, A.; Granda, P.; Legay, G.; Cuvelier, G.; Delarue, J. Consumer acceptance and sensory drivers of liking for high plant protein snacks. J. Sci. Food Agric. 2019, 99, 3983–3991. [Google Scholar] [CrossRef]
- The Carbon Trust. Quorn Footprint Comparison Report. 2018. Available online: https://www.quorn.co.uk/files/content/Carbon-Trust-Comparison%20Report-2018.pdf (accessed on 18 August 2020).
- CT. The Carbon Trust. Quorn-Product Carbon Footprinting and Labeling. 2019. Available online: https://www.carbontrust.com/our-clients/q/quorn-product-carbon-footprinting-and-labeling/ (accessed on 18 August 2020).
- Finnigan, T.; Abbott, C.; Thornton, H.; Needham, L. Mycoprotein Comparative Land Use and GWP Q&A. Unpublished work. 2020. [Google Scholar]
| Data Collation | Main Findings | |
|---|---|---|
| Animal-Derived Proteins | ||
| Guasch-Ferré et al. (2019) [14] | 36 studies | Substituting red meat with high-quality plant protein sources (but not fish or low-quality carbohydrates) resulted in favorable changes to blood lipids and lipoproteins. |
| Fan et al. (2019) [15] | 12 studies | T2D risk ↑ with ↑ consumption of total protein and animal protein, red meat, processed meat, milk, and eggs. Plant protein and yogurt had an inverse relationship. |
| Kim et al. (2017) [16] | 15 studies | Inter-relations between meat intake and stroke risk differ by type of meat. Red and processed meats replaced with white meat may be considered for stroke prevention. |
| Tian et al. (2017) [17] | 11 studies | Red meat and processed meat are risk factors for T2D whilst soy and dairy products appear protective. Egg and fish intake were not associated with decreased T2D risk. |
| Plant-Derived Proteins | ||
| Naghshi et al. (2020) [18] | 32 studies | Intake of plant-protein was associated with a lower risk of all-cause and cardiovascular disease mortality. |
| Zhao et al. (2020) [19] | 32 studies | Plant-protein could improve lipid profile in patients with hypercholesterolemia. |
| Blanco Mejia et al. (2019) [20] | 46 trials | Soy protein significantly reduced LDL cholesterol by approximately 3–4% in adults. |
| Messina et al. (2018) [21] | 9 studies | Soy protein supplementation produced similar gains in strength and LBM in response to RET as whey protein. |
| Shams-White et al. (2018) [22] | 7 trials | Soy protein consumption versus animal protein was not more advantageous at improving markers of bone health. |
| Kou et al. (2017) [23] | 12 trials | The ingestion of ≥25 g soy protein per day had BP-lowering effects, possibly due to isoflavones. |
| Li et al. (2017) [24] | 112 trials | Plant protein in substitution for animal protein ↓ LDL cholesterol by 0.16 mmol/L, non-high-density lipoprotein cholesterol by 0.18 mmol/L and apolipoprotein B by 0.05 g/L. |
| Eichelmann et al. (2016) [25] | 29 trials | Plant-based diets were associated with improved markers of inflammation, including CRP and IL-6. |
| Viguiliouk et al. (2015) [26] | 13 trials | Replacing animal with plant protein leas to modest improvements in glycemic control in individuals with diabetes. |
| Fungal-Derived Proteins | ||
| Cherta-Murillo et al. (2020) [27] | Five trials | Results showed that mycoprotein reduced insulin levels. Acute mycoprotein intake also decreased energy intake at an ad libitum meal and post-24 h in healthy lean, overweight and obese humans. |
| Dicks and Ellinger (2020) [28] | Eight trials | P. ostreatus intake may improve cardiometabolic health, but ongoing research is needed. |
| Cashman et al. (2016) [29] | Six trials | Consumption of ultraviolet (UV)-exposed mushrooms may increase serum 25(OH)D when baseline vitamin D status is low. |
| Fritz et al. (2015) [30] | 28 studies | Polysaccharide K may improve immune function, reduce tumor-associated symptoms, and extend survival in lung cancer patients. |
| Shitake, Dried, Raw | Oyster Mushrooms, Raw | White Mushrooms, Raw | Mycoprotein per 100 g (Wet Weight) * | |
|---|---|---|---|---|
| Energy, kcal | 296 | 8 | 7 | 85 |
| Protein, g | 9.6 | 1.6 | 1.0 | 11 |
| Total fat, g | 1.0 | 0.2 | 0.2 | 2.9 |
| Saturated fatty acids, g | 0.2 | Tr | 0.04 | 0.7 |
| Monounsaturated fatty acids, g | 0.3 | Tr | Tr | 0.5 |
| Polyunsaturated fatty acids, g | 0.1 | 0.10 | 0.11 | 1.8 |
| Total carbohydrate, g | 63.9 | Tr | 0.3 | 3.0 |
| Sugars, g | N | Tr | 0.3 | 0.5 |
| Dietary fiber, g | N | N | 1.2 | 6.0 |
| Riboflavin, mg | 1.27 | 0.4 | 0.27 | 0.26 |
| Folate (B9), μg | N | N | 40 | 114 |
| Vitamin B-12, μg | 0.0 | 0.0 | 0.0 | 0.71 |
| Choline, mg | - | - | - | 180 |
| Phosphorous, mg | 290 | 120 | 94 | 290 |
| Zinc, mg | N | Tr | 0.6 | 7.6 |
| Iron, mg | 1.7 | 1.9 | 0.21 | 0.39 |
| Manganese, mg | N | 3.6 | 0.05 | 4.9 |
| Sodium, mg | 13 | 77 | 4 | 5.0 |
| Animal-Based Protein | Plant-Based Protein | Mycoprotein | |||
|---|---|---|---|---|---|
| Beef | Cows Milk | Soy Isolate * | Soy Concentrate * | ||
| Histidine | 0.30 | 0.09 | 0.6 | 0.4 | 0.39 |
| Isoleucine | 0.87 | 0.20 | 1.1 | 0.8 | 0.57 |
| Leucine | 2.53 | 0.32 | 1.8 | 1.3 | 0.95 |
| Lysine | 1.60 | 0.26 | 1.4 | 1 | 0.91 |
| Methionine | 0.50 | 0.08 | 0.3 | 0.2 | 0.23 |
| Phenylalanine | 0.76 | 0.16 | 1.1 | 0.9 | 0.54 |
| Tryptophan | 0.22 | 0.05 | 0.3 | 0.2 | 0.18 |
| Threonine | 0.84 | 0.15 | 0.8 | 0.7 | 0.61 |
| Valine | 0.94 | 0.22 | 1.1 | 0.8 | 0.60 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Derbyshire, E.J. Is There Scope for a Novel Mycelium Category of Proteins alongside Animals and Plants? Foods 2020, 9, 1151. https://doi.org/10.3390/foods9091151
Derbyshire EJ. Is There Scope for a Novel Mycelium Category of Proteins alongside Animals and Plants? Foods. 2020; 9(9):1151. https://doi.org/10.3390/foods9091151
Chicago/Turabian StyleDerbyshire, Emma J. 2020. "Is There Scope for a Novel Mycelium Category of Proteins alongside Animals and Plants?" Foods 9, no. 9: 1151. https://doi.org/10.3390/foods9091151
APA StyleDerbyshire, E. J. (2020). Is There Scope for a Novel Mycelium Category of Proteins alongside Animals and Plants? Foods, 9(9), 1151. https://doi.org/10.3390/foods9091151





