Myrtus Communis Liquor Byproduct as a Source of Bioactive Compounds
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Standards
2.2. Samples Collection and Extracts Preparation
2.3. Chemical Composition
2.4. Fatty Acid Profile
2.5. Antioxidant Capacity
2.6. DPPH Radical Scavenging Activity
2.7. ABTS Radical Scavenging Assay
2.8. Determination of Total Phenols
2.9. ESI-MS and ESI-MS/MS Analysis
2.10. HPLC–ESI-MS and HPLC–ESI-MS/MS Analysis
2.11. Calibration and Quantification of Phenolic Compounds
2.12. Method Validation
2.13. Statistical Analysis
3. Results and Discussions
3.1. Chemical Composition
3.2. Fatty Acid Composition
3.3. Polyphenolic Compounds
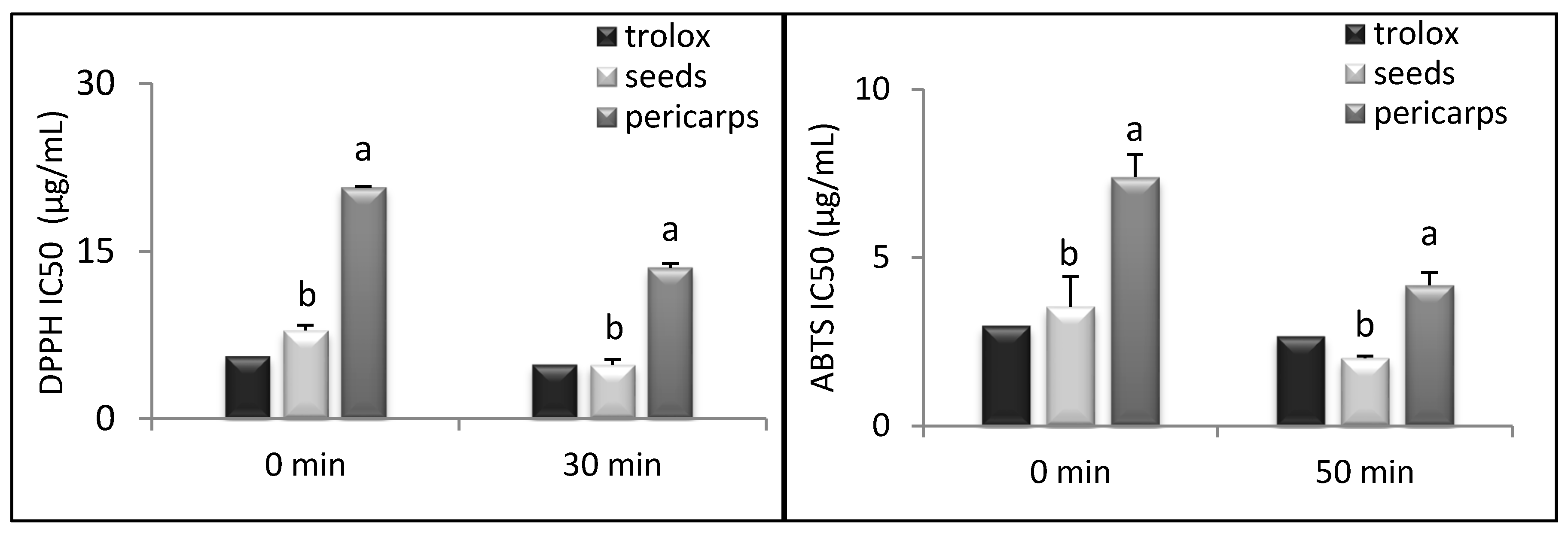
3.4. Antioxidant Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Federici, F.; Fava, F.; Kalogerakis, N.; Mantzavinos, D. Valorisation of agro-industrial by-products, effluents and waste: Concept, opportunities and the case of olive mill wastewaters. J. Chem. Technol. Biotechnol. 2009, 84, 895–900. [Google Scholar] [CrossRef]
- Mauricio, E.M.; Rosado, C.; Duarte, M.P.; Fernando, A.L.; Diaz-Lanza, A.M. Evaluation of Industrial Sour Cherry Liquor Wastes as an Ecofriendly Source of Added Value Chemical Compounds and Energy. Waste Biomass Valorization 2018, 1–10. [Google Scholar] [CrossRef]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouysegu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef] [PubMed]
- Tuberoso, C.I.G.; Rosa, A.; Bifulco, E.; Melis, M.P.; Atzeri, A.; Pirisi, F.M.; Dessì, M.A. Chemical composition and antioxidant activities of Myrtus communis L. berries extracts. Food Chem. 2010, 123, 1242–1251. [Google Scholar] [CrossRef]
- Quiñones, M.; Miguel, M.; Aleixandre, A. Beneficial effects of polyphenols on cardiovascular disease. Pharmacol. Res. 2013, 68, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Sakagami, H.; Fujisawa, S. Cytotoxicity and apoptosis induction by butylated hydroxyanisole (BHA) and butylated hydroxytoluene (BHT). Anticancer Res. 2002, 23, 4693–4701. [Google Scholar]
- Anunciato, T.P.; da Rocha Filho, P.A. Carotenoids and polyphenols in nutricosmetics, nutraceuticals, and cosmeceuticals. J. Cosmet. Dermatol. 2012, 11, 51–54. [Google Scholar] [CrossRef]
- Asensi, M.; Ortega, A.; Mena, S.; Feddi, F.; Estrela, J.M. Natural polyphenols in cancer therapy. Crit. Rev. Clin. Lab. Sci. 2011, 48, 197–216. [Google Scholar] [CrossRef]
- Aerts, R.J.; Barry, T.N.; McNabb, W.C. Polyphenols and agriculture: Beneficial effects of proanthocyanidins in forages. Agric. Ecosyst. Environ. 1999, 75, 1–12. [Google Scholar] [CrossRef]
- Grainger, C.; Clarke, T.; Auldist, M.; Beauchemin, K.; McGinn, S.; Waghorn, G.; Eckard, R. Potential use of Acacia mearnsii condensed tannins to reduce methane emissions and nitrogen excretion from grazing dairy cows. Can. J. Anim. Sci. 2009, 89, 241–251. [Google Scholar] [CrossRef]
- Vasta, V.; Yanez-Ruiz, D.R.; Mele, M.; Serra, A.; Luciano, G.; Lanza, M.; Biondi, L.; Priolo, A. Bacterial and protozoal communities and fatty acid profile in the rumen of sheep fed a diet containing added tannins. Appl. Environ. Microb. 2010, 76, 2549–2555. [Google Scholar] [CrossRef] [PubMed]
- Nudda, A.; Correddu, F.; Marzano, A.; Battacone, G.; Nicolussi, P.; Bonelli, P.; Pulina, G. Effects of diets containing grape seed, linseed or both on milk production traits, liver and kidney activities, and immunity of lactating dairy ewes. J. Dairy Sci. 2015, 98, 1157–1166. [Google Scholar] [CrossRef] [PubMed]
- Correddu, F.; Gaspa, G.; Pulina, G.; Nudda, A. Grape seed and linseed, alone and in combination, enhance unsaturated fatty acids in the milk of Sarda dairy sheep. J. Dairy Sci. 2016, 99, 1725–1735. [Google Scholar] [CrossRef] [PubMed]
- Dung, N.T.; Van Binh, D.; Mui, N.T.; Preston, T. Effect of cassava hay supplementation on milk production in lactating goats. Livest. Res. Rural Dev. 2010, 22, 45. [Google Scholar]
- Franco, M.A.; Versini, G.; Saba, R. Caratterizzazione del Liquore “Mirto di Sardegna Tradizionale”; Associazione Produttori Liquore Mirto di Sardegna Tradizionale/Confindustria; Litotipografia Valdes: Cagliari, Italy, 1998. [Google Scholar]
- Tuberoso, C.I.G.; Barra, A.; Cabras, P. Effect of different technological processes on the chemical composition of myrtle (Myrtus communis L.) alcoholic extracts. Eur. Food Res. Technol. 2008, 226, 801–808. [Google Scholar] [CrossRef]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 17th ed.; Association of Official Analytical Chemists: Arlington, VA, USA, 2000. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International, 18th ed.; Association of Official Analytical Chemists: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Guimarães, R.; Barros, L.; Carvalho, A.M.; Ferreira, I.C.F.R. Studies on chemical constituents and bioactivity of rosa micrantha: An alternative antioxidants source for food, pharmaceutical, or cosmetic applications. J. Agric. Food Chem. 2010, 58, 6277–6284. [Google Scholar] [CrossRef]
- Kramer, J.K.G.; Fellner, V.; Dugan, M.E.R.; Sauer, F.D.; Mossoba, M.M.; Yurawecz, M.P. Evaluating acid and base catalysts in the methylation of milk and rumen fatty acids with special emphasis on conjugated dienes and total trans fatty acids. Lipids 1997, 32, 1219–1228. [Google Scholar] [CrossRef]
- Kramer, J.K.; Cruz-Hernandez, C.; Deng, Z.; Zhou, J.; Jahreis, G.; Dugan, M.E. Analysis of conjugated linoleic acid and trans 18: 1 isomers in synthetic and animal products. Am. J. Clin. Nutr. 2004, 79, 1137S–1145S. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Maldini, M.; Montoro, P.; Addis, R.; Toniolo, C.; Petretto, G.L.; Foddai, M.; Nicoletti, M.; Pintore, G. A new approach to discriminate Rosmarinus officinalis L. plants with antioxidant activity, based on HPTLC fingerprint and targeted phenolic analysis combined with PCA. Ind. Crops Prod. 2016, 94, 665–672. [Google Scholar] [CrossRef]
- Petretto, G.L.; Maldini, M.; Addis, R.; Chessa, M.; Foddai, M.; Rourke, J.P.; Pintore, G. Variability of chemical composition and antioxidant activity of essential oils between Myrtus communis var. Leucocarpa DC and var. Melanocarpa DC. Food Chem. 2016, 197, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Lizcano, L.J.; Bakkali, F.; Ruiz-Larrea, M.B.; Ruiz-Sanz, J.I. Antioxidant activity and polyphenol content of aqueous extracts from Colombian Amazonian plants with medicinal use. Food Chem. 2010, 119, 1566–1570. [Google Scholar] [CrossRef]
- Maldini, M.; Foddai, M.; Natella, F.; Addis, R.; Chessa, M.; Petretto, G.L.; Tuberoso, C.I.G.; Pintore, G. Metabolomic study of wild and cultivated caper (Capparis spinosa L.) from different areas of Sardinia and their comparative evaluation. J. Mass Spectrom. 2016, 51, 716–728. [Google Scholar] [CrossRef] [PubMed]
- EMEA; European Medicines Agency. Quality Guidelines: Validation of Analytical Procedures: Text and Methodology (ICHQ2). 1995. Available online: http://www.ema.europa.eu/ema/index.jsp?curl=pages/regulation/general/general_content_000768.jsp&mid=WC0b01ac0580028e8d/ (accessed on 22 September 2016).
- Free Statistics Calculators. Available online: http://www.danielsoper.com/statcalc3/calc.aspx?id=103 (accessed on 1 February 2017).
- Nudda, A.; Correddu, F.; Atzori, A.S.; Marzano, A.; Battacone, G.; Nicolussi, P.; Bonelli, P.; Pulina, G. Whole exhausted berries of Myrtus communis L. supplied to dairy ewes: Effects on milk production traits and blood metabolites. Small Rumin. Res. 2017, 155, 33–38. [Google Scholar] [CrossRef]
- Nudda, A.; Buffa, G.; Atzori, A.S.; Cappai, M.G.; Caboni, P.; Fais, G.; Pulina, G. Small amounts of agro-industrial byproducts in dairy ewes diets affects milk production traits and hematological parameters. Anim. Feed Sci. Technol. 2019, 251, 76–85. [Google Scholar] [CrossRef]
- Wannes, W.A.; Marzouk, B. Characterization of myrtle seed (Myrtus communis var. baetica) as a source of lipids, phenolics, and antioxidant activities. J. Food Drug Anal. 2016, 24, 316–323. [Google Scholar] [CrossRef]
- Wannes, W.A.; Mhamdi, B.; Sriti, J.; Bettaieb, I.; Tounsi, M.S.; Marzouk, B. Fatty acid and glycerolipid changes during Tunisian myrtle (Myrtus communis var. italica) fruit ripening. J. Food Biochem. 2011, 35, 177–194. [Google Scholar] [CrossRef]
- Cakir, A. Essential oil and fatty acid composition of the fruits of Hippophae rhamnoides L. (Sea Buckthorn) and Myrtus communis L. from Turkey. Biochem. Syst. Ecol. 2004, 32, 809–816. [Google Scholar] [CrossRef]
- Wannes, W.A.; Mhamdi, B.; Marzouk, B. Variations in essential oil and fatty acid composition during Myrtus communis var. italica fruit maturation. Food Chem. 2009, 112, 621–626. [Google Scholar] [CrossRef]
- Nguyen, M.T.; Hanzelmann, D.; Härtner, T.; Peschel, A.; Götz, F. Skin-Specific unsaturated fatty acids boost the Staphylococcus aureus innate immune response. Infect. Immun. 2016, 84, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Shingfield, K.J.; Bonnet, M.; Scollan, N.D. Recent developments in altering the fatty acid composition of ruminant-derived foods. Animal 2013, 7, 132–162. [Google Scholar] [CrossRef] [PubMed]
- Ramos, M.J.; Fernández, C.M.; Casas, A.; Rodríguez, L.; Pérez, Á. Influence of fatty acid composition of raw materials on biodiesel properties. Bioresour. Technol. 2009, 100, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Correddu, F.; Nudda, A.; Battacone, G.; Boe, R.; Francesconi, A.H.D.; Pulina, G. Effects of grape seed supplementation, alone or associated with linseed, on ruminal metabolism in Sarda dairy sheep. Anim. Feed Sci. Technol. 2015, 199, 61–72. [Google Scholar] [CrossRef]
- Wannes, W.A.; Marzouk, B. Differences between myrtle fruit parts (Myrtus communis var. italica) in phenolics and antioxidant contents. J. Food Biochem. 2013, 37, 585–594. [Google Scholar]
- Correddu, F.; Fancello, F.; Chessa, L.; Atzori, A.S.; Pulina, G.; Nudda, A. Effects of supplementation with exhausted myrtle berries on rumen function of dairy sheep. Small Rumin. Res. 2019, 170, 51–61. [Google Scholar] [CrossRef]
- Montoro, P.; Tuberoso, C.I.; Piacente, S.; Perrone, A.; De Feo, V.; Cabras, P.; Pizza, C. Stability and antioxidant activity of polyphenols in extracts of Myrtus communis L. berries used for the preparation of myrtle liqueur. J. Pharm. Biomed. Anal. 2006, 41, 1614–1619. [Google Scholar] [CrossRef]
- Barboni, T.; Cannac, M.; Massi, L.; Perez-Ramirez, Y.; Chiaramonti, N. Variability of polyphenol compounds in Myrtus communis L. (Myrtaceae) berries from Corsica. Molecules 2010, 15, 7849–7860. [Google Scholar] [CrossRef]
- Barboni, T.; Venturini, N.; Paolini, J.; Desjobert, J.M.; Chiaramonti, N.; Costa, J. Characterisation of volatiles and polyphenols for quality assessment of alcoholic beverages prepared from Corsican Myrtus communis berries. Food Chem. 2010, 122, 1304–1312. [Google Scholar] [CrossRef]
- Martín, T.; Rubio, B.; Villaescusa, L.; Fernández, L.; Díaz, A.M. Polyphenolic compounds from pericarps of Myrtus communis. Pharm. Biol. 1999, 37, 28–31. [Google Scholar] [CrossRef]
- Vattem, D.A.; Shetty, K. Biological functionality of ellagic acid: A review. J. Food Biochem. 2005, 29, 234–266. [Google Scholar] [CrossRef]
- Petretto, G.L.; Tuberoso, C.I.G.; Vlahopoulou, G.; Atzei, A.; Mannu, A.; Zrira, S.; Pintore, G. Volatiles, color characteristics and other physico-chemical parameters of commercial Moroccan honeys. Nat. Prod. Res. 2016, 30, 286–292. [Google Scholar] [CrossRef] [PubMed]
| Chemical Composition 1 | Pericarps | Seeds | Whole EMB | p-Value |
|---|---|---|---|---|
| dry matter (DM), % | 88.58 ± 0.01 c | 91.37 ± 0.03 a | 89.73 ± 0.04 b | *** |
| organic matter, % of DM | 94.46 ± 0.02 c | 98.05 ± 0.03 a | 96.53 ± 0.02 b | *** |
| NDF, % of DM | 62.66 ± 1.81 b | 69.38 ± 0.17 a | 65.14 ± 1.61 b | ** |
| ADF, % of DM | 59.71 ± 1.16 a | 51.67 ± 0.16 b | 53.34 ± 1.37 b | *** |
| NFC, % of DM | 25.21 ± 1.83 a | 8.62 ± 0.17 c | 14.64 ± 2.05 b | *** |
| ADL, % of DM | 38.47 ± 0.05 a | 23.92 ± 0.45 c | 29.85 ± 0.93 b | *** |
| hemicelluloses, % of DM | 2.95 ± 0.65 c | 17.71 ± 0.00 a | 11.80 ± 0.25 b | *** |
| cellulose, % of DM | 21.24 ± 1.20 c | 27.75 ± 0.29 a | 23.49 ± 0.45 b | *** |
| carbohydrates, % of DM | 87.87 ± 0.03 a | 78.00 ± 0.16 c | 79.78 ± 0.48 b | *** |
| proteins, % of DM | 5.35 ± 0.02 b | 9.43 ± 0.15 a | 9.02 ± 0.50 a | *** |
| fat, % of DM | 1.24 ± 0.00 c | 10.61 ± 0.00 a | 7.73 ± 0.00 b | *** |
| total fatty acids, % of DM | 0.39 ± 0.02 c | 9.17 ± 0.07 a | 5.92 ± 0.05 b | *** |
| ash, % of DM | 5.54 ± 0.02 a | 1.95 ± 0.03 c | 3.47 ± 0.02 b | *** |
| gross energy, kcal/100 g DM | 384.04 ± 0.08 c | 445.23 ± 0.10 a | 424.74 ± 0.07 b | *** |
| Fatty Acids 1 (g/100 g of FAME) | Pericarps | Seeds | Whole EMB | p-Value |
|---|---|---|---|---|
| C10:0 | 1.56 ± 0.44 a | 0.04 ± 0.00 b | 0.14 ± 0.03 b | *** |
| C12:0 | 0.32 ± 0.06 a | 0.01 ± 0.00 b | 0.03 ± 0.00 b | *** |
| C14:0 | 2.47 ± 0.04 a | 0.05 ± 0.00 c | 0.21 ± 0.00 b | *** |
| C15:0 | 0.14 ± 0.02 a | 0.01 ± 0.00 b | 0.02 ± 0.00 b | *** |
| C16:0 (PA) | 24.47 ± 0.31 a | 8.30 ± 0.04 c | 9.34 ± 0.02 b | *** |
| C16:1 cis-9 | 0.36 ± 0.05 a | 0.02 ± 0.00 b | 0.04 ± 0.00 b | *** |
| C17:0 | 0.00 ± 0.00 c | 0.11 ± 0.00 a | 0.11 ± 0.00 b | *** |
| C18:0 (SA) | 8.12 ± 0.11 a | 3.99 ± 0.04 c | 4.26 ± 0.03 b | *** |
| C18:1 trans-5 | 0.68 ± 0.03 a | 0.06 ± 0.01 b | 0.10 ± 0.01 b | *** |
| C18:1 trans-6 + trans-8 | 1.03 ± 0.12 a | 0.11 ± 0.01 b | 0.17 ± 0.02 b | *** |
| C18:1 trans-11 | 0.95 ± 0.11 a | 0.03 ± 0.01 b | 0.09 ± 0.01 b | *** |
| C18:1 cis-9 (OA) | 11.69 ± 0.12 a | 9.25 ± 0.06 b | 9.41 ± 0.05 b | *** |
| C18:1 cis-11 | 0.88 ± 0.12 a | 0.45 ± 0.04 b | 0.48 ± 0.04 b | *** |
| C18:1 cis-16 | 0.00 ± 0.00 b | 0.07 ± 0.00 a | 0.07 ± 0.00 a | *** |
| C18:2 trans-11,trans-15 | 1.68 ± 0.39 a | 0.00 ± 0.00 b | 0.11 ± 0.03 b | *** |
| C18:2 n-6 (LA) | 17.31 ± 0.57 c | 75.09 ± 0.18 a | 71.38 ± 0.15 b | *** |
| C18:3 n-3 (LNA) | 4.24 ± 0.31 a | 0.42 ± 0.01 b | 0.67 ± 0.02 b | *** |
| C20:0 | 5.20 ± 0.09a | 0.58 ± 0.03 b | 0.88 ± 0.03 b | *** |
| C20:1 cis-5 | 0.02 ± 0.04 | 0.04 ± 0.01 | 0.04 ± 0.01 | NS |
| C20:1 cis-9 | 0.50 ± 0.31 a | 0.02 ± 0.00 b | 0.05 ± 0.02 ab | * |
| C20:1 cis-11 | 0.76 ± 0.25 a | 0.22 ± 0.00 b | 0.25 ± 0.02 b | ** |
| C20:2 n-6 | 0.30 ± 0.13 a | 0.11 ± 0.01 b | 0.13 ± 0.01 ab | * |
| C20:4 n-6 | 1.32 ± 0.26 a | 0.03 ± 0.00 b | 0.11 ± 0.02 b | *** |
| C22:0 | 4.03 ± 0.13 a | 0.11 ± 0.00 c | 0.36 ± 0.01 b | *** |
| C22:3 n-6 | 0.33 ± 0.05 a | 0.14 ± 0.00 b | 0.15 ± 0.00 b | *** |
| EPA | 0.00 ± 0.00 b | 0.02 ± 0.01 a | 0.02 ± 0.01 a | * |
| C24:0 | 3.77 ± 0.15 a | 0.10 ± 0.01 c | 0.34 ± 0.01 b | *** |
| C24:1 cis-15 | 0.43 ± 0.00 a | 0.20 ± 0.00 c | 0.21 ± 0.00 b | *** |
| C25:0 | 0.80 ± 0.09 a | 0.02 ± 0.00 b | 0.07 ± 0.01 b | *** |
| C26:0 | 3.61 ± 0.63a | 0.02 ± 0.00 b | 0.25 ± 0.04 b | *** |
| Σ SFA | 55.04 ± 0.97 a | 13.41 ± 0.09 c | 16.09 ± 0.02 b | *** |
| Σ MUFA | 17.31 ± 0.39 a | 10.47 ± 0.07 b | 10.91 ± 0.09 b | *** |
| Σ PUFA | 25.28 ± 1.13 c | 76.05 ± 0.14 a | 72.79 ± 0.12 b | *** |
| SFA:PUFA ratio | 2.18 ± 0.13 a | 0.18 ± 0.00 b | 0.22 ± 0.00 b | *** |
| Σ C18 1 trans | 2.66 ± 0.22 a | 0.20 ± 0.02 b | 0.36 ± 0.02 b | *** |
| Σ C18:2 (except LA) | 1.77 ± 0.55 a | 0.22 ± 0.05 b | 0.32 ± 0.03 b | ** |
| Σ C21:1 | 1.28 ± 0.07 a | 0.28 ± 0.01 b | 0.35 ± 0.01 b | ** |
| Extract Concentration (µg/µL) | Seeds (µg GAE 1) | Pericarps (µg GAE) | Correlation with Antioxidant Activity p-Value |
|---|---|---|---|
| 100 | 468.96 ± 2.95 | 158.99 ± 11.95 | p < 0.001 |
| 50 | 248.85 ± 2.59 | 82.19 ± 6.81 | |
| 25 | 136.56 ± 9.61 | 31.02 ± 2.09 | |
| 10 | 50.39 ± 1.62 | 19.70 ± 3.62 | |
| 5 | 30.99 ± 1.32 | 8.63 ± 0.60 | |
| 1 | 7.22 ± 0.57 | 1.60 ± 0.55 |
| Compound | tR | Pericarps | Seeds | Whole EMB | p-Value | |||
|---|---|---|---|---|---|---|---|---|
| mg/100 g ± SD | % | mg/100 g ± SD | % | mg/100 g ± SD | % | |||
| gallic acid | 3.27 | 78.49 ± 5.63 | 15.02 | 63.44 ± 4.52 | 11.22 | 65.50 ± 9.95 | 12.70 | NS |
| caffeic acid | 9.27 | 0.07 ± 0.01 | 0.01 | 0.06 ± 0.01 | 0.01 | 0.07 ± 0.01 | 0.01 | NS |
| p coumaric acid | 10.26 | 0.51 ± 0.02 a | 0.10 | 0.17 ± 0.01 | 0.03 | 0.27 ± 0.01 ab | 0.05 | ** |
| ellagic acid | 11.42 | 244.67 ± 14.63 c | 46.83 | 345.02 ± 5.95 a | 61.00 | 281.79 ± 19.16 b | 54.64 | *** |
| ferulic acid | 11.38 | 0.15 ± 0.00 b | 0.03 | 0.20 ± 0.02 a | 0.04 | 0.18 ± 0.01 a | 0.04 | *** |
| sinapic acid | 11.47 | 0.02 ± 0.01 c | 0.00 | 0.04 ± 0.01 a | 0.01 | 0.03 ± 0.01 b | 0.01 | *** |
| quinic acid | 1.30 | 120.82 ± 3.65 a | 23.13 | 77.11 ± 1.44 c | 13.63 | 96.36 ± 0.60 b | 18.68 | *** |
| siringic acid | 9.81 | 7.48 ± 0.81 a | 1.43 | 0.51 ± 0.06 b | 0.09 | 2.77 ± 0.08 ab | 0.54 | ** |
| chlorogenic acid | 8.93 | 0.09 ± 0.01 | 0.02 | 0.08 ± 0.01 | 0.01 | 0.08 ± 0.01 | 0.02 | NS |
| kaempferol | 16.11 | 1.81 ± 0.04 b | 0.35 | 2.07 ± 0.04 a | 0.37 | 1.80 ± 0.15 b | 0.35 | * |
| quercetin | 15.42 | 18.76 ± 0.45 b | 3.59 | 20.91 ± 1.01 a | 3.70 | 19.18 ± 0.31 b | 3.72 | * |
| isorhamnetin | 16.19 | 7.67 ± 0.51 c | 1.47 | 14.75 ± 0.37 a | 2.61 | 9.83 ± 0.38 b | 1.91 | *** |
| myricetin | 12.64 | 7.11 ± 0.31 a | 1.36 | 5.26 ± 0.30 b | 0.93 | 5.87 ± 0.56 b | 1.14 | ** |
| isoquercetin | 11.63 | 2.30 ± 0.08 a | 0.44 | 2.21 ± 0.06 a | 0.39 | 1.99 ± 0.05 b | 0.39 | ** |
| quercetin 3-O-rhamnoside | 12.74 | 17.24 ± 0.52 c | 3.30 | 23.78 ± 0.43 a | 4.20 | 19.15 ± 0.26 b | 3.71 | *** |
| robinin | 10.77 | 0.03 ± 0.01 a | 0.01 | 0.02 ± 0.01 b | 0.00 | 0.02 ± 0.01 b | 0.00 | ** |
| rutin | 11.43 | 0.02 ± 0.01 ab | 0.00 | 0.01 ± 0.01 b | 0.00 | 0.03 ± 0.01 a | 0.01 | * |
| isorhamnetin 3-O-rutinoside | 12.41 | 0.00 ± 0.00 a | 0.00 | 0.00 ± 0.00 c | 0.00 | 0.00 ± 0.00 b | 0.00 | *** |
| quercetin 3-O-galactoside | 11.71 | 0.32 ± 0.01 a | 0.06 | 0.13 ± 0.01 c | 0.02 | 0.18 ± 0.01 b | 0.04 | *** |
| myricitrin | 11.46 | 4.62 ± 0.10 c | 0.88 | 6.80 ± 0.16 a | 1.20 | 5.26 ± 0.28 b | 1.02 | *** |
| neohesperidin | 13.11 | 0.02 ± 0.01 c | 0.00 | 0.05 ± 0.01 a | 0.01 | 0.03 ± 0.01 b | 0.00 | *** |
| myricetin 3-O-galactoside | 10.60 | 9.55 ± 0.06 a | 1.83 | 2.65 ± 0.08 c | 0.47 | 4.91 ± 0.20 b | 0.95 | *** |
| phloridzin | 13.19 | 0.05 ± 0.01 a | 0.01 | 0.04 ± 0.01 b | 0.01 | 0.04 ± 0.01 b | 0.01 | *** |
| apigenin | 16.03 | 0.01 ± 0.00 a | 0.00 | 0.00 ± 0.00 b | 0.00 | 0.00 ± 0.00 b | 0.00 | ** |
| luteolin | 15.38 | 0.01 ± 0.00 | 0.00 | 0.01 ± 0.00 | 0.00 | 0.01 ± 0.00 | 0.00 | NS |
| epicatechin | 8.72 | 0.06 ± 0.01 a | 0.01 | 0.04 ± 0.01 b | 0.01 | 0.04 ± 0.01 b | 0.01 | *** |
| catechin | 9.61 | 0.11 ± 0.01 a | 0.02 | 0.09 ± 0.01 b | 0.02 | 0.08 ± 0.01 c | 0.02 | *** |
| epigallocatechin | 7.68 | 0.05 ± 0.01 a | 0.01 | 0.03 ± 0.01 b | 0.01 | 0.04 ± 0.01 b | 0.01 | ** |
| epigallocatechin 3-O-gallate | 9.28 | 0.40 ± 0.03 a | 0.08 | 0.13 ± 0.01 c | 0.02 | 0.21 ± 0.01 b | 0.04 | *** |
| procyanidin B1 | 7.96 | 0.02 ± 0.01 a | 0.00 | 0.00 ± 0.00 c | 0.00 | 0.02 ± 0.01 b | 0.00 | *** |
| procyanidin B2 | 8.82 | 0.02 ± 0.01 a | 0.00 | 0.00 ± 0.00 b | 0.00 | 0.02 ± 0.01 ab | 0.00 | ** |
| cyanidin 3,5-di-O-glucoside | 9.65 | ND | ND | ND | ||||
| cyanidin 3-O-glucoside | 10.50 | ND | ND | ND | ||||
| cyanidin 3-O-arabinoside | 11.86 | ND | ND | ND | ||||
| delphinidin 3-O-glucoside | 10.00 | ND | ND | ND | ||||
| malvidin 3-O-glucoside | 12.01 | ND | ND | ND | ||||
| pelargonidin 3-O-glucoside | 13.94 | ND | ND | ND | ||||
| pelargonidin 3-O-rutinoside | 14.41 | ND | ND | ND | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Correddu, F.; Maldini, M.; Addis, R.; Petretto, G.L.; Palomba, M.; Battacone, G.; Pulina, G.; Nudda, A.; Pintore, G. Myrtus Communis Liquor Byproduct as a Source of Bioactive Compounds. Foods 2019, 8, 237. https://doi.org/10.3390/foods8070237
Correddu F, Maldini M, Addis R, Petretto GL, Palomba M, Battacone G, Pulina G, Nudda A, Pintore G. Myrtus Communis Liquor Byproduct as a Source of Bioactive Compounds. Foods. 2019; 8(7):237. https://doi.org/10.3390/foods8070237
Chicago/Turabian StyleCorreddu, Fabio, Mariateresa Maldini, Roberta Addis, Giacomo Luigi Petretto, Michele Palomba, Gianni Battacone, Giuseppe Pulina, Anna Nudda, and Giorgio Pintore. 2019. "Myrtus Communis Liquor Byproduct as a Source of Bioactive Compounds" Foods 8, no. 7: 237. https://doi.org/10.3390/foods8070237
APA StyleCorreddu, F., Maldini, M., Addis, R., Petretto, G. L., Palomba, M., Battacone, G., Pulina, G., Nudda, A., & Pintore, G. (2019). Myrtus Communis Liquor Byproduct as a Source of Bioactive Compounds. Foods, 8(7), 237. https://doi.org/10.3390/foods8070237









