Effect of Brine Concentrations on the Bacteriological and Chemical Quality and Histamine Content of Brined and Dried Milkfish
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Reagents
2.3. Brine-Salting and Drying of Milkfish
2.4. Determination of pH Value, Moisture Content, Water Activity, and Salt Content
2.5. Determination of Total Volatile Basic Nitrogen (TVBN) and Thiobarbituric Acid (TBA)
2.6. Microbiological Analysis
2.7. Histamine Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Chemical and Bacteriological Quality of the Brined and Dried Milkfish Samples
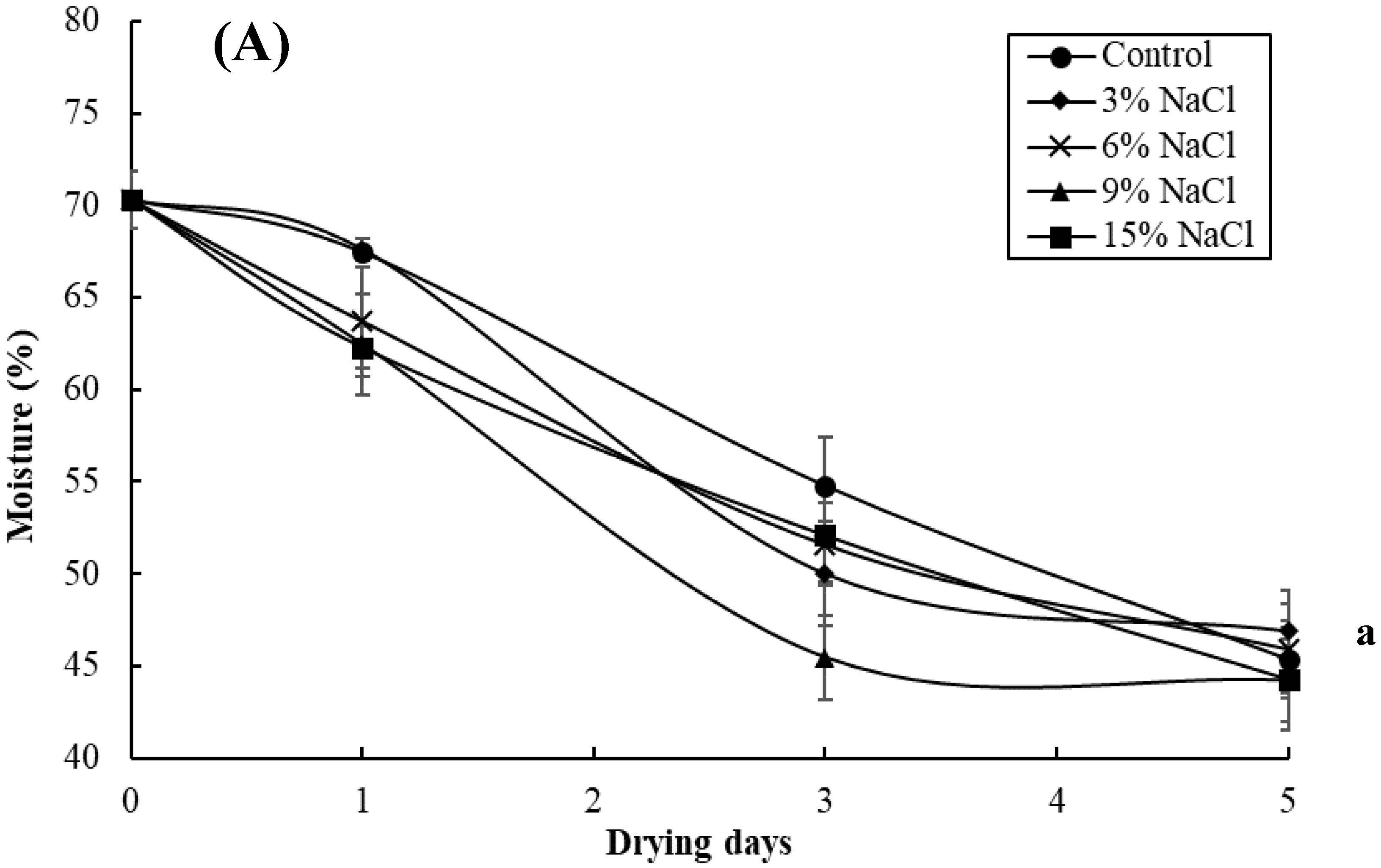
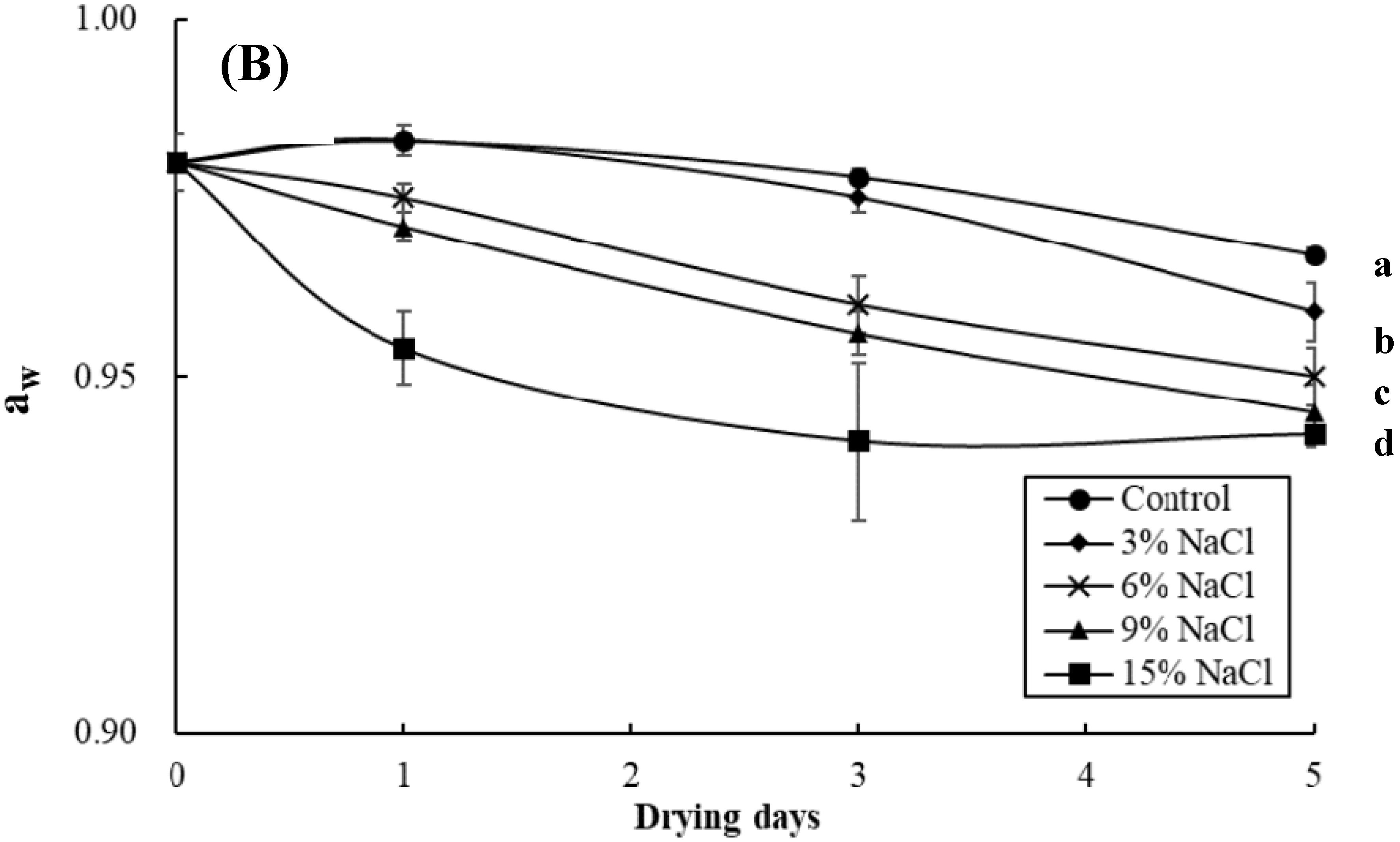
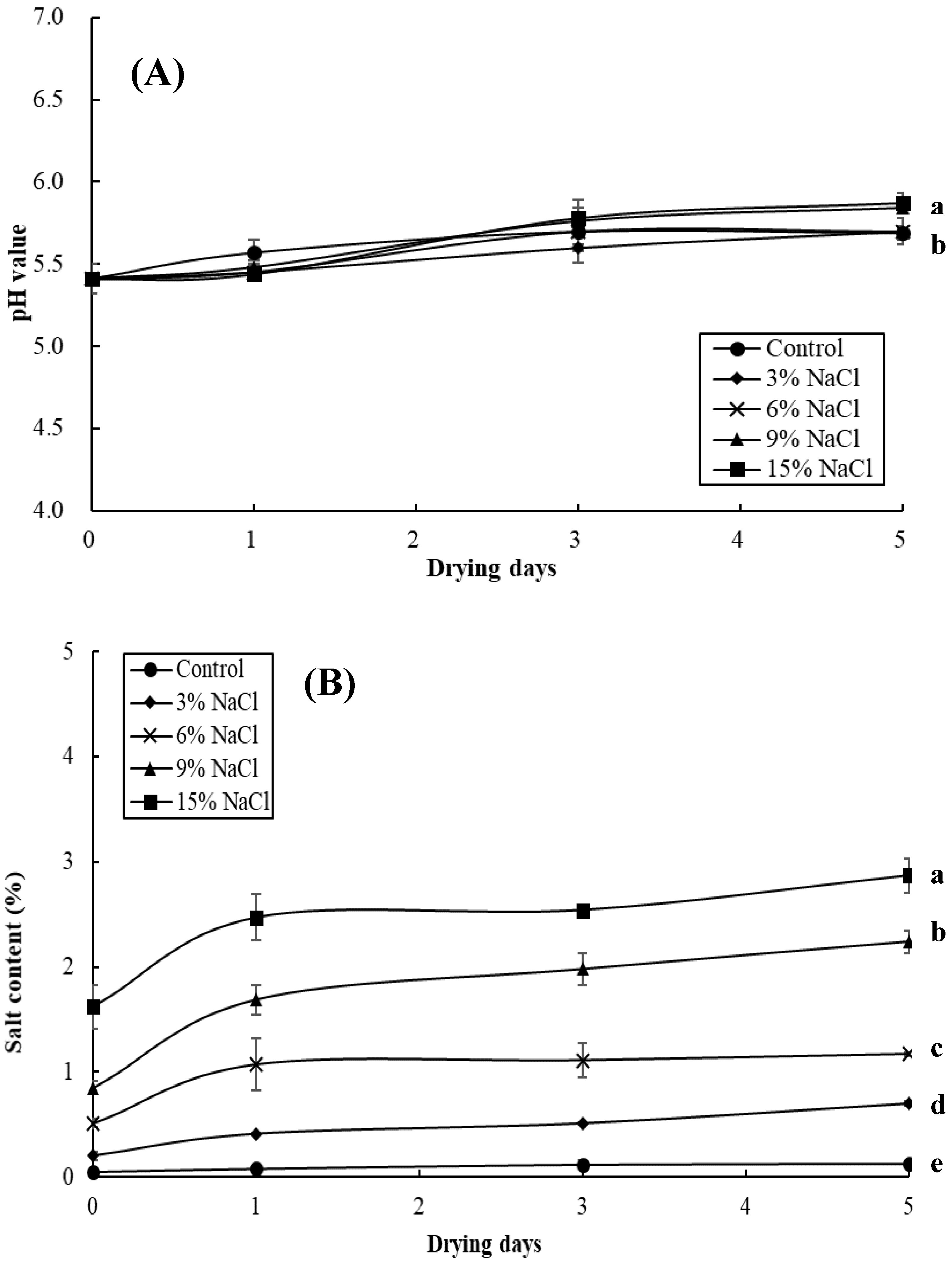
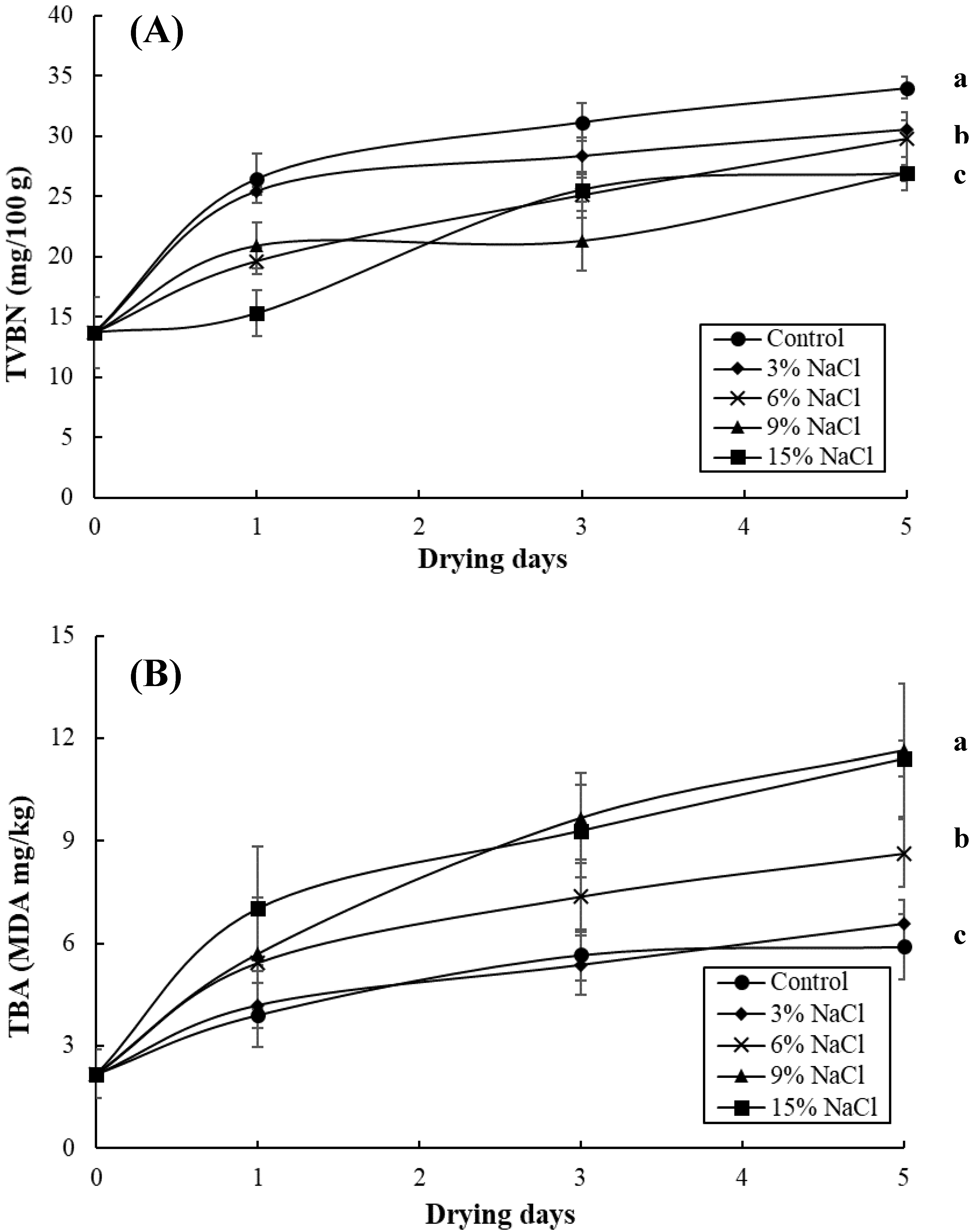
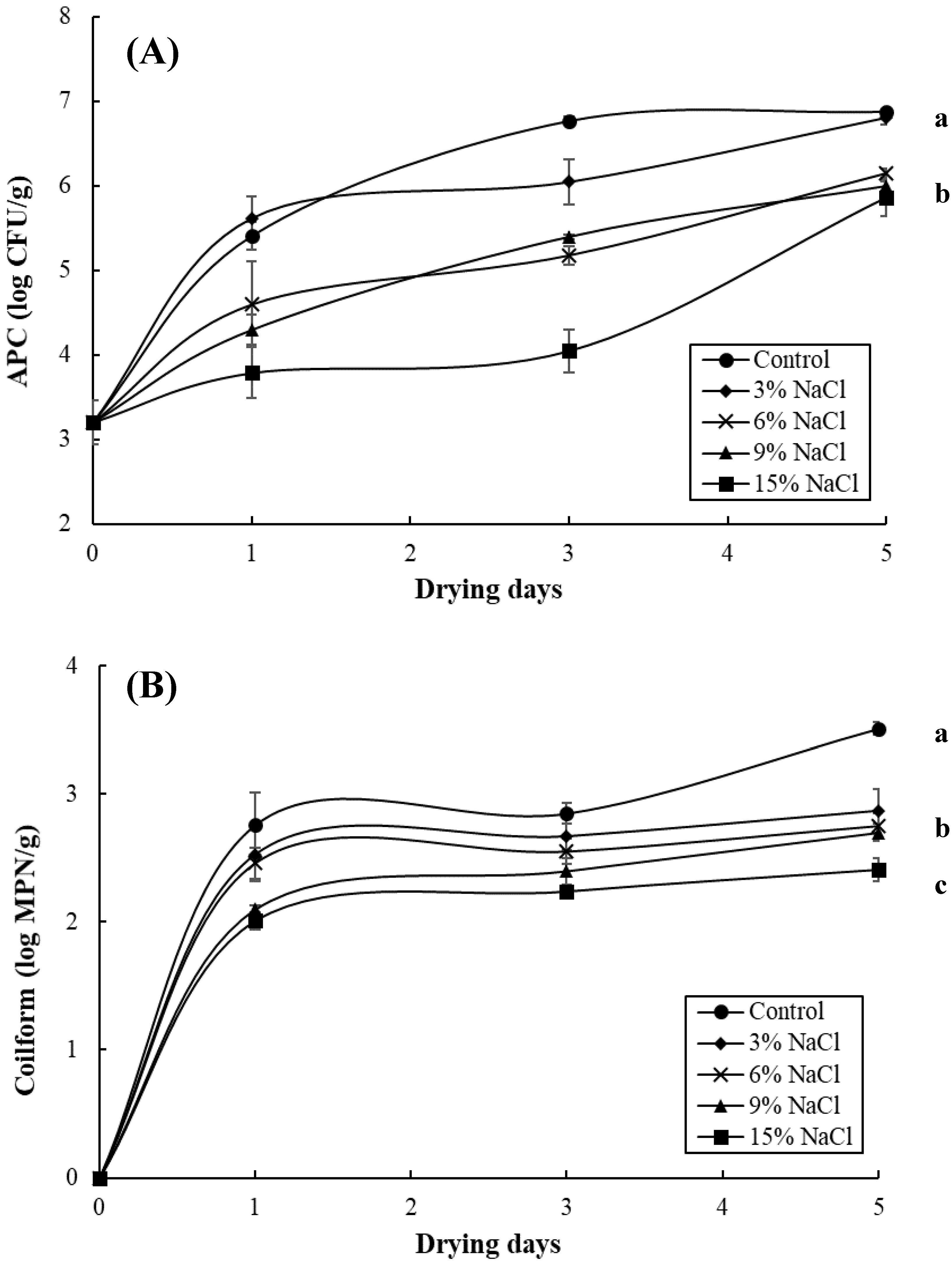
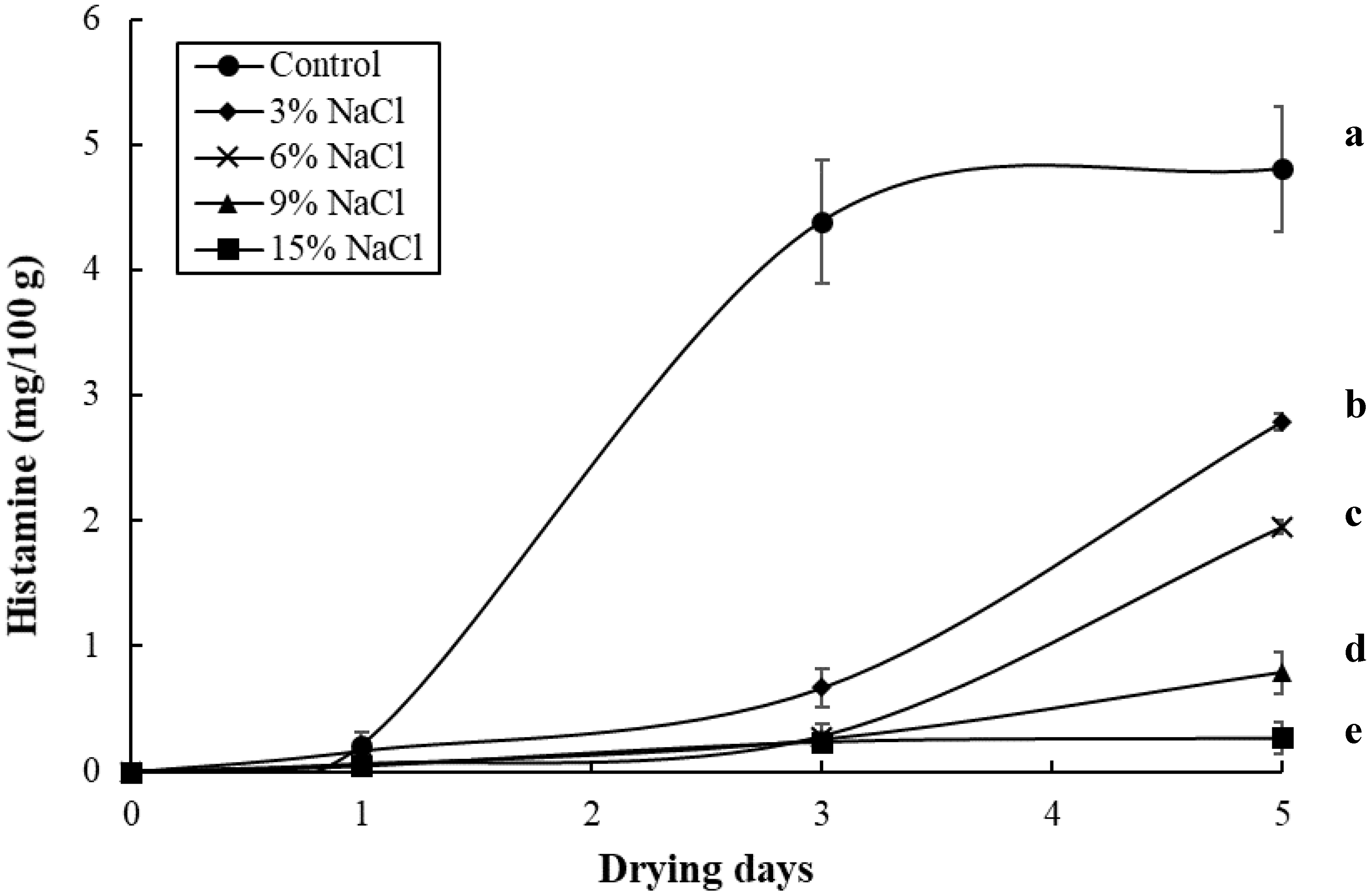
3.2. Effect of Brine Concentrations on the Quality of Brined and Dried Milkfish
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hugerford, J.M. Scombroid poisoning: A review. Toxicon 2010, 56, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.C.; Kung, H.F.; Chen, W.C.; Lin, W.F.; Hwang, D.F.; Lee, Y.C.; Tsai, Y.H. Determination of histamine and histamine-forming bacteria in tuna dumpling implicated in a food-borne poisoning. Food Chem. 2008, 106, 612–618. [Google Scholar] [CrossRef]
- Chen, H.C.; Huang, Y.R.; Hsu, H.H.; Lin, C.S.; Chen, W.C.; Lin, C.M.; Tsai, Y.H. Determination of histamine and biogenic amines in fish cubes (Tetrapturus angustirostris) implicated in a food-borne poisoning. Food Control 2010, 21, 13–18. [Google Scholar] [CrossRef]
- Chang, S.C.; Kung, H.F.; Chen, H.C.; Lin, C.S.; Tsai, Y.H. Determination of histamine and bacterial isolation in swordfish fillets (Xiphias gladius) implicated in a food borne poisoning. Food Control 2008, 19, 16–21. [Google Scholar] [CrossRef]
- Tsai, Y.H.; Kung, H.F.; Lee, T.M.; Chen, H.C.; Chou, S.S.; Wei, C.I.; Hwang, D.F. Determination of histamine in canned mackerel implicated in a food borne poisoning. Food Control 2005, 16, 579–585. [Google Scholar] [CrossRef]
- Tsai, Y.H.; Kung, H.F.; Chen, H.C.; Chang, S.C.; Hsu, H.H.; Wei, C.I. Determination of histamine and histamine-forming bacteria in dried milkfish (Chanos chanos) implicated in a food-borne poisoning. Food Chem. 2007, 105, 1289–1296. [Google Scholar] [CrossRef]
- Hsieh, S.L.; Chen, Y.N.; Kuo, C.M. Physiological responses, desaturase activity, and fatty acid composition in milkfish (Chanos chanos) under cold acclimation. Aquaculture 2003, 220, 903–918. [Google Scholar] [CrossRef]
- Chiang, F.S.; Sun, C.H.; Yu, J.M. Technical efficiency analysis of milkfish (Chanos chanos) production in Taiwan- an application of the stochastic frontier production function. Aquaculture 2004, 230, 99–116. [Google Scholar] [CrossRef]
- Chiou, T.K.; Shiau, C.Y.; Chai, T.J. Extractive nitrogenous components of cultured milkfish and tilapia. Nippon Suisan Gakk. 1990, 56, 1313–1317. [Google Scholar] [CrossRef]
- Lee, Y.C.; Kung, H.F.; Wu, C.H.; Hsu, H.M.; Chen, H.C.; Huang, T.C.; Tsai, Y.H. Determination of histamine in milkfish stick implicated in a foodborne poisoning. J. Food Drug Anal. 2016, 24, 63–71. [Google Scholar] [CrossRef]
- Hwang, C.C.; Kung, H.F.; Lee, Y.C.; Wen, S.Y.; Chen, P.Y.; Tsen, D.I.; Tsai, Y.H. Histamine fish poisoning and histamine production by Raoultella ornithinolytica in milkfish surimi. J. Food Prot. 2020, 83, 874–880. [Google Scholar] [CrossRef]
- Hsu, H.H.; Chuang, T.C.; Lin, H.C.; Huang, Y.R.; Lin, C.M.; Kung, H.F.; Tsai, Y.H. Histamine content and histamine-forming bacteria in dried milkfish (Chanos chanos) products. Food Chem. 2009, 114, 933–938. [Google Scholar] [CrossRef]
- Gallart-Jornet, L.; Rustad, T.; Barat, J.M.; Fito, P.; Escriche, I. Effect of superchilled storage on the freshness and salting behaviour of Atlantic salmon (Salmo salar) fillets. Food Chem. 2007, 103, 1268–1281. [Google Scholar] [CrossRef]
- Yang, W.; Shi, W.; Qu, Y.; Wang, Z.; Shen, S.; Tu, L.; Huang, H.; Wu, H. Research on the quality changes of grass carp during brine salting. Food Sci. Nutri. 2020, 8, 2968–2983. [Google Scholar] [CrossRef]
- Nooralabettu, K.P. Effect of sun drying and artificial drying of fresh, salted Bombay duck (Harpodon neherius) on the physical characteristics of the product. J. Aquat. Food Prod. Technol. 2008, 17, 99–116. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 21st ed.; AOAC International: Arlington, VA, USA, 2019. [Google Scholar]
- Cobb, B.F.; Alaniz, I.; Thompson, C.A. Biochemical and microbial studies on shrimp: Volatile nitrogen and amino nitrogen analysis. J. Food Sci. 1973, 38, 431–435. [Google Scholar] [CrossRef]
- Faustman, C.; Spechtm, S.M.; Malkus, L.A.; Kinsman, D.M. Pigment oxidation in ground veal: Influence of lipid oxidation, iron and zinc. Meat Sci. 1992, 31, 351–362. [Google Scholar] [CrossRef]
- FDA. Bacteriological Analytical Manual; AOAC International: Arlington, VA, USA, 1998.
- Bartholomev, B.A.; Berry, P.R.; Rodhouse, J.C.; Gilhouse, R.J. Scombrotoxic fish poisoning in Britain: Features of over 250 suspected incidents from 1976 to 1986. Epidemiol. Infect. 1987, 99, 775–782. [Google Scholar] [CrossRef]
- Hwang, C.C.; Lin, C.M.; Kung, H.F.; Huang, Y.L.; Hwang, D.F.; Su, Y.C.; Tsai, Y.H. Effect of salt concentrations and drying methods on the quality and formation of histamine in dried milkfish (Chanos chanos). Food Chem. 2012, 135, 839–844. [Google Scholar] [CrossRef]
- Arulkumar, A.; Paramasiam, S.; Rameshthangam, P.; Rabie, M.A. Changes on biogenic, volatile amines and microbial quality of the blue swimmer crab (Portunus pelagicus) muscle during storage. J. Food Sci. Technol. 2017, 54, 2503–2511. [Google Scholar] [CrossRef]
- Connell, J.J. Methods of assessing and selecting for quality. In Control of Fish Quality, 3rd ed.; Fishing News Books: Oxford, UK, 1990; pp. 122–150. [Google Scholar]
- Mohamed, S.B.; Mendes, R.; Slama, R.B.; Oliveira, P.; Silva, H.A.; Bakhrouf, A. Changes in bacterial counts and biogenic amines during the ripening of salted anchovy (Engraulis encrasicholus). J. Food Nutr. Res. 2016, 5, 318–325. [Google Scholar]
- Fernandez, J.; Perez-Alvarez, J.A.; Fernandez-Lopez, J.A. Thiobarbituric acid test for monitoring lipid oxidation in meat. Food Chem. 1997, 59, 343–353. [Google Scholar] [CrossRef]
- Yanar, Y.; Celik, M.; Akamca, E. Effects of brine concentration on shelf-life of hot-smoked tilapia (Oreochromis niloticus) stored at 4 °C. Food Chem. 2006, 97, 244–247. [Google Scholar] [CrossRef]
- Kanner, J.; Harel, S.; Jaffe, R. Lipid peroxidation in muscle food as affected by NaCl. J. Agri. Food Chem. 1991, 39, 1017–1021. [Google Scholar] [CrossRef]
- Gwak, H.; Eun, J.B. Changes in the chemical characteristics of Gulbi, salted and dried yellow Corvenia, during drying at different temperatures. J. Aquat. Food Prod. Technol. 2010, 19, 274–283. [Google Scholar] [CrossRef]
| Sample Sources | Number of Samples | pH | Moisture (%) | Water Activity | Salt Content (%) | TVBN (mg/100 g) | APC (log CFU/g) | Coliform (MPN/g) | E. coli (MPN/g) | Histamine (mg/100 g) |
|---|---|---|---|---|---|---|---|---|---|---|
| A | 6 | 5.74~5.83 (5.78 ± 0.04) A | 38.27~65.37 (51.78 ± 9.28) B | 0.89~0.97 (0.94 ± 0.03) B | 2.47~4.37 (3.23 ± 0.78) A | 8.86~17.36 (12.79 ± 3.25) B | 3.51~7.65 (5.50 ± 1.05) B | <3~70 (42 ± 25) B | <3 | 0.34~4.9 (1.3 ± 1.8) C |
| B | 5 | 5.67~6.05 (5.76 ± 0.17) A | 52.31~60.18 (55.41 ± 3.23) AB | 0.97~0.98 (0.98 ± 0.01) A | 0.16~1.88 (0.80 ± 0.77) C | 13.86~19.32 (16.06 ± 2.08) A | 5.87~8.03 (6.62 ± 0.83) A | <3~40 (25 ± 13) B | <3 | 0.62~301 (79 ± 57) A |
| C | 5 | 5.80~5.95 (5.87 ± 0.07) A | 54.68~69.78 (64.17 ± 5.95) A | 0.97~0.98 (0.98 ± 0.01) A | 0.61~1.02 (0.86 ± 0.18) C | 10.85~13.93 (12.35 ± 1.39) B | 4.18~5.92 (5.32 ± 0.66) B | <3~240 (90 ± 130) B | <3 | 0.16~0.31 (0.20 ± 0.11) C |
| D | 4 | 5.75~5.82 (5.78 ± 0.03) A | 51.07~54.12 (52.46 ± 1.26) B | 0.95~0.96 (0.96 ± 0.01) AB | 1.47~1.80 (1.68 ± 0.15) B | 15.40~19.88 (16.82 ± 2.66) A | 5.80~8.25 (6.33 ± 0.60) A | 20~>2400 (356 ± 47) A | <3 | 0.24~19 (4.9 ± 4.8) B |
| Content of Histamine (mg/100 g) | Brined and Dried Milkfish Products | |
|---|---|---|
| Number of Samples | % of Samples | |
| <4.9 | 15 | 75 |
| 5.0–49.9 | 3 | 15 |
| 50.0–99.9 | 1 | 5 |
| >100 | 1 | 5 |
| Total | 20 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, C.-C.; Lee, Y.-C.; Huang, C.-Y.; Kung, H.-F.; Cheng, H.-H.; Tsai, Y.-H. Effect of Brine Concentrations on the Bacteriological and Chemical Quality and Histamine Content of Brined and Dried Milkfish. Foods 2020, 9, 1597. https://doi.org/10.3390/foods9111597
Hwang C-C, Lee Y-C, Huang C-Y, Kung H-F, Cheng H-H, Tsai Y-H. Effect of Brine Concentrations on the Bacteriological and Chemical Quality and Histamine Content of Brined and Dried Milkfish. Foods. 2020; 9(11):1597. https://doi.org/10.3390/foods9111597
Chicago/Turabian StyleHwang, Chiu-Chu, Yi-Chen Lee, Chung-Yung Huang, Hsien-Feng Kung, Hung-Hui Cheng, and Yung-Hsiang Tsai. 2020. "Effect of Brine Concentrations on the Bacteriological and Chemical Quality and Histamine Content of Brined and Dried Milkfish" Foods 9, no. 11: 1597. https://doi.org/10.3390/foods9111597
APA StyleHwang, C.-C., Lee, Y.-C., Huang, C.-Y., Kung, H.-F., Cheng, H.-H., & Tsai, Y.-H. (2020). Effect of Brine Concentrations on the Bacteriological and Chemical Quality and Histamine Content of Brined and Dried Milkfish. Foods, 9(11), 1597. https://doi.org/10.3390/foods9111597






