Do Best-Selected Strains Perform Table Olive Fermentation Better than Undefined Biodiverse Starters? A Comparative Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Plan
2.2. Technological Characterisation
2.2.1. Tolerance to Low Temperatures, High Saline Concentrations, and Alkaline pH
2.2.2. Bacterial Growth and Acidification Ability
2.2.3. Tolerance to Olive Leaf Extract
2.2.4. Use of Oleuropein as Substrate
2.2.5. Hydrogen Peroxide Production
2.2.6. Lactic Acid Production
2.3. Starter Culture Origin and Preparation
2.4. Pilot Scale Fermentation Trials
2.5. Physical-Chemical Analyses
2.6. Phenolic Analysis
2.7. DPPH Scavenging Activity as Trolox Equivalent Antioxidant Capacity (TEAC)
2.8. Texture Analyses
2.9. Microbiological Analyses
2.10. Statistical Analysis
3. Results
3.1. Technological Characterisation
3.1.1. Tolerance to Low Temperatures, High Saline Concentrations, and Alkaline pH
3.1.2. Bacterial Growth and Acidification Ability
3.1.3. Olive Leaf Extract Tolerance and Use of Oleuropein as Substrate
3.1.4. Hydrogen Peroxide Production
3.1.5. Lactic Acid Production
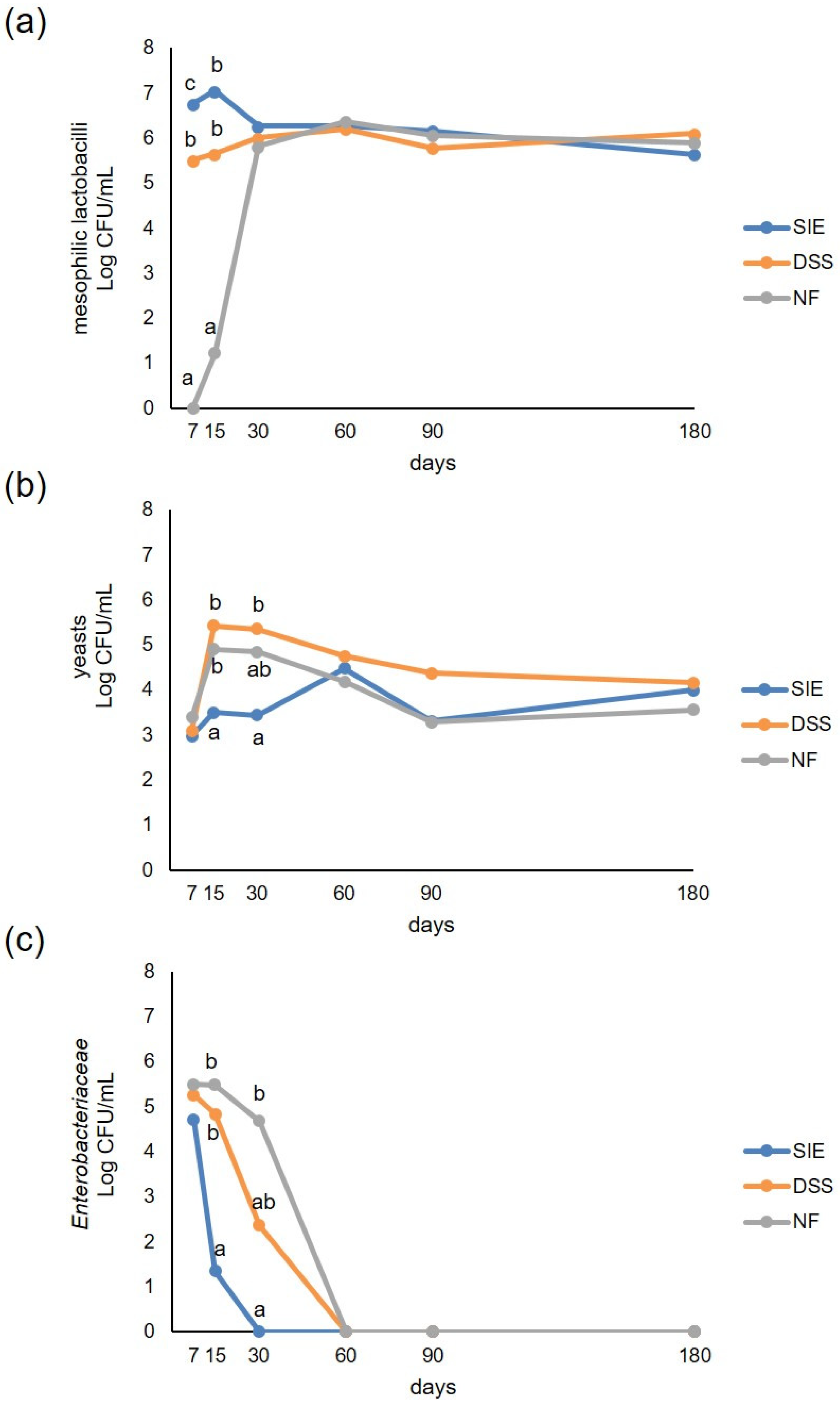
3.2. Microbiological Analyses
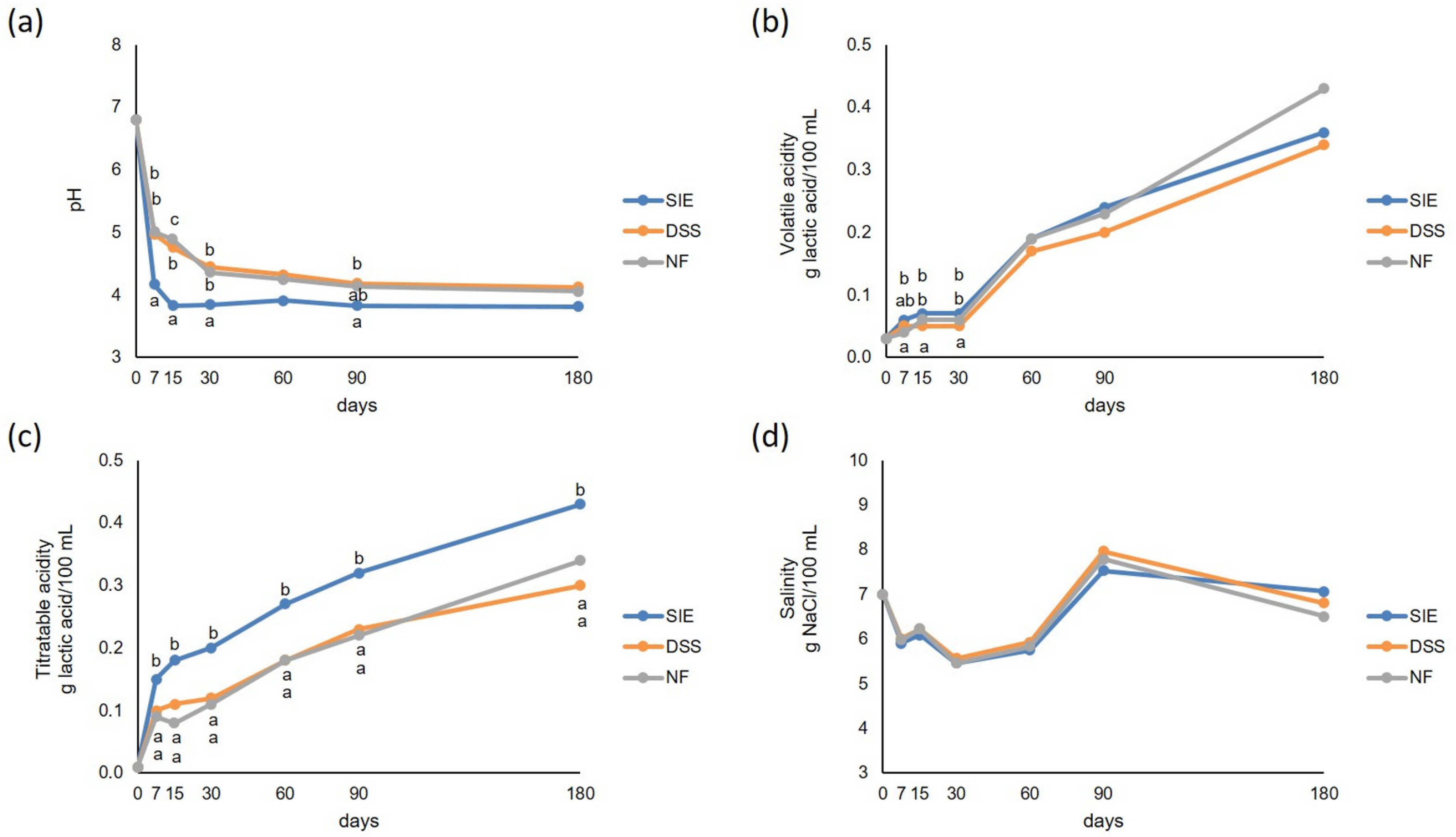
3.3. Physical-Chemical Analyses
3.4. Phenolic Compounds Concentration and Antioxidant Activity as TEAC (Trolox Equivalent Antioxidant Capacity)
3.5. Texture Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ciafardini, G.; Zullo, B.A. Use of selected yeast starter cultures in industrial-scale processing of brined Taggiasca black table olives. Food Microbiol. 2019, 84, 103250. [Google Scholar] [CrossRef] [PubMed]
- Campus, M.; Değirmencioğlu, N.; Comunian, R. Technologies and Trends to Improve Table Olive Quality and Safety. Front. Microbiol. 2018, 9, 617. [Google Scholar] [CrossRef] [PubMed]
- Aponte, M.; Blaiotta, G.; La Croce, F.; Mazzaglia, A.; Farina, V.; Settanni, L.; Moschetti, G. Use of selected autochthonous lactic acid bacteria for Spanish-style table olive fermentation. Food Microbiol. 2012, 30, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Corsetti, A.; Perpetuini, G.; Schirone, M.; Tofalo, R.; Suzzi, G. Application of starter cultures to table olive fermentation: An overview on the experimental studies. Front. Microbiol. 2012, 3, 248. [Google Scholar] [CrossRef] [PubMed]
- Lanza, B. Abnormal fermentations in table-olive processing: Microbial origin and sensory evaluation. Front. Microbiol. 2013, 4, 91. [Google Scholar] [CrossRef]
- Bonatsou, S.; Benitez, A.; Rodriguez-Gomez, F.; Panagou, E.Z.; Arroyo-Lopez, F.N. Selection of yeasts with multifunctional features for application as starters in natural black table olive processing. Food Microbiol. 2015, 46, 66–73. [Google Scholar] [CrossRef]
- Bonatsou, S.; Tassou, C.C.; Panagou, E.Z.; Nychas, G.E. Table Olive Fermentation Using Starter Cultures with Multifunctional Potential. Microorganisms 2017, 5, 30. [Google Scholar] [CrossRef]
- Hurtado, A.; Reguant, C.; Bordons, A.; Rozes, N. Lactic acid bacteria from fermented table olives. Food Microbiol. 2012, 31, 1–8. [Google Scholar] [CrossRef]
- Leroy, F.; De Vuyst, L. Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends Food Sci. Tech. 2004, 15, 67–78. [Google Scholar] [CrossRef]
- Martorana, A.; Alfonzo, A.; Settanni, L.; Corona, O.; La Croce, F.; Caruso, T.; Moschetti, G.; Francesca, N. An innovative method to produce green table olives based on “pied de cuve” technology. Food Microbiol. 2015, 50, 126–140. [Google Scholar] [CrossRef]
- Heperkan, D. Microbiota of table olive fermentations and criteria of selection for their use as starters. Front. Microbiol. 2013, 4, 143. [Google Scholar] [CrossRef]
- Bevilacqua, A.; Altieri, C.; Corbo, M.R.; Sinigaglia, M.; Ouoba, L.I. Characterization of lactic acid bacteria isolated from Italian Bella di Cerignola table olives: selection of potential multifunctional starter cultures. J. Food Sci. 2010, 75, M536–M544. [Google Scholar] [CrossRef]
- Bleve, G.; Tufariello, M.; Durante, M.; Grieco, F.; Ramires, F.A.; Mita, G.; Tasioula-Margari, M.; Logrieco, A.F. Physico-chemical characterization of natural fermentation process of Conservolea and Kalamata table olives and developement of a protocol for the pre-selection of fermentation starters. Food Microbiol. 2015, 46, 368–382. [Google Scholar] [CrossRef]
- Di Cagno, R.; Surico, R.F.; Paradiso, A.; De Angelis, M.; Salmon, J.C.; Buchin, S.; De Gara, L.; Gobbetti, M. Effect of autochthonous lactic acid bacteria starters on health-promoting and sensory properties of tomato juices. Int. J. Food Microbiol. 2009, 128, 473–483. [Google Scholar] [CrossRef]
- Bassi, D.; Puglisi, E.; Cocconcelli, P.S. Comparing natural and selected starter cultures in meat and cheese fermentations. Curr. Opin. Food Sci. 2015, 2, 118–122. [Google Scholar] [CrossRef]
- Campus, M.; Sedda, P.; Cauli, E.; Piras, F.; Comunian, R.; Paba, A.; Daga, E.; Schirru, S.; Angioni, A.; Zurru, R.; et al. Evaluation of a single strain starter culture, a selected inoculum enrichment, and natural microflora in the processing of Tonda di Cagliari natural table olives: Impact on chemical, microbiological, sensory and texture quality. LWT Food Sci. Tech. 2015, 64, 671–677. [Google Scholar] [CrossRef]
- Comunian, R.; Ferrocino, I.; Paba, A.; Daga, E.; Campus, M.; Di Salvo, R.; Cauli, E.; Piras, F.; Zurru, R.; Cocolin, L. Evolution of microbiota during spontaneous and inoculated Tonda di Cagliari table olives fermentation and impact on sensory characteristics. LWT 2017, 84, 64–72. [Google Scholar] [CrossRef]
- Bandino, G.; Moro, C.; Mulas, M.; Sedda, P. Survey on olive genetic resources of sardinia. Acta Hortic. 1999, 474, 151–154. [Google Scholar] [CrossRef]
- Bandino, G.; Sedda, P. Le Varietà di olivo della Sardegna. In L’olio in Sardegna; ILISSO, Ed.; ILISSO: Nuoro, Italy, 2013; pp. 171–222. ISBN 978-88-6202-309-2. [Google Scholar]
- Sawatari, Y.; Yokota, A. Diversity and Mechanisms of Alkali Tolerance in Lactobacilli. Appl. Environ. Microbiol. 2007, 73, 3909. [Google Scholar] [CrossRef]
- Ghabbour, N.; Lamzira, Z.; Thonart, P.; Cidalia, P.; Markaoui, M.; Asehraou, A. Selection of oleuropein-degrading lactic acid bacteria strains isolated from fermenting Moroccan green olives. Grasas y Aceites 2011, 62, 84–89. [Google Scholar] [CrossRef]
- Marshall, V.M. A Note on Screening Hydrogen Peroxide-producing Lactic Acid Bacteria Using a Non-toxic Chromogen. J. Appl. Bacteriol. 1979, 47, 327–328. [Google Scholar] [CrossRef]
- Berthier, F. On the screening of hydrogen peroxide-generating lactic acid bacteria. Lett. Appl. Micbobiol. 1993, 16, 150–153. [Google Scholar] [CrossRef]
- International Olive Council. Determination of Biophenols in Olive Oils by HPLC. Available online: https://www.oelea.de/downloads/COI-T20-DOC-29-2009-DETERMINATION-OF-BIOPHENOLS-IN-OLIVE-OILS-BY-HPLC.pdf (accessed on 17 December 2019).
- Szczesniak, A.S. Classification of Textural Characteristicsa. J. Food Sci. 1963, 28, 385–389. [Google Scholar] [CrossRef]
- Friedman, H.H.; Whitney, J.E.; Szczesniak, A.S. The texturometer—A new instrument for objective texture measurement. J. Food Sci. 1963, 28, 390–396. [Google Scholar] [CrossRef]
- Kiai, H.; Hafidi, A. Chemical composition changes in four green olive cultivars during spontaneous fermentation. LWT Food Sci. Technol. 2014, 57, 663–670. [Google Scholar] [CrossRef]
- International Olive Council. Trade Standard on Table Olives. Available online: https://www.internationaloliveoil.org/what-we-do/chemistry-standardisation-unit/standards-and-methods/ (accessed on 17 December 2019).
- De Angelis, M.; Campanella, D.; Cosmai, L.; Summo, C.; Rizzello, C.G.; Caponio, F. Microbiota and metabolome of un-started and started Greek-type fermentation of Bella di Cerignola table olives. Food Microbiol. 2015, 52, 18–30. [Google Scholar] [CrossRef]
- Medina-Pradas, E.; Pérez-Díaz, I.M.; Garrido-Fernández, A.; Arroyo-López, F.N. Review of Vegetable Fermentations With Particular Emphasis on Processing Modifications, Microbial Ecology, and Spoilage. In The Microbiological Quality of Food: Foodborne Spoilers; Elsevier: Duxford, UK, 2017; pp. 211–236. [Google Scholar]
- Campus, M.; Cauli, E.; Scano, E.; Piras, F.; Comunian, R.; Paba, A.; Daga, E.; Di Salvo, R.; Sedda, P.; Angioni, A.; et al. Towards Controlled Fermentation of Table Olives: LAB Starter Driven Process in an Automatic Pilot Processing Plant. Food Bioprocess Tech. 2017, 10, 1063–1073. [Google Scholar] [CrossRef]
- Tofalo, R.; Schirone, M.; Perpetuini, G.; Angelozzi, G.; Suzzi, G.; Corsetti, A. Microbiological and chemical profiles of naturally fermented table olives and brines from different Italian cultivars. Antonie van Leeuwenhoek 2012, 102, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-López, F.N.; Bautista-Gallego, J.; Rodríguez-Gómez, F.; Garrido-Fernández, A. Predictive microbiology and table olives. In Current Research, Technology and Education Topics in Applied Microbiology and Microbial Biotechnology; Formatex Research Center: Badajoz, Spain, 2010; pp. 1452–1461. [Google Scholar]
- Hernandez, A.; Martin, A.; Aranda, E.; Perez-Nevado, F.; Cordoba, M.G. Identification and characterization of yeast isolated from the elaboration of seasoned green table olives. Food Microbiol. 2007, 24, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-López, F.N.; Querol, A.; Bautista-Gallego, J.; Garrido-Fernández, A. Role of yeasts in table olive production. Int. J. Food Microbiol. 2008, 128, 189–196. [Google Scholar] [CrossRef]
- Fernandez, A.G.; Adams, M.R.; Fernandez-Diez, M.J. Table Olives, 1st ed.; Springer: Heidelberg, Germany, 1997. [Google Scholar]
- Lazarides, H. Reasons and possibilities to control solids uptake turing osmotic treatment of fruits and vegetables, osmotic dehydration and vacuum impregnation. In Osmotic Dehydration and Vacuum Impregnation; Taylor & Francis Group: London, UK, 2019; pp. 33–42. [Google Scholar]
- Akbarian, M.; Ghasemkhani, N.; Moayedi, F. Osmotic dehydration of fruits in food industrial: A review. Int. J. Biosci. 2013, 3, 234–237. [Google Scholar] [CrossRef]
- Maldonado, M.; Zuritz, C.; Miras, N. Influence of brine concentration on sugar and sodium chloride diffusion during the processing of the green olive variety Arauco. Grasas y Aceites 2008, 59, 267–273. [Google Scholar] [CrossRef]
- Cardoso, S.M.; Guyot, S.; Marnet, N.; Lopes-da-Silva, J.A.; Renard, C.M.; Coimbra, M.A. Characterisation of phenolic extracts from olive pulp and olive pomace by electrospray mass spectrometry. J. Sci. Food Agric. 2005, 85, 21–32. [Google Scholar] [CrossRef]
- Marsilio, V.; Seghetti, L.; Iannucci, E.; Russi, F.; Lanza, B.; Felicioni, M. Use of a lactic acid bacteria starter culture during green olive (Olea europaea L. cv Ascolana tenera) processing. J. Sci. Food Agric. 2005, 85, 1084–1090. [Google Scholar] [CrossRef]
- Coimbra, M.A.; Waldron, K.W.; Delgadillo, I.; Selvendran, R.R. Effect of Processing on Cell Wall Polysaccharides of Green Table Olives. J. Agric. Food Chem. 1996, 44, 2394–2401. [Google Scholar] [CrossRef]
- Servili, M.; Minnocci, A.; Veneziani, G.; Taticchi, A.; Urbani, S.; Esposto, S.; Sebastiani, L.; Valmorri, S.; Corsetti, A. Compositional and tissue modifications induced by the natural fermentation process in table olives. J. Agric. Food Chem. 2008, 56, 6389–6396. [Google Scholar] [CrossRef] [PubMed]
- Di Cagno, R.; Coda, R.; De Angelis, M.; Gobbetti, M. Exploitation of vegetables and fruits through lactic acid fermentation. Food Microbiol. 2013, 33, 1–10. [Google Scholar] [CrossRef]
- Bevilacqua, A.; de Stefano, F.; Augello, S.; Pignatiello, S.; Sinigaglia, M.; Corbo, M.R. Biotechnological innovations for table olives. Int. J. Food Sci. Nutr. 2015, 66, 127–131. [Google Scholar] [CrossRef]
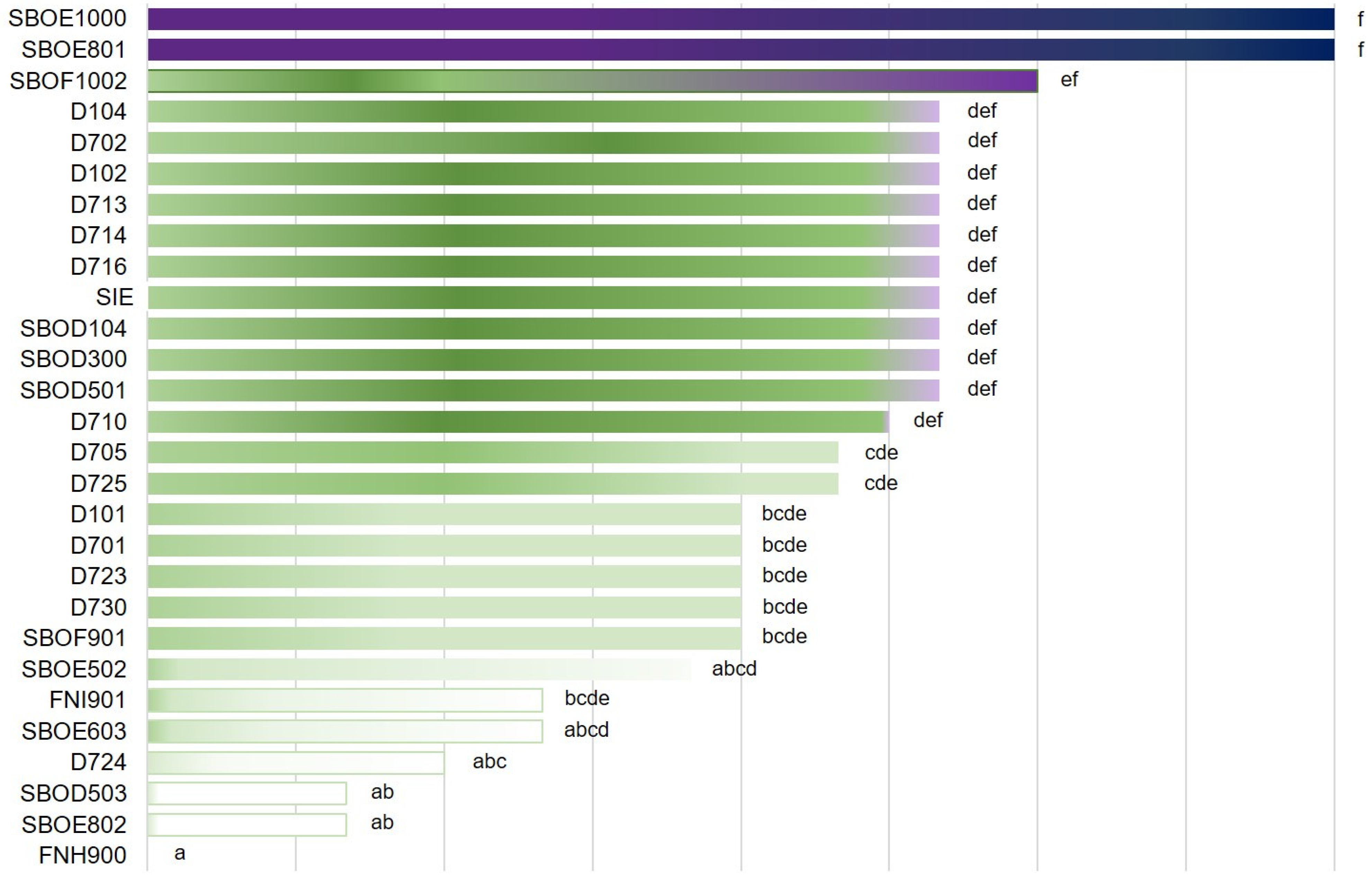
| Culture | Growth at 15 °C 7 day | Growth NaCl 8% 3 day | Growth pH 8 48 h | Growth 30 °C 24 h | pH 24 h | |
|---|---|---|---|---|---|---|
| OD600 | OD600 | OD600 | OD600 | CFU/mL | UpH | |
| D101 | 1.00 ± 1.86 abc | 1.07 ± 0.90 | 4.56 ± 0.36 abc | 6.09 ± 0.31 | 8.96 ± 0.28 | 4.29 ± 0.09 |
| D102 | 1.05 ± 1.20 abc | 2.04 ± 0.65 | 4.13 ± 0.23 abc | 6.14 ± 0.59 | 8.66 ± 0.49 | 4.15 ± 0.02 |
| D104 | 0.26 ± 0.33 a | 2.11 ± 0.27 | 4.33 ± 0.11 abc | 6.28 ± 0.82 | 9.07 ± 0.35 | 4.16 ± 0.05 |
| D701 | 3.56 ± 1.90 abc | 0.50 ± 0.87 | 4.07 ± 0.42 abc | 6.12 ± 1.00 | 8.41 ± 0.33 | 4.18 ± 0.07 |
| D702 | 0.36 ± 0.34 a | 2.01 ± 0.49 | 4.30 ± 0.10 abc | 6.27 ± 0.45 | 8.58 ± 0.50 | 4.15 ± 0.03 |
| D705 | 4.25 ± 0.33 abc | 0.99 ± 1.15 | 4.38 ± 0.05 abc | 6.49 ± 0.78 | 8.81 ± 0.50 | 4.05 ± 0.03 |
| D710 | 4.01 ± 0.77 abc | 1.02 ± 1.43 | 3.95 ± 0.29 abc | 6.51 ± 1.07 | 8.17 ± 0.85 | 4.07 ± 0.04 |
| D713 | 3.38 ± 2.71 abc | 0.80 ± 1.39 | 3.66 ± 0.32 a | 6.58 ± 2.13 | 8.74 ± 0.37 | 4.17 ± 0.19 |
| D714 | 4.02 ± 1.95 abc | 0.00 ± 0.00 | 3.95 ± 0.32 abc | 5.68 ± 2.61 | 8.42 ± 0.67 | 4.34 ± 0.33 |
| D716 | 4.33 ± 1.67 abc | 0.44 ± 0.75 | 4.20 ± 0.39 abc | 6.12 ± 1.11 | 8.28 ± 0.52 | 4.21 ± 0.13 |
| D723 | 4.67 ± 1.62 abc | 0.00 ± 0.00 | 4.00 ± 0.64 abc | 3.80 ± 2.89 | 7.73 ± 0.79 | 4.68 ± 0.59 |
| D724 | 4.62 ± 0.67 abc | 0.57 ± 0.98 | 4.48 ± 0.27 abc | 7.60 ± 0.55 | 8.34 ± 0.56 | 4.13 ± 0.05 |
| D725 | 4.12 ± 1.47 abc | 0.43 ± 0.74 | 3.97 ± 0.64 abc | 6.47 ± 2.27 | 8.33 ± 0.49 | 4.15 ± 0.11 |
| D730 | 3.43 ± 1.32 abc | 0.26 ± 0.45 | 4.34 ± 0.10 abc | 6.24 ± 1.26 | 8.53 ± 0.57 | 4.15 ± 0.17 |
| SIE | 3.74 ± 2.16 abc | 1.09 ± 0.78 | 4.45 ± 0.33 abc | 5.26 ± 0.82 | 8.19 ± 0.78 | 4.42 ± 0.16 |
| FNH900 | 2.80 ± 2.29 abc | 0.67 ± 0.58 | 4.58 ± 0.18 abc | 5.12 ± 1.70 | 8.46 ± 0.24 | 4.54 ± 0.36 |
| FNI901 | 0.44 ± 0.78 ab | 0.00 ± 0.00 | 4.01 ± 0.55 abc | 7.16 ± 0.43 | 8.05 ± 0.14 | 4.12 ± 0.02 |
| SBOD104 | 0.98 ± 0.91 abc | 1.91 ± 0.58 | 4.45 ± 0.11 abc | 5.78 ± 0.82 | 8.65 ± 0.50 | 4.32 ± 0.06 |
| SBOD300 | 1.09 ± 1.12 abc | 2.19 ± 0.29 | 4.47 ± 0.09 abc | 6.22 ± 0.61 | 8.91 ± 0.58 | 4.21 ± 0.08 |
| SBOD501 | 3.01 ± 2.18 abc | 1.85 ± 0.45 | 4.04 ± 0.44 abc | 5.70 ± 1.68 | 8.30 ± 0.31 | 4.29 ± 0.03 |
| SBOD503 | 5.09 ± 0.35 bc | 0.89 ± 1.14 | 4.00 ± 0.90 abc | 4.85 ± 3.23 | 8.57 ± 0.98 | 4.59 ± 0.74 |
| SBOE1000 | 4.60 ± 0.30 abc | 0.35 ± 0.61 | 4.85 ± 0.15 bc | 6.89 ± 1.12 | 8.36 ± 0.55 | 4.15 ± 0.30 |
| SBOE502 | 5.35 ± 0.92 c | 1.26 ± 0.79 | 4.57 ± 0.30 abc | 6.66 ± 1.81 | 7.98 ± 0.21 | 4.23 ± 0.08 |
| SBOE603 | 0.81 ± 1.53 abc | 1.00 ± 1.31 | 4.11 ± 0.46 abc | 6.19 ± 1.09 | 8.43 ± 0.45 | 4.14 ± 0.11 |
| SBOE801 | 2.42 ± 1.90 abc | 0.21 ± 0.32 | 4.78 ± 0.16 abc | 4.50 ± 3.00 | 7.99 ± 1.53 | 4.30 ± 0.14 |
| SBOE802 | 3.86 ± 2.68 abc | 0.35 ± 0.15 | 5.05 ± 0.25 c | 6.06 ± 1.25 | 8.32 ± 0.58 | 4.31 ± 0.05 |
| SBOF1002 | 1.99 ± 2.10 abc | 0.00 ± 0.00 | 4.59 ± 0.45 abc | 4.15 ± 3.46 | 8.40 ± 0.64 | 4.21 ± 0.09 |
| SBOF901 | 3.49 ± 1.74 abc | 0.00 ± 0.00 | 3.86 ± 0.32 ab | 4.38 ± 3.29 | 8.60 ± 0.26 | 4.26 ± 0.11 |
| Culture | Lactic acid D− (g/L) | Lactic acid L+ (g/L) | Total Lactic acid (g/L) |
|---|---|---|---|
| D101 | 6.41 ± 0.82 abcd | 2.53 ± 0.41 abc | 9.55 ± 0.93 ab |
| D102 | 7.30 ± 0.97 bcde | 3.09 ± 0.95 abc | 10.40 ± 0.61 ab |
| D104 | 5.69 ± 0.39 abc | 2.54 ± 0.25 abc | 8.21 ± 0.45 ab |
| D702 | 7.94 ± 1.04 cde | 2.82 ± 0.92 abc | 11.08 ± 0.01 cde |
| D705 | 4.21 ± 0.02 a | 3.43 ± 0.57 abc | 7.54 ± 0.75 cdef |
| D710 | 7.03 ± 0.79 abcde | 4.03 ± 0.94 abc | 11.27 ± 0.44 def |
| D724 | 4.81 ± 1.04 ab | 4.76 ± 0.21 c | 11.19 ± 1.09 def |
| D730 | 7.08 ± 1.00 bcde | 4.31 ± 0.48 bc | 11.39 ± 0.52 def |
| SIE | 4.66 ± 0.48 ab | 2.18 ± 0.94 ab | 8.48 ± 0.90 ab |
| FNI901 | 8.90 ± 0.95 def | 1.94 ± 0.56 a | 10.12 ± 0.15 def |
| SBOE1000 | 11.53 ± 0.23 f | 2.18 ± 0.95 ab | 12.71 ± 0.85 ef |
| SBOE603 | 9.46 ± 0.35e f | 2.36 ± 0.44 ab | 11.82 ± 0.74 f |
| Phenolic Compounds | SIE | DSS | NF |
|---|---|---|---|
| Elenolic acid | 44.44 ± 8.46 a | 31.76 ± 4.24 a | 41.64 ± 7.80 a |
| OH tyrosol | 264.22 ± 5.20 b | 214.51 ± 9.87 a | 217.08 ± 27.75 a |
| Tyrosol | 34.74 ± 2.08 b | 25.25 ± 1.99 a | 25.27 ± 2.78 a |
| 4 OH benzoic acid | 21.46 ± 1.82 a | 16.68 ± 2.64 a | 19.79 ± 6.23 a |
| unknown | 8.87 ± 0.47 b | 5.12 ± 0.51 a | 5.54 ± 1.04 a |
| Paracumaric acid | 9.59 ± 1.29 a | 11.69 ± 2.07 a | 18.85 ± 3.35 b |
| Ferulic acid | 6.11 ± 1.01 ab | 4.97 ± 0.11 b | 7.82 ± 1.09 a |
| Verbascoside | 175.14 ± 16.57 b | 124.57 ± 6.09 a | 130.96 ± 19.31 a |
| Luteolin 7-glucoside | 9.38 ± 2.21 | n.d. | n.d. |
| Oleuropein | n.d. | 17.05 ± 1.75 a | 21.01 ± 3.64 a |
| Quercetin dihydrate | 1.13 ± 0.28 a | 2.41 ± 0.45 a | 3.10 ± 0.55 a |
| Luteolin | 30.50 ± 3.52 b | 15.17 ± 1.25 a | 15.59 ± 3.11 a |
| Apigenin | 2.24 ± 0.23 a | 1.98 ± 0.34 a | 1.98 ± 0.41 a |
| Total phenolic compounds | 3942.93 ± 478.78 a | 3977.64 ± 612.15 a | 4182.20 ± 213.90 a |
| TEAC | 350.36 ± 33.82 a | 339.95 ± 43.38 a | 350.55 ± 63.12 a |
| TPA Parameters | SIE | DSS | NF |
|---|---|---|---|
| Hardness (g) | 2397.31 ± 506.84 a | 2185.96 ± 560.90 a | 2209.41 ± 530.11 a |
| Springiness | 0.64 ± 0.05 a | 0.62 ± 0.06 a | 0.62 ± 0.06 a |
| Cohesiveness | 0.52 ± 0.05 a | 0.50 ± 0.04 a | 0.51 ± 0.05 a |
| Gumminess | 1228.17 ± 236.15 b | 1084.29 ± 251.77 a | 1117.14 ± 239.66 a |
| Chewiness (g/mm) | 782.25 ± 157.29 b | 673.48 ± 168.16 a | 690.67 ± 154.63 a |
| Resilience | 0.27 ± 0.03 a | 0.26 ± 0.03 a | 0.26 ± 0.03 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paba, A.; Chessa, L.; Daga, E.; Campus, M.; Bulla, M.; Angioni, A.; Sedda, P.; Comunian, R. Do Best-Selected Strains Perform Table Olive Fermentation Better than Undefined Biodiverse Starters? A Comparative Study. Foods 2020, 9, 135. https://doi.org/10.3390/foods9020135
Paba A, Chessa L, Daga E, Campus M, Bulla M, Angioni A, Sedda P, Comunian R. Do Best-Selected Strains Perform Table Olive Fermentation Better than Undefined Biodiverse Starters? A Comparative Study. Foods. 2020; 9(2):135. https://doi.org/10.3390/foods9020135
Chicago/Turabian StylePaba, Antonio, Luigi Chessa, Elisabetta Daga, Marco Campus, Monica Bulla, Alberto Angioni, Piergiorgio Sedda, and Roberta Comunian. 2020. "Do Best-Selected Strains Perform Table Olive Fermentation Better than Undefined Biodiverse Starters? A Comparative Study" Foods 9, no. 2: 135. https://doi.org/10.3390/foods9020135
APA StylePaba, A., Chessa, L., Daga, E., Campus, M., Bulla, M., Angioni, A., Sedda, P., & Comunian, R. (2020). Do Best-Selected Strains Perform Table Olive Fermentation Better than Undefined Biodiverse Starters? A Comparative Study. Foods, 9(2), 135. https://doi.org/10.3390/foods9020135





