Use of Optical Oxygen Sensors in Non-Destructively Determining the Levels of Oxygen Present in Combined Vacuum and Modified Atmosphere Packaged Pre-Cooked Convenience-Style Foods and the Use of Ethanol Emitters to Extend Product Shelf-Life
Abstract
:1. Introduction
2. Experimental Section
2.1. Optical O2 Sensor and Analysis
2.2. Ethanol Emitter Preparation
2.3. Sample Preparation
| Sample | Abbreviation | No. of Units | Original Packaging | New Packaging |
|---|---|---|---|---|
| per Pack | Settings (MPa) | Settings (MPa) | ||
| Battered Sausages | BS | 6 | V (0.20)/G (0.06) | V (0.35)/G (0.02) |
| or V (0.01) | ||||
| Bacon Slices | BA | 8 | V (0.01) | V (0.35)/G (0.02) |
| Beef & Potato Pie | PP | 4 | V (0.01) | V (0.35)/G (0.02) |
2.4. Microbial Analysis
2.5. Sensory Analysis Design
2.6. Statistical Analysis
| Attribute | Description |
|---|---|
| Overall Appearance | 0 = Extremely Dislike, 10 = Extremely Like |
| Off Aroma | 0 = None, 10 = Extreme |
| Ethanol Aroma | 0 = None, 10 = Extreme |
| Acid Aroma | 0 = None, 10 = Extreme |
| Overall Flavour Liking | 0 = Extremely Dislike, 10 = Extremely Like |
| Off Flavour | 0 = None, 10 = Extreme |
| Sour Flavour | 0 = None, 10 = Extreme |
| Astringent Taste | 0 = None, 10 = Extreme |
| Ethanol Flavour | 0 = None, 10 = Extreme |
| Overall acceptability | 0 = Extremely Unacceptable, 10 = Extremely Unacceptable |

| Sample | Time | |||||||
|---|---|---|---|---|---|---|---|---|
| BS | BS + EE | PP | PP + EE | BA | BA + EE | Day 14 | Day 35 | |
| Overall Appearance | 0.93 ns | −0.33 ns | 0.27 ns | 0.23 ns | 0.47 ns | 0.78 ns | 0.64 ns | 0.58 ns |
| Off Aroma | −0.003 ** | 0.001 *** | 0.001 *** | −0.004 ** | −0.85 ns | −0.001 *** | −0.40 ns | 0.42 ns |
| Ethanol Aroma | 0.001 *** | 0.001 *** | 0.001 *** | −0.003 ** | −0.90 ns | −0.001 *** | −0.61 ns | 0.64 ns |
| Acid Aroma | −0.04 * | 0.02 * | −0.19 ns | −0.17 ns | −0.53 ns | −0.28 ns | −0.59 ns | 0.63 ns |
| Overall Flavour Liking | 0.002 ** | 0.001 *** | 0.002 ** | 0.004 ** | 0.99 ns | 0.001 *** | 0.67 ns | −0.61 ns |
| Off Flavour | −0.002 ** | 0.001 *** | −0.003 ** | −0.005 ** | −0.74 ns | −0.001 *** | −0.56 ns | 0.38 ns |
| Sour Flavour | −0.003 ** | 0.001 *** | −0.002 ** | −0.007 ** | −0.83 ns | −0.001 *** | −0.48 ns | 0.46 ns |
| Astringent Taste | −0.002 ** | 0.001 *** | −0.015 * | −0.02 * | −0.94 ns | −0.001 *** | −0.81 ns | 0.77 ns |
| Ethanol Flavour | 0.001 *** | 0.001 *** | 0.001 *** | −0.002 ** | −0.65 ns | −0.001 *** | −0.48 ns | 0.37 ns |
| Overall Acceptablility | 0.003 ** | 0.001 *** | 0.001 *** | 0.002 ** | 0.75 ns | 0.001 *** | 0.45 ns | −0.42 ns |
3. Results and Discussion
3.1. Packaging Assessment and Optical O2 Readings
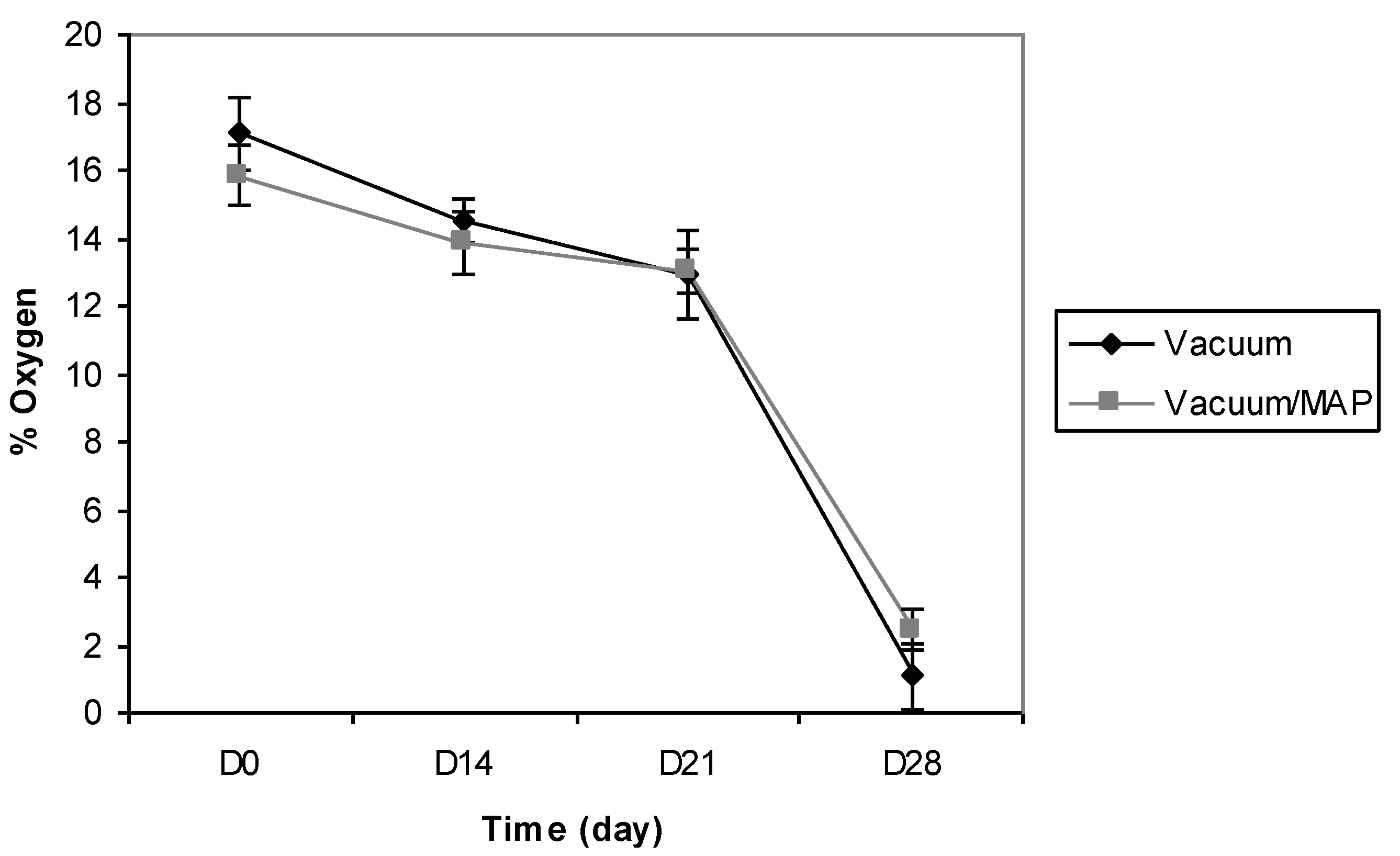
| Differential | |||||||
|---|---|---|---|---|---|---|---|
| Sample | D0 | D7 | D14 | D21 | D28 | D35 | D0–D35 |
| V (0.01)/G (0.02) | 14.7 | 13.5 | 13.3 | 12.5 | 10.4 | 4.1 | 10.7 |
| V (0.04)/G (0.02) | 15.4 | 13.7 | 13.5 | 13.5 | 12.6 | 5.0 | 10.4 |
| V (0.08)/G (0.02) | 14.7 | 13.5 | 13.2 | 12.4 | 11.8 | 7.0 | 7.7 |
| V (0.20)/G (0.02) | 12.2 | 11.5 | 10.4 | 9.8 | 8.1 | 6.8 | 5.4 |
| V (0.35)/G (0.02) | 8.2 | 6.9 | 6.7 | 6.3 | 6.0 | 4.5 | 3.7 |
| V (0.60)/G (0.02) | 8.1 | 6.5 | 6.3 | 6.1 | 6.0 | 4.4 | 3.7 |
| V (0.01)/G (0.03) | 14.3 | 13.5 | 13.1 | 12.2 | 10.1 | 4.0 | 10.3 |
| V (0.04)/G (0.03) | 15.1 | 13.7 | 13.5 | 13.5 | 12.6 | 5.0 | 10.1 |
| V (0.08)/G (0.03) | 15.0 | 13.5 | 13.0 | 12.2 | 11.5 | 7.1 | 7.9 |
| V (0.20)/G (0.03) | 11.4 | 11.4 | 10.1 | 9.5 | 7.8 | 6.8 | 4.6 |
| V (0.35)/G (0.03) | 8.4 | 6.7 | 6.5 | 6.3 | 6.1 | 4.6 | 3.8 |
| V (0.60)/G (0.03) | 8.2 | 6.4 | 6.3 | 6.0 | 5.8 | 4.5 | 3.7 |
3.2. Revised Packaging Treatment
3.3. Microbial Analysis
| Sample | Time (Day) | Differential % | |||||
|---|---|---|---|---|---|---|---|
| D0 | D7 | D14 | D21 | D28 | D35 | D0–D35 | |
| BS Normal 1 (V (0.01)) | 16.5 ± 0.81 | 13.2 ± 0.74 | 13.8 ± 0.75 | 12.6 ± 0.57 | 8.6 ± 0.62 | 0.9 ± 0.44 | 15.7 |
| BS Normal 1 (V (0.01)) + EE | 16.6 ± 0.70 | 15.3 ± 0.83 | 14.6 ± 0.52 | 13.9 ± 0.66 | 10.4 ± 0.87 | 5.7 ± 1.20 | 10.9 |
| BS Normal 2 (V (0.20)/G (0.06)) | 12.9 ± 1.00 | 11.1 ± 0.91 | 10.2 ± 0.61 | 9.7 ± 0.83 | 6.4 ± 0.76 | 4.1 ± 0.86 | 8.8 |
| BS Normal 2 (V (0.20)/G (0.06)) + EE | 12.8 ± 0.90 | 11.3 ± 0.40 | 10.4 ± 0.64 | 9.9 ± 0.85 | 8.7 ± 0.57 | 6.1 ± 0.77 | 6.7 |
| BS (V (0.35)/G (0.02)) | 7.5 ± 0.39 | 6.7 ± 0.48 | 6.8 ± 0.32 | 6.8 ± 0.36 | 7.0 ± 0.20 | 5.0 ± 0.27 | 2.5 |
| BS (V (0.35)/G (0.02)) + EE | 8.4 ± 0.41 | 6.8 ± 0.37 | 6.7 ± 0.38 | 6.5 ± 0.44 | 6.7 ± 0.22 | 7.4 ± 0.11 | 1.0 |
| PP Normal (V (0.01)) | 14.1 ± 1.10 | 13.1 ± 0.83 | 12.5 ± 0.64 | 8.4 ± 0.76 | 2.7 ± 0.46 | 0.9 ± 0.62 | 13.2 |
| PP Normal (V (0.01)) + EE | 14.4 ± 0.94 | 13.8 ± 0.73 | 12.9 ± 0.88 | 9.4 ± 0.72 | 6.4 ± 0.65 | 4.7 ± 0.80 | 10.7 |
| PP (V (0.35)/G (0.02)) | 7.3 ± 0.51 | 6.5 ± 0.47 | 6.4 ± 0.47 | 6.3 ± 0.56 | 5.5 ± 0.32 | 2.2 ± 0.43 | 5.1 |
| PP (V (0.35)/G (0.02)) + EE | 6.9 ± 0.33 | 6.6 ± 0.38 | 6.8 ± 0.13 | 5.9 ± 0.23 | 6.0 ± 0.27 | 4.3 ± 0.11 | 2.7 |
| BA Normal (0.01) | 16.9 ± 1.31 | 16.5 ± 0.73 | 16.4 ± 0.33 | 15.3 ± 0.37 | 14.4 ± 0.64 | 8.2 ± 0.89 | 10.7 |
| BA Normal (0.01) + EE | 16.8 ± 1.00 | 16.5 ± 0.84 | 16.2 ± 0.76 | 15.8 ± 0.53 | 15.1 ± 0.45 | 11.7 ± 0.44 | 5.1 |
| BA (V (0.35)/G (0.02)) | 9.5 ± 0.45 | 7.9 ± 0.53 | 7.7 ± 0.45 | 7.7 ± 0.27 | 7.7 ± 0.36 | 7.9 ± 0.40 | 1.6 |
| BA (V (0.35)/G (0.02)) + EE | 7.1 ± 0.22 | 6.3 ± 0.46 | 6.1 ± 0.32 | 6.1 ± 0.13 | 6.8 ± 0.12 | 6.3 ± 0.18 | 0.9 |
| Sample | Time | ||||||
|---|---|---|---|---|---|---|---|
| D0 | D7 | D14 | D21 | D28 | D35 | D42 | |
| BS Normal 1 (V (0.01)) | 2.8 ± 0.14 | 3.2 ± 0.21 | 3.9 ± 0.32 | 4.5 ± 0.17 | ^6.1 ± 0.10 | n/d | n/d |
| BS Normal 1 (V (0.01)) + EE | 2.9 ± 0.09 | 3.1 ± 0.15 | 3.7 ± 0.35 | 4.0 ± 0.22 | ^5.1 ± 0.09 | n/d | n/d |
| BS Normal 2 (V (0.2)/G (0.06)) | 2.6 ± 0.25 | 3.0 ± 0.07 | 3.5 ± 0.23 | 4.3 ± 0.27 | ^5.8 ± 0.10 | n/d | n/d |
| BS Normal 2 (V (0.2)/G (0.06)) + EE | 2.8 ± 0.18 | 3.2 ± 0.21 | 3.4 ± 0.32 | 3.8 ± 0.19 | ^5.0 ± 0.28 | n/d | n/d |
| BS (V (0.35)/G (0.02)) | 1.8 ± 0.29 | 2.2 ± 0.45 | 2.7 ± 0.49 | 2.9 ± 0.34 | 3.6 ± 0.37 | 4.8 ± 0.07 | ^6.0 ± 0.29 |
| BS (V (0.35)/G (0.02)) + EE | 1.7 ± 0.27 | 1.8 ± 0.29 | 2.0 ± 0.49 | 2.3 ± 0.27 | 3.1 ± 0.32 | 4.1 ± 0.29 | 4.8 ± 0.12 |
| CB Normal (V (0.01)) | 2.8 ± 0.20 | 3.2 ± 0.26 | 3.7 ± 0.15 | 4.2 ± 0.18 | ^6.8 ± 0.11 | n/d | n/d |
| CB Normal (V (0.01)) + EE | 2.7 ± 0.28 | 3.2 ± 0.37 | 3.6 ± 0.41 | 3.9 ± 0.50 | ^5.2 ± 0.07 | n/d | n/d |
| CB (V (0.35)/G (0.02)) | 1.7 ± 0.17 | 2.1 ± 0.12 | 2.4 ± 0.07 | 2.8 ± 0.04 | 3.4 ± 0.16 | 4.5 ± 0.21 | ^5.4 ± 0.29 |
| CB (V (0.35)/G (0.02)) + EE | 1.5 ± 0.04 | 2.0 ± 0.13 | 2.2 ± 0.09 | 2.5 ± 0.11 | 3.0 ± 0.27 | 4.4 ± 0.31 | ^5.0 ± 0.42 |
| BA Normal (V (0.01)) | 2.9 ± 0.10 | 3.1 ± 0.16 | 3.6 ± 0.19 | 4.2 ± 0.14 | ^6.8 ± 0.08 | n/d | n/d |
| BA Normal (V (0.01)) + EE | 2.8 ± 0.06 | 3.0 ± 0.12 | 3.2 ± 0.22 | 3.8 ± 0.15 | ^5.6 ± 0.12 | n/d | n/d |
| BA (V (0.35)/G (0.02)) | 1.4 ± 0.07 | 2.0 ± 0.09 | 2.4 ± 0.17 | 2.9 ± 0.21 | 3.7 ± 0.04 | 4.8 ± 0.05 | ^5.3 ± 0.17 |
| BA (V (0.35)/G (0.02)) + EE | 1.2 ± 0.05 | 1.3 ± 0.08 | 1.6 ± 0.14 | 1.9 ± 0.18 | 2.4 ± 0.10 | 3.9 ± 0.07 | 4.8 ± 0.11 |
3.4. Sensory and Statistical Analyses
4. Conclusions
Acknowledgements
Conflicts of Interest
References
- De Barcellos, M.D.; Grunert, K.G.; Scholderer, J. Processed Meat Products: Consumer and Emerging Markets. In Processed Meats; Improving Safety, Nutrition and Quality; Kerry, J.P., Kerry, J.F., Eds.; Woodhead Publishing: Cornwall, UK, 2011. [Google Scholar]
- Brunner, T.A.; van der Horst, K.; Siegrist, M. Convenience food products. Drivers for consumption. Appetite 2010, 55, 498–506. [Google Scholar] [CrossRef]
- Scholderer, J.; Grunert, K.G. Consumers, food and convenience: The long way from resource to actual consumption patterns. J. Econ. Psychol. 2005, 26, 105–128. [Google Scholar] [CrossRef]
- Subramaniam, P.J. Miscellaneous Applications. In Principles of Modified Atmosphere Packaging of Food; Parry, R.T., Ed.; Blackie Academic and Professional: Suffolk, UK, 1993. [Google Scholar]
- Kanatt, S.R.; Shobita Rao, M.; Chawla, S.P.; Sharma, A. Shelf life extension of convenience meat products sold in Indian supermarkets by radiation processing. Radiat. Phys. Chem. 2010, 79, 1259–1263. [Google Scholar] [CrossRef]
- Jofre, A.; Aymerich, T.; Grebol, N.; Garriga, M. Efficiency of high hydrostatic pressure at 600 MPa against food borne microorganisms by challenge tests on convenience meat products. LWT Food Sci. Technol. 2009, 42, 924–928. [Google Scholar] [CrossRef]
- Vercammen, A.; Vanoirbeek, K.G.A.; Lurquin, I.; Steen, L.; Goemaere, O.; Szczepaniak, S.; Paelinck, H.; Hendrickx, M.E.G.; Michiels, C.W. Shelf life extension of cooked ham model product by high hydrostatic pressure and natural preservatives. Innov. Food Sci. Emerg. Technol. 2011, 12, 407–415. [Google Scholar] [CrossRef]
- Yam, K.L. Smart Packaging/Intelligent Packaging/MAP. In Wiley Encyclopaedia of Food Packaging, 3rd ed.; John Wiley & Sons Publication: Hoboken, NJ, USA, 2009. [Google Scholar]
- Floros, J.D.; Matos, K.I. Introduction in Modified Atmosphere Packaging. In Innovations in Food Packaging; Han, J.H., Ed.; Elsevier Academic Press: London, UK, 2005. [Google Scholar]
- Lee, D.S.; Yam, K.L.; Piergiovanni, L. Vacuum and Modified Atmosphere Packaging. In Food Packaging Science and Technology; Lee, D.S., Yam, K.L., Piergiovanni, L., Eds.; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Hogan, S.A.; Kerry, J.P. Fluorescent Based Oxygen Sensors. In Smart Packaging Technologies for Fast Moving Consumer Goods; Kerry, J.P., Butler, P., Eds.; John Wiley & Sons Publication: Wiltshire, UK, 2008. [Google Scholar]
- Kerry, J.P.; O’Grady, M.N.; Hogan, S.A. Past, current and potential utilisations of active and intelligent packaging systems for meat and muscle-based products: A review. Meat Sci. 2006, 74, 113–130. [Google Scholar] [CrossRef]
- Papkovsky, D.B. Newoxygensensors and their application to bio sensing. Sensors Actuators B Chem. 1995, 29, 213–218. [Google Scholar] [CrossRef]
- Papkovsky, D.B. Methods in optical oxygen sensing: Protocols and critical Analyses. Methods Enzymol. 2004, 381, 715–735. [Google Scholar] [CrossRef]
- Kerry, J.P.; Papkovsky, D.B. Development and use of non-destructive, continuous assessment, chemical oxygen sensors in packs containing oxygen sensitive foodstuffs. Res. Adv. Food Sci. 2002, 3, 121–140. [Google Scholar]
- Papkovsky, D.B.; Papkovskaia, N.; Smyth, A.; Kerry, J.P.; Ogurtsov, I. Phosphorescent sensor approach for non-destructive measurement of oxygen in packaged foods. Optimisation of disposal sensors and their characterisation over a wide temperature range. Anal. Lett. 2000, 33, 1755–1777. [Google Scholar] [CrossRef]
- O’Mahoney, F.; O’Riordan, T.C.; Papkovskaia, N.; Kerry, J.P.; Papkovsky, D.B. Non-destructive assessment of oxygen levels in industrial modified atmosphere packaged cheddar cheese. Food Control 2006, 17, 286–292. [Google Scholar] [CrossRef]
- Hempel, A.W.; Gillanders, R.N.; Papkovsky, D.B.; Kerry, J.P. Detection of cheese packaging containment failures using reversible optical oxygen sensors. Int. J. Dairy Technol. 2012, 65, 456–460. [Google Scholar] [CrossRef]
- Smiddy, M.; Fitzgerald, M.; Kerry, J.P.; Papkovsky, D.B.; O’Sullivan, C.K.; Guilbault, G.G. Use of oxygen sensors to non-destructively measure the oxygen content in modified atmosphere and vacuum packed beef: Impact of oxygen content on lipid oxidation. Meat Sci. 2002, 61, 285–290. [Google Scholar] [CrossRef]
- Smiddy, M.; Papkovsky, D.; Kerry, J.P. Evaluation of oxygen content in commercial modified packs of processed cooked meats. Food Res. Int. 2002, 35, 571–575. [Google Scholar] [CrossRef]
- Smiddy, M.; Papkovskaia, N.; Papkovsky, D.P.; Kerry, J.P. Use of oxygen sensors for the non-destructive measurement of the oxygen content in modified atmosphere and vacuum packs of cooked chicken patties; impact of oxygen content on lipid oxidation. Food Res. Int. 2002, 35, 577–584. [Google Scholar] [CrossRef]
- O’Mahoney, F.; O’Riordan, T.C.; Papkovskaia, N.; Ogurtsov, V.I.; Kerry, J.P.; Papkovsky, D.B. Assessment of oxygen levels in convenience-style muscle based sous vide products through optical means and impact on shelf life stability. Packag. Technol. Sci. 2004, 12, 225–234. [Google Scholar]
- Hempel, A.W.; O’Sullivan, M.G.; Papkovsky, D.B.; Kerry, J.P. Use of optical oxygen sensors to monitor residual oxygen in pre- and post-pasteurised bottled beer and its affect on sensory attributes and product acceptability during simulated commercial storage. LWT Food Sci. Technol. 2013, 50, 226–231. [Google Scholar] [CrossRef]
- Robertson, G.L. Food Packaging—Principles and Practice, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Day, B.P.F. Ethanol Emitters. In Smart Packaging Technologies for Fast Moving Consumer Goods; Kerry, J.P., Butler, P., Eds.; John Wiley & Sons Publication: Wiltshire, UK, 2008. [Google Scholar]
- Brody, A.L.; Strupinsky, E.R.; Kline, L.R. Antimicrobial Packaging. In Active Packaging for Food Applications; Brody, A.L., Strupinsky, E.R., Kline, L.R., Eds.; A Technomic Publication: Lancaster, PA, USA, 2001. [Google Scholar]
- Commission Regulation (EC) No 2073/2005 of 15 November 2005 on Microbiological Criteria for Foodstuffs (Text with EEA Relevance). Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CONSLEG:2005R2073:20071227:EN:PDF (accessed on 11 December 2012).
- ISO 8589:2007. Sensory Analysis—General Guidance for the Design of Test Rooms; International Organization for Standardization: Geneva, Switzerland, 2007.
- Hempel, A.W.; O’Sullivan, M.G.; Papkovsky, D.B.; Kerry, J.P. Use of smart packaging technologies for monitoring and extending the shelf-life quality of modified atmosphere packaged (MAP) bread: Application of intelligent oxygen sensors and active ethanol emitters. Eur. Food Res. Technol. 2013, in press. [Google Scholar]
- Latou, E.; Mexis, S.F.; Badeka, A.V.; Kontominas, M.G. Shelf life extension of sliced wheat bread using either an ethanol emitter or an ethanol emitter combined with an oxygen absorber as alternatives to chemical preservatives. J. Cereal Sci. 2010, 52, 457–465. [Google Scholar] [CrossRef]
- ISO 2073:2005. Microbiological Criteria for Foodstuffs; International Organization for Standards: Geneva, Switzerland, 2005.
- Smith, J.P.; Hoshino, J.; Abe, Y. Interactive Packaging Involving Sachet Technology. In Active Food Packaging; Rooney, M.L., Ed.; Blackie Academic and Professional: Glasgow, UK, 1995. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hempel, A.W.; Papkovsky, D.B.; Kerry, J.P. Use of Optical Oxygen Sensors in Non-Destructively Determining the Levels of Oxygen Present in Combined Vacuum and Modified Atmosphere Packaged Pre-Cooked Convenience-Style Foods and the Use of Ethanol Emitters to Extend Product Shelf-Life. Foods 2013, 2, 507-520. https://doi.org/10.3390/foods2040507
Hempel AW, Papkovsky DB, Kerry JP. Use of Optical Oxygen Sensors in Non-Destructively Determining the Levels of Oxygen Present in Combined Vacuum and Modified Atmosphere Packaged Pre-Cooked Convenience-Style Foods and the Use of Ethanol Emitters to Extend Product Shelf-Life. Foods. 2013; 2(4):507-520. https://doi.org/10.3390/foods2040507
Chicago/Turabian StyleHempel, Andreas W., Dmitri B. Papkovsky, and Joseph P. Kerry. 2013. "Use of Optical Oxygen Sensors in Non-Destructively Determining the Levels of Oxygen Present in Combined Vacuum and Modified Atmosphere Packaged Pre-Cooked Convenience-Style Foods and the Use of Ethanol Emitters to Extend Product Shelf-Life" Foods 2, no. 4: 507-520. https://doi.org/10.3390/foods2040507
APA StyleHempel, A. W., Papkovsky, D. B., & Kerry, J. P. (2013). Use of Optical Oxygen Sensors in Non-Destructively Determining the Levels of Oxygen Present in Combined Vacuum and Modified Atmosphere Packaged Pre-Cooked Convenience-Style Foods and the Use of Ethanol Emitters to Extend Product Shelf-Life. Foods, 2(4), 507-520. https://doi.org/10.3390/foods2040507




