Effect of Physical and Chemical Treatments on Viability, Sub-Lethal Injury, and Release of Cellular Components from Bacillus clausii and Bacillus coagulans Spores and Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Culture Conditions
2.2. Spore Production
2.3. Chemical and Physical Treatments
2.3.1. Target of the Experiments
2.3.2. Chemical Treatments
2.3.3. Physical Treatments
2.3.4. Experiments
- Chemical: Sterile saline solution (0.9% NaCl), supplemented with lemon extract or citrus extract (250 ppm), and inoculated to 5–6 log cfu/mL; each strain was evaluated separately.
- Physical: Sterile saline solution, inoculated to 5–6 log cfu/mL, treated with high homogenization pressure (150 MPa for 1 time; 150 MPa for 2 times, with a rest time of 90 min and 150 MPa for 3 times), ultrasound (power/time/pulse: 60% 4 min 4 s), or heat stress (95 °C for 5 min).
2.3.5. Modeling and Statistics
2.4. Injury Characterization
2.4.1. Leakage of UV-Absorbing Substances
2.4.2. BSA Protein Assay
2.4.3. DPA Protein Assay
2.4.4. Statistic Indices
3. Results and Discussion
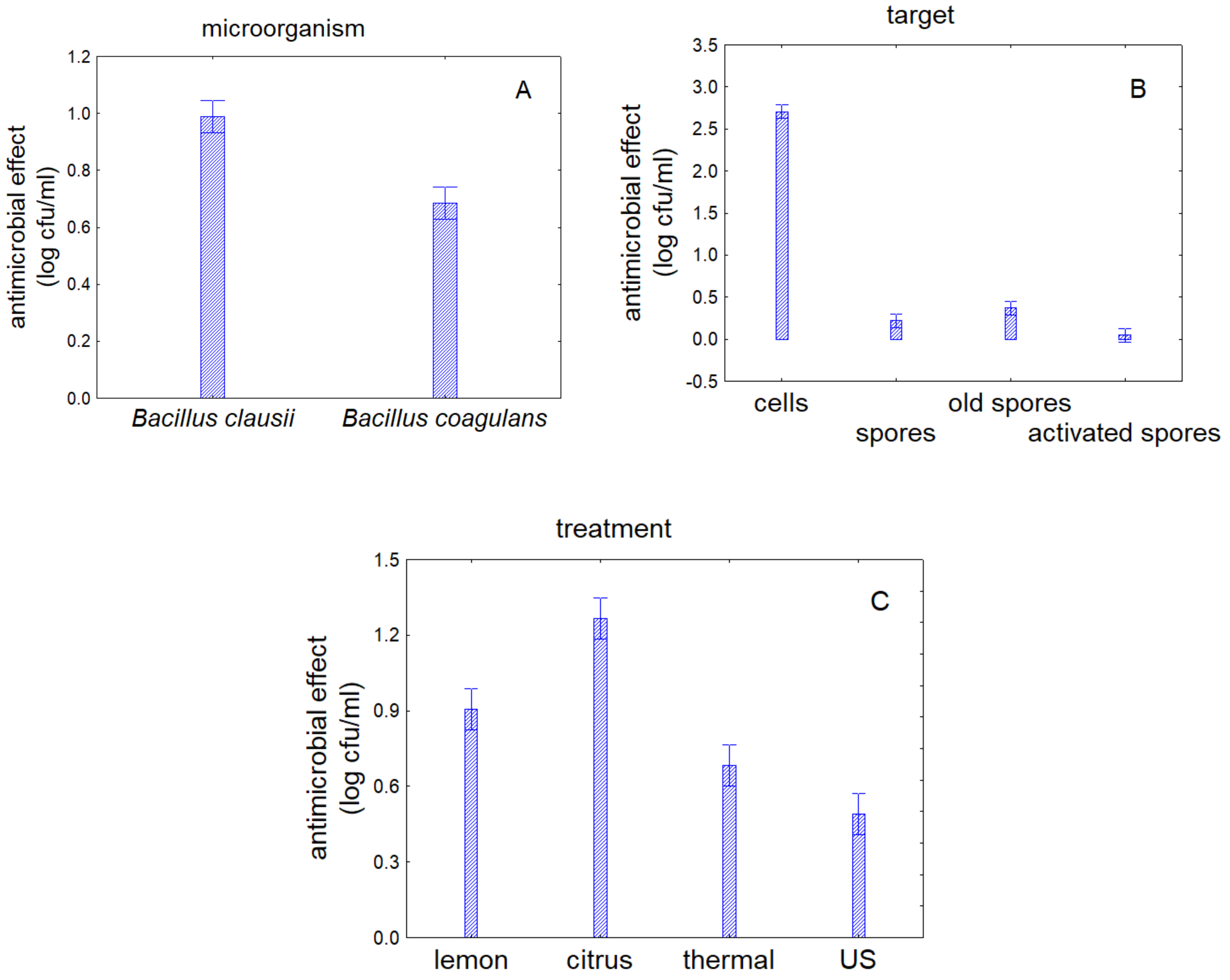
3.1. Antimicrobial Effect
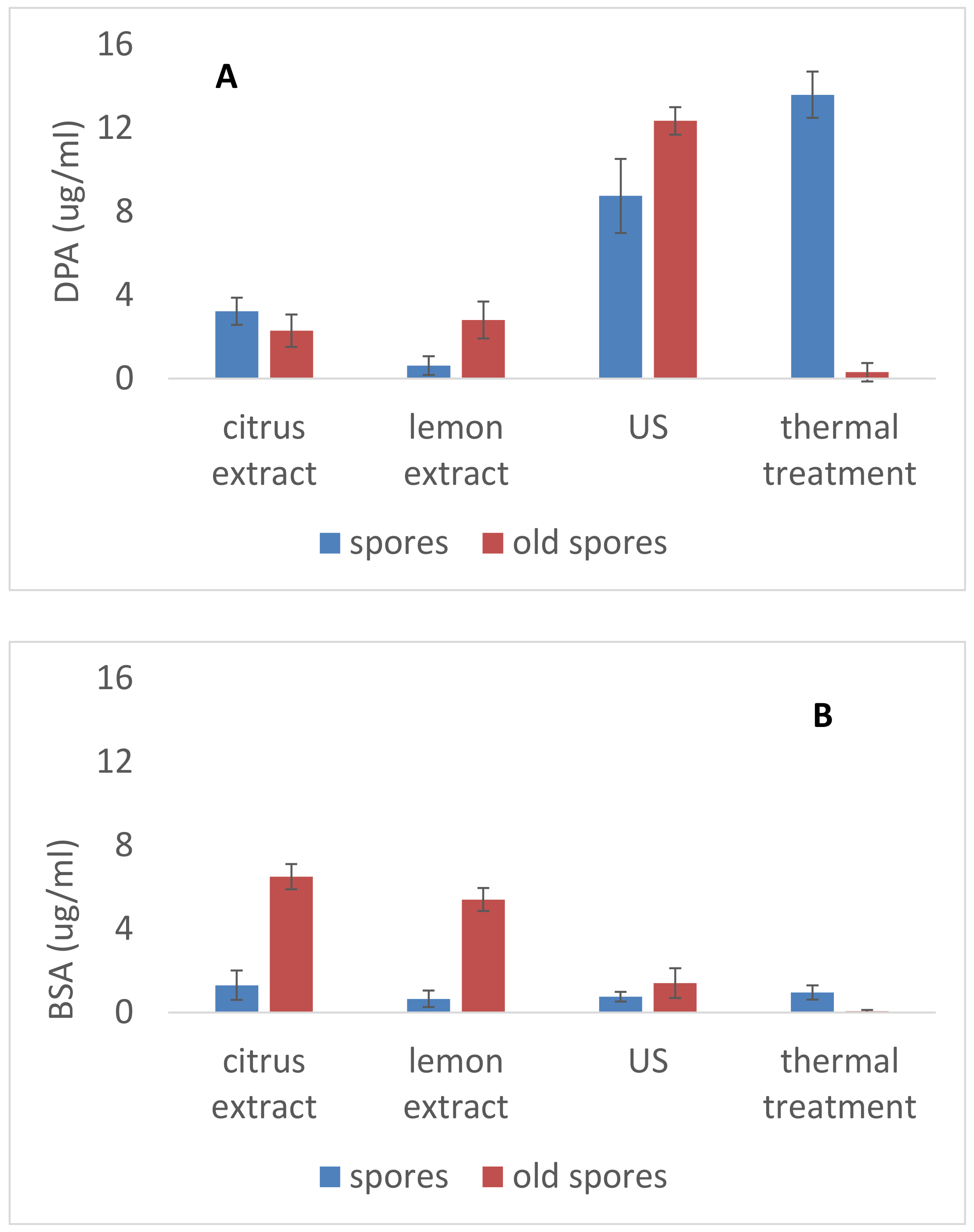
3.2. Sub-Lethal Injury and Injury Characterization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hussain, M.A. Food Contamination: Major challenges of the future. Foods 2016, 5, 21. [Google Scholar] [CrossRef]
- Wu, V.C.H. A review of microbial injury and recovery methods in food. Food Microbiol. 2008, 25, 735–744. [Google Scholar] [CrossRef]
- Hurst, A. Revival of vegetative bacteria after sublethal heating. In The Revival of Injured Microbes; Andrew, M.H., Russell, A.D., Eds.; Academic Press Inc.: Orlando, FL, USA, 1984; pp. 77–103. [Google Scholar]
- Remize, F. Spore-forming bacteria. In The Microbiological Quality of Food. Foodborne Spoilers; Bevilacqua, A., Corbo, M.R., Sinigaglia, M., Eds.; Woodhead Publishing: Duxford, UK, 2017; pp. 99–120. [Google Scholar]
- Soni, A.; Oey, I.; Silcock, P.; Bremer, P. Bacillus spores in the food industry: A review on resistance and response to novel inactivation technologies. Compr. Rev. Food Sci. Food Saf. 2016, 15, 1139–1148. [Google Scholar] [CrossRef]
- Busta, F.F. Practical implications of injured microorganisms in food. J. Milk Food Technol. 1976, 39, 138–145. [Google Scholar] [CrossRef]
- Achir, N.; Dhuique-Mayer, C.; Hadjal, T.; Madani, K.; Pain, J.P.; Dornier, M. Pasteurization of citrus juices with ohmic heating to preserve the carotenoid profile. Innov. Food Sci. Emerg. Technol. 2016, 33, 397–404. [Google Scholar] [CrossRef]
- Petruzzi, L.; Campaniello, D.; Speranza, B.; Corbo, M.R.; Sinigaglia, M.; Bevilacqua, A. Thermal treatments for fruit and vegetable juices and beverages: A literature overview. Compr. Rev. Food Sci. Food Saf. 2017, 16, 668–691. [Google Scholar] [CrossRef]
- Miller, F.A.; Silva, C.L.M. Thermal treatment effects in fruit juices. In Advances in Fruit Processing Technologies; Rodrigues, S., Fernandes, F.A.N., Eds.; CRC Press: Boca Raton, FL, USA, 2012; pp. 363–383. [Google Scholar]
- Lv, R.; Wang, D.; Zou, M.; Wang, W.; Ma, X.; Chen, W.; Zhou, J.; Ding, T.; Ye, X.; Liu, D. Analysis of Bacillus cereus cell viability, sublethal injury, and death induced by mild thermal treatment. J. Food Saf. 2018, 39, e12581. [Google Scholar]
- Patrignani, F.; Lanciotti, R. Applications of high and ultra high pressure homogenization for food safety. Front. Microbiol. 2016, 7, 1132. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, A.; Campaniello, D.; Speranza, B.; Altieri, C.; Sinigaglia, M.; Corbo, M.R. Two non-thermal technologies for food safety and quality: Ultrasound in comparison with high-pressure homogenization. Effects on microorganisms, advances and possibilities. J. Food Prot. 2019, 12, 2049–2064. [Google Scholar] [CrossRef] [PubMed]
- Prakash, B.; Kedia, A.; Kumar Mishra, P.; Dubey, N.K. Plant essential oils as food preservatives to control moulds, mycotoxin contamination and oxidative deterioration of agri-food commodities—Potentials and challenges. Food Control 2015, 47, 381–391. [Google Scholar] [CrossRef]
- Amor, G.; Caputo, L.; La Storia, A.; De Feo, V.; Mauriello, G.; Fechtali, T. Chemical composition and antimicrobial activity of Artemisia herba-alba and Origanum majorana essential oils from Morocco. Molecules 2019, 24, 4021. [Google Scholar] [CrossRef] [PubMed]
- Ungaretti Haberbeck, L.; da Silva Riehl, C.A.; Martins Salomão, B.C.; Falcão de Aragão, G.M. Bacillus coagulans spore inactivation through the application of oregano essential oil and heat. LWT-Food Sci. Technol. 2012, 46, 267–273. [Google Scholar] [CrossRef][Green Version]
- De Castro, J.A.; Villa-Real Guno, M.J.; Perez, M.O. Bacillus clausii as adjunctive treatment for acute community-acquired diarrhea among Filipino children: A large-scale, multicenter, open-label study (CODDLE). Trop. Dis. Travel Med. Vaccines 2019, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Virto, R.; Manas, P.; Alvarez, I.; Condon, S.; Raso, J. Membrane damage and microbial inactivation by chlorine in the absence and presence of a chlorine-demanding substrate. Appl. Environ. Microbiol. 2005, 71, 5022–5028. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Yu, X.; Hu, A.; Zhang, L.; Jin, Y.; Abid, M. Ultrasonic disruption of yeast cells: Underlying mechanism and effects of processing parameters. Innov. Food Sci. Emerg. Technol. 2015, 28, 59–65. [Google Scholar] [CrossRef]
- Chaves-Lòpez, C.; Lanciotti, R.; Serio, A.; Paparella, A.; Guerzoni, M.E.; Suzzi, G. Effect of high-pressure homogenization applied alone or in combination with other mild physical or chemical stresses on Bacillus cereus and Bacillus subtilis spore viability. Food Control 2009, 20, 691–695. [Google Scholar] [CrossRef]
- Fisher, K.; Phillips, C. Potential antimicrobial uses of essential oils in food: Is citrus the answer? Trends Food Sci. Technol. 2008, 19, 156–164. [Google Scholar] [CrossRef]
- Settanni, L.; Palazzolo, E.; Guarrasi, V.; Aleo, A.; Mammina, C.; Moschetti, G.; Germanà, M.A. Inhibition of foodborne pathogen bacteria by essential oils extracted from citrus fruits cultivated in Sicily. Food Control 2012, 26, 326–330. [Google Scholar] [CrossRef]
- Bevilacqua, A.; Campaniello, D.; Speranza, B.; Sinigaglia, M.; Corbo, M.R. Control of Alicyclobacillus acidoterrestris in apple juice by citrus extracts and a mild heat-treatment. Food Control 2013, 31, 553–559. [Google Scholar] [CrossRef]
- Joyce, E.; Phull, S.S.; Lorimer, J.P.; Mason, T.J. The development and evaluation of ultrasound for the treatment of bacterial suspensions. A study of frequency, power and sonication time on cultured Bacillus species. Ultrason. Sonochem. 2003, 10, 315–318. [Google Scholar] [CrossRef]
- Riesz, P.; Kondo, T. Free radical formation induced by ultrasound and its biological implications. Free Radic. Biol. Med. 1992, 13, 247–270. [Google Scholar] [CrossRef]
- Ojha, K.S.; Mason, T.J.; O’Donnell, C.P.; Kerry, J.P.; Tiwari, B.K. Ultrasound technology for food fermentation applications. Ultrason. Sonochem. 2017, 34, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Hayley, A.L.; Palombo, E.A. Activity of essential oils against Bacillus subtilis spores. J. Microbiol. Biotechnol. 2009, 19, 1590–1595. [Google Scholar]
- Cox, S.D.; Mann, C.M.; Markham, J.L.; Gustafson, J.E.; Warmington, J.R.; Wyllie, S.G. Determining the antimicrobial actions of tea tree oil. Molecules 2001, 6, 87–91. [Google Scholar] [CrossRef]
- Cortezzo, D.E.; Koziol-Dube, K.; Setlow, B.; Setlow, P. Treatment with oxidizing agents damages the inner membrane of spores of Bacillus subtilis and sensitizes spores to subsequent stress. J. Appl. Microbiol. 2004, 97, 838–852. [Google Scholar] [CrossRef]
- Hurst, A. Bacterial injury: A review. Can. J. Microbiol. 1977, 23, 935–944. [Google Scholar] [CrossRef]
- Palumbo, S.A. Injury in emerging foodborne pathogens and their detection. In Injured Index and Pathogenic Bacteria: Occurrence and Detection in Food, Water and Feeds; Ray, B., Ed.; CRC Press, Inc.: Boca Raton, FL, USA, 1989; pp. 115–132. [Google Scholar]
- Jay, J.M.; Loessner, M.J.; Golden, D.A. Modern Food Microbiology; Springer: New York, NY, USA, 2008. [Google Scholar]
- Bevilacqua, A.; Ciuffreda, E.; Sinigaglia, M.; Corbo, M.R. Spore inactivation and DPA release in Alicyclobacillus acidoterrestris under different stress conditions. Food Microbiol. 2015, 46, 299–306. [Google Scholar] [CrossRef]
- Klijn, N.; Herman, L.; Langeveld, L.; Vaerewijck, M.; Wagendorp, A.A.; Huemer, I.; Weerkamp, A.H. Genotypical and phenotypical characterization of Bacillus sporothermodurans strains, surviving UHT sterilisation. Int. Dairy J. 1997, 7, 421–428. [Google Scholar] [CrossRef]
- Setlow, P. Spores of Bacillus subtilis: Resistance of bacterial spores. In Bacterial Stress Response; Storz, G., Hengge-Aronis, R., Eds.; American Society of Microbiology: Washington, DC, USA, 2000; pp. 217–230. [Google Scholar]
- Warth, A.D. Heat-stability of Bacillus-cereus enzymes within spores and in extracts. J. Bacteriol. 1980, 143, 27–34. [Google Scholar] [CrossRef]

| Treatments | Conditions |
|---|---|
| Lemon extract | Saline solution supplemented with lemon extract (250 ppm) |
| Citrus extract | Saline solution supplemented with citrus extract (250 ppm) |
| Heating | Saline solution treated at 95 °C for 5 min |
| Ultrasound | Saline solution treated through US at a power of 60% for 4 min; pulse set to 4 s |
| Homogenization | Saline solution treated at 150 MPa for 1 time |
| Saline solution treated at 150 MPa for 2 times | |
| Saline solution treated at 150 MPa for 3 times | |
| Targets and experiments | Each treatment was tested on spores, old spores, activated spores, and cells; thus, 20 experiments were done (5 treatments × 4 targets) |
| Analysis | The microbiological analyses were done immediately after the treatment (T0), and after 1 (T1) or 2 days (T2); counts were done on two media: Optimal media and restrictive media (the details are in the text) |
| SS | Degree of Freedom | MS | Fisher Test | |
|---|---|---|---|---|
| Microorganism (1) | 89.76 | 1 | 89.76 | 1689.04 |
| Target (2) | 2.96 | 1 | 2.96 | 55.62 |
| Treatment (3) | 150.99 | 3 | 50.33 | 947.03 |
| Time (4) | 10.65 | 3 | 3.55 | 66.78 |
| Microorganism × target | 1.87 | 1 | 1.87 | 35.20 |
| Microorganism × treatment | 0.75 | 3 | 0.25 | 4.71 |
| Target × treatment | 6.10 | 3 | 2.03 | 38.26 |
| Microorganism × time | - 1 | - | - | - |
| Target × time | 0.11 | 1 | 0.11 | 2.10 |
| Treatment × time | 0.77 | 3 | 0.26 | 4.86 |
| Microorganism × target × treatment | 0.46 | 3 | 0.15 | 2.90 |
| Microorganism × target × time | 5.78 | 9 | 0.64 | 12.08 |
| Microorganism × treatment × time | 0.48 | 3 | 0.16 | 2.99 |
| Target × treatment × time | 0.86 | 3 | 0.29 | 5.37 |
| 1 × 2 × 3 × 4 | 1.16 | 9 | 0.13 | 2.42 |
| Error | 1.87 | 9 | 0.21 | 3.92 |
| B. coagulans | |||||
| Treatment | Time (day) | Cells | Spores | Old Spores | Activated Spores |
| Lemon extract | 0 | - 1 | - | 4.85 ± 0.40 | - |
| 2 | 33.37 ± 3.82 | - | - | - | |
| Citrus extract | 0 | 44.50 ± 10.27 | - | - | - |
| 2 | - | - | - | 18.27 ± 0.33 | |
| Thermal treatment | 0 | - | 4.45 ± 0.11 | 2.40 ± 0.40 | - |
| 2 | - | 10.51 ± 0.90 | - | - | |
| US | 0 | 19.20 ± 3.11 | - | 3.61 ± 0.13 | - |
| 2 | - | - | - | - | |
| B. clausii | |||||
| Treatment | Time (day) | Cells | Spores | Old Spores | Activated Spores |
| Lemon extract | 0 | - | - | - | - |
| 2 | - | - | - | - | |
| Citrus extract | 0 | / 2 | - | - | - |
| 2 | / | - | 7.77 ± 1.67 | - | |
| Thermal treatment | 0 | - | - | 7.72 ± 0.41 | - |
| 2 | 24.08 ± 6.45 | - | - | - | |
| US | 0 | 24.50 ± 1.15 | - | 15.69 ± 0.94 | - |
| 2 | 39.54 ± 2.81 | - | - | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bevilacqua, A.; Petruzzi, L.; Sinigaglia, M.; Speranza, B.; Campaniello, D.; Ciuffreda, E.; Corbo, M.R. Effect of Physical and Chemical Treatments on Viability, Sub-Lethal Injury, and Release of Cellular Components from Bacillus clausii and Bacillus coagulans Spores and Cells. Foods 2020, 9, 1814. https://doi.org/10.3390/foods9121814
Bevilacqua A, Petruzzi L, Sinigaglia M, Speranza B, Campaniello D, Ciuffreda E, Corbo MR. Effect of Physical and Chemical Treatments on Viability, Sub-Lethal Injury, and Release of Cellular Components from Bacillus clausii and Bacillus coagulans Spores and Cells. Foods. 2020; 9(12):1814. https://doi.org/10.3390/foods9121814
Chicago/Turabian StyleBevilacqua, Antonio, Leonardo Petruzzi, Milena Sinigaglia, Barbara Speranza, Daniela Campaniello, Emanuela Ciuffreda, and Maria Rosaria Corbo. 2020. "Effect of Physical and Chemical Treatments on Viability, Sub-Lethal Injury, and Release of Cellular Components from Bacillus clausii and Bacillus coagulans Spores and Cells" Foods 9, no. 12: 1814. https://doi.org/10.3390/foods9121814
APA StyleBevilacqua, A., Petruzzi, L., Sinigaglia, M., Speranza, B., Campaniello, D., Ciuffreda, E., & Corbo, M. R. (2020). Effect of Physical and Chemical Treatments on Viability, Sub-Lethal Injury, and Release of Cellular Components from Bacillus clausii and Bacillus coagulans Spores and Cells. Foods, 9(12), 1814. https://doi.org/10.3390/foods9121814







