Effects of Animal, Climatic, Hunting and Handling Conditions on the Hygienic Characteristics of Hunted Roe Doer (Caprelous capreolus L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Hunting Remarks and Carcass Sampling
2.2. Microbial Analysis
2.3. Statistical Analysis
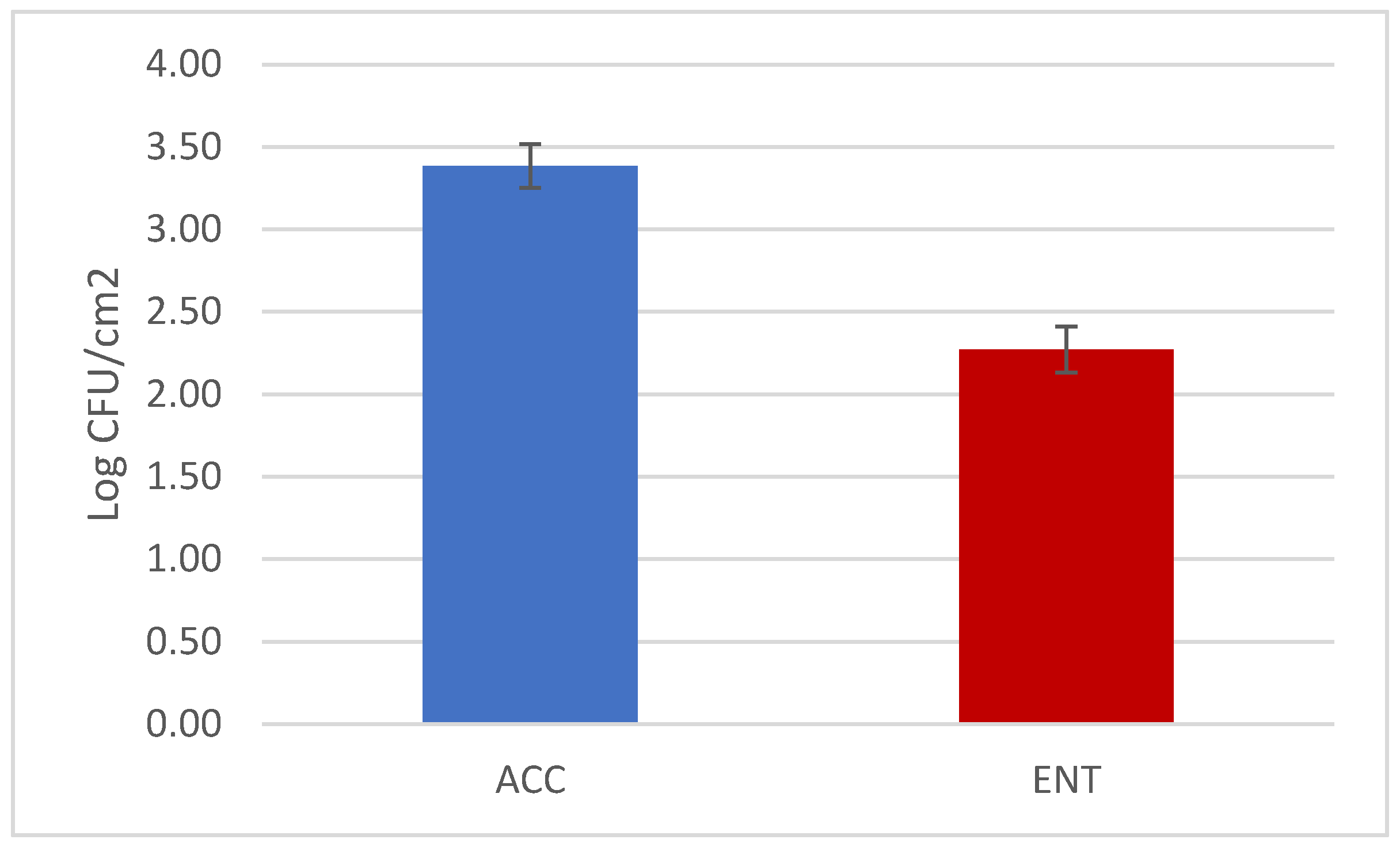
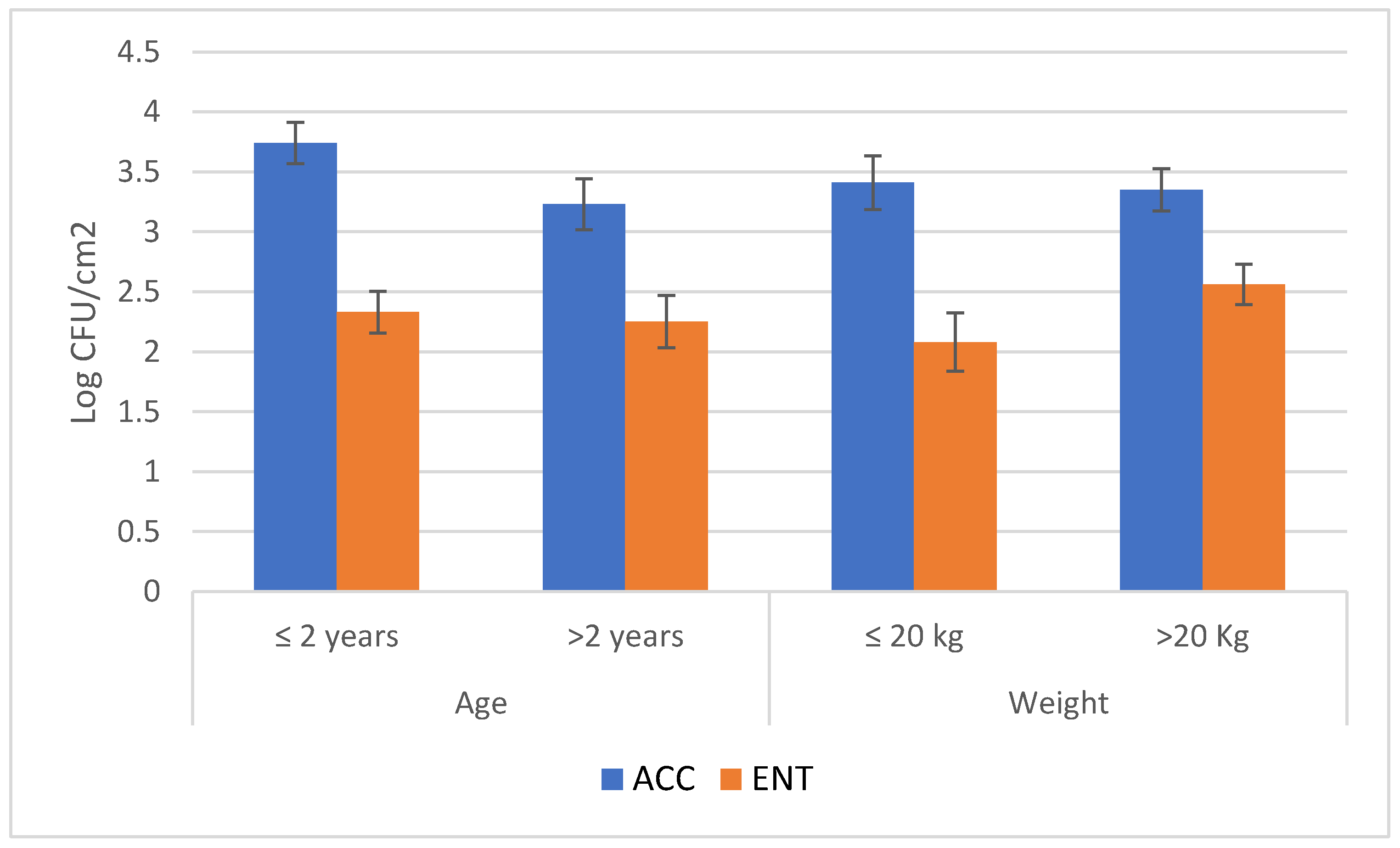
3. Results
4. Discussions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pedrazzoli, M.; Dal Bosco, A.; Castellini, C.; Ranucci, D.; Mattioli, S.; Pauselli, M.; Roscini, V. Effect of age and feeding area on meat quality of wild boars. Ital. J. Anim. Sci. 2017, 16, 353–362. [Google Scholar] [CrossRef]
- Ferroglio, E.; Gortazar, C.; Vicente, J. Wild ungulate diseases and the risk for livestock and public health. In Ungulate Management in Europe: Problems and Practices; Putman, R., Apollonio, M., Andersen, R., Eds.; Cambridge University Press: Cambridge, UK, 2011; pp. 192–214. [Google Scholar]
- Langbein, J.; Putman, R.; Pokorny, B. Traffic collisions involving deer and other ungulates in Europe and available measures for mitigation. In Ungulate Management in Europe: Problems and Practices; Putman, R., Apollonio, M., Andersen, R., Eds.; Cambridge University Press: Cambridge, UK, 2011; pp. 215–259. [Google Scholar]
- Hagen, R.; Haydn, A.; Suchant, R. Estimating red deer (Cervus elaphus) population size in the Southern Black Forest: The role of hunting in population control. Eur. J. Wildl. Res. 2018, 64, 42. [Google Scholar] [CrossRef]
- Winkelmayer, R.; Stangl, P.-V.; Paulsen, P. Assurance of food safety along the game meat production chain: Inspection of meat from wild game and education of official veterinarians and ‘trained persons’ in Austria. In Game Meat Hygiene in Focus; Paulsen, P., Bauer, A., Vodnansky, M., Winkelmayer, R., Smulders, F.J.M., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2011; pp. 245–258. [Google Scholar]
- Hoffman, L.C.; Wiklund, E. Game and venison–meat for the modern consumer. Meat Sci. 2006, 74, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Ranucci, D.; Roila, R.; Andoni, E.; Braconi, P.; Branciari, R. Punica granatum and Citrus spp. extract mix affects spoilage microorganisms growth rate in vacuum-packaged cooked sausages made from pork meat, emmer wheat (Triticum dicoccum Schübler), almond (Prunus dulcis Mill.) and hazelnut (Corylus avellana L.). Foods 2019, 8, 664. [Google Scholar] [CrossRef] [PubMed]
- European Parliament and of the Council Regulation (EC) of 29 April 2004 laying down specific hygiene rules for food of animal origin 853/2004/CE. Off. J. 2004, L139, 55.
- Ranucci, D.; Roila, R.; Miraglia, D.; Arcangeli, C.; Vercillo, F.; Bellucci, S.; Branciari, R. Microbial, chemical-physical, rheological and organoleptic characterisation of roe deer (Capreolus capreolus) salami. Ital. J. Food Saf. 2019, 8, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Marescotti, M.E.; Caputo, V.; Demartini, E.; Gaviglio, A. Discovering market segments for hunted wild game meat. Meat Sci. 2019, 149, 163–176. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Regulation (EC) of 15 November 2005 on microbial criteria for foodstuffs 2073/2005/CE. Off. J. 2005, 338, 1–26. [Google Scholar]
- International Organization for Standardization. Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Colony Count at 30 Degrees C by the Pour Plate Technique; ISO 4833-1:2013; International Organization for Standardization: Geneva, Switzerland, 2013. [Google Scholar]
- International Organization for Standardization. Microbiology of the Food Chain—Horizontal Method for the Detection and Enumeration of Enterobacteriaceae—Part 2: Colony-Cunt Technique. Enumeration of Microorganisms; ISO 21528-2:2017; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- Lindblad, L. Microbiological sampling of swine carcasses: A comparison of data obtained by swabbing with medical gauze and data collected routinely by excision at Swedish abattoirs. Int. J. Food Microbiol. 2007, 118, 180–185. [Google Scholar] [CrossRef]
- International Organization for Standardization. Microbiology of the Food Chain—Horizontal Method for the Detection, Enumeration and Serotyping of Salmonella—Part 1: Detection of Salmonella spp.; ISO 6579-1:2017; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- International Organization for Standardization. Microbiology of the Food Chain—Horizontal Method for the Detection and Enumeration of Listeria Monocytogenes and of Listeria Spp.—Part 1: Detection Method; ISO 11290-1:2017; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- Branciari, R.; Ortenzi, R.; Roila, R.; Miraglia, D.; Ranucci, D.; Valiani, A. Listeria Monocytogenes in Soft Spreadable Salami: Study of the Pathogen Behavior and Growth Prediction During Manufacturing Process and Shelf Life. Appl. Sci. 2020, 10, 4438. [Google Scholar] [CrossRef]
- Akaike, H. A new look at the statistical identification model. IEEE Trans. Autom. Control 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Avagnina, A.; Nucera, D.; Grassi, M.A.; Ferroglio, E.; Dalmasso, A.; Civera, T. The microbiological conditions of carcasses from large game animals in Italy. Meat Sci. 2012, 91, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Klupsaite, D.; Buckiuniene, V.; Sidlauskiene, S.; Lele, V.; Sakiene, V.; Zavistanaviciute, P.; Klementaviciute, J.; Viskontaite, E.; Bartkiene, E. Comparison studies of the chemical, physical, technological, and microbiological characteristics of the European roe deer, boar, red deer, and beaver hunted wild game meat. Anim. Sci. J. 2020, 91, e13346. [Google Scholar] [CrossRef] [PubMed]
- Atanassova, V.; Apelt, J.; Reich, F.; Klein, G. Microbiological quality of freshly shot game in Germany. Meat Sci. 2008, 78, 414–419. [Google Scholar] [CrossRef]
- Eglezos, S.; Huang, B.; Stuttard, E. A survey of the microbiological quality of kangaroo carcasses processed for human consumption in two processing plants in Queensland, Australia. J. Food Prot. 2007, 70, 1249–1251. [Google Scholar] [CrossRef]
- Eglezos, S.; Stuttard, E.; Huang, B.; Dykes, G.A.; Fegan, N. A survey of the microbiological quality of feral pig carcasses processed for human consumption in Queensland, Australia. Foodborne Pathog. Dis. 2008, 5, 105–109. [Google Scholar] [CrossRef]
- Membré, J.M.; Laroche, M.; Magras, C. Assessment of levels of bacterial contamination of large wild game meat in Europe. Food Microbiol. 2011, 28, 1072–1079. [Google Scholar] [CrossRef]
- Gill, C.O. Microbiological conditions of meats from large game animals and birds. Meat Sci. 2007, 77, 149–160. [Google Scholar] [CrossRef]
- Giuggioli, G.; Olivastri, A.; Pennisi, L.; Paludi, D.; Ianieri, A.; Vergara, A. The hygiene-sanitary control in the wild game meats. Ital. J. Food Saf. 2017, 6, 222–224. [Google Scholar] [CrossRef][Green Version]
- Ranucci, D.; Branciari, R.; Miraglia, D.; Stocchi, R.; Rea, S.; Loschi, A.R. Evaluation of carcass hygiene in sheep subjected to gas de-pelting with different skinning procedures. Ital. J. Food Saf. 2014, 3, 188–191. [Google Scholar] [CrossRef][Green Version]
- Milios, K.; Mataragas, M.; Pantouvakis, A.; Drosinos, E.H.; Zoiopoulos, P.E. Evaluation of control over the microbiological contamination of carcass in lamb carcass dressing process operated with or without pasteurizing treatment. Int. J. Food Microbiol. 2011, 146, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Stella, S.; Tirloni, E.; Castelli, E.; Colombo, F.; Bernardi, C. Microbiological evaluation of carcasses of wild boar hunted in a hill area of Northern Italy. J. Food Prot. 2018, 81, 1519–1525. [Google Scholar] [CrossRef]
- Paulsen, P. Hygiene and microbiology of meat from wild game: An Austrian view. In Game Meat Hygiene in Focus; Paulsen, P., Bauer, A., Vodnansky, M., Winkelmayer, R., Smulders, F.J.M., Eds.; Academic Publishers: Wageningen, The Netherlands, 2011; pp. 20–37. [Google Scholar]
- Decastelli, L.; Giaccone, V.; Mignone, W. Bacteriologica examination of meat of wild boars shot down in Piedmont and Liguria, Italy. IBEX J. Mt. Ecol. 1995, 3, 88–89. [Google Scholar]
- Van Schalkwyk, D.L.; Hoffman, L.C.; Laubscher, L.A. Game harvesting procedures and their effect on meat quality: The Africa experience. In Game Meat Hygiene in Focus; Paulsen, P., Bauer, A., Vodnansky, M., Winkelmayer, R., Smulders, F.J.M., Eds.; Academic Publishers: Wageningen, The Netherlands, 2011; pp. 67–92. [Google Scholar]
- Gremse, C.; Rieger, S.; Lahrssen-Wiederholt, M.; Ball, J.P.; Gremse, F. Risk analysis of game meat-borne hazards induced by hunting rifle bullets: Intermediate report on German field studies. In Trends in Game Meat Hygiene; Paulsen, P., Bauer, A., Smulders, F.M.J., Eds.; Academic Publishers: Wageningen, The Netherlands, 2014; pp. 353–362. [Google Scholar]
- Alberto, J.R.; Serejo, J.P.; Vieira-Pinto, M. Dog bites in hunted large game: A hygienic and economical problem for game meat production. In Game Meat Hygiene in Focus; Paulsen, P., Bauer, A., Vodnansky, M., Winkelmayer, R., Smulders, F.J.M., Eds.; Academic Publishers: Wageningen, The Netherlands, 2011; pp. 101–105. [Google Scholar]
- Ramanzin, M.; Amici, A.; Casoli, C.; Esposito, L.; Lupi, P.; Marsico, G.; Mattiello, S.; Olivieri, O.; Ponzetta, M.P.; Russo, C.; et al. Meat from wild ungulates: Ensuring quality and hygiene of an increasing resource. Ital. J. Anim. Sci. 2010, 9, e61. [Google Scholar]
- Mirceta, J.; Petrovic, J.; Malesevic, M.; Blagojevic, B.; Antic, D. Assessment of microbial carcass contamination of hunted wild boars. Eur. J. Wildl. Res. 2017, 63, 37. [Google Scholar] [CrossRef]
- Mirceta, J.; Petrovic, J.; Blagojevic, B.; Malesevic, M.; Antic, D. The microbiological status of carcasses from wild boar in Serbia. Procedia Food Sci. 2015, 5, 199–202. [Google Scholar] [CrossRef]
- Casoli, C.; Duranti, E.; Cambiotti, F.; Avellini, P. Wild ungulate slaughtering and meat inspection. Vet. Res. Commun. 2005, 29, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Orsoni, F.; Romeo, C.; Ferrari, N.; Bardasi, L.; Merialdi, G.; Barbani, R. Factors affecting the microbiological load of Italian hunted wild boar meat (Sus scrofa). Meat Sci. 2020, 160, 107967. [Google Scholar] [CrossRef] [PubMed]
- Borilova, G.; Hulankova, R.; Svobodova, I.; Jezek, F.; Hutarova, Z.; Vecerek, V.; Steinhauserova, I. The effect of storage conditions on the hygiene and sensory status of wild boar meat. Meat Sci. 2016, 118, 71–77. [Google Scholar] [CrossRef]
- Coburn, H.L.; Snary, E.L.; Kelly, L.A.; Wooldridge, M. Qualitative risk assessment of the hazards and risks from wild game. Vet. Rec. 2005, 157, 321–322. [Google Scholar] [CrossRef]
- Paulsen, P.; Smulders, F.J.M.; Hilbert, F. Salmonella in meat from hunted game: A Central European perspective. Food Res. Int. 2012, 45, 609–616. [Google Scholar] [CrossRef]
- Lillehaug, A.; Bergsjø, B.; Schau, J.; Bruheim, T.; Vikøren, T.; Handeland, K. Campylobacter spp., Salmonella spp., verocytotoxic Escherichia coli, and antibiotic resistance in indicator organisms in wild cervids. Acta Vet. Scand. 2015, 46, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Peruzy, M.F.; Murru, N.; Yu, Z.; Kerkhof, P.J.; Neola, B.; Joossens, M.; Proroga, Y.T.R.; Houf, K. Assessment of microbial communities on freshly killed wild boar meat by MALDI-TOF MS and 16S rRNA amplicon sequencing. Int. J. Food Microbiol. 2019, 301, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Weindl, L.; Frank, E.; Ullrich, U.; Heurich, M.; Kleta, S.; Ellerbroek, L.; Gareis, M. Listeria monocytogenes in different specimens from healthy red deer and wild boars. Foodborne Pathog. Dis. 2016, 13, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Lucchini, R.; Armani, M.; Novelli, E.; Rodas, S.; Masiero, A.; Minenna, J.; Drigo, I.; Bacchin, C.; Piovesana, A.; Favretti, M.; et al. Listeria monocytogenes in game meat cured sausages. In Trends in Game Meat Hygiene; Paulsen, P., Bauer, A., Smulders, F.M.J., Eds.; Academic Publishers: Wageningen, The Netherlands, 2014; pp. 167–174. [Google Scholar]
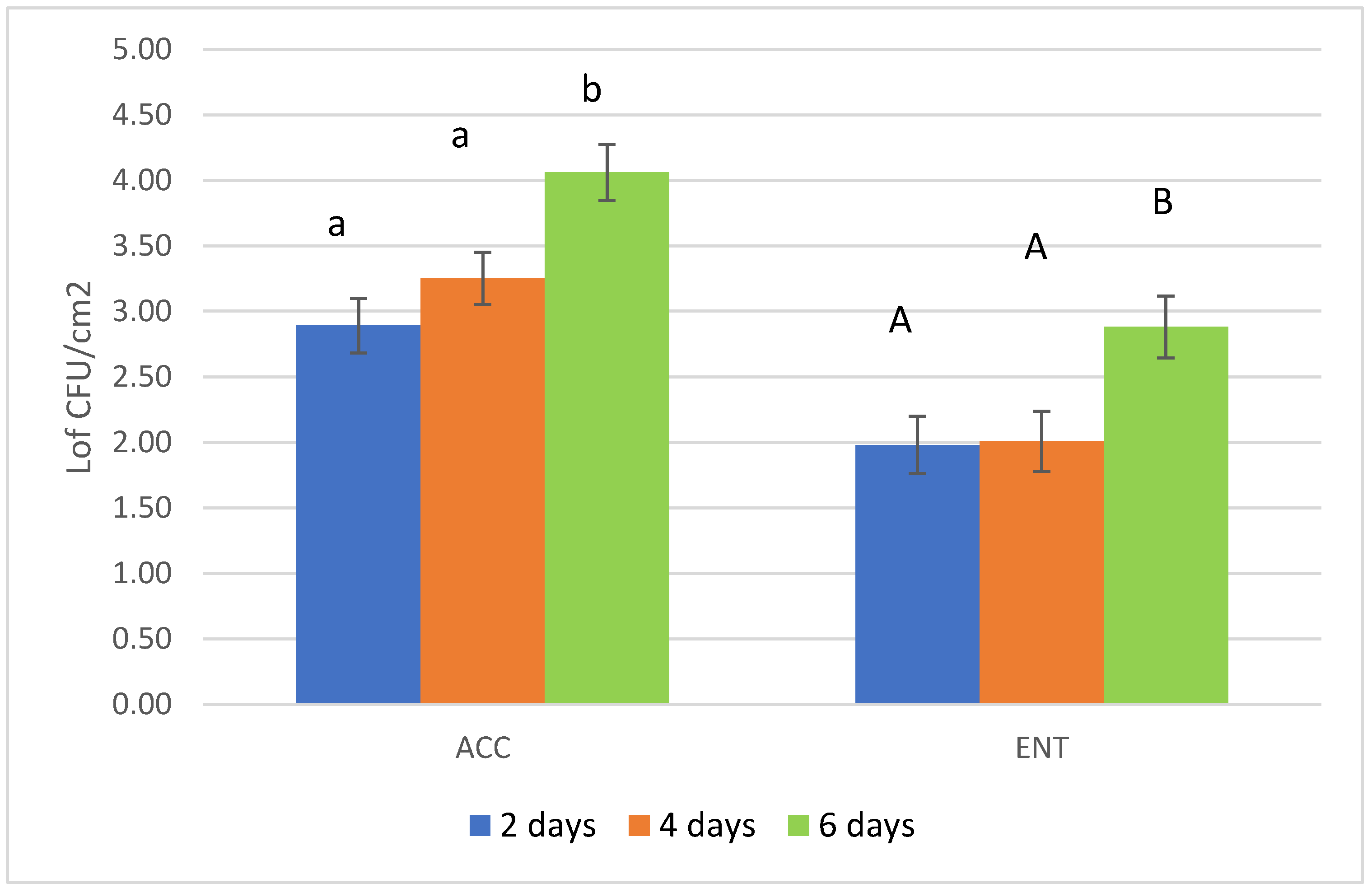
| Questions | Selected Answers | ||||
|---|---|---|---|---|---|
| General question | |||||
| Date and time (hour) | |||||
| Environmental temperature °C | <10 °C | 10–15 °C | >15 °C | ||
| Roe deer generality | |||||
| Gender | male | female | |||
| Age (presumptive) | |||||
| Weight (after shooting) in kg | |||||
| Animal condition | good | bad | |||
| Shooting conditions | |||||
| Shotgun | Smooth bore | Rifled bore | |||
| Bullet | Single | Buckshot | |||
| Ammunition caliber | 7.0 mm | 7.8 mm | |||
| Shooting site | Head | Neck | Shoulder/ hearth | Low thorax | Abdomen |
| Hunting condition | |||||
| Time between shooting and collection | <15 min | 15–29 min | 30–59 min | 60–90 min | |
| Time between collection and bleeding | <15 min | 15–29 min | 30–59 min | 60–90 min | |
| Time between bleeding and evisceration | <30 min | 30–59 min | 60–90 min | ||
| Rupture of the intestine | yes | no | |||
| Questions | Classes | |||
|---|---|---|---|---|
| Environmental temperature °C | <10 °C | 10–15 °C | >15 °C | |
| numbers of samples | 13 | 21 | 30 | |
| Age (presumptive) | ≤2 year | >2 year | ||
| numbers of samples | 39 | 25 | ||
| Weight (after shooting) in kg | 10–20 kg | 20–30 kg | ||
| numbers of samples | 20 | 44 | ||
| Ammunition caliber | 7.0 mm | 7.8 mm | ||
| numbers of samples | 28 | 36 | ||
| Shooting site | Head | Neck | Shoulder/Heart | Low Thorax |
| numbers of samples | 18 | 13 | 24 | 9 |
| Time between shooting and collection | <15 min | 15–29 min | 30–60 min | |
| numbers of samples | 27 | 28 | 9 | |
| Time between shooting and bleeding | <30 min | 31–60 min | 61–90 min | |
| numbers of samples | 27 | 27 | 10 | |
| Time between shooting and evisceration | 30–59 min | 1.0–2.5 h | 2.5–4 h | |
| numbers of samples | 46 | 11 | 7 | |
| Time of storage before skinning | 2 days | 4 days | 6 days | |
| numbers of samples | 21 | 21 | 20 | |
| Environmental Temperature | p Value | |||
|---|---|---|---|---|
| <10 °C | 10–15 °C | >15 °C | ||
| ACC | 3.32 ± 0.298 | 3.38 ± 0.234 | 3.42 ± 0.196 | 0.963 |
| ENT | 1.48 ± 0.29 a | 2.43 ± 0.19 b | 2.53 ± 0.23 b | 0.012 |
| Ammunition Caliber | p Value | ||||||
|---|---|---|---|---|---|---|---|
| 7 mm | 7.8 mm | ||||||
| ACC | 3.38 ± 0.202 | 3.39 ± 0.18 | 0.969 | ||||
| ENT | 2.20 ± 0.21 | 2.33 ± 0.19 | 0.641 | ||||
| Shooting point | |||||||
| Head | Neck | Heart | Thorax | ||||
| ACC | 3.14 ± 0.21 | 3.21 ± 0.21 | 3.52 ± 0.25 | 4.01± 0.35 | 0.164 | ||
| ENT | 2.43 ± 0.27 | 2.07 ± 0.31 | 2.23 ± 0.23 | 2.34 ± 0.38 | 0.841 | ||
| Time between shooting and collection | |||||||
| <15 min | 15–29 min | 30–60 min | |||||
| ACC | 3.25 ± 0.20 | 3.31 ± 0.19 | 4.02 ± 0.35 | 0.151 | |||
| ENT | 2.23 ± 0.22 | 2.29 ± 0.21 | 2.33 ± 0.38 | 0.969 | |||
| Time between shooting and bleeding | |||||||
| <30 min | 31–60 min | 61–90 min | |||||
| ACC | 3.25 ± 0.20 | 3.27± 0.20 | 4.04± 0.33 | 0.098 | |||
| ENT | 2.23 ± 0.22 | 2.28 ± 0.22 | 2.36 ± 0.36 | 0.955 | |||
| Time between shooting and evisceration | |||||||
| 30–59 min | 1.0–2.5 h | 2.5–4 h | |||||
| ACC | 3.19 ± 0.151 a | 3.73 ± 0.309 a,b | 4.10 ± 0.387 b | 0.049 | |||
| ENT | 2.24 ± 0.17 | 2.26 ± 0.34 | 2.51 ± 0.43 | 0.840 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Branciari, R.; Onofri, A.; Cambiotti, F.; Ranucci, D. Effects of Animal, Climatic, Hunting and Handling Conditions on the Hygienic Characteristics of Hunted Roe Doer (Caprelous capreolus L.). Foods 2020, 9, 1076. https://doi.org/10.3390/foods9081076
Branciari R, Onofri A, Cambiotti F, Ranucci D. Effects of Animal, Climatic, Hunting and Handling Conditions on the Hygienic Characteristics of Hunted Roe Doer (Caprelous capreolus L.). Foods. 2020; 9(8):1076. https://doi.org/10.3390/foods9081076
Chicago/Turabian StyleBranciari, Raffaella, Andrea Onofri, Fausto Cambiotti, and David Ranucci. 2020. "Effects of Animal, Climatic, Hunting and Handling Conditions on the Hygienic Characteristics of Hunted Roe Doer (Caprelous capreolus L.)" Foods 9, no. 8: 1076. https://doi.org/10.3390/foods9081076
APA StyleBranciari, R., Onofri, A., Cambiotti, F., & Ranucci, D. (2020). Effects of Animal, Climatic, Hunting and Handling Conditions on the Hygienic Characteristics of Hunted Roe Doer (Caprelous capreolus L.). Foods, 9(8), 1076. https://doi.org/10.3390/foods9081076






