Functionalization of Sodium Caseinate for Production of Neat Films: Effects of Casein Crosslinking Induced by Heating at Alkaline pH or Light Exposure
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Sodium Caseinate Powder
2.2. Preparation of Control NaCas Solution (Untreated)
2.3. Preparation of LAL-Functionalized NaCas Solution
2.4. Preparation of Di-Tyr-Functionalized NaCas Solutions
2.5. Determination of the Crosslinks
2.5.1. Acid Hydrolysis
2.5.2. Determination of LAL Content by LC-MS
2.5.3. Determination of Di-Tyr Content by HPLC
2.6. Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis (SDS-PAGE)
2.7. Preparation and Characterization of NaCas Films
2.7.1. Film Thickness
2.7.2. Fourier-Transform Infrared (FTIR) Spectroscopy
2.7.3. Mechanical Properties
2.8. Statistical Analysis
3. Results and Discussion
3.1. Lysinoalanine (LAL) Formation in NaCas Solutions Heated Under Alkaline Conditions
3.2. Di-Tyr Formation in NaCas Solutions Exposed to LED Light
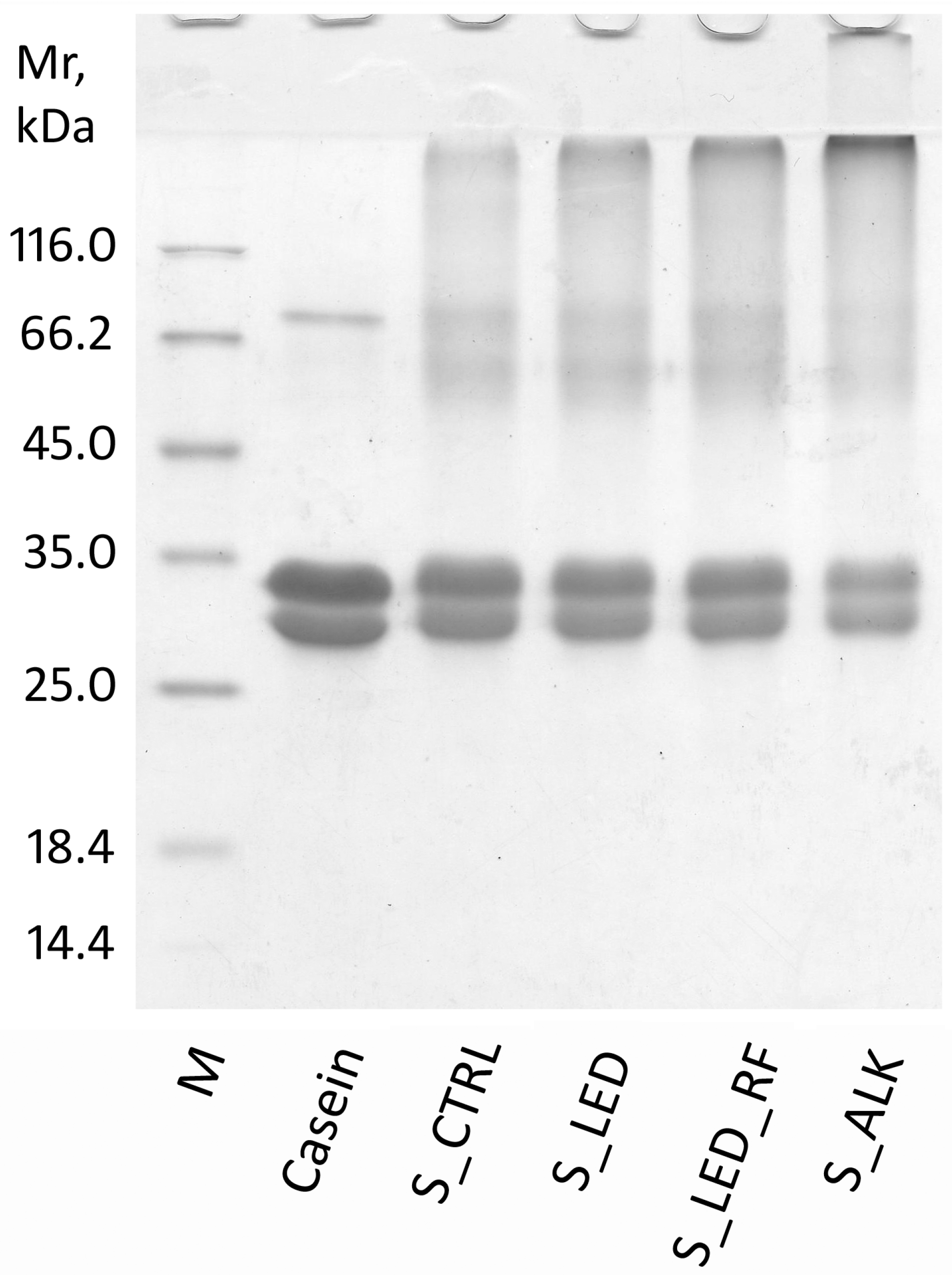
3.3. SDS-PAGE of Functionalized NaCas Solutions
3.4. Film Characterization
3.4.1. Thickness of Films from Functionalized NaCas
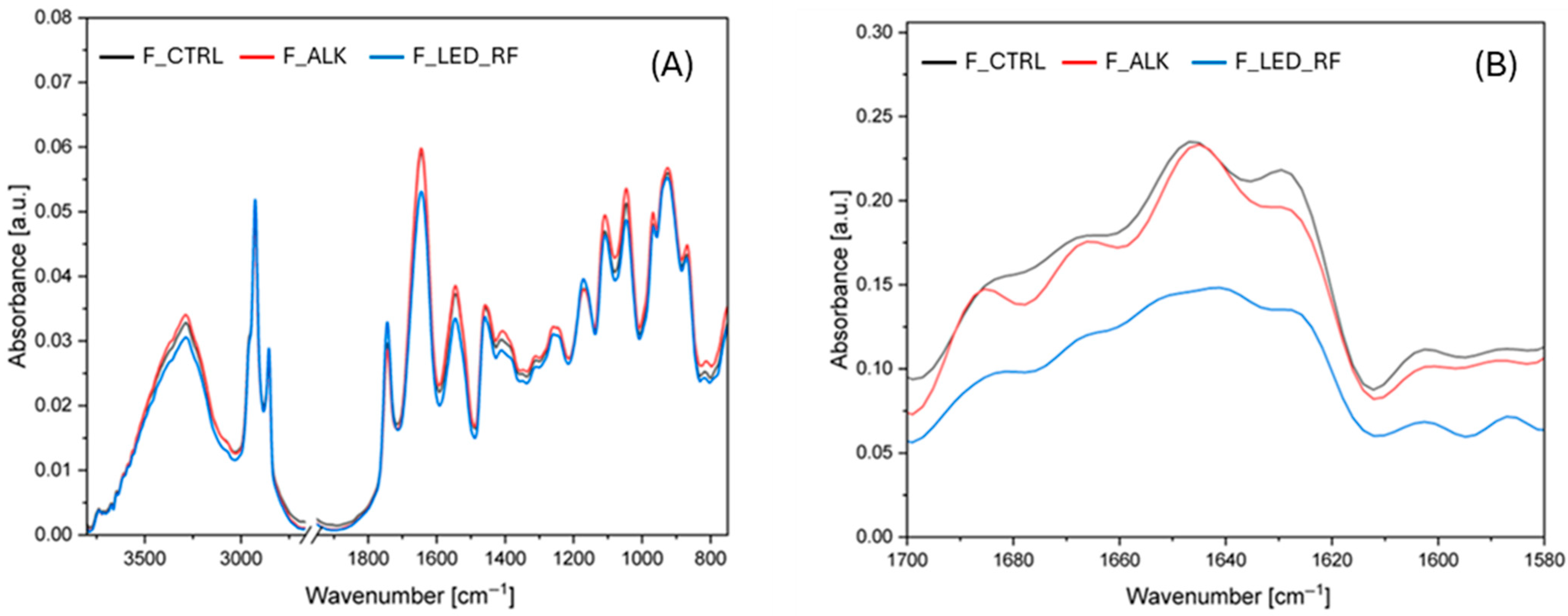
3.4.2. FTIR Analysis of Functionalized NaCas Films
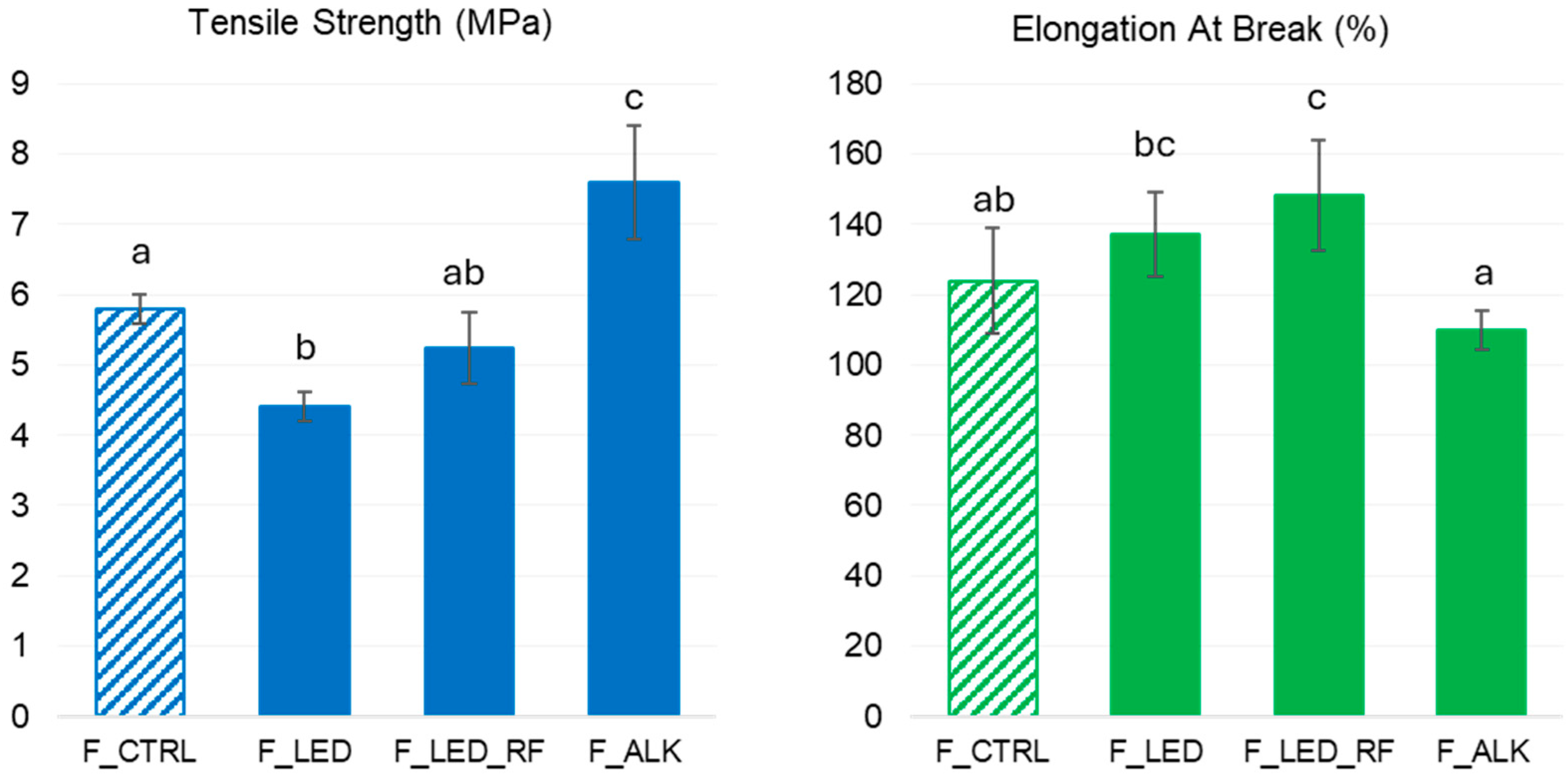
3.4.3. Mechanical Properties of the Films
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FTIR | Fourier-transform infrared spectroscopy |
| TS | Tensile strength |
| EAB | Elongation at break |
References
- Huppertz, T. Chemistry of the caseins. In Advanced Dairy Chemistry, 4th ed.; Volume 1A: Proteins: Basic Aspects; Springer: Boston, MA, USA, 2012; pp. 135–160. [Google Scholar]
- Rehan, F.; Ahemad, N.; Gupta, M. Casein nanomicelle as an emerging biomaterial—A comprehensive review. Colloids Surf. B Biointerfaces 2019, 179, 280–292. [Google Scholar] [CrossRef]
- Gerna, S.; D’Incecco, P.; Limbo, S.; Sindaco, M.; Pellegrino, L. Strategies for exploiting Milk protein properties in making films and coatings for food packaging: A review. Foods 2023, 12, 1271. [Google Scholar] [CrossRef]
- Khwaldia, K. Water vapor barrier and mechanical properties of paper—Sodium caseinate and paper—Sodium caseinate—Paraffin wax films. J. Food Biochem. 2010, 34, 998–1013. [Google Scholar] [CrossRef]
- Renoldi, N.; Calligaris, S.; Nicoli, M.C.; Marino, M.; Rossi, A.; Innocente, N. Effect of the shifting from multi-layer systems towards recyclable mono-material packaging solutions on the shelf-life of portioned semi-hard cheese. Food Packag. Shelf Life 2024, 46, 101363. [Google Scholar] [CrossRef]
- Chevalier, E.; Assezat, G.; Prochazka, F.; Oulahal, N. Development and characterization of a novel edible extruded sheet based on different casein sources and influence of the glycerol concentration. Food Hydrocoll. 2018, 75, 182–191. [Google Scholar] [CrossRef]
- Gerna, S.; D’Incecco, P.; Limbo, S.; Alamprese, C.; Pellegrino, L. Upcycling casein from expired pasteurized milk into films–Development and characterization of films made from sodium caseinate. Appl. Food Res. 2025, 5, 100901. [Google Scholar] [CrossRef]
- Audic, J.L.; Chaufer, B. Caseinate based biodegradable films with improved water resistance. J. Appl. Polym. Sci. 2010, 117, 1828–1836. [Google Scholar] [CrossRef]
- Lin, H.C.; Wang, B.J.; Weng, Y.M. Development and characterization of sodium caseinate edible films cross-linked with genipin. LWT 2020, 118, 108813. [Google Scholar] [CrossRef]
- Viora, L.; Tichané, T.; Nottelet, B.; Mouton, J.; Garric, X.; Van Den Berghe, H.; Coudane, J. Casein—Based conjugates and graft copolymers. Synthesis, properties, and applications. Compr. Rev. Food Sci. Food Saf. 2024, 23, 13306. [Google Scholar] [CrossRef]
- Kumar, A.P.; Singh, R.P. Biocomposites of cellulose reinforced starch: Improvement of properties by photo-induced crosslinking. Biores. Technol. 2008, 99, 8803–8809. [Google Scholar] [CrossRef]
- Cardoso, H.B.; Wierenga, P.A.; Gruppen, H.; Schols, H.A. Maillard induced aggregation of individual milk proteins and interactions involved. Food Chem. 2019, 276, 652–661. [Google Scholar] [CrossRef]
- Raak, N.; Abbate, R.A.; Lederer, A.; Rohm, H.; Jaros, D. Size separation techniques for the characterisation of cross-linked casein: A review of methods and their applications. Separations 2018, 5, 14. [Google Scholar] [CrossRef]
- Pellegrino, L.; Resmini, P.; De Noni, I.; Masotti, F. Sensitive determination of lysinoalanine for distinguishing natural from imitation mozzarella cheese. J. Dairy Sci. 1996, 79, 725–734. [Google Scholar] [CrossRef]
- Friedman, M. Chemistry, biochemistry, nutrition, and microbiology of lysinoalanine, lanthionine, and histidinoalanine in food and other proteins. J. Agric. Food Chem. 1999, 47, 1295–1319. [Google Scholar] [CrossRef] [PubMed]
- Picchio, M.L.; Linck, Y.G.; Monti, G.A.; Gugliotta, L.M.; Minari, R.J.; Igarzabal, C.I.A. Casein films crosslinked by tannic acid for food packaging applications. Food Hydrocoll. 2018, 84, 424–434. [Google Scholar] [CrossRef]
- Raak, N.; Rohm, H.; Jaros, D. Enzymatically cross-linked sodium caseinate as techno-functional ingredient in acid-induced milk gels. Food Bioprocess Technol. 2020, 13, 1857–1865. [Google Scholar] [CrossRef]
- Dalsgaard, T.K.; Nielsen, J.H.; Brown, B.E.; Stadler, N.; Davies, M.J. Dityrosine, 3, 4-dihydroxyphenylalanine (DOPA), and radical formation from tyrosine residues on milk proteins with globular and flexible structures as a result of riboflavin-mediated photo-oxidation. J. Agric. Food Chem. 2011, 59, 7939–7947. [Google Scholar] [CrossRef]
- Fuentes-Lemus, E.; Silva, E.; Leinisch, F.; Dorta, E.; Lorentzen, L.G.; Davies, M.J.; López-Alarcón, C. α-and β-casein aggregation induced by riboflavin-sensitized photo-oxidation occurs via di-tyrosine cross-links and is oxygen concentration dependent. Food Chem. 2018, 256, 119–128. [Google Scholar] [CrossRef]
- Lee, Y.B.; Lim, S.; Lee, Y.; Park, C.H.; Lee, H.J. Green chemistry for crosslinking biopolymers: Recent advances in riboflavin-mediated photochemistry. Materials 2023, 16, 1218. [Google Scholar] [CrossRef]
- Chen, Z.; Leinisch, F.; Greco, I.; Zhang, W.; Shu, N.; Chuang, C.Y.; Lund, M.N.; Davies, M.J. Characterisation and quantification of protein oxidative modifications and amino acid racemisation in powdered infant milk formula. Free Radic. Res. 2019, 53, 68–81. [Google Scholar] [CrossRef]
- D’Incecco, P.; Dallavalle, S.; Musso, L.; Rosi, V.; Sindaco, M.; Pellegrino, L. Formation of di-Tyrosine in pasteurized milk during shelf storage. Food Chem. 2024, 435, 137566. [Google Scholar] [CrossRef]
- Nielsen, S.D.; Knudsen, L.J.; Bækgaard, L.T.; Rauh, V.; Larsen, L.B. Influence of lactose on the Maillard reaction and dehydroalanine-mediated protein cross-linking in casein and whey. Foods 2022, 11, 897. [Google Scholar] [CrossRef]
- Cattaneo, S.; Masotti, F.; Pellegrino, L. Chemical modifications of casein occurring during industrial manufacturing of milk protein powders. Eur. Food Res. Technol. 2012, 235, 315–323. [Google Scholar] [CrossRef]
- Calabrese, M.G.; Mamone, G.; Caira, S.; Ferranti, P.; Addeo, F. Quantitation of lysinoalanine in dairy products by liquid chromatography–mass spectrometry with selective ion monitoring. Food Chem. 2009, 116, 799–805. [Google Scholar] [CrossRef]
- Akıllıoğlu, H.G.; Lund, M.N. Quantification of advanced glycation end products and amino acid cross-links in foods by high-resolution mass spectrometry: Applicability of acid hydrolysis. Food Chem. 2022, 366, 130601. [Google Scholar] [CrossRef] [PubMed]
- Markoska, T.; Daniloski, D.; Vasiljevic, T.; Huppertz, T. Structural changes of β-casein induced by temperature and pH analysed by nuclear magnetic resonance, Fourier-transform infrared spectroscopy, and chemometrics. Molecules 2021, 26, 7650. [Google Scholar] [CrossRef]
- ASTM D882-18; Standard Test Method for Tensile Properties of Thin Plastic Sheeting. ASTM International: West Conshohocken, PA, USA, 2018.
- Anema, S.G. Heat-induced changes in caseins and casein micelles, including interactions with denatured whey proteins. Int. Dairy J. 2021, 122, 105136. [Google Scholar] [CrossRef]
- Resmini, P.; Pellegrino, L.; Cattaneo, S. Furosine and other heat-treatment indicators for detecting fraud in milk and milk products. Ital. J. Food Sci. 2003, 15, 473–484. [Google Scholar]
- Zhang, Z.; Wang, Y.; Jiang, H.; Dai, C.; Xing, Z.; Mintah, B.K.; Dabbour, M.; He, R.; Ma, H. Effect of dual-frequency ultrasound on the formation of lysinoalanine and structural characterization of rice dreg protein isolates. Ultrason. Sonochem. 2020, 67, 105124. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Z.; Ren, W.; Wang, Y.; Mintah, B.K.; Dabbour, M.; Hou, Y.; He, R.; Cheng, Y.; Ma, H. Inhibition effect of ultrasound on the formation of lysinoalanine in rapeseed protein isolates during pH shift treatment. J. Agric. Food Chem. 2021, 69, 8536–8545. [Google Scholar] [CrossRef]
- Poulsen, N.A.; Rybicka, I.; Larsen, L.B.; Buitenhuis, A.J.; Larsen, M.K. Genetic variation of riboflavin content in bovine milk. J. Dairy Sci. 2015, 98, 3496–3501. [Google Scholar] [CrossRef]
- Fracassetti, D.; Limbo, S.; D’Incecco, P.; Tirelli, A.; Pellegrino, L. Development of a HPLC method for the simultaneous analysis of riboflavin and other flavin compounds in liquid milk and milk products. Eur. Food Res. Technol. 2018, 244, 1545–1554. [Google Scholar] [CrossRef]
- Masum, A.K.M.; Chandrapala, J.; Huppertz, T.; Adhikari, B.; Zisu, B. Production and characterization of infant milk formula powders: A review. Dry. Technol. 2021, 39, 1492–1512. [Google Scholar] [CrossRef]
- Monogioudi, E.; Creusot, N.; Kruus, K.; Gruppen, H.; Buchert, J.; Mattinen, M.L. Cross-linking of β-casein by Trichoderma reesei tyrosinase and Streptoverticillium mobaraense transglutaminase followed by SEC–MALLS. Food Hydrocoll. 2009, 23, 2008–2015. [Google Scholar] [CrossRef]
- Diak, O.A.; Bani-Jaber, A.; Amro, B.; Jones, D.; Andrews, G.P. The manufacture and characterization of casein films as novel tablet coatings. Food Bioprod. Process. 2007, 85, 284–290. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Peltzer, M.A.; del Carmen Garrigós, M.; Jiménez, A. Structure and mechanical properties of sodium and calcium caseinate edible active films with carvacrol. J. Food Eng. 2013, 114, 486–494. [Google Scholar] [CrossRef]
- Indrie, L.; McNeil, S.; Mutlu, M.M.; Bota, S.; Ilieș, D.C.; Karavana, H.A. The Initial Development and Evaluation of Cross-Linked Casein Films for Sustainable Footwear. Coatings 2023, 13, 217. [Google Scholar] [CrossRef]
- Fernández, C.; Ausar, S.F.; Badini, R.G.; Castagna, L.F.; Bianco, I.D.; Beltramo, D.M. An FTIR spectroscopy study of the interaction between αs-casein-bound phosphoryl groups and chitosan. Int. Dairy J. 2003, 13, 897–901. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, Y.; Dai, C.; He, R.; Ma, H. Alkali extraction of rice residue protein isolates: Effects of alkali treatment conditions on lysinoalanine formation and structural characterization of lysinoalanine-containing protein. Food Chem. 2018, 261, 176–183. [Google Scholar] [CrossRef]
- Haddad-Khoozani, R.; Soltanizadeh, N. The effect of polysaccharide type on dielectric barrier discharge (DBD) plasma glycosylation of sodium caseinate-part I: Physicochemical, structural and thermal properties. Int. J. Biol. Macromol. 2024, 278, 135061. [Google Scholar] [CrossRef]
- Khan, H.H.; McCarthy, U.; Esmonde-White, K.; Casey, I.; O’Shea, N. Potential of Raman spectroscopy for in-line measurement of raw milk composition. Food Control 2023, 152, 109862. [Google Scholar] [CrossRef]
- Whittaker, M.M.; DeVito, V.L.; Asher, S.A.; Whittaker, J.W. Resonance Raman evidence for tyrosine involvement in the radical site of galactose oxidase. J. Biol. Chem. 1989, 264, 7104–7106. [Google Scholar] [CrossRef]

| Sample | LAL (µg/g Protein) | LAL (µmol/g Protein) |
|---|---|---|
| S_CTRL | 867 ± 22 a | 3.88 |
| S_LED | 907 ± 27 b | 4.06 |
| S_LED_RF | 722 ± 11 c | 3.46 |
| S_ALK | 3540 ± 64 d | 15.86 |
| Sample | di-Tyr (µg/g Protein) | di-Tyr (µmol/g Protein) |
|---|---|---|
| S_CTRL | n.d. | n.d. |
| S_LED | 4 ± 0.1 | 0.0111 |
| S_LED_RF | 501 ± 7.5 | 1.39 |
| S_ALK | 1 ± 0.0 | 0.0003 |
| Sample | Thickness (µm) |
|---|---|
| F_CRTL | 145 ± 30 a |
| F_LED | 145 ± 14 a |
| F_LED_RF | 152 ± 15 a |
| F_ALK | 142 ± 4 a |
| Band Position (cm−1) | Band Assignment | F_CTRL (%) | F_ALK (%) | F_LED_RF (%) |
|---|---|---|---|---|
| 1600–1610 | Side chain | 7.1 ± 0.6 a | 7.0 ± 0.7 a | 6.9 ± 0.2 a |
| 1615–1635 | β-sheet | 23.6 ± 2.5 a | 17.1 ± 2.7 b | 19.7 ± 1.5 bc |
| 1640–1645 | Random coil | 6.0 ± 1.0 a | 10.1 ± 1.6 b | 9.3 ± 1.3 b |
| 1645–1660 | α-Helix | 22.8 ± 2.5 a | 23.9 ± 3.2 a | 23.4 ± 2.9 a |
| 1660–1682 | β-turn | 25.3 ± 1.5 a | 21.8 ± 1.7 b | 25.5 ± 1.9 a |
| 1682–1699 | Aggregated β-sheet | 15.2 ± 3.1 a | 20.0 ± 4.7 b | 15.2 ± 2.8 a |
| Band Position (cm−1) | Band Assignment | F_CTRL | F_ALK | F_LED_RF |
|---|---|---|---|---|
| 1615–1635 | β-sheet | 1629 | 1625 | 1628 |
| 1640–1645 | Random coil | 1647 | 1645 | 1640 |
| 1660–1682 | β-turn | 1667 | 1666 | 1667 |
| 1682–1699 | Aggregated β-sheet | 1683 | 1686 | 1682 |
| 1470–1450 | Tryptophan | 1459 | 1459 | 1461 |
| 1190–1155 | Tyr/di-Tyr | 1171 | 1171 | 1174 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Incecco, P.; Gerna, S.; Sindaco, M.; Pellegrino, L.; Barbiroli, A.; Rosi, V.; Limbo, S. Functionalization of Sodium Caseinate for Production of Neat Films: Effects of Casein Crosslinking Induced by Heating at Alkaline pH or Light Exposure. Foods 2025, 14, 2764. https://doi.org/10.3390/foods14162764
D’Incecco P, Gerna S, Sindaco M, Pellegrino L, Barbiroli A, Rosi V, Limbo S. Functionalization of Sodium Caseinate for Production of Neat Films: Effects of Casein Crosslinking Induced by Heating at Alkaline pH or Light Exposure. Foods. 2025; 14(16):2764. https://doi.org/10.3390/foods14162764
Chicago/Turabian StyleD’Incecco, Paolo, Stefano Gerna, Marta Sindaco, Luisa Pellegrino, Alberto Barbiroli, Veronica Rosi, and Sara Limbo. 2025. "Functionalization of Sodium Caseinate for Production of Neat Films: Effects of Casein Crosslinking Induced by Heating at Alkaline pH or Light Exposure" Foods 14, no. 16: 2764. https://doi.org/10.3390/foods14162764
APA StyleD’Incecco, P., Gerna, S., Sindaco, M., Pellegrino, L., Barbiroli, A., Rosi, V., & Limbo, S. (2025). Functionalization of Sodium Caseinate for Production of Neat Films: Effects of Casein Crosslinking Induced by Heating at Alkaline pH or Light Exposure. Foods, 14(16), 2764. https://doi.org/10.3390/foods14162764








