Sustainable Extraction and Multimodal Characterization of Fungal Chitosan from Agaricus bisporus
Abstract
1. Introduction
2. Materials and Physical–Chemical Characterization
2.1. Sample Processing
2.2. Chitin Extraction
2.2.1. Demineralization
2.2.2. Deproteinization
2.3. Chitin Deacetylation
2.4. Characterization Analysis
2.4.1. X-Ray Diffraction (illustratedXRD)
- I002 = Maximum diffraction intensity;
- Iam = Amorphous diffraction intensity.
2.4.2. Scanning Electron Microscopy (SEM)
2.4.3. Fourier Transform Infrared Spectroscopy (FTIR) and Degree of Deacetylation
2.4.4. Thermogravimetric Analysis (TGA)
2.4.5. Zeta Potential
2.4.6. Color Analysis
2.4.7. Statistical Analysis
3. Results and Discussion
3.1. Yield
3.2. X-Ray Diffraction (XRD) Data
3.3. Scanning Electron Microscopy Data (SEM)
3.4. FT-IR Data
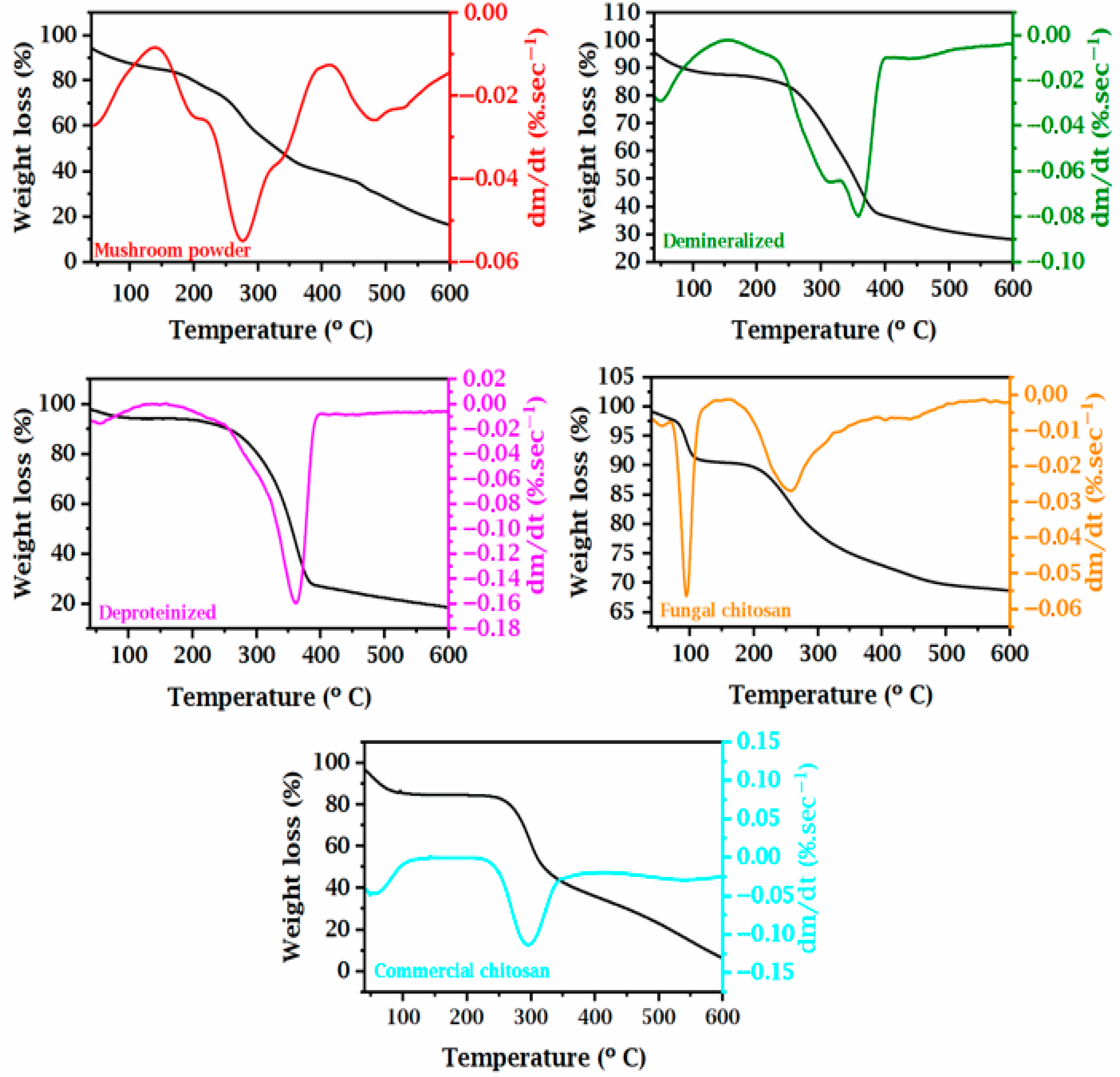
3.5. Thermogravimetric (TGA) Data
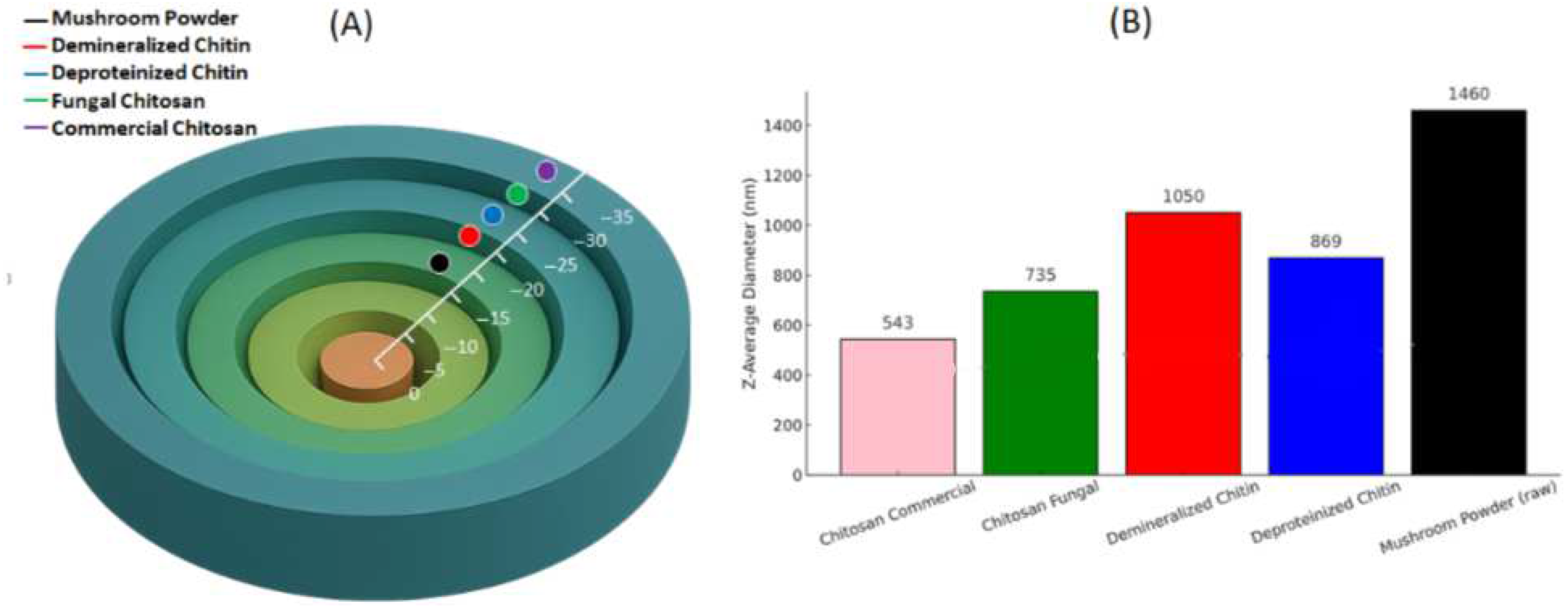
3.6. Zeta Potential Data
3.7. Color Analysis Data
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Batista, A.C.L.; Bandeira, M.G.L.; Souza Neto, F.E.D.; Paiva, W.D.S.; Rodrigues, D.N.R.; Costa, A.C.A.D.A. Obtenção de Quitosana Fúngica Crescida em Meio Alternativo Constituído com Farinha de Carapaça de Camarão. Available online: https://rsc.revistas.ufcg.edu.br/index.php/rsc/article/view/309 (accessed on 21 April 2025).
- Wang, H.; Roman, M.; Wang, H.; Roman, M. Effects of Chitosan Molecular Weight and Degree of Deacetylation on Chitosan−Cellulose Nanocrystal Complexes and Their Formation. Molecules 2023, 28, 1361. [Google Scholar] [CrossRef]
- Huq, T.; Khan, A.; Brown, D.; Dhayagude, N.; He, Z.; Ni, Y. Sources, Production, and Commercial Applications of Fungal Chitosan: A Review. J. Bioresour. Bioprod. 2022, 7, 85–98. [Google Scholar] [CrossRef]
- Islam, N.; Hoque, M.; Taharat, S.F. Recent advances in extraction of chitin and chitosan. World J. Microbiol. Biotechnol. 2023, 39, 28. [Google Scholar] [CrossRef]
- Alimi, B.A.; Pathania, S.; Wilson, J.; Duffy, B.; Frias, J.M.C. Extraction, quantification, characterization, and application in food packaging of chitin and chitosan from mushrooms: A review. Int. J. Biol. Macromol. 2023, 237, 124195. [Google Scholar] [CrossRef]
- Rebouillat, S.; Pla, F. A Review: On Smart Materials Based on Some Polysaccharides; within the Contextual Bigger Data, Insiders, “Improvisation” and Said Artificial Intelligence Trends. J. Biomater. Nanobiotechnol. 2019, 10, 41–77. [Google Scholar] [CrossRef]
- Ozel, N.; Elibol, M. Chitin and chitosan from mushroom (Agaricus bisporus) using deep eutectic solvents. Int. J. Biol. Macromol. 2024, 262, 130110. [Google Scholar] [CrossRef] [PubMed]
- Hassainia, A.; Satha, H.; Boufi, S. Chitin from Agaricus bisporus: Extraction and characterization. Int. J. Biol. Macromol. 2018, 117, 1334–1342. [Google Scholar] [CrossRef] [PubMed]
- Del Rocío Urbina-Salazar, A.; Inca-Torres, A.R.; Aguilar, M.D.P.C.; Palomas, J.B. Preparación de quitina fúngica a partir de subproductos de hongos comestibles (Agaricus bisporus). Polo Del Conoc. Rev. Científico-Prof. 2020, 5, 115–140. [Google Scholar]
- Zargar, V.; Asghari, M.; Dashti, A. A Review on Chitin and Chitosan Polymers: Structure, Chemistry, Solubility, Derivatives, and Applications. ChemBioEng Rev. 2015, 2, 204–226. [Google Scholar] [CrossRef]
- Yen, M.-T.; Mau, J.-L. Physico-chemical characterization of fungal chitosan from shiitake stipes. LWT—Food Sci. Technol. 2007, 40, 472–479. [Google Scholar] [CrossRef]
- de Moura Rodrigues, G.; Okura, M.H. Avalição da composição centesimal e temperatura de secagem de cogumelos Shiitake. Res. Soc. Dev. 2022, 11, e594111134144. [Google Scholar] [CrossRef]
- AOAC. Official Method 930.04—Moisture in Dried Fruits and Vegetables. In Official Methods of Analysis of the AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- AOAC. Official Method 981.12—pH of Fruit Products. In Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Erdogan, S.; Kaya, M.; Akata, I. Chitin extraction and chitosan production from cell wall of two mushroom species (Lactarius vellereus and Phyllophora ribis). AIP Conf. Proc. 2017, 1809, 020012. [Google Scholar] [CrossRef]
- Ospina Álvarez, S.P.; Ramírez Cadavid, D.A.; Escobar Sierra, D.M.; Ossa Orozco, C.P.; Rojas Vahos, D.F.; Zapata Ocampo, P.; Atehortúa, L. Comparison of Extraction Methods of Chitin from Ganoderma lucidum Mushroom Obtained in Submerged Culture. BioMed Res. Int. 2014, 2014, 169071. [Google Scholar] [CrossRef]
- Liao, J.; Huang, H. A fungal chitin derived from Hericium erinaceus residue: Dissolution, gelation and characterization. Int. J. Biol. Macromol. 2020, 152, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Segal, L.; Creely, J.J.; Martin, A.E.; Conrad, C.M. An Empirical Method for Estimating the Degree of Crystallinity of Native Cellulose Using the X-Ray Diffractometer. Text. Res. J. 1959, 29, 786–794. [Google Scholar] [CrossRef]
- Kim, H.; Kang, S.; Li, K.; Jung, D.; Park, K.; Lee, J. Preparation and characterization of various chitin-glucan complexes derived from white button mushroom using a deep eutectic solvent-based ecofriendly method. Int. J. Biol. Macromol. 2021, 169, 122–129. [Google Scholar] [CrossRef]
- Moriana, R.; Vilaplana, F.; Ek, M. Cellulose Nanocrystals from Forest Residues as Reinforcing Agents for Composites: A Study from Macro- to Nano-Dimensions. Carbohydr. Polym. 2016, 139, 139–149. [Google Scholar] [CrossRef] [PubMed]
- de Souza Soares, L.; Perim, R.B.; de Alvarenga, E.S.; de Moura Guimarães, L.; de Carvalho Teixeira, A.V.N.; dos Reis Coimbra, J.S.; de Oliveira, E.B. Insights on physicochemical aspects of chitosan dispersion in aqueous solutions of acetic, glycolic, propionic or lactic acid. Int. J. Biol. Macromol. 2019, 128, 140–148. [Google Scholar] [CrossRef]
- Sousa, M.M.D.; Soares, N.D.F.F.; Silva, D.D.J.; Coimbra, J.S.D.R. Desenvolvimento De Nanocompósito Biodegradável Ativo Incorporado Com Nanocristais De Celulose Modificados E Com Lisozima. Master’s Thesis, Universidade Federal de Viçosa, Viços, Brazil, 2020. [Google Scholar]
- Souza Neto, F.E.D. Caracterização e Avaliação do Potencial da Quitosana Extraída de Cunninghamella Elegans na Formação de Cristais de Oxalato de cálcio e Atividade Antioxidante. Master’s Thesis, Universidade Federal Rural do Semi-Árido, Mossoró, Brazil, 2017. [Google Scholar]
- Paiva, W.D.S. Quitosana Fúngica na Produção de Biomaterial Membranoso Modificado por Plasma de Descarga em Barreira Dielétrica (DBD). Master’s Thesis, Universidade Federal Rural do Semi-Árido, Mossoró, Brazil, 2017. [Google Scholar]
- Espitia, P.J.P.; Soares, N.D.F.F.; Teófilo, R.F.; dos Reis Coimbra, J.S.; Vitor, D.M.; Batista, R.A.; Ferreira, S.O.; de Andrade, N.J.; Medeiros, E.A.A. Physical–mechanical and antimicrobial properties of nanocomposite films with pediocin and ZnO nanoparticles. Carbohydr. Polym. 2013, 94, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Gil, I.M. Cultivo Submerso do Fungo Hericium Erinaceus: Avaliação do Meio de Cultivo na Produção de Biomassa Micelial e Exopolissacarídeo. Bachelor’s Thesis, Universidade Tecnológica Federal do Paraná, Paraná, Brazil, 16 June 2023. [Google Scholar]
- Di Mario, F.; Rapanà, P.; Tomati, U.; Galli, E. Chitin and chitosan from Basidiomycetes. Int. J. Biol. Macromol. 2008, 43, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Zinavovic, S.; Draughon, F.A.; Sams, C.E. Chtin and Chitosan—Value-Added Products from Mushroom Waste. J. Agric. Food Chem. 2004, 52, 7905–7910. [Google Scholar] [CrossRef] [PubMed]
- Delezuk, J.A.D.M.; Pavinatto, A.; Campana-Filho, S.P. Influence of the process parameters on β-chitin and α-chitin extraction: Probing about the grinding and particles size. Mater. Today Proc. 2019, 14, 722–732. [Google Scholar] [CrossRef]
- Wang, C.; Esker, A.R. Nanocrystalline chitin thin films. Carbohydr. Polym. 2014, 102, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, J.; Hu, A.; Nie, T.; Cheng, Z.; Liu, W. A Critical Review on Engineering of d-Mannitol Crystals: Properties, Applications, and Polymorphic Control. Crystals 2022, 12, 1080. [Google Scholar] [CrossRef]
- Poerio, A.; Petit, C.; Jehl, J.-P.; Arab-Tehrany, E.; Mano, J.F.; Cleymand, F. Extraction and Physicochemical Characterization of Chitin from Cicada orni Sloughs of the South-Eastern French Mediterranean Basin. Molecules 2020, 25, 2543. [Google Scholar] [CrossRef]
- Almeida, C.F.; Amorim, I.; Silva, C.G.; Lopes, J.C.B.; Manrique, Y.A.; Dias, M.M. Recovery of Chitin from Agaricus bisporus Mushrooms: Influence of Extraction Parameters and Supercritical CO2 Treatment on Fresh Mushrooms and Production Residues. Molecules 2025, 30, 1479. [Google Scholar] [CrossRef]
- Rasweefali, M.K.; Sabu, S.; Muhammed Azad, K.S.; Raseel Rahman, M.K.; Sunooj, K.V.; Sasidharan, A.; Anoop, K.K. Influence of deproteinization and demineralization process sequences on the physicochemical and structural characteristics of chitin isolated from Deep-sea mud shrimp (Solenocera hextii). Adv. Biomark. Sci. Technol. 2022, 4, 12–27. [Google Scholar] [CrossRef]
- Yuan, Y.; Chesnutt, B.M.; Haggard, W.O.; Bumgardner, J.D. Deacetylation of Chitosan: Material Characterization and in vitro Evaluation via Albumin Adsorption and Pre-Osteoblastic Cell Cultures. Materials 2011, 4, 1399–1416. [Google Scholar] [CrossRef] [PubMed]
- Kanani, N.; Kurniawan, T.; Kosimaningrum, W.; Meliana, Y.; Jayanudin; Wardhono, E. Ultrasonic irradiation as a mild and efficient protocol for the demineralization of chitin from shrimp shell wastes. S. Afr. J. Chem. Eng. 2023, 43, 359–368. [Google Scholar] [CrossRef]
- Kumari, S.; Kumar Annamareddy, S.H.; Abanti, S.; Kumar Rath, P. Physicochemical properties and characterization of chitosan synthesized from fish scales, crab and shrimp shells. Int. J. Biol. Macromol. 2017, 104, 1697–1705. [Google Scholar] [CrossRef]
- Sul, Y.; Ezati, P.; Rhim, J.-W. Preparation of chitosan/gelatin-based functional films integrated with carbon dots from banana peel for active packaging application. Int. J. Biol. Macromol. 2023, 246, 125600. [Google Scholar] [CrossRef]
- Rasti, H.; Parivar, K.; Baharara, J.; Iranshahi, M.; Namvar, F. Chitin from the Mollusc Chiton: Extraction, Characterization and Chitosan Preparation. Iran. J. Pharm. Res. 2017, 16, 366. [Google Scholar]
- Valverde, M.E.; Hernández-Pérez, T.; Paredes-López, O. Edible Mushrooms: Improving Human Health and Promoting Quality Life. Int. J. Microbiol. 2015, 2015, 376387. [Google Scholar] [CrossRef] [PubMed]
- Al-khattawi, A.; Alyami, H.; Townsend, B.; Ma, X.; Mohammed, A.R. Evidence-Based Nanoscopic and Molecular Framework for Excipient Functionality in Compressed Orally Disintegrating Tablets. PLoS ONE 2014, 9, e101369. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Kalonia, D.S. Effect of vacuum drying on protein-mannitol interactions: The physical state of mannitol and protein structure in the dried state. AAPS PharmSciTech 2004, 5, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Triunfo, M.; Tafi, E.; Guarnieri, A.; Salvia, R.; Scieuzo, C.; Hahn, T.; Zibek, S.; Gagliardini, A.; Panariello, L.; Coltelli, M.B.; et al. Characterization of chitin and chitosan derived from Hermetia illucens, a further step in a circular economy process. Sci. Rep. 2022, 12, 6613. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Zhu, H.; Huang, J.; Zhao, J.; Yan, B.; Ma, S.; Zhang, H.; Fan, D. The physicochemical properties of chitosan prepared by microwave heating. Food Sci. Nutr. 2020, 8, 1987–1994. [Google Scholar] [CrossRef]
- Kaya, M.; Baran, T.; Erdoğan, S.; Menteş, A.; Aşan Özüsağlam, M.; Çakmak, Y.S. Physicochemical comparison of chitin and chitosan obtained from larvae and adult Colorado potato beetle (Leptinotarsa decemlineata). Mater. Sci. Eng. C 2014, 45, 72–81. [Google Scholar] [CrossRef]
- Kaya, M.; Baran, T. Description of a new surface morphology for chitin extracted from wings of cockroach (Periplaneta americana). Int. J. Biol. Macromol. 2015, 75, 7–12. [Google Scholar] [CrossRef]
- Hosseinnejad, M.; Jafari, S.M. Evaluation of different factors affecting antimicrobial properties of chitosan. Int. J. Biol. Macromol. 2016, 85, 467–475. [Google Scholar] [CrossRef]
- Li, J.; Pu, T.; Wang, Z.; Liu, T. Thermal Behavior and Pyrolysis Kinetics of Mushroom Residue with the Introduction of Waste Plastics. Polymers 2023, 15, 3824. [Google Scholar] [CrossRef]
- Tǎnase, C.; Odochian, L.; Balaeş, T.; Lisǎ, G.; Gherca, D.; Pui, A. Study of thermal behaviour of some edible mushrooms. J. Therm. Anal. Calorim. 2014, 115, 947–953. [Google Scholar] [CrossRef]
- Ramírez, M.Á.; González, P.; Fagundo, J.R.; Suarez, M.; Melian, C.; Rodríguez, T.; Peniche, C. Chitin Preparation by Demineralizing Deproteinized Lobster Shells with CO2 and a Cationite. J. Renew. Mater. 2017, 5, 30–37. [Google Scholar] [CrossRef]
- Alehosseini, E.; Tabarestani, H.S.; Kharazmi, M.S.; Jafari, S.M. Physicochemical, Thermal, and Morphological Properties of Chitosan Nanoparticles Produced by Ionic Gelation. Foods 2022, 11, 3841. [Google Scholar] [CrossRef]
- Corazzari, I.; Nisticò, R.; Turci, F.; Faga, M.G.; Franzoso, F.; Tabasso, S.; Magnacca, G. Advanced physico-chemical characterization of chitosan by means of TGA coupled on-line with FTIR and GCMS: Thermal degradation and water adsorption capacity. Polym. Degrad. Stab. 2015, 112, 1–9. [Google Scholar] [CrossRef]
- Zasoski, R.J. Zeta potential. In Encyclopedia of Earth Sciences Series; Springer: Dordrecht, The Netherlands, 2008; pp. 841–845. [Google Scholar] [CrossRef]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.R. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Mohanta, Y.K.; Nayak, D.; Biswas, K.; Singdevsachan, S.K.; Abd Allah, E.F.; Hashem, A.; Alqarawi, A.A.; Yadav, D.; Mohanta, T.K. Silver Nanoparticles Synthesized Using Wild Mushroom Show Potential Antimicrobial Activities against Food Borne Pathogens. Mol. A J. Synth. Chem. Nat. Prod. Chem. 2018, 23, 655. [Google Scholar] [CrossRef]
- Zhang, M.; Zhao, K.; Zhang, K.; Wang, W.; Xing, J.; Li, Y. Influence of glucan on physicochemical and rheology properties of chitin nanofibers prepared from Shiitake stipes. Carbohydr. Polym. 2022, 294, 119762. [Google Scholar] [CrossRef] [PubMed]
- Mohd Affandy, M.A.; Rovina, K. Characterization of chitosan derived from mushroom sources: Physicochemical, morphological, thermal analysis. Sustain. Chem. Pharm. 2024, 40, 101624. [Google Scholar] [CrossRef]
- Psarianos, M.; Marzban, N.; Ojha, S.; Schneider, R.; Schluter, O.K. Functional and bioactive properties of chitosan produced from Acheta domesticus with fermentation, enzymatic and microwave-assisted extraction. Sustain. Food Technol. 2024, 3, 277–285. [Google Scholar] [CrossRef]
- Boonmahitthisud, A.; Thongdonson, K.; Tanpichai, S. Preparation of Chitin Nanofibers from Shrimp Shell Waste by Partial Deacetylation and Mechanical Treatment. J. Nat. Fibers 2023, 20, 2229515. [Google Scholar] [CrossRef]
- Mun, S.; Decker, E.A.; McClements, D.J. Effect of molecular weight and degree of deacetylation of chitosan on the formation of oil-in-water emulsions stabilized by surfactant-chitosan membranes. J. Colloid Interface Sci. 2006, 296, 581–590. [Google Scholar] [CrossRef]
- Yang, H.C.; Hon, M.H. The effect of the degree of deacetylation of chitosan nanoparticles and its characterization and encapsulation efficiency on drug delivery. Polymer 2010, 49, 1292–1296. [Google Scholar] [CrossRef]
- Pellis, A.; Guebitz, G.M.; Nyanhongo, G.S. Chitosan: Sources, Processing and Modification Techniques. Gels 2022, 8, 393. [Google Scholar] [CrossRef]
- Maciel, V.B.V.; Franco, T.T.; Yoshida, C.M.P. Sistemas inteligentes de embalagens utilizando filmes de quitosana como indicador colorimétrico de temperatura. Polímeros 2012, 22, 318–324. [Google Scholar] [CrossRef]
- Junior, L.B.; Bertuci, M.L.; Fakhouri, F.M.; Martelli, S.M. Caracterização e biodegradação de filmes de quitosana e proteína isolada de soja incorporados com nanocristais de celulose/Characterization and biodegradation of chitosan and soy protein isolate films incorporated with cellulose nanocrystals. Braz. J. Dev. 2022, 8, 16681–16718. [Google Scholar] [CrossRef]
- Mapari, S.A.S.; Meyer, A.S.; Thrane, U. Colorimetric Characterization for Comparative Analysis of Fungal Pigments and Natural Food Colorants. J. Agric. Food Chem. 2006, 54, 7027–7035. [Google Scholar] [CrossRef] [PubMed]
- Kita, D.M.; Giovanella, P.; Yoshinaga, T.T.; Pellizzer, E.P.; Sette, L.D. Antarctic fungi applied to textile dye bioremediation. An. Acad. Bras. Ciênc. 2022, 94 (Suppl. S1), e20210234. [Google Scholar] [CrossRef] [PubMed]
- Mohan, K.; Ganesan, A.R.; Ezhilarasi, P.N.; Kondamareddy, K.K.; Rajan, D.K.; Sathishkumar, P.; Rajarajessaran, J.; Conterno, L. Green and eco-friendly approaches for the extraction of chitin and chitosan: A review. Carbohydr. Polym. 2022, 287, 119349. [Google Scholar] [CrossRef] [PubMed]
- Oladzadabbasabadi, N.; Nafchi, A.M.; Ariffin, F.; Wijekoon, M.M.J.; Al-Hassan, A.A.; Dheyab, M.A.; Ghasemlou, M. Recent advances in extraction, modification, and application of chitosan in packaging industry. Carbohydr. Polym. 2022, 277, 118876. [Google Scholar] [CrossRef]




| Sample | DA (%) | DD (%) |
|---|---|---|
| Fungal Chitosan | 38.43 | 61.57 |
| Commercial Chitosan | 22.73 | 77.27 |
| Sample | L* | a* | b* |
|---|---|---|---|
| Commercial Chitosan | 86.90 ± 0.28 a | 0.87 ± 0.07 c | 14.75 ± 0.14 a |
| Fungal Chitosan | 71.97 ± 0.44 b | 3.35 ± 0.04 b | 13.03 ± 0.12 ab |
| Deproteinized Chitosan | 47.92 ± 0.83 c | 5.96 ± 0.12 a | 14.99 ± 0.34 a |
| Demineralized Chitosan | 23.11 ± 0.02 d | 3.21 ± 0.27 b | 6.77 ± 0.24 c |
| Mushroom Powder | 30.50 ± 3.36 e | 5.36 ± 0.70 a | 11.36 ± 2.19 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sousa, I.C.G.; Teixeira, S.C.; Souza, M.V.d., Jr.; Conde, M.B.M.; Bailon, G.R.; Cardoso, S.H.S.; Araújo, L.D.; Oliveira, E.B.d.; Ferreira, S.O.; Oliveira, T.V.d.; et al. Sustainable Extraction and Multimodal Characterization of Fungal Chitosan from Agaricus bisporus. Foods 2025, 14, 2785. https://doi.org/10.3390/foods14162785
Sousa ICG, Teixeira SC, Souza MVd Jr., Conde MBM, Bailon GR, Cardoso SHS, Araújo LD, Oliveira EBd, Ferreira SO, Oliveira TVd, et al. Sustainable Extraction and Multimodal Characterization of Fungal Chitosan from Agaricus bisporus. Foods. 2025; 14(16):2785. https://doi.org/10.3390/foods14162785
Chicago/Turabian StyleSousa, Inês C. G., Samiris C. Teixeira, Marinaldo V. de Souza, Jr., Maria B. M. Conde, Gabriela R. Bailon, Samuel H. S. Cardoso, Leandro D. Araújo, Eduardo B. de Oliveira, Sukarno O. Ferreira, Taíla V. de Oliveira, and et al. 2025. "Sustainable Extraction and Multimodal Characterization of Fungal Chitosan from Agaricus bisporus" Foods 14, no. 16: 2785. https://doi.org/10.3390/foods14162785
APA StyleSousa, I. C. G., Teixeira, S. C., Souza, M. V. d., Jr., Conde, M. B. M., Bailon, G. R., Cardoso, S. H. S., Araújo, L. D., Oliveira, E. B. d., Ferreira, S. O., Oliveira, T. V. d., & Soares, N. d. F. F. (2025). Sustainable Extraction and Multimodal Characterization of Fungal Chitosan from Agaricus bisporus. Foods, 14(16), 2785. https://doi.org/10.3390/foods14162785








