Glyceridic and Unsaponifiable Components of Microencapsulated Sacha Inchi (Plukenetia huayllabambana L. and Plukenetia volubilis L.) Edible Oils
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Solvents and Reactants
2.3. Preparation of Sacha Inchi Oil Microparticles by Spray-Drying
2.4. Extraction of the Surface and Total Oil
2.5. Analytical Determinations
2.5.1. Fatty Acid Composition Analysis
2.5.2. Minor Glyceride Polar Compounds Analysis
2.5.3. Tocopherol Analysis
2.5.4. Sterol Composition Analysis
2.6. Data Analysis
3. Results and Discussion
3.1. Fatty Acid Composition
3.2. Minor Glyceride Polar Compound Composition
3.3. Tocopherol Composition
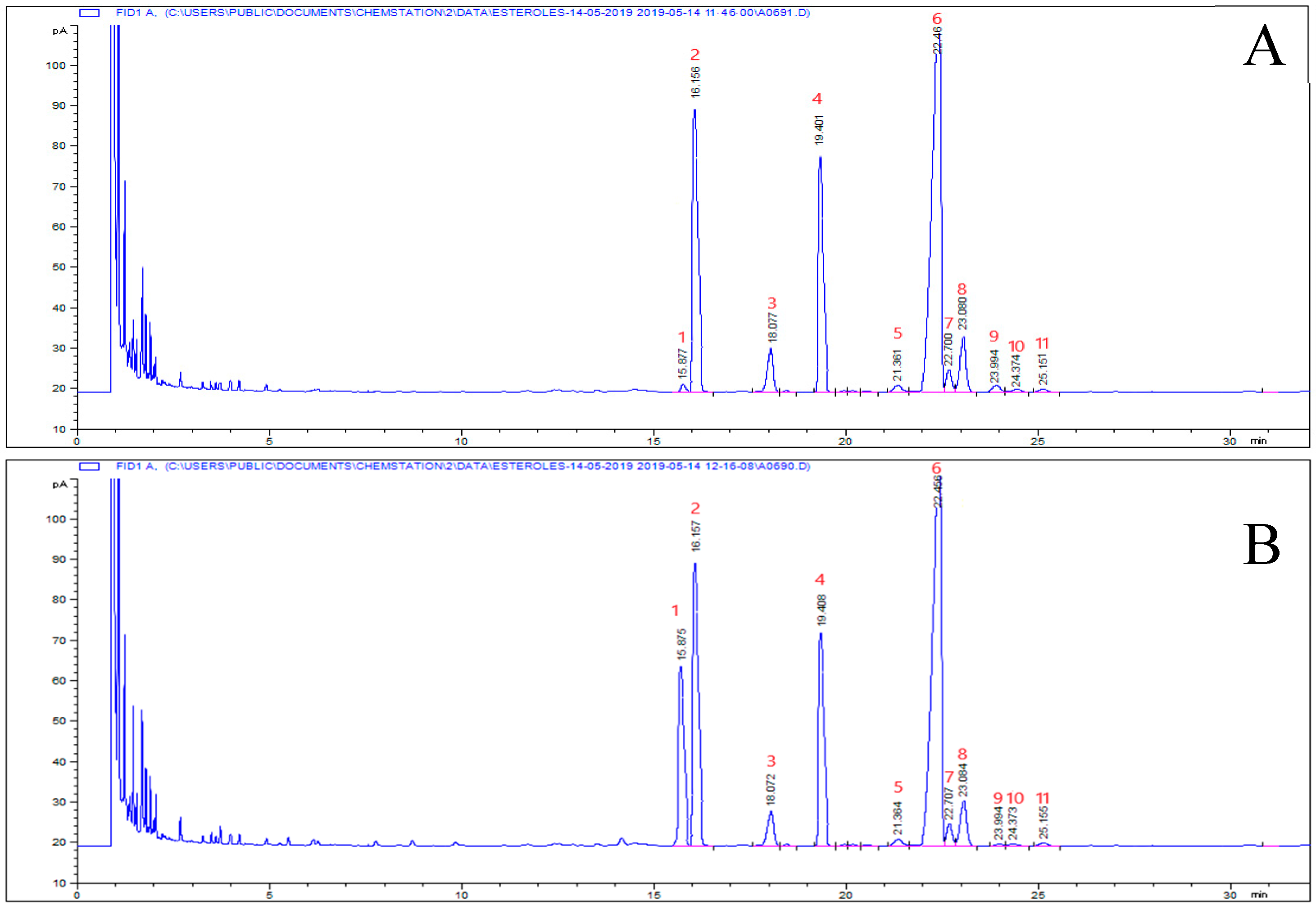
3.4. Sterol Composition
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Champagne, C.P.; Fustier, P. Microencapsulation for the improved delivery of bioactive compounds into foods. Curr. Opin. Biotech. 2007, 18, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, P.; Dowling, K.; Barrow, C.J.; Adhikari, B. Microencapsulation of omega-3 fatty acids: A review of microencapsulation and characterization methods. J. Funct. Food 2015, 19, 868–881. [Google Scholar] [CrossRef]
- Martínez, M.L.; Curti, M.I.; Roccia, P.; Llabot, J.M.; Penci, M.C.; Bodoira, R.M.; Ribotta, P.D. Oxidative stability of walnut (Juglans regia L.) and chia (Salvia hispanica L.) oils microencapsulated by spray drying. Powder Technol. 2015, 270, 271–277. [Google Scholar] [CrossRef]
- Carneiro, H.C.F.; Tonon, R.V.; Grosso, C.R.F.; Hubinger, M.D. Encapsulation efficiency and oxidative stability of flaxseed oil microencapsulated by spray drying using different combinations of wall materials. J. Food Eng. 2013, 115, 443–451. [Google Scholar] [CrossRef]
- Gallardo, G.; Guida, L.; Martínez, V.; López, M.C.; Bernhardt, D.; Blasco, R. Microencapsulation of linseed oil by spray drying for functional food application. Food Res. Int. 2013, 52, 473–482. [Google Scholar] [CrossRef]
- Comunian, T.A.; Favaro-Trindade, C.S. Microencapsulation using biopolymers as an alternative to produce food enhanced with phytosterols and omega-3 fatty acids: A review. Food Hydrocol. 2016, 61, 442–457. [Google Scholar] [CrossRef]
- Alcântara, M.A.; de Lima, A.E.A.; Mattos Braga, A.L.; Valeriano Tonon, R.; Galdeano, M.C.; da Costa Mattos, M.; Santa Brígida, A.I.; Rosenhaim, R.; Albuquerque dos Santos, N.; Tribuzy de Magalhães Cordeiro, A.M. Influence of the emulsion homogenization method on the stability of chia oil microencapsulated by spray drying. Powder Technol. 2019, 354, 877–885. [Google Scholar] [CrossRef]
- Maeda Rojas, V.; da Costa, L.F.; Guimarães-Inácio, A.; Leimann, F.V.; Tanamati, A.; Gozzo, Â.M.; Hernandez, R.; Barreiro, M.F.; Barros, L.; Ferreira, I.; et al. Formulation of mayonnaises containing PUFAs by the addition of microencapsulated chia seeds, pumpkin seeds and baru oils. Food Chem. 2019, 274, 220–227. [Google Scholar] [CrossRef]
- Cortés-Camargo, S.; Acuña-Avila, P.E.; Rodríguez-Huezo, M.E.; Román-Guerrero, A.; Varela-Guerrero, V.; Pérez-Alonso, C. Effect of chia mucilage addition on oxidation and release kinetics of lemon essential oil microencapsulated using mesquite gum—Chia mucilage mixtures. Food Res. Int. 2019, 116, 1010–1019. [Google Scholar] [CrossRef]
- Lavanya, M.N.; Kathiravan, T.; Moses, J.A.; Anandharamakrishnan, C. Influence of spray-drying conditions on microencapsulation of fish oil and chia oil. Drying Technol. 2019. [Google Scholar] [CrossRef]
- Timilsena, Y.P.; Vongsvivut, J.; Tobin, M.J.; Adhikari, R. Investigation of oil distribution in spray-dried chia seed oil microcapsules using synchrotron-FTIR microspectroscopy. Food Chem. 2019, 275, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, E.S.; Julio, L.M.; Henning, C.; Diehl, B.W.K.; Tomás, M.C.; Ixtaina, V.Y. Effect of natural antioxidants on the physicochemical properties and stability of freeze-dried microencapsulated chia seed oil. J. Sci. Food Agric. 2019, 99, 1682–1690. [Google Scholar] [CrossRef] [PubMed]
- Chirinos, R.; Pedreschi, R.; Domínguez, G.; Campos, D. Comparison of the physico-chemical and phytochemical characteristics of the oil of two Plukenetia species. Food Chem. 2015, 173, 1203–1206. [Google Scholar] [CrossRef] [PubMed]
- Chasquibol, N.A.; del Aguila, C.; Yácono, J.C.; Guinda, A.; Moreda, W.; Gómez-Coca, R.B.; Pérez-Camino, M.C. Characterization of glyceridic and unsaponifiable compounds of sacha inchi (Plukenetia huayllabambana L.) oils. J. Agric. Food Chem. 2014, 62, 10162–11016. [Google Scholar] [CrossRef]
- Triana-Maldonado, D.M.; Torijano-Gutiérrez, S.A.; Giraldo-Estrada, C. Supercritical CO2 extraction of oil and omega-3 concentrate from sacha inchi (Plukenetia volubilis L.) from Antioquia, Colombia. Grasas Aceites 2017, 68, e172. [Google Scholar] [CrossRef]
- Wang, S.; Zhub, F.; Kakuda, Y. Sacha inchi (Plukenetia volubilis L.): Nutritional composition, biologicalactivity, and uses. Food Chem. 2018, 265, 316–328. [Google Scholar] [CrossRef]
- Quispe-Condori, S.; Saldaña, M.D.A.; Temelli, F. Microencapsulation of flax oil with zein using spray and freeze drying. LWT Food Sci. Technol. 2011, 44, 1880–1887. [Google Scholar] [CrossRef]
- Vicente, J.; de Souza Cezarino, T.; Barreto Pereira, L.J.; Pinto da Rocha, E.; Raymundo Sá, G.; Domingues Gamallo, O.; de Carvalho, M.G.; García-Rojas, E.E. Microencapsulation of sacha inchi oil using emulsion-based delivery systems. Food Res. Int. 2017, 99, 612–622. [Google Scholar] [CrossRef]
- Sánchez-Reinoso, Z.; Gutiérrez, L.F. Effects of the emulsion composition on the physical properties and oxidative stability of sacha inchi (Plukenetia volubilis L.) oil microcapsules produced by spray drying. Food Bioproc. Technol. 2017, 10, 1354–1366. [Google Scholar] [CrossRef]
- Pastuña-Pullutasig, A.; López-Hernández, O.; Debut, A.; Vaca, A.; Rodríguez-Leyes, E.; Vicente, R.; Gonzalez, V.; González-Sanabia, M.; Tapia-Hernández, F. Microencapsulation of oil sacha inchi (Plukenetia volubilis L.) by spray drying. Rev. Colomb. Cienc. Quimic. Farmac. 2016, 45, 422–437. [Google Scholar] [CrossRef]
- Landoni, L.; Vilca Calderón, L.; Chasquibol, N.; Pérez-Camino, M.C.; Gallardo, G. Physicochemical characterization and oxidative stability of microencapsulated sacha inchi seed oils by spray drying. Grasas Aceites 2020, 71, e387, in press. [Google Scholar]
- Oxley, J.D. Spray cooling and spray chilling for food ingredient and nutraceutical encapsulation. In Encapsulation Technologies and Delivery Systems for Food Ingredients and Nutraceuticals; Garti, N., Mc Clements, D.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 110–130. [Google Scholar]
- Fadini, A.L.; Dutra Alvima, I.; Porto Ribeiro, I.; Geraldini Ruzene, L.; Bataglia da Silva, L.; Bonifácio Queiroz, M.; Rauen de Oliveira, A.M.; Maia Chaves, F.C.; Ferreira Rodrigues, R.A. Innovative strategy based on combined microencapsulation technologies for food application and the influence of wall material composition. LWT-Food Sci. Technol. 2018, 91, 345–352. [Google Scholar] [CrossRef]
- Da Silva Soares, B.; Pinto Siqueira, R.; de Carvalho, M.G.; Juarez, V.; Garcia-Rojas, E.E. Microencapsulation of sacha inchi oil (Plukenetia volubilis L.) using complex coacervation: Formation and structural characterization. Food Chem. 2019, 298, 125045. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.K.; Jessie, L.Y.L.; Tan, C.P.; Long, K.; Nyam, K.L. Effect of accelerated storage on microencapsulated Kenaf seed oil. J. Am. Oil Chem. Soc. 2013, 90, 1023–1029. [Google Scholar] [CrossRef]
- Encina, C.; Márquez-Ruiz, G.; Holgado, F.; Giménez, B.; Vergara, C.; Robert, P. Effect of spray-drying with organic solvents on the encapsulation, release and stability of fish oil. Food Chem. 2018, 263, 283–291. [Google Scholar] [CrossRef]
- Holgado, F.; García-Martínez, M.C.; Velasco, J.; Ruiz-Méndez, M.V.; Márquez-Ruiz, G. Microencapsulation of conjugated linoleic acid (CLA)-rich oil with skimmed milk components protects against polymerization. J. Am. Oil Chem. Soc. 2018, 95, 1399–1408. [Google Scholar] [CrossRef]
- Velasco, J.; Dobarganes, M.C.; Márquez, G. Oxidation of free and encapsulated oil fractions in dried microencapsulated fish oil. Grasas Aceites 2000, 51, 439–446. [Google Scholar]
- Márquez-Ruiz, G.; Velasco, J.; Dobarganes, C. Evaluation of oxidation in dried microencapsulated fish oils by a combination of adsorption and size exclusion chromatography. Eur. Food Res. Technol. 2000, 211, 13–18. [Google Scholar] [CrossRef]
- Sankarikutty, B.M.; Sreekumar, M.; Nrayanan, C.S.; Mathew, A.G. Studies on microcapsulation of cardamon oil by spray drying technique. J. Food Sci. Technol. 1988, 26, 352–356. [Google Scholar]
- Richardson, G.H. Standard Methods for the Examination of Dairy Products, 15th ed.; American Public Health Association: Washington, DC, USA, 1985; p. 358. [Google Scholar]
- IUPAC Standard Method 2.301. Standard methods for the analysis of oils, fats and derivatives. In Preparation of Fatty Acid Methyl Ester; Blackwell Scientific: Oxford, UK, 1987. [Google Scholar]
- IUPAC Standard Method 2.432. Standard methods for the analysis of oils, fats and derivatives. In Determination of Tocopherol and Tocotrienols in Vegetable Oils and Fats by HPLC; Blackwell Scientific: Oxford, UK, 1987. [Google Scholar]
- García-González, A.; Velasco, J.; Velasco, L.; Ruiz-Méndez, M.V. An analytical simplification for faster determination of fatty acid composition and phytosterols in seed oils. Food Anal. Method. 2018, 11, 1234–1242. [Google Scholar] [CrossRef]
- NTP. Norma Tećnica Peruana 151.400, Amendment. NTP 151.400, 2009; Requisitos Aceite Sacha Inchi, INDECOPI: Lima, Peru, 2014. [Google Scholar]
- Aquilani, C.; Pérez-Palacios, T.; Sirtori, F.; Jiménez-Martín, E.; Antequera, T.; Franci, O.; Bozzi, R.; Acciaioli, A.; Pugliese, C. Enrichment of Cinta Senese burger with omega-3 fatty acids. Effect of type of addition and storage condition on quality characteristics. Grasas Aceites 2018, 69, e235. [Google Scholar] [CrossRef]
- Ilyasoglu, H. Production of structured lipid with a low omega-6/omega-3 fatty acids ratio by enzymatic interesterification. Grasas Aceites 2017, 68, e191. [Google Scholar] [CrossRef]
- Liu, W.; Lu, G.H. Cis-trans isomerization of unsaturated fatty acids in edible oils to prepare trans fat. Grasas Aceites 2018, 69, e268. [Google Scholar] [CrossRef]
- Jiménez, M.; Garcia, H.S.; Beristain, C.I. Spray-drying microencapsulation and oxidative stability of conjugated linoleic acid. Europ. Food Res. Technol. 2004, 219, 588–592. [Google Scholar] [CrossRef]
- Costa, A.M.M.; Nunes, J.C.; Lima, B.N.B.; Pedrosa, C.; Calado, V.; Torres, A.G.; Pierucci, A.P.R. Effective stabilization of CLA by microencapsulation in pea protein. Food Chem. 2015, 168, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Dobarganes, M.C.; Pérez-Camino, M.C.; Márquez-Ruiz, G. High Performance Size Exclusion Chromatography of Polar Compounds in Heated and Non-Heated Fats. Fatt Sci. Technol. 1988, 90, 308–311. [Google Scholar] [CrossRef]
- Velasco, J.; Marmesat, S.; Dobarganes, C.; Márquez-Ruiz, G. Heterogeneous Aspects of Lipid Oxidation in Dried Microencapsulated Oils. J. Agric. Food Chem. 2006, 54, 1722–1729. [Google Scholar] [CrossRef]
- Tomassi, G.; Simano, V. An assessment of the safety of tocopherols as food additives. Food Chem. Toxicol. 1986, 24, 1051–1061. [Google Scholar] [CrossRef]
- Chasquibol, N.A.; Gómez-Coca, R.B.; Yácono, J.C.; Guinda, A.; Moreda, W.; del Aguila, C.; Pérez-Camino, M.C. Markers of quality and genuineness of commercial extra virgin sacha inchi oils. Grasas Aceites 2016, 67, e169. [Google Scholar] [CrossRef]
- Bruscatto, M.H.; Pestana-Bauer, V.R.; Otero, D.M.; Zambiazi, R.C. Effects of heating temperature on the tocopherol contents of chemically and physically refined rice bran oil. Grasas Aceites 2019, 70, e294. [Google Scholar] [CrossRef]
- Deng, R. Food and Food Supplements with Hypocholesterolemic Effects. Recent Patents on Food, Nutrit. Agric. 2009, 1, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, F.; Poli, A. Phytosterols and cardiovascular health. Pharmacol. Res. 2010, 61, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Alvim, I.D.; de Souza, F.; Koury, I.P.; Jurt, T.; Dantas, F.B.H. Use of the spray chilling method to deliver hydrophobic components: Physical characterization of microparticles. Cienc. Tecnol. Aliment. 2013, 33, 34–39. [Google Scholar] [CrossRef]
- Di Battista, C.A.; Constenla, D.; Ramírez-Rigo, M.V.; Piña, J. The use of arabic gum, maltodextrin and surfactants in the microencapsulation of phytosterols by spray drying. Powder Technol. 2015, 286, 193–201. [Google Scholar] [CrossRef]
- Hue, W.L.; Nyam, K.L. Physiochemical properties of kenaf seed oil microcapsules before and after freeze drying and its storage stability. Int. Food Res. J. 2018, 25, 1502–1509. [Google Scholar]
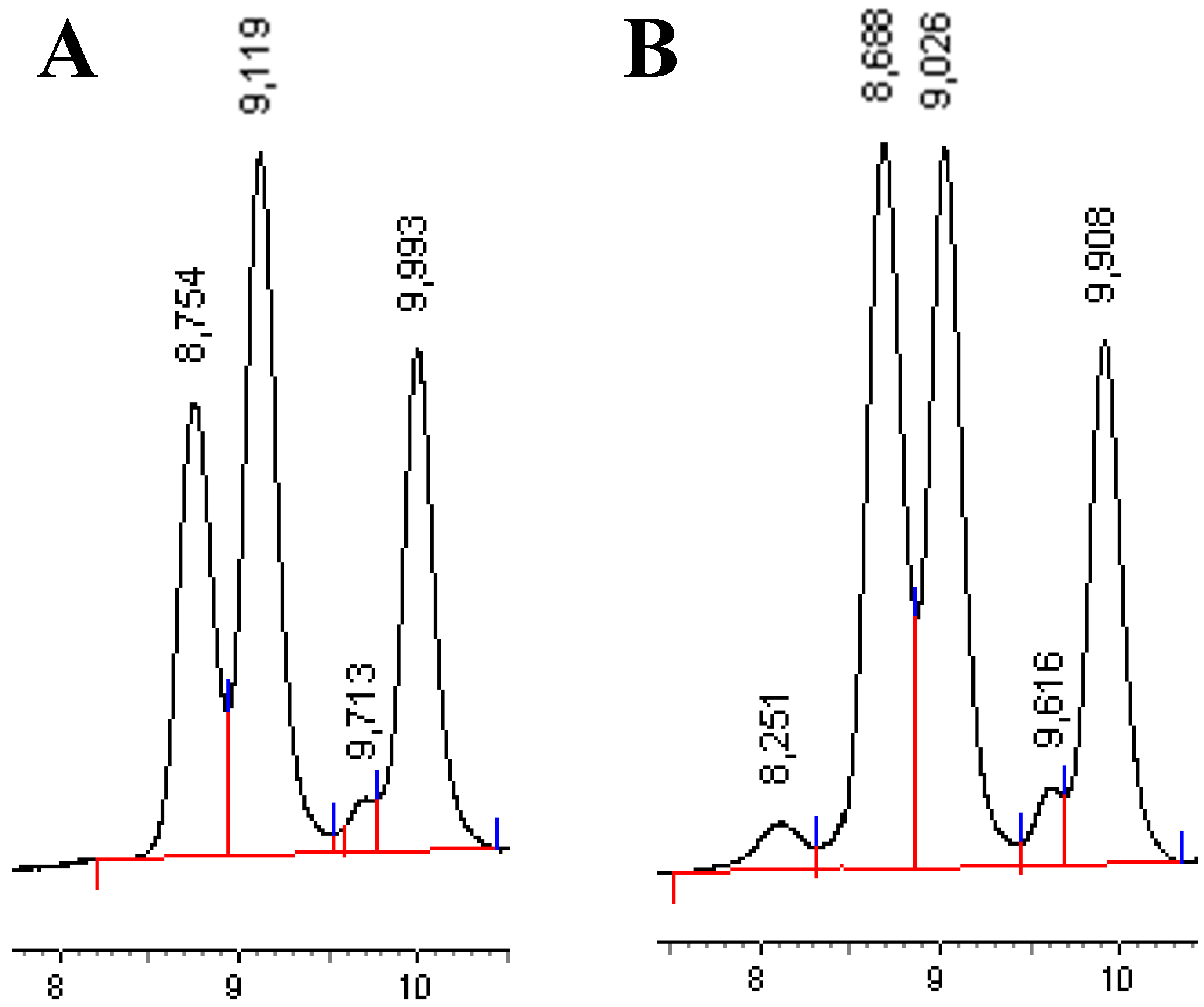
 Total oil and
Total oil and  Surface oil.
Surface oil.
 Total oil and
Total oil and  Surface oil.
Surface oil.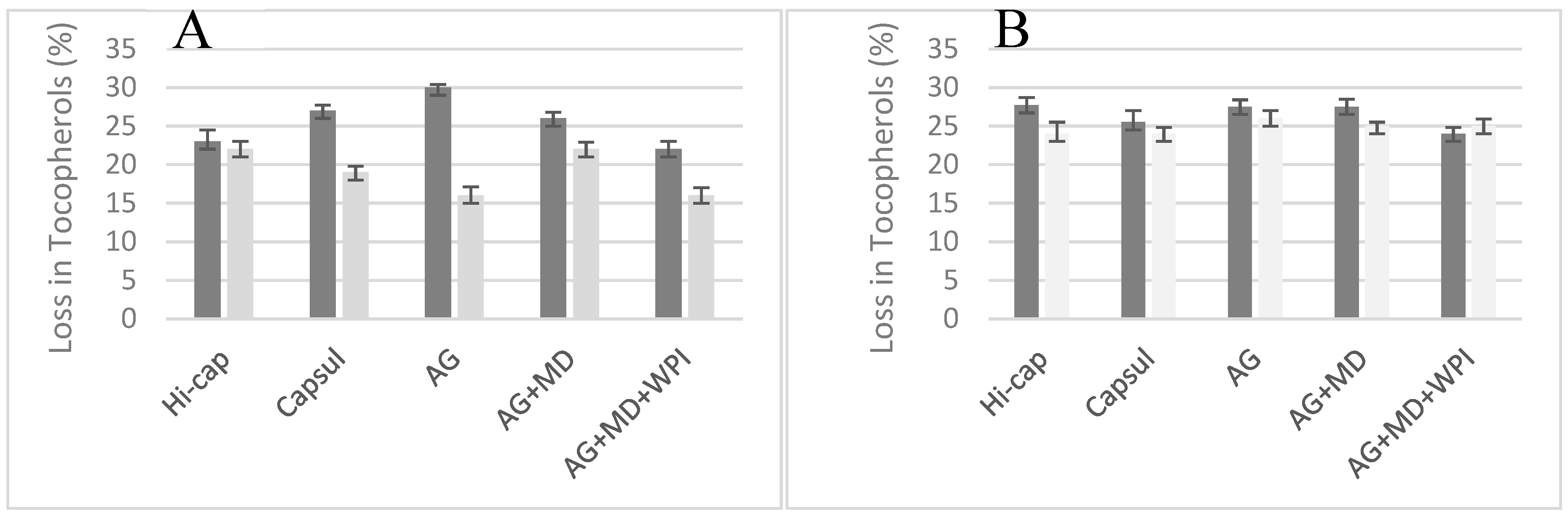
| Oil | Wall Material | SO (%w/w) | EE (%) |
|---|---|---|---|
| P. huayllabambana | Hi-cap | 1.3 ± 0.1 f | 93.3 ± 0.5 C |
| Capsul | 0.5 ± 0.1 h | 97.6 ± 0.5 A | |
| AG | 5.4 ± 0.1 f | 73.0 ± 1.5 E | |
| AG + MD | 4.8 ± 0.1 e | 75.8 ± 0.9 D | |
| AG+ MD + WPI | 7.7 ± 0.1 a | 61.1 ± 0.6 G | |
| P. volubilis | Hi-cap | 0.7 ± 0.1 g,h | 96.5 ± 0.7 A,B |
| Capsul | 0.8 ± 0.1 g | 96.0 ± 0.7 B | |
| AG | 6.5 ± 0.1 b | 67.4 ± 0.5 F | |
| AG + MD | 6.0 ± 0.1 c | 67.9 ± 0.5 F | |
| AG+ MD + WPI | 7.6 ± 0.1 a | 61.7 ± 0.6 G |
| Samples | Fatty Acid Composition (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| C16:0 | C16:1 | C17:0 | C17:1 | C18:0 | C18:1 | C18:2 | C18:3 | C20:0 | C20:1 | Trans | |
| P. huayllabambana | |||||||||||
| Initial oil | 4.50 ± 0.24 | 0.07 ± 0.01 | 0.06 ± 0.01 | 0.04 ± 0.01 | 1.75 ± 0.05 | 7.95 ± 0.30 | 26.10 ± 0.50 | 58.12 ± 0.55 a | 0.31 ± 0.15 | 0.29 ± 0.15 | nd * |
| Total encapsulated oils | |||||||||||
| SIHO + Hi-cap | 4.58 ± 0.23 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.04 ± 0.01 | 1.77 ± 0.09 | 7.88 ± 0.30 | 26.57 ± 0.50 | 58.61 ± 0.56 a | 0.31 ± 0.15 | 0.28 ± 0.15 | nd * |
| SIHO + Capsul | 4.82 ± 0.25 | 0.03 ± 0.01 | 0.04 ± 0.01 | 0.04 ± 0.01 | 1.84 ± 0.09 | 8.57 ± 0.30 | 26.56 ± 0.50 | 57.58 ± 0.52 a | 0.34 ± 0.15 | 0.30 ± 0.14 | nd * |
| SIHO + AG | 4.57 ± 0.25 | 0.06 ± 0.01 | 0.07 ± 0.01 | 0.04 ± 0.01 | 2.08 ± 0.08 | 8.62 ± 0.30 | 28.52 ± 0.50 | 55.46 ± 0.55 b | 0.30 ± 0.15 | 0.28 ± 0.14 | nd * |
| SIHO + AG + MD | 5.20 ± 0.23 | 0.03 ± 0.01 | 0.07 ± 0.01 | 0.05 ± 0.01 | 1.78 ± 0.09 | 11.08 ± 0.30 | 25.56 ± 0.52 | 55.60 ± 0.54 b | 0.30 ± 0.15 | 0.22 ± 0.15 | nd * |
| SIHO + AG + MD + WPI | 5.38 ± 0.24 | 0.04 ± 0.01 | 0.06 ± 0.01 | 0.04 ± 0.01 | 1.94 ± 0.09 | 12.38 ± 0.30 | 25.21 ± 0.53 | 54.50 ± 0.55 b | 0.31 ± 0.15 | 0.22 ± 0.14 | nd * |
| Surface oils | |||||||||||
| SIHO + Hi-cap | 4.65 ± 0.24 | 0.07 ± 0.01 | 0.06 ± 0.02 | 0.04 ± 0.00 | 1.70 ± 0.09 | 7.85 ± 0.35 | 26.20 ± 0.51 | 58.02 ± 0.55 a | 0.31 ± 0.15 | 0.29 ± 0.15 | nd * |
| SIHO + Capsul | 4.55 ± 0.23 | 0.07 ± 0.02 | 0.06 ± 0.02 | 0.04 ± 0.00 | 1.75 ± 0.09 | 7.90 ± 0.36 | 26.40 ± 0.50 | 58.00 ± 0.56 a | 0.33 ± 0.16 | 0.29 ± 0.10 | nd * |
| SIHO + AG | 4.52 ± 0.23 | 0.09 ± 0.01 | 0.08 ± 0.02 | 0.04 ± 0.01 | 2.13 ± 0.09 | 8.35 ± 0.35 | 28.98 ± 0.50 | 55.29 ± 0.54 b | 0.33 ± 0.15 | 0.23 ± 0.11 | nd * |
| SIHO+ AG + MD | 4.65 ± 0.24 | 0.06 ± 0.01 | 0.07 ± 0.02 | 0.04 ± 0.01 | 1.73 ± 0.09 | 7.84 ± 0.35 | 26.41 ± 0.58 | 58.71 ± 0.55 a | 0.30 ± 0.14 | 0.23 ± 0.12 | nd * |
| SIHO + AG + MD + WPI | 4.72 ± 0.25 | 0.07 ± 0.01 | 0.10 ± 0.01 | 0.04 ± 0.01 | 1.83 ± 0.09 | 8.07 ± 0.40 | 26.41 ± 0.58 | 58.26 ± 0.54 a | 0.32 ± 0.14 | 0.22 ± 0.10 | nd * |
| P. volubilis | |||||||||||
| Initial oil | 3.95 ± 0.21 | 0.06 ± 0.01 | 0.09 ± 0.01 | 0.05 ± 0.01 | 3.05 ± 0.15 | 9.90 ± 0.35 | 34.42 ± 0.58 | 47.88 ± 0.44 A,B | 0.32 ± 0.15 | 0.28 ± 0.15 | nd * |
| Total encapsulated oils | |||||||||||
| SIVO + Hi-cap | 4.06 ± 0.24 | 0.06 ± 0.01 | 0.09 ± 0.01 | 0.05 ± 0.01 | 3.06 ± 0.16 | 10.34 ± 0.34 | 34.27 ± 0.58 | 47.66 ± 0.45 B | 0.33 ± 0.15 | 0.28 ± 0.15 | nd * |
| SIVO + Capsul | 4.43 ± 0.23 | 0.08 ± 0.02 | 0.09 ± 0.01 | 0.05 ± 0.01 | 3.07 ± 0.16 | 12.04 ± 0.36 | 33.52 ± 0.58 | 46.18 ± 0.45 D | 0.31 ± 0.15 | 0.28 ± 0.15 | nd * |
| SIVO + AG | 4.21 ± 0.23 | 0.10 ± 0.02 | 0.79 ± 0.01 | 0.05 ± 0.01 | 3.34 ± 0.16 | 9.85 ± 0.35 | 33.91 ± 0.58 | 47.18 ± 0.41 C | 0.39 ± 0.01 | 0.23 ± 0.15 | nd * |
| SIVO + AG + MD | 4.10 ± 0.23 | 0.06 ± 0.02 | 0.05 ± 0.01 | 0.11 ± 0.01 | 3.12 ± 0.16 | 10.03 ± 0.35 | 34.27 ± 0.58 | 47.77 ± 045 B | 0.32 ± 0.01 | 0.23 ± 0.15 | nd * |
| SIV O+ AG + MD + WPI | 4.67 ± 0.23 | 0.09 ± 0.02 | 0.09 ± 0.01 | 0.05 ± 0.01 | 3.16 ± 0.16 | 11.44 ± 0.36 | 33.55 ± 0.58 | 46.40 ± 0.46 C,D | 0.32 ± 0.01 | 0.28 ± 0.15 | nd * |
| Surface oils | |||||||||||
| SIVO + Hi-cap | 4.09 ± 0.20 | 0.09 ± 0.02 | 0.09 ± 0.01 | 0.09 ± 0.01 | 3.06 ± 0.01 | 10.45 ± 0.29 | 34.33 ± 0.56 | 47.38 ± 0.44 B,C | 0.32 ± 0.15 | 0.27 ± 0.15 | nd * |
| SIVO + Capsul | 4.09 ± 0.20 | 0.09 ± 0.02 | 0.09 ± 0.01 | 0.09 ± 0.01 | 3.08 ± 0.01 | 9.42 ± 0.25 | 34.66 ± 0.56 | 48.06 ± 0.43 A,B | 0.31 ± 0.15 | 0.28 ± 0.15 | nd * |
| SIVO + AG | 3.99 ± 0.20 | 0.09 ± 0.02 | 0.09 ± 0.01 | 0.09 ± 0.01 | 3.04 ± 0.01 | 9.25 ± 0.25 | 34.73 ± 0.54 | 48.47 ± 0.45 A | 0.30 ± 0.16 | 0.21 ± 0.15 | nd * |
| SIVO + AG + MD | 3.95 ± 0.20 | 0.06 ± 0.02 | 0.09 ± 0.01 | 0.09 ± 0.01 | 3.05 ± 0.01 | 9.28 ± 0.25 | 34.70 ± 0.54 | 48.48 ± 0.44 A | 0.32 ± 0.15 | 0.21 ± 0.15 | nd * |
| SIVO + AG + MD + WPI | 4.18 ± 0.20 | 0.06 ± 0.01 | 0.09 ± 0.01 | 0.09 ± 0.01 | 3.10 ± 0.01 | 10.16 ± 0.29 | 34.22 ± 0.55 | 47.71 ± 0.45 A,B | 0.34 ± 0.15 | 0.28 ± 0.15 | nd * |
| Samples | Minor Glyceride Polar Compounds (mg·g−1) | |||
|---|---|---|---|---|
| TG-P | ox-TG | DG + MG + FA | Total PC | |
| P. huayllabambana | ||||
| Initial Oil | nd * | 7.8 ± 0.14 | 18.2 ± 0.15 | 26.0 ± 0.21 i |
| Total Encapsulated Oils | ||||
| SIHO + Hi-cap | 1.1 ± 0.02 | 13.7 ± 0.11 | 19.1 ± 0.16 | 33.9 ± 0.26 f |
| SIHO + Capsul | 2.0 ± 0.03 | 11.7 ± 0.12 | 19.1 ± 0.15 | 32.8 ± 0.32 h |
| SIHO + AG | 5.5 ± 0.06 | 20.5 ± 0.15 | 19.1 ± 0.15 | 45.1 ± 0.34 a |
| SIHO + AG + MD | 4.4 ± 0.07 | 20.3 ± 0.26 | 19.3 ± 0.15 | 44.0 ± 0.36 a |
| SIHO + AG + MD + WPI | 3.0 ± 0.05 | 17.7 ± 0.12 | 19.1 ± 0.15 | 39.8 ± 0.31 c |
| Surface Oils | ||||
| SIHO + Hi-cap | 0.6 ± 0.01 | 18.2 ± 0.10 | 19.3 ± 0.15 | 38.1 ± 0.22 d |
| SIHO + Capsul | 0.6 ± 0.05 | 11.0 ± 0.14 | 19.4 ± 0.17 | 31.0 ± 0.30 g |
| SIHO + AG | 0.6 ± 0.05 | 25.1 ± 0.20 | 19.3 ± 0.17 | 45.0 ± 0.38 a |
| SIHO + AG + MD | 0.8 ± 0.03 | 22.1 ± 0.10 | 19.2 ± 0.14 | 42.1 ± 0.28 b |
| SIHO + AG + MD + WPI | 1.0 ± 0.03 | 17.0 ± 0.11 | 19.0 ± 0.16 | 37.0 ± 0.24 e |
| P. volubilis | ||||
| Initial Oil | nd * | 6.5± 0.10 | 16.5± 0.16 | 23.0 ± 0.26 G |
| Total encapsulated Oils | ||||
| SIVO + Hi-cap | 0.4 ± 0.26 | 8.3 ± 0.26 | 17.0 ± 0.14 | 25.7 ± 0.35 F |
| SIVO + Capsul | 0.5 ± 0.26 | 8.1 ± 0.20 | 17.4 ± 0.16 | 26.0 ± 0.28 F |
| SIVO + AG | 0.9 ± 0.26 | 11.9 ± 0.12 | 17.4 ± 0.15 | 30.2 ± 0.28 B,C |
| SIVO + AG + MD | 0.2 ± 0.26 | 10.5 ± 0.26 | 17.4 ± 0.16 | 28.1 ± 0.28 D |
| SIVO + AG + MD + WPI | 0.7 ± 0.26 | 8.8 ± 0.26 | 17.4 ± 0.13 | 26.9 ± 0.24 E |
| Surface Oils | ||||
| SIVO + Hi-cap | 0.3 ± 0.26 | 11.3 ± 0.26 | 17.3 ± 0.16 | 28.9 ± 0.30 D |
| SIVO + Capsul | 0.9 ± 0.26 | 12.6 ± 0.14 | 17.4 ± 0.17 | 30.9 ± 0.29 B |
| SIVO + AG | 0.4 ± 0.26 | 14.4 ± 0.26 | 17.4 ± 0.15 | 32.2 ± 0.30 A |
| SIVO + AG + MD | 0.6 ± 0.26 | 14.9 ± 0.26 | 17.4 ± 0.11 | 32.9 ± 0.32 A |
| SIVO + AG + MD + WPI | 0.5 ± 0.26 | 13.4 ± 0.26 | 17.1 ± 0.18 | 31.0 ± 0.29 B |
| Tocopherols (mg·kg−1) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total Extracted Oil | Surface Oil | |||||||||||
| α- | β- | γ- | δ- | Total | Ratio γ/δ | α- | β- | γ- | δ- | Total | Ratio γ/δ | |
| P. huayllabambana | ||||||||||||
| Initial oil | nd * | nd * | 1811 ± 20 | 849 ± 15 | 2660 ± 45 a | 2.1 | ||||||
| SIHO + Hi-cap | nd * | nd * | 1295 ± 22 | 746 ± 14 | 2041 ± 40 d | 1.7 | nd * | nd * | 1221 ± 18 | 837 ± 14 | 2058 ± 32 d | 1.5 |
| SIHO + Capsul | nd * | nd * | 1155 ± 25 | 762 ± 15 | 1917 ± 30 e | 1.5 | nd * | nd * | 1300 ± 17 | 840 ± 20 | 2140 ± 32 c | 1.5 |
| SIHO + AG | nd * | nd * | 1104 ± 24 | 760 ± 14 | 1864 ± 35 f | 1.5 | nd * | nd * | 1375 ± 19 | 848 ± 15 | 2223 ± 36 b | 1.6 |
| SIHO + AG + MD | nd * | nd * | 1204 ± 22 | 764 ± 14 | 1968 ± 35 e | 1.6 | nd * | nd * | 1200 ± 19 | 848 ± 15 | 2048 ± 31 d | 1.4 |
| SIHO + AG + MD + WPI | nd * | nd * | 1290 ± 23 | 749 ± 16 | 2039 ± 35 d | 1.7 | nd * | nd * | 1406 ± 18 | 842 ± 12 | 2248 ± 30 b | 1.7 |
| P. volubilis | ||||||||||||
| Initial Oil | nd * | nd * | 2767 ± 32 | 1626 ± 30 | 4393 ± 45 A | 1.7 | ||||||
| SIVO + Hi-cap | nd * | nd * | 1596 ± 33 | 1585 ± 32 | 3181 ± 35 D | 1.0 | nd * | nd * | 1720 ± 31 | 1624 ± 30 | 3344 ± 35 B | 1.1 |
| SIVO + Capsul | nd * | nd * | 1699 ± 30 | 1558 ± 30 | 3257 ± 35 C | 1.1 | nd * | nd * | 1710 ± 31 | 1625 ± 35 | 3335 ± 30 B | 1.1 |
| SIVO + AG | nd * | nd * | 1550 ± 31 | 1615 ± 30 | 3165 ± 32 D | 1.0 | nd * | nd * | 1613 ± 33 | 1614 ± 32 | 3227 ± 33 C | 1.0 |
| SIVO + AG + MD | nd * | nd * | 1586 ± 30 | 1591 ± 30 | 3177 ± 33 D | 1.0 | nd * | nd * | 1675 ± 33 | 1614 ± 33 | 3289 ± 35 C | 1.0 |
| SIVO + AG + MD+ WPI | nd * | nd * | 1765 ± 32 | 1581 ± 30 | 3346 ± 35 B | 1.1 | nd * | nd * | 1657 ± 31 | 1622 ± 34 | 3279 ± 35 C | 1.0 |
| Sterols from Total Microencapsulated Oil | Sterols from the Surface Oil | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Samples | β-sitosterol (Mayor Phytosterol) % on Total Phytosterols | Stigmasterol/ Campesterol Ratio | Cholesterol (mg·kg−1) | Total Phytosterols (mg·kg−1) | β-sitosterol (Mayor Phytosterol) % on Total Phytosterols) | Stigmasterol/ Campesterol Ratio | Cholesterol (mg·kg−1) | Total Phytosterols (mg·kg−1) | |
| P. huayllabambana | Initial oil | 56.7 ± 1.5 | 5.9 ± 0,6 | * | 2056 ± 97 a,b | ||||
| SIHO + Hi-cap | 60.7 ± 3.0 | 5.1 ± 0.5 | * | 2119 ± 90 a | 62.2 ± 3.5 | 5.2 ± 0.6 | * | 1934 ± 75 b,c | |
| SIHO + Capsul | 61.8 ± 3.8 | 4.9 ± 0.5 | * | 2154 ± 95 a | 63.7 ± 3.5 | 5.3 ± 0.6 | * | 1888 ± 79 c | |
| SIHO + AG | 60.8 ± 3.1 | 5.2 ± 0.5 | * | 2101 ± 92 a | 61.6 ± 3.0 | 5.2 ± 0.5 | * | 1893 ± 80 c | |
| SIHO + AG + MD | 63.7 ± 6.0 | 5.1 ± 0.5 | * | 2050 ± 75 a | 61.2 ± 3.2 | 5.3 ± 0.5 | * | 1855 ± 80 c | |
| SIHO + AG + MD + WPI | 62.5 ± 4.5 | 4.3 ± 0.5 | 1014 ± 30 | 2104 ± 90 a | 61.5 ± 3.1 | 5.4 ± 0.6 | * | 1901 ± 75 c | |
| P. volubilis | Initial oil | 52.8 ± 2.0 | 3.3 ± 0.6 | * | 2225 ± 65 A | ||||
| SIVO + Hi-cap | 58.2 ± 2.5 | 3.7 ± 0.5 | * | 2255 ± 65 A | 56.0 ± 2.9 | 3.4 ± 0.5 | * | 2269 ± 85 A | |
| SIVO + Capsul | 58.6 ± 3.9 | 3.8 ± 0.5 | * | 2282 ± 60 A | 56.0 ± 2.9 | 3.4 ± 0.5 | * | 2290 ± 90 A | |
| SIVO + AG | 55.8 ± 3.0 | 4.1 ± 0.5 | * | 2356 ± 70 A | 56.5 ± 3.0 | 3.8 ± 0.5 | * | 2277 ± 95 A | |
| SIVO + AG + MD | 56.3 ± 3.4 | 3.1 ± 0.6 | * | 2348 ± 65 A | 55.8 ± 3.0 | 3.9 ± 0.5 | * | 2240 ± 95 A | |
| SIVO + AG + MD + WPI | 56.0 ± 3.2 | 3.0 ± 0.7 | 1072 ± 32 | 2321 ± 55 A | 55.8 ± 3.0 | 4.0 ± 0.6 | * | 2279 ± 95 A | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chasquibol, N.A.; Gallardo, G.; Gómez-Coca, R.B.; Trujillo, D.; Moreda, W.; Pérez-Camino, M.C. Glyceridic and Unsaponifiable Components of Microencapsulated Sacha Inchi (Plukenetia huayllabambana L. and Plukenetia volubilis L.) Edible Oils. Foods 2019, 8, 671. https://doi.org/10.3390/foods8120671
Chasquibol NA, Gallardo G, Gómez-Coca RB, Trujillo D, Moreda W, Pérez-Camino MC. Glyceridic and Unsaponifiable Components of Microencapsulated Sacha Inchi (Plukenetia huayllabambana L. and Plukenetia volubilis L.) Edible Oils. Foods. 2019; 8(12):671. https://doi.org/10.3390/foods8120671
Chicago/Turabian StyleChasquibol, Nancy A., Gabriela Gallardo, Raquel B. Gómez-Coca, Diego Trujillo, Wenceslao Moreda, and M. Carmen Pérez-Camino. 2019. "Glyceridic and Unsaponifiable Components of Microencapsulated Sacha Inchi (Plukenetia huayllabambana L. and Plukenetia volubilis L.) Edible Oils" Foods 8, no. 12: 671. https://doi.org/10.3390/foods8120671
APA StyleChasquibol, N. A., Gallardo, G., Gómez-Coca, R. B., Trujillo, D., Moreda, W., & Pérez-Camino, M. C. (2019). Glyceridic and Unsaponifiable Components of Microencapsulated Sacha Inchi (Plukenetia huayllabambana L. and Plukenetia volubilis L.) Edible Oils. Foods, 8(12), 671. https://doi.org/10.3390/foods8120671




