First Evidence of the Potential of Postharvest Hesperidin Treatments: Effects on Strawberry Quality During Storage
Abstract
1. Introduction
2. Materials and Methods
2.1. Fruit Material and Experimental Conditions
2.2. Postharvest Quality Traits
2.3. Total Chlorophylls and Polyphenolic Content
2.4. Statistical Analysis
3. Results and Discussion
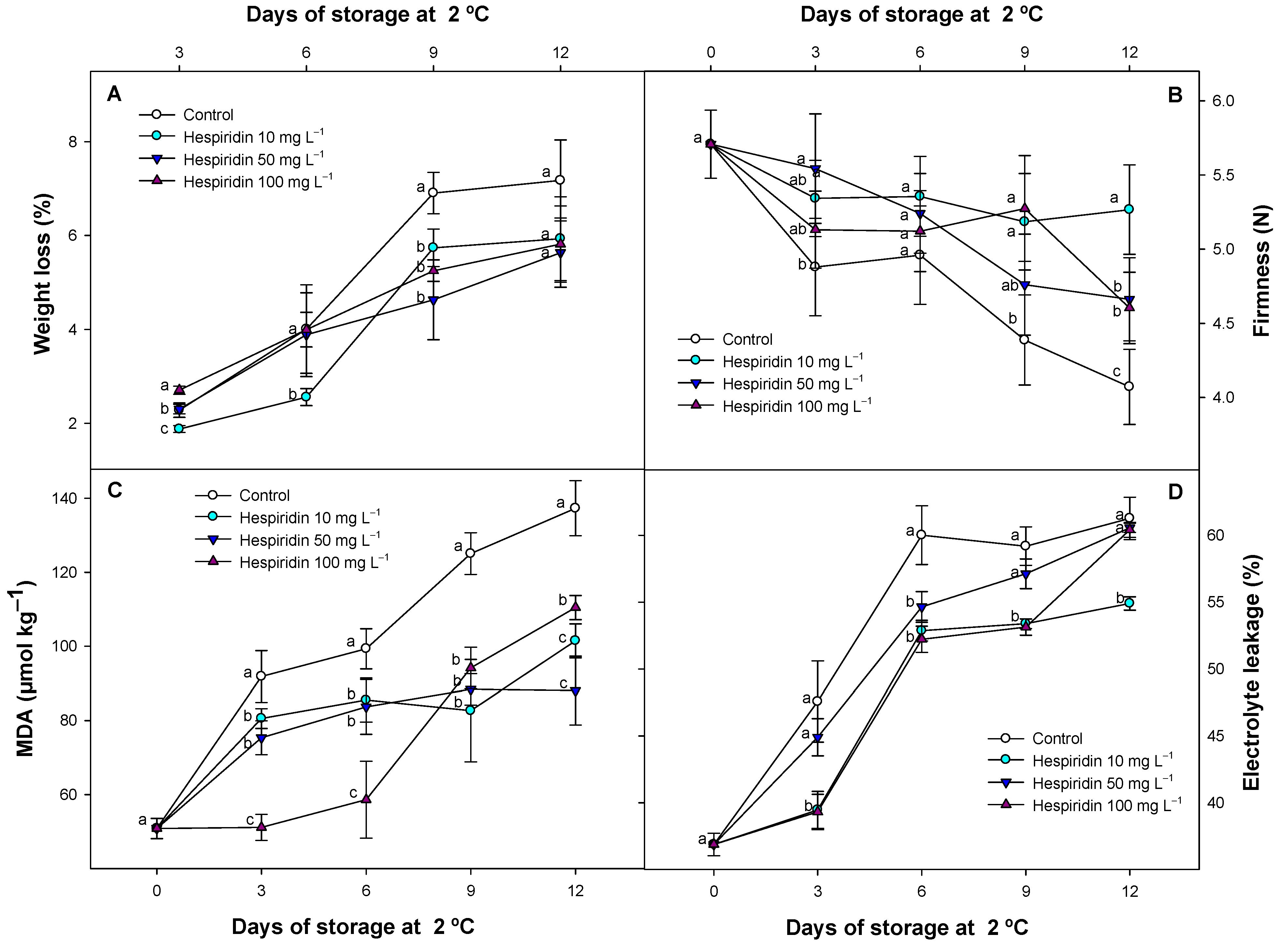
3.1. Effect of Hesperidin Treatments on Weight Loss, Fruit Firmness, MDA, and EL
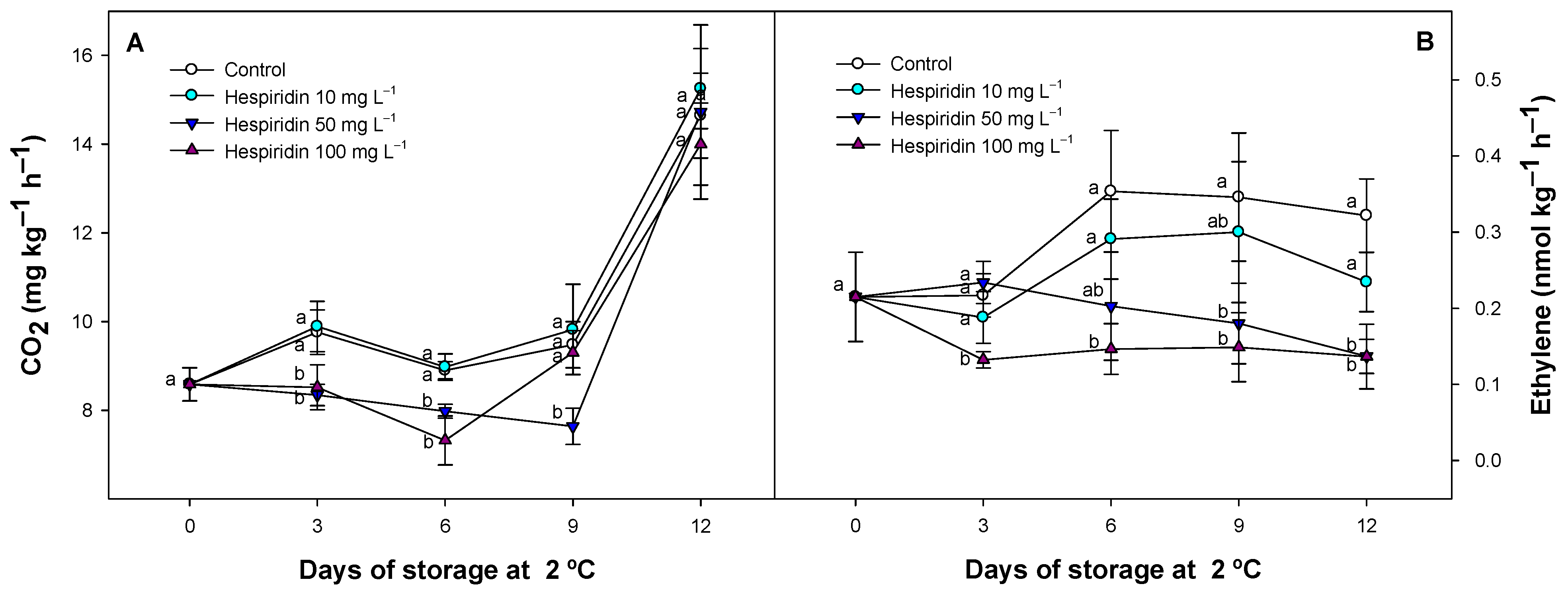
3.2. Effect of Hesperidin Treatments on Respiration and Ethylene Production
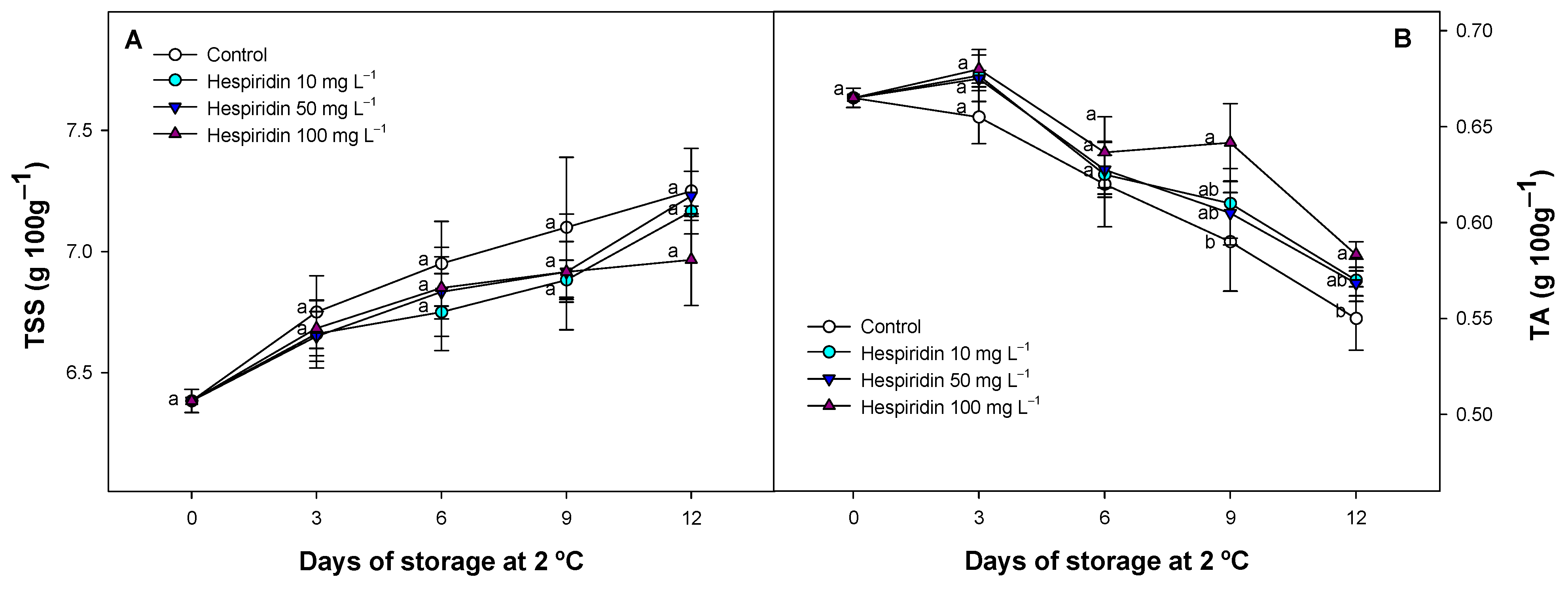
3.3. Effect of Hesperidin Treatments on TSS and TA
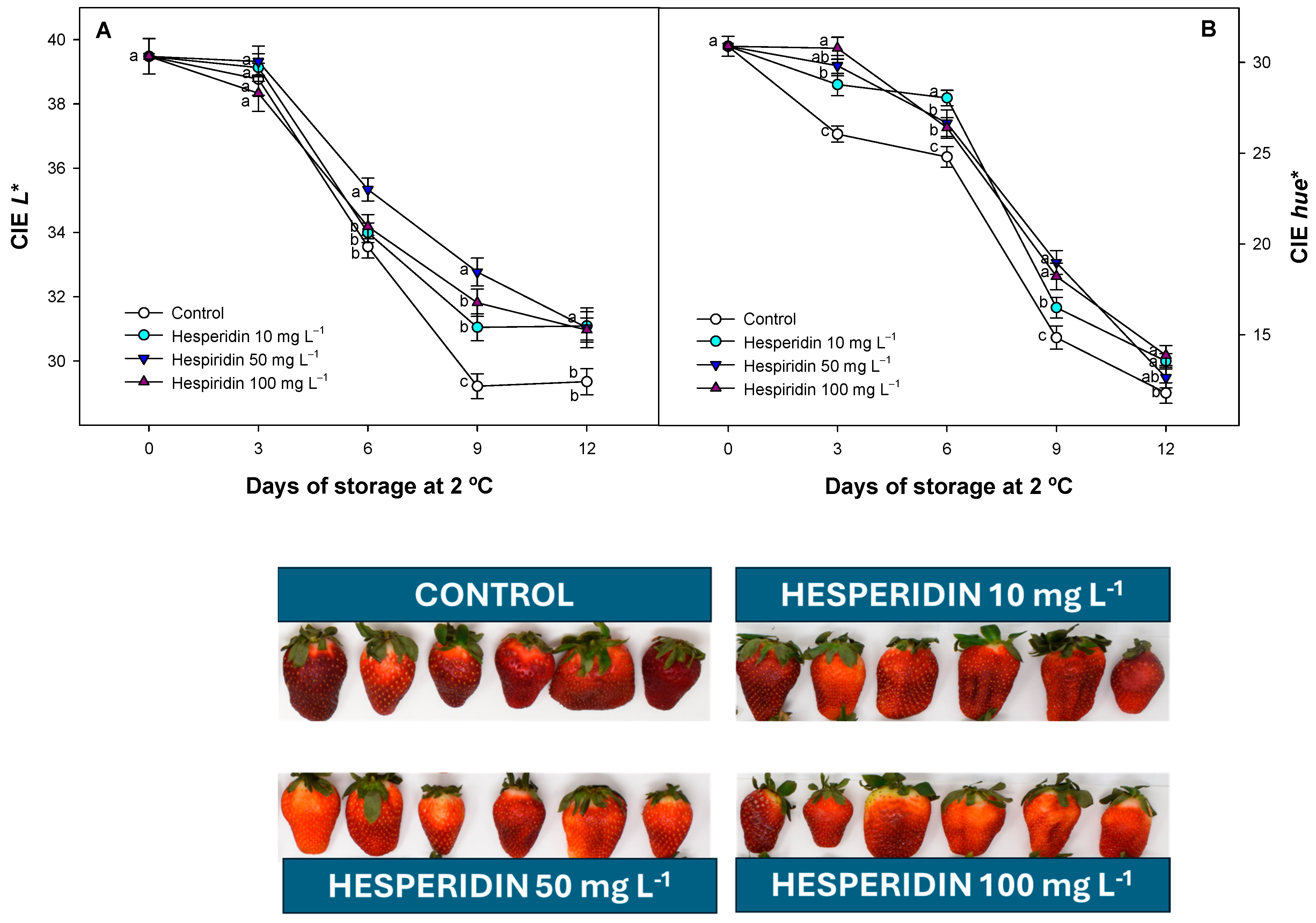
3.4. Effect of Hesperidin Treatments on Strawberry Colour Evolution
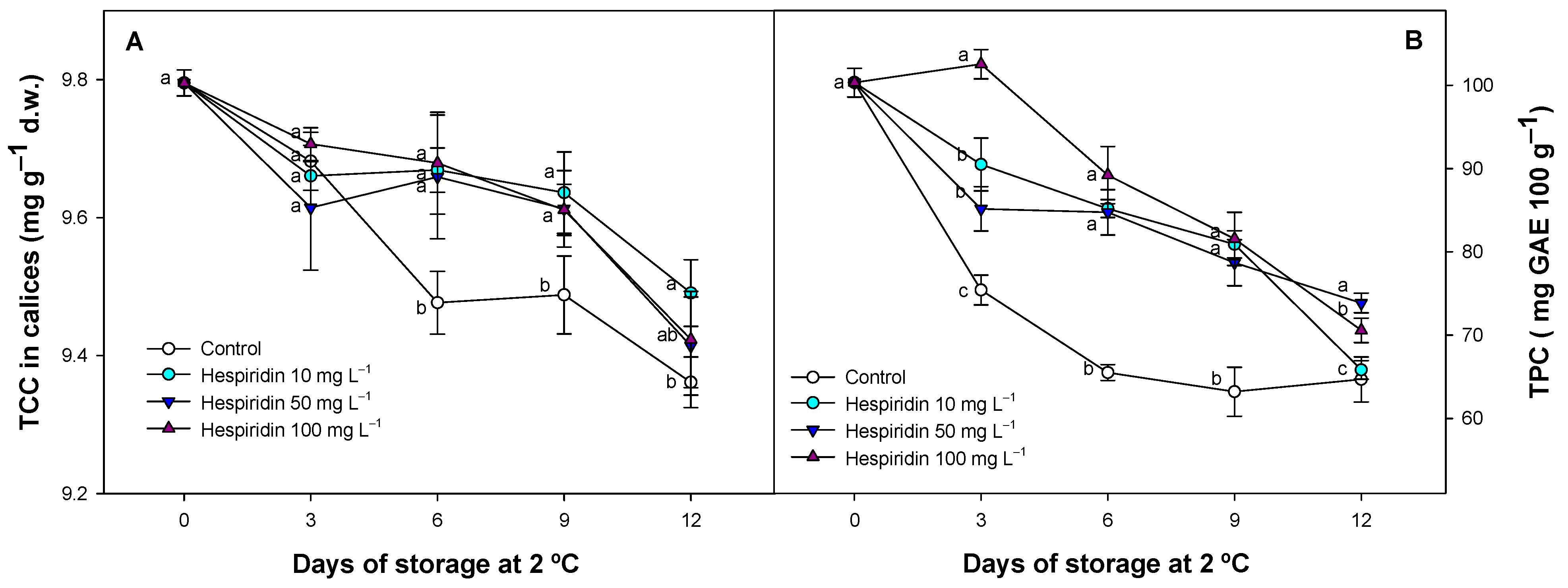
3.5. Effect of Hesperidin Treatments on TCC in Strawberry Calyces and TPC in the Fruit Flesh
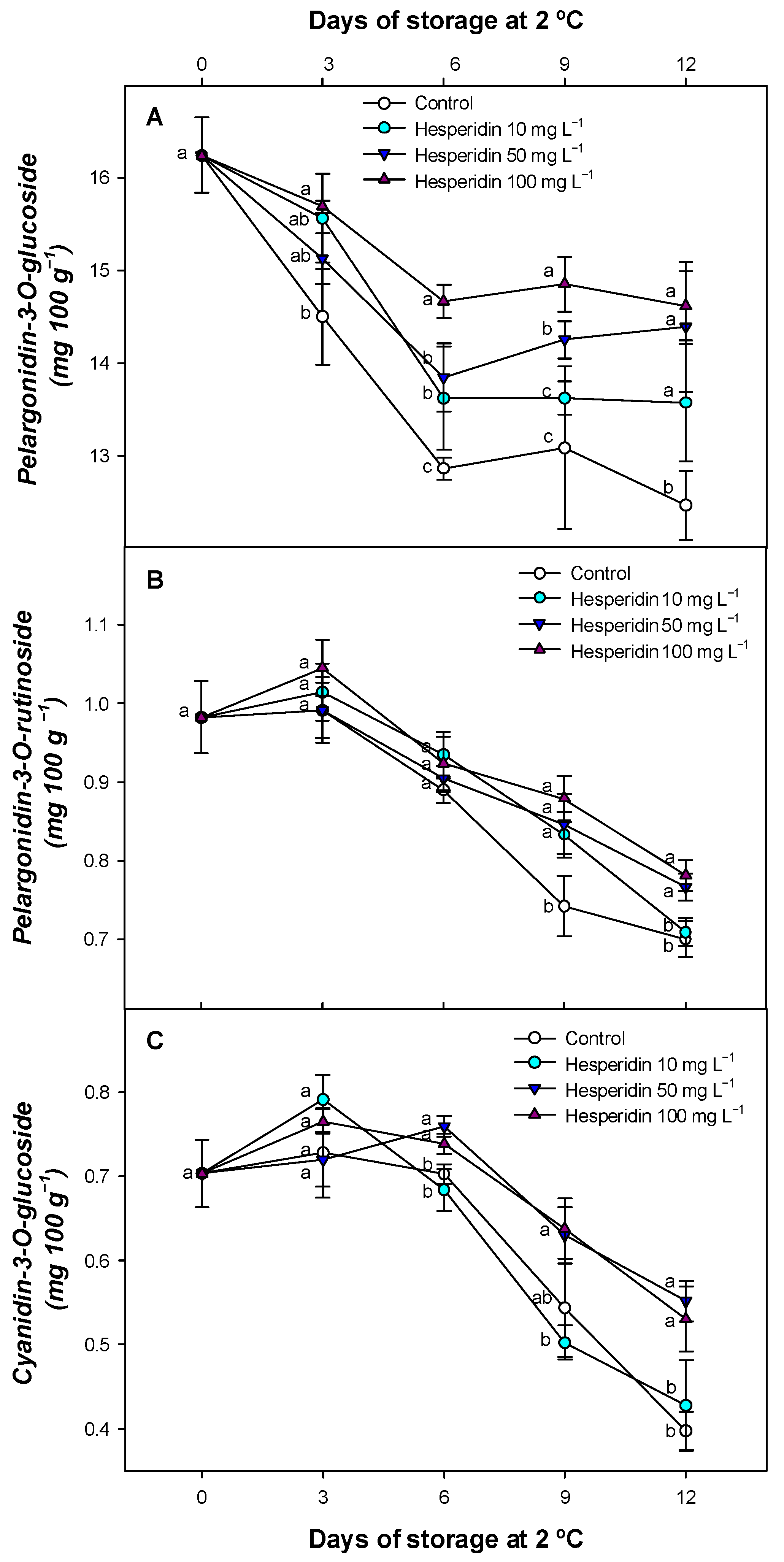
3.6. Effect of Hesperidin Treatments on Individual Anthocyanins During Storage
4. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ilea, M.I.M.; Díaz-Mula, H.M.; García-Molina, A.; Ruiz-Aracil, M.C.; Fernández-Picazo, C.; Guillén, F. Comparative effect of GABA and 1-MCP in maintaining strawberry fruit quality during cold storage. Horticulturae 2025, 11, 370. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Jayakumar, A.; de Souza, C.K.; Rhim, J.-W.; Kim, J.T. Advances in strawberry postharvest preservation and packaging: A comprehensive review. Compr. Rev. Food Sci. Food Saf. 2024, 23, e13417. [Google Scholar] [CrossRef]
- Wang, F.; Wang, J.; Ji, G.; Kang, X.; Li, Y.; Hu, J.; Qian, C.; Wang, S. Regulation of anthocyanins and quality in strawberries based on light quality. Horticulturae 2025, 11, 377. [Google Scholar] [CrossRef]
- Pinto, L.; Palma, A.; Cefola, M.; Pace, B.; D’Aquino, S.; Carboni, C.; Baruzzi, F. Effect of modified atmosphere packaging (MAP) and gaseous ozone pre-packaging treatment on the physico-chemical, microbiological and sensory quality of small berry fruit. Food Packag. Shelf Life 2020, 26, 100573. [Google Scholar] [CrossRef]
- Adiletta, G.; Di Matteo, M.; De Filippis, G.; Di Grazia, A.; Ciambelli, P.; Petriccione, M. Sustainable bamboo-based packaging and passive modified atmosphere: A strategy to preserve strawberry quality during cold storage. Processes 2025, 13, 2262. [Google Scholar] [CrossRef]
- Zhou, Z.; Yi, W.; Dong, T.; Song, L.; Pan, P.; Bai, J.; Yun, X. Enzyme-based respiration kinetics and MAP strategy for extending strawberry shelf life. Food Biosci. 2025, 71, 107169. [Google Scholar] [CrossRef]
- Salazar-Orbea, G.L.; García-Villalba, R.; Bernal, M.J.; Hernández, A.; Tomás-Barberán, F.A.; Sánchez-Siles, L.M. Stability of phenolic compounds in apple and strawberry: Effect of different processing techniques in industrial set up. Food Chem. 2023, 401, 134099. [Google Scholar] [CrossRef]
- Langer, S.E.; Marina, M.; Francese, P.; Civello, P.M.; Martínez, G.A.; Villarreal, N.M. New insights into the cell wall preservation by 1-methylcyclopropene treatment in harvest-ripe strawberry fruit. Sci. Hortic. 2022, 299, 111032. [Google Scholar] [CrossRef]
- Liu, C.; Zheng, H.; Sheng, K.; Liu, W.; Zheng, L. Effects of melatonin treatment on the postharvest quality of strawberry fruit. Postharvest Biol. Technol. 2018, 139, 47–55. [Google Scholar] [CrossRef]
- Manda-Hakki, K.; Hassanpour, H. Changes in postharvest quality and physiological attributes of strawberry fruits influenced by -L-phenylalanine. Food Sci. Nutr. 2024, 12, 10262–10274. [Google Scholar] [CrossRef]
- Kebriti, I.; Solgi, M.; Velashjerdi, M. Improving quality of strawberry by novel essential oil nanoemulsions of Echinophora platyloba combined with Aloe vera gel and gum arabic. Sci. Rep. 2025, 15, 1731. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, C.; Du, Y.; Liu, Q.; Yuan, S.; Yu, H.; Guo, Y.; Cheng, Y.; Qian, H.; Yao, W. Complex coacervation of ε-polylysine and glutenin: Phase behavior, interaction and as antimicrobial edible coatings on strawberry. Food Chem. 2025, 493, 145686. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Seo, J.; Kim, H.J.; Jeong, Y.; Lee, H.; Park, C.; Eom, Y. Synergistic design of Pickering emulsion inks of nanochitosan–alginate–beeswax for edible coating in fruit preservation. Food Hydrocoll. 2025, 167, 111462. [Google Scholar] [CrossRef]
- Abdalla, G.; Mussagy, C.U.; Brasil, G.S.P.; Scontri, M.; Sasaki, J.C.S.; Su, Y.; Bebber, C.; Rocha, R.R.; Abreu, A.P.S.; Goncalves, R.P.; et al. Eco-sustainable coatings based on chitosan, pectin, and lemon essential oil nanoemulsion and their effect on strawberry preservation. Int. J. Biol. Macromol. 2023, 249, 126016. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, M.; Fancello, F.; Zara, S.; Pace, B.; Ricci, I.; Quintieri, L.; Lippolis, V.; Forleo, T.; Sorrentino, A.; Oliviero, M.; et al. Citral-releasing active patches preserve the overall quality and extend strawberry shelf-life. Appl. Food Res. 2025, 5, 100903. [Google Scholar] [CrossRef]
- Maslov Bandić, L.; Vuković, M.; Gavranić, D.; Tanuwidjaja, I.; Mrkonjić Fuka, M.; Duralija, B.; Jurić, S. Elevating the bioactive potential of strawberries with apple pomace extract-infused chitosan edible coating. Pol. J. Food Nutr. Sci. 2025, 75, 24–36. [Google Scholar] [CrossRef]
- Zantar, Y.; Noutfia, Y.; Laglaoui, A.; Maurady, A.; Alfeddy, M.N.; El Abbassi, N.; Hassani Zerrouk, M.; Zantar, S. Impact of a coating of pectin and Cinnamomum zeylanicum essential oil on the postharvest quality of strawberries packaged under different modified atmosphere conditions. Discov. Food 2025, 5, 11. [Google Scholar] [CrossRef]
- Ilea, M.I.M.; Zapata, P.J.; Fernández-Picazo, C.; Díaz-Mula, H.M.; Castillo, S.; Guillén, F. Chlorogenic acid as a promising tool for mitigating chilling injury: Cold tolerance and the ripening effect on tomato fruit (Solanum lycopersicum L.). Plants 2024, 13, 2055. [Google Scholar] [CrossRef]
- Jayamali, N.A.I.; Wijesinghe, W.A.J.P.; Silva, P.A.P.M.D. Green tea incorporated edible coating extends the postharvest life of strawberry fruits (Fragaria × ananassa). Adv. Technol. 2022, 2, 382–393. [Google Scholar] [CrossRef]
- Karabekir, S.Ç.; Sözen, M.E.; Ayan, I.Ç.; Savaş, H.B.; Cüce, G.; Kalkan, S. The ameliorative effects of hesperidin in rats developed hepatotoxicity with deltamethrin. Iran. J. Basic Med. Sci. 2025, 28, 929–936. [Google Scholar] [CrossRef]
- Sun, K.X.; Tan, W.S.; Wang, H.Y.; Gao, J.M.; Wang, S.Y.; Xie, M.L.; Deng, W.L. Hesperidin suppressed colorectal cancer through inhibition of glycolysis. Chin. J. Integr. Med. 2025, 31, 529–540. [Google Scholar] [CrossRef]
- Ziyadanoğullari, M.O.; Tuncer, M.C.; Özdemir, I. Antitumor effects of hesperidin and cisplatin on human osteosarcoma cells through inhibiting proliferation and inducing mitochondrial-mediated apoptosis. Medicina 2025, 61, 960. [Google Scholar] [CrossRef]
- He, J.; Zhou, Z.; Zhu, W.; Chen, X.; Zhu, S.; Sun, J.; Zuo, Y.; Yang, M.; Yu, M.; Yang, L.; et al. Hesperidin derivatives contained hydrogel dressing for photothermal treatment of melanoma and postoperative tissue regeneration. ACS Appl. Bio Mater. 2025, 8, 5321–5336. [Google Scholar] [CrossRef]
- Shaernejad, S.; Nosrat, A.; Baeeri, M.; Hashemi Goradel, N.; SeyedSadeghi, M.; Akbariani, M.; Arabzadeh, A.; Rahimifard, M.; Haghi-Aminjan, H. Role of hesperidin/hesperetin against chemotherapy-induced cardiotoxicity: A systematic review of non-clinical studies. Cancer Cell Int. 2025, 25, 186. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, H.S.; Hosny, E.N.; Sawie, H.G.; Khadrawy, Y.A. Assessment of the therapeutic potential of hesperidin and quercetin nanoparticles on oxidative stress, enzyme activities, trophic factors, and monoamines in an animal model of depression. Neurochem. Res. 2025, 50, 203. [Google Scholar] [CrossRef] [PubMed]
- Asif, F.; Mahmood, H.; Jiangtao, L.; Ce, S.; Haiying, C.; Yuxiao, W.; Rentang, Z.; Yaqoob, S.; Jianbo, X.; Lin, L.; et al. Development of eco-friendly chitosan films incorporated with pomelo peel (Citrus paradisi cv. changshanhuyou) extract and application to prolong the shelf life of grapes. Int. J. Biol. Macromol. 2025, 304, 140547. [Google Scholar] [CrossRef] [PubMed]
- Hassan, D.; Sani, A. Chitosan films developed using all-natural resources for fruit preservation and the impact of lemon peel extract mediated nickel ferrite nanoparticles on films’ physical and barrier properties. Food Chem. 2025, 482, 144068. [Google Scholar] [CrossRef]
- Hasanin, M.S.; Emam, M.; Ahmed, M.A.; Rohim, F.M.; Mohamed, M.A.A.; El Saied, H.; Hussein, H.A.Z.; Abdelkhalek, A. Bioactive preservative nano-packaging films based on food wastes of orange peels and shrimp for apple (Malus domestica var. anna) fruit quality and storage. Bioresour. Bioprocess. 2025, 12, 54. [Google Scholar] [CrossRef]
- Patidar, S.; Kumari, P.; Singh, A.; Shahi, N.C.; Chand, K.; Hussain, A.; Ahmad, W.; Lohani, U.C.; Kumar, S. Formulation and optimization of plant-based organic coating with citrus peel extract for enhancing shelf life and quality of postharvest litchi fruit. J. Food Meas. Charact. 2025, 19, 2350–2365. [Google Scholar] [CrossRef]
- Ningrum, A.; Bagus, H.; Santoso, U.; Munawaroh, H.S.H.; Susanto, E.; Yilmaz, B.; Show, P.L. Effect of edible coating from gelatin-κ-carrageenan composites with the addition of orange peel extract (Citrus sinensis (L.) Osbeck) on the quality attributes of tomato (Solanum lycopersicum). J. Food Meas. Charact. 2025, 19, 4062–4084. [Google Scholar] [CrossRef]
- Van, T.T.; Tanaka, F.; Wardak, M.H.; Jothi, J.S.; Phuong, N.T.H.; Yan, X.; Zdunek, A.; Tanaka, F. Effect of coatings containing 1-methylcyclopropane or mandarin peel extract on the freshness and metabolic profiles of cold stored strawberry. Food Chem. 2024, 461, 140819. [Google Scholar] [CrossRef] [PubMed]
- Razola-Díaz, M.d.C.; Guerra-Hernández, E.J.; García-Villanova, B.; Verardo, V. Recent developments in extraction and encapsulation techniques of orange essential oil. Food Chem. 2021, 354, 129575. [Google Scholar] [CrossRef] [PubMed]
- Pyrzynska, K. Hesperidin: A review on extraction methods, stability and biological activities. Nutrients 2022, 14, 2387. [Google Scholar] [CrossRef] [PubMed]
- Bertolo, M.R.V.; Pereira, T.S.; Dos Santos, F.V.; Facure, M.H.M.; Dos Santos, F.; Teodoro, K.B.R.; Mercante, L.A.; Correa, D.S. Citrus wastes as sustainable materials for active and intelligent food packaging: Current advances. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70144. [Google Scholar] [CrossRef]
- Martínez-Romero, D.; Serrano, M.; Carbonell, A.; Burgos, L.; Riquelme, F.; Valero, D. Effects of postharvest putrescine treatment on extending shelf life and reducing mechanical damage in apricot. J. Food Sci. 2002, 67, 1706–1712. [Google Scholar] [CrossRef]
- Vu, H.T.; Scarlett, C.J.; Vuong, Q.V. Changes of phytochemicals and antioxidant capacity of banana peel during the ripening process; with and without ethylene treatment. Sci. Hortic. 2019, 253, 255–262. [Google Scholar] [CrossRef]
- Lezoul, N.E.H.; Belkadi, M.; Habibi, F.; Guillén, F. Extraction processes with several solvents on total bioactive compounds in different organs of three medicinal plants. Molecules 2020, 25, 4672. [Google Scholar] [CrossRef]
- del Olmo, I.; Romero, I.; Álvarez, M.D.; Tarradas, R.; Sánchez-Ballesta, M.T.; Escribano, M.I.; Merodio, C. Transcriptomic analysis of CO2-treated strawberries (Fragaria vesca) with enhanced resistance to softening and oxidative stress at consumption. Front. Plant Sci. 2022, 13, 983976. [Google Scholar] [CrossRef]
- Valero, D.; Serrano, M. Postharvest Biology and Technology for Preserving Fruit Quality, 1st ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar] [CrossRef]
- Zhang, L.; Li, S.; Dong, Y.; Zhi, H.; Zong, W. Tea polyphenols incorporated into alginate-based edible coating for quality maintenance of chinese winter jujube under ambient temperature. LWT 2016, 70, 155–161. [Google Scholar] [CrossRef]
- Bai, X.Y.; Yang, Z.M.; Shen, W.J.; Shao, Y.Z.; Zeng, J.K.; Li, W. Polyphenol treatment delays the browning of litchi pericarps and promotes the total antioxidant capacity of litchi fruit. Sci. Hortic. 2022, 291, 110563. [Google Scholar] [CrossRef]
- Strano, M.C.; Altieri, G.; Allegra, M.; Di Renzo, G.C.; Paterna, G.; Matera, A.; Genovese, F. Postharvest technologies of fresh citrus fruit: Advances and recent developments for the loss reduction during handling and storage. Horticulturae 2022, 8, 612. [Google Scholar] [CrossRef]
- Merchante, C.; Vallarino, J.G.; Aragüez, I.; Villarreal, N.; Ariza, M.T.; Martínez, G.A.; Medina-Escobar, N.; Civello, M.P.; Fernie, A.R.; Botella, M.A.; et al. Ethylene is involved in strawberry fruit ripening in an organ-specific manner. J. Exp. Bot. 2013, 64, 4421–4439. [Google Scholar] [CrossRef] [PubMed]
- Aghdam, M.S.; Jannatizadeh, A.; Luo, Z.; Paliyath, G. Ensuring sufficient intracellular ATP supply and friendly extracellular ATP signalling attenuates stresses, delays senescence, and maintains quality in horticultural crops during postharvest life. Trends Food Sci. Technol. 2018, 76, 67–81. [Google Scholar] [CrossRef]
- Ansari, M.I.; Jalil, S.U.; Ansari, S.A.; Hasanuzzaman, M. GABA Shunt: A key-player in mitigation of ros during stress. Plant Growth Regul. 2021, 94, 131–149. [Google Scholar] [CrossRef]
- Zhang, H.; Li, K.; Zhang, X.; Dong, C.; Ji, H.; Ke, R.; Ban, Z.; Hu, Y.; Lin, S.; Chen, C. Effects of ozone treatment on the antioxidant capacity of postharvest strawberry. RSC Adv. 2020, 10, 38142–38157. [Google Scholar] [CrossRef]
- Cheng, Z.; Li, R.; Jiang, Z.; Li, W.; Shao, Y. Combined effect of Bacillus siamensis and chlorogenic acid on maintenance of quality and control of disease in stored wax apple fruit. Food Qual. Saf. 2022, 6, 475–486. [Google Scholar] [CrossRef]
- Kumar, S.; Shukla, P.; Das, K.; Katiyar, V. Chitosan/water caltrop pericarp extract reinforced active edible film and its efficacy as strawberry coating for prolonging shelf life. Int. J. Biol. Macromol. 2025, 307, 142115. [Google Scholar] [CrossRef]
- Tian, S.; Qin, G.; Li, B. Reactive oxygen species involved in regulating fruit senescence and fungal pathogenicity. Plant Mol. Biol. 2013, 82, 593–602. [Google Scholar] [CrossRef]
- Martínez-González, M.d.C.; Bautista-Baños, S.; Correa-Pacheco, Z.N.; Corona-Rangel, M.L.; Ventura-Aguilar, R.I.; Del Río-García, J.C.; Ramos-García, M.d.L. Effect of nanostructured chitosan/propolis coatings on the quality and antioxidant capacity of strawberries during storage. Coatings 2020, 10, 90. [Google Scholar] [CrossRef]
- Bertolo, M.R.V.; de Oliveira Filho, J.G.; Lamonica, G.C.; de Oliveira Nobre Bezerra, C.C.; da Conceição Amaro Martins, V.; Ferreira, M.D.; de Guzzi Plepis, A.M.; Bogusz Junior, S. Improvement of the physical-chemical, microbiological, volatiles and sensory quality of strawberries covered with chitosan/gelatin/pomegranate peel extract-based coatings. Food Chem. 2025, 471, 142755. [Google Scholar] [CrossRef]
- Fan, P.; Huber, D.J.; Su, Z.; Hu, M.; Gao, Z.; Li, M.; Shi, X.; Zhang, Z. Effect of postharvest spray of apple polyphenols on the quality of fresh-cut red pitaya fruit during shelf life. Food Chem. 2018, 243, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Jiao, W.; Cao, J.; Jiang, W. Effects of chlorogenic acid on the capacity of free radical scavenging and proteomic changes in postharvest fruit of nectarine. PLoS ONE 2017, 12, e0182494. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhang, X.; Li, L.; Aghdam, M.S.; Wei, X.; Liu, J.; Xu, Y.; Luo, Z. Elevated CO2 delayed the chlorophyll degradation and anthocyanin accumulation in postharvest strawberry fruit. Food Chem. 2019, 285, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lin, B.; Tang, G.; Chen, Y.; Deng, M.; Lin, Y.; Li, M.; He, W.; Wang, Y.; Zhang, Y.; et al. Application of γ-aminobutyric acid improves the postharvest marketability of strawberry by maintaining fruit quality and enhancing antioxidant system. Food Chem. X 2024, 21, 101252. [Google Scholar] [CrossRef]
- Watkins, C.B. The use of 1-methylcyclopropene (1-MCP) on fruits and vegetables. Biotechnol. Adv. 2006, 24, 389–409. [Google Scholar] [CrossRef]
- Azam, M.; Ejaz, S.; Rehman, R.N.U.; Khan, M.; Qadri, R. Postharvest Quality Management of Strawberries. In Strawberry Pre- and Post-Harvest Management Techniques for Higher Fruit Quality; Asao, T., Asaduzzaman, M., Eds.; IntechOpen: London, UK, 2019; p. 22. [Google Scholar] [CrossRef]
- Babarabie, M.; Zarei, H.; Eskandari, A. The impact of pre-harvest treatment with gamma-aminobutyric acid (GABA) and salicylic acid on vase life and post-harvest traits of tuberose cut flowers. Acta Sci. Pol. Hortorum Cultus 2019, 18, 83–92. [Google Scholar] [CrossRef]
- Lezoul, N.E.H.; Serrano, M.; Ruiz-Aracil, M.C.; Belkadi, M.; Castillo, S.; Valero, D.; Guillén, F. Melatonin as a new postharvest treatment for increasing cut carnation (Dianthus caryophyllus L.) vase life. Postharvest Biol. Technol. 2022, 184, 111759. [Google Scholar] [CrossRef]
- Li, D.; Zhang, X.; Xu, Y.; Aghdam, M.S.; Luo, Z. Effect of exogenous sucrose on anthocyanin synthesis in postharvest strawberry fruit. Foods 2019, 289, 112–120. [Google Scholar] [CrossRef]
- Haynes, B.; Fernandez, G.; Ma, G.; Chen, H.; Perkins-Veazie, P. Strawberry germplasm influences fruit physicochemical composition more than harvest date or location. Horticulturae 2025, 11, 55. [Google Scholar] [CrossRef]
- Chaves, V.C.; Calvete, E.; Reginatto, F.H. Quality properties and antioxidant activity of seven strawberry (Fragaria × ananassa Duch) cultivars. Sci. Hortic. 2017, 225, 293–298. [Google Scholar] [CrossRef]
- Octavia, L.; Choo, W.S. Folate, ascorbic acid, anthocyanin and colour changes in strawberry (Fragaria × ananassa) during refrigerated storage. LWT 2017, 86, 652–659. [Google Scholar] [CrossRef]
- Riaz, A.; Aadil, R.M.; Amoussa, A.M.O.; Bashari, M.; Abid, M.B.M.; Hashim, M.M. Application of chitosan-based apple peel polyphenols edible coating on the preservation of strawberry (Fragaria ananassa cv. Hongyan) fruit. J. Food Process. Preserv. 2021, 45, e15018. [Google Scholar] [CrossRef]
- Benavides, S.; Franco, W. Innovative integration of arrayan (Luma apiculata) extracts in chitosan coating for fresh strawberry preservation. Int. J. Mol. Sci. 2023, 24, 14681. [Google Scholar] [CrossRef]
- Kaynarca, G.B.; Kamer, D.D.A.; Yucel, E.; Yılmaz, O.S.; Henden, Y.; Kaymaz, E.; Gumus, T. The potential of pectin-based films enriched with bioactive components for strawberry preservation: A sustainable and innovative coating. Sci. Hortic. 2024, 334, 113294. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ilea, M.I.M.; Díaz-Mula, H.M.; Fernández-Picazo, C.; Zapata, P.J.; Dobón-Suárez, A.; Castillo, S.; Guillén, F. First Evidence of the Potential of Postharvest Hesperidin Treatments: Effects on Strawberry Quality During Storage. Foods 2025, 14, 2837. https://doi.org/10.3390/foods14162837
Ilea MIM, Díaz-Mula HM, Fernández-Picazo C, Zapata PJ, Dobón-Suárez A, Castillo S, Guillén F. First Evidence of the Potential of Postharvest Hesperidin Treatments: Effects on Strawberry Quality During Storage. Foods. 2025; 14(16):2837. https://doi.org/10.3390/foods14162837
Chicago/Turabian StyleIlea, Mihaela Iasmina Madalina, Huertas María Díaz-Mula, Christian Fernández-Picazo, Pedro Javier Zapata, Alicia Dobón-Suárez, Salvador Castillo, and Fabián Guillén. 2025. "First Evidence of the Potential of Postharvest Hesperidin Treatments: Effects on Strawberry Quality During Storage" Foods 14, no. 16: 2837. https://doi.org/10.3390/foods14162837
APA StyleIlea, M. I. M., Díaz-Mula, H. M., Fernández-Picazo, C., Zapata, P. J., Dobón-Suárez, A., Castillo, S., & Guillén, F. (2025). First Evidence of the Potential of Postharvest Hesperidin Treatments: Effects on Strawberry Quality During Storage. Foods, 14(16), 2837. https://doi.org/10.3390/foods14162837









