Tangerines Cultivated on Madeira Island—A High Throughput Natural Source of Bioactive Compounds
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Tangerine Samples
2.3. HS-SPME Procedure
2.4. Gas Chromatography–Mass Spectrometry Analysis (GC–MS)
2.5. Multivariate Statistical Analysis
3. Results and Discussion
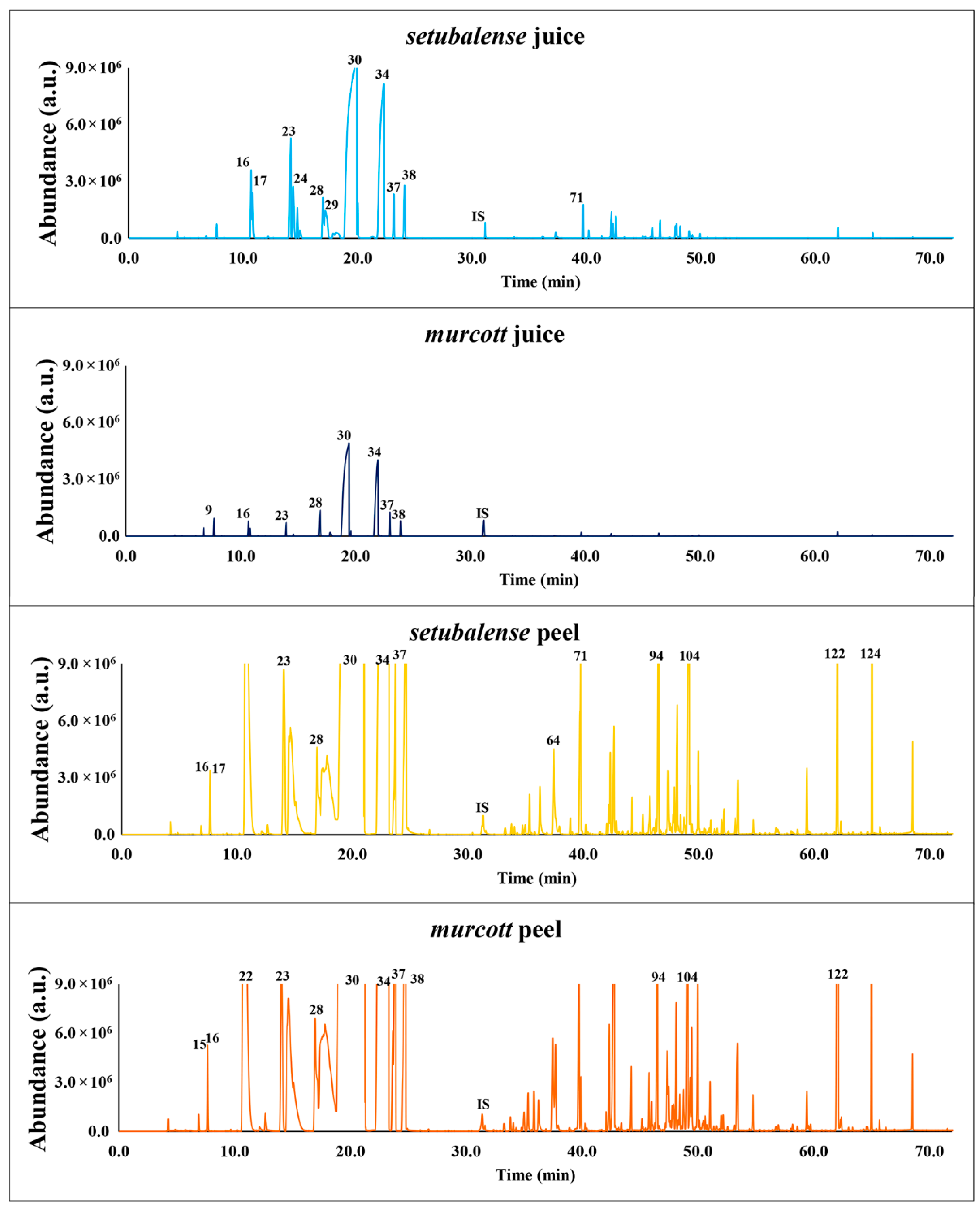
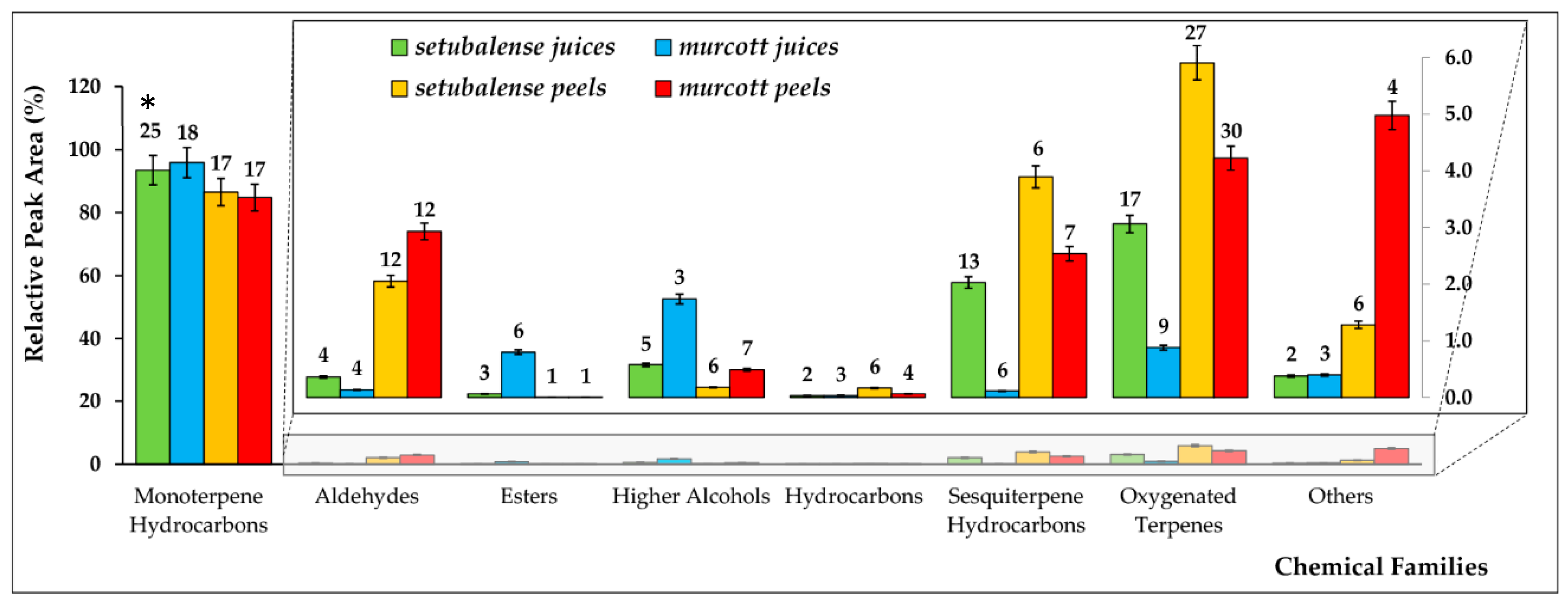
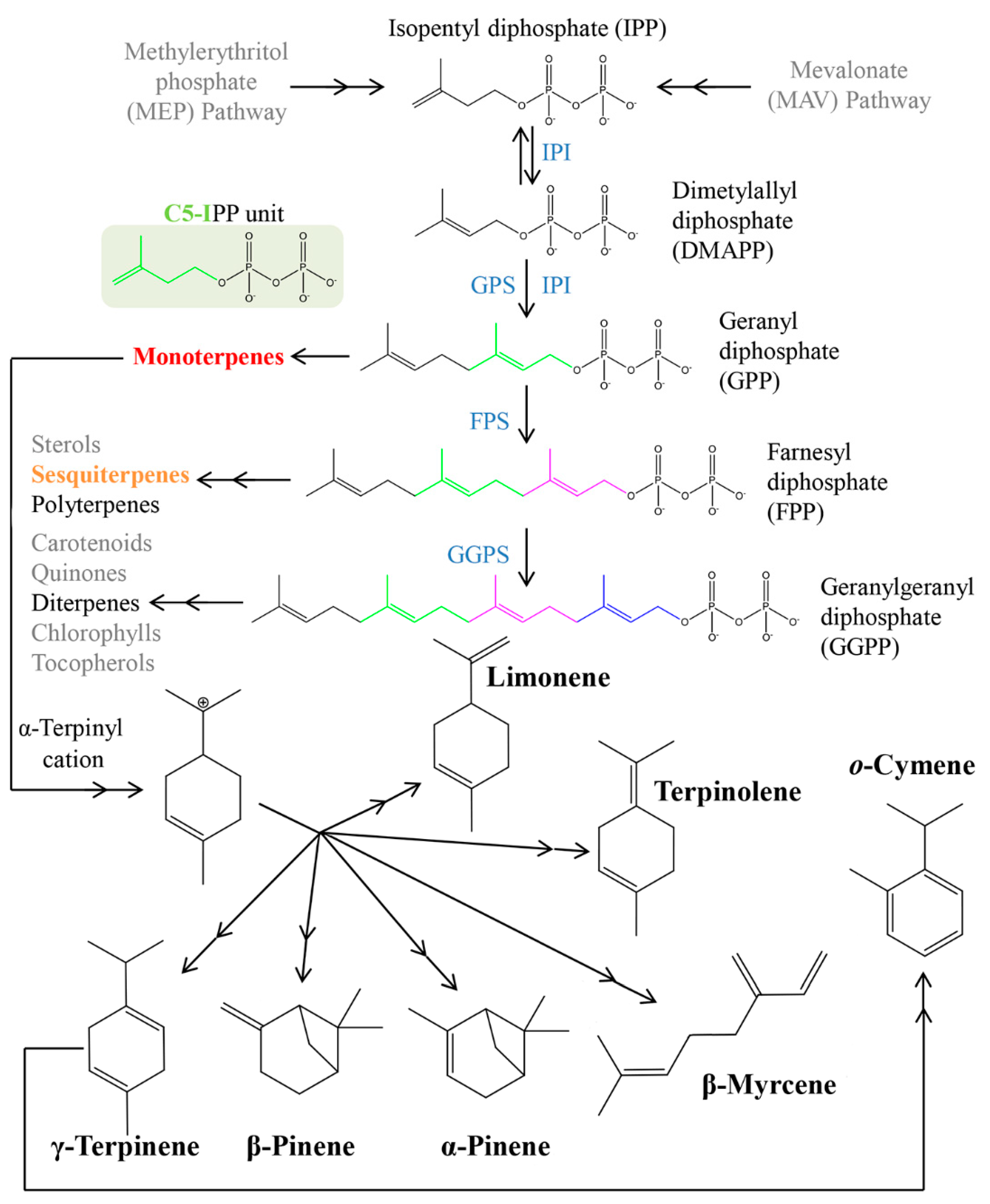
3.1. Volatomic Profile of Tangerines
3.1.1. Volatomic Profile of Tangerine Juices
3.1.2. Volatomic Profile of Tangerine Peels
3.2. Tangerines Bioactive Potential
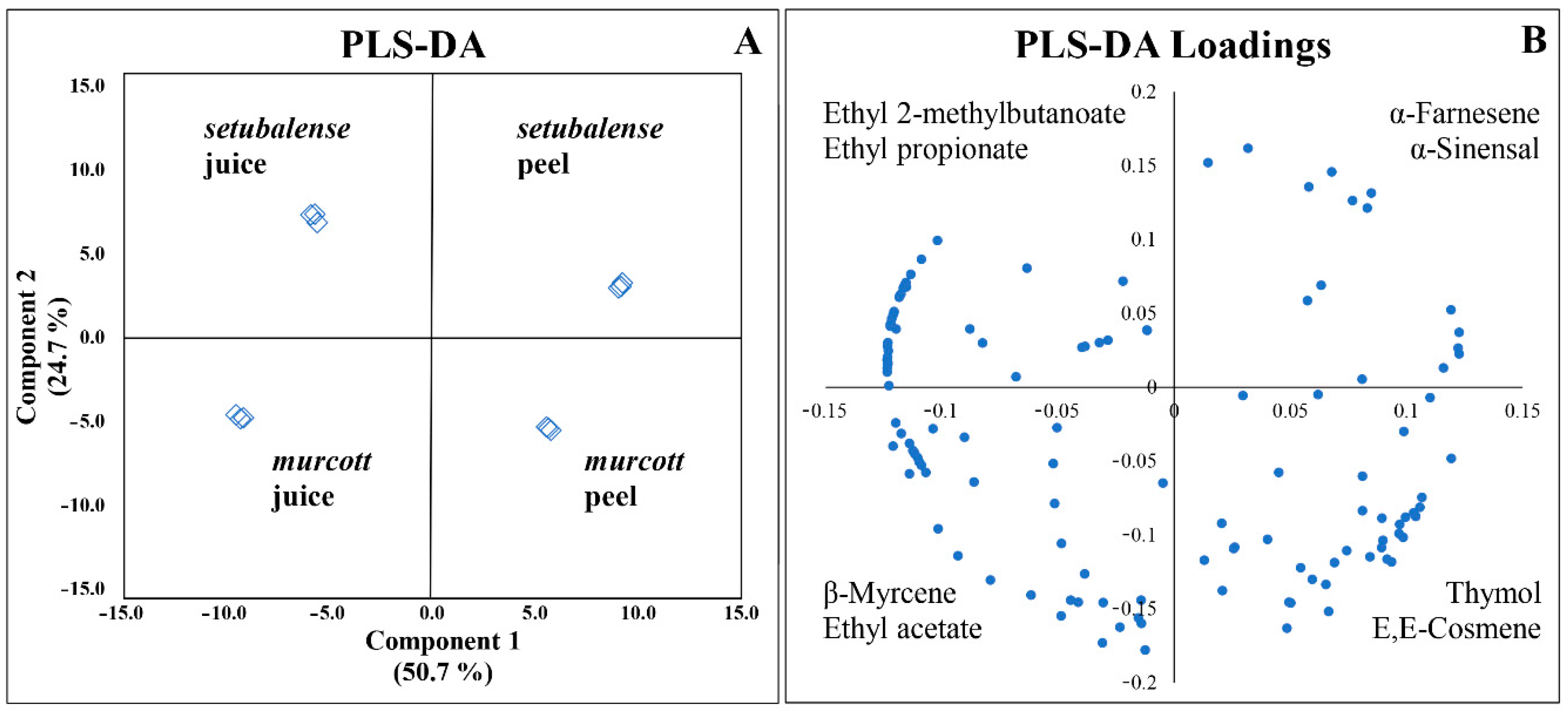
3.3. Classification of Tangerine Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| EI | electron: impact mode |
| CAR | carboxen |
| DBV | divinylbenzene |
| GC-MS | gas chromatography-mass spectrometry |
| HCA | hierarchical cluster analysis |
| HS-SPME | head space solid phase microextraction |
| IS | internal standard |
| MVSA | multivariate statistical analysis |
| NIST | National Institute of Standards and Technology |
| PDMS | polydimethylsiloxane |
| PLS-DA | partial least square discriminant analysis |
| PTFE | polytetrafluoroethylene |
| RT | retention time |
| VIP | variable importance in projection |
| VOMs | volatile organic metabolites |
References
- Geraci, A.; Di Stefano, V.; Di Martino, E.; Schillaci, D.; Schicchi, R. Essential oil components of orange peels and antimicrobial activity. Nat. Prod. Res. 2017, 31, 653–659. [Google Scholar] [CrossRef]
- USDA. Citrus: World Markets and Trades. Available online: https://apps.fas.usda.gov/psdonline/circulars/citrus.pdf (accessed on 25 September 2020).
- Kaur, J.; Kaur, G. An insight into the role of citrus bioactives in modulation of colon cancer. J. Funct. Foods 2015, 13, 239–261. [Google Scholar] [CrossRef]
- Rodríguez, A.; Peris, J.E.; Redondo, A.; Shimada, T.; Costell, E.; Carbonell, I.; Rojas, C.; Peña, L. Impact of d-limonene synthase up-or down-regulation on sweet orange fruit and juice odor perception. Food Chem. 2017, 217, 139–150. [Google Scholar] [CrossRef]
- Miyazaki, T.; Plotto, A.; Goodner, K.; Gmitter, F.G., Jr. Distribution of aroma volatile compounds in tangerine hybrids and proposed inheritance. J. Sci. Food Agric. 2011, 91, 449–460. [Google Scholar] [CrossRef]
- Goldenberg, L.; Yaniv, Y.; Porat, R.; Carmi, N. Mandarin fruit quality: A review. J. Sci. Food Agric. 2018, 98, 18–26. [Google Scholar] [CrossRef]
- Tietel, Z.; Plotto, A.; Fallik, E.; Lewinsohn, E.; Porat, R. Taste and aroma of fresh and stored mandarins. J. Sci. Food Agric. 2011, 91, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Maarse, H. Volatile Compounds in Foods and Beverages; CRC Press: New York, NY, USA, 1991; p. 784. [Google Scholar]
- Figueira, J.; Camara, H.; Pereira, J.; Camara, J.S. Evaluation of volatile metabolites as markers in Lycopersicon esculentum L. cultivars discrimination by multivariate analysis of headspace solid phase microextraction and mass spectrometry data. Food Chem. 2014, 145, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Porto-Figueira, P.; Figueira, J.A.; Berenguer, P.; Câmara, J.S. Exploring a volatomic-based strategy for a fingerprinting approach of Vaccinium padifolium L. berries at different ripening stages. Food Chem. 2018, 245, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Figueira, J.A.; Porto-Figueira, P.; Pereira, J.A.M.; Câmara, J.S. A comprehensive methodology based on NTME/GC-MS data and chemometric tools for lemons discrimination according to geographical origin. Microchem. J. 2020, 157, 104933. [Google Scholar] [CrossRef]
- Porto-Figueira, P.; Freitas, A.; Cruz, C.J.; Figueira, J.; Camara, J.S. Profiling of passion fruit volatiles: An effective tool to discriminate between species and varieties. Food Res. Int. 2015, 77, 408–418. [Google Scholar] [CrossRef]
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef]
- El Hadi, M.; Zhang, F.-J.; Wu, F.-F.; Zhou, C.-H.; Tao, J. Advances in fruit aroma volatile research. Molecules 2013, 18, 8200–8229. [Google Scholar] [CrossRef]
- Degenhardt, J.; Kollner, T.G.; Gershenzon, J. Monoterpene and sesquiterpene synthases and the origin of terpene skeletal diversity in plants. Phytochemistry 2009, 70, 1621–1637. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Bai, J.; Chen, C.; Plotto, A.; Baldwin, E.A.; Gmitter, F.G. Comparative analysis of juice volatiles in selected mandarins, mandarin relatives and other citrus genotypes. J. Sci. Food Agric. 2018, 98, 1124–1131. [Google Scholar] [CrossRef]
- Li, H.; Ge, Y.; Luo, Z.; Zhou, Y.; Zhang, X.; Zhang, J.; Fu, Q. Evaluation of the chemical composition, antioxidant and anti-inflammatory activities of distillate and residue fractions of sweet basil essential oil. J. Food Sci. Technol. 2017, 54, 1882–1890. [Google Scholar] [CrossRef] [PubMed]
- Lubinska-Szczygieł, M.; Różańska, A.; Namieśnik, J.; Dymerski, T.; Shafreen, R.B.; Weisz, M.; Ezra, A.; Gorinstein, S. Quality of limes juices based on the aroma and antioxidant properties. Food Control 2018, 89, 270–279. [Google Scholar] [CrossRef]
- Fawzi Mahomoodally, M.; Mollica, A.; Stefanucci, A.; Zakariyyah Aumeeruddy, M.; Poorneeka, R.; Zengin, G. Volatile components, pharmacological profile, and computational studies of essential oil from Aegle marmelos (Bael) leaves: A functional approach. Ind. Crops Prod. 2018, 126, 13–21. [Google Scholar] [CrossRef]
- Ahmed, H.M.; Tavaszi-Sarosi, S. Identification and quantification of essential oil content and composition, total polyphenols and antioxidant capacity of Perilla frutescens (L.) Britt. Food Chem. 2019, 275, 730–738. [Google Scholar] [CrossRef]
- Alam, F.; Shafique, Z.; Amjad, S.T.; Bin Asad, M.H.H. Enzymes inhibitors from natural sources with antidiabetic activity: A review. Phytother. Res. 2019, 33, 41–54. [Google Scholar] [CrossRef]
- da Silva, V.D.; Almeida-Souza, F.; Teles, A.M.; Neto, P.A.; Mondego-Oliveira, R.; Mendes Filho, N.E.; Taniwaki, N.N.; Abreu-Silva, A.L.; Calabrese, K.d.S.; Mouchrek Filho, V.E. Chemical composition of Ocimum canum Sims. essential oil and the antimicrobial, antiprotozoal and ultrastructural alterations it induces in Leishmania amazonensis promastigotes. Ind. Crops Prod. 2018, 119, 201–208. [Google Scholar] [CrossRef]
- da Silva, V.P.; Alves, C.C.F.; Miranda, M.L.D.; Bretanha, L.C.; Balleste, M.P.; Micke, G.A.; Silveira, E.V.; Martins, C.H.G.; Ambrosio, M.A.L.V.; de Souza Silva, T.; et al. Chemical composition and in vitro leishmanicidal, antibacterial and cytotoxic activities of essential oils of the Myrtaceae family occurring in the Cerrado biome. Ind. Crops Prod. 2018, 123, 638–645. [Google Scholar] [CrossRef]
- Fadil, M.; Fikri-Benbrahim, K.; Rachiq, S.; Ihssane, B.; Lebrazi, S.; Chraibi, M.; Haloui, T.; Farah, A. Combined treatment of Thymus vulgaris L., Rosmarinus officinalis L. and Myrtus communis L. essential oils against Salmonella typhimurium: Optimization of antibacterial activity by mixture design methodology. Eur. J. Pharm. Biopharm. 2018, 126, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Dănilă, E.; Moldovan, Z.; Popa, M.; Chifiriuc, M.C.; Kaya, A.D.; Kaya, M.A. Chemical composition, antimicrobial and antibiofilm efficacy of C. limon and L. angustifolia EOs and of their mixtures against Staphylococcus epidermidis clinical strains. Ind. Crops Prod. 2018, 122, 483–492. [Google Scholar] [CrossRef]
- Assmann, C.E.; Cadoná, F.C.; Bonadiman, B.d.S.R.; Dornelles, E.B.; Trevisan, G.; da Cruz, I.B.M. Tea tree oil presents in vitro antitumor activity on breast cancer cells without cytotoxic effects on fibroblasts and on peripheral blood mononuclear cells. Biomed. Pharmacother. 2018, 103, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Elkady, W.M.; Ayoub, I.M. Chemical profiling and antiproliferative effect of essential oils of two Araucaria species cultivated in Egypt. Ind. Crops Prod. 2018, 118, 188–195. [Google Scholar] [CrossRef]
- Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Lampri, E.; Fitsiou, E.; Vasileiadis, S.; Vamvakias, M.; Bardouki, H.; Goussia, A.; Malamou-Mitsi, V.; Panayiotidis, M.I.; et al. Dietary mastic oil extracted from Pistacia lentiscus var. chia suppresses tumor growth in experimental colon cancer models. Sci. Rep. 2017, 7, 3782. [Google Scholar] [CrossRef]
- Satou, T.; Hanashima, Y.; Mizutani, I.; Koike, K. The effect of inhalation of essential oil from Rosmarinus officinalis on scopolamine-induced Alzheimer‘s type dementia model mice. Flavour Fragr. J. 2018, 33, 230–234. [Google Scholar] [CrossRef]
- Sieniawska, E.; Sawicki, R.; Swatko-Ossor, M.; Napiorkowska, A.; Przekora, A.; Ginalska, G.; Swatko-Ossor, M.; Augustynowicz-Kopec, E. The effect of combining natural terpenes and antituberculous agents against reference and clinical Mycobacterium tuberculosis strains. Molecules 2018, 23, 176. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Mousavi Khaneghah, A.; Gavahian, M.; Marszałek, K.; Eş, I.; Munekata, P.E.; Ferreira, I.C.; Barba, F.J. Understanding the potential benefits of thyme and its derived products for food industry and consumer health: From extraction of value-added compounds to the evaluation of bioaccessibility, bioavailability, anti-inflammatory, and antimicrobial activities. Crit. Rev. Food Sci. Nutr. 2019, 59, 2879–2895. [Google Scholar] [CrossRef]
- Salehi, B.; Mishra, A.P.; Shukla, I.; Sharifi-Rad, M.; Contreras, M.d.M.; Segura-Carretero, A.; Fathi, H.; Nasrabadi, N.N.; Kobarfard, F.; Sharifi-Rad, J. Thymol, thyme, and other plant sources: Health and potential uses. Phytother. Res. 2018, 32, 1688–1706. [Google Scholar] [CrossRef]
- Ladanyia, M.; Ladaniya, M. Citrus Fruit: Biology, Technology and Evaluation; Academic Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Ahluwalia, V.; Sisodia, R.; Walia, S.; Sati, O.P.; Kumar, J.; Kundu, A. Chemical analysis of essential oils of Eupatorium adenophorum and their antimicrobial, antioxidant and phytotoxic properties. J. Pest Sci. 2013, 87, 341–349. [Google Scholar] [CrossRef]
- Galvez, C.E.; Jimenez, C.M.; Gomez, A.d.l.A.; Lizarraga, E.F.; Sampietro, D.A. Chemical composition and antifungal activity of essential oils from Senecio nutans, Senecio viridis, Tagetes terniflora and Aloysia gratissima against toxigenic Aspergillus and Fusarium species. Nat. Prod. Res. 2020, 34, 1442–1445. [Google Scholar] [CrossRef] [PubMed]
- Elghwaji, W.; El-Sayed, A.M.; El-Deeb, K.S.; ElSayed, A.M. Chemical composition, antimicrobial and antitumor potentiality of essential oil of Ferula tingitana L. Apiaceae grow in Libya. Pharmacogn. Mag. 2017, 13, S446. [Google Scholar] [PubMed]
- Nie, J.Y.; Li, R.; Jiang, Z.T.; Wang, Y.; Tan, J.; Tang, S.H.; Zhang, Y. Antioxidant activity screening and chemical constituents of the essential oil from rosemary by ultra-fast GC electronic nose coupled with chemical methodology. J. Sci. Food Agric. 2020, 100, 3481–3487. [Google Scholar] [CrossRef] [PubMed]
- Safar, A.A.; Ghafoor, A.O.; Dastan, D. Screening of Chemical Characterization, Antifungal and Cytotoxic Activities of Essential Oil Constituents of Tagetes erecta L. from Erbil, Kurdistan Region-Iraq. Pol. J. Environ. Stud. 2020, 29, 2317–2326. [Google Scholar] [CrossRef]
- Song, Y.; Sun, R.; Ji, Z.; Li, X.; Fu, Q.; Ma, S. Perilla aldehyde attenuates CUMS-induced depressive-like behaviors via regulating TXNIP/TRX/NLRP3 pathway in rats. Life Sci. 2018, 206, 117–124. [Google Scholar] [CrossRef]
- Okla, M.K.; Alamri, S.A.; Salem, M.Z.; Ali, H.M.; Behiry, S.I.; Nasser, R.A.; Alaraidh, I.A.; Al-Ghtani, S.M.; Soufan, W. Yield, phytochemical constituents, and antibacterial activity of essential oils from the leaves/twigs, branches, branch wood, and branch bark of Sour Orange (Citrus aurantium L.). Processes 2019, 7, 363. [Google Scholar] [CrossRef]

| Juice | Peels | VOMs Potential Bioactive Effects 3 | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PN 1 | VOMs | Set/MurRatio 2 | T Test (p < 0.05) | Set/Mur Ratio 2 | T Test (p < 0.05) | Antibacterial | Antidepressant | Antidiabetic | Anti-inflammatory | Antileishmanial | Antifungal | Antioxidant | Antiproliferative | Antitumor | Cytotoxic | References |
| 16 | α-pinene | 8.50 | 0.0218 | 0.40 | 0.0367 | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | [18,21,22,23,24,26,27,28] | |||
| 17 | α-thujene | 10.0 | 0.0135 | 39.8 | 0.0002 | ǂ | ǂ | ǂ | ǂ | [35,36] | ||||||
| 21 | camphene | 15.8 | 0.0079 | 0.10 | 0.0025 | ǂ | ǂ | ǂ | [18,24,28] | |||||||
| 23 | β-pinene | 17.5 | 0.0182 | 1.00 | 0.3719 | ǂ | ǂ | ǂ | ǂ | [18,24,26,28] | ||||||
| 25 | sabinene | 23.3 | 0.0115 | – | – | ǂ | ǂ | ǂ | ǂ | [22,24,26] | ||||||
| 28 | β-myrcene | 5.60 | 0.0054 | 0.70 | 0.3413 | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | [17,19,22,24,26,28] | ||
| 29 | α-terpinene | 7.50 | 0.0076 | 0.90 | 0.0854 | ǂ | ǂ | ǂ | ǂ | ǂ | [22,24,26] | |||||
| 30 | limonene (isomer) | 4.40 | 0.0086 | 1.20 | 0.0979 | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | [18,19,20,21,22,23,24,25,26,27,28] | ||
| 31 | β-phellandrene | 7.90 | 0.0201 | 0.70 | 0.0827 | ǂ | ǂ | ǂ | ǂ | ǂ | [19,22,26] | |||||
| 33 | (E)-ocimene | 31.3 | 0.0052 | – | – | ǂ | ǂ | ǂ | ǂ | [17,19] | ||||||
| 34 | γ-terpinene | 4.90 | 0.0001 | 2.10 | 0.0283 | ǂ | ǂ | ǂ | [18,24,26] | |||||||
| 37 | o-cymene | 3.00 | 0.0033 | 0.20 | 0.0015 | ǂ | ǂ | ǂ | ǂ | ǂ | [35,37,38] | |||||
| 38 | terpinolene | 6.00 | 0.0001 | 1.60 | 0.0013 | ǂ | ǂ | [24,26] | ||||||||
| 53 | p-cymenene | 7.00 | 0.0004 | 1.90 | 0.2046 | ǂ | ǂ | ǂ | ǂ | [37,38] | ||||||
| 55 | Cosmene 4 | – | – | 3.40 | 0.0000 | ǂ | ǂ | ǂ | [34] | |||||||
| 71 | linalool | 17.3 | 0.0000 | 1.90 | 0.0000 | ǂ | ǂ | ǂ | ǂ | ǂ | [17,20,24,25,28] | |||||
| 79 | thymol methyl ether | 8.34 | 0.0004 | 3.00 | 0.0000 | ǂ | ǂ | ǂ | ǂ | ǂ | [31,32] | |||||
| 82 | β-caryophyllene | 62.5 | 0.0009 | 1.20 | 0.0000 | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | [20,22,23,24,27,28] | ||||
| 94 | α-terpineol | 13.7 | 0.0010 | 1.60 | 0.0030 | ǂ | ǂ | ǂ | ǂ | ǂ | ǂ | [18,23,24,25,26] | ||||
| 107 | perilla aldehyde | 9.40 | 0.0017 | 1.40 | 0.0164 | ǂ | ǂ | ǂ | ǂ | [39] | ||||||
| 122 | dimethyl anthranilate | 4.94 | 0.0105 | 0.6 | 0.0012 | ǂ | ǂ | [40] | ||||||||
| 124 | thymol | 11.5 | 0.0073 | 1.9 | 0.0004 | ǂ | ǂ | ǂ | ǂ | [31,32] | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Figueira, J.A.; Porto-Figueira, P.; Pereira, J.A.M.; Câmara, J.S. Tangerines Cultivated on Madeira Island—A High Throughput Natural Source of Bioactive Compounds. Foods 2020, 9, 1470. https://doi.org/10.3390/foods9101470
Figueira JA, Porto-Figueira P, Pereira JAM, Câmara JS. Tangerines Cultivated on Madeira Island—A High Throughput Natural Source of Bioactive Compounds. Foods. 2020; 9(10):1470. https://doi.org/10.3390/foods9101470
Chicago/Turabian StyleFigueira, José A., Priscilla Porto-Figueira, Jorge A. M. Pereira, and José S. Câmara. 2020. "Tangerines Cultivated on Madeira Island—A High Throughput Natural Source of Bioactive Compounds" Foods 9, no. 10: 1470. https://doi.org/10.3390/foods9101470
APA StyleFigueira, J. A., Porto-Figueira, P., Pereira, J. A. M., & Câmara, J. S. (2020). Tangerines Cultivated on Madeira Island—A High Throughput Natural Source of Bioactive Compounds. Foods, 9(10), 1470. https://doi.org/10.3390/foods9101470






