Impact of Commercial Oenotannin and Mannoprotein Products on the Chemical and Sensory Properties of Shiraz Wines Made from Sequentially Harvested Fruit
Abstract
:1. Introduction
2. Materials and Methods
2.1. Grapes and Wine
2.2. Addition of Oenotannin and Mannoprotein Supplements
- (1)
- No additives (control)
- (2)
- 300 mg/L skin oenotannin (skin)
- (3)
- 300 mg/L seed oenotannin (seed)
- (4)
- 400 mg/L mannoprotein (MP400)
- (5)
- 1000 mg/L mannoprotein (MP1000)
- (6)
- 300 mg/L skin oenotannin and 1000 mg/L mannoprotein (skin MP1000)
- (7)
- 300 mg/L seed oenotannin and 400 mg/L mannoprotein (seed MP400)
- (8)
- 300 mg/L seed oenotannin and 1000 mg/L mannoprotein (seed MP1000)
2.3. Chemical Analysis of Wines
2.4. Sensory Analysis of Wines
2.5. Data Analysis
3. Results and Discussion
3.1. Influence of Tannin and Mannoprotein Additions on Wine Composition
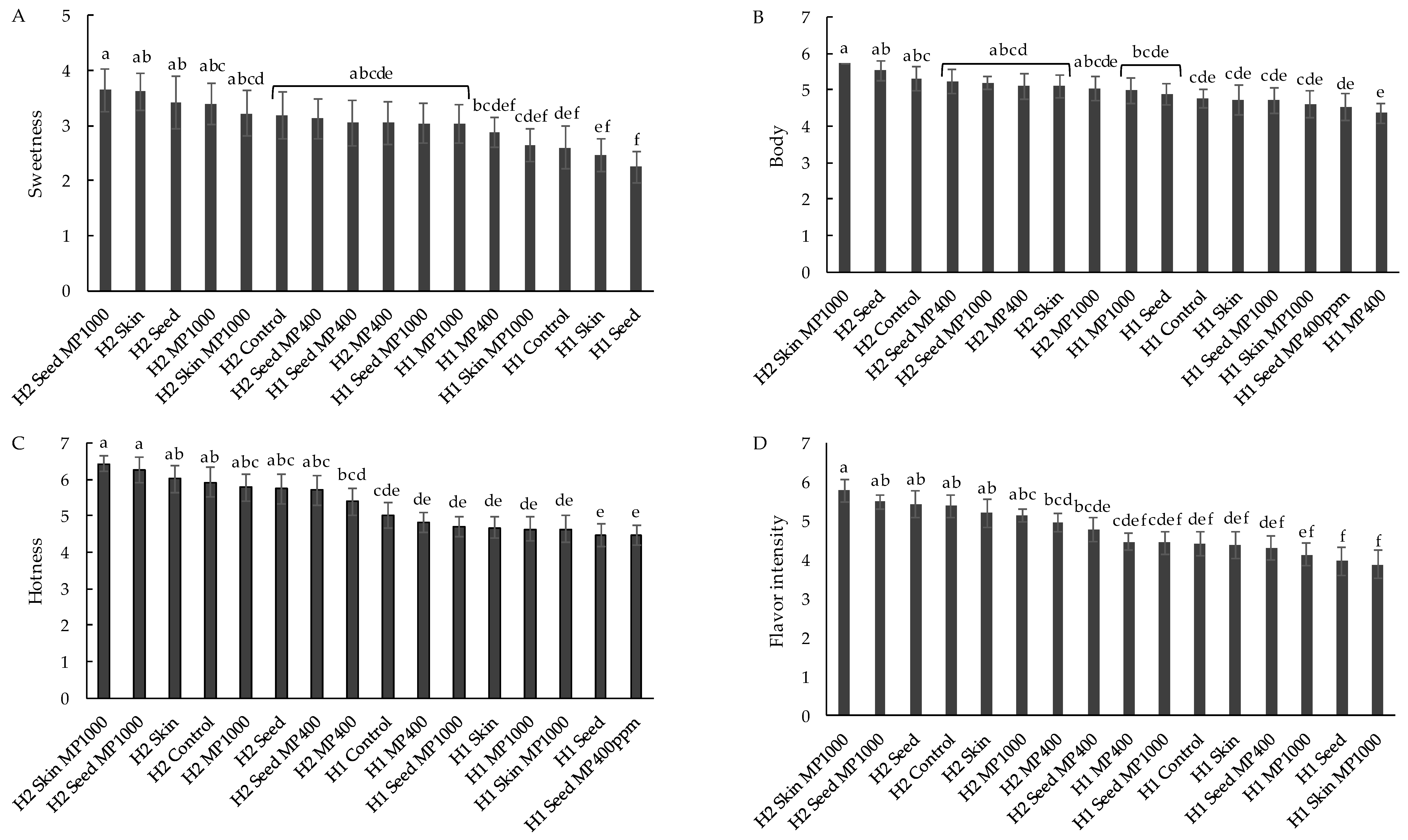
3.2. Influence of Tannin and Mannoprotein Additions on Wine Sensory Properties
3.2.1. Sensory Profiles of H1 and H2 Wines Following Addition of Wine Supplements
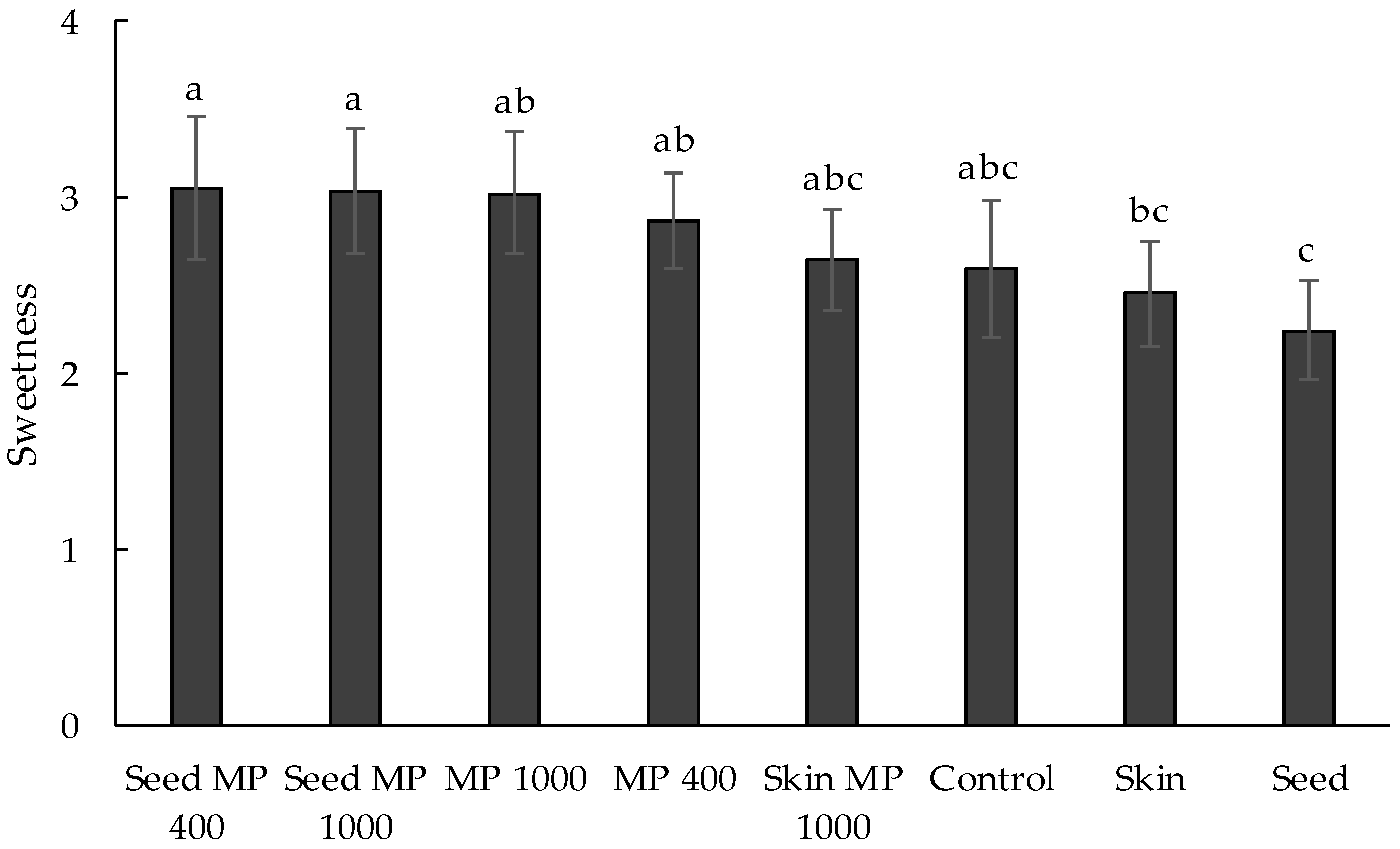
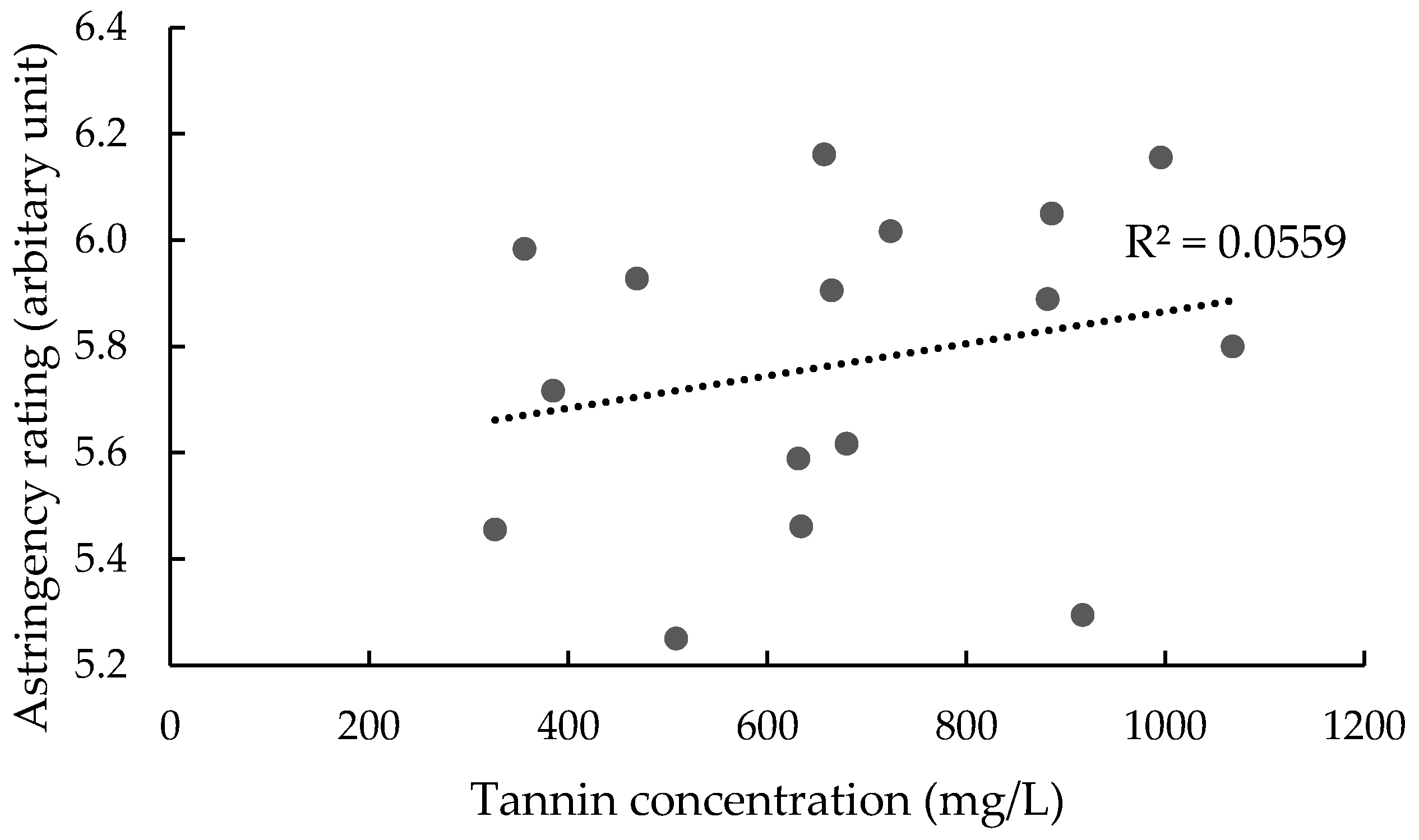
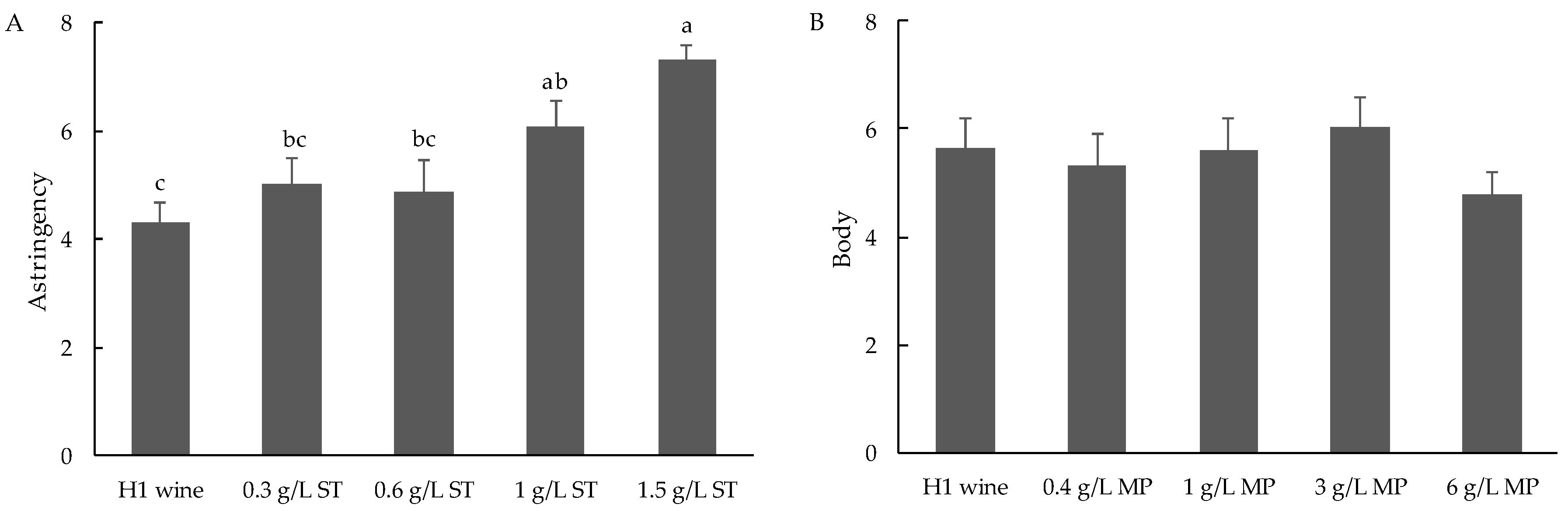
3.2.2. Influence of Oenotannin Supplementation on Perceived Astringency in Wine
3.2.3. Influence of Mannoprotein Supplementation on Perceived Body and Astringency in Wine
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alston, J.M.; Fuller, K.B.; Lapsley, J.T.; Soleas, G.; Tumber, K.P. The high and rising alcohol content of wine. In Wine Economics: Quantitative Studies and Empirical Applications; Giraud-Héraud, E., Pichery, M.-C., Eds.; Palgrave Macmillan UK: London, UK, 2013; pp. 350–364. ISBN 978-1-137-28952-0. [Google Scholar]
- Saliba, A.J.; Ovington, L.A.; Moran, C.C. Consumer demand for low-alcohol wine in an Australian sample. Int. J. Wine Res. 2013, 5, 1–8. [Google Scholar] [CrossRef]
- Bisson, L.F. Stuck and sluggish fermentations. Am. J. Enol. Vitic. 1999, 50, 107–119. [Google Scholar]
- Bucher, T.; Deroover, K.; Stockley, C. Low-alcohol wine: A narrative review on consumer perception and behaviour. Beverages 2018, 4, 82. [Google Scholar] [CrossRef]
- Longo, R.; Blackman, J.W.; Torley, P.J.; Rogiers, S.Y.; Schmidtke, L.M. Changes in volatile composition and sensory attributes of wines during alcohol content reduction. J. Sci. Food Agric. 2017, 97, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Bindon, K.; Holt, H.; Williamson, P.O.; Varela, C.; Herderich, M.; Francis, I.L. Relationships between harvest time and wine composition in Vitis vinifera L. cv. Cabernet Sauvignon 2. Wine sensory properties and consumer preference. Food Chem. 2014, 154, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Schelezki, O.J.; Šuklje, K.; Boss, P.K.; Jeffery, D.W. Comparison of consecutive harvests versus blending treatments to produce lower alcohol wines from Cabernet Sauvignon grapes: Impact on wine volatile composition and sensory properties. Food Chem. 2018, 259, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Longo, R.; Blackman, J.W.; Antalick, G.; Torley, P.J.; Rogiers, S.Y.; Schmidtke, L.M. Volatile and sensory profiling of Shiraz wine in response to alcohol management: Comparison of harvest timing versus technological approaches. Food Res. Int. 2018, 109, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Šuklje, K.; Zhang, X.; Antalick, G.; Clark, A.C.; Deloire, A.; Schmidtke, L.M. Berry shriveling significantly alters Shiraz (Vitis vinifera L.) grape and wine chemical composition. J. Agric. Food Chem. 2016, 64, 870–880. [Google Scholar] [CrossRef]
- Bindon, K.; Varela, C.; Kennedy, J.; Holt, H.; Herderich, M. Relationships between harvest time and wine composition in Vitis vinifera L. cv. Cabernet Sauvignon 1. Grape and wine chemistry. Food Chem. 2013, 138, 1696–1705. [Google Scholar] [CrossRef]
- Schelezki, O.J.; Smith, P.A.; Hranilovic, A.; Bindon, K.A.; Jeffery, D.W. Comparison of consecutive harvests versus blending treatments to produce lower alcohol wines from Cabernet Sauvignon grapes: Impact on polysaccharide and tannin content and composition. Food Chem. 2018, 244, 50–59. [Google Scholar] [CrossRef]
- Nurgel, C.; Pickering, G. Contribution of glycerol, ethanol and sugar to the perception of viscosity and density elicited by model white wines. J. Texture Stud. 2005, 36, 303–323. [Google Scholar] [CrossRef]
- Fischer, U.; Noble, A.C. The effect of ethanol, catechin concentration, and pH on sourness and bitterness of wine. Am. J. Enol. Vitic. 1994, 45, 6–10. [Google Scholar]
- Meillon, S.; Urbano, C.; Schlich, P. Contribution of the Temporal Dominance of Sensations (TDS) method to the sensory description of subtle differences in partially dealcoholized red wines. Food Qual. Prefer. 2009, 20, 490–499. [Google Scholar] [CrossRef]
- DeMiglio, P.; Pickering, G.J. The influence of ethanol and pH on the taste and mouthfeel sensations elicited by red wine. J. Food Agric. Environ. 2008, 6, 143–150. [Google Scholar]
- Lattey, K.A.; Bramley, B.R.; Francis, I.L. Consumer acceptability, sensory properties and expert quality judgements of Australian Cabernet Sauvignon and Shiraz wines. Aust. J. Grape Wine Res. 2010, 16, 189–202. [Google Scholar] [CrossRef]
- Schultz, H.R.; Jones, G.V. Climate induced historic and future changes in viticulture. J. Wine Res. 2010, 21, 137–145. [Google Scholar] [CrossRef]
- Gawel, R.; Smith, P.A.; Cicerale, S.; Keast, R. The mouthfeel of white wine. Crit. Rev. Food Sci. Nutr. 2017, 5, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Laguna, L.; Bartolomé, B.; Moreno-Arribas, M.V. Mouthfeel perception of wine: Oral physiology, components and instrumental characterization. Trends Food Sci. Technol. 2017, 59, 49–59. [Google Scholar] [CrossRef]
- Quijada-Morín, N.; Williams, P.; Rivas-Gonzalo, J.C.; Doco, T.; Escribano-Bailón, M.T. Polyphenolic, polysaccharide and oligosaccharide composition of Tempranillo red wines and their relationship with the perceived astringency. Food Chem. 2014, 154, 44–51. [Google Scholar] [CrossRef]
- Watrelot, A.A.; Schulz, D.L.; Kennedy, J.A. Wine polysaccharides influence tannin-protein interactions. Food Hydrocoll. 2017, 63, 571–579. [Google Scholar] [CrossRef]
- Del Barrio-Galán, R.; Pérez-Magariño, S.; Ortega-Heras, M.; Guadalupe, Z.; Ayestarán, B. Polysaccharide characterization of commercial dry yeast preparations and their effect on white and red wine composition. LWT Food Sci. Technol. 2012, 48, 215–223. [Google Scholar] [CrossRef] [Green Version]
- Versari, A.; Toit, W.; Parpinello, G.P. Oenological tannins: A review. Aust. J. Grape Wine Res. 2012, 19, 1–10. [Google Scholar] [CrossRef]
- Hill, M.; Kaine, G. The market potential of tannin related innovations in the wine industry. Enhancing the Uptake of Wine Industry Innovations through the Development of Targeted Extension Programs; Victorian Department of Primary Industries: Tatura, Australia, 2007. [Google Scholar]
- Smith, P.A.; Mcrae, J.M.; Bindon, K.A. Impact of winemaking practices on the concentration and composition of tannins in red wine. Aust. J. Grape Wine Res. 2015, 21, 601–614. [Google Scholar] [CrossRef]
- McRae, J.M.; Falconer, R.J.; Kennedy, J.A. Thermodynamics of grape and wine tannin interaction with polyproline: Implications for red wine astringency. J. Agric. Food Chem. 2010, 58, 12510–12518. [Google Scholar] [CrossRef] [PubMed]
- McRae, J.M.; Schulkin, A.; Kassara, S.; Holt, H.E.; Smith, P.A. Sensory properties of wine tannin fractions: Implications for in-mouth sensory properties. J. Agric. Food Chem. 2013, 61, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Guadalupe, Z.; Ayestarán, B.; Williams, P.; Doco, T. Determination of must and wine polysaccharides by gas chromatography-mass spectrometry (GC-MS) and Size-Exclusion Chromatography (SEC). In Polysaccharides: Bioactivity and Biotechnology; Springer International Publishing: Cham, Switzerland, 2015; pp. 1265–1297. ISBN 9783319162980. [Google Scholar]
- Vidal, S.; Francis, L.; Williams, P.; Kwiatkowski, M.; Gawel, R.; Cheynier, V.; Waters, E. The mouth-feel properties of polysaccharides and anthocyanins in a wine like medium. Food Chem. 2004, 85, 519–525. [Google Scholar] [CrossRef]
- Guadalupe, Z.; Palacios, A.; Ayestarán, B. Maceration enzymes and mannoproteins: A possible strategy to increase colloidal stability and color extraction in red wines. J. Agric. Food Chem. 2007, 55, 4854–4862. [Google Scholar] [CrossRef]
- Li, S.; Wilkinson, K.L.; Bindon, K.A. Compositional variability in commercial tannin and mannoprotein products. Am. J. Enol. Vitic. 2018, 176–181. [Google Scholar] [CrossRef]
- Bindon, K.A.; Kennedy, J.A. Ripening-induced changes in grape skin proanthocyanidins modify their interaction with cell walls. J. Agric. Food Chem. 2011, 59, 2696–2707. [Google Scholar] [CrossRef]
- Kassara, S.; Kennedy, J.A. Relationship between red wine grade and phenolics. 2. Tannin composition and size. J. Agric. Food Chem. 2011, 59, 8409–8412. [Google Scholar] [CrossRef]
- Kennedy, J.A.; Jones, G.P. Analysis of proanthocyanidin cleavage products following acid-catalysis in the presence of excess phloroglucinol. J. Agric. Food Chem. 2001, 49, 1740–1746. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J.A.; Taylor, A.W. Analysis of proanthocyanidins by high-performance gel permeation chromatography. J. Chromatogr. A 2003, 995, 99–107. [Google Scholar] [CrossRef]
- Li, S.; Bindon, K.; Bastian, S.E.P.; Jiranek, V.; Wilkinson, K.L. Use of winemaking supplements to modify the composition and sensory properties of Shiraz wine. J. Agric. Food Chem. 2017, 65, 1353–1364. [Google Scholar] [CrossRef] [PubMed]
- Niimi, J.; Danner, L.; Li, L.; Bossan, H.; Bastian, S.E.P. Wine consumers’ subjective responses to wine mouthfeel and understanding of wine body. Food Res. Int. 2017, 99, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Lawless, H.T.; Heymann, H. Sensory Evaluation of Food: Principles and Practices; Springer: Boston, MA, USA, 1999; pp. 301–340. ISBN 978-1-4419-7452-5. [Google Scholar]
- Mercurio, M.D.; Smith, P.A. Tannin quantification in red grapes and wine: Comparison of polysaccharide- and protein-based tannin precipitation techniques and their ability to model wine astringency. J. Agric. Food Chem. 2008, 56, 5528–5537. [Google Scholar] [CrossRef] [PubMed]
- Bindon, K.A.; Smith, P.A.; Holt, H.; Kennedy, J.A. Interaction between grape-derived proanthocyanidins and cell wall material. 2. implications for vinification. J. Agric. Food Chem. 2010, 58, 10736–10746. [Google Scholar] [CrossRef] [PubMed]
- McRae, J.M.; Kassara, S.; Kennedy, J.A.; Waters, E.J.; Smith, P.A. Effect of wine pH and bottle closure on tannins. J. Agric. Food Chem. 2013, 61, 11618–11627. [Google Scholar] [CrossRef] [PubMed]
- Robichaud, J.L.; Noble, A.C. Astringency and bitterness of selected phenolics in wine. J. Sci. Food Agric. 1990, 53, 343–353. [Google Scholar] [CrossRef]
- Kallithraka, S.; Kim, D.; Tsakiris, A.; Paraskevopoulos, I.; Soleas, G. Sensory assessment and chemical measurement of astringency of Greek wines: Correlations with analytical polyphenolic composition. Food Chem. 2011, 126, 1953–1958. [Google Scholar] [CrossRef]
- Vidal, S.; Courcoux, P.; Francis, L.; Kwiatkowski, M.; Gawel, R.; Williams, P.; Waters, E.; Cheynier, V. Use of an experimental design approach for evaluation of key wine components on mouth-feel perception. Food Qual. Prefer. 2004, 15, 209–217. [Google Scholar] [CrossRef]
- Bautista-Ortín, A.B.; Martínez-Cutillas, A.; Ros-García, J.M.; López-Roca, J.M.; Gómez-Plaza, E. Improving colour extraction and stability in red wines: The use of maceration enzymes and enological tannins. Int. J. Food Sci. Technol. 2005, 40, 867–878. [Google Scholar] [CrossRef]
- Harbertson, J.F.; Parpinello, G.P.; Heymann, H.; Downey, M.O. Impact of exogenous tannin additions on wine chemistry and wine sensory character. Food Chem. 2012, 131, 999–1008. [Google Scholar] [CrossRef]
- Kennedy, J.A.; Ferrier, J.; Harbertson, J.F.; Peyrot Des Gachons, C. Analysis of tannins in red wine using multiple methods: Correlation with perceived astringency. Am. J. Enol. Vitic. 2006, 57, 481–485. [Google Scholar]
- Landon, J.L.; Weller, K.; Harbertson, J.F.; Ross, C.F. Chemical and sensory evaluation of astringency in Washington State red wines. Am. J. Enol. Vitic. 2008, 2, 153–158. [Google Scholar]
- McRae, J.M.; Dambergs, R.G.; Kassara, S.; Parker, M.; Jeffery, D.W.; Herderich, M.J.; Smith, P.A. Phenolic compositions of 50 and 30 year sequences of Australian red wines: The impact of wine age. J. Agric. Food Chem. 2012, 60, 10093–10102. [Google Scholar] [CrossRef] [PubMed]
- Quijada-Morín, N.; Regueiro, J.; Simal-Gándara, J.; Tomás, E.; Rivas-Gonzalo, J.C.; Escribano-Bailón, M.T. Relationship between the sensory-determined astringency and the flavanolic composition of red wines. J. Agric. Food Chem. 2012, 60, 12355–12361. [Google Scholar] [CrossRef] [PubMed]
- Aleixandre-Tudo, J.L.; Buica, A.; Nieuwoudt, H.; Aleixandre, J.L.; Du Toit, W. Spectrophotometric analysis of phenolic compounds in grapes and wines. J. Agric. Food Chem. 2017, 65, 4009–4026. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Noble, A.C. Temporal perception of astringency and sweetness in red wine. Food Qual. Prefer. 1995, 6, 27–33. [Google Scholar] [CrossRef]
- Poncet-Legrand, C.; Doco, T.; Williams, P.; Vernhet, A. Inhibition of grape seed tannin aggregation by wine mannoproteins: Effect of polysaccharide molecular weight. Am. J. Enol. Vitic. 2007, 58, 87–91. [Google Scholar]
- Rinaldi, A.; Gambuti, A.; Moio, L. Precipitation of salivary proteins after the interaction with wine: The effect of ethanol, pH, fructose, and mannoproteins. J. Food Sci. 2012, 77, C485–C490. [Google Scholar] [CrossRef]
- Escot, S.; Feuillat, M.; Dulau, L.; Charpentier, C. Release of polysaccharides by yeasts and the influence of released polysaccharides on colour stability and wine astringency. Aust. J. Grape Wine Res. 2001, 7, 153–159. [Google Scholar] [CrossRef]
- Guadalupe, Z.; Ayestarán, B. Polysaccharide profile and content during the vinification and aging of Tempranillo red wines. J. Agric. Food Chem. 2007, 55, 10720–10728. [Google Scholar] [CrossRef] [PubMed]
- Gawel, R.; Oberholster, A.; Francis, I.L. A “Mouth-feel Wheel”: Terminology for communicating the mouth-feel characteristics of red wine. Aust. J. Grape Wine Res. 2000, 6, 203–207. [Google Scholar] [CrossRef]
- Gawel, R.; Van Sluyter, S.; Waters, E.J. The effects of ethanol and glycerol on the body and other sensory characteristics of Riesling wines. Aust. J. Grape Wine Res. 2007, 13, 38–45. [Google Scholar] [CrossRef]
- Gawel, R.; Smith, P.A.; Waters, E.J. Influence of polysaccharides on the taste and mouthfeel of white wine. Aust. J. Grape Wine Res. 2016, 22, 350–357. [Google Scholar] [CrossRef]
- Food Standards Australia New Zealand. Final Assessment Report: Application A605. Yeast Mannoproteins as a Food Additive for Wine. Available online: www.foodstandards.gov.au/code/applications/Documents/A605_Mannoproteins_FAR.pdf (accessed on 16 October 2018).
| Harvest | TSS (°Brix) | pH | TA 1 (g/L) | Malic Acid (g/L) | YAN 2 (g/L) |
|---|---|---|---|---|---|
| H1 | 20.8 | 3.5 | 5.2 | 3.5 | 0.21 |
| H2 | 24.5 | 3.7 | 4.4 | 2.9 | 0.25 |
| Harvest | Alcohol (% v/v) | pH | TA 1 (g/L) | Malic Acid (g/L) | Residual Sugar (g/L) | VA 2 (g/L) |
|---|---|---|---|---|---|---|
| H1 | 11.5 | 3.9 | 4.9 | <0.05 | 0.3 | <0.25 |
| H2 | 14.5 | 4.2 | 4.5 | 0.08 | 0.1 | 0.32 |
| Measurement | Control | Skin | Seed | MP400 | MP1000 | Skin MP1000 | Seed MP400 | Seed MP1000 | p |
|---|---|---|---|---|---|---|---|---|---|
| Total tannin after bottling (mg/L) | 329 ± 10 c | 557 ± 9 a | 608 ± 36 a | 381 ± 20 c | 373 ± 4 c | 476 ± 7 b | 608 ± 3 a | 605 ± 2 a | <0.001 |
| Total tannin (mg/L) | 326 ± 5 d | 469 ± 56 bc | 665 ± 22 a | 356 ± 32 d | 385 ± 14 cd | 508 ± 38 b | 634 ± 36 a | 631 ± 13 a | 0.001 |
| Extension subunits 1 | |||||||||
| C | 3.7 ± 0.1c | 4.3 ± 0.3 bc | 6.5 ± 0.2 a | 4.4 ± 0.3 bc | 4.3 ± 0.1 bc | 4.5 ± 0.2 b | 6.8 ± 0.2 a | 6.6 ± 6.6 a | <0.001 |
| EC | 47.6 ± 0.0 bcd | 49.8 ± 0.4 a | 47.8 ± 0.8 bc | 48.4 ± 0.9 ab | 47.1 ± 1.5 bcd | 48.1 ± 0.3 bc | 47.5 ± 0.3 bcd | 46.3 ± 0.4 d | 0.013 |
| EGC | 26.3 ± 0.8 a | 25.5 ± 0.2 a | 20.4 ± 0.1 b | 25.2 ±0.2 a | 26.6 ± 0.9 a | 26.2 ± 0.4 a | 20.3 ± 0.4 b | 21.5 ± 0.2 b | <0.001 |
| EC-G | 1.7 ± 0.1 bc | 1.6 ± 0.1 bc | 3.1 ± 1.8 a | 1.8 ± 0.0 b | 1.4 ± 0.1 c | 1.5 ± 0.1 bc | 2.8 ± 0.1 a | 2.8 ± 0.1 a | <0.001 |
| Terminal subunits 1 | |||||||||
| C | 16.2 ± 0.5 | 13.9 ± 0.1 | 14.4 ± 0.4 | 15.7 ± 0.7 | 15.5 ± 0.0 | 14.5 ± 0.2 | 14.8 ± 0.2 | 14.8 ± 0.5 | 0.089 |
| EC | 4.8 ± 0.2 b | 4.7 ± 0.1 b | 7.2 ± 0.2 a | 4.4 ± 0.4 b | 4.7 ± 0.3 b | 5.1 ± 0.1 b | 7.2 ± 0.3 a | 7.6 ± 0.2 a | <0.001 |
| EC-G | 0.2 ± 0.0 cd | 0.2 ± 0.0 c | 0.6 ± 0.0 a | 0.1 ± 0.0 cd | 0.2 ± 0.0 cd | 0.1 ± 0.1 d | 0.5 ± 0.1 ab | 0.4 ± 0.1 b | <0.001 |
| % Yield 2 | 22.0 ± 0.4 a | 15.2 ± 0.1 b | 16.0 ± 0.1 b | 21.2 ± 2.2 a | 18.6 ± 1.1 ab | 16.1 ± 1.5 b | 17.1 ± 1.3 b | 16.6 ± 0.8 b | 0.021 |
| mDP | 4.84 ± 0.16 bcd | 5.32 ± 0.03 a | 4.51 ± 0.12 cde | 4.96 ± 0.28 abc | 4.89 ± 0.09 abcd | 5.08 ± 0.08 ab | 4.44 ± 0.05 de | 4.38 ± 0.14 e | 0.014 |
| Tannin molecular mass (g/mol) 3 | 1670 ± 49 bc | 1745 ± 30 bc | 1931 ± 81 a | 1644 ± 58 c | 1621 ± 32 c | 1673 ± 49 bc | 1836 ± 55 ab | 1815 ± 50 ab | 0.027 |
| % Colored (520:280) 4 | 14.6 ± 0.1 | 14.7 ± 0.1 | 11.7 ± 0.0 | 12.1 ± 2.2 | 14.3 ± 0.0 | 14.2 ± 0.3 | 11.5 ± 0.2 | 11.7 ± 0.2 | 0.05 |
| Measurement | Control | Skin | Seed | MP400 | MP1000 | Skin MP1000 | Seed MP400 | Seed MP1000 | p |
|---|---|---|---|---|---|---|---|---|---|
| Total tannin after bottling (mg/L) | 686 ± 21 d | 824 ± 12 bc | 903 ± 12 ab | 733 ± 65 cd | 761 ± 18 cd | 825 ± 6 bc | 1006 ± 52 a | 976 ± 4 a | 0.001 |
| Total tannin (mg/L) | 679 ± 11 d | 916 ± 10 c | 995 ± 17 b | 724 ± 20 d | 657 ± 17d | 886 ± 10 c | 881 ± 14 c | 1067 ± 47 a | <0.001 |
| Extension subunits 1 | |||||||||
| C | 4.0 ± 0.5 | 4.4 ± 0.3 | 5.1 ± 0.4 | 4.2 ± 0.5 | 4.1 ± 0.5 | 4.3 ± 0.1 | 5.2 ± 0.0 | 5.4 ± 0.3 | 0.165 |
| EC | 48.8 ± 1.7 ab | 49.8 ± 0.1 a | 50.1 ± 0.0 a | 49.0 ± 1.1 ab | 46.7 ± 0.4 bc | 45.7 ± 0.5 c | 46.4 ± 0.8 bc | 46.4 ± 0.9 bc | 0.032 |
| EGC | 29.0 ± 2.2 ab | 28.2 ± 0.6 b | 23.7 ± 1.3 c | 28.2 ±1.0 b | 30.1 ± 0.6 ab | 32.1 ± 0.1 a | 26.6 ± 0.0 bc | 27.6 ± 1.1 b | 0.018 |
| EC-G | 2.1 ± 0.0 cd | 2.1 ± 0.1 cd | 2.9 ± 0.1 a | 2.2 ± 0.0 c | 2.1 ± 0.0 cd | 1.9 ± 0.0 d | 2.8 a ± 0.1 a | 2.4 ± 0.1 b | <0.001 |
| Terminal subunits 1 | |||||||||
| C | 11.2 ± 0.0 | 10.7 ± 0.2 | 12.0 ± 0.5 | 11.3 ± 0.3 | 11.8 ± 0.4 | 10.8 ± 0.3 | 11.9 ± 0.2 | 11.5 ± 0.1 | 0.085 |
| EC | 4.8 ± 0.1 b | 4.8 ± 0.3 b | 6.1 ± 0.3 a | 5.0 ± 0.2 b | 5.1 ± 0.0 b | 5.2 ± 0.2 b | 6.9 ± 0.4 a | 6.5 ± 0.3 a | 0.002 |
| EC-G | nd | nd | 0.2 ± 0.0 a | nd | nd | nd | 0.2 ± 0.0 a | 0.2 ± 0.0 b | <0.001 |
| % Yield 2 | 19.2 ± 3.9 | 18.2 ± 1.7 | 17.4 ± 0.6 | 21.1 ± 1.2 | 21.9 ± 1.3 | 18.1 ± 0.0 | 23.0 ± 0.9 | 18.3 ± 1.9 | 0.330 |
| mDP | 6.23 ± 0.01 ab | 6.46 ± 0.20 a | 5.47 ± 0.24 cd | 6.12 ± 0.17 ab | 5.89 ± 0.13 bc | 6.27 ± 0.20 ab | 5.28 ± 0.19 d | 5.50 ± 0.13 cd | 0.009 |
| Tannin molecular mass (g/mol) 3 | 1746 ± 3 | 1807 ± 26 | 1889 ± 19 | 1696 ± 54 | 1735 ± 41 | 1746 ± 75 | 1811 ± 12 | 1837 ± 52 | 0.132 |
| % Colored (520:280) 4 | 12.9 ± 0.1 ab | 13.0 ± 0.1 a | 11.4 ± 0.1 c | 12.7 ± 0.1 b | 12.8 ± 0.1 ab | 13.0 ± 0.0 a | 11.3 ± 0.0 c | 11.4 ± 0.0 c | <0.001 |
| Measurement | Control | Skin | Seed | MP400 | MP1000 | Skin MP1000 | Seed MP400 | Seed MP1000 | p |
|---|---|---|---|---|---|---|---|---|---|
| H1 wines | |||||||||
| Total polysaccharides (mg/L) | 401 ± 45 cd | 387 ± 3 d | 398 ± 4 d | 520 ±51 b | 801 ± 15 a | 755 ± 24 a | 495 ± 18 bc | 754 ± 29a | <0.001 |
| Mannose 1 | 72 ± 7 c | 68 ± 1 c | 69 ± 0 c | 186 ± 18 b | 409 ± 11 a | 378 ± 9 a | 173 ± 8 b | 397 ± 10 a | <0.001 |
| Glucose 2 | 20 ± 2 c | 21 ± 0 c | 21 ± 0 c | 40 ± 4 b | 80 ± 0 a | 78 ± 5 a | 37 ± 1 b | 71 ± 4 a | <0.001 |
| H2 wines | |||||||||
| Total polysaccharides (mg/L) | 403 ± 4 bcd | 395 ± 9 cd | 383 ± 23 d | 518 ± 27 b | 720 ± 28 a | 782 ± 67 a | 504 ± 14 bc | 820 ± 64 a | <0.001 |
| Mannose 1 | 98 ± 2 cd | 92 ± 3 d | 91 ± 6 d | 214 ± 10 b | 404 ± 4 a | 432 ± 36 a | 210 ± 7 bc | 452 ± 42 a | <0.001 |
| Glucose | 19 ± 1 b | 24 ± 3 b | 20 ± 1 b | 29 ± 2 b | 53 ± 4 a | 60 ± 6 a | 27 ± 3 b | 62 ± 2 a | <0.001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Bindon, K.; Bastian, S.; Wilkinson, K. Impact of Commercial Oenotannin and Mannoprotein Products on the Chemical and Sensory Properties of Shiraz Wines Made from Sequentially Harvested Fruit. Foods 2018, 7, 204. https://doi.org/10.3390/foods7120204
Li S, Bindon K, Bastian S, Wilkinson K. Impact of Commercial Oenotannin and Mannoprotein Products on the Chemical and Sensory Properties of Shiraz Wines Made from Sequentially Harvested Fruit. Foods. 2018; 7(12):204. https://doi.org/10.3390/foods7120204
Chicago/Turabian StyleLi, Sijing, Keren Bindon, Susan Bastian, and Kerry Wilkinson. 2018. "Impact of Commercial Oenotannin and Mannoprotein Products on the Chemical and Sensory Properties of Shiraz Wines Made from Sequentially Harvested Fruit" Foods 7, no. 12: 204. https://doi.org/10.3390/foods7120204
APA StyleLi, S., Bindon, K., Bastian, S., & Wilkinson, K. (2018). Impact of Commercial Oenotannin and Mannoprotein Products on the Chemical and Sensory Properties of Shiraz Wines Made from Sequentially Harvested Fruit. Foods, 7(12), 204. https://doi.org/10.3390/foods7120204






