Evaluation of Starch–Protein Interactions as a Function of pH
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
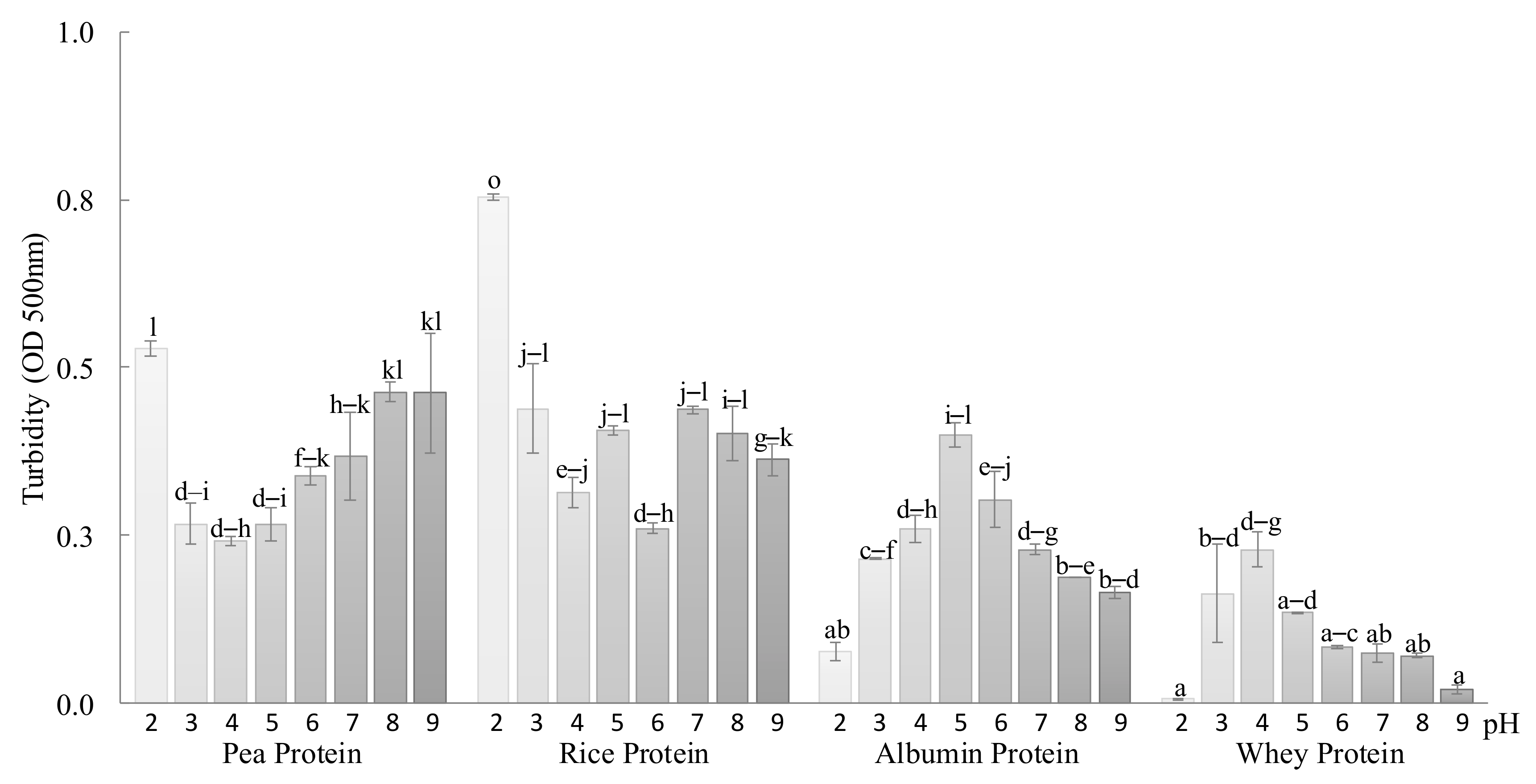
2.2.1. Protein Turbidity and Water Binding Capacity of the Starch, Proteins, and Starch–Protein Mixtures
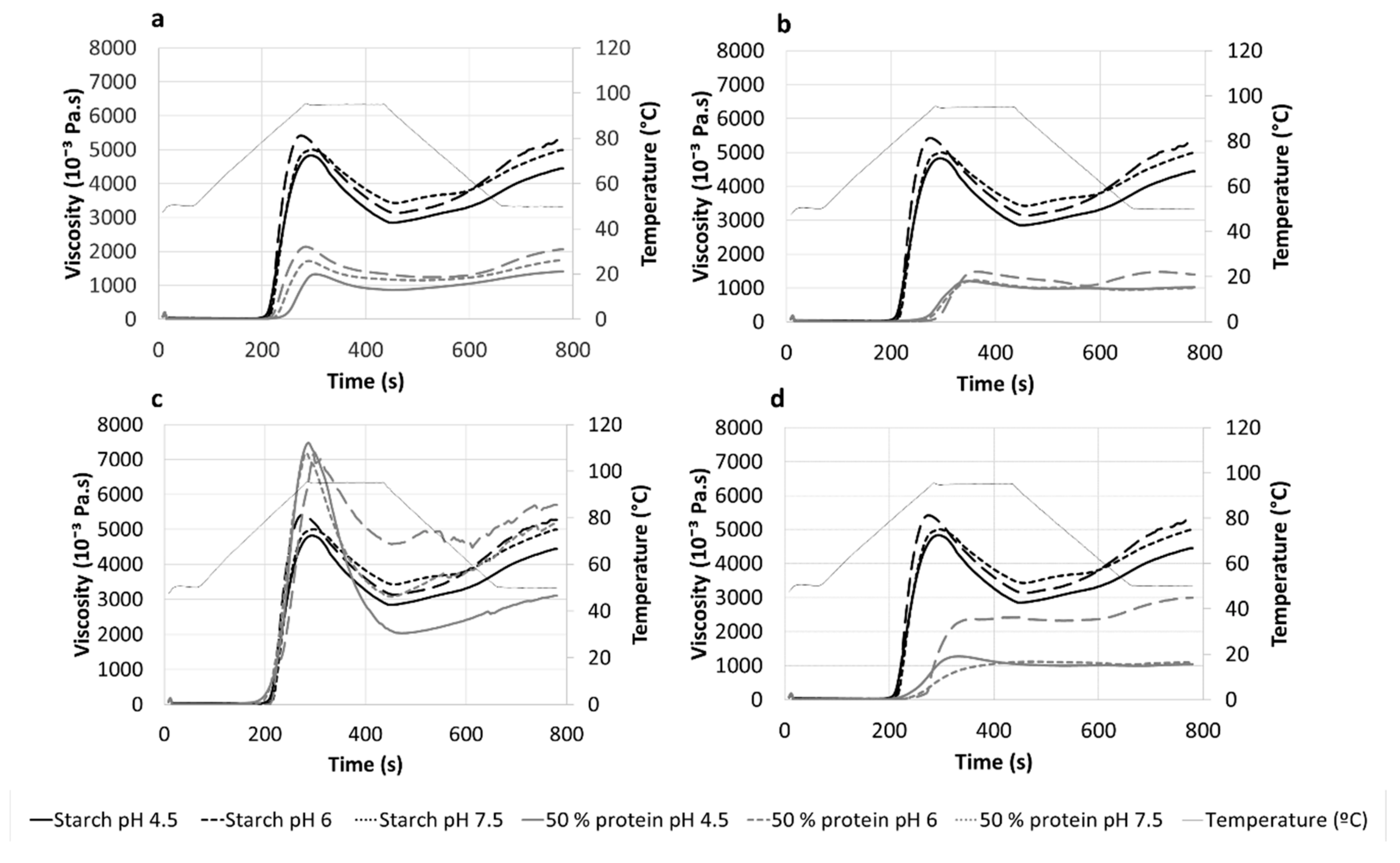
2.2.2. Pasting Properties
2.2.3. Gel Microstructure
2.2.4. Gel Texture
2.2.5. Statistical Analysis
3. Results and Discussion
3.1. Protein Solubility and Water Binding Capacity of the Mixtures
3.2. Pasting Properties
3.3. Gel Microstructure
3.4. Gel Texture
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Research and Markets. Protein Ingredients Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2018–2023. Available online: https://www.researchandmarkets.com/reports/4514555/protein-ingredients-market-global-industry (accessed on 10 March 2019).
- Oñate Narciso, J.; Brennan, C. Whey and pea protein fortification of rice starches: Effects on protein and starch digestibility and starch pasting properties. Starch 2018, 70, 1700315. [Google Scholar] [CrossRef]
- Joshi, M.; Aldred, P.; Panozzo, J.F.; Kasapis, S.; Adhikari, B. Rheological and microstructural characteristics of lentil starch-lentil protein composite pastes and gels. Food Hydrocoll. 2014, 35, 226–237. [Google Scholar] [CrossRef]
- Ribotta, P.D.; Colombo, A.; León, A.E.; Añón, M.C. Effects of soy protein on physical and rheological properties of wheat starch. Starch 2007, 59, 614–623. [Google Scholar] [CrossRef]
- Sun, Q.; Xiong, C.S.L. Functional and pasting properties of pea starch and peanut protein isolate blends. Carbohydr Polym. 2014, 101, 1134–1139. [Google Scholar]
- Jekle, M.; Mühlberger, K.; Becker, T. Starch-gluten interactions during gelatinization and its functionality in dough like model systems. Food Hydrocoll. 2016, 54, 196–201. [Google Scholar] [CrossRef]
- Chen, B.; Zhang, B.; Li, M.N.; Xie, Y.; Chen, H.Q. Effects of glutenin and gliadin modified by protein-glutaminase on pasting, rheological properties and microstructure of potato starch. Food Chem. 2018, 253, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Kett, A.P.; Chaurin, V.; Fitzsimons, S.M.; Morris, E.R.; O’Mahony, J.A.; Fenelon, M.A. Influence of milk proteins on the pasting behaviour and microstructural characteristics of waxy maize starch. Food Hydrocoll. 2013, 30, 661–671. [Google Scholar] [CrossRef]
- Onwulata, C.I.; Tunick, M.H.; Thomas-Gahring, A.E. Pasting and extrusion properties of mixed carbohydrate and whey protein isolate matrices. J. Food Process. Preserv. 2014, 38, 1577–1591. [Google Scholar] [CrossRef]
- Noisuwan, A.; Hemar, Y.; Wilkinson, B.; Bronlund, J.E. Adsorption of milk proteins onto rice starch granules. Carbohydr. Polym. 2011, 84, 247–254. [Google Scholar] [CrossRef]
- Considine, T.; Noisuwan, A.; Hemar, Y.; Wilkinson, B.; Bronlund, J.; Kasapis, S. Rheological investigations of the interactions between starch and milk proteins in model dairy systems: A review. Food Hydrocoll. 2011, 25, 2008–2017. [Google Scholar] [CrossRef]
- Shim, J.; Mulvaney, S.J. Effect of heating temperature, pH, concentration and starch/whey protein ratio on the viscoelastic properties of corn starch/whey protein mixed gels. J. Sci. Food Agric. 2001, 81, 706–717. [Google Scholar] [CrossRef]
- Bravo-Núñez, Á.; Gómez, M. Physicochemical properties of native and extruded maize flours in the presence of animal proteins. J. Food Eng. 2019, 243, 49–56. [Google Scholar] [CrossRef]
- Lopes da Silva, J.A.; Rao, M.A. Rheological behaviour of food gels. In Rheology of Fluid and Semisolid Foods Principles and Applications; Rao, M.A., Ed.; Springer: New York, NY, USA, 2007; pp. 339–401. [Google Scholar]
- Damodaran, S. Interfaces, Protein Films, and Foams. In Advance Food Nutrition Research; Kinsella, J.E., Ed.; Academic Press: San Diego, CA, USA, 1990; Volume 34, pp. 1–79. [Google Scholar]
- Zhang, F.; Skoda, M.W.A.; Jacobs, R.M.J.; Martin, R.A.; Martin, C.M.; Schreiber, F. Protein interactions studied by SAXS: Effect of ionic strength and protein concentration for BSA in aqueous solutions. J. Phys. Chem. B 2007, 111, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Mantzourani, I.; Plessas, S.; Odatzidou, M.; Alexopoulos, A.; Galanis, A.; Bezirtzoglou, E.; Bekatorou, A. Effect of a novel Lactobacillus paracasei starter on sourdough bread quality. Food Chem. 2019, 271, 259–265. [Google Scholar] [CrossRef]
- Gómez, M.; Doyagüe, M.J.; de la Hera, E. Addition of pin-milled pea flour and air-classified fractions in layer and sponge cakes. LWT-Food Sci. Technol. 2012, 46, 142–147. [Google Scholar] [CrossRef]
- Babiker, E.E. Effect of transglutaminase treatment on the functional properties of native and chymotrypsin-digested soy protein. Food Chem. 2000, 70, 139–145. [Google Scholar] [CrossRef]
- AACC. Approved Methods of the American Association of Cereal Chemists, 11th ed.; American Association of Cereal Chemist: St. Paul, MI, USA, 2012. [Google Scholar]
- Martínez, M.M.; Pico, J.; Gómez, M. Effect of different polyols on wheat and maize starches paste and gel properties. Food Hydrocoll. 2015, 44, 81–85. [Google Scholar] [CrossRef]
- Guzey, D.; McClements, D.J. Characterization of β-lactoglobulin–chitosan interactions in aqueous solutions: A calorimetry, light scattering, electrophoretic mobility and solubility study. Food Hydrocoll. 2006, 20, 124–131. [Google Scholar] [CrossRef]
- Gharbi, N.; Labbafi, M. Effect of processing on aggregation mechanism of egg white proteins. Food Chem. 2018, 252, 126–133. [Google Scholar] [CrossRef]
- Li, Q.; Zhao, Z. Interaction between lactoferrin and whey proteins and its influence on the heat-induced gelation of whey proteins. Food Chem. 2018, 252, 92–98. [Google Scholar] [CrossRef]
- Ferreira Machado, F.; Coimbra, J.S.R.; Garcia Rojas, E.E.; Minim, L.A.; Oliveira, F.C.; Sousa, R.d.C.S. Solubility and density of egg white proteins: Effect of pH and saline concentration. LWT-Food Sci. Technol. 2007, 40, 1304–1307. [Google Scholar] [CrossRef]
- Pelegrine, D.H.G.; Gasparetto, C.A. Whey proteins solubility as function of temperature and pH. LWT-Food Sci. Technol. 2005, 38, 77–80. [Google Scholar] [CrossRef]
- Vojdani, F. Solubility. In Methods of Testing Protein Functionality; Hall, G.M., Ed.; Blackie Academic & Profesional: London, UK, 1996; pp. 11–49. [Google Scholar]
- Shand, P.J.; Ya, H.; Pietrasik, Z.; Wanasundara, P.K.J.P.D. Physicochemical and textural properties of heat-induced pea protein isolate gels. Food Chem. 2007, 102, 1119–1130. [Google Scholar] [CrossRef]
- Amagliani, L.; O’Regan, J.; Kelly, A.L.; O’Mahony, J.A. Composition and protein profile analysis of rice protein ingredients. J. Food Compos. Anal. 2017, 59, 126–133. [Google Scholar] [CrossRef]
- Wang, S.; Copeland, L. Effect of acid hydrolysis on starch structure and functionality: A Review. Crit. Rev. Food Sci. Nutr. 2015, 55, 1081–1097. [Google Scholar] [CrossRef]
- Shin, M.; Gang, D.O.; Song, J.Y. Effects of protein and transglutaminase on the preparation of gluten-free rice bread. Food Sci. Biotechnol. 2010, 19, 951–956. [Google Scholar] [CrossRef]
- Singh, H.; Sodhi, N.S.; Singh, N. Structure and functional properties of acid thinned sorghum starch. Int. J. Food Prop. 2009, 12, 713–725. [Google Scholar] [CrossRef]
- Polesi, L.F.; Sarmento, S.B.S. Structural and physicochemical characterization of RS prepared using hydrolysis and heat treatments of chickpea starch. Starch 2011, 63, 226–235. [Google Scholar] [CrossRef]
- Colonna, P.; Leloup, V.; Buléon, A. Limiting factors of starch hydrolysis. Eur. J. Clin. Nutr. 1992, 46, S17–S32. [Google Scholar]
- Donovan, J.W.; Mapes, C.J. Multiple phase transitions of starches and nägeli amylodextrins. Starch 1980, 32, 190–193. [Google Scholar] [CrossRef]
- Donovan, J.W. Phase transitions of the starch-water system. Biopolymers 1979, 18, 263–275. [Google Scholar] [CrossRef]
- Rasper, V. Theoretical aspests of amylographology. In The Amylograph Handbook; Shuey, W.C., Tipples, K.H., Eds.; American Association of Cereal Chemists: St. Paul, MI, USA, 1980; pp. 1–6. [Google Scholar]
- Ferreira, M.; Hofer, C.; Raemy, A. A calorimetric study of egg white proteins. J. Therm. Anal. 1997, 48, 683–690. [Google Scholar] [CrossRef]
- Biliaderis, C.G.; Maurice, T.J.; Vose, J.R. Starch gelatinization phenomena studied by differential scanning calorimetry. J. Food Sci. 1980, 45, 1669–1674. [Google Scholar] [CrossRef]
- Van Kleef, F. Thermally induced protein gelation: gelation and rheological characterization of highly concentrated ovalbumin and soybean protein gels. Biopolymers 1986, 25, 31–59. [Google Scholar] [CrossRef]
- Matsudomi, N.; Rector, D.; Kinsella, J.E. Gelation of bovine serum albumin and β-lactoglobulin; effects of pH, salts and thiol reagents. Food Chem. 1991, 40, 55–69. [Google Scholar] [CrossRef]
- Villanueva, M.; Ronda, F.; Moschakis, T.; Lazaridou, A.; Biliaderis, C.G. Impact of acidification and protein fortification on thermal properties of rice, potato and tapioca starches and rheological behaviour of their gels. Food Hydrocoll. 2018, 79, 20–29. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.J. Structures and physicochemical properties of acid-thinned corn, potato and rice starches. Starch 2001, 53, 570–576. [Google Scholar] [CrossRef]
- Wang, Y.J.; Truong, V.D.; Wang, L. Structures and rheological properties of corn starch as affected by acid hydrolysis. Carbohydr. Polym. 2003, 52, 327–333. [Google Scholar] [CrossRef]
- Yang, H.; Irudayaraj, J.; Otgonchimeg, S.; Walsh, M. Rheological study of starch and dairy ingredient-based food systems. Food Chem. 2004, 86, 571–578. [Google Scholar] [CrossRef]

| WBC | Hardness (N) | |||||
|---|---|---|---|---|---|---|
| pH 4.5 | pH 6.0 | pH 7.5 | pH 4.5 | pH 6.0 | pH 7.5 | |
| Starch | 0.72 ± 0.00 d | 0.75 ± 0.01 d | 0.75 ± 0.02 d | 0.79 ± 0.10 cd | 1.00 ± 0.06 d | 0.77 ± 0.03 cd |
| Pea protein | 2.69 ± 0.16 ij | 3.37 ± 0.06 k | 4.09 ± 0.04 l | nd | nd | nd |
| Rice protein | 2.6 ± 0.04 hi | 2.52 ± 0.04 h | 2.75 ± 0.05 j | nd | nd | nd |
| Egg albumin protein | nd | nd | nd | nd | nd | nd |
| Whey protein | nd | nd | nd | nd | nd | nd |
| Starch–pea protein | 1.46 ± 0.00 e | 1.60 ± 0.01 f | 2.00 ± 0.03 g | 0.25 ± 0.03 a | 0.15 ± 0.00 a | 0.09 ± 0.00 a |
| Starch–rice protein | 1.48 ± 0.05 ef | 1.39 ± 0.01 e | 1.48 ± 0.03 ef | nd | nd | nd |
| Starch–egg albumin protein | 0.35 ± 0.07 c | 0.08 ± 0.03 ab | 0.14 ± 0.08 b | 0.34 ± 0.04 ab | 0.23 ± 0.01 a | 0.37 ± 0.03 ab |
| Starch–whey protein | 0.02 ± 0.00 a | nd | nd | 0.10 ± 0.02 a | 0.09 ± 0.00 a | 0.55 ± 0.44 bc |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bravo-Núñez, Á.; Garzón, R.; Rosell, C.M.; Gómez, M. Evaluation of Starch–Protein Interactions as a Function of pH. Foods 2019, 8, 155. https://doi.org/10.3390/foods8050155
Bravo-Núñez Á, Garzón R, Rosell CM, Gómez M. Evaluation of Starch–Protein Interactions as a Function of pH. Foods. 2019; 8(5):155. https://doi.org/10.3390/foods8050155
Chicago/Turabian StyleBravo-Núñez, Ángela, Raquel Garzón, Cristina M. Rosell, and Manuel Gómez. 2019. "Evaluation of Starch–Protein Interactions as a Function of pH" Foods 8, no. 5: 155. https://doi.org/10.3390/foods8050155
APA StyleBravo-Núñez, Á., Garzón, R., Rosell, C. M., & Gómez, M. (2019). Evaluation of Starch–Protein Interactions as a Function of pH. Foods, 8(5), 155. https://doi.org/10.3390/foods8050155






