Physicochemical Properties and Pollen Profile of Oak Honeydew and Evergreen Oak Honeydew Honeys from Spain: A Comparative Study
Abstract
1. Introduction
2. Material and Methods
2.1. Honey Samples
2.2. Reagents and Standards
2.3. Melissopalynological Analysis
2.4. Physicochemical Analysis
2.5. Determination of The Total Polyphenol and Flavonoid Content
2.6. Radical Scavenging Activity
2.7. Sugar Composition
2.8. Statistical Analysis
3. Results and Discussion
3.1. Microscopic Analysis of The Sediment of Honeydew Honeys
3.1.1. Oak Honeydew Honeys
3.1.2. Evergreen Oak Honeydew Honeys
3.2. Physicochemical Profile of Honeydew Honeys
3.2.1. Oak Honeydew Honeys
3.2.2. Evergreen Oak Honeydew Honeys
3.3. Differentiation Between Both Honeydew Honeys
3.3.1. Differences in The Pollen Profile of The Two Groups of Honeydew Honeys
3.3.2. Differences in the Physicochemical Profile of the Two Groups of Honeydew Honeys
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodríguez-Flores, M.S.; Escuredo, O.; Seijo, M.C. Assessment of physicochemical and antioxidant characteristics of Quercus pyrenaica honeydew honeys. Food Chem. 2015, 166, 101–106. [Google Scholar] [CrossRef]
- Pita-Calvo, C.; Vázquez, M. Honeydew honeys: A Review on the characterization and authentication of botanical and geographical origins. J. Agric. Food Chem. 2018, 66, 2523–2537. [Google Scholar] [CrossRef]
- European Commission. Council directive 2001/110/EC of 20 December 2001 relating to honey. Off. J. Eur. Communities 2002, 10, 47–52. [Google Scholar]
- Bentabol-Manzanares, A.; Hernández-García, Z.; Rodríguez-Galdón, B.; Rodríguez-Rodríguez, E.; Díaz-Romero, C. Differentiation of blossom and honeydew honeys using multivariate analysis on the physicochemical parameters and sugar composition. Food Chem. 2011, 126, 664–672. [Google Scholar] [CrossRef]
- Escuredo, O.; Fernández-González, M.; Seijo, M.C. Differentiation of blossom honey and honeydew honey from Northwest Spain. Agriculture 2012, 2, 25–37. [Google Scholar]
- Escuredo, O.; Míguez, M.; Fernández-González, M.; Seijo, M.C. Nutritional value and antioxidant activity of honeys produced in a European Atlantic area. Food Chem. 2013, 138, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Simova, S.; Atanassov, A.; Shishiniova, M.; Bankova, A. A rapid differentiation between oak honeydew honey and nectar and other honeydew honeys by NMR spectroscopy. Food Chem. 2012, 134, 1706–1710. [Google Scholar] [CrossRef] [PubMed]
- Castiglioni, S.; Stefano, M.; Astolfi, P.; Carloni, P. Chemometric approach to the analysis of antioxidant properties and colour of typical Italian monofloral honeys. Int. J. Food Sci. Technol. 2017, 52, 1138–1146. [Google Scholar] [CrossRef]
- Kolayli, S.Z.C.; Çakir, H.E.; Okan, O.T.; Yildiz, O. An investigation on Trakya region Oak (Quercus spp.) honeys of Turkey: Their physico-chemical, antioxidant and phenolic compounds properties. Turk. J. Biochem. 2018, 43, 362–374. [Google Scholar] [CrossRef]
- Terrab, A.; Berjano, R.; Sanchez, J.A.; Gómez-Pajuelo, A.; Díez, M.J. Palynological and geographical characterisation of Spanish oak honeydew honeys. Grana 2019, 58, 63–77. [Google Scholar] [CrossRef]
- Castro-Vázquez, L.; Díaz-Maroto, M.C.; Pérez-Coello, M.S. Volatile composition and contribution to the aroma of spanish honeydew honeys. Identification of a new chemical marker. J. Agric. Food Chem. 2006, 54, 4809–4813. [Google Scholar] [CrossRef] [PubMed]
- Nayik, G.A.; Suhag, Y.; Majid, I.; Nanda, V. Discrimination of high altitude Indian honey by chemometric approach according to their antioxidant properties and macro minerals. J. Saudi Soc. Agric. Sci. 2018, 17, 200–207. [Google Scholar] [CrossRef]
- Bacandritsos, N.; Sabatini, A.G.; Papanastasiou, I.; Saitanis, C. Physico-chemical characteristics of greek fir honeydew honey from Marchalina hellenica (gen.) in comparison to other mediterranean honeydew honeys. Ital. J. Food Sci. 2006, 18, 21–31. [Google Scholar]
- De Miguel, S.; Pukkala, T.; Yeşil, A. Integrating pine honeydew honey production into forest management optimization. Eur. J. For. Res. 2014, 133, 423–432. [Google Scholar] [CrossRef]
- Rybak-Chmielewska, H.; Szczęsna, T.; Waś, E.; Jaśkiewicz, K.; Teper, D. Characteristics of Polish unifloral honeys IV. Honeydew honey, mainly Abies alba L. J. Apic. Sci. 2013, 57, 51–59. [Google Scholar] [CrossRef]
- Karabagias, I.K.; Vlasiou, M.; Kontakos, S.; Drouza, C.; Kontominas, M.G.; Keramidas, A.D. Geographical discrimination of pine and fir honeys using multivariate analyses of major and minor honey components identified by 1 H NMR and HPLC along with physicochemical data. Eur. Food Res. Technol. 2018, 244, 1249–1259. [Google Scholar] [CrossRef]
- Alía-Miranda, R.; García Del Barrio, J.M.; Iglesias-Sauce, S.; Mancha-Núñez, J.A.; De Miguel, J.; Nicolás-Peragón, J.L.; Pérez-Martín, F.; Sánchez, D. Regiones de Procedencia de Especies Forestales en España; Ed. Organismo Autónomo Parques Nacionales: Madrid, Spain, 2009. [Google Scholar]
- Sagata, K.; Gibb, H. The effect of temperature increases on an ant-hemiptera-plant interaction. PLoS ONE 2016, 11, e0155131. [Google Scholar] [CrossRef]
- Bertoncelj, J.; Polak, T.; Kropf, U.; Korošec, M.; Golob, T. LCDAD-ESI/MS analysis of flavonoids and abscisic acid with chemometric approach for the classification of Slovenian honey. Food Chem. 2011, 127, 296–302. [Google Scholar] [CrossRef]
- Persano-Oddo, L.; Piro, R. Main European unifloral honeys: Descriptive sheets. Apidologie 2004, 35, S38–S81. [Google Scholar] [CrossRef]
- Dobre, I.; Escuredo, O.; Rodríguez-Flores, M.S.; Seijo, M.C. Evaluation of several Romanian honeys based on their palynological and biochemical profiles. Int. J. Food Prop. 2014, 17, 1850–1860. [Google Scholar] [CrossRef][Green Version]
- Escuredo, O.; Dobre, I.; Fernández-González, M.; Seijo, M.C. Contribution of botanical origin and sugar composition of honeys on the crystallization phenomenon. Food Chem. 2014, 149, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Escuredo, O.; Fernández-González, M.; Rodríguez-Flores, M.S.; Seijo-Rodríguez, A.; Seijo, M.C. Influence of the botanical origin of honey from North western Spain in some antioxidant components. J. Apic. Sci. 2013, 57, 5–14. [Google Scholar] [CrossRef]
- Rodríguez-Flores, M.S.; Escuredo, O.; Seijo-Rodríguez, A.; Seijo, M.C. Characterization of the honey produced in heather communities (NW Spain). J. Apic. Res. 2019, 58, 84–91. [Google Scholar] [CrossRef]
- Bonta, V.; Marghitas, L.A.; Stanciu, O.; Laslo, L.; Dezmirean, D.; Bobis, O. High-performance liquid chromatographic analysis of sugars in Transylvanian honeydew honey. Bulletin of University of Agricultural Sciences and Veterinary Medicine Cluj-Napoca. Anim. Sci. Biotechnol. 2008, 65, 229–232. [Google Scholar]
- Piana, M.L.; Persano-Oddo, L.; Bentabol, A.; Bruneau, E.; Bogdanov, S.; Guyot Declerck, C. Sensory analysis applied to honey: State of the art. Apidologie 2004, 35, 26–37. [Google Scholar] [CrossRef]
- León-Ruiz, V.; Vera, S.; González-Porto, A.V.; San Andrés, M.P. Vitamin C and sugar levels as simple markers for discriminating Spanish honey sources. J. Food Sci. 2011, 76, C356–C361. [Google Scholar] [CrossRef]
- Perna, A.; Simonetti, A.; Intaglietta, I.; Sofo, A.; Gambacorta, E. Metal content of southern Italy honey of different botanical origins and its correlation with polyphenol content and antioxidant activity. Int. J. Food Sci. Technol. 2012, 47, 1909–1917. [Google Scholar] [CrossRef]
- Bilandžić, N.; Gajger, I.T.; Kosanović, M.; Čalopek, B.; Sedak, M.; Kolanović, B.S.; Varenina, I.; Luburić, D.B.; Varga, I.; Đokić, M. Essential and toxic element concentrations in monofloral honeys from southern Croatia. Food Chem. 2017, 234, 245–253. [Google Scholar] [CrossRef]
- Stanek, N.; Jasicka-Misiak, I. HPTLC phenolic profiles as useful tools for the authentication of honey. Food Anal. Methods 2018, 11, 2979–2989. [Google Scholar] [CrossRef]
- Dimou, M.; Katsaros, J.; Tzavella, K.; Thrasyvoulou, A. Discriminating pine and fir honeydew honeys by microscopic characteristics. J. Apic. Res. 2006, 45, 16–21. [Google Scholar] [CrossRef]
- Seijo, M.C.; Escuredo, O.; Fernández-González, M. Fungal diversity in honeys from northwest Spain and their relationship to the ecological origin of the product. Grana 2011, 50, 55–62. [Google Scholar] [CrossRef]
- Magyar, D.; Gönczöl, J.; Révay, Á.; Grillenzoni, F.; Seijo, M.C. Stauro- and scolecoconidia in floral and honeydew honeys. Fungal Divers. 2005, 20, 103–120. [Google Scholar]
- Magyar, D.; Mura-Mészáros, A.; Grillenzoni, F. Fungal diversity in floral and honeydew honeys. Acta Bot. Hung. 2016, 58, 145–166. [Google Scholar] [CrossRef]
- Mura-Mészáros, A.; Magyar, D. Fungal honeydew elements as potential indicators of the botanical and geographical origin of honeys. Food Anal. Methods 2017, 10, 3079–3087. [Google Scholar] [CrossRef]
- Louveaux, J.; Maurizio, A.; Vorwohl, G. Methods of melissopalynology. Bee World 1978, 59, 139–157. [Google Scholar] [CrossRef]
- Bogdanov, S.; Martin, P.; Lullmann, C. Harmonized methods of the international honey commission. Apidologie 1997, extra issue. 1–59. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Arvouet-Grand, A.; Vennat, B.; Pourrat, A.; Legret, P. Standardization of propolis extract and identification of principal constituents. J. Pharm. Belg. 1994, 49, 462–468. [Google Scholar]
- Brand-Williams, W.; Culivier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Atanassova, J.; Lazarova, M.; Yurukova, L. Significant parameters of Bulgarian honeydew honey. J. Cent. Eur. Agric. 2016, 17, 640–651. [Google Scholar] [CrossRef][Green Version]
- Flanjak, I.; Kenjerić, D.; Bubalo, D.; Primorac, L. Characterisation of selected Croatian honey types based on the combination of antioxidant capacity, quality parameters, and chemometrics. Eur. Food Res. Technol. 2016, 242, 467–475. [Google Scholar] [CrossRef]
- Vasić, V.; Gašić, U.; Stanković, D.; Lušić, D.; Vukić-Lušić, D.; Milojković-Opsenica, D.; Tešića, Ž.; Trifković, J. Towards better quality criteria of European honeydew honey: Phenolic profile and antioxidant capacity. Food Chem. 2019, 274, 629–641. [Google Scholar] [CrossRef]
- Muñoz, O.; Copaja, S.; Speisky, H.; Peña, R.C.; Montenegro, G. Contenido de flavonoides y compuestos fenólicos de mieles chilenas e índice antioxidante. Quim. Nova 2007, 30, 848–851. [Google Scholar] [CrossRef]
- Vela, L.; de Lorenzo, C.; Perez, R.A. Antioxidant capacity of Spanish honeys and its correlation with polyphenol content and other physicochemical properties. J. Sci. Food Agric. 2007, 87, 1069–1075. [Google Scholar] [CrossRef]
- Seraglio, S.K.T.; Silva, B.; Bergamo, G.; Brugnerotto, P.; Gonzaga, L.V.; Fett, R.; Oliveira, A.C. An overview of physicochemical characteristics and health-promoting properties of honeydew honey. Food Res. Int. 2019, 119, 44–66. [Google Scholar] [CrossRef]
- Manikis, I.; Thrasyvoulou, A. The relation of physicochemical characteristics of honey and the crystallization sensitive parameters. Apiacta 2001, 36, 106–112. [Google Scholar]
- Pita-Calvo, C.; Vázquez, M. Differences between honeydew and blossom honeys: A review. Trends Food Sci. Technol. 2017, 59, 79–87. [Google Scholar] [CrossRef]
- Dobre, I.; Georgescu, L.A.; Alexe, P.; Escuredo, O.; Seijo, M.C. Rheological behavior of different honey types from Romania. Food Res. Int. 2012, 49, 126–132. [Google Scholar] [CrossRef]
- Pascual-Maté, A.; Osés, S.M.; Marcazzan, G.L.; Gardini, S.; Fernández Muiño, M.A.; Sancho, M.T. Sugar composition and sugar-related parameters of honeys from the northern Iberian Plateau. J. Food Comp. Anal. 2018, 74, 34–43. [Google Scholar] [CrossRef]
- Primorac, L.; Flanjak, I.; Kenjeric, D.; Bubalo, D.; Topolnjak, Z. Specific rotation and carbohydrate profile of Croatian unifloral honeys. Czech J. Food Sci. 2011, 29, 515–519. [Google Scholar] [CrossRef]
- Primorac, L.J.; Angelkov, B.; Mandić, M.; Kenjerić, D.; Nedeljko, M.; Flanjak, I.; Pirički, A.P.; Arapceska, M. Comparison of the Croatian and Macedonian honeydew honey. J. Cent. Eur. Agric. 2009, 10, 263–270. [Google Scholar]
- Ouchemoukh, S.; Schweitzer, P.; Bachir-Bey, M.; Djoudad-Kadji, H.; Louaileche, H. HPLC sugar profiles of Algerian honeys. Food Chem. 2010, 121, 561–568. [Google Scholar] [CrossRef]
- Weston, R.J.; Brocklebank, L.K. The oligosaccharide composition of some New Zealand honeys. Food Chem. 1999, 64, 33–37. [Google Scholar] [CrossRef]

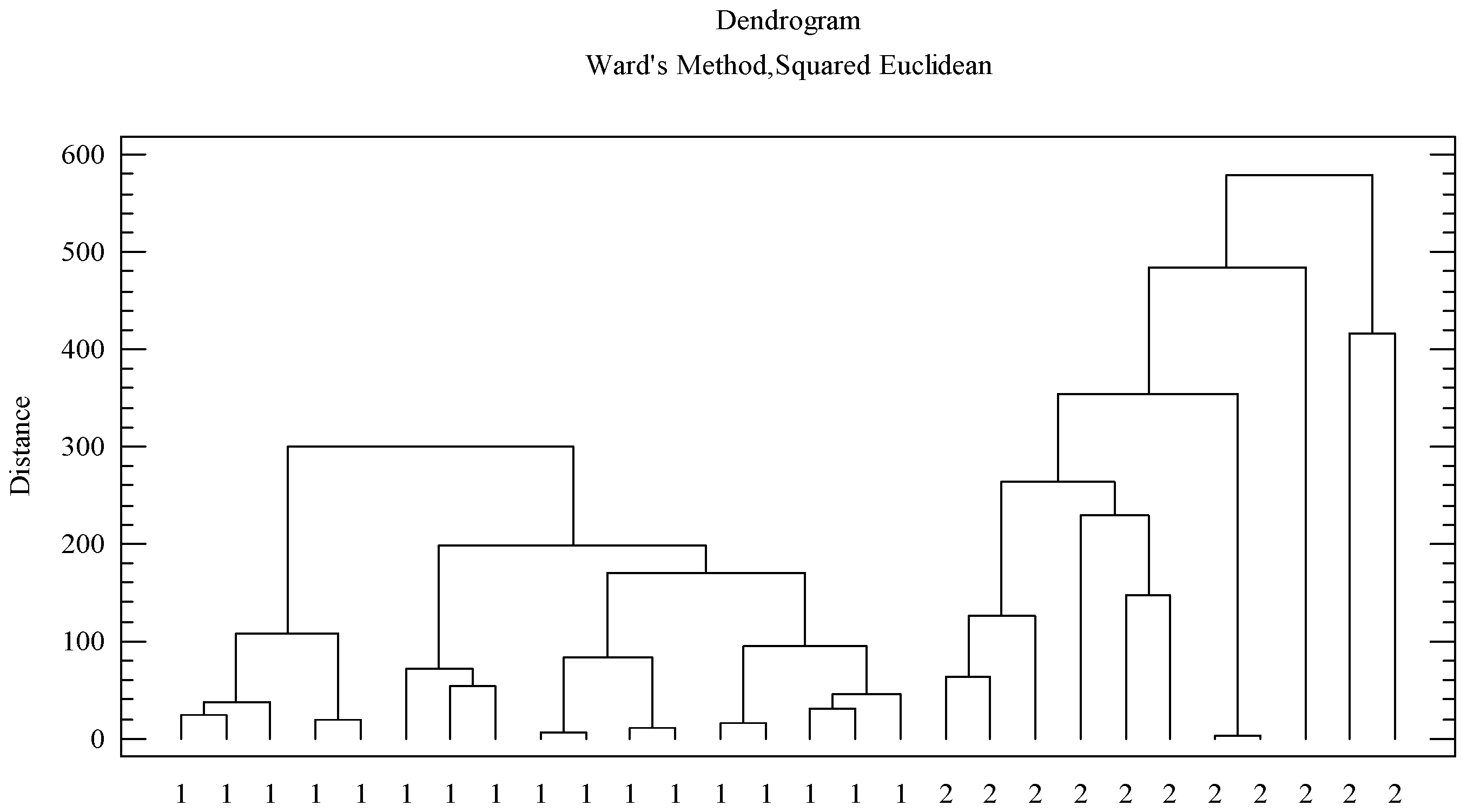
| Family | Pollen type | % | P | R | I | A | D |
|---|---|---|---|---|---|---|---|
| Ericaceae | Erica ** | 100.0 | 5.9 | 47.1 | 41.2 | 5.9 | - |
| Fabaceae | Cytisus type | 100.0 | - | 17.6 | 82.4 | - | - |
| Fagaceae | Castanea sativa | 100.0 | - | - | - | 64.7 | 35.3 |
| Rosaceae | Rubus | 100.0 | - | - | - | 58.8 | 41.2 |
| Fagaceae | Quercus | 82.4 | 76.5 | 5.9 | - | - | - |
| Boraginaceae | Echium * | 76.5 | 52.9 | 11.8 | 11.8 | - | - |
| Caprifoliaceae | Frangula alnus * | 64.7 | 47.1 | 5.9 | 5.9 | 5.9 | - |
| Rosaceae | Crataegus monogyna type * | 64.7 | 41.2 | 17.6 | 5.9 | - | - |
| Salicaceae | Salix | 64.7 | 64.7 | - | - | - | - |
| Fabaceae | Trifolium type | 58.8 | 52.9 | 5.9 | - | - | - |
| Myrtaceae | Eucalyptus | 58.8 | 23.5 | 29.4 | 5.9 | - | - |
| Plantaginaceae | Plantago | 52.9 | 52.9 | - | - | - | - |
| Brassicaceae | Brassica type | 47.1 | 47.1 | - | - | - | - |
| Campanulaceae | Campanula type | 41.2 | 41.2 | - | - | - | - |
| Cistaceae | Cistus psilosepalus | 41.2 | 35.3 | 5.9 | - | - | - |
| Poaceae | Poaceae * | 41.2 | 41.2 | - | - | - | - |
| Poaceae | Zea mays | 35.3 | 35.3 | - | - | - | - |
| Resedaceae | Sesamoides | 35.3 | 23.5 | 5.9 | - | 5.9 | - |
| Rosaceae | Prunus type | 35.3 | 35.3 | - | - | - | - |
| Umbelliferae | Conium maculatum type | 35.3 | 35.3 | - | - | - | - |
| Labiatae | Lavandula stoechas ** | 29.4 | 29.4 | - | - | - | - |
| Compositae | Centaurea | 23.5 | 23.5 | - | - | - | - |
| Fabaceae | Lotus type | 23.5 | 23.5 | - | - | - | - |
| 1 Pollen grain/g: 18451 ± 12895 | |||||||
| Family | Pollen type | % | P | R | I | A | D |
|---|---|---|---|---|---|---|---|
| Boraginaceae | Echium ** | 100.0 | 9.1 | 18.2 | 27.3 | 27.3 | 18.2 |
| Fagaceae | Quercus * | 100.0 | 45.5 | 27.3 | 27.3 | - | - |
| Brassicaceae | Brassica type * | 90.9 | 36.4 | 45.5 | 9.1 | - | - |
| Cistaceae | Cistus ladanifer type * | 90.9 | 54.5 | 27.3 | 9.1 | - | - |
| Fabaceae | Cytisus type ** | 90.9 | 27.3 | 36.4 | 27.3 | - | - |
| Labiatae | Lavandula stoechas type ** | 90.9 | 63.6 | 27.3 | - | - | - |
| Rosaceae | Rubus ** | 90.9 | 9.1 | - | 36.4 | 36.4 | 9.1 |
| Campanulaceae | Campanula type * | 81.8 | 36.4 | 27.3 | 18.2 | - | - |
| Compositae | Taraxacum oficcinale * | 81.8 | 72.7 | 9.1 | - | - | - |
| Fabaceae | Trifolium type | 81.8 | 54.5 | 18.2 | 9.1 | - | - |
| Fagaceae | Castanea sativa | 81.8 | 9.1 | - | 18.2 | 27.3 | 27.3 |
| Myrtaceae | Eucalyptus | 81.8 | 45.5 | 27.3 | 9.1 | - | - |
| Rosaceae | Prunus type | 72.7 | 45.5 | 18.2 | 9.1 | - | - |
| Umbelliferae | Conium maculatum type | 72.7 | 63.6 | 9.1 | - | - | - |
| Compositae | Carduus type * | 63.6 | 63.6 | - | - | - | - |
| Ericaceae | Erica * | 63.6 | 18.2 | 45.5 | - | - | - |
| Poaceae | Poaceae ** | 63.6 | 63.6 | - | - | - | - |
| Salicaceae | Salix | 63.6 | 63.6 | - | - | - | - |
| Oleaceae | Olea europaea * | 54.5 | 27.3 | 27.3 | - | - | - |
| Plantaginaceae | Plantago | 45.5 | 27.3 | 18.2 | - | - | - |
| Brassicaceae | Diplotaxis | 36.4 | 27.3 | 9.1 | - | - | - |
| Chenopodiaceae | Chenopodium | 36.4 | 36.4 | - | - | - | - |
| Cistaceae | Cistus salviifolius | 36.4 | 36.4 | - | - | - | - |
| Cistaceae | Halimium type | 36.4 | 36.4 | - | - | - | - |
| Compositae | Anthemis | 36.4 | 27.3 | - | 9.1 | - | - |
| Compositae | Helianthus annuus type * | 36.4 | 27.3 | 9.1 | - | - | - |
| Fabaceae | Galega officinalis * | 36.4 | - | - | 36.4 | - | - |
| Fabaceae | Vicia type | 36.4 | 36.4 | - | - | - | - |
| Labiatae | Mentha type | 36.4 | 36.4 | - | - | - | - |
| Fabaceae | Anthyllis cytisoides | 27.3 | 9.1 | 9.1 | - | - | 9.1 |
| Fabaceae | Ceratonia | 27.3 | 18.2 | 9.1 | - | - | - |
| Fabaceae | Onobrychis | 27.3 | 18.2 | 9.1 | - | - | - |
| Lythraceae | Lythrum | 27.3 | 27.3 | - | - | - | - |
| Papaveraceae | Hypecoum | 27.3 | 18.2 | - | 9.1 | - | - |
| Scrophulariaceae | Scrophularia type | 27.3 | 27.3 | - | - | - | - |
| Caprifoliaceae | Frangula alnus | 18.2 | 9.1 | 9.1 | - | - | - |
| 1 Pollen grain/g: 38519 ± 41187 | |||||||
| Mean ± SD | ||
|---|---|---|
| Oak Honeydew | Evergreen Oak Honeydew | |
| Physicochemical parameters | ||
| Moisture (%) | 17.4 ± 1.0 | 16.7 ± 1.0 |
| EC (mS/cm) | 1.0 ± 0.1 | 1.1 ± 0.2 |
| pH | 4.4 ± 0.2 | 4.4 ± 0.2 |
| HMF (mg/100 g) | 0.1 ± 0.1 | 0.3 ± 0.6 |
| DI | 24.6 ± 4.2 | 27.3 ± 8.3 |
| Colour (mm Pfund) | 142 ± 11 ** | 120 ± 19 ** |
| L | 52.1 ± 3.4 | 53.8 ± 3.4 |
| a * | 7.2 ± 1.5 ** | 13.9 ± 1.6 ** |
| b * | 4.8 ± 1.9 ** | 14.7 ± 4.8** |
| Antioxidant properties | ||
| Phenols (mg/100 g) | 134.8 ± 26.7 * | 111.3 ± 26.0 * |
| Flavonoids (mg/100 g) | 9.7 ± 1.8 ** | 7.5 ± 1.3 ** |
| RSA (%) | 72.4 ± 6.84 ** | 63.3 ± 8.9 ** |
| Sugar composition (g/100 g) | ||
| Fructose | 35.4 ± 1.8 * | 32.0 ± 3.9 * |
| Glucose | 27.2 ±1.3 ** | 24.1 ±1.7 ** |
| Turanose | 3.1 ± 0.8 | 2.4 ± 1.1 |
| Maltose | 0.7 ± 0.4 * | 1.2 ± 1.0 * |
| Sucrose | 0.2 ± 0.1 | 0.2 ± 0.1 |
| Melezitose | 0.1 ± 0.1 | 0.9 ± 2.2 |
| Trehalose | nd | 0.1 ± 0.1 |
| Fructose + Glucose | 62.5 ± 2.4 ** | 56.2 ± 4.8 ** |
| Ratio sugar/water content | ||
| F+G/W | 3.6 ± 0.2 | 3.4 ± 0.4 |
| F/W | 2.0 ± 0.2 | 1.9 ± 0.3 |
| G/W | 1.6 ± 0.1 * | 1.4 ± 0.1 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seijo, M.C.; Escuredo, O.; Rodríguez-Flores, M.S. Physicochemical Properties and Pollen Profile of Oak Honeydew and Evergreen Oak Honeydew Honeys from Spain: A Comparative Study. Foods 2019, 8, 126. https://doi.org/10.3390/foods8040126
Seijo MC, Escuredo O, Rodríguez-Flores MS. Physicochemical Properties and Pollen Profile of Oak Honeydew and Evergreen Oak Honeydew Honeys from Spain: A Comparative Study. Foods. 2019; 8(4):126. https://doi.org/10.3390/foods8040126
Chicago/Turabian StyleSeijo, María Carmen, Olga Escuredo, and María Shantal Rodríguez-Flores. 2019. "Physicochemical Properties and Pollen Profile of Oak Honeydew and Evergreen Oak Honeydew Honeys from Spain: A Comparative Study" Foods 8, no. 4: 126. https://doi.org/10.3390/foods8040126
APA StyleSeijo, M. C., Escuredo, O., & Rodríguez-Flores, M. S. (2019). Physicochemical Properties and Pollen Profile of Oak Honeydew and Evergreen Oak Honeydew Honeys from Spain: A Comparative Study. Foods, 8(4), 126. https://doi.org/10.3390/foods8040126







