Effects of a Small Increase in Carbon Dioxide Pressure during Fermentation on Wine Aroma
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Trials
- Atmospheric pressure (0 bar, the control sample—the P0 sample),
- Four levels of pressures slightly higher than the atmospheric pressure (0.2, 0.5, 0.7, and 1 bar—the P02, P05, P07, and P1 samples, respectively)
2.2. Determination of Volatile Compounds
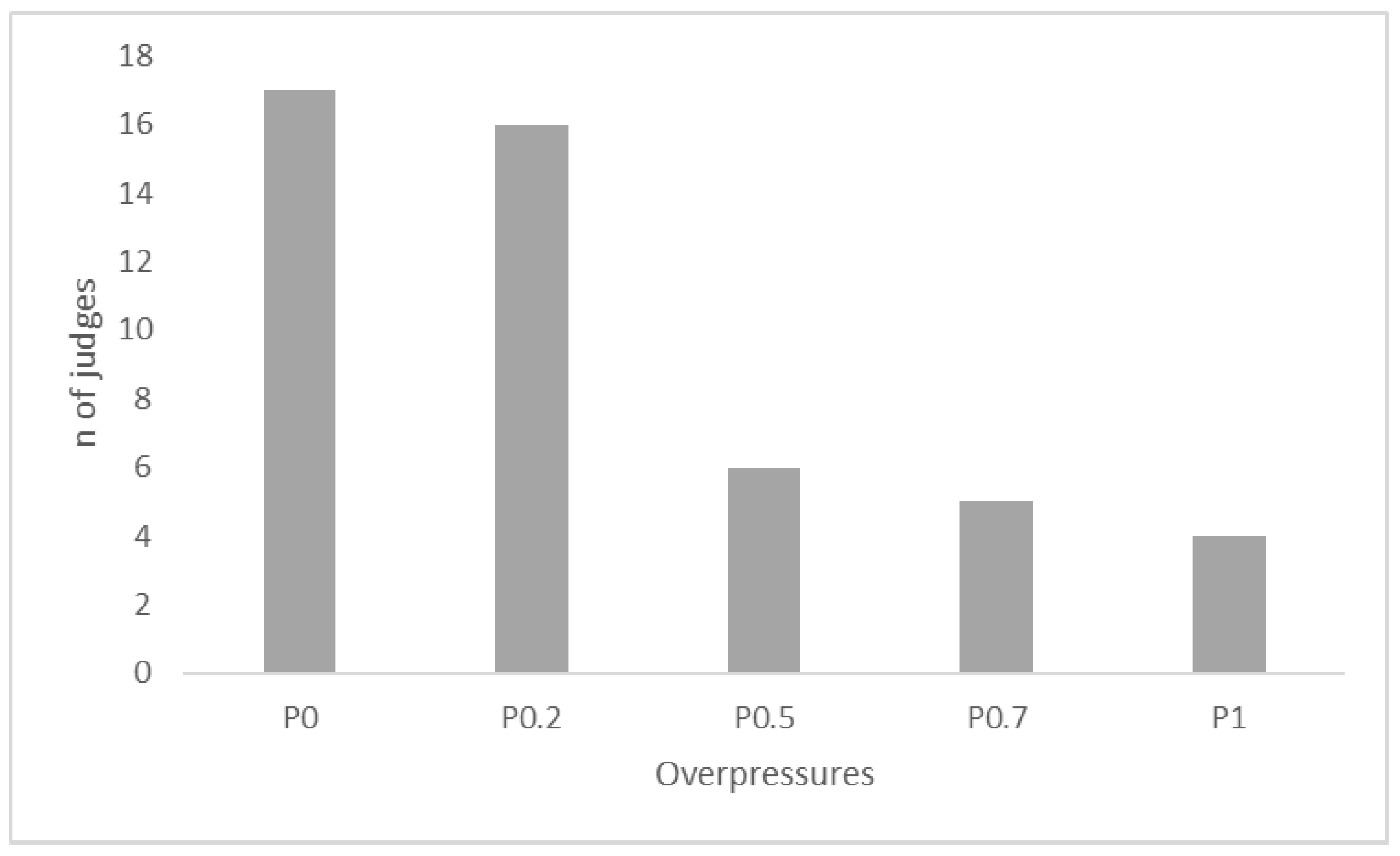
2.3. Sensory Tests
2.4. Statistical Analysis
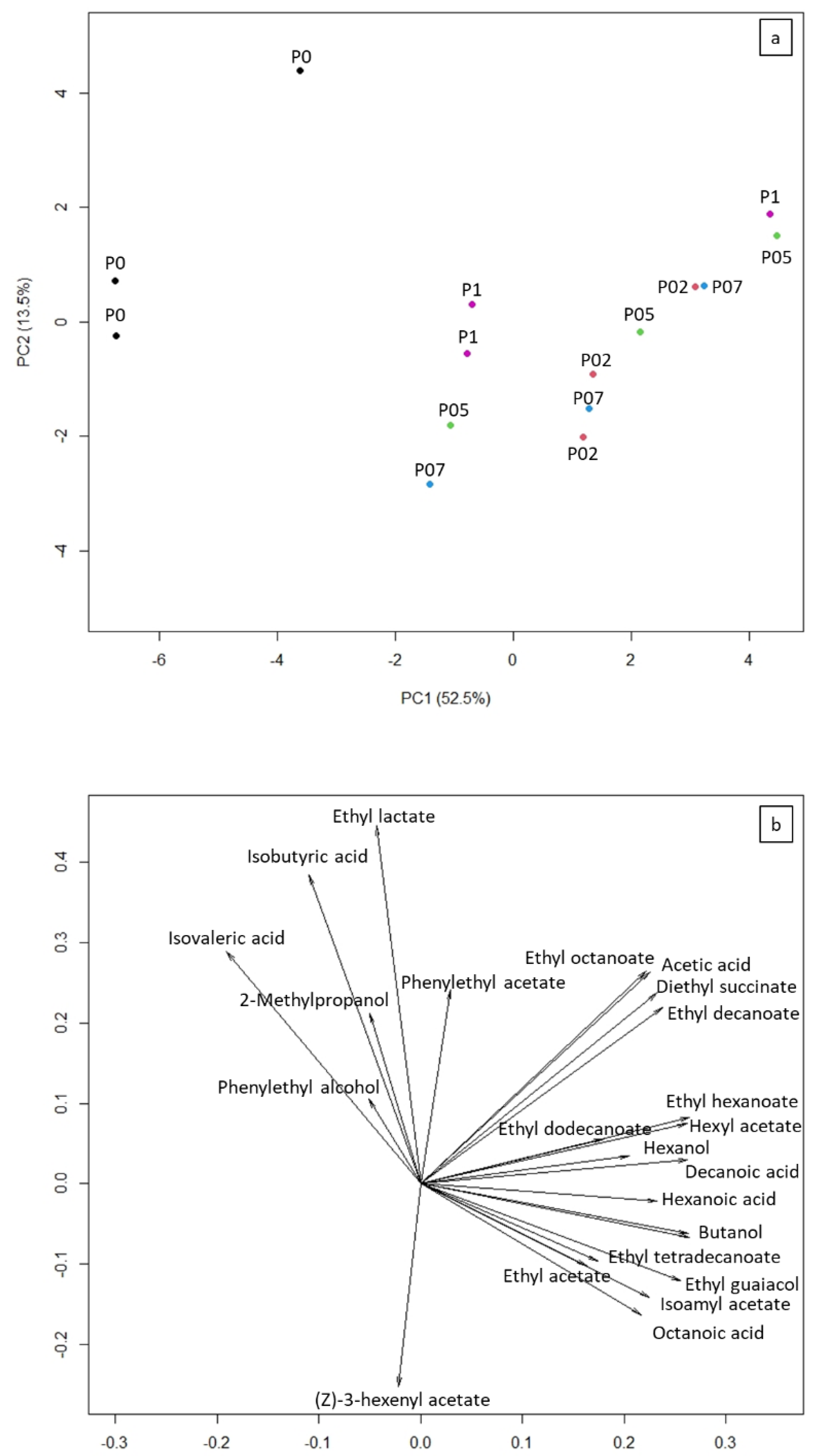
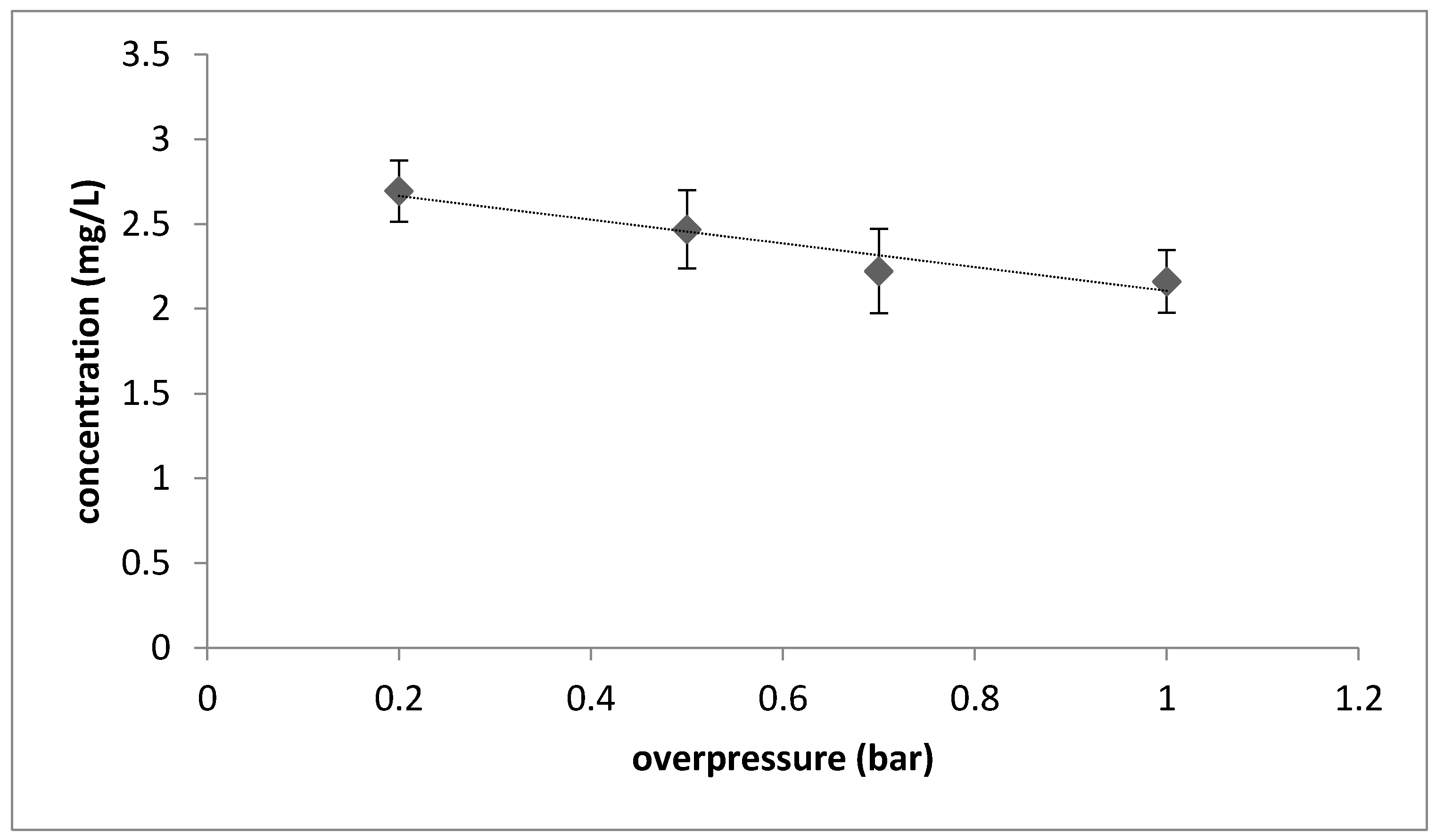
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sablayrolles, J.-M. Control of alcoholic fermentation in winemaking: Current situation and prospect. Food Res. Int. 2009, 42, 418–424. [Google Scholar] [CrossRef]
- Moreno, J.J.; Cerpa-Calderón, F.; Cohen, S.D.; Fang, Y.; Qian, M.; Kennedy, J.A. Effect of postharvest dehydration on the composition of pinot noir grapes (Vitis vinifera L.) and wine. Food Chem. 2008, 109, 755–762. [Google Scholar] [CrossRef]
- Guerrini, L.; Angeloni, G.; Baldi, F.; Parenti, A. Thermal effects of pump-overs during red wine fermentation. Appl. Therm. Eng. 2017, 112, 621–626. [Google Scholar] [CrossRef]
- Guerrini, L.; Masella, P.; Angeloni, G.; Calamai, L.; Spinelli, S.; Di Blasi, S.; Parenti, A. Harvest of Sangiovese grapes: The influence of material other than grape and unripe berries on wine quality. Eur. Food Res. Tech. 2018, 244, 1487–1496. [Google Scholar] [CrossRef]
- Spranger, M.I.; Clímaco, M.C.; Sun, B.; Eiriz, N.; Fortunato, C.; Nunes, A.; Belchior, A.P. Differentiation of red winemaking technologies by phenolic and volatile composition. Anal. Chim. Act. 2004, 513, 151–161. [Google Scholar] [CrossRef]
- Ward, S.C.; Petrie, P.R.; Johnson, T.E.; Boss, P.K.; Bastian, S.E.P. Unripe Berries and Petioles in Vitis vinifera cv. Cabernet Sauvignon fermentations affect sensory and chemical profiles. Am. J. Enol. Vitic. 2015, 4, 435–443. [Google Scholar] [CrossRef]
- Guerrini, L.; Masella, P.; Spugnoli, P.; Spinelli, S.; Calamai, L.; Parenti, A. A condenser to recover organic volatile compounds during vinification. Am. J. Enol. Vitic. 2016, 67, 163–168. [Google Scholar] [CrossRef]
- Lezaeta, A.; Bordeu, E.; Agosin, E.; Pérez-Correa, J.R.; Varela, P. White wines aroma recovery and enrichment: Sensory-led aroma selection and consumer perception. Food Res. Int. 2018, 108, 595–603. [Google Scholar] [CrossRef]
- Río Segade, S.; Vilanova, M.; Giacosa, S.; Perrone, I.; Chitarra, W.; Pollon, M.; Rolle, L. Ozone improves the aromatic fingerprint of white grapes. Sci. Rep. 2017, 7, 1–16. [Google Scholar] [CrossRef]
- Robinson, A.L.; Boss, P.K.; Solomon, P.S.; Trengove, R.D.; Heymann, H.; Ebeler, S.E. Origins of grape and wine aroma. Part 1. Chemical components and viticultural impacts. Am. J. Enol. Vitic. 2014, 65, 1–24. [Google Scholar] [CrossRef]
- Salinas, M.R.; Alonso, G.L.; Navarro, G.; Pardo, F.; Jimeno, J.; Huerta, M.D. Evolution of the aromatic composition of wines undergoing carbonic maceration under different aging conditions. Am. J. Enol. Vitic. 1996, 47, 134–144. [Google Scholar]
- Tesniere, C.; Flanzy, C. Carbonic maceration wines: Characteristics and winemaking process. Adv. Food Nutr. Res. 2011, 63, 1–15. [Google Scholar] [PubMed]
- Kemp, B.; Alexandre, H.; Robillard, B.; Marchal, R. Effect of production phase on bottle-fermented sparkling wine quality. J. Agric. Food Chem. 2015, 63, 19–38. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, R.; García-Martínez, T.; Puig-Pujol, A.; Mauricio, J.C.; Moreno, J. Changes in sparkling wine aroma during the second fermentation under CO2 pressure in sealed bottle. Food Chem. 2017, 237, 1030–1040. [Google Scholar] [CrossRef]
- Welke, J.E.; Zanus, M.; Lazzarotto, M.; Pulgati, F.H.; Zini, C.A. Main differences between volatiles of sparkling and base wines accessed through comprehensive two-dimensional gas chromatography with time-of-flight mass spectrometric detection and chemometric tools. Food Chem. 2014, 164, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Renger, B.R.S.; Van Hateren, S.H.; Luyben, K.C.A.M. The formation of esters and higher alcohols during brewery fermentation; the effect of carbon dioxide pressure. J. Instit. Brew. 1992, 98, 509–513. [Google Scholar] [CrossRef]
- Sacchi, K.L.; Bisson, L.F.; Adams, D.O. A review of the effect of winemaking techniques on phenolic extraction in red wines. Am. J. Enol. Vitic. 2005, 56, 197–206. [Google Scholar]
- Domizio, P.; Lencioni, L.; Calamai, L.; Portaro, L.; Bisson, L.F. Evaluation of the yeast Schizosaccharomyces japonicus for use in wine production. Am. J. Enol Vitic 2018, 69, 266–277. [Google Scholar] [CrossRef]
- Francis, I.L.; Newton, J.L. Determining wine aroma from compositional data. Aust. J. Grape Wine R 2005, 11, 114–126. [Google Scholar] [CrossRef]
- UNI. U590A2520; Analisi Sensoriale. Metodo Triangolare. (Regulation in Italian); Italian Unification Institute: Milano, Italy, 2001. [Google Scholar]
- Mamede, M.E.O.; Cardello, H.M.A.B.; Pastore, G.M. Evaluation of an aroma similar to that of sparkling wine: Sensory and gas chromatography analyses of fermented grape musts. Food Chem. 2005, 89, 63–68. [Google Scholar] [CrossRef]
- Pineau, B.; Barbe, J.C.; Van Leeuwen, C.; Dubourdieu, D. Examples of perceptive interactions involved in specific “Red-” and “Black-berry” aromas in red wines. J. Agric. Food Chem. 2009, 57, 3702–3708. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, L.; Angeloni, G.; Masella, P.; Calamai, L.; Parenti, A. A technological solution to modulate the aroma profile during beer fermentation. Food Bioprocess. Technol. 2018, 11, 1259–1266. [Google Scholar] [CrossRef]
- Knatchbull, F.B.; Slaughter, J.C. The effect of low CO2 pressures on the absorption of amino acids and production of flavour-active volatiles by yeast. J. Inst. Brew. 1987, 93, 420–424. [Google Scholar] [CrossRef]
- Landaud, B.S.; Latrille, E.; Corrieu, G.; Genie, U.M.R.; Pug, I.I.N.A. Top pressure and temperature control the fusel alcohol/Ester ratio through yeast growth in beer fermentation. J. Inst. Brew. 2001, 107, 107–117. [Google Scholar] [CrossRef]
- Kruger, L.; Pickerell, A.T.W.; Axcell, B. The sensitivity of different brewing yeast strains to carbon dioxide inhibition: Fermentation and production of flavor-active volatile compounds. J. Inst. Brew. 1992, 98, 133–138. [Google Scholar] [CrossRef]
- Dennis, E.G.; Keyzers, R.A.; Kalua, C.M.; Maffei, S.M.; Nicholson, E.L.; Boss, P.K. Grape contribution to wine aroma: Production of hexyl acetate, octyl acetate, and benzyl acetate during yeast fermentation is dependent upon precursors in the must. J. Agric. Food Chem. 2012, 60, 2638–2646. [Google Scholar] [CrossRef] [PubMed]
- Gürbüz, O.; Rouseff, J.M.; Rouseff, R.L. Comparison of aroma volatiles in commercial merlot and Cabernet Sauvignon wines using gas chromatography-olfactometry and gas chromatography-mass spectrometry. J. Agric. Food Chem. 2006, 54, 3990–3996. [Google Scholar] [CrossRef] [PubMed]
- Riu-Aumatell, M.; Bosch-Fusté, J.; López-Tamames, E.; Buxaderas, S. Development of volatile compounds of cava (Spanish sparkling wine) during long ageing time in contact with lees. Food Chem. 2006, 95, 237–242. [Google Scholar] [CrossRef]
| Parameter | Concentration |
|---|---|
| Total sugar (g/L) | 215 |
| Glucose (g/L) | 113 |
| Fructose (g/L) | 102 |
| pH | 3.37 |
| Titratable acidity (g/L sulfuric acid) | 4.43 |
| Free SO2 (mg/L) | 0 |
| Total SO2 (mg/L) | 22 |
| Malic acid (g/L) | 1.5 |
| Ammonia nitrogen (mg/kg) | 56 |
| Amino acid nitrogen (mg/kg) | 117 |
| Compound Name | Calibration Equation | R2 | ISTD | OT (mg/L) | Ref. | Descriptors |
|---|---|---|---|---|---|---|
| Ethyl Acetate | y = 0.014675x + 0.066574 | 0.99 | i | 12.26 | [7] | pineapple, varnish, balsamic |
| 2-Methylpropanol | y = 0.039772x − 0.003727 | 0.98 | ii | 830 | [14] | fusel alcohol, ripe fruit |
| Isoamyl acetate | y = 0.100689 | 0.92 | iii | 0.03 | [7] | sweet, fruity, banana, solvent |
| 1-butanol | y = 0.089765x − 0.040793 | 0.99 | ii | - | - | - |
| Ethyl Hexanoate | y = 2.209025x + 0.590842 | 0.96 | iv | 0.014 | [7] | fruit, fat |
| p-cymene | y = 0.120378x − 0.002342 | 0.97 | iii | [18,19] | citrus, tymus | |
| Hexyl Acetate | y = 0.263802x + 0.008024 | 0.98 | iv | 1.5 | [14] | green apple |
| (Z)-3- Hexenyl Acetate | y = 52.46746x + 1.253990 | 0.98 | v | 1.9 | [18,19] | herbaceous |
| Ethyl Lactate | y = 0.036195x − 0.029498 | 0.99 | iv | 154.7 | [7] | |
| 1-hexanol | y = 0.262175x + 0.006559 | 0.99 | vi | 8 | [7] | grass just cut |
| Etyhl Octanoate | y = 2.037874x − 0.694484 | 0.99 | iv | 0.005 | [7] | pineapple, pear, soapy |
| Acetic Acid | y = 0.049014x − 0.002893 | 0.99 | vii | 200 | [20] | sour, pungent, vinegar |
| Isobutyric Acid | y = 0.027054x − 0.006984 | 0.98 | iv | 2.3 | [20] | rancid, butter, cheese |
| Ethyl Decanoate | y = 0.020602x + 0.001370 | 0.99 | viii | 0.2 | [7] | sweet, fruity, nuts and dried fruit |
| Diethyl Succinate | y = 0.109274x + 0.009381 | 0.92 | viii | 200 | [7] | overripe melon, lavender |
| Isovaleric Acid | y = 0.039342x + 0.006740 | 0.87 | iv | 0.03 | [20] | sweat, acid, rancid |
| Hexanoic Acid | y = 0.123790x − 0.069588 | 0.99 | ix | 0.43 | [20] | sweat |
| 2-Phenylethyl Acetate | y = 3.255536x − 1.461896 | 0.99 | ii | 0.25 | [7] | fruity, rose, sweet, honey |
| Ethyl Dodecanoate | y = 0.076558x + 0.414303 | 0.96 | iv | 1.5 | [14] | sweet |
| Phenylethyl Alcohol | y = 0.767459x − 1.055210 | 0.99 | x | 10 | [14] | rose talc, honey |
| b-ionone | y = 2.266933x + 0.034350 | 0.99 | iii | 0.45 | [18,19] | flowers, violet |
| Ethyl guaiacol | y = 2.111488x + 0.032071 | 0.98 | x | 0.03 | [20] | spice, clove |
| Octanoic Acid | y = 0.590406x + 0.210770 | 0.97 | ix | 0.5 | [14] | fatty, waxy, rancid, oily |
| Ethyl Tetradecanoate | y = 0.703345x − 0.001324 | 0.99 | iv | 2 | [14] | sweet fruit, butter, fatty odor |
| Compound Name | Samples | ||||
|---|---|---|---|---|---|
| P0 (0.0 Bar) | P02 (0.2 Bar) | P05 (0.5 Bar) | P07 (0.7 Bar) | P1 (1.0 Bar) | |
| Alcohols | |||||
| 1-Butanol | 1.77 ± 0.55 a | 8.88 ± 1.71 b | 9.15 ± 2.82 b | 7.95 ± 1.70 b | 7.69 ± 1.58 b |
| 2-Methylpropanol | 127.37 ± 52.25 a | 74.02 ± 32.38 a | 53.99 ± 2.95 a | 120.13 ± 71.37 a | 110.22 ± 44.90 a |
| 1-hexanol | 0.62 ± 0.19 a | 0.91 ± 0.14 a | 0.91 ± 0.06 a | 0.92 ± 0.07 a | 1.11 ± 0.15 a |
| Phenylethyl alcohol | 85.96 ± 19.08 a | 76.76 ± 24.38 a | 73.50 ± 18.56 a | 69.51 ± 6.98 a | 91.01 ± 15.64 a |
| Acids | |||||
| Acetic acid | 74.03 ± 42.55 a | 114.21 ± 34.10 a | 119.19 ± 55.04 a | 109.07 ± 22.33 a | 122.87 ± 34.11 a |
| Hexanoic acid | 0.07 ± 0.12 a | 0.96 ± 0.17 b | 0.86 ± 0.24 b | 0.79 ± 0.48 b | 0.85 ± 0.67 b |
| Octanoic acid | 0.36 ± 0.11 a | 2.27 ± 0.62 b | 2.04 ± 0.45 b | 2.08 ± 0.62 b | 1.95 ± 0.92 b |
| Decanoic acid | 0.78 ± 0.53 a | 3.14 ± 0.47 b | 2.95 ± 0.89 b | 2.47 ± 1.15 b | 2.46 ± 1.10 b |
| Isobutyric acid | 2.55 ± 0.43 a | 1.91 ± 0.24 b | 1.65 ± 0.29 b | 1.23 ± 0.49 b | 1.73 ± 0.22 b |
| Isovaleric acid | 2.05 ± 0.35 a | 1.01 ± 0.43 ab | 0.80 ± 0.17 ab | 0.65 ± 0.24 c | 0.83 ± 0.09 bc |
| Esters | |||||
| Ethyl acetate | 6.78 ± 5.61 a | 22.42 ± 2.26 b | 16.57 ± 0.72 b | 21.72 ± 1.93 b | 16.66 ± 6.44 b |
| Ethyl hexanoate | 3.15 ± 3.17 a | 9.86 ± 1.55 b | 10.54 ± 2.76 b | 9.01 ± 3.18 b | 9.95 ± 1.42 b |
| Hexyl acetate | 0.73 ± 1.11 a | 3.39 ± 0.90 b | 3.57 ± 1.31 b | 2.92 ± 1.04 b | 2.96 ± 0.41 b |
| (Z)-3-hexenyl acetate | 0.03 ± 0.01 a | 0.05 ± 0.03 a | 0.03 ± 0.00 a | 0.03 ± 0.02 a | 0.03 ± 0.01 a |
| Ethyl lactate | 5.65 ± 2.47 a | 3.18 ± 1.23 a | 4.48 ± 1.57 a | 3.14 ± 0.61 a | 5.14 ± 0.91 a |
| Ethyl octanoate | 1.33 ± 0.77 a | 2.06 ± 0.68 a | 2.16 ± 1.09 a | 1.99 ± 0.42 a | 2.23 ± 0.66 a |
| Ethyl decanoate | 28.60 ± 20.82 a | 66.91 ± 27.35 a | 76.89 ± 60.00 a | 61.53 ± 28.90 a | 70.28 ± 42.93 a |
| Ethyl dodecanoate | 0.37 ± 0.08 b | 0.49 ± 0.07 a | 0.47 ± 0.08 a | 0.42 ± 0.10 a | 0.43 ± 0.07 a |
| Ethyl tetradecanoate | 0.12 ± 0.03 b | 0.32 ± 0.15 a | 0.29 ± 0.05 a | 0.22 ± 0.06 a | 0.26 ± 0.05 a |
| Diethyl succinate | 0.03 ± 0.03 a | 0.07 ± 0.04 a | 0.08 ± 0.07 a | 0.07 ± 0.04 a | 0.08 ± 0.06 a |
| 2-Phenylethyl acetate | 3.77 ± 2.77 a | 3.48 ± 0.88 a | 2.94 ± 1.17 a | 2.48 ± 0.93 b | 2.93 ± 2.14 a |
| Ethyl guaiacol | 0.08 ± 0.01 a | 0.16 ± 0.08 a | 0.17 ± 0.08 a | 0.22 ± 0.09 a | 0.13 ± 0.07 a |
| Isoamyl acetate | 0.48 ± 0.18 a | 2.69 ± 0.36 b | 2.47 ± 0.46 b | 2.22 ± 0.5 b | 2.16 ± 0.37 b |
| Aldehydes | |||||
| Furfural | 0.00 ± 0.01 a | 0.01 ± 0.01 a | 0.01 ± 0.02 a | 0.01 ± 0.01 a | 0.01 ± 0.01 a |
| Terpenes and C-13 norisoprenoids | |||||
| p-cymene | 0.02 ± 0.00 a | 0.02 ± 0.00 a | 0.02 ± 0.00 a | 0.02 ± 0.00 a | 0.02 ± 0.00 a |
| β-ionone | 0.01 ± 0.00 a | 0.01 ± 0.00 a | 0.01 ± 0.00 a | 0.01 ± 0.00 a | 0.01 ± 0.00 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guerrini, L.; Masella, P.; Angeloni, G.; Sacconi, A.; Calamai, L.; Parenti, A. Effects of a Small Increase in Carbon Dioxide Pressure during Fermentation on Wine Aroma. Foods 2020, 9, 1496. https://doi.org/10.3390/foods9101496
Guerrini L, Masella P, Angeloni G, Sacconi A, Calamai L, Parenti A. Effects of a Small Increase in Carbon Dioxide Pressure during Fermentation on Wine Aroma. Foods. 2020; 9(10):1496. https://doi.org/10.3390/foods9101496
Chicago/Turabian StyleGuerrini, Lorenzo, Piernicola Masella, Giulia Angeloni, Andrea Sacconi, Luca Calamai, and Alessandro Parenti. 2020. "Effects of a Small Increase in Carbon Dioxide Pressure during Fermentation on Wine Aroma" Foods 9, no. 10: 1496. https://doi.org/10.3390/foods9101496
APA StyleGuerrini, L., Masella, P., Angeloni, G., Sacconi, A., Calamai, L., & Parenti, A. (2020). Effects of a Small Increase in Carbon Dioxide Pressure during Fermentation on Wine Aroma. Foods, 9(10), 1496. https://doi.org/10.3390/foods9101496







