Valorisation of Grape Stems as a Source of Phenolic Antioxidants by Using a Sustainable Extraction Methodology
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Material
2.3. Experimental Design
2.4. PLE
2.5. Identification and Quantification of Phenolic Compounds by RP-HPLC-PAD-MS
2.6. Analysis of Total Flavan-3-ol Monomers and Oligomers and Total Polymers by NP-HPLC
2.7. Determination of Mean Degree of Procyanidin Polymerization (mDP)
2.8. Total Phenolic Content (TPC)
2.9. Antioxidant Activity
2.10. Statistical Analyses
3. Results and Discussion
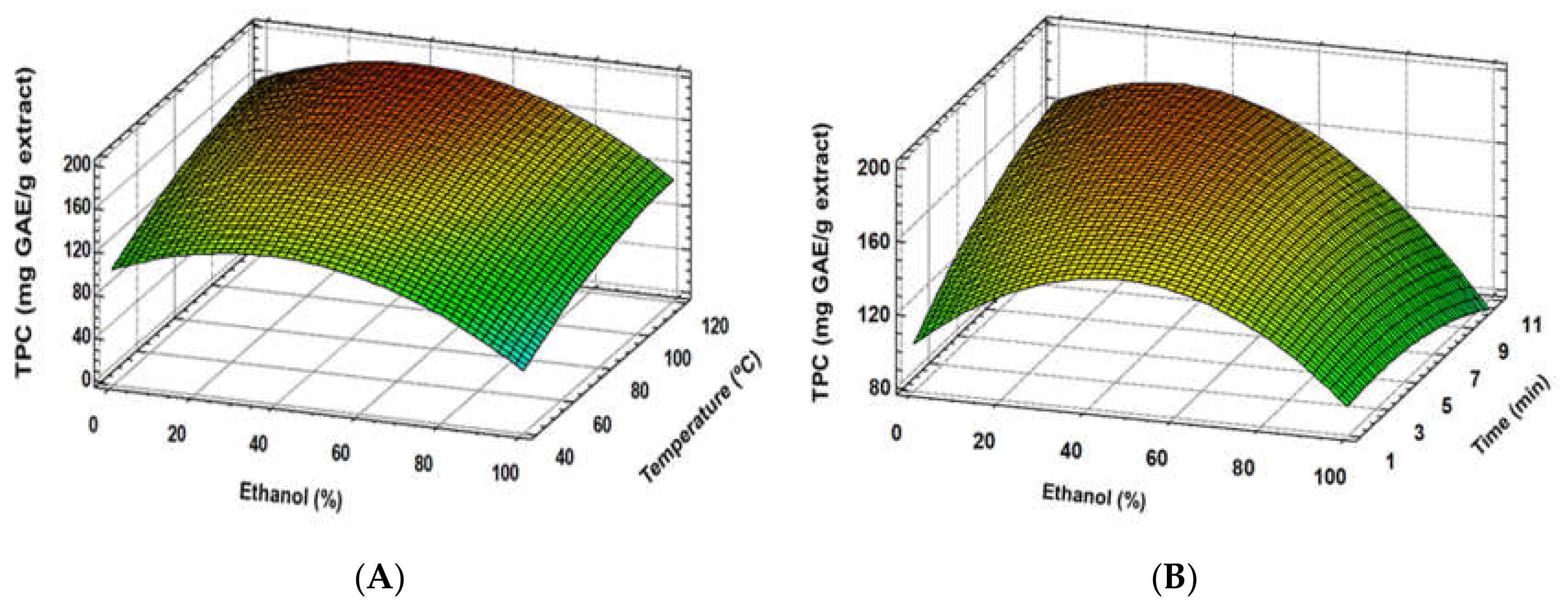
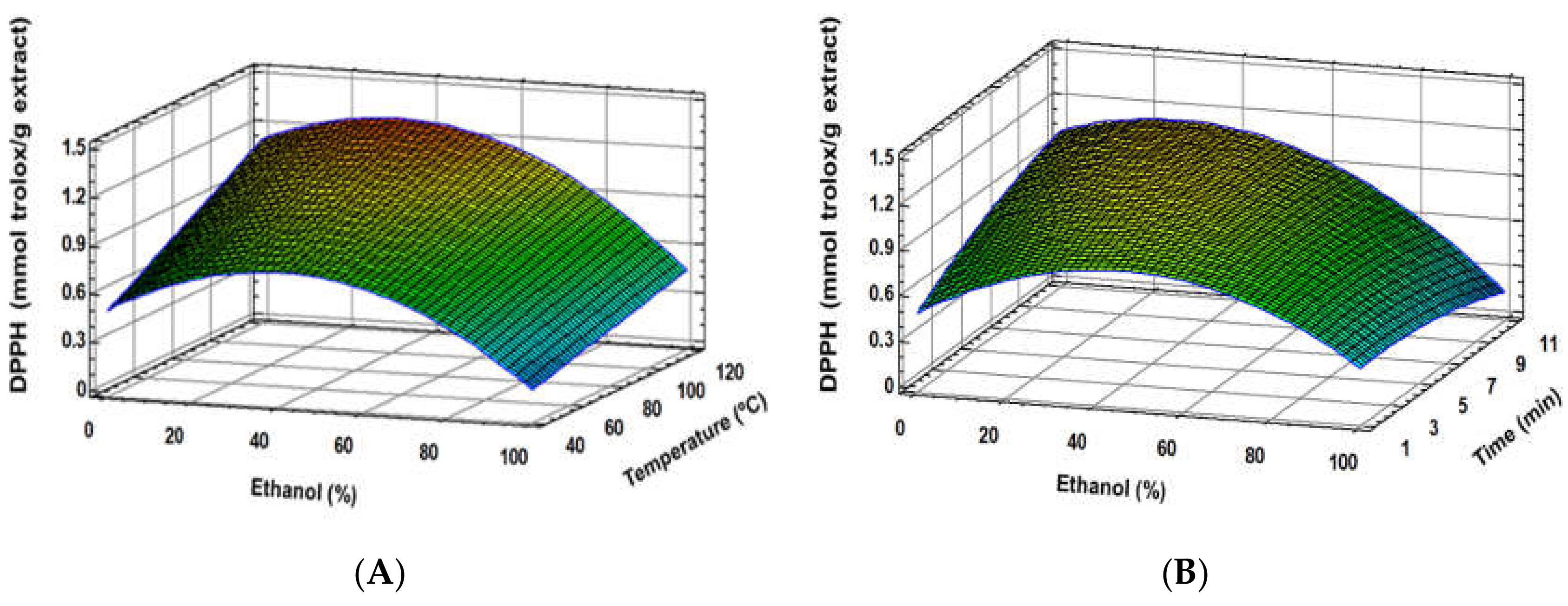
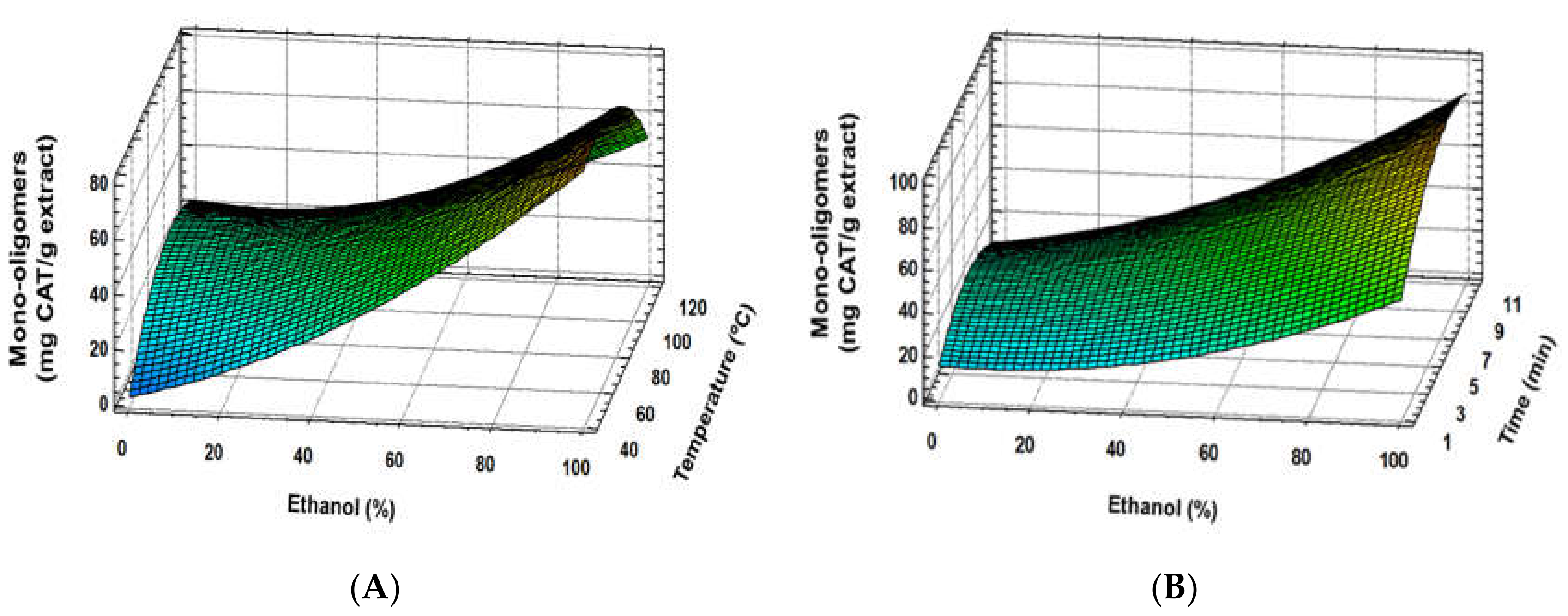
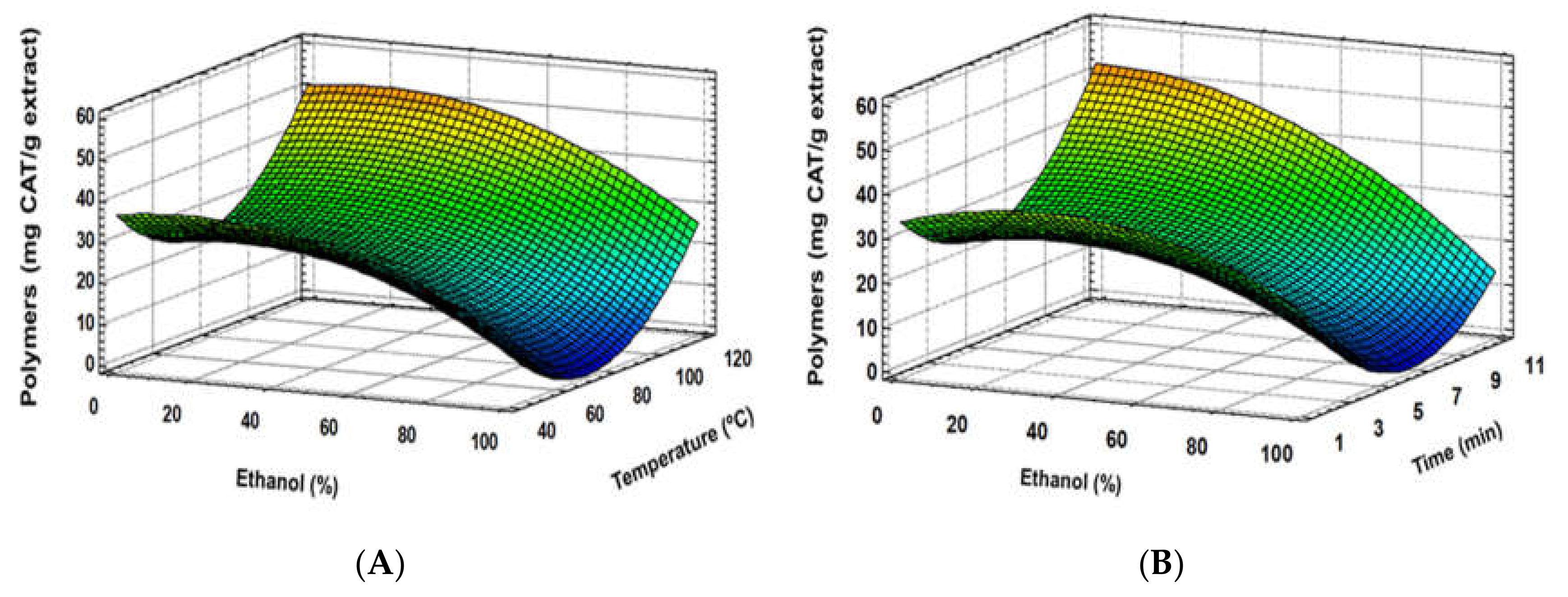
3.1. Experimental Model Fitting
3.2. Optimal Conditions and Validation of the Developed Model
3.3. Correlation between Response Variables
3.4. Phenolic Composition of the Optimum Extract
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations (FAO). 2019. Available online: http://www.fao.org/faostat/en/#home (accessed on 11 April 2019).
- Gouvinhas, I.; Queiroz, M.; Rodrigues, M.; Barros, A.I. Evaluation of the Phytochemistry and Biological Activity of Grape (Vitis vinifera L.) Stems: Toward a Sustainable Winery Industry. In Polyphenols in Plants; Watson, R.R., Ed.; Academic Press Inc.: San Diego, CA, USA, 2019; pp. 381–394. [Google Scholar]
- Barros, A.I.; Gouvinhas, I.; Machado, N.; Pinto, J.; Cunha, M.; Rosa, E.; Domínguez-Perles, R. New grape stems-based liqueur: Physicochemical and phytochemical evaluation. Food Chem. 2016, 190, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Moreno, M.J.; Raposo, R.; Cayuela, J.M.; Zafrilla, P.; Piñeiro, Z.; Moreno-Rojas, J.M.; Mulero, J.; Puertas, B.; Girón, F.; Guerrero, R.F.; et al. Valorization of grape stems. Ind. Crop. Prod. 2015, 63, 152–157. [Google Scholar] [CrossRef]
- Goutzourelas, N.; Stagos, D.; Spanidis, Y.; Liosi, M.; Apostolou, A.; Priftis, A.; Haroutounian, S.; Spandidos, D.A.; Tsatsakis, A.M.; Kouretas, D. Polyphenolic composition of grape stem extracts affects antioxidant activity in endothelial and muscle cells. Mol. Med. Rep. 2015, 12, 5846–5856. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Igrejas, G.; Falco, V.; Santos, T.P.; Torres, C.; Oliveira, A.M.P.; Pereira, J.E.; Amaral, J.S.; Poeta, P. Chemical composition, antioxidant and antimicrobial activity of phenolic compounds extracted from wine industry by-products. Food Control. 2018, 92, 516–522. [Google Scholar] [CrossRef]
- Souquet, J.M.; Labarbe, B.; Le Guernevé, C.; Cheynier, V.Y. Phenolic composition of grape stems. J. Agric. Food. Comp. Chem. 2000, 48, 1076–1080. [Google Scholar] [CrossRef] [PubMed]
- Barros, A.; Gironés-Vilaplana, A.; Teixeira, A.; Collado-González, J.; Moreno, D.A.; Gil-Izquierdo, A.; Rosa, E.; Domínguez-Perles, R. Evaluation of grape (Vitis vinifera L.) stems from Portuguese varieties as a resource of (poly)phenolic compounds: A comparative study. Food Res. Int. 2014, 65, 375–384. [Google Scholar] [CrossRef]
- Pugajeva, I.; Perkons, I.; Górnaś, P. Identification and determination of stilbenes by Q-TOF in grape skins, seeds, juice and stems. J. Food Compos. Anal. 2018, 74, 44–52. [Google Scholar] [CrossRef]
- Püssa, T.; Floren, J.; Kuldkeep, P.; Raal, A. Survey of grape wine stem polyphenols by liquid chromatography-diode array detection tandem mass spectrometry. J. Agric. Food Chem. 2006, 54, 7488–7994. [Google Scholar] [CrossRef]
- Luo, L.; Cui, Y.; Zhang, S.; Li, L.; Suo, H.; Sun, B. Detailed phenolic composition of Vidal grape pomace by ultrahigh-performance liquid chromatography–tandem mass spectrometry. J. Chromatogr. B 2017, 1068, 201–209. [Google Scholar] [CrossRef]
- Domínguez-Perles, R.; Teixeira, A.; Rosa, E.; Barros, A. Assessment of (poly)phenols in grape (Vitis vinifera L.) stems by using food/pharma industry compatible solvents and response surface methodology. Food Chem. 2014, 164, 339–346. [Google Scholar]
- Karvela, E.; Makris, D.P.; Kalogeropoulosa, N.; Karathanosa, V.T. Deployment of response surface methodology to optimize recovery of grape (Vitis vinifera) stem and seed polyphenols. Procedia Food Sci. 2011, 1, 1686–1693. [Google Scholar] [CrossRef]
- Llaudy, M.C.; Canals, R.; Canals, J.M.; Zamora, F. Influence of ripening stage and maceration length on the contribution of grape skins, seeds and stems to phenolic composition and astringency in wine-simulated macerations. Eur. Food Res. Technol. 2008, 226, 337–344. [Google Scholar] [CrossRef]
- Choi, M.P.K.; Chan, K.K.C.; Leung, H.W.; Huie, C.W. Pressurized liquid extraction of active ingredients (ginsenosides) from medicinal plants using non-ionic surfactant solutions. J. Chromatogr. A 2003, 983, 153–162. [Google Scholar] [CrossRef]
- Arapitsas, P.; Turner, C. Pressurized solvent extraction and monolithic column HPLC/DAD analysis of anthocyanins in red cabbage. Talanta 2008, 74, 1218–1223. [Google Scholar] [CrossRef]
- Wijngaard, H.; Hossain, M.B.; Rai, D.K.; Brunton, N. Techniques to extract bioactive compounds from food by-products of plant origin. Food Res. Int. 2012, 46, 505–513. [Google Scholar] [CrossRef]
- Llobera, A. Study on the Antioxidant Activity of Grape Stems (Vitis vinifera). A Preliminary Assessment of Crude Extracts. Food Nutr. Sci. 2012, 3, 500–504. [Google Scholar]
- Anastasiadi, M.; Pratsinis, H.; Kletsas, D.; Skaltsounis, A.; Haroutounian, S.A. Grape stem extracts: Polyphenolic content and assessment of their in vitro antioxidant properties. Lwt Food Sci. Technol. 2012, 48, 316–322. [Google Scholar] [CrossRef]
- Casas, L.; Mantell, C.; Rodríguez, M.; Martínez de la Ossa, E.J.; Roldán, A.; De Ory, I.; Caro, I.; Blandino, A. Extraction of resveratrol from the pomace of Palomino fino grapes by supercritical carbon dioxide. J. Food Eng. 2010, 96, 304–308. [Google Scholar] [CrossRef]
- Piñeiro, Z.; Marrufo-Curtido, A.; Vela, C.; Palma, M. Microwave-assisted extraction of stilbenes from woody vine material. Food Bioprod. Process. 2017, 103, 18–26. [Google Scholar] [CrossRef]
- González-Centeno, M.R.; Jourdes, M.; Femenia, A.; Simal, S.; Rosselló, C.; Teissedre, P.L. Proanthocyanidin composition and antioxidant potential of the stem winemaking byproducts from 10 different grape varieties (Vitis vinifera L.). J. Agric. Food Chem. 2012, 60, 11850–11858. [Google Scholar]
- Wenzel, J.; Storer-Samaniego, C.; Wang, L.; Nelson, L.; Ketchum, K.; Ammerman, M.; Zand, A. Superheated liquid and supercritical denatured ethanol extraction of antioxidants from Crimson red grape stems. Food Sci. Nutr. 2015, 3, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Prieto, R.M.; Fernández-Cabot, R.A.; Costa-Bauzá, A.; Sánchez, A.M.; Prodanov, M. Effect of consuming a grape seed supplement with abundant phenolic compounds on the oxidative status of healthy human volunteers. Nutr. J. 2015, 14, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Prodanov, M.; Vacas, V.; Hernández, T.; Estrella, I.; Amador, B.; Winterhalter, P. Chemical characterisation of Malvar grape seeds (Vitis vinifera L.) by ultrafiltration and RP-HPLC-PAD-MS. J. Food Comp. Anal. 2013, 31, 284–292. [Google Scholar] [CrossRef]
- Muñoz-Labrador, A.; Prodanov, M.; Villamiel, M. Effects of high intensity ultrasound on disaggregation of a macromolecular procyanidin-rich fraction from Vitis vinifera L. seed extract and evaluation of its antioxidant activity. Ultrason. Sonochem. 2019, 50, 74–81. [Google Scholar] [CrossRef]
- Sun, B.S.; Leandro, M.C.; Ricardo-da-Silva, J.M.; Spranger, M.I. Separation of grape and wine proanthocyanidins according to their degree of polymerisation. J. Agric. Food Chem. 1998, 46, 1390–1396. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu Reagent. Methods Enzym. 1999, 299, 152–178. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuveleir, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Lwt-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Huang, D.J.; Ou, D.X.; Hampsch-Woodill, M.; Flanagan, J.A.; Prior, R.L. High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 2002, 50, 4437–4444. [Google Scholar] [CrossRef]
- Li, Y.; Skouroumounis, J.K.; Elsey, G.M.; Taylor, D.K. Microwave-assistance provides very rapid and efficient extraction of grape seed polyphenols. Food Chem. 2011, 129, 570–576. [Google Scholar] [CrossRef]
- Silva, E.M.; Rogez, H.; Larondelle, Y. Optimization of extraction of phenolic from Inga edulis leaves using response surface methodology. Sep. Purif. Technol. 2007, 55, 381–387. [Google Scholar] [CrossRef]
- Mandal, V.; Mohan, Y.; Hemalatha, S. Microwave assisted extraction-an innovative and promising extraction tool for medicinal plant research. Pharm. Rev. 2007, 1, 7–18. [Google Scholar]
- Aliakbarian, B.; Fathi, A.; Perego, P.; Dehghani, F. Extraction of antioxidants from winery wastes using subcritical water. J. Supercrit. Fluids 2012, 65, 18–24. [Google Scholar] [CrossRef]
- Sun, B.; Spranger, M.I. Changes in phenolic composition of Tinta Miúda red wines after 2 years of ageing in bottle: effect of winemaking technologies. Eur. Food Res. Technol. 2005, 221, 305–312. [Google Scholar] [CrossRef]
- Hawthorne, S.B.; Yang, Y.; Miller, D.J. Extraction of organic pollutans from environmental solids with subcritical and supercritical water. Anal. Chem. 1994, 66, 2912–2920. [Google Scholar] [CrossRef]
- Makris, D.P.; Boskou, G.; Andrikopoulos, K. Recovery of antioxidant phenolics from white vinification solid by-prodcts employing water/ethanol mixtures. Bioresour. Technol. 2007, 98, 2963–2967. [Google Scholar] [CrossRef]
- Mulero, J.; Martínez, G.; Oliva, J.; Cermeño, S.; Cayuela, J.M.; Zafrilla, P.; Martínez-Cachá, A.; Barba, A. Phenolic compounds and antioxidant activity of red wine made from grapes treated with different fungicides. Food Chem. 2015, 180, 25–31. [Google Scholar] [CrossRef]
- Marchante, L.; Loarce, L.; Izquierdo-Cañas, M.P.; Alañón, M.E.; García-Romero, E.; Pérez-Coello, M.S.; Díaz-Maroto, M.C. Natural extracts from grape seed and stem by-products in combination with colloidal silver as alternative preservatives to SO2 for white wines: Effects on chemical composition and sensorial properties. Food Res. Int. 2019, 108594. [Google Scholar] [CrossRef]



| Factor | Coded Symbol | Coded Levels | ||||
|---|---|---|---|---|---|---|
| −1.68 | −1 | 0 | 1 | 1.68 | ||
| Ethanol concentration (%) | Et | 0 | 20 | 50 | 80 | 100 |
| Temperature (°C) | T | 40 | 56 | 80 | 104 | 120 |
| Time (min) | t | 1 | 3 | 6 | 9 | 11 |
| Factor | Response Variables | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Run | X1, Ethanol | X2, Temperature | X3, Time | Yield | Total Phenolic Compounds (TPC) | ABTS | DPPH | Total Flavan-3-ol Mono- and Oligomers | Total Polymer Procyanidins | |
| (%) | (°C) | (min) | (g Extract/100 g Stem) | (mg GAE/g Extract) | (mmol Trolox/g Extract) | (mmol Trolox/g Extract) | (mg catechin/g Extract) | (mg catechin/g Extract) | ||
| 1 | 20 | 56 | 3 | 21.5 | 118.1 | 2.06 | 0.65 | 19.53 | 30.02 | |
| 2 | 80 | 56 | 3 | 9.4 | 114.0 | 1.89 | 0.55 | 52.72 | 14.55 | |
| 3 | 20 | 104 | 3 | 27.5 | 164.0 | 2.89 | 0.97 | 24.48 | 42.64 | |
| 4 | 80 | 104 | 3 | 23.3 | 145.9 | 2.50 | 0.81 | 31.51 | 32.10 | |
| 5 | 20 | 56 | 9 | 21.0 | 148.7 | 2.63 | 0.86 | 27.16 | 41.97 | |
| 6 | 80 | 56 | 9 | 10.3 | 113.2 | 1.94 | 0.56 | 69.97 | 21.26 | |
| 7 | 20 | 104 | 9 | 28.5 | 178.7 | 3.33 | 1.19 | 26.59 | 44.58 | |
| 8 | 80 | 104 | 9 | 14.8 | 135.2 | 2.42 | 0.72 | 52.64 | 19.65 | |
| 9 | 0 | 80 | 6 | 24.9 | 147.8 | 2.54 | 0.87 | 17.81 | 21.68 | |
| 10 | 100 | 80 | 6 | 12.4 | 87.4 | 1.39 | 0.29 | 98.73 | 4.06 | |
| 11 | 50 | 40 | 6 | 18.8 | 129.4 | 2.24 | 0.77 | 30.67 | 34.53 | |
| 12 | 50 | 120 | 6 | 30.6 | 186.6 | 3.43 | 1.22 | 26.51 | 49.78 | |
| 13 | 50 | 80 | 1 | 21.2 | 147.8 | 2.60 | 0.88 | 26.26 | 38.42 | |
| 14 | 50 | 80 | 11 | 24.8 | 172.7 | 2.96 | 1.06 | 27.73 | 44.51 | |
| 15 | 50 | 80 | 6 | 22.8 | 167.6 | 2.98 | 1.00 | 47.45 | 22.05 | |
| 16 | 50 | 80 | 6 | 25.0 | 171.8 | 3.00 | 1.05 | 45.43 | 21.62 | |
| 17 | 50 | 80 | 6 | 24.3 | 167.9 | 2.94 | 1.03 | 45.67 | 21.30 | |
| 18 | 50 | 80 | 6 | 24.1 | 169.7 | 2.95 | 0.98 | 45.34 | 21.82 | |
| 19 | 50 | 80 | 6 | 24.0 | 160.2 | 2.77 | 1.01 | 46.65 | 21.48 | |
| Variable | Polynomial Equation of Fitted Model | R2 | Lack-of-Fit (p-Value) |
|---|---|---|---|
| Yields (g extract/g stem) | Y = −2.05097 + 0.188423(Et) + 0.24681(T) + 2.91554(t) − 0.00271887(Et)2 − 0.0115812(Et × t) − 0.0144409(T × t) − 0.0984341(t)2 | 0.951 | 0.15 |
| TPC (mg GAE/g extract) | Y = −15.954 + 2.10974(Et) + 1.94514(T) + 10.808(t) − 0.0212643(Et)2 − 0.0807058(Et × t) − 0.00779276(T)2 − 0.408779(t)2 | 0.97 | 0.21 |
| ABTS (mmol Trolox/g extract) | Y = 0.578714 + 0.0369343(Et) + 0.0142628(T) + 0.112674(t) − 0.000382393(Et)2 − 0.00147263(Et × t) | 0.97 | 0.33 |
| DPPH (mmol Trolox/g extract) | Y = −0.238343 + 0.0209687(Et) + 0.00766806(T) + 0.0851506(t) − 0.000183361(Et)2 − 0.0000407129(Et × T) − 0.00071712(Et × t) − 0.00273786(t)2 | 0.98 | 0.19 |
| Optimal Conditions | Optimal Extract Values (30% Et, 120 °C, 10 min) | ||||
|---|---|---|---|---|---|
| Et (%) | T (°C) | T (min) | Experimental | Estimated | |
| Yields (g extract/100 g stem) | 25 | 120 | 4.5 | 28.9 | 29.2 |
| TPC (mg GAE/g extract) | 30 | 120 | 10 | 187.3 | 192.4 |
| ABTS (mmol Trolox/g extract) | 27 | 120 | 11 | 3.69 | 3.81 |
| DPPH (mmol Trolox/g extract) | 22 | 120 | 11 | 1.32 | 1.37 |
| Phenolic Compound | UV-Vis Max. | [M − H]−1 | [M + H]+1 | MS/MS Fragments | mg/g dry Extract |
|---|---|---|---|---|---|
| No Flavonoids | |||||
| Hydroxybenzoic acids | |||||
| Gallic acid | 270 | 169 | 125 | 0.541 ± 0.029 | |
| Protocatechuic acid | 260/290 | 153 | 117 | 0.008 ± 0.000 | |
| Monogalloyl glucoside | 257/298 | 331 | 169 | <LOQ | |
| 4-Hydroxybenzoic acid | 256 | 137 | 0.048 ± 0.001 | ||
| Vanillic acid | 259/292 | 167 | 153 | 0.224 ± 0.010 | |
| Syringic acid | 278 | 197 | 183 | 0.202 ± 0.015 | |
| Ethyl gallate | 277 | 197 | 169 | 0.010 ± 0.001 | |
| Ellagic acid | 256/353 | 301 | 229 | 0.073 ± 0.004 | |
| Hydroxycinnamic acids | |||||
| trans-caftaric acid | 296/328 | 311 | 179 | 0.357 ± 0.003 | |
| trans-caffeic acid | 300/324 | 179 | 161 | 0.006 ± 0.000 | |
| 4-Coumaric acid | 290/310 | 163 | 119 | 0.004 ± 0.000 | |
| 3-Coumaric acid | 289/309 | 163 | 119 | 0.003 ± 0.000 | |
| Coumaroyl-O-glucoside | 280/308 | 325 | 163,119 | 0.003 ± 0.000 | |
| trans- ferulic acid | 298/322 | 193 | 149 | 0.008 ± 0.000 | |
| Stilbenes | |||||
| trans-Piceid | 295/324 | 389 | 227 | 0.016 ± 0.000 | |
| trans-Resveratrol | 303/328 | 227 | 185 | 0.141 ± 0.003 | |
| ε-viniferin | 262/308/322 | 453 | 359 | 0.879 ± 0.065 | |
| cis-resveratrol trimer | 286 | 679 | 585 | 0.031 ± 0.002 | |
| trans-resveratrol trimer | 296/320 | 679 | 587,575 | 0.012 ± 0.001 | |
| trans-resveratrol trimer | 288/326 | 679 | 587,575 | 0.042 ± 0.003 | |
| trans-resveratrol tetramer | 306/316 | 905 | 811 | 0.136 ± 0.011 | |
| trans-resveratrol tetramer | 306/316 | 905 | 811 | 0.086 ± 0.007 | |
| cis-resveratrol tetramer | 284 | 905 | 811,717 | 0.038 ± 0.003 | |
| trans-resveratrol tetramer | 306/318 | 905 | 811,799 | <LOQ | |
| Flavonoids | |||||
| Flavan-3-ols | |||||
| Catechin | 278 | 289 | 245 | 2.422 ± 0.034 | |
| Epicatechin | 278 | 289 | 245 | 1.293 ± 0.039 | |
| Epicatechin gallate | 280 | 441 | 289,169 | 0.245 ± 0.005 | |
| Procyanidin B1 | 278 | 577 | 425 | 1.410 ± 0.034 | |
| Procyanidin B2 | 278 | 577 | 425 | 0.015 ± 0.003 | |
| Procyanidin B3 | 278 | 577 | 425 | 0.349 ± 0.025 | |
| Procyanidin B4 | 279 | 577 | 425 | 0.036 ± 0.002 | |
| Procyanidin B7 | 280 | 577 | 425 | 0.025 ± 0.003 | |
| Procyanidin C1 | 280 | 865 | 577 | 0.016 ± 0.001 | |
| Flavonols | |||||
| Kaempferol-3-O-glucoside | 287/358 | 447 | 285 | <LOQ | |
| Quercetin-3-O-galactoside | 256/354 | 463 | 301 | 0.047 ± 0.000 | |
| Quercetin-3-O-rutinoside | 256/354 | 609 | 301 | 0.029 ± 0.000 | |
| Quercetin-3-O-glucuronide | 252/354 | 477 | 301 | 1.425 ± 0.001 | |
| Quercetin-3-O-glucoside | 254/354 | 463 | 301 | 0.106 ± 0.004 | |
| Quercetin | 256/368 | 301 | 0.005 ± 0.000 | ||
| Anthocyanins | |||||
| Delphinidin-3-O-glucoside | 292/535 | 465 | 303 | <LOQ | |
| Cyanidin-3-O-glucoside | 290/530 | 449 | 287 | 0.010 ± 0.001 | |
| Malvidin-3-O-glucoside | 293/537 | 493 | 331 | 0.079 ± 0.002 |
| Terminal Units (%) | Extension Units (%) | mDP | Galloilated Units (%) | |||||
|---|---|---|---|---|---|---|---|---|
| Cat | EC | ECG | Cat | EC | ECG | EGC | ||
| 6.32 | 1.07 | 0.81 | 8.27 | 71.27 | 11.59 | 0.68 | 12.22 | 12.40 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nieto, J.A.; Santoyo, S.; Prodanov, M.; Reglero, G.; Jaime, L. Valorisation of Grape Stems as a Source of Phenolic Antioxidants by Using a Sustainable Extraction Methodology. Foods 2020, 9, 604. https://doi.org/10.3390/foods9050604
Nieto JA, Santoyo S, Prodanov M, Reglero G, Jaime L. Valorisation of Grape Stems as a Source of Phenolic Antioxidants by Using a Sustainable Extraction Methodology. Foods. 2020; 9(5):604. https://doi.org/10.3390/foods9050604
Chicago/Turabian StyleNieto, Juan Antonio, Susana Santoyo, Marin Prodanov, Guillermo Reglero, and Laura Jaime. 2020. "Valorisation of Grape Stems as a Source of Phenolic Antioxidants by Using a Sustainable Extraction Methodology" Foods 9, no. 5: 604. https://doi.org/10.3390/foods9050604
APA StyleNieto, J. A., Santoyo, S., Prodanov, M., Reglero, G., & Jaime, L. (2020). Valorisation of Grape Stems as a Source of Phenolic Antioxidants by Using a Sustainable Extraction Methodology. Foods, 9(5), 604. https://doi.org/10.3390/foods9050604








