Chemical Characterization and Bioactive Properties of Different Extracts from Fibigia clypeata, an Unexplored Plant Food
Abstract
1. Introduction
2. Material and Methods
2.1. Plant Material and Preparation of Extracts
2.2. Profiling of Bioactive Compounds in the Different Extracts
2.3. Determination of Antioxidant and Enzyme Inhibitory Effects
2.4. Cell Assays
2.4.1. Cell Lines and Reagents
2.4.2. Cell Cultures
2.4.3. Evaluation of Cytotoxic Properties
2.5. Statistical Analysis
3. Results and Discussion
3.1. Phytochemical Profile Determination
3.2. Enzyme Inhibition Activity
3.3. In Vitro Antioxidant Activity of the Tested Extracts
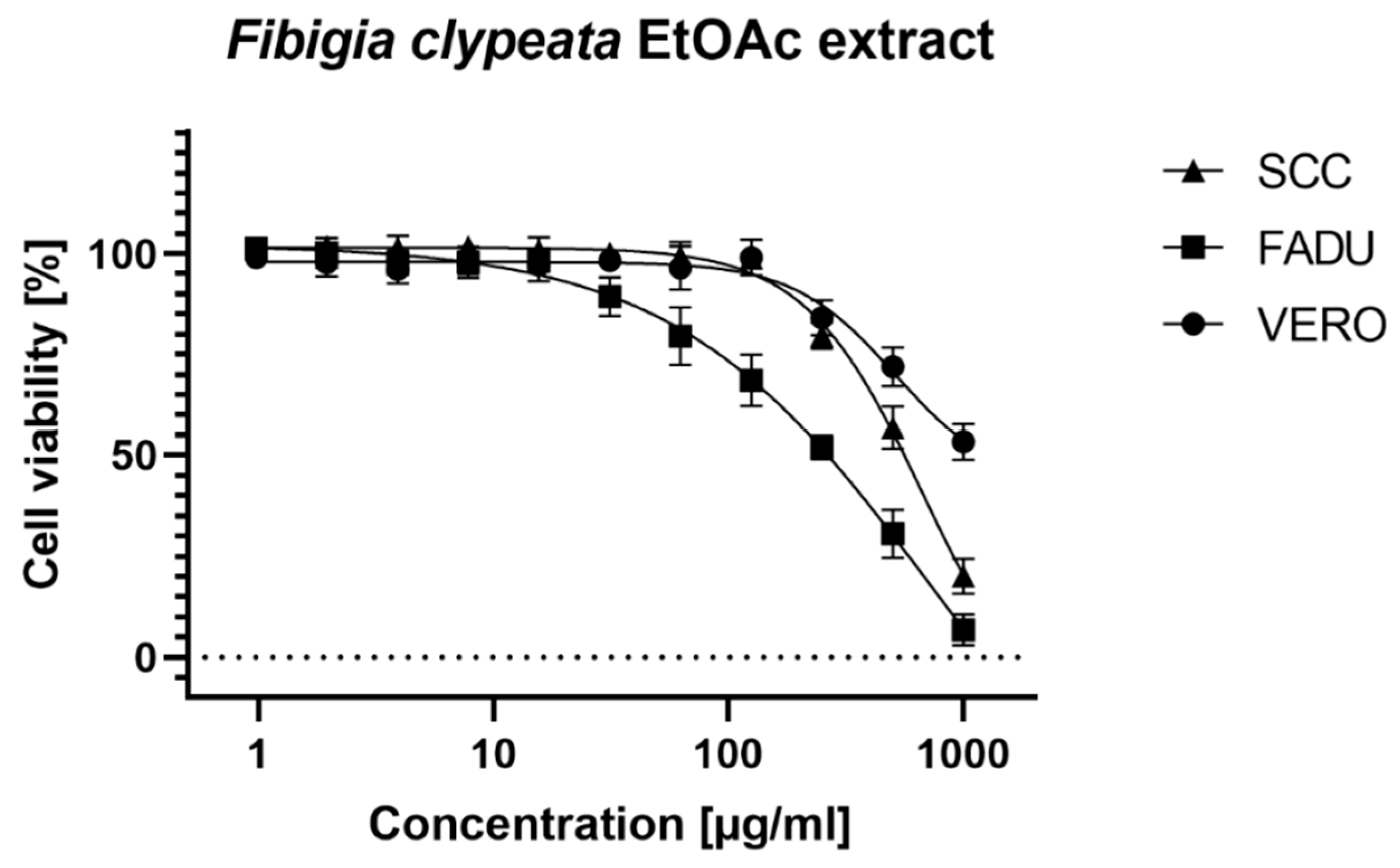
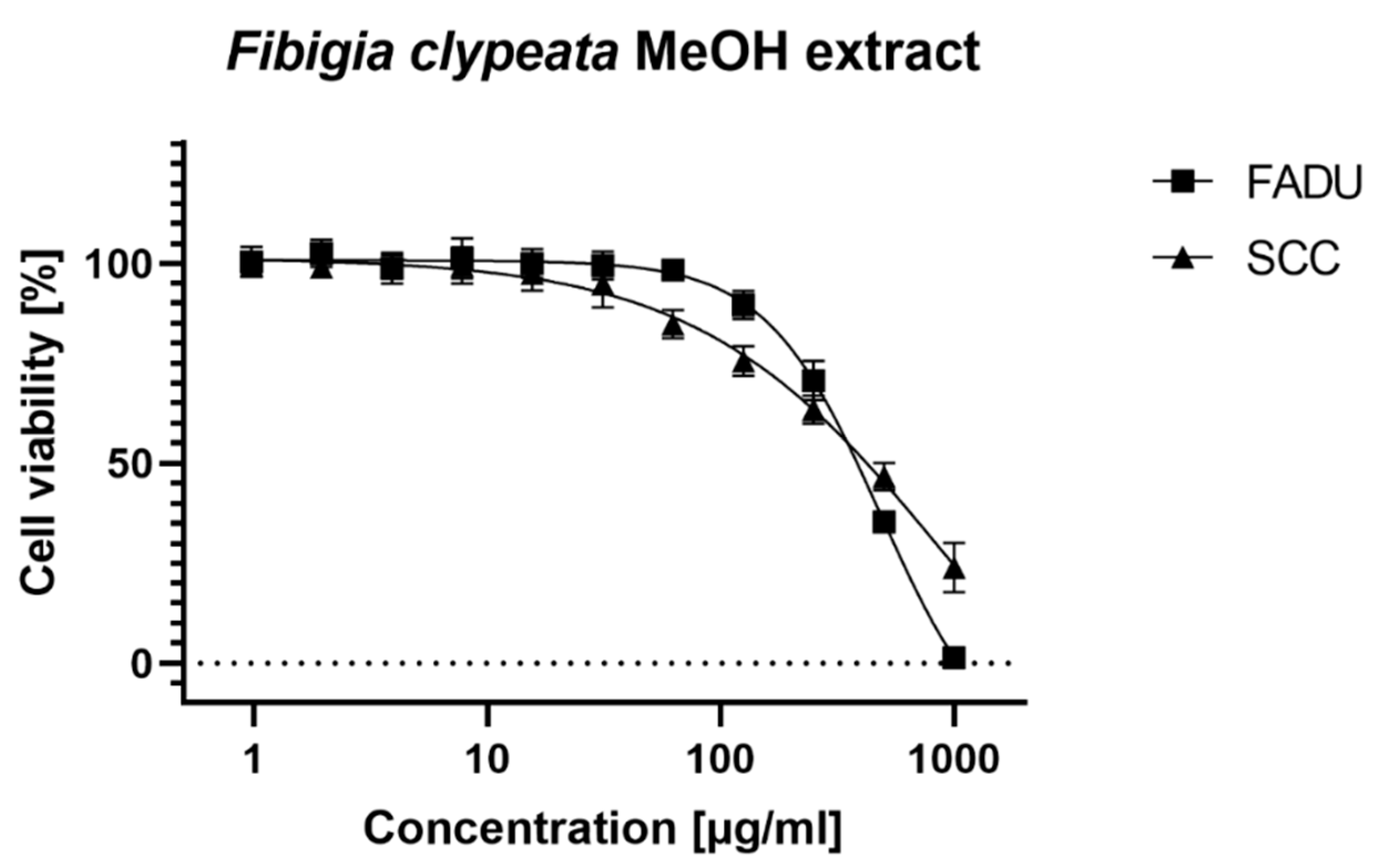
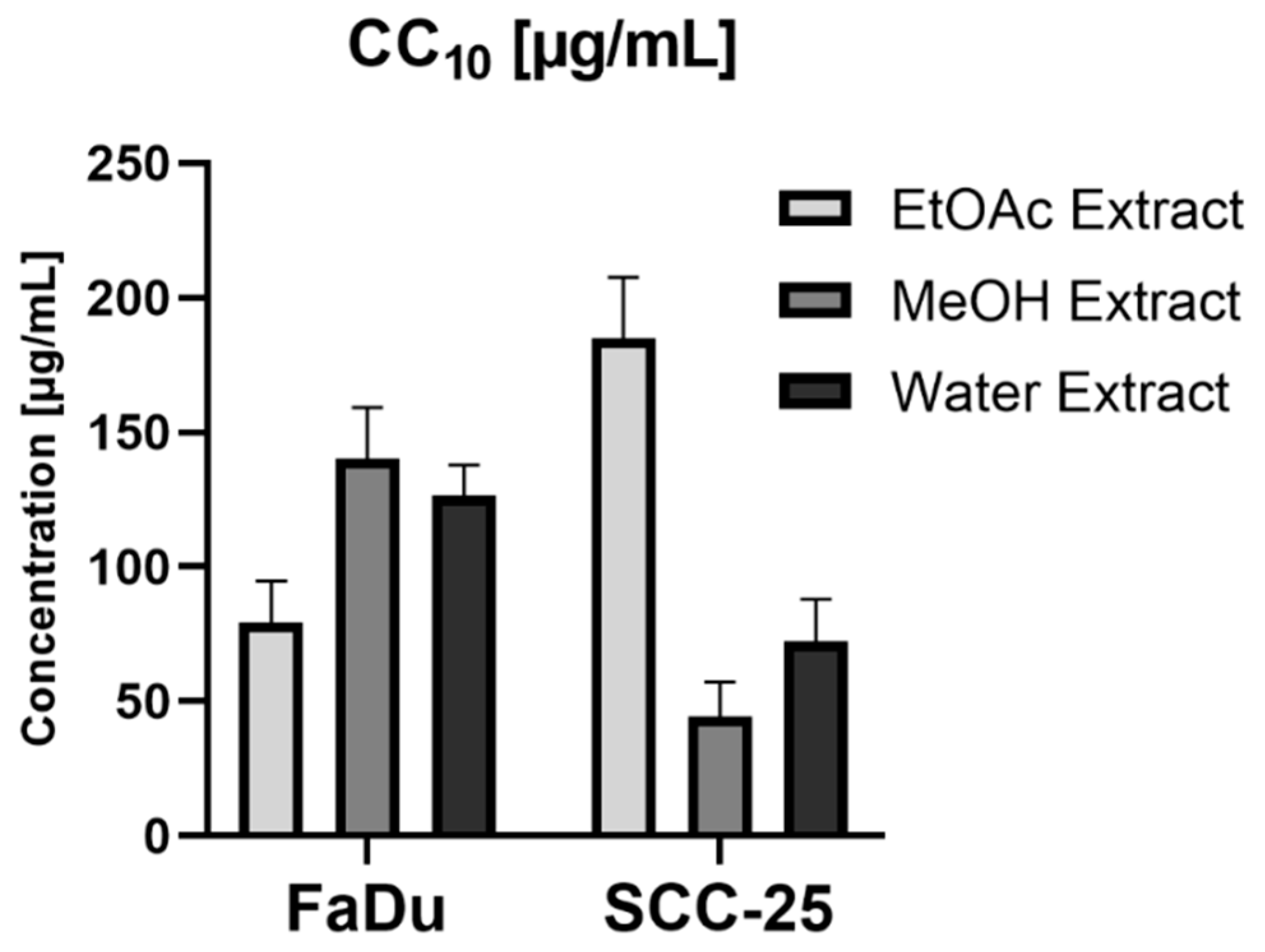
3.4. Cell Assays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Borbone, N.; Borrelli, F.; Montesano, D.; Izzo, A.A.; De Marino, S.; Capasso, R.; Zollo, F. Identification of a new sesquiterpene polyol ester from Celastrus paniculatus. Planta Med. 2007, 73, 792–794. [Google Scholar] [CrossRef]
- De Almeida, E.M.; Ferreira, H.J.; Alves, D.R.; da Silva, W.M.B. Therapeutic potential of medicinal plants indicated by the Brazilian public health system in treating the collateral effects induced by chemotherapy, radiotherapy, and chemoradiotherapy: A systematic review. Complement. Ther. Med. 2020, 49, 102293. [Google Scholar] [CrossRef]
- Rocchetti, G.; Giuberti, G.; Busconi, M.; Marocco, A.; Trevisan, M.; Lucini, L. Pigmented sorghum polyphenols as potential inhibitors of starch digestibility: An in vitro study combining starch digestion and untargeted metabolomics. Food Chem. 2020, 312, 126077. [Google Scholar] [CrossRef]
- Senizza, B.; Rocchetti, G.; Okur, M.A.; Zengin, G.; Yıldıztugay, E.; Ak, G.; Montesano, D.; Lucini, L. Phytochemical Profile and Biological Properties of Colchicum triphyllum (Meadow Saffron). Foods 2020, 9, 457. [Google Scholar] [CrossRef]
- Borrelli, F.; Borbone, N.; Capasso, R.; Montesano, D.; De Marino, S.; Aviello, G.; Aprea, G.; Masone, S.; Izzo, A.A. Potent relaxant effect of a Celastrus paniculatus extract in the rat and human ileum. J. Ethnopharmacol. 2009, 122, 434–438. [Google Scholar] [CrossRef]
- Rocchetti, G.; Lucini, L.; Ahmed, S.R.; Saber, F.R. In vitro cytotoxic activity of six Syzygium leaf extracts as related to their phenolic profiles: An untargeted UHPLC-QTOF-MS approach. Food Res. Int. 2019, 126, 108715. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, G.; Pagnossa, J.P.; Blasi, F.; Cossignani, L.; Piccoli, R.H.; Zengin, G.; Montesano, D.; Cocconcelli, P.S.; Lucini, L. Phenolic profiling and in vitro bioactivity of Moringa oleifera leaves as affected by different extraction solvents. Food Res. Int. 2020, 127, 108712. [Google Scholar] [CrossRef] [PubMed]
- Güneş, S.; Savran, A.; Paksoy, M.Y.; Koşar, M.; Çakılcıoğlu, U. Ethnopharmacological survey of medicinal plants in Karaisalı and its surrounding (Adana-Turkey). J. Herb. Med. 2017, 8, 68–75. [Google Scholar] [CrossRef]
- Baydoun, S.; Chalak, L.; Dalleh, H.; Arnold, N. Ethnopharmacological survey of medicinal plants used in traditional medicine by the communities of Mount Hermon, Lebanon. J. Ethnopharmacol. 2015, 173, 139–156. [Google Scholar] [CrossRef]
- Mosaddegh, M.; Naghibi, F.; Moazzeni, H.; Pirani, A.; Esmaeili, S. Ethnobotanical survey of herbal remedies traditionally used in Kohghiluyeh va Boyer Ahmad province of Iran. J. Ethnopharmacol. 2012, 141, 80–95. [Google Scholar] [CrossRef]
- Di Giorgio, C.; Delmas, F.; Tueni, M.; Cheble, E.; Khalil, T.; Balansard, G. Alternative and Complementary Antileishmanial Treatments: Assessment of the Antileishmanial Activity of 27 Lebanese Plants, Including 11 Endemic Species. J. Altern. Complement. Med. 2008, 14, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L.D.; Hidalgo, F.J.; Miyashita, K.; van Camp, J.; Alasalvar, C.; Ismail, A.B. Antioxidant activity, total phenolics and flavonoids contents: Should we ban in vitro screening methods? Food Chem. 2018, 264, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Zengin, G.; Aktumsek, A. Investigation of antioxidant potentials of solvent extracts from different anatomical parts of Asphodeline anatolica E. Tuzlaci: An endemic plant to Turkey. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Zengin, G.; Sieniawska, E.; Senkardes, I.; Picot-Allain, M.C.N.; Sinan, K.I.; Mahomoodally, M.F. Antioxidant abilities, key enzyme inhibitory potential and phytochemical profile of Tanacetum poteriifolium Grierson. Ind. Crops Prod. 2019, 140, 111629. [Google Scholar] [CrossRef]
- Grochowski, D.M.; Uysal, S.; Aktumsek, A.; Granica, S.; Zengin, G.; Ceylan, R.; Locatelli, M.; Tomczyk, M. In vitro enzyme inhibitory properties, antioxidant activities, and phytochemical profile of Potentilla thuringiaca. Phytochem. Lett. 2017, 20, 365–372. [Google Scholar] [CrossRef]
- Zengin, G.; Lobine, D.; Mollica, A.; Locatelli, M.; Carradori, S.; Mahomoodally, M.F. Multiple pharmacological approaches on Fibigia eriocarpa extracts by in vitro and computational assays. Fundam. Clin. Pharmacol. 2018, 32, 400–413. [Google Scholar] [CrossRef] [PubMed]
- El Atki, Y.; Aouam, I.; El kamari, F.; Taroq, A.; Lyoussi, B.; Taleb, M.; Abdellaoui, A. Total phenolic and flavonoid contents and antioxidant activities of extracts from Teucrium polium growing wild in Morocco. Mater. Today Proc. 2019, 13, 777–783. [Google Scholar] [CrossRef]
- Ahmed, D.; Khan, M.M.; Saeed, R. Comparative Analysis of Phenolics, Flavonoids, and Antioxidant and Antibacterial Potential of Methanolic, Hexanic and Aqueous Extracts from Adiantum caudatum Leaves. Antioxidants 2015, 4, 394–409. [Google Scholar] [CrossRef]
- Chávez-González, M.L.; Sepúlveda, L.; Verma, D.K.; Luna-García, H.A.; Rodríguez-Durán, L.V.; Ilina, A.; Aguilar, C.N. Conventional and Emerging Extraction Processes of Flavonoids. Processes 2020, 8, 434. [Google Scholar] [CrossRef]
- Tang, J.; Dunshea, F.R.; Suleria, H.A. LC-ESI-QTOF/MS Characterization of Phenolic Compounds from Medicinal Plants (Hops and Juniper Berries) and Their Antioxidant Activity. Foods 2020, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.R. Application of HPLC and ESI-MS techniques in the analysis of phenolic acids and flavonoids from green leafy vegetables (GLVs). J. Pharm. Anal. 2017, 7, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Bennett, R.N.; Mellon, F.A.; Kroon, P.A. Screening crucifer seeds as sources of specific intact glucosinolates using ion-pair high-performance liquid chromatography negative ion electrospray mass spectrometry. J. Agric. Food Chem. 2004, 52, 428–438. [Google Scholar] [CrossRef] [PubMed]
- Metlin Database. Available online: https://metlin.scripps.edu/landing_page.php?pgcontent=mainPage (accessed on 4 January 2020).
- Linić, I.; Šamec, D.; Grúz, J.; Vujčić Bok, V.; Strnad, M.; Salopek-Sondi, B. Involvement of phenolic acids in short-term adaptation to salinity stress is species-specific among brassicaceae. Plants 2019, 8, 155. [Google Scholar] [CrossRef] [PubMed]
- Oszmiański, J.; Kolniak-Ostek, J.; Wojdyło, A. Application of ultra performance liquid chromatography-photodiode detector-quadrupole/time of flight-mass spectrometry (UPLC-PDA-Q/TOF-MS) method for the characterization of phenolic compounds of Lepidium sativum L. sprouts. Eur. Food Res. Technol. 2013, 236, 699–706. [Google Scholar] [CrossRef]
- Copeland, R.A.; Harpel, M.R.; Tummino, P.J. Targeting enzyme inhibitors in drug discovery. Expert Opin. Ther. Targets 2007, 11, 967–978. [Google Scholar] [CrossRef]
- World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/dementia (accessed on 31 December 2019).
- Alzheimer Association. 2019 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2019, 15, 321–387. [Google Scholar] [CrossRef]
- Sabbagh, M.N.; Hendrix, S.; Harrison, J.E. FDA position statement “Early Alzheimer’s disease: Developing drugs for treatment, Guidance for Industry”. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2019, 5, 13–19. [Google Scholar] [CrossRef]
- Li, Q.; Yang, H.; Chen, Y.; Sun, H. Recent progress in the identification of selective butyrylcholinesterase inhibitors for Alzheimer’s disease. Eur. J. Med. Chem. 2017, 132, 294–309. [Google Scholar] [CrossRef]
- Mesulam, M.M.; Guillozet, A.; Shaw, P.; Levey, A.; Duysen, E.G.; Lockridge, O. Acetylcholinesterase knockouts establish central cholinergic pathways and can use butyrylcholinesterase to hydrolyze acetylcholine. Neuroscience 2002, 110, 627–639. [Google Scholar] [CrossRef]
- Altamura, C.; Ventriglia, M.; Martini, M.G.; Montesano, D.; Errante, Y.; Piscitelli, F.; Scrascia, F.; Quattrocchi, C.; Palazzo, P.; Seccia, S. Elevation of plasma 2-arachidonoylglycerol levels in alzheimer’s disease patients as a potential protective mechanism against neurodegenerative decline. J. Alzheimer’s Dis. 2015, 46, 497–506. [Google Scholar] [CrossRef]
- Kirkan, B.; Sarikurkcu, C.; Ozer, M.S.; Cengiz, M.; Atılgan, N.; Ceylan, O.; Tepe, B. Phenolic profile, antioxidant and enzyme inhibitory potential of Onosma tauricum var. tauricum. Ind. Crops Prod. 2018, 125, 549–555. [Google Scholar] [CrossRef]
- Sut, S.; Dall’Acqua, S.; Uysal, S.; Zengin, G.; Aktumsek, A.; Picot-Allain, C.; Mahomoodally, F. LC-MS, NMR fingerprint of Potentilla argentea and Potentilla recta extracts and their in vitro biopharmaceutical assessment. Ind. Crops Prod. 2019, 131, 125–133. [Google Scholar] [CrossRef]
- Elufioye, T.O.; Chinaka, C.G.; Oyedeji, A.O. Antioxidant and Anticholinesterase Activities of Macrosphyra Longistyla (DC) Hiern Relevant in the Management of Alzheimer’s Disease. Antioxidants 2019, 8, 400. [Google Scholar] [CrossRef] [PubMed]
- Farràs, A.; Cásedas, G.; Les, F.; Terrado, E.M.; Mitjans, M.; López, V. Evaluation of anti-tyrosinase and antioxidant properties of four fern species for potential cosmetic applications. Forests 2019, 10, 179. [Google Scholar] [CrossRef]
- Tlili, N.; Kirkan, B.; Sarikurkcu, C. LC–ESI–MS/MS characterization, antioxidant power and inhibitory effects on α-amylase and tyrosinase of bioactive compounds from hulls of Amygdalus communis: The influence of the extracting solvents. Ind. Crops Prod. 2019, 128, 147–152. [Google Scholar] [CrossRef]
- He, L.; Li, H.T.; Guo, S.W.; Liu, L.F.; Qiu, J.B.; Li, F.; Cai, B.C. Inhibitory effects of sinapine on activity of acetylcholinesterase in cerebral homogenate and blood serum of rats. Zhongguo Zhong Yao Za Zhi 2008, 33, 813–815. [Google Scholar]
- Santos-Sánchez, N.F.; Salas-Coronado, R.; Villanueva-Cañongo, C.; Hernández-Carlos, B. Antioxidant compounds and their antioxidant mechanism. In Antioxidants; IntechOpen: London, UK, 2019. [Google Scholar]
- Mahomoodally, M.F.; Zengin, G.; Aumeeruddy, M.Z.; Sezgin, M.; Aktumsek, A. Phytochemical profile and antioxidant properties of two Brassicaceae species: Cardaria draba subsp. draba and Descurainia sophia. Biocatal. Agric. Biotechnol. 2018, 16, 453–458. [Google Scholar] [CrossRef]
- Wang, L.-S.; Sun, X.-D.; Cao, Y.; Wang, L.; Li, F.-J.; Wang, Y.-F. Antioxidant and pro-oxidant properties of acylated pelargonidin derivatives extracted from red radish (Raphanus sativus var. niger, Brassicaceae). Food Chem. Toxicol. 2010, 48, 2712–2718. [Google Scholar] [CrossRef]
- Alqahtani, F.Y.; Aleanizy, F.S.; Mahmoud, A.Z.; Farshori, N.N.; Alfaraj, R.; Al-sheddi, E.S.; Alsarra, I.A. Chemical composition and antimicrobial, antioxidant, and anti-inflammatory activities of Lepidium sativum seed oil. Saudi J. Biol. Sci. 2019, 26, 1089–1092. [Google Scholar] [CrossRef] [PubMed]
- Chen, C. Sinapic Acid and Its Derivatives as Medicine in Oxidative Stress-Induced Diseases and Aging. Oxid. Med. Cell. Longev. 2016, 2016, 3571614. [Google Scholar] [CrossRef]
- Khan, F.A.; Maalik, A.; Murtaza, G. Inhibitory mechanism against oxidative stress of caffeic acid. J. Food Drug Anal. 2016, 24, 695–702. [Google Scholar] [CrossRef]
- Geran, R.; Greenberg, N.; Macdonald, M.; Schumacher, A. Protocols for screening chemical agents and natural products against animal tumors and other biological systems. Cancer Chemother. Rep. 1972, 3, 1–103. [Google Scholar]
- Łaska, G.; Sieniawska, E.; Świątek, Ł.; Zjawiony, J.; Khan, S.; Boguszewska, A.; Stocki, M.; Angielczyk, M.; Polz-Dacewicz, M. Phytochemistry and biological activities of Polemonium caeruleum L. Phytochem. Lett. 2019, 30, 314–323. [Google Scholar] [CrossRef]
- Srisawat, T.; Chumkaew, P.; Heed-Chim, W.; Sukpondma, Y.; Kanokwiroon, K. Phytochemical screening and cytotoxicity of crude extracts of Vatica diospyroides Symington Type LS. Trop. J. Pharm. Res. 2013, 12, 71–76. [Google Scholar] [CrossRef]
- Thienthiti, K.; Tuchinda, P.; Wongnoppavich, A.; Anantachoke, N.; Soonthornchareonnon, N. Cytotoxic effect of compounds isolated from Goniothalamus marcanii Craib stem barks. Pharm. Sci. Asia 2017, 44, 86–95. [Google Scholar] [CrossRef]

| Extracts | Total Phenolic Content (mg GAE/g) | Total Flavonoid Content (mg RE/g) |
|---|---|---|
| Ethyl acetate | 21.04 ± 0.68 a | 10.13 ± 0.39 b |
| Methanolic | 33.42 ± 3.79 b | 21.58 ± 0.16 c |
| Water | 24.43 ± 0.21 a | 5.81 ± 0.07 a |
| Compound | Tentative Identification | RT (Min) | Molecular Formula | Molecular Weight | [M-H] | Fragments (m/z) | Extracts | References |
|---|---|---|---|---|---|---|---|---|
| 1 | Caffeic acid-O-hexoside derivative | 1.725 | 388.1247 | 387.1247 | 341.1180; 179.0121 | EA, MeOH, Water | [20] | |
| 2 | Ethyl 2-hydroxy-3-(hydroxyphenyl)propanoate | 2.120 | C11H14O4 | 210.0051 | 209.0051 | 190.9851; 172.9663; 146.9663; 128.9377; 102.9125 | Water | [21] |
| 3 | Quinic acid | 2.773 | C7H12O6 | 192.0205 | 191.0205 | 173.0034; 127.0437; 111.0463 | MeOH, Water | [20] |
| 4 | p-Methoxy-2-hydroxy-2-phenylethyl glucosinolate | 4.825 | C16H23NO11S2 | 469.0042 | 468.0042 | 388.0458; 274.9727; 259.0033; 240.9840; 225.9687; 194.9916; 139.3081; 135.9009 | MeOH, Water | [22] |
| 5 | p-Methoxy-2-hydroxy-2-phenylethyl glucosinolate–desulfo | 5.018 | C16H23NO8S | 389.0527 | 388.0455 | 274.9727; 259.0033; 240.9840; 225.9687; 194.9916; 139.3081; 135.9009 | MeOH, Water | [22] |
| 6 | Dihydroxybenzoic acid hexoside | 15.386 | C13H16O9 | 316.0741 | 315.0741 | 153.0440; 109.0311 | Water | [20] |
| 7 | Dihydroxybenzoic acid hexoside | 17.781 | C13H16O9 | 316.0772 | 315.0772 | 153.0440; 109.0311 | Water | [20] |
| 8 | p-Coumaric acid ethyl ester derivative | 18.460 | 314.0613 | 313.0613 | 191.0031; 172.9730; 1146.9803; 128.9525 | Water | [20] | |
| 9 | p-Coumaric acid ethyl ester derivative | 19.622 | 314.0613 | 313.0613 | 191.9672; 172.9696; 146.9676; 128.9426 | Water | [20] | |
| 10 | 2-hydroxy-3-phenylpropanoic acid hexoside | 20.062 | C15H20O8 | 328.1154 | 327.1154 | 165.0021; 146.9606; 128.9594; 100.9476 | EA | [23] |
| 11 | Rhamnoside of ethyl 2-hydroxy-3-(hydroxyphenyl)propanoate isomer | 20.466 | C17H24O8 | 356.0780 | 355.0780 | 209.0037; 191.0097; 181.3211; 163.0022; 146.9911 | Water | [23] |
| 12 | Rhamnoside of ethyl 2-hydroxy-3-(hydroxyphenyl)propanoate isomer | 21.598 | C17H24O8 | 356.0780 | 355.0780 | 209.0401; 191.0162; 181.3317; 163.0043; 146.9924 | Water | [23] |
| 13 | Benzoic acid | 21.641 | C7H6O2 | 122.0369 | 121.0369 | - | EA | [23] |
| 14 | Feruloyl, ethyl 2-hydroxy-3-(hydroxyphenyl)propanoate | 22.395 | C17H22O10 | 386.0912 | 385.0912 | 209.0082; 190.9877; 146.9696; 129.9443 | Water | [23] |
| 15 | Sinapic acid hexoside derivative | 22.488 | 432.2047 | 431.2047 | 385.2424; 223.1204 | MeOH, Water | [24,25] | |
| 16 | p-Coumaric acid hexoside | 22.687 | C15H18O8 | 326.0987 | 325.0987 | 162.9889.;144.9626; 119.0003 | EA | [20] |
| 17 | p-Coumaric acid ethyl ester derivative | 23.203 | 314.0613 | 313.0613 | 190.9426; 172.9625; 146.9600; 128.9344; 120.9409 | Water | [20] | |
| 18 | p-coumaroyl acetic acid derivative | 23.407 | 388.1763 | 387.1763 | 207.0850; 163.0775 | MeOH | [21] | |
| 19 | Protocatechuic acid derivative | 23.613 | 432.2013 | 431.2013 | 385.1909; 223.1168; 153.0360 | EA | [21] | |
| 20 | Rhamnoside of ethyl 2-hydroxy-3-(hydroxyphenyl)propanoate | 23.976 | C17H24O8 | 356.0780 | 355.0780 | 209.0074; 190.9903 | Water | [23] |
| 21 | (Epi)catechin | 24.569 | C15H14O6 | 290.0440 | 289.0440 | 245.0665; 205.0202; 125.9405 | MeOH | [20] |
| 22 | Phenylacetic acid | 24.773 | C8H8O2 | 136.0057 | 135.0057 | - | EA | [20] |
| 23 | Feruloyl, ethyl 2-hydroxy-3-(hydroxyphenyl)propanoate | 24.994 | C17H22O10 | 386.0912 | 385.0912 | 223.01146; 209.0082; 190.9877; 146.9696; 129.9443 | Water | [23] |
| 24 | O-sinapoyl-glucose | 25.870 | C17H22O10 | 386.1268 | 385.1268 | 223.0670; | MeOH, Water | [25] |
| 25 | Vanilic acid derivative hexoside | 26.244 | 508.2029 | 507.2029 | 345.1089; 327.1215; 315.1409; 167.0116 | EA | [21] | |
| 26 | Ellagic acid | 26.397 | C14H6O8 | 302.1221 | 301.1221 | 283.1089; 271.1000; 160.9988; 125.9795 | EA | [21] |
| 27 | Sinapic acid derivative | 27.423 | 312.1533 | 311.1533 | 223.8222; 208.0325 | EA | [24,25] | |
| 28 | Caffeic acid derivative | 28.138 | 416.2092 | 415.2092 | 179.0079; 137.0253 | MeOH | [21] | |
| 29 | Quercetin-O-hexoside | 29.159 | C21H20O12 | 464.1014 | 463.1014 | 301.0300; 178.9275; 150.9379 | MeOH | [20] |
| 30 | Dimethoxy-trihydroxyflavone | 30.177 | C17H14O7 | 300.1051 | 299.1051 | 269.0865 | EA | [23] |
| 31 | Ferulic acid | 30.672 | C10H10O4 | 194.0153 | 193.0153 | 160.9938; 151.0017; 133.9934 | MeOH | [21,23] |
| 32 | Kaempferol glucoside | 30.710 | C21H20O11 | 448.1060 | 447.1060 | 285.0817 | MeOH | [14] |
| 33 | Caffeic acid derivative | 30.788 | 362.1790 | 361.1790 | 193.0563; 179.0297; 165.0047; 146.9725; 135.9938; 120.9468 | Water | [20] | |
| 34 | Quercetin derivative | 30.905 | 550.0995 | 549.0995 | 505.1463; 301.0477 | MeOH | [20] | |
| 35 | Gallic acid derivative | 31.085 | 188.0603 | 187.0603 | 169.0445; 125.0163 | EA | [21] | |
| 36 | Caffeic acid derivative | 32.040 | 362.1765 | 361.1765 | 301.1126; 285.2249; 179.0294; 165.0075; 146.9873; 120.9487 | EA | [21] | |
| 37 | Unidentified | 32.203 | 306.0734 | 305.0734 | 225.0952 | MeOH, Water | ||
| 38 | Unidentified dimer | 33.506 | 896.3362 | 895.3362 | 447.1766; 403.1865; 343.1615 | MeOH, Water | ||
| 39 | Dihydrocoumaric acid derivative | 34.565 | 246.0071 | 244.9989 | 165.0111 | MeOH, Water | [20] | |
| 40 | Trihydroxy-methoxyflavone derivative hexoside | 35.032 | 920.2537 | 919.2537 | 757.2071; 461.1288; 299.0536; 284.0217 | Water | [14] | |
| 41 | Unidentified | 36.106 | 534.1841 | 533.1841 | 445.2067; 385.1792; 343.1590; 163.0220 | Water | ||
| 42 | Tetrahydroxy-dimethoxyflavone | 36.744 | C17H14O8 | 346.0740 | 345.0740 | 330.0496; 315.0293 | Water | [14] |
| 43 | Caffeyl alcohol O-glucopyranoside | 37.102 | C15H20O8 | 328.2227 | 327.2227 | 229.1231; 165.0727; 121.0198 | EA, MeOH, Water | [23] |
| 44 | Sesquiterpene-derivative | 38.691 | 330.2386 | 329.2386 | 289.1532; 229.1252; 211.1104; 129.0162 | MeOH, Water | ||
| 45 | Trihydroxy-methoxyflavone | 39.570 | C16H12O6 | 300.0572 | 299.0572 | 284.0313 | EA | [14] |
| 46 | Sesquiterpene-derivative | 43.427 | 308.1944 | 307.1944 | 289.1829; 235.1171; 209.0913; 185.0827; 120.9816 | MeOH, Water | ||
| 47 | Ferulic acid derivative | 44.078 | 294.1770 | 293.1770 | 236.0898; 221.1337; 193.1262 | EA, MeOH, Water | [21] | |
| 48 | Phenylacetic acid derivative | 45.795 | 306.1786 | 305.1786 | 249.1359; 135.0091; 125.0171; | EA | [20] | |
| 49 | Sinapic acid derivative | 45.927 | 312.2265 | 311.2265 | 223.1479; 208.4343 | MeOH, Water | [24,25] | |
| 50 | Sinapine | 46.770 | C16H24NO5 | 310.2099 | 309.2099 | 291.2122; 251.1664; 223.1588; 208.6589 | EA | [25] |
| 51 | Sesquiterpene-derivative | 47.632 | 310.2099 | 309.2099 | 291.1943; 225.1275; 197.0855; 110.9822 | EA |
| Extracts | AChE Inhibition (mg GALAE/g) | BChE Inhibition (mg GALAE/g) | Tyrosinase Inhibition (mg KAE/g) | Amylase Inhibition (mmol ACAE/g) | Glucosidase Inhibition (mmol ACAE/g) |
|---|---|---|---|---|---|
| Ethyl acetate | 3.49 ± 0.16 b | 3.54 ± 0.43 b | 115.07 ± 3.14 b | 0.73 ± 0.04 c | 22.32 ± 1.15 b |
| Methanolic | 4.87 ± 0.57 c | 3.52 ± 0.11 b | 126.80 ± 0.39 c | 0.55 ± 0.03 b | 24.68 ± 0.03 c |
| Water | na a | na a | 43.11 ± 5.14 a | 0.11 ± 0.01 a | 1.06 ± 0.20 a |
| Extracts | DPPH (mg TE/g) | ABTS (mg TE/g) | CUPRAC (mg TE/g) | FRAP (mg TE/g) | Phosphomolybdenum (mmol TE/g) | Chelating Ability (mg EDTAE/g) |
|---|---|---|---|---|---|---|
| Ethyl acetate | 22.38 ± 0.37 a | 15.61 ± 0.58 a | 78.41 ± 4.04 a | 28.53 ± 1.47 a | 1.91 ± 0.05 b | 25.21 ± 1.20 b |
| Methanolic | 96.52 ± 0.10 c | 109.10 ± 2.57 c | 154.02 ± 4.58 c | 104.85 ± 0.57 c | 1.79 ± 0.15 b | 21.18 ± 1.72 a |
| Water | 40.21 ± 1.24 b | 90.98 ± 1.00 b | 108.02 ± 1.01 b | 75.18 ± 0.77 b | 1.25 ± 0.11 a | 31.64 ± 0.87 c |
| CC50 (µg/mL) | |||
|---|---|---|---|
| Ethyl Acetate | Methanol | Water | |
| VERO | >500 | na | na |
| FaDu | 231.6 ± 13.97 | 363.13 ± 19.56 | >500 |
| SCC-25 | 554.57 ± 62.34 | 408.43 ± 48.52 | 412.43 ± 51.61 |
| CC50 Value | <20 μg/mL | 21–200 μg/mL | 201–500 μg/mL | >500 μg/mL |
|---|---|---|---|---|
| Cytotoxic activity | high | moderate | weak | no activity |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zengin, G.; Mahomoodally, M.F.; Rocchetti, G.; Lucini, L.; Sieniawska, E.; Świątek, Ł.; Rajtar, B.; Polz-Dacewicz, M.; Senkardes, I.; Aktumsek, A.; et al. Chemical Characterization and Bioactive Properties of Different Extracts from Fibigia clypeata, an Unexplored Plant Food. Foods 2020, 9, 705. https://doi.org/10.3390/foods9060705
Zengin G, Mahomoodally MF, Rocchetti G, Lucini L, Sieniawska E, Świątek Ł, Rajtar B, Polz-Dacewicz M, Senkardes I, Aktumsek A, et al. Chemical Characterization and Bioactive Properties of Different Extracts from Fibigia clypeata, an Unexplored Plant Food. Foods. 2020; 9(6):705. https://doi.org/10.3390/foods9060705
Chicago/Turabian StyleZengin, Gokhan, Mohamad Fawzi Mahomoodally, Gabriele Rocchetti, Luigi Lucini, Elwira Sieniawska, Łukasz Świątek, Barbara Rajtar, Małgorzata Polz-Dacewicz, Ismail Senkardes, Abdurrahman Aktumsek, and et al. 2020. "Chemical Characterization and Bioactive Properties of Different Extracts from Fibigia clypeata, an Unexplored Plant Food" Foods 9, no. 6: 705. https://doi.org/10.3390/foods9060705
APA StyleZengin, G., Mahomoodally, M. F., Rocchetti, G., Lucini, L., Sieniawska, E., Świątek, Ł., Rajtar, B., Polz-Dacewicz, M., Senkardes, I., Aktumsek, A., Picot-Allain, M. C. N., & Montesano, D. (2020). Chemical Characterization and Bioactive Properties of Different Extracts from Fibigia clypeata, an Unexplored Plant Food. Foods, 9(6), 705. https://doi.org/10.3390/foods9060705












