Recent Advances in the Use of Sourdough Biotechnology in Pasta Making
Abstract
1. Sourdough Fermentation: Innovations from the Past
1.1. The Key Role of Lactic Acid Bacteria
1.2. Nutritional Effects of Sourdough Fermentation
1.3. Functional Effects of Sourdough Fermentation
2. Cereal-Based Sourdough in Pasta Making
2.1. Nutritional Aspects
2.2. The Celiac Issue and Gluten Reduction
2.3. Textural and Cooking Properties and Sensory Profile
3. Fermented Legumes and Pseudocereals in Pasta Making
3.1. Effect on Chemical, Nutritional, and Functional Quality
3.2. Sensory Acceptability and Textural Properties of Fortified Products
3.3. Cooking Properties of Fortified Products
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Leroy, F.; De Vuyst, L. Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends Food Sci. Technol. 2004, 15, 67–78. [Google Scholar] [CrossRef]
- Gobbetti, M.; De Angelis, M.; Di Cagno, R.; Calasso, M.; Archetti, G.; Rizzello, C.G. Novel insights on the functional/nutritional features of the sourdough fermentation. Int. J. Food Microbiol. 2018, in press. [Google Scholar]
- Gobbetti, M.; Rizzello, C.G.; Di Cagno, R.; De Angelis, M. How the sourdough may affect the functional features of leavened baked goods. Food Microbiol. 2014, 37, 30–40. [Google Scholar] [CrossRef]
- Coda, R.; Rizzello, C.G.; Gobbetti, M. Use of sourdough fermentation and pseudo-cereals and leguminous flours for the making of a functional bread enriched of γ-aminobutyric acid (GABA). Int. J. Food Microbiol. 2010, 137, 236–245. [Google Scholar] [CrossRef]
- Coda, R.; Di Cagno, R.; Rizzello, C.G.; Nionelli, L.; Edema, M.O.; Gobbetti, M. Utilization of African grains for sourdough bread making. J. Food Sci. 2011, 76, M329–M335. [Google Scholar] [CrossRef]
- Moroni, A.V.; Zannini, E.; Sensidoni, G.; Arendt, E.K. Exploitation of buckwheat sourdough for the production of wheat bread. Eur. Food Res. Technol. 2012, 235, 659–668. [Google Scholar] [CrossRef]
- Rizzello, C.G.; Coda, R.; Gobbetti, M. Use of sourdough fermentation and nonwheat flours for enhancing nutritional and healthy properties of wheat-based foods. In Fermented Foods in Health and Disease Prevention; Academic Press: Cambridge, MA, USA, 2017; pp. 433–452. [Google Scholar]
- Chavan, R.S.; Chavan, S.R. Sourdough technology-a traditional way for wholesome foods: A review. Compr. Rev. Food Sci. Food Saf. 2011, 10, 169–182. [Google Scholar] [CrossRef]
- Hammes, W.P.; Gänzle, M.G. Sourdough breads and related products. In Microbiology of Fermented Foods, 2nd ed.; Woods, B.J.B., Ed.; Blackie Academic: London, UK, 1998; pp. 199–216. [Google Scholar]
- De Vuyst, L.; Neysens, P. The sourdough microflora: Biodiversity and metabolic interactions. Trends Food Sci. Technol. 2005, 16, 43–56. [Google Scholar] [CrossRef]
- Minervini, F.; De Angelis, M.; Di Cagno, R.; Pinto, D.; Siragusa, S.; Rizzello, C.G.; Gobbetti, M. Robustness of Lactobacillus plantarum starters during daily propagation of wheat flour sourdough type I. Food Microbiol. 2010, 27, 897–908. [Google Scholar] [CrossRef] [PubMed]
- Gänzle, M.G.; Vermeulen, N.; Vogel, R.F. Carbohydrate, peptide and lipid metabolism of lactic acid bacteria in sourdough. Food Microbiol. 2007, 24, 128–138. [Google Scholar] [CrossRef]
- Coda, R.; Di Cagno, R.; Gobbetti, M.; Rizzello, C.G. Sourdough lactic acid bacteria: Exploration of non-wheat cereal-based fermentation. Food Microbiol. 2014, 37, 51–58. [Google Scholar] [CrossRef]
- Speranza, B.; Bevilacqua, A.; Corbo, M.R.; Sinigaglia, M. Starter Cultures in Food Production; Wiley Blackwell: Hoboken, NJ, USA, 2017. [Google Scholar]
- Ludwig, D.S. The glycemic index: Physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA 2002, 287, 2414–2423. [Google Scholar] [CrossRef]
- Di Cagno, R.; De Angelis, M.; Lavermicocca, P.; De Vincenzi, M.; Giovannini, C.; Faccia, M.; Gobbetti, M. Proteolysis by sourdough lactic acid bacteria: Effects on wheat flour protein fractions and gliadin peptides involved in human cereal intolerance. Appl. Environ. Microbiol. 2002, 68, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Pessione, E.; Cirrincione, S. Bioactive molecules released in food by lactic acid bacteria: Encrypted peptides and biogenic amines. Front. Microbiol. 2016, 7, 876. [Google Scholar] [CrossRef] [PubMed]
- Silow, C.; Axel, C.; Zannini, E.; Arendt, E.K. Current status of salt reduction in bread and bakery products—A review. J. Cereal Sci. 2016, 72, 135–145. [Google Scholar] [CrossRef]
- De Wardener, H.E.; MacGregor, G.A. Harmful effects of dietary salt in addition to hypertension. J. Hum. Hypertens. 2002, 16, 213. [Google Scholar] [CrossRef]
- Kopeć, A.; Pysz, M.; Borczak, B.; Sikora, E.; Rosell, C.M.; Collar, C.; Sikora, M. Effects of sourdough and dietary fibers on the nutritional quality of breads produced by bake-off technology. J. Cereal Sci. 2011, 54, 499–505. [Google Scholar] [CrossRef]
- Curiel, J.A.; Coda, R.; Centomani, I.; Summo, C.; Gobbetti, M.; Rizzello, C.G. Exploitation of the nutritional and functional characteristics of traditional Italian legumes: The potential of sourdough fermentation. Int. J. Food Microbiol. 2015, 196, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Katina, K.; Salmenkallio-Marttila, M.; Partanen, R.; Forssell, P.; Autio, K. Effects of sourdough and enzymes on staling of high-fiber wheat bread. LWT Food Sci. Technol. 2006, 39, 479–491. [Google Scholar] [CrossRef]
- Lu, Z.X.; Walker, K.Z.; Muir, J.G.; Mascara, T.; O’Dea, K. Arabinoxylan fiber, a byproduct of wheat flour processing, reduces the postprandial glucose response in normoglycemic subjects. Am. J. Clin. Nutr. 2000, 71, 1123–1128. [Google Scholar] [CrossRef]
- Lu, Z.X.; Walker, K.Z.; Muir, J.G.; O’dea, K. Arabinoxylan fiber improves metabolic control in people with Type II diabetes. Eur. J. Clin. Nut. 2004, 58, 621. [Google Scholar] [CrossRef]
- Möhlig, M.; Koebnick, C.; Weickert, M.O.; Lueder, W.; Otto, B.; Steiniger, J.; Twilfert, M.; Meuser, F.; Pfeiffer, A.F.H.; Zunft, H.J. Arabinoxylan-enriched meal increases serum ghrelin levels in healthy humans. Horm. Metab. Res. 2005, 37, 303–308. [Google Scholar] [CrossRef]
- Broekaert, W.F.; Courtin, C.M.; Verbeke, K.; van de Wiele, T.; Verstraete, W.; Delcour, J.A. Prebiotic and other health-related effects of cereal-derived arabinoxylans, arabinoxylan-oligosaccharides, and xylooligosaccharides. Crit. Rev. Food Sci. Nutr. 2011, 51, 178–194. [Google Scholar] [CrossRef] [PubMed]
- Lynch, K.M.; Coffey, A.; Arendt, E.K. Exopolysaccharide producing lactic acid bacteria: Their techno-functional role and potential application in gluten-free bread products. Food Res. Int. 2018, 110, 52–61. [Google Scholar] [CrossRef]
- Ryan, P.M.; Ross, R.P.; Fitzgerald, G.F.; Caplice, N.M.; Stanton, C. Sugar-coated: Exopolysaccharide producing lactic acid bacteria for food and human health applications. Food Funct. 2015, 6, 679–693. [Google Scholar] [CrossRef] [PubMed]
- Corsetti, A.; Settanni, L. Lactobacilli in sourdough fermentation. Food Res. Int. 2007, 40, 539–558. [Google Scholar] [CrossRef]
- Inoue, K.; Shirai, T.; Ochiai, H.; Kassao, M.; Hayakawa, K.; Rimura, M.; Sansawa, H. Blood-pressure-lowering effect of a novel fermented milk containing gamma aminobutyric acid (GABA) in mild hypertensive. Eur. J. Clin. Nutr. 2003, 57, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Naramoto, K.; Koyama, M. Blood-pressure-lowering effect of fermented buckwheat sprouts in spontaneously hypertensive rats. J. Funct. Foods 2013, 5, 406–415. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Bioactive peptides. J. AOAC Int. 2008, 91, 914–931. [Google Scholar] [PubMed]
- Gobbetti, M.; Minervini, F.; Rizzello, C.G. Bioactive peptides in dairy products. In Handbook of Food Products Manufacturing; Hui, Y.H., Ed.; Wiley & Sons Publication: New York, NY, USA, 2007; pp. 489–517. [Google Scholar]
- Rizzello, C.G.; Cassone, A.; Di Cagno, R.; Gobbetti, M. Synthesis of angiotensin I-converting enzyme (ACE)-inhibitory peptides and γ-aminobutyric acid (GABA) during sourdough fermentation by selected lactic acid bacteria. J. Agric. Food Chem. 2008, 56, 6936–6943. [Google Scholar] [CrossRef]
- Nionelli, L.; Coda, R.; Curiel, J.A.; Poutanen, K.; Gobbetti, M.; Rizzello, C.G. Manufacture and characterization of a yogurt-like beverage made with oat flakes fermented by selected lactic acid bacteria. Int. J. Food Microbiol. 2014, 185, 17–26. [Google Scholar]
- Rizzello, C.G.; Nionelli, L.; Coda, R.; Gobbetti, M. Synthesis of the cancer preventive peptide lunasin by lactic acid bacteria during sourdough fermentation. Nutr. Cancer 2012, 64, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Rizzello, C.G.; Lorusso, A.; Russo, V.; Pinto, D.; Marzani, B.; Gobbetti, M. Improving the antioxidant properties of quinoa flour through fermentation with selected autochthonous lactic acid bacteria. Int. J. Food Microbiol. 2017, 241, 252–261. [Google Scholar] [CrossRef] [PubMed]
- Malaguti, M.; Dinelli, G.; Leoncini, E.; Bregola, V.; Bosi, S.; Cicero, A.F.; Hrelia, S. Bioactive peptides in cereals and legumes: Agronomical, biochemical and clinical aspects. Int. J. Mol. Sci. 2014, 15, 21120–21135. [Google Scholar] [CrossRef] [PubMed]
- Coda, R.; Rizzello, C.G.; Pinto, D.; Gobbetti, M. Selected lactic acid bacteria synthesize antioxidant peptides during sourdough fermentation of cereal flours. Appl. Environ. Microbiol. 2012, 78, 1087–1096. [Google Scholar] [CrossRef]
- Rizzello, C.G.; Tagliazucchi, D.; Babini, E.; Rutella, G.S.; Saa, D.L.T.; Gianotti, A. Bioactive peptides from vegetable food matrices: Research trends and novel biotechnologies for synthesis and recovery. J. Funct. Foods 2016, 27, 549–569. [Google Scholar] [CrossRef]
- Giacco, R.; Vitale, M.; Riccardi, G. Pasta: Role in diet. In The Encyclopedia of Food and Health; Caballero, B., Finglas, P., Toldrá, F., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 242–245. [Google Scholar]
- Capozzi, V.; Menga, V.; Digesu, A.M.; De Vita, P.; van Sinderen, D.; Cattivelli, L.; Fares, C.; Spano, G. Biotechnological production of vitamin B2-enriched bread and pasta. J. Agric. Food Chem. 2011, 59, 8013–8020. [Google Scholar] [CrossRef] [PubMed]
- Nisha, P.; Singhal, R.S.; Pandit, A.B. A study on degradation kinetics of riboflavin in green gram whole (Vigna radiate L.). Food Chem. 2005, 89, 577–582. [Google Scholar] [CrossRef]
- Jung, M.Y.; Oh, Y.S.; Kim, D.K.; Kim, H.J.; Min, D.B. Photoinduced generation of 2,3-butanedione from riboflavin. J. Agric. Food Chem. 2007, 55, 170–174. [Google Scholar] [CrossRef]
- Thakur, K.; Tomar, S.K.; De, S. Lactic acid bacteria as a cell factory for riboflavin production. Microb. Biotechnol. 2016, 9, 441–451. [Google Scholar] [CrossRef]
- Food and Nutrition Board. Riboflavin. Dietary Reference Intakes: Thiamin, Riboflavin, Niacin, Vitamin B6, Vitamin B12, Pantothenic Acid, Biotin, Folate and Choline; National Academies Press: Washington, DC, USA, 1998. [Google Scholar]
- Brand-Miller, J.; Hayne, S.; Petocz, P.; Colagiuri, S. Low-glycemic index diets in the management of diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 2003, 26, 2261–2267. [Google Scholar] [CrossRef]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.; Manson, J.; Liu, S. Glycemic index, glycemic load, and risk of type 2 diabetes. Am. J. Clin. Nutr. 2002, 76, 274S–280S. [Google Scholar] [CrossRef] [PubMed]
- Di Cagno, R.; De Angelis, M.; Alfonsi, G.; de Vincenzi, M.; Silano, M.; Vincentini, O.; Gobbetti, M. Pasta made from durum wheat semolina fermented with selected lactobacilli as a tool for a potential decrease of the gluten intolerance. J. Agric. Food Chem. 2005, 53, 4393–4402. [Google Scholar] [CrossRef]
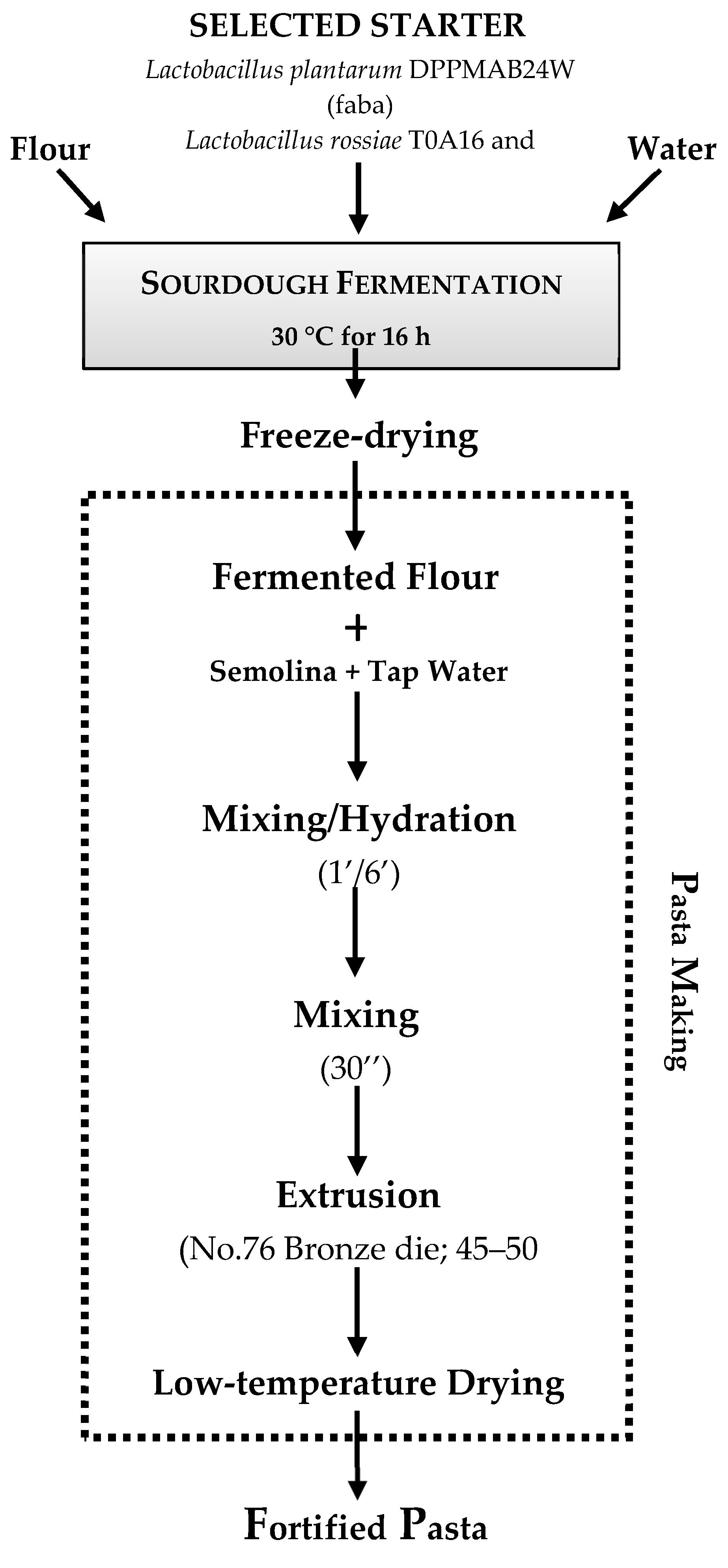
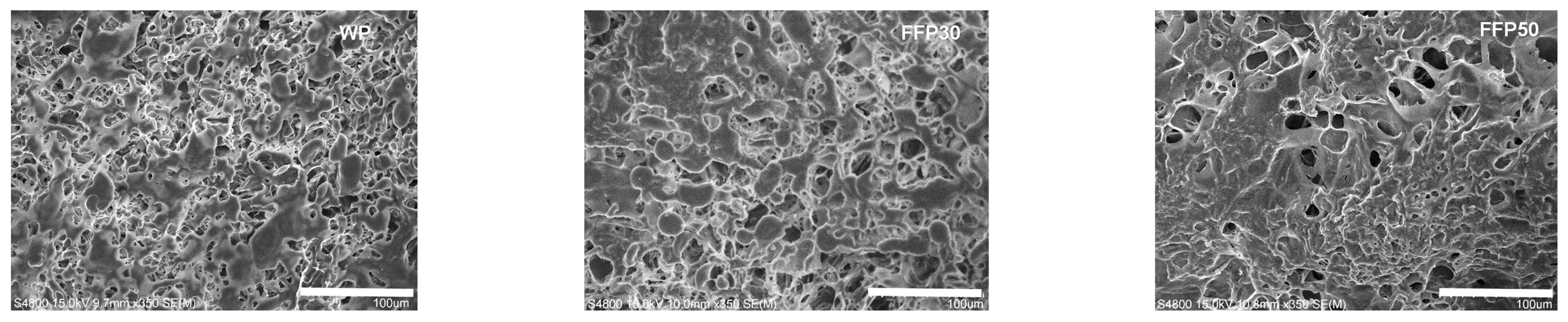
- Rizzello, C.G.; Verni, M.; Koivula, H.; Montemurro, M.; Seppa, L.; Kemell, M.; Katina, K.; Coda, R.; Gobbetti, M. Influence of fermented faba bean flour on the nutritional, technological and sensory quality of fortified pasta. Food Funct. 2017, 8, 860–871. [Google Scholar] [CrossRef]
- Rosa-Sibakov, N.; Heiniö, R.L.; Cassan, D.; Holopainen-Mantila, U.; Micard, V.; Lantto, R.; Sozer, N. Effect of bioprocessing and fractionation on the structural, textural and sensory properties of gluten-free faba bean pasta. LWT Food Sci. Technol. 2016, 67, 27–36. [Google Scholar] [CrossRef]
- Östman, E.M.; Nilsson, M.; Elmståhl, H.L.; Molin, G.; Björck, I.M.E. On the effect of lactic acid on blood glucose and insulin responses to cereal products: Mechanistic studies in healthy subjects and in vitro. J. Cereal Sci. 2002, 36, 339–346. [Google Scholar] [CrossRef]
- Fois, S.; Piu, P.P.; Sanna, M.; Roggio, T.; Catzeddu, P. Starch digestibility and properties of fresh pasta made with semolina-based liquid sourdough. LWT Food Sci. Technol. 2018, 89, 496–502. [Google Scholar] [CrossRef]
- Elli, L.; Branchi, F.; Tomba, C.; Villalta, D.; Norsa, L.; Ferretti, F.; Roncoroni, L.; Bardella, M.T. Diagnosis of gluten related disorders: Celiac disease, wheat allergy and non-celiac gluten sensitivity. World J. Gastroenterol. 2015, 21, 7110. [Google Scholar] [CrossRef] [PubMed]
- Calasso, M.; Francavilla, R.; Cristofori, F.; De Angelis, M.; Gobbetti, M. New protocol for production of reduced-gluten wheat bread and pasta and clinical effect in patients with irritable bowel syndrome: A randomised, double-blind, cross-over study. Nutrients 2018, 10, 1873. [Google Scholar] [CrossRef]
- Catassi, C.; Aleadini, A.; Bojarski, C.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; De Magistris, L.; Dieterich, W.; Di Liberto, D.; et al. The overlapping area of non-celiac gluten sensitivity (NCGS) and wheat-sensitive irritable bowel syndrome (IBS): An update. Nutrients 2017, 9, 1268. [Google Scholar] [CrossRef]
- Reg. EU 828/2014. Commission Implementing Regulation (EU) No 828/2014 of 30 July 2014 on the Requirements for the Provision of Information to Consumers on the Absence or Reduced Presence of Gluten in Food. Available online: https://eur-lex.europa.eu (accessed on 27 February 2019).
- FDA Guidance for Industry Gluten-Free Labeling of Foods—Small Entity Compliance Guide 2014. Available online: http://www.fda.gov/FoodGuidances (accessed on 27 February 2019).
- Shan, L.; Qiao, S.W.; Arentz-Hansen, H.; Molberg, Ø.; Gray, G.M.; Sollid, L.M.; Khosla, C. Identification and analysis of multivalent proteolytically resistant peptides from gluten: Implications for celiac sprue. J. Proteome Res. 2005, 4, 1732–1741. [Google Scholar] [CrossRef] [PubMed]
- Curiel, J.A.; Coda, R.; Limitone, A.; Katina, K.; Raulio, M.; Giuliani, G.; Rizzello, C.G.; Gobbetti, M. Manufacture and characterization of pasta made with wheat flour rendered gluten-free using fungal proteases and selected sourdough lactic acid bacteria. J. Cereal Sci. 2014, 59, 79–87. [Google Scholar] [CrossRef]
- Mastromatteo, M.; Chillo, S.; Iannetti, M.; Civica, V.; Del Nobile, M.A. Formulation optimisation of gluten-free functional spaghetti based on quinoa, maize and soy flours. Int. J. Food Sci. Technol. 2011, 46, 1201–1208. [Google Scholar] [CrossRef]
- Taylor, J.R.N.; Belton, P.S.; Beta, T.; Duodu, K.G. Increasing the utilisation of sorghum, millets and pseudocereals: Developments in the science of their phenolic phytochemicals, biofortification and protein functionality. J. Cereal Sci. 2014, 59, 257–275. [Google Scholar] [CrossRef]
- Taylor, J.R.N.; Emmambux, N.M. Developments in our understanding of sorghum polysaccharides and their health benefits. Cereal Chem. 2010, 87, 263–271. [Google Scholar] [CrossRef]
- Marengo, M.; Bonomi, F.; Marti, A.; Pagani, M.A.; Elkhalifa, A.E.O.; Iametti, S. Molecular features of fermented and sprouted sorghum flours relate to their suitability as components of enriched gluten-free pasta. LWT Food Sci. Technol. 2015, 63, 511–518. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Zee, S.Y.; Corke, H. Physicochemical properties related to quality of rice noodles. Cereal Chem. 1999, 76, 861–867. [Google Scholar] [CrossRef]
- Chillo, S.; Laverse, J.; Falcone, P.M.; Del Nobile, M.A. Quality of spaghetti in base amaranthus wholemeal flour added with quinoa, broad bean and chick pea. J. Food Eng. 2008, 84, 101–107. [Google Scholar] [CrossRef]
- Lopez-Garcia, R. Quinoa: A traditional Andean crop with new horizons. Cereal Food World 2007, 52, 88. [Google Scholar] [CrossRef]
- Torres, A.; Frias, J.; Granito, M.; Guerra, M.; Vidal-Valverde, C. Chemical, biological and sensory evaluation of pasta products supplemented with agalactoside-free lupin flours. J. Sci. Food Agric. 2007, 87, 74–81. [Google Scholar] [CrossRef]
- Torres, A.; Frias, J.; Granito, M.; Vidal-Valverde, C. Fermented pigeon pea (Cajanus cajan) ingredients in pasta products. J. Agric. Food Chem. 2006, 54, 6685–6691. [Google Scholar] [CrossRef] [PubMed]
- Torres, A.; Frias, J.; Granito, M.; Vidal-Valverde, C. Germinated Cajanus cajan seeds as ingredients in pasta products: Chemical, biological and sensory evaluation. Food Chem. 2007, 101, 202–211. [Google Scholar] [CrossRef]
- Zhang, J.H.; Tatsumi, E.; Ding, C.H.; Li, L.T. Angiotensin I-converting enzyme inhibitory peptides in douchi, a Chinese traditional fermented soybean product. Food Chem. 2006, 98, 551–557. [Google Scholar] [CrossRef]
- Song, Y.S.; Frias, J.; Martinez-Villaluenga, C.; Vidal-Valverde, C.; Gonzalez de Mejia, E. Immunoreactivity reduction of soybean meal by fermentation, effect on amino acid composition and antigenicity of commercial soy products. Food Chem. 2008, 108, 571–581. [Google Scholar] [CrossRef]
- Coda, R.; Melama, L.; Rizzello, C.G.; Curiel, J.A.; Sibakov, J.; Holopainen, U.; Pulkkinen, M.; Sozer, N. Effect of air classification and fermentation by Lactobacillus plantarum VTT E-133328 on faba bean (Vicia faba L.) flour nutritional properties. Int. J. Food Microbiol. 2015, 193, 34–42. [Google Scholar] [PubMed]
- Giménez, M.A.; Drago, S.R.; Bassett, M.N.; Lobo, M.O.; Sammán, N.C. Nutritional improvement of corn pasta-like product with broad bean (Vicia faba) and quinoa (Chenopodium quinoa). Food Chem. 2016, 199, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Rizzello, C.G.; Losito, I.; Facchini, L.; Katina, K.; Palmisano, F.; Gobbetti, M.; Coda, R. Degradation of vicine, convicine and their aglycones during fermentation of faba bean flour. Sci. Rep. 2016, 6, 32452. [Google Scholar] [CrossRef]
- Crisan, E.V.; Sands, A. Edible Mushrooms, Nutritional Value. In The Biology and Cultivation of Edible Mushrooms; Chang, S.T., Hayes, W.A., Eds.; Academic Press: Cambridge, MA, USA, 1978. [Google Scholar]
- Petitot, M.; Boyer, L.; Minier, C.; Micard, V. Fortification of pasta with split pea and faba bean flours: Pasta processing and quality evaluation. Food Res. Int. 2010, 43, 634–641. [Google Scholar] [CrossRef]
- De Angelis, M.; Damiano, N.; Rizzello, C.G.; Cassone, A.; Di Cagno, R.; Gobbetti, M. Sourdough fermentation as a tool for the manufacture of low-glycemic index white wheat bread enriched in dietary fiber. Eur. Food Res. Technol. 2009, 229, 593–601. [Google Scholar] [CrossRef]
- Lorusso, A.; Verni, M.; Montemurro, M.; Coda, R.; Gobbetti, M.; Rizzello, C.G. Use of fermented quinoa flour for pasta making and evaluation of the technological and nutritional features. LWT Food Sci. Technol. 2017, 78, 215–221. [Google Scholar] [CrossRef]
- Martínez-Villaluenga, C.; Torres, A.; Frias, J.; Vidal-Valverde, C. Semolina supplementation with processed lupin and pigeon pea flours improve protein quality of pasta. LWT Food Sci. Technol. 2010, 43, 617–622. [Google Scholar] [CrossRef]
- Lampart-Szczapa, E.; Obuchowski, W.; Czaczyk, K.; Pastuszewska, B.; Buraczewska, L. Effect of lupine flour on the quality and oligosaccharides of pasta and crisps. Mol. Nutr. Food Res. 1997, 41, 219–223. [Google Scholar] [CrossRef]
- Frías, J.; Granito, M.; Vidal-Valverde, C. Fermentation as a process to improve the nutritional quality of grain legumes. In Proceedings of the 5th European Conference on Grain Legumes. Legumes for the Benefit of Agriculture, Nutrition and the Environment: Their Genomics, Their Products and Their Improvement, Dijon, France, 7–11 June 2004. [Google Scholar]
- Schoenlechner, R.; Drausinger, J.; Ottenschlaeger, V.; Jurackova, K.; Berghofer, E. Functional properties of gluten-free pasta produced from amaranth, quinoa and buckwheat. Plant Foods Hum. Nutr. 2010, 65, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Petitot, M.; Barron, C.; Morel, M.H.; Micard, V. Impact of legume flour addition on pasta structure: Consequences on its in vitro starch digestibility. Food Biophys. 2010, 5, 284–299. [Google Scholar] [CrossRef]
- Dexter, J.E.; Matsou, R.R. Changes in spaghetti protein solubility during cooking. Cereal Chem. 1979, 56, 394–398. [Google Scholar]
| Product | Fermentation Type | Main Results | Reference |
|---|---|---|---|
| Semolina pasta enriched in vitamin B2 | Fermentation of semolina (partial and total substitution) with a selected Lactobacillus plantarum strain at 42 and 60% w/w | Vitamin B2 synthesis. | [42] |
| Pasta with fermented semolina | Semolina fermentation by Saccharomyces cerevisiae and Lactobacillus alimentarius, addition to pasta formulation at 30% w/w | Low glycemic index. | [54] |
| Low cooking loss. | |||
| Semolina pasta with reduced gluten content | Semolina fermentation by a pool of four selected lactic acid bacteria (LAB) at 30% w/w | Gluten degradation (ca. 83%). | [50] |
| No changes in sensory attributes. | |||
| Pasta made with semolina rendered gluten free and pre-gelatinized rice flour | Semolina fermentation by a pool of selected LAB in presence of fungal proteases at 50% w/w | Gluten degradation. | [61] |
| Increase of the acidic perception. | |||
| Pasta made with parboiled brown rice flour and pre-gelatinized rice flour with sorghum sourdough | Spontaneous fermentation of sorghum, addition to pasta formulation at 15% w/w | Low cooking loss, optimal protein network. | [65] |
| Product | Fermentation Type | Main Results | Reference |
|---|---|---|---|
| Gluten free faba bean pasta | Faba bean flour fermented with selected starters at 100% w/w | Increase of protein and resistant starch content. | [52] |
| Low beany-flavor perception. | |||
| Pasta fortified with fermented faba bean | Fermentation of faba bean flour with selected starter, addition to pasta formulation at 10, 30, and 50% w/w | Increase of protein digestibility and protein quality indexes, reduction of glycemic index degradation of trypsin inhibitors, and condensed tannins. | [51] |
| Pasta fortified with fermented quinoa | Fermentation of quinoa flour with selected LAB, addition to pasta formulation at 20% w/w | Increase of peptides, free amino acids, and total phenol content. | [80] |
| Increased of all nutritional indexes and decrease of hydrolysis index. | |||
| Increase of cooking loss and decrease of water absorption | |||
| Pasta fortified with fermented pigeon pea | Spontaneous fermentation of pigeon pea flour, addition to pasta formulation at 10% w/w | Increase of protein quality indexes and chemical scores. | [70,81] |
| Sensory acceptability at substitution levels below 10%. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montemurro, M.; Coda, R.; Rizzello, C.G. Recent Advances in the Use of Sourdough Biotechnology in Pasta Making. Foods 2019, 8, 129. https://doi.org/10.3390/foods8040129
Montemurro M, Coda R, Rizzello CG. Recent Advances in the Use of Sourdough Biotechnology in Pasta Making. Foods. 2019; 8(4):129. https://doi.org/10.3390/foods8040129
Chicago/Turabian StyleMontemurro, Marco, Rossana Coda, and Carlo Giuseppe Rizzello. 2019. "Recent Advances in the Use of Sourdough Biotechnology in Pasta Making" Foods 8, no. 4: 129. https://doi.org/10.3390/foods8040129
APA StyleMontemurro, M., Coda, R., & Rizzello, C. G. (2019). Recent Advances in the Use of Sourdough Biotechnology in Pasta Making. Foods, 8(4), 129. https://doi.org/10.3390/foods8040129





