Antioxidant, Angiotensin-Converting Enzyme Inhibitory Properties and Blood-Pressure-Lowering Effect of Rice Bran Protein Hydrolysates
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Defatted Rice Bran Protein Concentrates
2.3. Preparation of Defatted Rice Bran Protein Hydrolysates
2.4. Ultrafiltration Process of Rice Bran Protein Hydrolysates
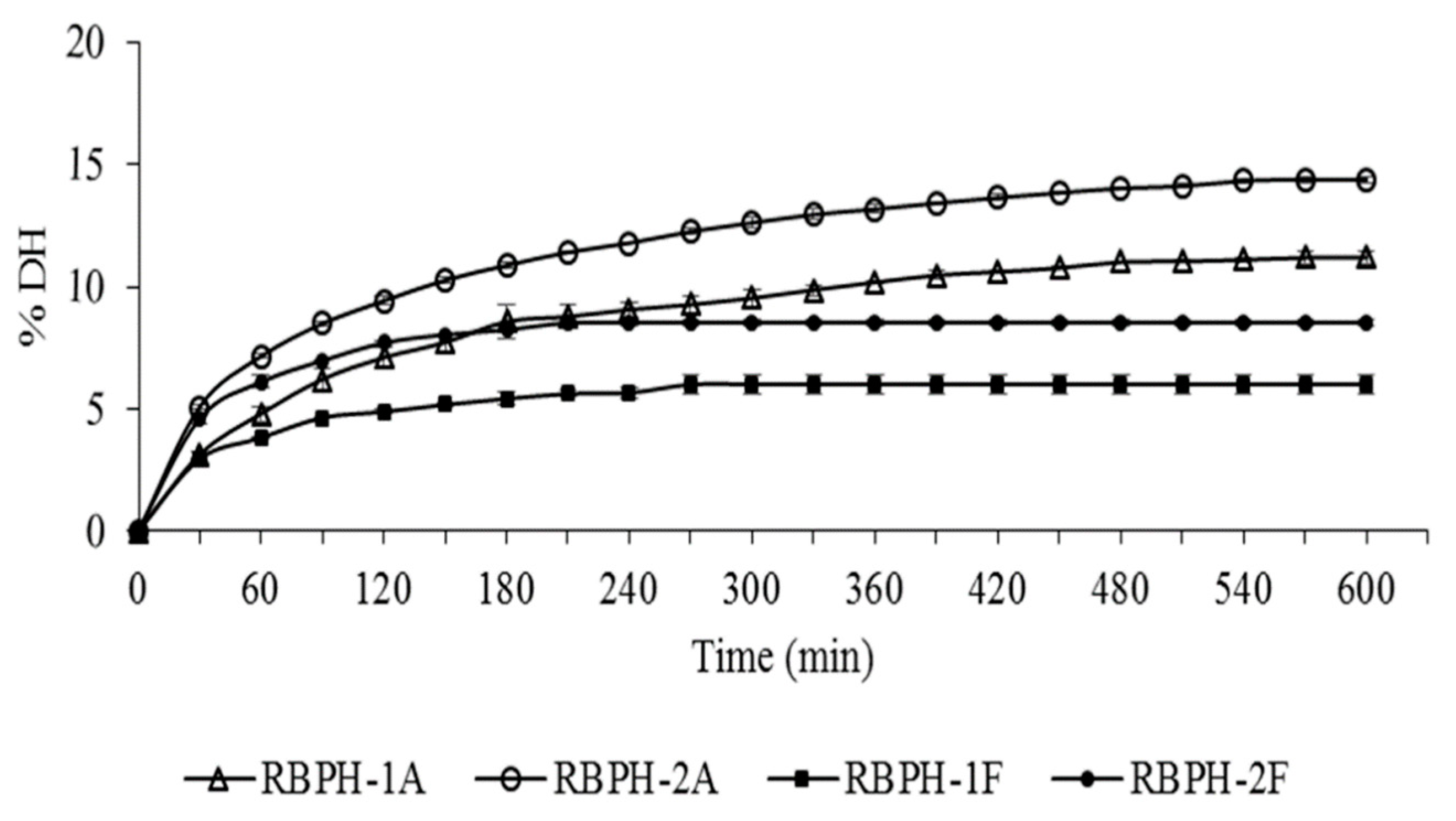
2.5. Degree of Hydrolysis Determination
2.6. In Vitro Assays
2.6.1. Oxygen Radical Absorbance Capacity (ORAC)
2.6.2. Reducing Power
2.6.3. Total Phenolic Compounds Analysis
2.6.4. Angiotensin-Converting Enzyme (ACE) Inhibitory Activity
2.7. In Vivo Assays
2.7.1. Animal Ethical Approval
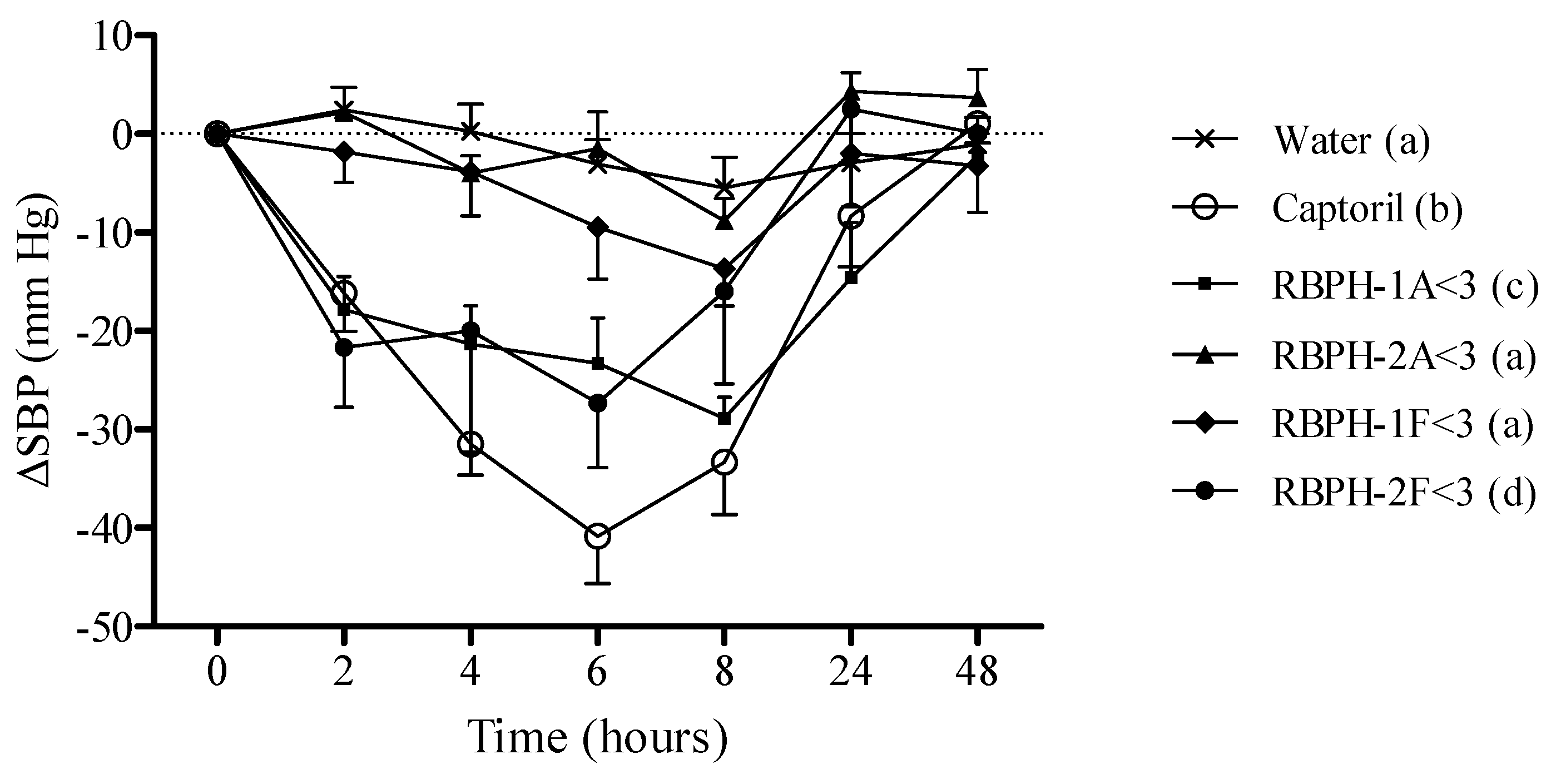
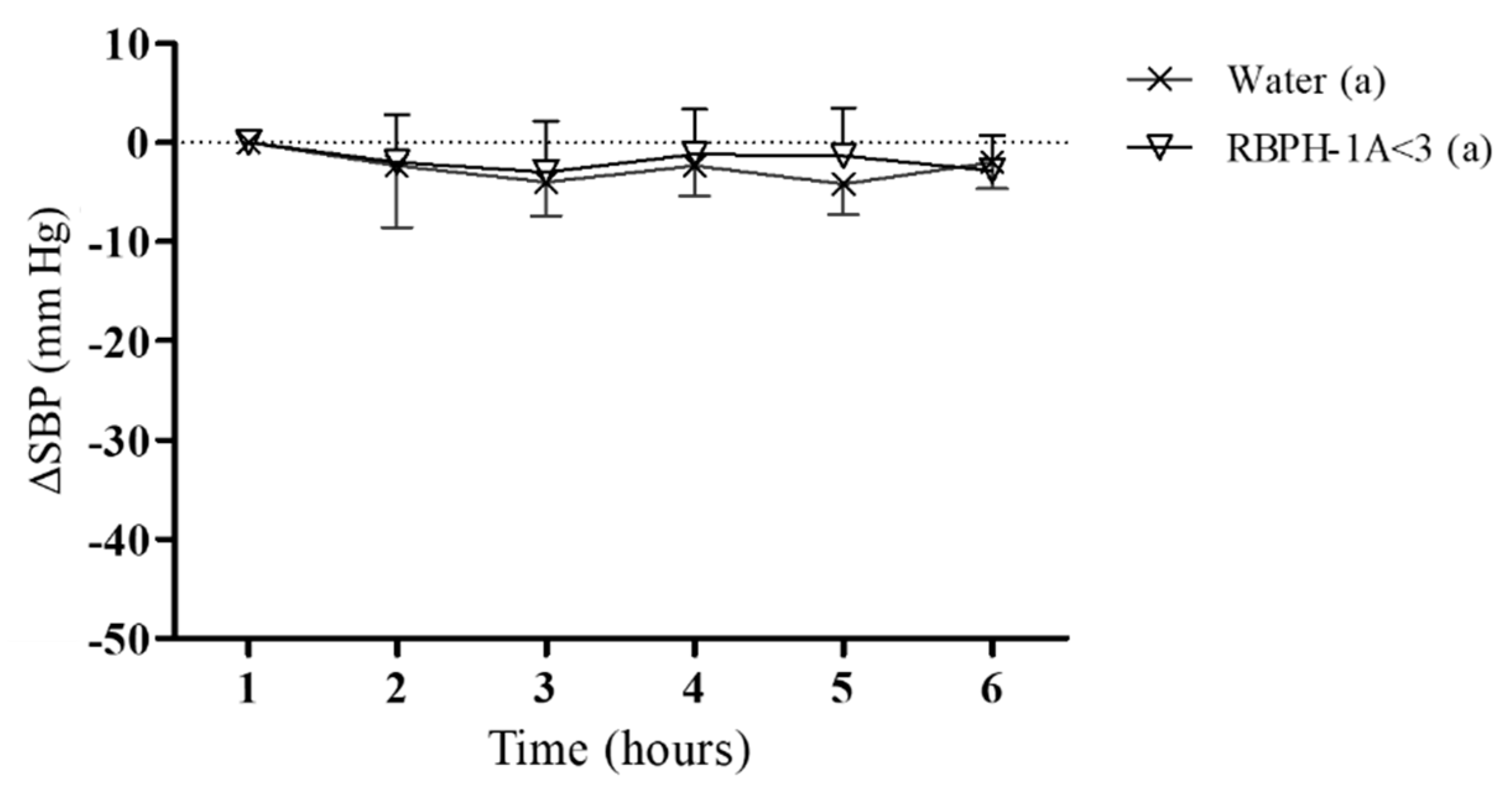
2.7.2. Experimental Procedure in Spontaneously Hypertensive Rats
2.8. Statistical Analysis of In Vitro and In Vivo Assays
3. Results and Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. World Health Organization. Q&As on hypertension. 2015. Available online: http://www.who.int/features/qa/82/en/ (accessed on 28 April 2020).
- Torruco-Uco, J.G.; Dominguéz-Magaña, M.A.; Martínez-Ayala, A.; Chel-Guerrero, L.A.; Betancur-Ancona, D.A. Péptidos antihipertensivos, una alternativa de tratamiento de origen natural: Una revisión. CYTA-J. Food 2008, 6, 158–168. [Google Scholar]
- Boschin, G.; Scigliuolo, G.M.; Resta, D.; Arnoldi, A. ACE-inhibitory activity of enzymatic protein hydrolysates from lupin and other legumes. Food Chem. 2014, 145, 34–40. [Google Scholar] [CrossRef]
- Csányi, G.; Miller, F.J. Oxidative stress in cardiovascular diseases. Int. J. Mol. Sci. 2014, 15, 6002–6008. [Google Scholar] [CrossRef]
- Wan, W.A.A.Q.I.; Hamid, A.A.; Abd-Aziz, S.; Muhamad SK, S.; Saari, N. Preparation of bioactive peptides with high angiotensin converting enzyme inhibitory activity from winged bean [Psophocarpus tetragonolobus (L.) DC.] seed. J. Food Sci. Technol. 2014, 51, 3658–3668. [Google Scholar]
- Uraipong, C.; Zhao, J. Rice bran protein hydrolysates exhibit strong in vitro α-amylase, β-glucosidase and ACE-inhibition activities. J. Sci. Food Agric. 2016, 96, 1101–1110. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Martinez, M.; Winkler, R.; García-Lara, S. Preventive and therapeutic potential of peptides from cereals against cancer. J. Proteome 2014, 111, 165–183. [Google Scholar] [CrossRef] [PubMed]
- Agyei, D. Bioactive Proteins and Peptides from Soybeans. Recent Pat. Food Nut. Agric. 2015, 7, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Minh, N.P. Alcalase hydrolysis of bioactive peptides from soybean. Int. J. Pure App. Biosci. 2015, 3, 19–29. [Google Scholar]
- Wattanasiritham, L.; Theerakulkait, C.; Wickramasekara, S.; Maier, C.S.; Stevens, J.F. Isolation and identification of antioxidant peptides from enzymatically hydrolyzed rice bran protein. Food Chem. 2016, 192, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Nagendra Prasad, M.N.; Sanjay, K.R.; Shravya Khatokar, M.; Vismaya, M.N.; Nanjunda Swamy, S. Health Benefits of Rice Bran—A Review. J. Nutr. Food Sci. 2011, 1, 1–7. [Google Scholar] [CrossRef]
- Cao, X.; Wen, H.; Li, C.; Gu, Z. Differences in functional properties and biochemical characteristics of congenetic rice proteins. J. Cereal Sci. 2009, 50, 184–189. [Google Scholar] [CrossRef]
- Chanput, W.; Theerakulkait, C.; Nakai, S. Antioxidative properties of partially purified barley hordein, rice bran protein fractions and their hydrolysates. J. Cereal Sci. 2009, 49, 422–428. [Google Scholar] [CrossRef]
- Liu, Y.Q.; Strappe, P.; Shang, W.T.; Zhou, Z.K. Functional peptides derived from rice bran proteins. Crit. Rev. Food Sci. Nutr. 2019, 59, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Piotrowicz, I.B.B.; Salas-Mellado, M.M. Protein concentrates from defatted rice bran: Preparation and characterization. Food Sci. Technol. 2017, 37, 165–172. [Google Scholar] [CrossRef][Green Version]
- Adler-Nissen, J. Enzymic Hydrolysis of Food Proteins; Elsevier Applied Science Publishers LTD: New York, NY, USA, 1986. [Google Scholar]
- Garcés-Rimón, M.; López-Expósito, I.; López-Fandiño, R.; Miguel, M. Egg white hydrolysates with in vitro biological multiactivities to control complications associated with the metabolic syndrome. Eur. Food Res. Technol. 2015, 242, 61–69. [Google Scholar] [CrossRef]
- Oyaizu, M. Antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Buñag, R.D.; Butterfield, J. Tail-cuff blood-pressure measurement without external preheating in awake rats. Hypertension 1982, 4, 898–903. [Google Scholar] [CrossRef]
- Tavano, O.L. Protein hydrolysis using proteases: An important tool for food biotechnology. J. Mol. Catal. B-Enzym. 2013, 90, 1–11. [Google Scholar] [CrossRef]
- Thamnarathip, P.; Jangchud, K.; Jangchud, A.; Vardhanabhuti, B. Functional properties of protein hydrolysates from Rice berry rice bran. Int. J. Food Sci. Technol. 2016, 51, 1110–1119. [Google Scholar] [CrossRef]
- Maragno, V.P.; Kuhn, R.C. Estabilidade do farelo de arroz sob diferentes tratamentos: Micro-ondas, peletização e desengorduramento. Cienc. Biol. Saude 2013, 15, 47–53. [Google Scholar]
- Santé-Lhoutellier, V.; Astruc, T.; Marinova, P.; Greve, E.; Gatellier, P. Effect of Meat Cooking on Physicochemical State and in Vitro Digestibility of Myofibrillar Proteins. J. Agric. Food Chem. 2008, 56, 1488–1494. [Google Scholar] [CrossRef] [PubMed]
- Sbroggio, M.F.; Montilha, M.S.; Figueiredo, V.R.G.; Georgetti, S.R.; Kurozawa, L.E. Influence of the degree of hydrolysis and type of enzyme on antioxidant activity of okara protein hydrolysates. Food Sci. Technol. 2016, 36, 375–381. [Google Scholar] [CrossRef]
- Pedroche, J.; Yust, M.M.; Girón-Calle, J.; Alaiz, M.; Millán, F.; Vioque, J. Utilization of chickpea protein isolate for production of peptides with angiotensin I-converting enzyme (ACE)-inhibitory activity. J. Sci. Food Agric. 2002, 82, 960–965. [Google Scholar] [CrossRef]
- Klompong, V.; Benjakul, S.; Kantachote, D.; Shahidi, F. Antioxidative activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as influenced by the degree of hydrolysis and enzyme type. Food Chem. 2007, 102, 1317–1327. [Google Scholar] [CrossRef]
- Samaranayaka, A.G.P.; Li-Chan, E.C.Y. Food-Derived Peptidic Antioxidants: A Review of Their Production, Assessment, and Potential Applications. J. Funct. Foods 2011, 3, 229–254. [Google Scholar] [CrossRef]
- Rajapakse, N.; Mendis, E.; Jung, W.K.; Je, J.Y.; Kim, S.K. Purification of a radical scavenging peptide from fermented mussel sauce and its antioxidant properties. Food Res. Int. 2005, 38, 175–182. [Google Scholar] [CrossRef]
- Qian, Z.J.; Jung, W.K.; Kim, S.K. Free radical scavenging activity of a novel antioxidative peptide purified from hydrolysate of bullfrog skin, Rana catesbeiana Shaw. Bioresour. Technol. 2008, 99, 1690–1698. [Google Scholar] [CrossRef]
- Matoba, T.; Kenkyu, D.T. How does the radical-scavenging activity of soy protein food change during heating. Daizu Tanpakushitsu Kenkyu 2002, 5, 47–50. [Google Scholar]
- Prior, R.L.; Wu, X.L.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolic in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Sun, T.; Tanumihardjo, S.A. An Integrated approach to evaluate food antioxidant capacity. J. Food Sci. 2007, 72, 159–165. [Google Scholar] [CrossRef]
- Frankel, E.N.; Meyer, A.S. The problems of using one dimensional methods to evaluate multifunctional food and biological antioxidants. J. Sci. Food Agric. 2000, 80, 1925–1941. [Google Scholar] [CrossRef]
- Ou, B.; Huang, D.; Hampsch-Woodill, M.; Flanagan, J.A.; Deemer, E.K. Analysis of Antioxidant Activities of Common Vegetables Employing Oxygen Radical Absorbance Capacity (ORAC) and Ferric Reducing Antioxidant Power (FRAP) Assays: A Comparative Study. J. Agric. Food Chem. 2002, 50, 3122–3128. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.J.; Wang, J.; Zhang, B.H.; Zhang, H. Antioxidant activities of the fractionated protein hydrolysates from heat stable defatted rice bran. Int. J. Food Sci. Technol. 2014, 49, 1330–1336. [Google Scholar] [CrossRef]
- Adom, K.K.; Liu, R.H. Antioxidant activity of grains. J. Agric. Food Chem. 2002, 50, 6182–6187. [Google Scholar] [CrossRef]
- Heuberger, A.L.; Lewis, M.R.; Chen, M.H.; Brick, M.A.; Leach, J.E.; Ryan, E.P. Metabolomic and functional genomic analyses reveal varietal differences in bioactive compounds of cooked rice. PLoS ONE 2010, 5, e12915. [Google Scholar] [CrossRef]
- Li, G.; Qu, M.; Wan, J.; You, J. Antihypertensive effect of rice protein hydrolysate with in vitro angiotensin I-converting enzyme inhibitory activity in spontaneously hypertensive rats. Asia Pac. J. Clin. Nutr. 2007, 16, 275–280. [Google Scholar]
- Chen, J.; Liu, S.; Ye, R.; Cai, G.; Ji, B.; Wu, Y. Angiotensin-I converting enzyme (ACE) inhibitory tripeptides from rice protein hydrolysate: Purification and characterization. J. Funct. Foods 2013, 5, 1684–1692. [Google Scholar] [CrossRef]
- Wang, X.; Chen, H.; Fu, X.; Li, S.; Wei, J. A novel antioxidant and ACE inhibitory peptide from rice bran protein: Biochemical characterization and molecular docking study. LTW-Food Sci. Technol. 2017, 75, 93–99. [Google Scholar] [CrossRef]
- Sanjuliani, A.F. Fisiopatologia da hipertensão arterial: Conceitos teóricos úteis para a prática clínica. Rev. Soc. Cardiol. Est. 2002, 2, 210–218. [Google Scholar]
- Fazan, R.; Silva, V.J.D.; Salgado, H.C. Modelos de hipertensão arterial. Rev. Bras Hipertens 2001, 8, 19–29. [Google Scholar]
- Miguel, M.; Gómez-Ruiz, J.Á.; Recio, I.; Aleixandre, A. Changes in arterial blood pressure after single oral administration of milk-casein-derived peptides in spontaneously hypertensive rats. Mol. Nutr. Food Res. 2010, 54, 1422–1427. [Google Scholar] [CrossRef] [PubMed]
- Iwaniak, A.; Minkiewicz, P.; Darewicz, M. Food-originating ace inhibitors, including antihypertensive peptides, as preventive food components in blood pressure reduction. Comp. Rev. Food Sci. F 2014, 13, 114–134. [Google Scholar] [CrossRef]
- Okamoto, K.; Aoki, K. Development of a strain of spontaneously hypertensive rats. Jpn. Circ. J. 1963, 27, 282–293. [Google Scholar] [CrossRef]
- Fujita, H.; Yoshikawa, M. LKPNM: A prodrug-type ACE-inhibitory peptide derived from fish protein. Immunopharmacology 1999, 44, 123–127. [Google Scholar] [CrossRef]
- Miguel, M.; Alonso, M.J.; Salaices, M.; Aleixandre, A.; López-Fandiño, R. Antihypertensive, ACE-inhibitory and vasodilator properties of an egg white hydrolysate: Effect of a simulated intestinal digestion. Food Chem. 2007, 104, 163–168. [Google Scholar] [CrossRef]
- Chen, G.W.; Tsai, J.S.; Sun Pan, B. Purification of angiotensin I-converting enzyme inhibitory peptides and antihypertensive effect of milk produced by protease-facilitated lactic fermentation. Int. Dairy J. 2007, 17, 641–647. [Google Scholar] [CrossRef]
- Leoncini, G.; Viazzi, F.; Storace, G.; Deferrari, G.; Pontremoli, R. Blood pressure variability and multiple organ damage in primary hypertension. J. Hum. Hypertens. 2013, 27, 663–670. [Google Scholar] [CrossRef]
- Miguel, M. Actividad Antihipertensiva y Antioxidante de Péptidos Derivados de Proteínas de Huevo. Ph.D. Thesis, Universidad Autónoma de Madrid, Madrid, Spain, 2004. 189 f. [Google Scholar]
- Liu, F.; Ma, C.; Gao, Y.; McClements, D.J. Food-Grade Covalent Complexes and Their Application as Nutraceutical Delivery Systems: A Review. Comp. Rev. Food Sci. F 2017, 16, 76–95. [Google Scholar] [CrossRef]
| Sample | ORAC (µmol eq Trolox/g Sample) | Reducing Power ABS (700 nm) |
|---|---|---|
| DRBPC-1 | 111.15 ± 5.30 g | 0.32 ± 0.03 c |
| DRBPC-2 | 256.50 ± 10.26 f | 0.55 ± 0.02 b |
| RBPH-1A | 385.81 ± 12.02 d,e | 0.33 ± 0.02 c |
| RBPH-2A | 508.02 ± 14.42 d | 0.51 ± 0.03 b |
| RBPH-1F | 273.88 ± 13.99 e,f | 0.33 ± 0.02 c |
| RBPH-2F | 391.68 ± 53.36 d,e | 0.53 ± 0.03 b |
| RBPH-1A<3 | 886.76 ± 32.13 b | 0.47 ± 0.01 b |
| RBPH-2A<3 | 1029.03 ± 83.72 a | 0.53 ± 0.01 b |
| RBPH-1F<3 | 720.03 ± 20.06 c | 0.96 ± 0.07 a |
| RBPH-2F<3 | 1040.27 ± 73.16 a | 0.88 ± 0.01 a |
| Sample | mg GAE/g Sample |
|---|---|
| DRBPC-1 | 4.78 ± 0.63 e |
| DRBPC-2 | 7.90 ± 0.52 d,e |
| RBPH-1A | 15.81 ± 0.65 c |
| RBPH-2A | 19.38 ± 1.33 c |
| RBPH-1F | 9.43 ± 0.87 d |
| RBPH-2F | 15.50 ± 1.75 c |
| RBPH-1A<3 | 36.17 ± 1.90 a |
| RBPH-2A<3 | 37.24 ± 2.58 a |
| RBPH-1F<3 | 29.01 ± 1.80 b |
| RBPH-2F<3 | 34.23 ± 0.77 a |
| Sample | IC50 |
|---|---|
| (mg Sample/mL) | |
| RBPH-1A<3 | 0.15 ± 0.02 d |
| RBPH-2A<3 | 0.32 ± 0.09 c |
| RBPH-1F<3 | 1.70 ± 0.16 a |
| RBPH-2F<3 | 1.42 ± 0.06 b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piotrowicz, I.B.B.; Garcés-Rimón, M.; Moreno-Fernández, S.; Aleixandre, A.; Salas-Mellado, M.; Miguel-Castro, M. Antioxidant, Angiotensin-Converting Enzyme Inhibitory Properties and Blood-Pressure-Lowering Effect of Rice Bran Protein Hydrolysates. Foods 2020, 9, 812. https://doi.org/10.3390/foods9060812
Piotrowicz IBB, Garcés-Rimón M, Moreno-Fernández S, Aleixandre A, Salas-Mellado M, Miguel-Castro M. Antioxidant, Angiotensin-Converting Enzyme Inhibitory Properties and Blood-Pressure-Lowering Effect of Rice Bran Protein Hydrolysates. Foods. 2020; 9(6):812. https://doi.org/10.3390/foods9060812
Chicago/Turabian StylePiotrowicz, Inajara Beatriz Brose, Marta Garcés-Rimón, Silvia Moreno-Fernández, Amaya Aleixandre, Myriam Salas-Mellado, and Marta Miguel-Castro. 2020. "Antioxidant, Angiotensin-Converting Enzyme Inhibitory Properties and Blood-Pressure-Lowering Effect of Rice Bran Protein Hydrolysates" Foods 9, no. 6: 812. https://doi.org/10.3390/foods9060812
APA StylePiotrowicz, I. B. B., Garcés-Rimón, M., Moreno-Fernández, S., Aleixandre, A., Salas-Mellado, M., & Miguel-Castro, M. (2020). Antioxidant, Angiotensin-Converting Enzyme Inhibitory Properties and Blood-Pressure-Lowering Effect of Rice Bran Protein Hydrolysates. Foods, 9(6), 812. https://doi.org/10.3390/foods9060812






