Occurrence, Dissipation and Risk Assessment of Widespread Pesticides and Their Metabolites in Pomegranates
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Field Trials and Sample Collection
2.3. Analytical Procedures
2.4. Dietary Risk Assessment
2.5. Data Analysis
3. Results
3.1. Method Validation
3.2. Occurrence and Dissipation of Four Pesticides in Both Pomegranate Arils and Whole Pomegranate Matrices
3.3. Terminal Levels and MRL Comparison of DIF, PRO, SPI, DIN and Their Metabolites in Pomegranates
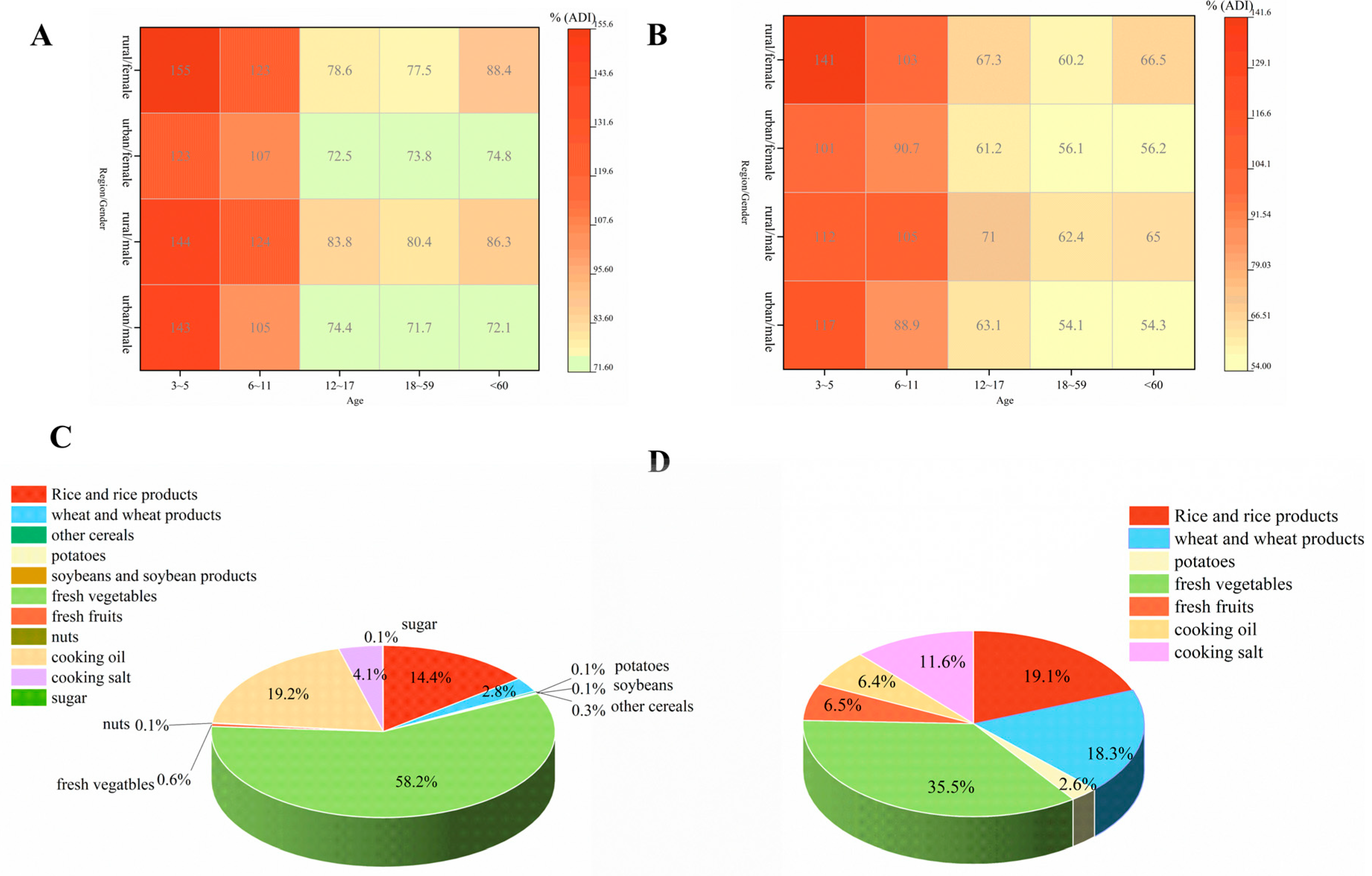
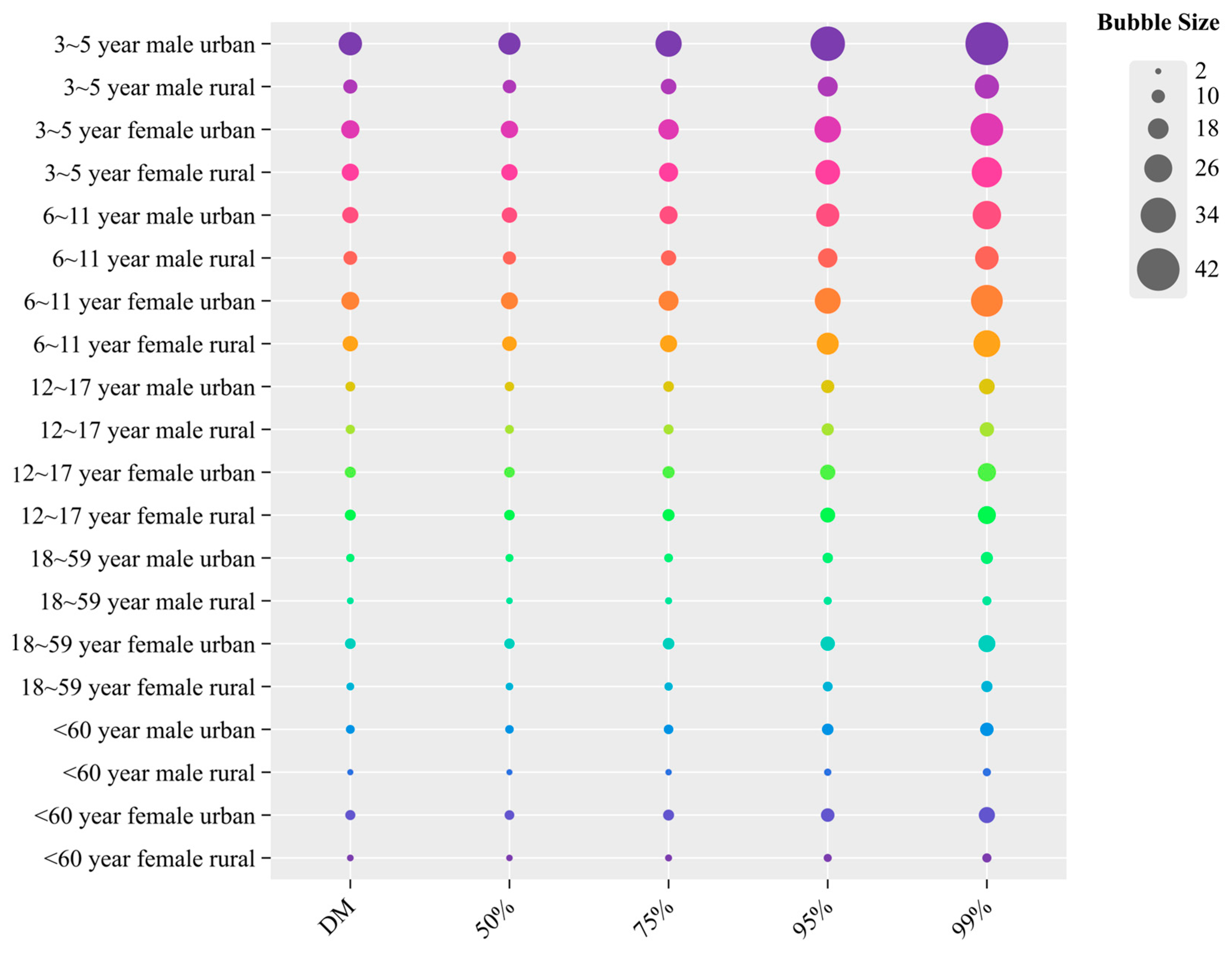
3.4. Dietary Risk Assessment of DIF and PRO Using Deterministic and Probabilistic Models
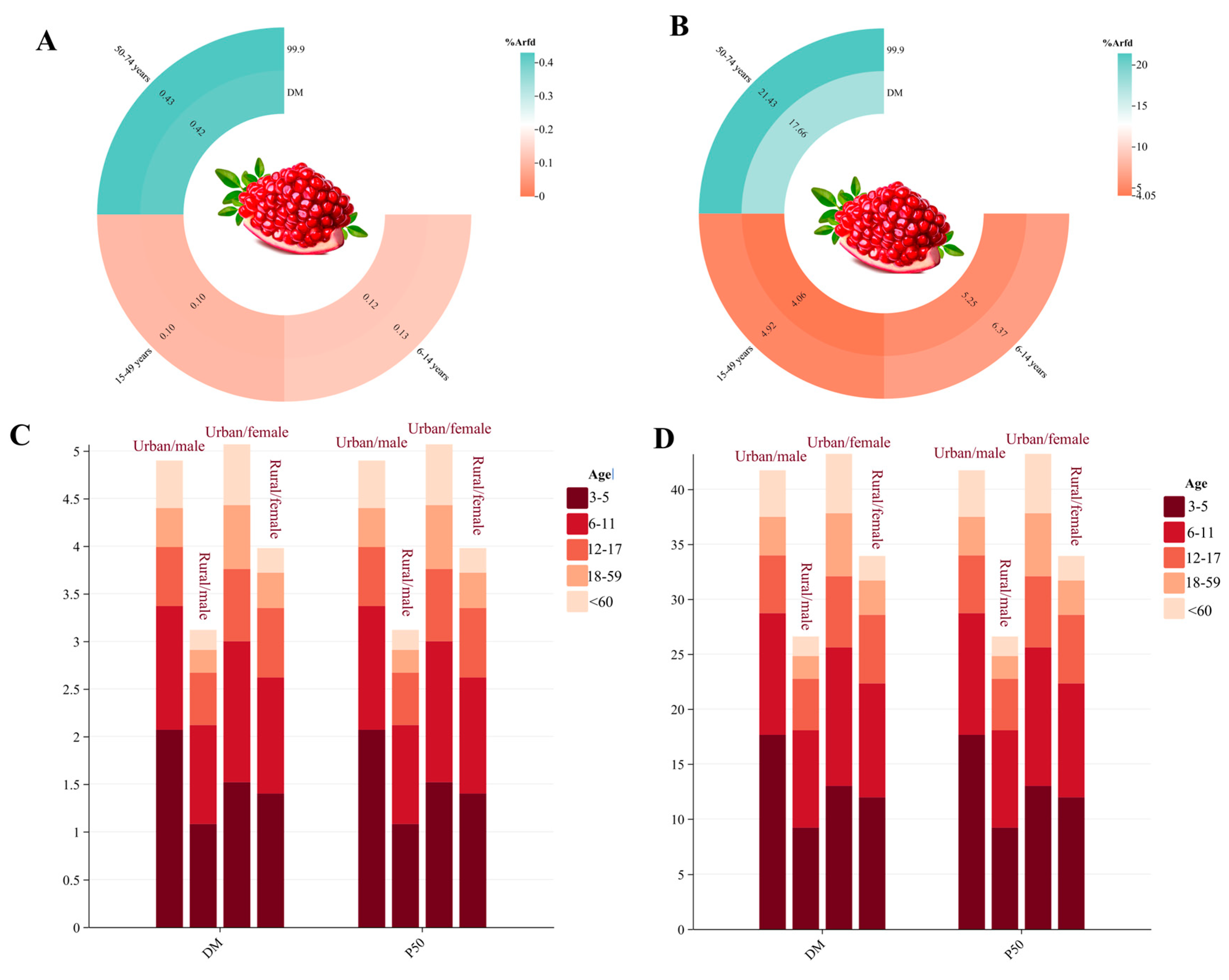
3.4.1. Acute Dietary Risk Assessment of DIF and PRO
3.4.2. Chronic Dietary Risk Assessment of DIF and PRO
3.5. Analysis of Non-Carcinogenic Effects of PRO Using Deterministic and Probabilistic Models
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DIF | Difenoconazole |
| PRO | Prochloraz |
| SPI | Spinosad |
| DIN | Dinotefuran |
| UF | UF (Metabolite of DIN) |
| DN | DN (Metabolite of DIN) |
| MRL | Maximum Residue Limit |
| ARfD | Acute Reference Dose |
| ADI | Acceptable Daily Intake |
| IESTI | International Estimated Short-Term Intake |
| NEDI | National Estimated Daily Intake |
| HQ | Hazard Quotient |
| RfD | Reference Dose |
| STMR | Supervised Trials Median Residue |
| HR | Highest Residue |
| UHPLC-MS/MS | Ultra-High Performance Liquid Chromatography-Tandem Mass Spectrometry |
| GC-ECD | Gas Chromatography with Electron Capture Detector |
| HPLC | High Performance Liquid Chromatography |
| QuEChERS | Quick, Easy, Cheap, Effective, Rugged, Safe |
| MRM | Multiple Reaction Monitoring |
| ESI | Electrospray Ionization |
| JMPR | Joint FAO/WHO Meeting on Pesticide Residues |
| OECD | Organisation for Economic Co-operation and Development |
| CAC | Codex Alimentarius Commission |
| FAO | Food and Agriculture Organization of the United Nations |
| PHI | Pre-Harvest Interval |
| LOQ | Limit of Quantification |
| ME | Matrix Effect |
| bw | Body Weight |
| SFO | Single First-Order |
| DFOP | Double First-Order in Parallel |
| FOMC | First-Order Multi-Compartment |
References
- Shahbazi, N.; Azizi, A.; Sayyari, M.; Seyedi, F.S.; Pourhosseini, S.H. Valorisation of pomegranate waste: A sustainable approach to harnessing natural antioxidants. Bioresour. Technol. Rep. 2025, 31, 102178. [Google Scholar] [CrossRef]
- Melgarejo-Sánchez, P.; Núñez-Gómez, D.; Martínez-Nicolás, J.J.; Hernández, F.; Legua, P.; Melgarejo, P. Pomegranate variety and pomegranate plant part, relevance from bioactive point of view: A review. Bioresour. Bioprocess. 2021, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.-H.; Li, J.; Adili, G.; Sun, G.-Y.; Abuduaini, M.; Abdulla, R.; Maiwulanjiang, M.; Aisa, H.A. Phenolic Compounds and Bioactivities from Pomegranate (Punica granatum L.) Peels. J. Agric. Food Chem. 2022, 70, 3678–3686. [Google Scholar] [CrossRef] [PubMed]
- Gormez, E.; Odabas, E.; Golge, O.; González-Curbelo, M.Á.; Kabak, B. Assessment of pesticide contamination in pomegranates: A multivariate approach and health risk evaluation. Food Chem. Toxicol. 2025, 200, 115363. [Google Scholar] [CrossRef] [PubMed]
- Munhuweyi, K.; Lennox, C.L.; Meitz-Hopkins, J.C.; Caleb, O.J.; Opara, U.L. Major diseases of pomegranate (Punica granatum L.), their causes and management—A review. Sci. Hortic. 2016, 211, 126–139. [Google Scholar] [CrossRef]
- Naik, R.H.; Pallavi, M.S.; Nandini; Shwetha, A.; Bheemanna, M.; Nidoni, R.U. Simultaneous determination of pesticide residues in pomegranate whole fruit and arils using LC-MS/MS. Food Chem. 2022, 387, 132865. [Google Scholar] [CrossRef]
- Mohapatra, S.; Siddamallaiah, L.; Matadha, N.Y.; Udupi, V.R.; Raj, D.P.; Gadigeppa, S. Dissipation of neonicotinoid insecticides imidacloprid, indoxacarb and thiamethoxam on pomegranate (Punica granatum L.). Ecotoxicol. Environ. Saf. 2019, 171, 130–137. [Google Scholar] [CrossRef]
- Chen, M.; Tao, L.; McLean, J.; Lu, C. Quantitative Analysis of Neonicotinoid Insecticide Residues in Foods: Implication for Dietary Exposures. J. Agric. Food Chem. 2014, 62, 6082–6090. [Google Scholar] [CrossRef]
- Tian, F.; Qiao, C.; Wang, C.; Pang, T.; Guo, L.; Li, J.; Pang, R.; Xie, H. Dissipation behavior of prochloraz and its metabolites in grape under open-field, storage and the wine-making process. J. Food Compos. Anal. 2022, 114, 104846. [Google Scholar] [CrossRef]
- Li, J.; Liu, Q.; Zhou, C.; Xie, J. Antifungal activity, physiological disruption, and toxicity mechanisms of difenoconazole in Sclerotinia sclerotiorum. Crop Prot. 2025, 197, 107329. [Google Scholar] [CrossRef]
- Wang, W.-G.; Xiong, S.-Q.; Lu, J.; Zhu, L.-H.; Zhang, C.; Cheng, J.-G.; Li, Z.; Xu, W.-P.; Tao, L.-M.; Zhang, Y. The effects of Spinosad on zebrafish larvae and THP-1 cells: Associated with immune cell damage and NF-kappa B signaling pathway activation. Chemosphere 2023, 343, 140237. [Google Scholar] [CrossRef]
- Kim, I.; Seok, S.H.; Lee, H.-Y. Evaluation of cardiac toxicity of a neonicotinoid insecticide, dinotefuran, in a zebrafish heart failure animal model. Ecotoxicol. Environ. Saf. 2025, 305, 119246. [Google Scholar] [CrossRef]
- Shi, G.; Zhang, Y.; Nie, M.; Huang, W.; Long, J.; Long, H.; Duan, T.; Cui, X. Dissipation, processing factors and risk assessment of chlorothalonil, chlorfenapyr and difenoconazole residues in chilli peppers from field to table. J. Food Compos. Anal. 2025, 137, 106885. [Google Scholar] [CrossRef]
- Si, W.; Song, Y.; Guo, C.; Huang, Z.; Chen, L.; Xu, F.; Bai, B.; Huo, K.; Wang, S. Multi-model risk assessment of pesticide residues in seasonal fruits based on integrated targeted/non-targeted screening: Implications for vulnerable populations. Food Chem. X 2025, 28, 102599. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Wei, Y.; Wang, D.; Huang, J.; Wan, N.; Wei, J.; Li, B. Risk assessment of 369 pesticide residues in banana from Hainan province of China through UPLC-Q-TOF/MS. J. Food Compos. Anal. 2023, 123, 105638. [Google Scholar] [CrossRef]
- Yu, W.; Wu, R.; Zhang, L.; Pan, Y.; Ling, J.; Yang, D.; Qu, J.; Tao, Z.; Meng, R.; Shen, Y.; et al. Identification of key factors affecting neonicotinoid residues in crops and risk of dietary exposure. Environ. Pollut. 2024, 345, 123489. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Liu, K.; Chen, L.; Wang, Y.; Ni, Y.; Hu, Y.; Jia, X.; Han, F.; Liu, Y.; Zhou, D. Determination of 118 pesticide residues in urine by ultra-high performance liquid chromatography-tandem mass spectrometry. Chin. J. Chromatogr. 2024, 42, 52–63. [Google Scholar] [CrossRef]
- Peng, F.-J.; Palazzi, P.; Mezzache, S.; Adelin, E.; Bourokba, N.; Bastien, P.; Appenzeller, B.M.R. Cross-Sectional Examination of Thyroid Hormones and Environmental Exposure to Multiclass Pesticides in Women of Reproductive Age in China. Environ. Health Perspect. 2024, 132, 107005. [Google Scholar] [CrossRef]
- Ueyama, J.; Aoi, A.; Ueda, Y.; Oya, N.; Sugiura, Y.; Ito, Y.; Ebara, T.; Kamijima, M. Biomonitoring method for neonicotinoid insecticides in urine of non-toilet-trained children using LC-MS/MS. Food Addit. Contam. Part A 2019, 37, 304–315. [Google Scholar] [CrossRef]
- Peng, F.-J.; Palazzi, P.; Mezzache, S.; Adelin, E.; Bourokba, N.; Bastien, P.; Appenzeller, B.M.R. Association between Environmental Exposure to Multiclass Organic Pollutants and Sex Steroid Hormone Levels in Women of Reproductive Age. Environ. Sci. Technol. 2023, 57, 19383–19394. [Google Scholar] [CrossRef]
- Dong, B. Recent advances in the toxicological effects of difenoconazole: A focus on toxic mechanisms in fish and mammals. Chemosphere 2024, 368, 143751. [Google Scholar] [CrossRef] [PubMed]
- Benchikh, I.; Ziani, K.; Gonzalez Mateos, A.; Khaled, B.M. Non-acute exposure of neonicotinoids, health risk assessment, and evidence integration: A systematic review. Crit. Rev. Toxicol. 2024, 54, 194–213. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Meng, Z.; Tian, S.; Teng, M.; Yan, J.; Jia, M.; Li, R.; Zhou, Z.; Zhu, W. Neonicotinoid insecticides exposure cause amino acid metabolism disorders, lipid accumulation and oxidative stress in ICR mice. Chemosphere 2020, 246, 125661. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Lu, J.; Zhao, Y.; Guo, Z.; Hu, Y.; Liu, Y.; Zhu, C. Photochemical reactions between superoxide ions and 2,4,6-trichlorophenol in atmospheric aqueous environments. Chemosphere 2021, 279, 130537. [Google Scholar] [CrossRef]
- Lin, Q.; Huang, Y.; Li, G.; Luo, Z.; Wang, L.; Li, D.; Xiang, Y.; Liu, L.; Ban, Z.; Li, L. The journey of prochloraz pesticide in Citrus sinensis: Residual distribution, impact on transcriptomic profiling and reduction by plasma-activated water. J. Hazard. Mater. 2023, 448, 130931. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, R.; Zeng, X.; Wang, X.; Wang, D.; Jia, H.; Xu, W.; Gao, Y. Exposure to neonicotinoid insecticides and their characteristic metabolites: Association with human liver cancer. Environ. Res. 2022, 208, 112703. [Google Scholar] [CrossRef]
- Tian, F.; Zhou, Z.; Lu, J.; Qiao, C.; Wang, C.; Pang, T.; Guo, L.; Li, J.; Pang, R.; Xie, H. Residual behaviors and health risk assessment of dinotefuran, flonicamid, and their metabolites during apple growth, storage, and processing. Food Res. Int. 2025, 205, 115970. [Google Scholar] [CrossRef]
- Li, T.; Li, L.; Li, S.; Ren, X.; Wu, M.; Liu, F.; Cheng, Y.; Chen, Z. Integrating processing factors and large-scale cabbage cultivation to understand the fate tendency and health risks of tolfenpyrad using deterministic and probabilistic models. J. Hazard. Mater. 2025, 486, 137131. [Google Scholar] [CrossRef]
- Floare-Avram, C.V.; Molnár, C.; Covaciu, F.-D. Integrated UHPLC-PDA and GC-ECD approaches for detecting pesticide residues in frozen fruits and vegetables: Unveiling contaminants and assessing health risks. J. Food Compos. Anal. 2025, 145, 107715. [Google Scholar] [CrossRef]
- Cui, K.; Guan, S.; Liang, J.; Ni, R.; Ding, R.; Li, T.; Wang, J.; Liu, J.; Fang, L.; Dong, Z.; et al. Residue behavior, processing factors and risk assessment of four new fungicides in grapes from field to processing. Food Control 2025, 177, 111454. [Google Scholar] [CrossRef]
- Zhang, W.; Zhou, C.; Zhou, F.; Zalán, Z.; Shi, H.; Kan, J.; Cai, T.; Chen, K. Determination of twelve neonicotinoid pesticides in chili using an improved QuEChERS method with UPLC-Q-TOF/MS. Food Chem. 2024, 452, 139463. [Google Scholar] [CrossRef]
- Zhu, Y.; Zheng, Y.; Dong, F.; Wu, X.; Pan, X.; Xu, J. Insight into abiotic and biotic transformations of fenaminstrobin in water and soil: Kinetics, transformation mechanism and ecotoxicity. J. Hazard. Mater. 2025, 493, 138324. [Google Scholar] [CrossRef]
- Kang, S.; Li, L.; Ren, X.; Zhang, M.; Li, W.; Chen, Z. Occurrence and fate characteristics of isoproturon from garlic cultivation to household processing: Implication for human exposure. J. Hazard. Mater. 2023, 448, 130936. [Google Scholar] [CrossRef]
- Utture, S.C.; Banerjee, K.; Kolekar, S.S.; Dasgupta, S.; Oulkar, D.P.; Patil, S.H.; Wagh, S.S.; Adsule, P.G.; Anuse, M.A. Food safety evaluation of buprofezin, dimethoate and imidacloprid residues in pomegranate. Food Chem. 2012, 131, 787–795. [Google Scholar] [CrossRef]
- Harischandra Naik, R.; Pavankumar, K.; Pallavi, M.S.; Shwetha, A.; Saraswati, M.; Paramasivam, M.; Patil, N.; Naveen Kumar, P.; Biradar, R.; Ratnamma, P.; et al. Simultaneous determination of persistent pesticide residues in pomegranate matrices using GC–MS/MS and its dietary risk assessment. Microchem. J. 2025, 218, 115193. [Google Scholar] [CrossRef]
- Chen, X.; Liu, X.; Dong, B.; Hu, J. Simultaneous determination of pyridaben, dinotefuran, DN and UF in eggplant ecosystem under open-field conditions: Dissipation behaviour and residue distribution. Chemosphere 2018, 195, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Van Der Voet, H.; Slob, W. Integration of Probabilistic Exposure Assessment and Probabilistic Hazard Characterization. Risk Anal. 2007, 27, 351–371. [Google Scholar] [CrossRef]
- Fan, Z.; Liu, F.; Ren, X.; Cheng, Y.; Wu, M.; Zhang, H.; Zhao, L.; Chen, Z. Large-scale fate tendency of lufenuron during cabbage cultivation: New insights into dietary risk and models comparison. Ecotoxicol. Environ. Saf. 2025, 303, 118824. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, C.; Xu, Z.; Wu, M.; Cheng, Y.; Zhao, L.; Chen, Z. Insights into the occurrence, distribution and dissipation of widespread agrochemicals in celery agrosystems for joint risk assessment. Environ. Res. 2024, 263, 120036. [Google Scholar] [CrossRef]
- Li, T.; Wu, X.; Zheng, L.; Cheng, Y.; Zhao, L.; Chen, Z. Quantitative tracing of typical herbicides and their metabolites in sorghum agrosystems for fate tendency and cumulative risk. Food Chem. 2025, 464, 141638. [Google Scholar] [CrossRef]

| Compounds | Matrix | Regression Equation | R2 | ME (%) | Spiked Levels (mg/kg) | Mean Recoveries (%) | Intraday RSD (%) | Interday RSD (%) | U (%) |
|---|---|---|---|---|---|---|---|---|---|
| DIF | MeCN | Y = 941,170X + 235 | 0.9957 | -- | -- | -- | |||
| Pomegranate aril | Y = 933,507X + 90.4 | 0.9964 | −0.8 | 0.02 | 90 | 4.4 | 3.6 | 5.7 | |
| 0.1 | 96 | 1.3 | 2.9 | 3.5 | |||||
| 1 | 93 | 4.5 | 5.7 | 7.7 | |||||
| Whole pomegranate | Y = 1,007,740X + 798 | 0.9969 | 8.0 | 0.02 | 99 | 1.8 | 1.9 | 2.7 | |
| 0.1 | 98 | 4.1 | 2.8 | 4.9 | |||||
| 1 | 93 | 1.6 | 8.3 | 9.7 | |||||
| 2,4,6-trichlorophenol (PRO) | Petroleum ether | Y = 49,349X − 3104 | 0.9983 | -- | -- | -- | |||
| Pomegranate aril | Y = 48,874x − 3428 | 0.9956 | −1.0 | 0.05 | 105 | 1.4 | 3.0 | 3.7 | |
| 0.5 | 108 | 0.9 | 7.1 | 8.2 | |||||
| 7 | 73 | 4.4 | 3.6 | 5.7 | |||||
| Whole pomegranate | Y = 48,723x − 3041 | 0.9967 | −1.3 | 0.05 | 91 | 5.2 | 8.5 | 10.9 | |
| 0.5 | 73 | 1.6 | 9.1 | 10.6 | |||||
| 7 | 80 | 3.5 | 8.7 | 10.5 | |||||
| SPI-A | MeCN | Y = 2,374,850X + 2235 | 0.9963 | -- | -- | -- | |||
| Pomegranate aril | Y = 2,311,220X + 1383 | 0.9966 | −2.7 | 0.02 | 88 | 6.7 | 5.8 | 9.0 | |
| 0.3 | 87 | 4.2 | 7.6 | 9.5 | |||||
| 1 | 96 | 0.6 | 3.9 | 4.5 | |||||
| Whole pomegranate | Y = 2,424,380X + 1811 | 0.9958 | 2.1 | 0.02 | 78 | 2.3 | 3.8 | 4.8 | |
| 0.3 | 82 | 2.6 | 3.6 | 4.8 | |||||
| 1 | 101 | 3.2 | 4.8 | 6.2 | |||||
| SPI-D | MeCN | Y = 4,472,690X + 27.5 | 0.9995 | -- | -- | -- | |||
| Pomegranate aril | Y = 4,392,520X − 45.8 | 0.9985 | −1.8 | 0.02 | 79 | 6.9 | 7.4 | 10.5 | |
| 0.3 | 85 | 5.7 | 2.5 | 5.9 | |||||
| 1 | 95 | 0.6 | 1.3 | 1.6 | |||||
| Whole pomegranate | Y = 4,595,450X + 1.91 | 0.9996 | 2.7 | 0.02 | 79 | 1.9 | 2.1 | 3.0 | |
| 0.3 | 78 | 5.0 | 2.4 | 5.3 | |||||
| 1 | 100 | 2.3 | 3.6 | 4.6 | |||||
| DIN | MeCN | Y = 548,810 X + 7618 | 0.9959 | -- | -- | -- | |||
| Pomegranate aril | Y = 514,009X + 9388 | 0.9956 | −6.3 | 0.02 | 91 | 12.0 | 9.2 | 15.1 | |
| 0.2 | 85 | 7.4 | 7.6 | 11.0 | |||||
| 0.4 | 85 | 7.1 | 7.3 | 10.6 | |||||
| Whole pomegranate | Y = 642,332X + 13,745 | 0.9906 | 17.0 | 0.02 | 99 | 5.1 | 4.8 | 7.2 | |
| 0.2 | 87 | 7.9 | 3.6 | 8.2 | |||||
| 0.4 | 77 | 7.6 | 9.1 | 12.5 | |||||
| UF | MeCN | Y = 228,605X + 37,826 | 0.9914 | -- | -- | -- | |||
| Pomegranate aril | Y = 221,594X + 38,766 | 0.9910 | −3.1 | 0.02 | 94 | 2.5 | 3.6 | 4.7 | |
| 0.2 | 81 | 1.2 | 2.7 | 3.3 | |||||
| 0.4 | 80 | 1.1 | 3.2 | 3.8 | |||||
| Whole pomegranate | Y =268,593.X + 49,573 | 0.9908 | 17.5 | 0.02 | 93 | 1.6 | 3.5 | 4.3 | |
| 0.2 | 82 | 2.7 | 2.4 | 3.7 | |||||
| 0.4 | 78 | 0.9 | 2.5 | 3.0 | |||||
| DN | MeCN | Y = 224,225X + 15,339 | 0.9975 | -- | -- | -- | |||
| Pomegranate aril | Y = 235,574X + 21,881 | 0.9969 | 5.1 | 0.02 | 98 | 4.5 | 9.8 | 12.0 | |
| 0.2 | 78 | 1.9 | 4.9 | 5.9 | |||||
| 0.4 | 86 | 2.4 | 5.2 | 6.4 | |||||
| Whole pomegranate | Y = 249,911X + 24,626 | 0.9965 | 11.5 | 0.02 | 97 | 4.7 | 9.3 | 11.5 | |
| 0.2 | 79 | 1.1 | 3.9 | 4.6 | |||||
| 0.4 | 84 | 3.1 | 4.0 | 5.4 |
| Dissipation patterns | Parameters | Trial #1 | Trial #2 | Trial #5 | |||
|---|---|---|---|---|---|---|---|
| PRO | DIF | PRO | DIF | PRO | DIF | ||
| SFO | DT50 (d) | 11.7 | 9.07 | 12.9 | 12.3 | 9.6 | 8.56 |
| R2 | 0.9932 | 0.9791 | 0.9394 | 0.9947 | 0.9549 | 0.9469 | |
| χ2 error (%) | 3.82 | 8.17 | 11.3 | 3.15 | 13.2 | 13.7 | |
| DFOP | DT50 (d) | 11.7 | 6.68 | 12.9 | 11.4 | 9.61 | 4.91 |
| R2 | 0.9932 | 0.9996 | 0.9395 | 0.9996 | 0.9549 | 0.9814 | |
| χ2 error (%) | 5.45 | 1.65 | 16.1 | 1.24 | 18.9 | 11.5 | |
| DT50 (d) | 11.7 | 7.1 | 12.8 | 11.3 | 9.6 | 6.1 | |
| FOMC | R2 | 0.9932 | 0.9967 | 0.9394 | 0.9992 | 0.9548 | 0.9691 |
| χ2 error (%) | 4.37 | 3.59 | 12.9 | 1.34 | 15.1 | 11.8 | |
| 7-day dissipation rate | 36 | 51 | 29 | 37 | 27 | 58 | |
| 14-day dissipation rate | 53 | 66 | 38 | 57 | 56 | 63 | |
| 21-day dissipation rate | 70 | 76 | 71 | 68 | 87 | 76 | |
| 28-day dissipation rate | 85 | 85 | 87 | 78 | 93 | 91 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Y.; Li, R.; Liu, T.; Li, R.; Fang, F.; Liang, H. Occurrence, Dissipation and Risk Assessment of Widespread Pesticides and Their Metabolites in Pomegranates. Foods 2025, 14, 3901. https://doi.org/10.3390/foods14223901
Zhu Y, Li R, Liu T, Li R, Fang F, Liang H. Occurrence, Dissipation and Risk Assessment of Widespread Pesticides and Their Metabolites in Pomegranates. Foods. 2025; 14(22):3901. https://doi.org/10.3390/foods14223901
Chicago/Turabian StyleZhu, Yuxiao, Rumei Li, Tongjin Liu, Ruijuan Li, Feng Fang, and Hui Liang. 2025. "Occurrence, Dissipation and Risk Assessment of Widespread Pesticides and Their Metabolites in Pomegranates" Foods 14, no. 22: 3901. https://doi.org/10.3390/foods14223901
APA StyleZhu, Y., Li, R., Liu, T., Li, R., Fang, F., & Liang, H. (2025). Occurrence, Dissipation and Risk Assessment of Widespread Pesticides and Their Metabolites in Pomegranates. Foods, 14(22), 3901. https://doi.org/10.3390/foods14223901





