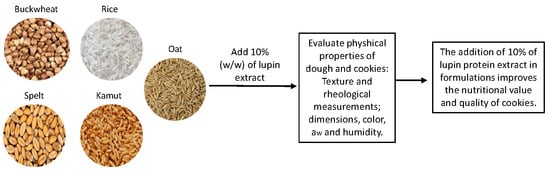
Lupin Seed Protein Extract Can Efficiently Enrich the Physical Properties of Cookies Prepared with Alternative Flours
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Lupin Seed Protein Extract
2.2. Flour Composition
2.3. Cookies Preparation
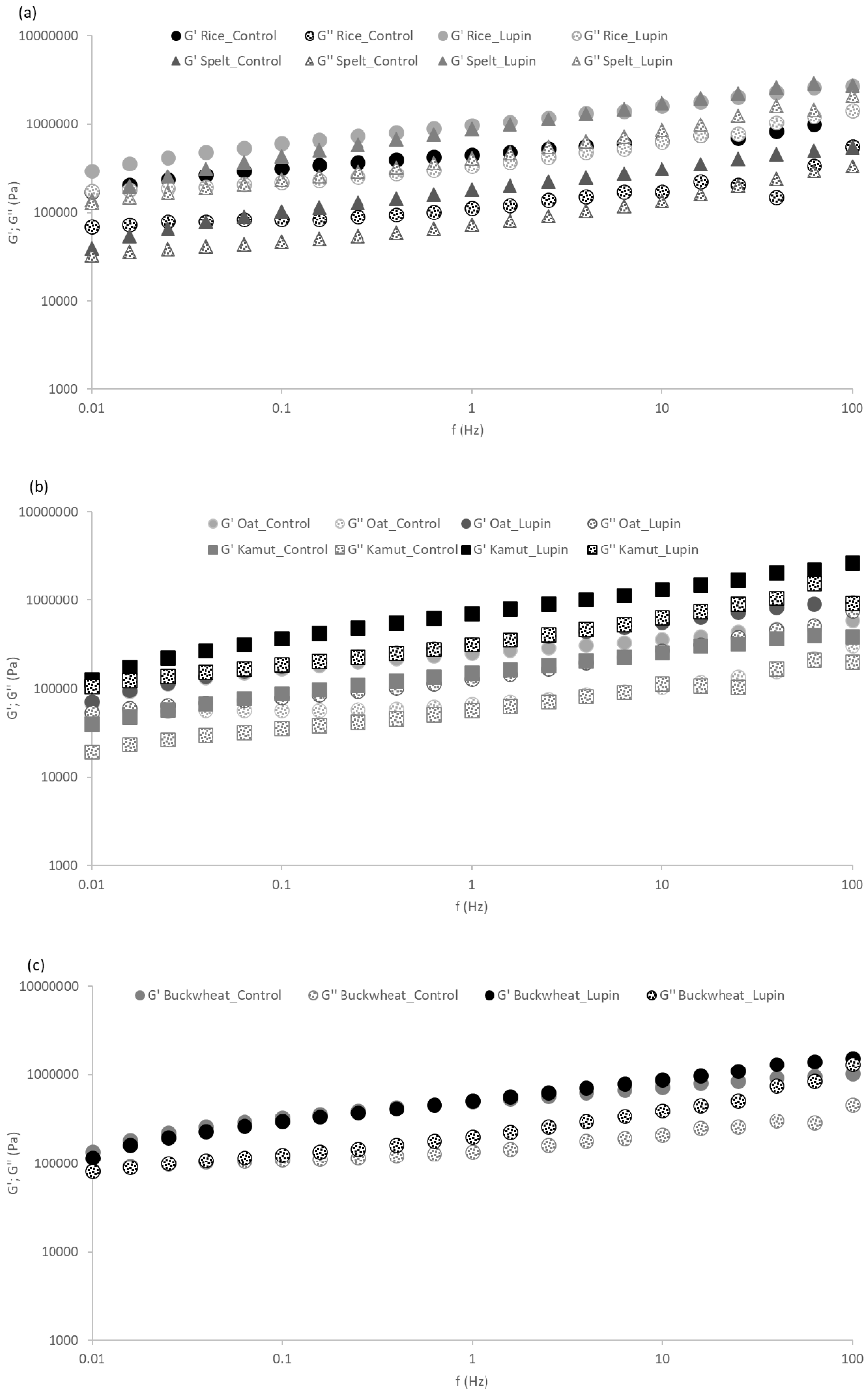
2.4. Dough Rheology
2.5. Dimensions
2.6. Color Analysis
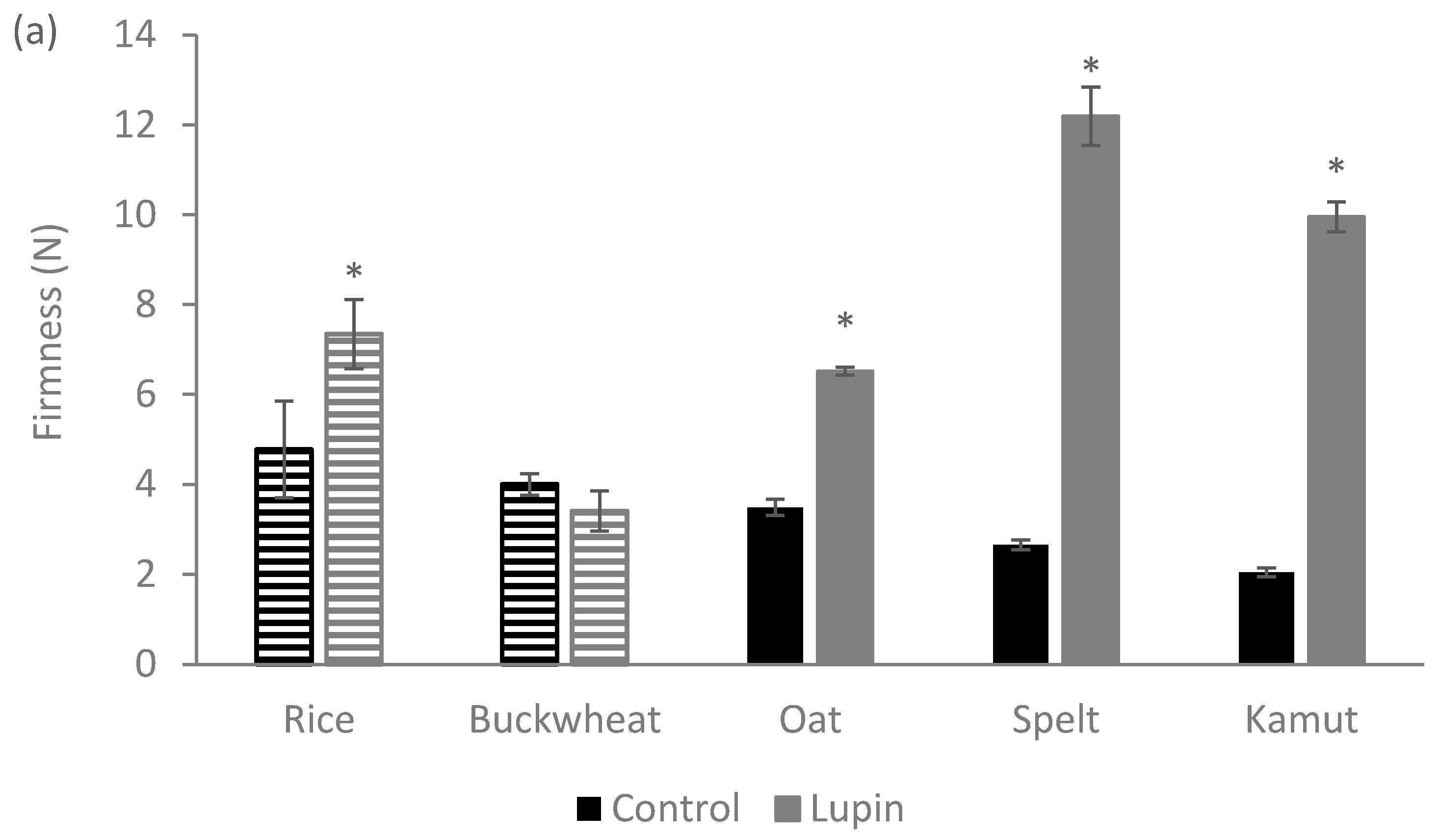
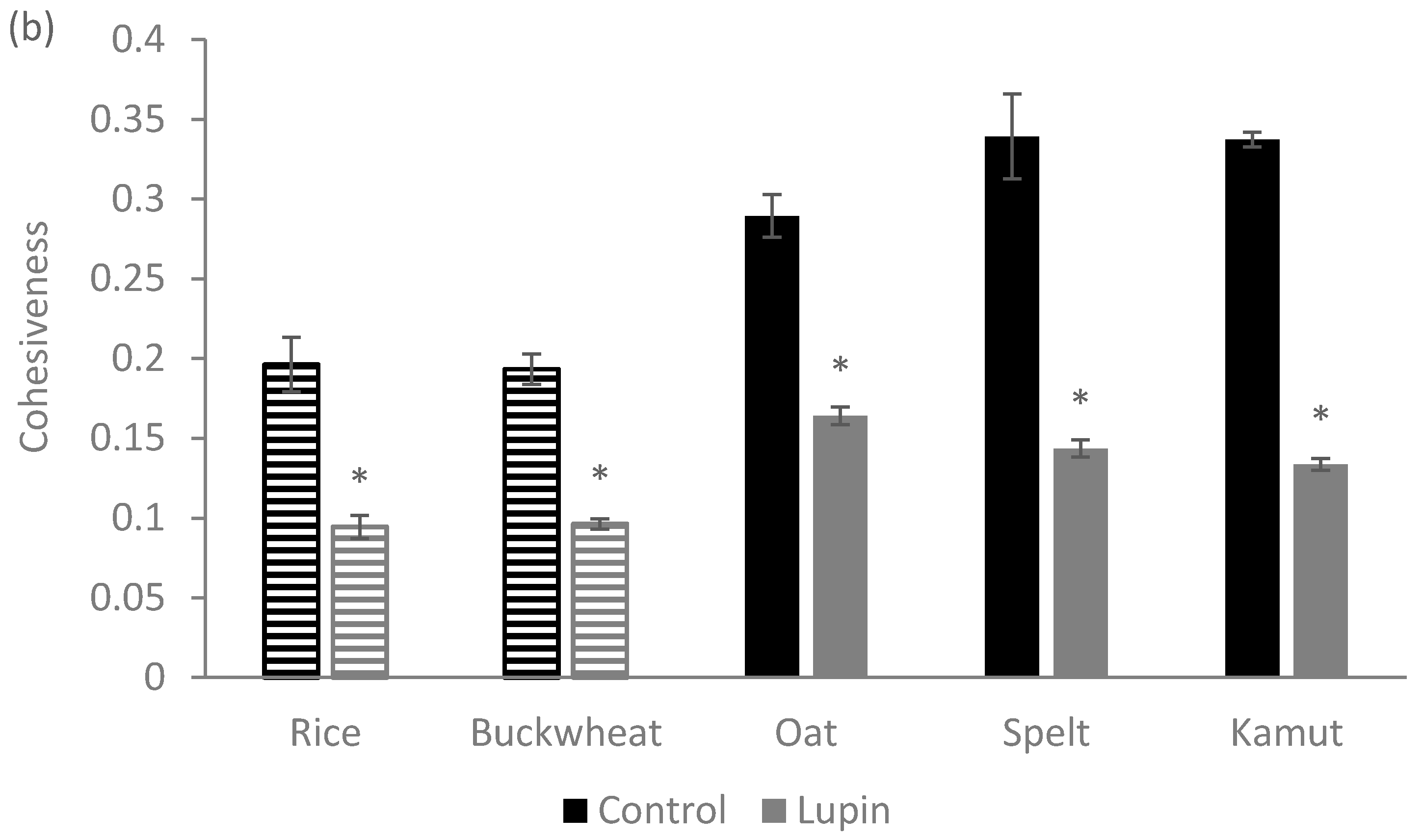
2.7. Texture Analysis
2.7.1. Dough Texture
2.7.2. Cookie Texture
2.8. Water Activity Determination
2.9. Moisture Content
2.10. Statistical Analyses
3. Results and Discussion
3.1. Physical Properties of the Dough
3.2. Physical Properties of Cookies
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Top Ten Trends for 2019 by Innova Market Insights. Available online: https://retailreport.at/sites/default/files/2019-06/Innova%20Market%20Trends.pdf (accessed on 23 January 2020).
- Morales-Polanco, E.; Campos-Veja, R.; Gaytán-Martínez, M.; Enriquez, L.G.; Loarca-Piña, G. Functional and textural properties of a dehulled oat (Avena sativa L.) and pea (Pisum sativum) protein isolate cracker. LWT Food Sci. Technol. 2017, 86, 418–423. [Google Scholar] [CrossRef]
- Sahagún, M.; Gómez, M. Influence of protein source on characteristics and quality of gluten-free cookies. J. Food Sci. Technol. 2018, 55, 4131–4138. [Google Scholar] [CrossRef] [PubMed]
- Zucco, F.; Borsuk, Y.; Arntfield, S.D. Physical and nutritional evaluation of wheat cookies supplemented with pulse flours of different particle sizes. LWT Food Sci. Technol. 2011, 44, 2070–2076. [Google Scholar] [CrossRef]
- Duodu, K.G.; Minnaar, A. Legume composite flours and baked goods: Nutritional, functional, sensory, and phytochemical qualities. In Flour and Breads and Their Fortification in Health and Disease Prevention, 2nd ed.; Preedy, V.R., Watson, R.R., Patel, V.B., Eds.; Academic Press: San Diego, CA, USA, 2011; pp. 193–203. [Google Scholar]
- De Punder, K.; Pruimboom, L. The dietary intake of wheat and other cereal grains and their role in inflammation. Nutrients 2013, 5, 771–787. [Google Scholar] [CrossRef] [PubMed]
- Dinu, M.; Whittaker, A.; Pagliai, G.; Benedettelli, S.; Sofi, F. Ancient wheat species and human health: Biochemical and clinical implications. J. Nutr. Biochem. 2018, 52, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Valli, V.; Taccari, A.; Di Nunzio, M.; Danesi, F.; Bordoni, A. Health benefits of ancient grains. Comparison among bread made with ancient, heritage and modern grain flours in human cultured cells. Food Res. Int. 2018, 107, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, S.; Primiterra, M.; Tagliamonte, M.C.; Carnevali, A.; Gianotti, A.; Bordoni, A.; Canestrari, F. Counteraction of oxidative damage in the rat liver by an ancient grain (Kamut brand khorasan wheat). Nutrition 2012, 28, 436–441. [Google Scholar] [CrossRef]
- Hoffmanová, I.; Sánchez, D.; Szczepanková, A.; Tlaskalová-Hogenová, H. The Pros and cons of using oat in a gluten-free diet for celiac patients. Nutrients 2019, 11, 2345. [Google Scholar] [CrossRef]
- Singh, R.; De, S.; Belkheir, A. Avena sativa (Oat), a potential neutraceutical and therapeutic agent: An overview. Crit. Rev. Food Sci. Nutr. 2013, 53, 126–144. [Google Scholar] [CrossRef]
- Gilissen, L.J.; Van der Meer, I.M.; Smulders, M.J. Why oats are safe and healthy for celiac disease patients. Med. Sci. 2016, 4, 21. [Google Scholar] [CrossRef]
- Hager, A.S.; Wolter, A.; Jacob, F.; Zannini, E.; Arendt, E.K. Nutritional properties and ultra-structure of commercial gluten free flours from different botanical sources compared to wheat flours. J. Cereal Sci. 2012, 56, 239–247. [Google Scholar] [CrossRef]
- Starowicz, M.; Koutsidis, G.; Zieliński, H. Sensory analysis and aroma compounds of buckwheat containing products—A review. Crit. Rev. Food Sci. 2018, 58, 1767–1779. [Google Scholar] [CrossRef] [PubMed]
- Barton, S.H.; Kelly, D.G.; Murray, J.A. Nutritional deficiencies in celiac disease. Gastroenterol. Clin. N. Am. 2007, 36, 93–108. [Google Scholar] [CrossRef]
- Taetzsch, A.; Das, S.K.; Brown, C.; Krauss, A.; Silver, R.E.; Roberts, S.B. Are gluten-free diets more nutritious? An evaluation of self-selected and recommended gluten-free and gluten-containing dietary patterns. Nutrients 2018, 10, 1881. [Google Scholar] [CrossRef] [PubMed]
- Gimenez-Bastida, J.A.; Zielinski, H. Buckwheat as a functional food and its effects on health. J. Agric. Food Chem. 2015, 63, 7896–7913. [Google Scholar] [CrossRef] [PubMed]
- Llavata, B.; Albors, A.; Martin-Esparza, M.E. High fibre gluten-free fresh pasta with tiger nut, chickpea and fenugreek: Technofunctional, sensory and nutritional properties. Foods 2020, 9, 11. [Google Scholar] [CrossRef]
- Paraskevopoulou, A.; Provatidou, E.; Tsotsiou, D.; Kiosseoglou, V. Dough rheology and baking performance of wheat flour-lupin protein isolate blends. Food Res. Int. 2010, 43, 1009–1016. [Google Scholar] [CrossRef]
- Lima, A.I.G.; Mota, J.; Monteiro, S.A.V.S.; Ferreira, R.M.S.B. Legume seeds and colorectal cancer revisited: Protease inhibitors reduce MMP-9 activity and colon cancer cell migration. Food Chem. 2016, 197, 30–38. [Google Scholar] [CrossRef]
- Capraro, J.; Magni, C.; Scarafoni, A.; Caramanico, R.; Rossi, F.; Morlacchini, M.; Duranti, M. Pasta supplemented with isolated lupin protein fractions reduces body weight gain and food intake of rats and decreases plasma glucose concentration upon glucose overload trial. Food Funct. 2014, 5, 375–380. [Google Scholar] [CrossRef]
- Erbaş, M.; Certel, M.; Uslu, M.K. Some chemical properties of white lupin seeds (Lupinus albus L.). Food Chem. 2005, 89, 341–345. [Google Scholar] [CrossRef]
- Favier, J.C.; Ripert, J.I.; Toque, C.; Feinberg, M. Répertoire Général des Aliments. Composition Tables, 2nd ed.; INRA Editions: Paris, France, 1995. [Google Scholar]
- Gupta, S.; Liu, C.; Sathe, S.K. Quality of a chickpea-based high protein snack. J. Food Sci. 2019, 84, 1621–1630. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.P.; Mori, T.A.; Sipsas, S.; Barden, A.; Puddey, I.B.; Burke, V.; Hall, R.S.; Hodgson, J.M. Lupin enriched bread increases satiety and reduces energy intake acutely. Am. J. Clin. Nutr. 2006, 84, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Jayasena, V.; Nasar-Abbas, S.M. Effect of lupin flour incorporation on the physical characteristics of dough and biscuits. Qual. Assur. Saf. Crop. 2011, 3, 140–147. [Google Scholar] [CrossRef]
- Lovati, M.R.; Manzoni, C.; Castiglioni, S.; Parolari, A.; Magni, C.; Duranti, M. Lupin seed γ-conglutin lowers blood glucose in hyperglycaemic rats and increases glucose consumption of HepG2 cells. Br. J. Nutr. 2012, 107, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Batista, A.P.; Niccolai, A.; Fradinho, P.; Fragoso, S.; Bursic, I.; Rodolfi, L.; Biondi, N.; Tredici, M.R.; Sousa, I.; Raymundo, A. Microalgae biomass as an alternative ingredient in cookies: Sensory, physical and chemical properties, antioxidant activity and In Vitro digestibility. Algal Res. 2017, 26, 161–171. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Fradinho, P.; Nunes, M.C.; Raymundo, A. Developing consumer acceptable biscuits enriched with Psyllium fibre. J. Food Sci. Technol. 2015, 52, 4830–4840. [Google Scholar] [CrossRef]
- de Penna, E.W.; Carreno, P.; Urrutia, X.; Lopez, L.; Ballester, D. Sensory evaluation and acceptability of cookies enriched with sweet lupine flour (Lupinus albus cv Multolupa). J. Food Sci. 1987, 52, 1434–1435. [Google Scholar] [CrossRef]
- Gorecka, D.; Lampart-Szczapa, E.; Janitz, W.; Sokolowska, B. Composition of fractional and functional properties of dietary fiber of lupines (L. luteus and L. albus). Food Nahr. 2000, 44, 229–232. [Google Scholar] [CrossRef]
- Szczesniak, A.S. Texture is a sensory property. Food Qual. Prefer. 2002, 13, 215–225. [Google Scholar] [CrossRef]
- Elgeti, D.; Jekle, M.; Becker, T. Strategies for the aeration of gluten-free bread–A review. Trends Food Sci. Technol. 2015, 46, 75–84. [Google Scholar] [CrossRef]
- Yano, H. Recent practical researches in the development of gluten-free breads. NPJ Sci. Food 2019, 3, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Tomotake, H.; Shimaoka, I.; Kayashita, J.; Nakajoh, M.; Kato, N. Physicochemical and functional properties of buckwheat protein product. J. Agric. Food Chem. 2002, 50, 2125–2129. [Google Scholar] [CrossRef]
- Jeddou, K.B.; Bouaziz, F.; Zouari-Ellouzi, S.; Chaari, F.; Ellouz-Chaabouni, S.; Ellouz-Ghorbel, R.; Nouri-Ellouz, O. Improvement of texture and sensory properties of cakes by addition of potato peel powder with high level of dietary fiber and protein. Food Chem. 2017, 217, 668–677. [Google Scholar] [CrossRef] [PubMed]
- Jooyandeh, H. Effect of fermented whey protein concentrate on texture of Iranian white cheese. J. Texture Stud. 2009, 40, 497–510. [Google Scholar] [CrossRef]
- Ortolan, F.; Steel, C.J. Protein characteristics that affect the quality of vital wheat gluten to be used in baking: A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 369–381. [Google Scholar] [CrossRef]
- Köhn, C.R.; Almeida, J.C.; Schmidt, M.M.; Vidal, A.R.; Kempka, A.P.; Demiate, I.M.; Rosa, C.S.; Kubota, E.H.; Mello, R.O.; Prestes, R.C. Evaluation of water absorption capacity of ingredients and additives used in the meat industry submitted to different saline concentrations and ultrasound. Int. Food Res. J. 2016, 23, 653–659. [Google Scholar]
- Raymundo, A.; Fradinho, P.; Nunes, M.C. Effect of Psyllium fibre content on the textural and rheological characteristics of biscuit and biscuit dough. Bioact. Carbohydr. Diet. Fibre 2014, 3, 96–105. [Google Scholar] [CrossRef]
- Graça, C.; Raymundo, A.; Sousa, I. Wheat bread with dairy products—Technology, nutritional, and sensory properties. Appl. Sci. Basel 2019, 9, 4101. [Google Scholar] [CrossRef]
- Fradinho, P.; Niccolai, A.; Soares, R.; Rodolfi, L.; Biondi, N.; Tredici, M.R.; Sousa, I.; Raymundo, A. Effect of Arthrospira platensis (spirulina) incorporation on the rheological and bioactive properties of gluten-free fresh pasta. Algal Res. 2020, 45, 101743. [Google Scholar] [CrossRef]
- Bilgiçli, N.; Levent, H. Utilization of lupin (Lupinus albus L.) flour and bran with xylanase enzyme in cookie production. Legum. Res. 2014, 37, 264–271. [Google Scholar] [CrossRef]
- Tsen, C.C.; Peters, E.M.; Schaffer, T.; Hoover, W.J. High protein cookies: Effect of soy fortification and surfactants. Bak. Dig. 1973, 47, 34–39. [Google Scholar]
- Jaworska, D.; Hoffmann, M. Relative importance of texture properties in the sensory quality and acceptance of commercial crispy products. J. Sci. Food Agric. 2008, 88, 1804–1812. [Google Scholar] [CrossRef]
- Obeidat, B.A.; Abdul-Hussain, S.S.; Al Omari, D.Z. Effect of addition of germinated lupin flour on the physiochemical and organoleptic properties of cookies. J. Food Process. Preserv. 2013, 37, 637–643. [Google Scholar] [CrossRef]
- Hegazy, N.A.; Faheid, S.M. Rheological and sensory characteristics of doughs and cookies based on wheat, soybean, chickpea and lupine flour. Food Nahr. 1990, 34, 835–841. [Google Scholar] [CrossRef]
- Oliver, C.M.; Melton, L.D.; Stanley, R.A. Creating proteins with novel functionality via the Maillard reaction: A review. Crit. Rev. Food Sci. 2006, 46, 337–350. [Google Scholar] [CrossRef]
- Cervenka, L.; Brožková, I.; Vytřasová, J. Effects of the principal ingredients of biscuits upon water activity. J. Food Nutr. Res. 2006, 45, 39–43. [Google Scholar]
- Uysal, H.; Bilgiçli, N.; Elgün, A.; İbanoğlu, Ş.; Herken, E.N.; Demir, M.K. Effect of dietary fibre and xylanase enzyme addition on the selected properties of wire-cut cookies. J. Food Eng. 2007, 78, 1074–1078. [Google Scholar] [CrossRef]
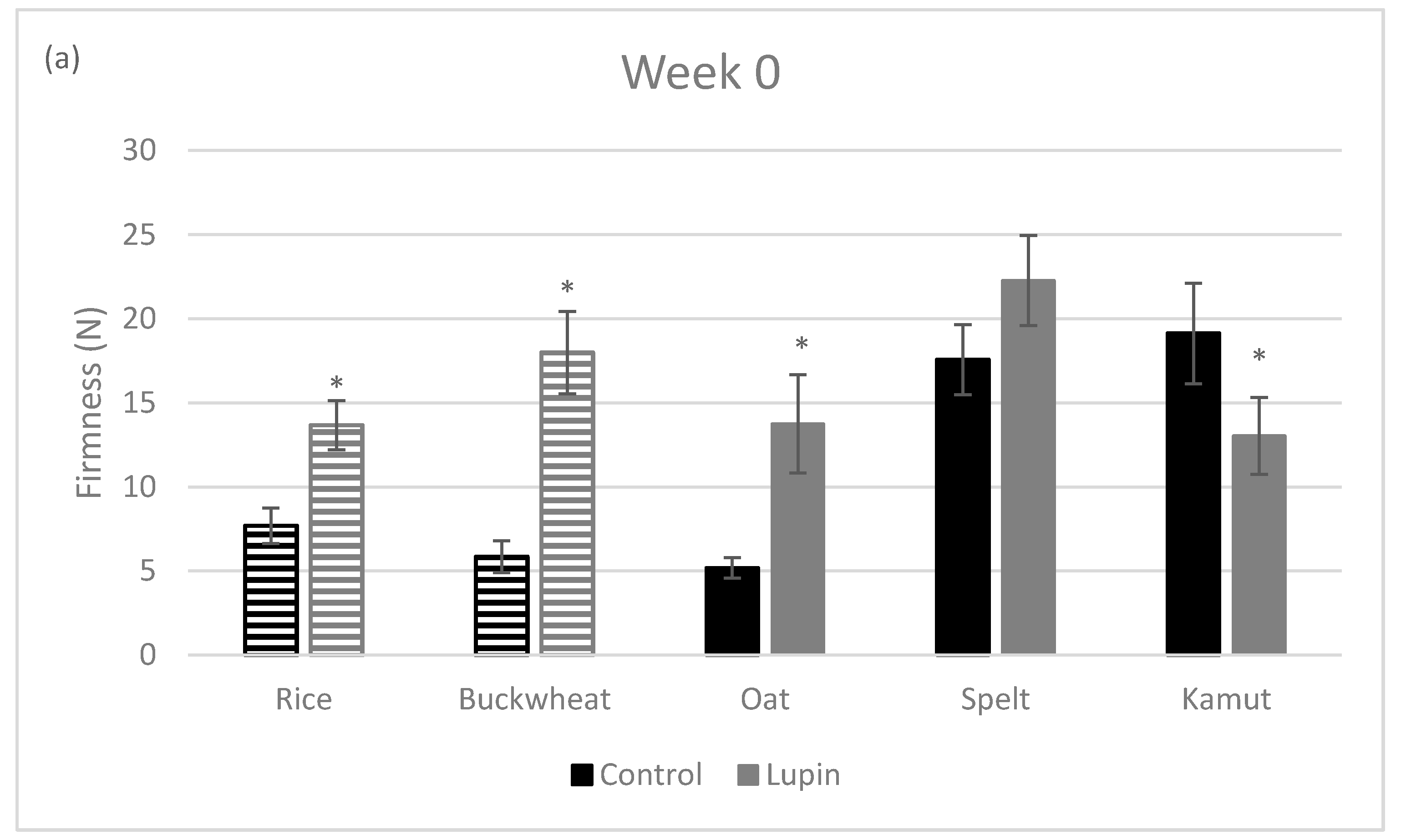
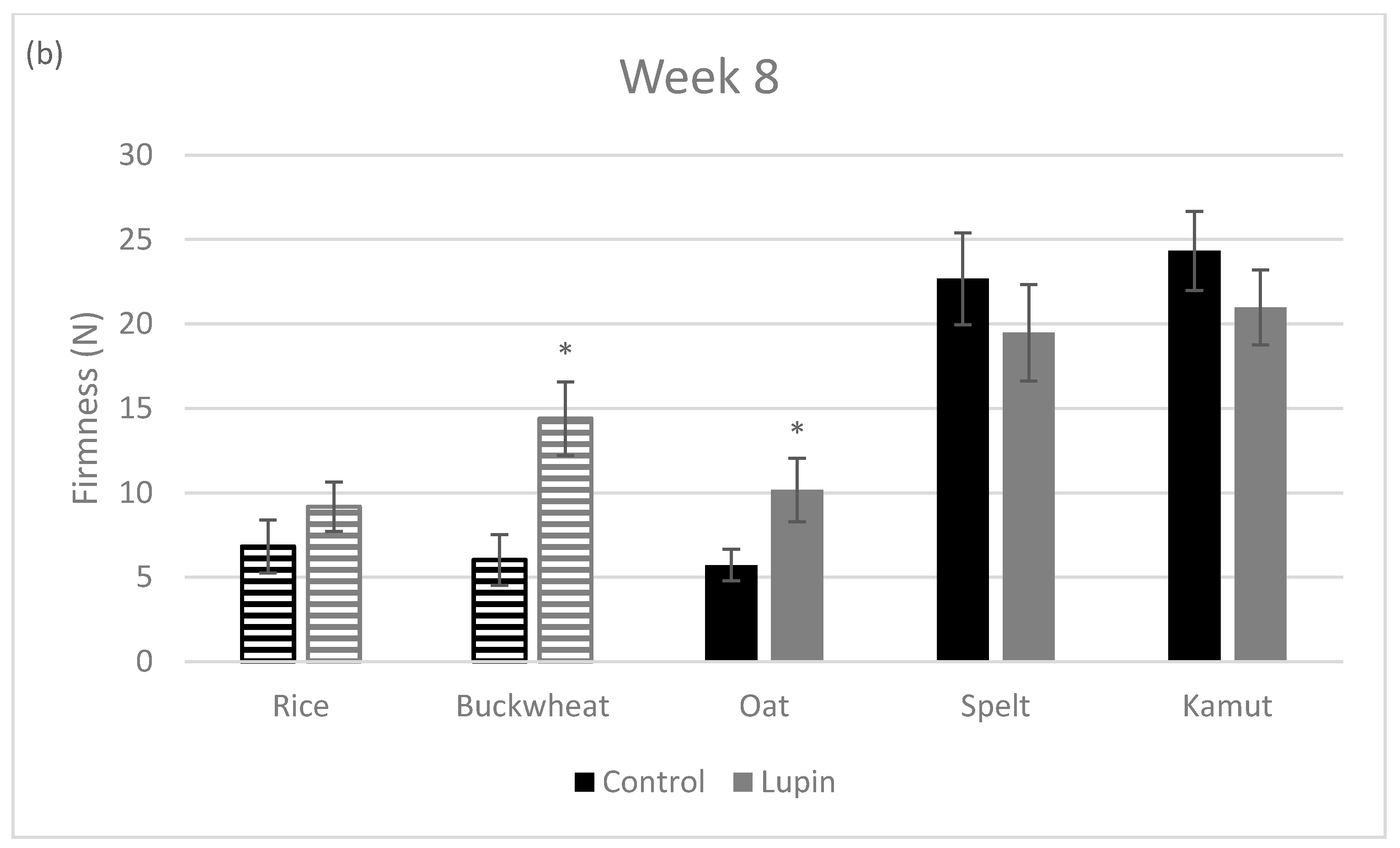
| Rice | Oat | Spelt | Kamut | Buckwheat | |
|---|---|---|---|---|---|
| Energy (kcal/100 g) | 350 | 370 | 295 | 385 | 366 |
| Protein | 7.6 | 14.0 | 13.0 | 15.0 | 13.3 |
| Total lipid | 0.7 | 7.6 | 1.8 | 15.0 | 3.4 |
| Of which saturated | 0.3 | 1.3 | 0.3 | 0.6 | 0.7 |
| Total carbohydrate | 78.5 | 56.0 | 55.0 | 60.0 | 61.5 |
| Of which sugars | 0.1 | 1.1 | 6.8 | 2.0 | 2.0 |
| Of which fiber | 2.4 | 10.0 | 4.0 | 11.0 | 10.0 |
| Salt | <0.1 | 0.01 | 0.02 | 0.03 | 0.08 |
| G’ 1 Hz (Pa) | ||
|---|---|---|
| Control | Lupin | |
| Rice | 4.6 × 105 ± 9.1 × 104 | 8.3 × 105 ± 1.3 × 105 * |
| Buckwheat | 5.0 × 105 ± 2.9 × 104 | 5.2 × 105 ± 1.5 × 104 |
| Oat | 2.5 × 105 ± 8.3 × 104 | 2.9 × 105 ± 2.6 × 104 |
| Spelt | 2.0 × 105 ± 5.1 × 104 | 6.2 × 105 ± 9.6 × 104 * |
| Kamut | 1.6 × 105 ± 1.4 × 104 | 7.6 × 105 ± 8.5 × 104 * |
| Cookie Formulation | Area (cm2) | Thickness (mm) | |
|---|---|---|---|
| Rice | Control | 15.63 ± 0.06 | 2.55 ± 0.14 |
| LE | 15.08 ± 0.12 ** | 2.94 ± 0.08 * | |
| Buckwheat | Control | 13.26 ± 0.11 | 2.62 ± 0.11 |
| LE | 16.05 ± 0.16 ** | 3.14 ± 0.10 * | |
| Oat | Control | 15.61 ± 0.10 | 3.25 ± 0.34 |
| LE | 15.44 ± 0.21 | 2.79 ± 0.12 | |
| Spelt | Control | 13.00 ± 0.03 | 3.57 ± 0.10 |
| LE | 15.94 ± 0.11 ** | 3.35 ± 0.28 | |
| Kamut | Control | 15.30 ± 0.07 | 2.94 ± 0.20 |
| Lupin | 16.69 ± 0.06 ** | 2.68 ± 0.16 | |
| Rice | Buckwheat | Oat | Spelt | Kamut | |||
|---|---|---|---|---|---|---|---|
| ΔE* | Week 0 | 21.52 | 20.58 | 12.22 | 31.19 | 15.80 | |
| Week 8 | 24.99 | 20.18 | 11.97 | 20.20 | 22.03 | ||
| L* | Week 0 | Control | 80.40 ± 1.28 | 72.84 ± 0.81 | 71.27 ± 0.92 | 55.16 ± 3.04 | 73.43 ± 0.69 |
| Lupin | 65.74 ± 2.73 ** | 56.78 ± 1.53 ** | 61.21 ± 1.47 ** | 64.68 ± 2.05 * | 59.72 ± 1.11 ** | ||
| Week 8 | Control | 80.81 ± 1.06 | 69.37 ± 2.89 | 71.41 ± 0.97 | 78.43 ± 3.57 | 72.79 ± 1.70 | |
| Lupin | 62.10 ± 1.51 ** | 55.28 ± 3.02 * | 63.11 ± 2.00 * | 60.50 ± 2.21 * | 53.41 ± 2.80 ** | ||
| aw | Week 0 | Control | 0.36 ± 0.02 | 0.21 ± 0.01 | 0.04 ± 0.02 | 0.09 ± 0.01 | 0.50 ± 0.03 |
| Lupin | 0.59 ± 0.01 ** | 0.35 ± 0.01 ** | 0.09 ± 0.00 * | 0.40 ± 0.02 ** | 0.39 ± 0.01 * | ||
| Week 8 | Control | 0.17 ± 0.00 | 0.12 ± 0.01 | 0.12 ± 0.00 | 0.22 ± 0.00 | 0.15 ± 0.01 | |
| Lupin | 0.23 ± 0.01 ** | 0.37 ± 0.00** | 0.33 ± 0.00 ** | 0.36 ± 0.00 ** | 0.35 ± 0.00 ** | ||
| H (%) | Week 0 | Control | 3.42 ± 0.14 | 5.61 ± 0.61 | 1.04 ± 0.11 | 2.75 ± 0.22 | 1.97 ± 0.08 |
| Lupin | 2.88 ± 0.11 * | 3.88 ± 0.08 * | 2.29 ± 0.16 ** | 4.33 ± 0.08 ** | 2.85 ± 0.06 ** | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mota, J.; Lima, A.; B. Ferreira, R.; Raymundo, A. Lupin Seed Protein Extract Can Efficiently Enrich the Physical Properties of Cookies Prepared with Alternative Flours. Foods 2020, 9, 1064. https://doi.org/10.3390/foods9081064
Mota J, Lima A, B. Ferreira R, Raymundo A. Lupin Seed Protein Extract Can Efficiently Enrich the Physical Properties of Cookies Prepared with Alternative Flours. Foods. 2020; 9(8):1064. https://doi.org/10.3390/foods9081064
Chicago/Turabian StyleMota, Joana, Ana Lima, Ricardo B. Ferreira, and Anabela Raymundo. 2020. "Lupin Seed Protein Extract Can Efficiently Enrich the Physical Properties of Cookies Prepared with Alternative Flours" Foods 9, no. 8: 1064. https://doi.org/10.3390/foods9081064
APA StyleMota, J., Lima, A., B. Ferreira, R., & Raymundo, A. (2020). Lupin Seed Protein Extract Can Efficiently Enrich the Physical Properties of Cookies Prepared with Alternative Flours. Foods, 9(8), 1064. https://doi.org/10.3390/foods9081064