Effect of Jasmonic Acid, Yeast Extract Elicitation, and Drying Methods on the Main Bioactive Compounds and Consumer Quality of Lovage (Levisticum officinale Koch)
Abstract
1. Introduction
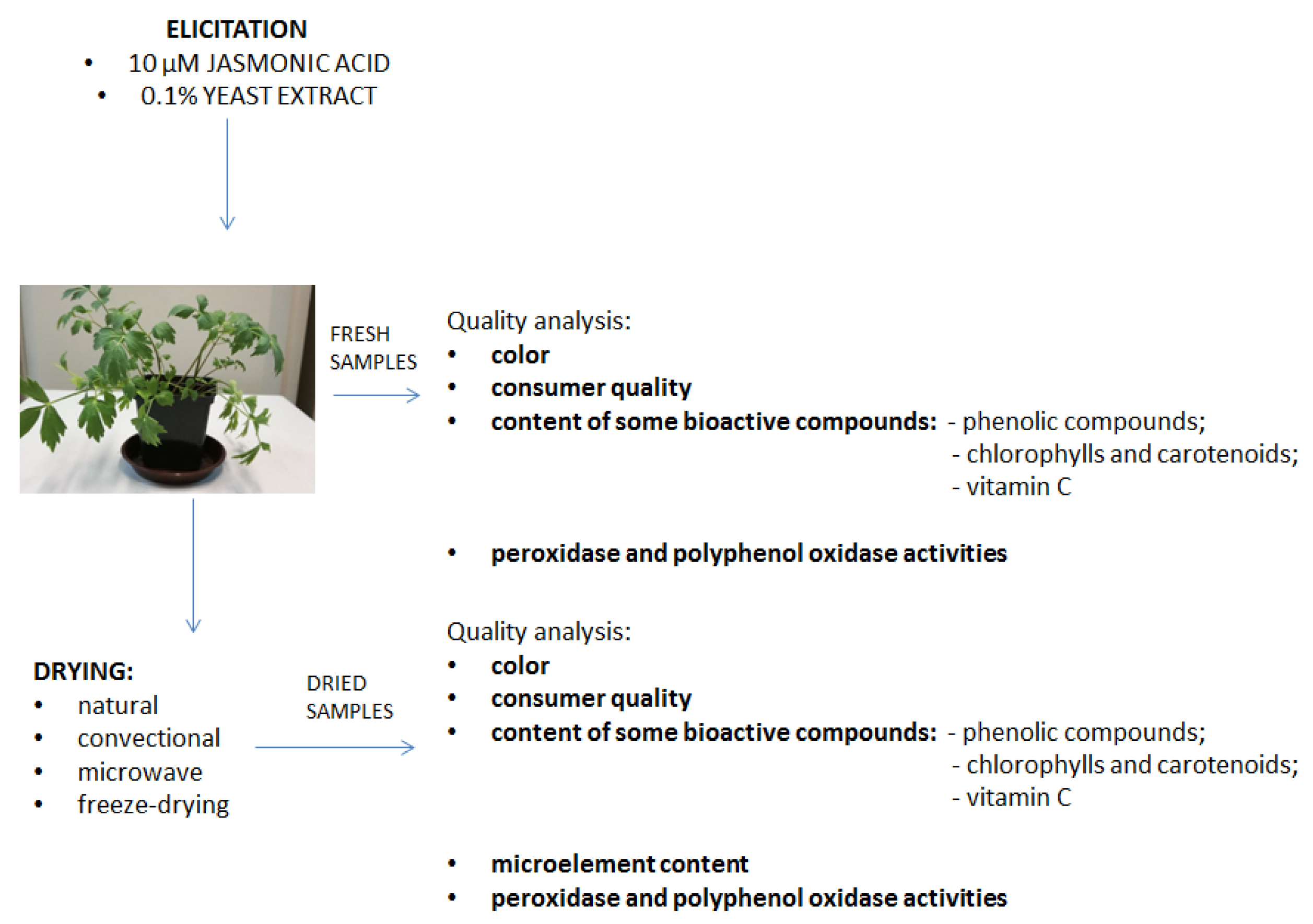
2. Materials and Methods
2.1. Plant Material
2.1.1. Growth Conditions
2.1.2. Determination of Plant Yield
2.1.3. Drying Techniques
2.2. Quality Analysis
2.2.1. Color
2.2.2. Consumer Quality
2.2.3. Content of Some Bioactive Compounds
Extraction and Determination of Total Phenolic Compounds in Lovage
Extraction and Determination of the Levels of Chlorophylls (Chl) and Carotenoids (Car)
Extraction and Determination of Vitamin C Content
Microelement Content
2.3. Peroxidase and Polyphenol Oxidase Activities
2.3.1. Polyphenol Oxidase (PPO)
2.3.2. Guaiacol Peroxidase (POD)
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Amiri, M.S.; Joharchi, M.R. Ethnobotanical knowledge of Apiaceae family in Iran: A review. J. Phytomedicine 2016, 6, 621–635. [Google Scholar]
- Rohwer, C.L.; Erwin, J.E. Horticultural applications of jasmonates: A review. J. Hortic. Sci. Biotechnol. 2008, 83, 283–304. [Google Scholar] [CrossRef]
- Taha, L.S.; Ibrahim, S.M.M.; Abdel Aziz, N.G. Vegetative growth, chemical composition, and flavonoids content of Azadirachta indica plants as affected by application of yeast natural extract. J. Appl. Pharm. Sci. 2016, 6, 93–97. [Google Scholar] [CrossRef]
- Złotek, U. Effect of Jasmonic Acid and yeast extract elicitation on low-molecular antioxidants and antioxidant activity of Marjoram (Origanum Majorana L.). Acta Sci. Pol. Technol. Aliment. 2017, 16. [Google Scholar]
- Złotek, U.; Świeca, M. Elicitation effect of Saccharomyces cerevisiae yeast extract on main health-promoting compounds and antioxidant and anti-inflammatory potential of butter lettuce (Lactuca sativa L.). J. Sci. Food Agric. 2016, 96, 2565–2572. [Google Scholar] [CrossRef]
- Złotek, U.; Szymanowska, U.; Pecio, U.; Kozachok, S.; Jakubczyk, A. Antioxidative and Potentially Anti-inflammatory Activity of Phenolics from Lovage Leaves Levisticum officinale Koch Elicited with Jasmonic Acid and Yeast Extract. Molecules 2019, 24, 1441. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Bioactivity and protective effects of natural carotenoids. Biochim. Biophys. Acta Mol. Basis Dis. 2005, 1740, 101–107. [Google Scholar] [CrossRef]
- Terefe, N.S.; Buckow, R.; Versteeg, C. Quality-Related Enzymes in Fruit and Vegetable Products: Effects of Novel Food Processing Technologies, Part 1: High-Pressure Processing. Crit. Rev. Food Sci. Nutr. 2014, 54, 24–63. [Google Scholar] [CrossRef]
- Baenas, N.; García-viguera, C.; Moreno, D.A. Elicitation: A Tool for Enriching the Bioactive Composition of Foods. Molecules 2014, 19, 13541–13563. [Google Scholar] [CrossRef]
- Malekpoor, F.; Salimi, A.; Pirbalouti, A.G. Effect of jasmonic acid on total phenolic content and antioxidant activity of extract from the green and purple landraces of sweet basil. Acta Pol. Pharm. Drug Res. 2016, 73, 1229–1234. [Google Scholar]
- Tamagnone, L.; Merida, A.; Stacey, N.; Plaskitt, K.; Parr, A.; Chang, C.F.; Lynn, D.; Dow, J.M.; Roberts, K.; Martin, C. Inhibition of phenolic acid metabolism results in precocious cell death and altered cell morphology in leaves of transgenic tobacco plants. Plant Cell 1998, 10, 1801–1816. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.Y.; Murtijaya, J. Antioxidant properties of Phyllanthus amarus extracts as affected by different drying methods. LWT Food Sci. Technol. 2007, 40, 1664–1669. [Google Scholar] [CrossRef]
- Król, B.; Kiełtyka-Dadasiewicz, A. Wpływ Metody Suszenia Na Cechy Sensoryczne Oraz Skład Olejku Eterycznego Tymianku Właściwego (Thymus vulgaris L.). Zywn. Nauk. Technol. Jakosc Food. Sci. Technol. Qual. 2015, 22, 162–175. [Google Scholar]
- Ansorena, M.R.; Agüero, M.V.; Grabriela Goñi, M.; Roura, S.; Ponce, A.; Del, M.; Moreira, R.; Scala, K. Di Assessment of lettuce quality during storage at low relative humidity using Global Stability Index methodology. Ciência e Tecnol. Aliment. 2012, 32, 366–373. [Google Scholar] [CrossRef]
- Capecka, E.; Mareczek, A.; Leja, M. Antioxidant activity of fresh and dry herbs of some Lamiaceae species. Food Chem. 2005, 93, 223–226. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Lin, K.-H.; Huang, M.-Y.; Huang, W.-D.; Hsu, M.-H.; Yang, Z.-W.; Yang, C.-M. The effects of red, blue, and white light-emitting diodes on the growth, development, and edible quality of hydroponically grown lettuce (Lactuca sativa L. var. capitata). Sci. Hortic. 2013, 150, 86–91. [Google Scholar] [CrossRef]
- Mazurek, A.; Jamroz, J. Precision of dehydroascorbic acid quantitation with the use of the subtraction method—Validation of HPLC–DAD method for determination of total vitamin C in food. Food Chem. 2015, 173, 543–550. [Google Scholar] [CrossRef]
- Wissemann, K.W.; Lee, C.Y. Purification of Grape Polyphenoloxidase with Hydrophobic Chromatography; Elsevier: Amsterdam, The Netherlands, 1980; Volume 192. [Google Scholar]
- Chance, B.; Maehly, A.C. Assay of catalases and peroxidases. Methods Enzymol. 1955, 2, 764–775. [Google Scholar]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef]
- Złotek, U.; Michalak-Majewska, M.; Szymanowska, U. Effect of jasmonic acid elicitation on the yield, chemical composition, and antioxidant and anti-inflammatory properties of essential oil of lettuce leaf basil (Ocimum basilicum L.). Food Chem. 2016, 213, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gholizadeh, A.; Kohnehrouz, B.B. Activation of phenylalanine ammonia lyase as a key component of the antioxidative system of salt-challenged maize leaves. Brazilian J. Plant Physiol. 2010, 22, 217–223. [Google Scholar] [CrossRef]
- Nafie, E.; Hathout, T.; Al Mokadem, A.S. Jasmonic acid elicits oxidative defense and detoxification systems in Cucumis melo L. cells. Brazilian J. Plant Physiol. 2011, 23, 161–174. [Google Scholar] [CrossRef]
- Kiczorowska, B.; Klebaniuk, R.; Bąkowski, M.; Al-Yasiry, A.R.M.H. Culinary herbs—The nutritive value and content of minerals. J. Elem. 2015, 20, 599–608. [Google Scholar] [CrossRef]
- Martínez-Ballesta, M.C.; Dominguez-Perles, R.; Moreno, D.A.; Muries, B.; Alcaraz-López, C.; Bastías, E.; García-Viguera, C.; Carvajal, M. Minerals in plant food: Effect of agricultural practices and role in human health. A review. Agron. Sustain. Dev. 2010, 30, 295–309. [Google Scholar] [CrossRef]
- Mbondo, N.N.; Owino, W.O.; Ambuko, J.; Sila, D.N. Effect of drying methods on the retention of bioactive compounds in African eggplant. Food Sci. Nutr. 2018, 6, 814–823. [Google Scholar] [CrossRef]
- Balan, D.; Israel-Roming, F.; Luta, G.; Gherghina, E. Changes in the nutrients content of some green vegetables during storage and thermal processing. Rom. Biotechnol. Lett. 2016, 21, 11857–11865. [Google Scholar]
- Śledz, M.; Witrowa-Rajchert, D. Influence of microwave-convective drying of chlorophyll content and colour of herbs. Acta Agrophysica 2012, 19, 865–876. [Google Scholar]
- Złotek, U. Effect of abiotic elicitation on the quality and antioxidant potential of lettuce and endive during storage. J. Food Biochem. 2017, 41, e12428. [Google Scholar] [CrossRef]
- Złotek, U.; Świeca, M.; Jakubczyk, A. Effect of abiotic elicitation on main health-promoting compounds, antioxidant activity and commercial quality of butter lettuce (Lactuca sativa L.). Food Chem. 2014, 148, 253–260. [Google Scholar] [CrossRef]
- Ferruzzi, M.G.; Blakeslee, J. Digestion, absorption, and cancer preventative activity of dietary chlorophyll derivatives. Nutr. Res. 2007, 27, 1–12. [Google Scholar] [CrossRef]
- Śledź, M.; Nowacka, M.; Wiktor, A.; Witrowa-Rajchert, D. Selected chemical and physico-chemical properties of microwave-convective dried herbs. Food Bioprod. Process. 2013, 91, 421–428. [Google Scholar] [CrossRef]
- Stępień, A.E.; Gorzelany, J.; Matłok, N.; Lech, K.; Figiel, A. The effect of drying methods on the energy consumption, bioactive potential and colour of dried leaves of Pink Rock Rose (Cistus creticus). J. Food Sci. Technol. 2019, 56, 2386–2394. [Google Scholar] [CrossRef] [PubMed]
- Pourcel, L.; Routaboul, J.-M.; Cheynier, V.; Lepiniec, L.; Debeaujon, I. Flavonoid oxidation in plants: From biochemical properties to physiological functions. Trends Plant Sci. 2007, 12, 29–36. [Google Scholar] [CrossRef]
- Raju, S.; Jayalakshmi, S.K.; Sreeramulu, K. Comparative study on the induction of defense related enzymes in two different cultivars of chickpea (Cicer arietinum L.) genotypes by salicylic acid, spermine and Fusarium oxysporum f. sp. ciceri. Aust. J. Crop Sci. 2008, 2, 121–140. [Google Scholar]
- Złotek, U.; Gawlik-Dziki, U. Selected biochemical properties of polyphenol oxidase in butter lettuce leaves (Lactuca sativa L. var. capitata) elicited with dl-β-amino-n-butyric acid. Food Chem. 2015, 168, 423–429. [Google Scholar] [CrossRef]
| - | Plant Height [cm] | Plant Fresh Weight [g/Plant] | Plant Dry Weight [g/Plant] |
|---|---|---|---|
| C | 15.27 ± 0.64 a | 1.09 ± 0.35 a | 0.17 ± 0.02 a |
| JA | 14.67 ± 1.53 a | 1.68 ± 0.32 a | 0.25 ± 0.01 b |
| YE | 15.00 ± 1.73 a | 1.09 ± 0.02 a | 0.16 ± 0.01 a |
| Sample | Color Parameters | Consumer Quality | ||||
|---|---|---|---|---|---|---|
| L* | a* | b* | Colour | Flavor | Appearance | |
| Fresh material | ||||||
| C | 35.57 ± 2.05 a | −7.42 ± 2.61 ab | 14.55 ± 1.50 c | 3.00 ± 0.00 ab | 4.00 ± 0.00 a | 4.00 ± 0.00 a |
| JA | 39.20 ± 0.45 ab | −9.26 ± 0.76 a | 18.54 ± 3.06 d | 4.33 ± 0.58 ab | 5.00 ± 0.00 a | 4.67 ± 0.58 a |
| YE | 41.97 ± 1.43 b | −5.86 ± 1.48 b | 24.28 ± 3.07 e | 4.00 ± 0.00 ab | 4.67 ± 0.58 a | 4.00 ± 0.00 a |
| Freeze-dried samples | ||||||
| CL | 50.11 ± 0.68 cd | −1.92 ± 0.32 cd | 5.58 ± 1.15 ab | 4.5 ± 0.71 b | 4.2 ± 0.79 a | 4.3 ± 0.82 a |
| JAL | 49.90 ± 1.67 cd | −1.22 ± 0.17 cde | 4.40 ± 0.42 ab | 3.8 ± 0.79 ab | 4.2 ± 0.79 a | 3.6 ± 1.07 a |
| YEL | 51.83 ± 0.43 cd | −3.14 ± 0.38 c | 5.87 ± 0.45 ab | 4.4 ± 0.97 ab | 4.3 ± 0.95 a | 4.6 ± 0.52 a |
| Microwave-dried samples | ||||||
| CM | 48.55 ± 3.91 cd | 0.31 ± 0.08 de | 3.24 ± 0.55 a | 2.6 ± 1.35 a | 3.2 ± 1.55 a | 3.1 ± 1.29 a |
| JAM | 54.01 ± 2.85 d | 0.71 ± 0.29 e | 5.99 ± 0.97 ab | 3.6 ± 1.26 ab | 3.3 ± 1.06 a | 3.5 ± 1.35 a |
| YEM | 52.55 ± 1.10 cd | 0.60 ± 0.09 e | 5.32 ± 0.10 ab | 3.4 ± 1.17 ab | 3.1 ± 1.10 a | 3.7 ± 0.82 a |
| Traditionally dried samples | ||||||
| CT | 49.37 ± 0.43 cd | −0.24 ± 0.12 de | 6.97 ± 0.16 ab | 3.4 ± 1.07 ab | 3.4 ± 1.17 a | 3.1 ± 1.66 a |
| JAT | 51.77 ± 1.48 cd | 0.53 ± 0.01 de | 7.75 ± 0.33 ab | 4.5 ± 0.53 b | 3.6 ± 0.52 a | 4.6 ± 0.52 a |
| YET | 47.79 ± 3.60 c | −1.12 ± 0.78 cde | 4.79 ± 0.46 ab | 3.3 ± 1.16 ab | 3.6 ± 0.84 a | 3.1 ± 1.37 a |
| Convectionally dried samples | ||||||
| CC | 50.12 ± 0.20 cd | −1.61 ± 0.30 cde | 6.11 ± 0.11 ab | 4.2 ± 0.92 b | 3.2 ± 0.63 a | 3.3 ± 0.82 a |
| JAC | 50.93 ± 0.24 cd | −1.02 ± 0.15 cde | 7.20 ± 0.05 b | 4.3 ± 0.95 b | 3.8 ± 0.79 a | 4.1 ± 0.89 a |
| YEC | 51.20 ± 0.65 cd | −0.73 ± 0.15 cde | 6.09 ± 0.11 ab | 4.1 ± 0.74 ab | 3.7 ± 0.67 a | 4.4 ± 0.84 a |
| Elicitor | Compounds | |||||
|---|---|---|---|---|---|---|
| Chl a (μg/g FW) | Chl b (μg/g FW) | Chl a + b (μg/g FW) | Car (μg/g FW) | Vit C mg/100 g FW | TPC mg/g FW | |
| Fresh material | ||||||
| C | 14.14 ± 1.97 b | 5.33 ± 0.43 f | 19.47 ± 2.39 c | 1.37 ± 0.28 c | 49.13 ± 0.63 c | 2.40 ± 0.18 bcd |
| JA | 18.00 ± 3.35 c | 11.45 ± 0.05 h | 29.45 ± 3.35 e | 0.89 ± 0.03 b | 57.93 ± 0.21 d | 4.01 ± 0.31 e |
| YE | 17.07 ± 1.71 bc | 6.57 ± 0.64 g | 23.64 ± 2.35 d | 1.55 ± 0.19 c | 49.80 ± 4.62 c | 2.74 ± 0.65 d |
| Freeze-dried samples | ||||||
| CL | 3.33 ± 0.01 a | 1.88 ± 0.02 e | 5.21 ± 0.03 b | 0.33 ± 0.00 a | 9.69 ± 0.81 b | 2.22 ± 0.12 bcd |
| JAL | 1.81 ± 0.13 a | 0.85 ± 0.20 bcd | 2.66 ± 0.34 ab | 0.18 ± 0.05 a | 3.58 ± 0.21 ab | 1.87 ± 0.17 ab |
| YEL | 3.17 ± 0.13 a | 1.91 ± 0.20 e | 5.08 ± 0.33 b | 0.25 ± 0.01 a | 5.31 ± 0.14 ab | 2.32 ± 0.17 bcd |
| Microwave-dried samples | ||||||
| CM | 1.42 ± 0.20 a | 0.39 ± 0.10 abc | 1.80 ± 0.30 ab | 0.15 ± 0.03 a | 6.51 ± 0.75 ab | 2.27 ± 0.22 bcd |
| JAM | 0.84 ± 0.01 a | 0.16 ± 0.00 a | 1.00 ± 0.01 a | 0.09 ± 0.00 a | 3.52 ± 0.06 ab | 1.78 ± 0.23 ab |
| YEM | 1.27 ± 0.09 a | 0.25 ± 0.02 ab | 1.52 ± 0.12 ab | 0.07 ± 0.00 a | 5.07 ± 0.77 ab | 1.94 ± 0.12 abc |
| Traditionally dried samples | ||||||
| CT | 2.36 ± 0.07 a | 0.95 ± 0.04 cd | 3.31 ± 0.11 ab | 0.22 ± 0.01 a | 4.84 ± 0.11 ab | 1.79 ± 0.34 ab |
| JAT | 1.26 ± 0.10 a | 0.42 ± 0.05 abc | 1.68 ± 0.15 ab | 0.14 ± 0.01 a | 1.58 ± 0.03 ab | 1.28 ± 0.16 a |
| YET | 1.99 ± 0.07 a | 0.90 ± 0.04 bcd | 2.89 ± 0.11 ab | 0.16 ± 0.01 a | 1.24 ± 0.08 a | 2.66 ± 0.14 d |
| Convectionally dried samples | ||||||
| CC | 1.84 ± 0.14 a | 0.81 ± 0.13 abcd | 2.66 ± 0.27 ab | 0.23 ± 0.03 a | 1.37 ± 0.00 ab | 2.55 ± 0.20 cd |
| JAC | 1.12 ± 0.13 a | 0.52 ± 0.09 abcd | 1.64 ± 0.21 ab | 0.14 ± 0.02 a | 0.77 ± 0.00 a | 1.81 ± 0.17 ab |
| YEC | 2.22 ± 0.07 a | 1.16 ± 0.05 d | 3.38 ± 0.12 ab | 0.23 ± 0.01 a | 0.87 ± 0.06 a | 2.08 ± 0.27 bcd |
| Microelement | Sample | ||
|---|---|---|---|
| C | JA | YE | |
| Fe [mg/kg] | 164.67 ± 2.52 b | 92.83 ± 3.43 a | 299.00 ± 3.61 c |
| Cu [mg/kg] | 8.55 ± 0.79 b | 4.66 ± 0.95 a | 5.11 ± 0.75 a |
| Zn [mg/kg] | 21.73 ± 0.49 a | 28.20 ± 0.56 b | 33.60 ± 0.17 c |
| Mn [mg/kg] | 55.40 ± 1.01 b | 41.73 ± 1.61 a | 71.20 ± 1.18 c |
| Ni [mg/kg] | 9.79 ± 0.46 a | 12.53 ± 0.61 b | 13.47 ± 0.83 b |
| Cd [mg/kg] | 0.34 ± 0.02 a | 0.52 ± 0.04 b | 0.83 ± 0.04 c |
| Mg [g/kg] | 2.14 ± 0.02 a | 2.25 ± 0.04 b | 2.65 ± 0.04 c |
| K [g/kg] | 66.10 ± 3.45 b | 68.53 ± 1.75 b | 51.07 ± 0.45 a |
| Ca [g/kg] | 27.20 ± 0.17 c | 21.10 ± 0.17 a | 24.30 ± 0.30 b |
| Na [mg/kg] | 46.13 ± 0.64 a | 56.87 ± 0.50 b | 58.97 ± 0.67 c |
| Pb | N.d. | N.d. | N.d. |
| Co | N.d. | N.d. | N.d. |
| Ag | N.d. | N.d. | N.d. |
| Sample | PPO [U/mg Protein] | POD [U/mg Protein] |
|---|---|---|
| Fresh material | ||
| C | 102.75 ± 0.60 def | 286.53 ± 0.08 b |
| JA | 203.81 ± 19.97 h | 494.55 ± 60.73 c |
| YE | 159.26 ± 7.80 gh | 461.87 ± 36.00 c |
| Freeze-dried samples | ||
| CL | 17.21 ± 4.28 ab | 9.39 ± 4.69 a |
| JAL | 13.77 ± 4.77 a | 6.35 ± 2.75 a |
| YEL | 38.25 ± 18.23 abc | 5.29 ± 0.01 a |
| Microwave-dried samples | ||
| CM | 88.89 ± 38.49 cde | 6.73 ± 0.1 2 a |
| JAM | 144.44 ± 19.25 fgh | 5.31 ± 0.75 a |
| YEM | 74.07 ± 32.08 bcd | 6.76 ± 0.04 a |
| Traditionally dried samples | ||
| CT | 44.44 ± 19.25 abcd | 5.95 ± 0.00 a |
| JAT | 39.22 ± 16.98 abc | 9.11 ± 6.31 a |
| YET | n.d. | 19.32 ± 0.00 a |
| Convectionally dried samples | ||
| CC | 30.30 ± 0.00 abc | 9.75 ± 3.38 a |
| JAC | 55.56 ± 27.49 abcd | 8.23 ± 3.56 a |
| YEC | 58.82 ± 19.61 abcd | 11.11 ± 0.00 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Złotek, U.; Szymanowska, U.; Rybczyńska-Tkaczyk, K.; Jakubczyk, A. Effect of Jasmonic Acid, Yeast Extract Elicitation, and Drying Methods on the Main Bioactive Compounds and Consumer Quality of Lovage (Levisticum officinale Koch). Foods 2020, 9, 323. https://doi.org/10.3390/foods9030323
Złotek U, Szymanowska U, Rybczyńska-Tkaczyk K, Jakubczyk A. Effect of Jasmonic Acid, Yeast Extract Elicitation, and Drying Methods on the Main Bioactive Compounds and Consumer Quality of Lovage (Levisticum officinale Koch). Foods. 2020; 9(3):323. https://doi.org/10.3390/foods9030323
Chicago/Turabian StyleZłotek, Urszula, Urszula Szymanowska, Kamila Rybczyńska-Tkaczyk, and Anna Jakubczyk. 2020. "Effect of Jasmonic Acid, Yeast Extract Elicitation, and Drying Methods on the Main Bioactive Compounds and Consumer Quality of Lovage (Levisticum officinale Koch)" Foods 9, no. 3: 323. https://doi.org/10.3390/foods9030323
APA StyleZłotek, U., Szymanowska, U., Rybczyńska-Tkaczyk, K., & Jakubczyk, A. (2020). Effect of Jasmonic Acid, Yeast Extract Elicitation, and Drying Methods on the Main Bioactive Compounds and Consumer Quality of Lovage (Levisticum officinale Koch). Foods, 9(3), 323. https://doi.org/10.3390/foods9030323








