Evaluation of the Quality Changes in Three Commercial Pastourma Samples during Refrigerated Storage Using Physicochemical, Microbiological, and Image Analyses Combined with Chemometrics
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Image Acquisition
2.3. Discriminant Analysis
2.4. Determination of Physicochemical Parameters during Pastourma Storage
2.5. Thiobarbituric Acid Reactive Substance (TBARS) Assay
2.6. Fourier-Transform Infrared Spectroscopy with Attenuated Total Reflectance (ATR-FTIR)
2.7. Microbiological Analyses
2.8. Statistical Analysis
3. Results and Discussion
3.1. Image Texture Evaluation of Pastourma Samples during Storage
3.2. Storage Effect on the Physicochemical Features of Pastourma Samples
3.3. Attenuated Total Reflection–Fourier-Transform Infrared (ATR-FTIR) Spectra of Pastourma Samples during Storage
3.4. Microbiological Analyses
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Aksu, M.İ.; Erdemir, E.; Turan, E.; Sat, İ.G. Effects of Red Beet Extracts on Protein and Lipid Oxidation, Colour, Microbial, Sensory Properties and Storage Stability of Turkish Pastırma. J. Stored Prod. Res. 2020, 89, 101721. [Google Scholar] [CrossRef]
- Akköse, A.; Kaban, G.; Karaoğlu, M.M.; Kaya, M. Manda Etinden Üretilen Pastırma Çeşitlerinin Özellikleri. Kafkas Univ. Vet. Fak. Derg. 2018, 24, 179–185. [Google Scholar] [CrossRef]
- Ren, W.; Bekhit, A.E.A.; Li, F.; Yang, H.; Jiang, X.; Zhang, W.; Kong, L. Physicochemical Properties of Pastirma from Horse Meat, Beef, Mutton and Pork. J. Food Qual. 2015, 38, 369–376. [Google Scholar] [CrossRef]
- Abdallah, M.R.S.; Mohamed, M.A.; Mohamed, H.M.H.; Emara, M.M.T. Improving the Sensory, Physicochemical and Microbiological Quality of Pastirma (A Traditional Dry Cured Meat Product) Using Chitosan Coating. LWT 2017, 86, 247–253. [Google Scholar] [CrossRef]
- Aksu, M.İ.; Turan, E.; Şat, İ.G. Effects of Lyophilized Red Cabbage Water Extract and pH Levels on the Quality Properties of Pastırma Cemen Paste during Chilled Storage. J. Stored Prod. Res. 2020, 89, 101696. [Google Scholar] [CrossRef]
- Aksu, M.İ.; Turan, E.; Şat, İ.G.; Erdemir, E. Red Raspberry (Rubus ideaus L.) Extracts: A Novel Ingredient in Cemen Paste. Food Process. Preserv. 2021, 45, e15930. [Google Scholar] [CrossRef]
- Gök, V.; Obuz, E.; Akkaya, L. Effects of Packaging Method and Storage Time on the Chemical, Microbiological, and Sensory Properties of Turkish Pastirma—A Dry Cured Beef Product. Meat Sci. 2008, 80, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Turan, E.; Şimşek, A. Determination of physicochemical, antioxidant, textural and sensory features of pastirma cemen paste produced by using different concentrations of black garlic. Gıda 2022, 47, 980–991. [Google Scholar] [CrossRef]
- Erdemir, E. Free Amino Acids Profile and Quality Properties of Turkish Pastırma Cured with Potassium Lactate and Sodium Chloride Combinations. Food Process. Preserv. 2022, 46, e16263. [Google Scholar] [CrossRef]
- Demirok, E.; Kiralan, M.; Carbonell-Barrachina, Á.A. Determination and classification of volatile compounds of pastirma using solid phase microextraction/gas chromatography/mass spectrometry. J. Microbiol. Biotechnol. Food Sci. 2013, 3, 105–109. [Google Scholar]
- Aksu, M.I.; Dogan, M.; Sirkecioglu, A.N. Changes in the Total Lipid, Neutral Lipid, Phospholipid and Fatty Acid Composition of Phospholipid Fractions during Pastirma Processing, a Dry-Cured Meat Product. Korean J. Food Sci. Anim. Resour. 2017, 37, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Abou-Taleb, M.; Talab, A.S.; Ibrahim, M.A.; Anees, F.R.; Genina, M.E.; Mahmoud, M.M.; Abou-Taleb, S.M. Quality Evaluation of Three Types of Fish Pastirma during Frozen Storage. Egypt. J. Aquat. Biol. Fish. 2018, 22, 261–273. [Google Scholar] [CrossRef][Green Version]
- Aksu, M.İ.; Kaya, M. Effect of Storage Temperatures and Time on Shelf-life of Sliced and Modified Atmosphere Packaged Pastırma, a Dried Meat Product, Produced from Beef. J. Sci. Food Agric. 2005, 85, 1305–1312. [Google Scholar] [CrossRef]
- Aksu, M.I.; Kaya, M.; Ockerman, H.W. Effect of modified atmosphere packaging and temperature on the shelf life of sliced pastirma produced from frozen/thawed meat. J. Muscle Foods 2005, 16, 192–206. [Google Scholar] [CrossRef]
- Turan, E.; Şimşek, A. Black Garlic as a Substitute for Fresh Garlic to Reduce Off-flavor and Enhance Consumer Acceptance and Bioactive Properties in Cemen Paste. Food Process. Preserv. 2022, 46, 16246. [Google Scholar] [CrossRef]
- Çakıcı, N.; Aksu, M.İ.; Erdemir, E. A Survey of the Physico-Chemical and Microbiological Quality of Different Pastırma Types: A Dry-Cured Meat Product. CyTA J. Food 2015, 13, 196–203. [Google Scholar] [CrossRef]
- Sinanoglou, V.; Cavouras, D.; Xenogiannopoulos, D.; Proestos, C.; Zoumpoulakis, P. Quality Assessment of Pork and Turkey Hams Using FT-IR Spectroscopy, Colorimetric, and Image Analysis. Foods 2018, 7, 152. [Google Scholar] [CrossRef] [PubMed]
- Sinanoglou, V.J.; Tsiaka, T.; Aouant, K.; Mouka, E.; Ladika, G.; Kritsi, E.; Konteles, S.J.; Ioannou, A.-G.; Zoumpoulakis, P.; Strati, I.F.; et al. Quality Assessment of Banana Ripening Stages by Combining Analytical Methods and Image Analysis. Appl. Sci. 2023, 13, 3533. [Google Scholar] [CrossRef]
- Tappi, S.; Rocculi, P.; Ciampa, A.; Romani, S.; Balestra, F.; Capozzi, F.; Dalla Rosa, M. Computer Vision System (CVS): A Powerful Non-Destructive Technique for the Assessment of Red Mullet (Mullus barbatus) Freshness. Eur. Food Res. Technol. 2017, 243, 2225–2233. [Google Scholar] [CrossRef]
- Rahman, U.U.; Sahar, A.; Pasha, I.; Rahman, S.U.; Ishaq, A. Assessing the Capability of Fourier Transform Infrared Spectroscopy in Tandem with Chemometric Analysis for Predicting Poultry Meat Spoilage. PeerJ 2018, 6, e5376. [Google Scholar] [CrossRef]
- Papastergiadis, A.; Mubiru, E.; Van Langenhove, H.; De Meulenaer, B. Malondialdehyde Measurement in Oxidized Foods: Evaluation of the Spectrophotometric Thiobarbituric Acid Reactive Substances (TBARS) Test in Various Foods. J. Agric. Food Chem. 2012, 60, 9589–9594. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, A.G.; Kritsi, E.; Sinanoglou, V.J.; Cavouras, D.; Tsiaka, T.; Houhoula, D.; Zoumpoulakis, P.; Strati, I.F. Highlighting the Potential of Attenuated Total Reflectance—Fourier Transform Infrared (ATR-FTIR) Spectroscopy to Characterize Honey Samples with Principal Component Analysis (PCA). Anal. Lett. 2023, 56, 789–806. [Google Scholar] [CrossRef]
- Aksu, M.İ.; Erdemir, E.; Turan, E.; Öz, F. Chemical, Microbial, Color, Oxidative and Sensory Properties of Clean-Label Pastırma Produced with Raspberry Water Extracts as a Novel Ingredient. Meat Sci. 2022, 186, 108737. [Google Scholar] [CrossRef] [PubMed]
- Ceylan, S.; Aksu, M.İ. Free Amino Acids Profile and Quantities of ‘Sırt’, ‘Bohca’ and ‘Sekerpare’ Pastirma, Dry Cured Meat Products: Free Amino Acid Profile and Quantities in Pastirma, Dry Cured Meat Products. J. Sci. Food Agric. 2011, 91, 956–962. [Google Scholar] [CrossRef] [PubMed]
- Gençcelep, H.; İhtiyar, B.; Yüzer, M.O. Determination of Quality Properties of Kastamonu Pastırma: A Dry-Cured Meat Product. Harran Tarım Gıda Bilim. Derg. 2022, 26, 491–500. [Google Scholar] [CrossRef]
- Wibowo, A.; Suhardi, S.; Indana, K.; Purnawan Chandra, K.; Chaijan, M.; Hanum, Z. Critical Factors Affecting the Quality of the Longissimus Lumborum from Native Thai Cattle (Bos indicus). Adv. Anim. Vet. Sci. 2021, 10, 107–113. [Google Scholar] [CrossRef]
- Ran, M.; He, L.; Li, C.; Zhu, Q.; Zeng, X. Quality Changes and Shelf-Life Prediction of Cooked Cured Ham Stored at Different Temperatures. J. Food Prot. 2021, 84, 1252–1264. [Google Scholar] [CrossRef]
- Lu, N.; Zhang, L.; Zhang, X.; Li, J.; Labuza, T.P.; Zhou, P. Molecular Migration in High-Protein Intermediate-Moisture Foods during the Early Stage of Storage: Variations between Dairy and Soy Proteins and Effects on Texture. Food Res. Int. 2016, 82, 34–43. [Google Scholar] [CrossRef]
- Chaijan, M. Review: Lipid and Myoglobin Oxidations in Muscle Foods. Songklanakarin J. Sci. Technol. 2008, 30, 47–53. [Google Scholar]
- Cincotta, F.; Tripodi, G.; Merlino, M.; Nalbone, L.; Condurso, C.; Verzera, A. FTIR Spectroscopy vs. Sensory Analyses for the Sensory Shelf-Life Definition of Hamburger Buns. Int. J. Food Sci. Technol. 2023, 58, 795–801. [Google Scholar] [CrossRef]
- Candogan, K.; Evrim; Gunes Altuntas, E.; İğci, N. Authentication and Quality Assessment of Meat Products by Fourier-Transform Infrared (FTIR) Spectroscopy. Food Eng. Rev. 2021, 13, 66–91. [Google Scholar] [CrossRef]
- Deniz, E.; Güneş Altuntaş, E.; Ayhan, B.; İğci, N.; Özel Demiralp, D.; Candoğan, K. Differentiation of Beef Mixtures Adulterated with Chicken or Turkey Meat Using FTIR Spectroscopy. J. Food Process Preserv. 2018, 42, e13767. [Google Scholar] [CrossRef]
- Lestari, D.; Rohman, A.; Syofyan, S.; Yuliana, N.D.; Abu Bakar, N.K.B.; Hamidi, D. Analysis of Beef Meatballs with Rat Meat Adulteration Using Fourier Transform Infrared (FTIR) Spectroscopy in Combination with Chemometrics. Int. J. Food Prop. 2022, 25, 1446–1457. [Google Scholar] [CrossRef]
- Sohail, A.; Al-Dalali, S.; Wang, J.; Xie, J.; Shakoor, A.; Asimi, S.; Shah, H.; Patil, P. Aroma Compounds Identified in Cooked Meat: A Review. Food Res. Int. 2022, 157, 111385. [Google Scholar] [CrossRef]
- Lamyaa, M.A. Discrimination of pork content in mixtures with raw minced camel and buffalo meat using FTIR spectroscopic technique. Int. Food Res. J. 2013, 20, 1389–1394. [Google Scholar]
- Carbonaro, M.; Nucara, A. Secondary Structure of Food Proteins by Fourier Transform Spectroscopy in the Mid-Infrared Region. Amino Acids 2010, 38, 679–690. [Google Scholar] [CrossRef]
- Vanga, S.K.; Singh, A.; Kalkan, F.; Gariepy, Y.; Orsat, V.; Raghavan, V. Effect of Thermal and High Electric Fields on Secondary Structure of Peanut Protein. Int. J. Food Prop. 2016, 19, 1259–1271. [Google Scholar] [CrossRef]
- Pissia, M.-A.; Matsakidou, A.; Paraskevopoulou, A.; Kiosseoglou, V. Structural Characteristics and Physicochemical Properties of Freeze-Dried Snail Meat. LWT 2022, 155, 112980. [Google Scholar] [CrossRef]
- Chou, K.-C.; Pottle, M.; Némethy, G.; Ueda, Y.; Scheraga, H.A. Structure of β-Sheets: Origin of the Right-Handed Twist and of the Increased Stability of Antiparallel over Parallel Sheets. J. Mol. Biol. 1982, 162, 89–112. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, Y.; Zhou, P.; Zhang, X.; Wang, J. Effects of High Pressure Modification on Conformation and Gelation Properties of Myofibrillar Protein. Food Chem. 2017, 217, 678–686. [Google Scholar] [CrossRef]
- Qian, S.; Li, X.; Wang, H.; Mehmood, W.; Zhang, C.; Blecker, C. Effects of Frozen Storage Temperature and Duration on Changes in Physicochemical Properties of Beef Myofibrillar Protein. J. Food Qual. 2021, 2021, 8836749. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Y.; Xia, X.; Sun, Q.; Sun, F.; Kong, B. Changes in Protein Oxidation, Structure, and Thermal Stability of Chicken Breast Subjected to Ultrasound-Assisted Immersion Freezing during Frozen Storage. Food Chem. 2023, 398, 133874. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Sun, D.-W.; Pu, H.; Wei, Q. Interpretation and Rapid Detection of Secondary Structure Modification of Actomyosin during Frozen Storage by Near-Infrared Hyperspectral Imaging. J. Food Eng. 2019, 246, 200–208. [Google Scholar] [CrossRef]
- Aksu, M.; Kaya, M. Some Microbiological, Chemical and Physical Characteristics of Pastirma Marketed in Erzurum (Erzurum Piyasasında Tüketime Sunulan Pastırmaların Bazı Fiziksel, Kimyasal ve Mikrobiyolojik Özellikleri). Turk. J. Vet. Anim. Sci. 2001, 25, 319–326. [Google Scholar]
- Öz, E.; Kaban, G.; Barış, Ö.; Kaya, M. Isolation and Identification of Lactic Acid Bacteria from Pastırma. Food Control 2017, 77, 158–162. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; García Fontán, M.C.; Cachaldora, A.; Franco, I.; Carballo, J. Study of the Lactic Acid Bacteria throughout the Manufacture of Dry-Cured Lacón (a Spanish Traditional Meat Product). Effect of Some Additives. Food Microbiol. 2010, 27, 229–235. [Google Scholar] [CrossRef]
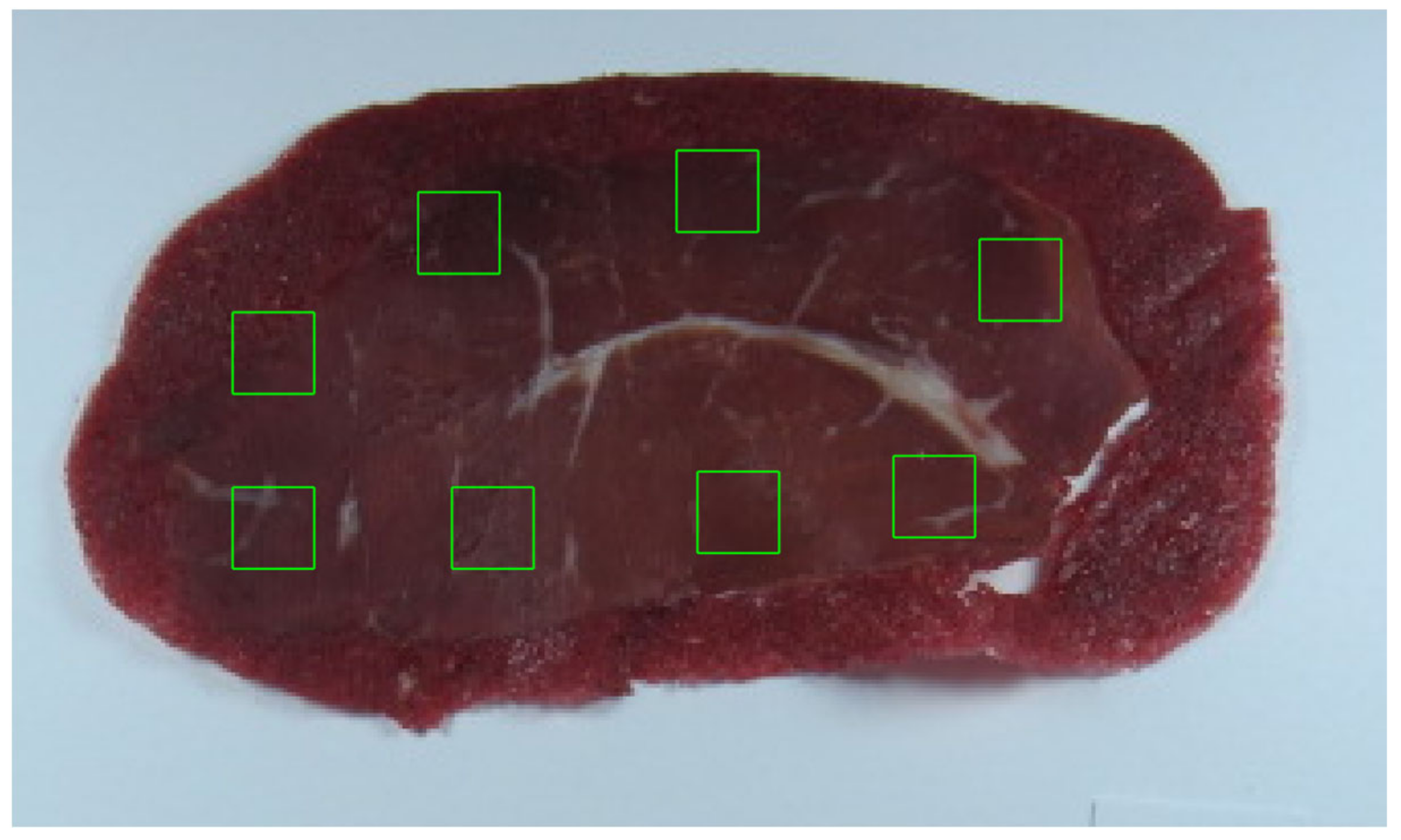




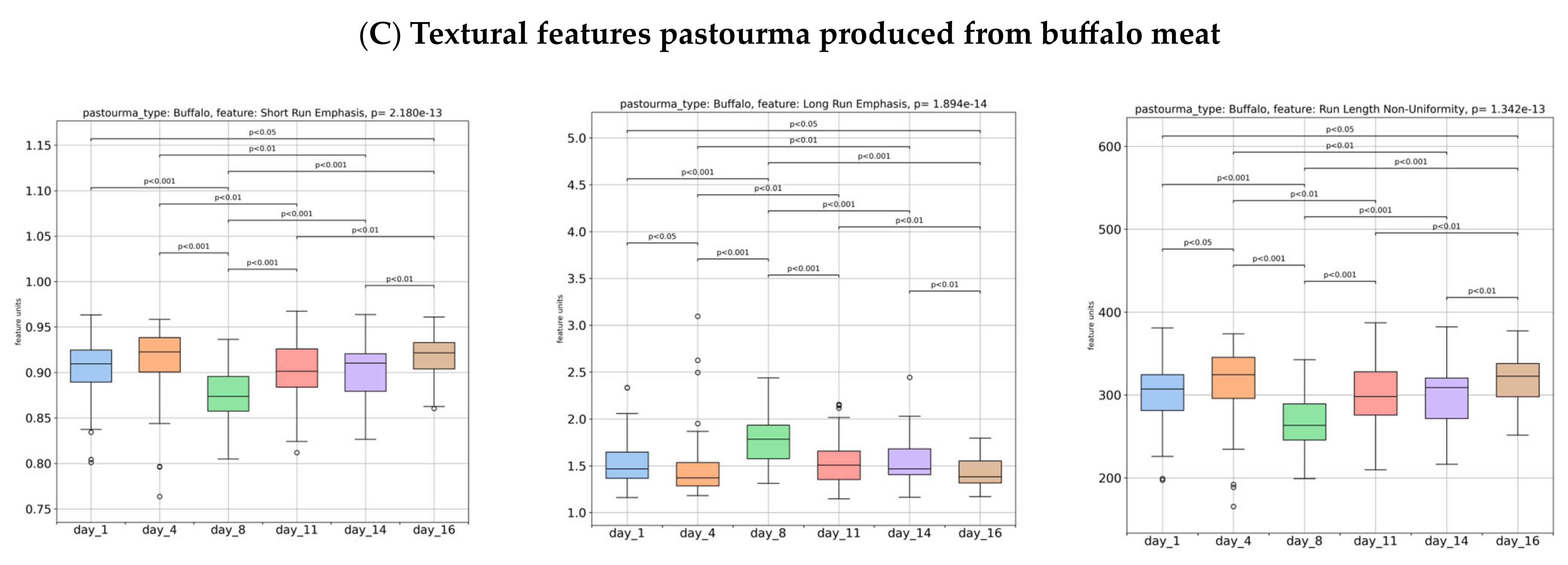

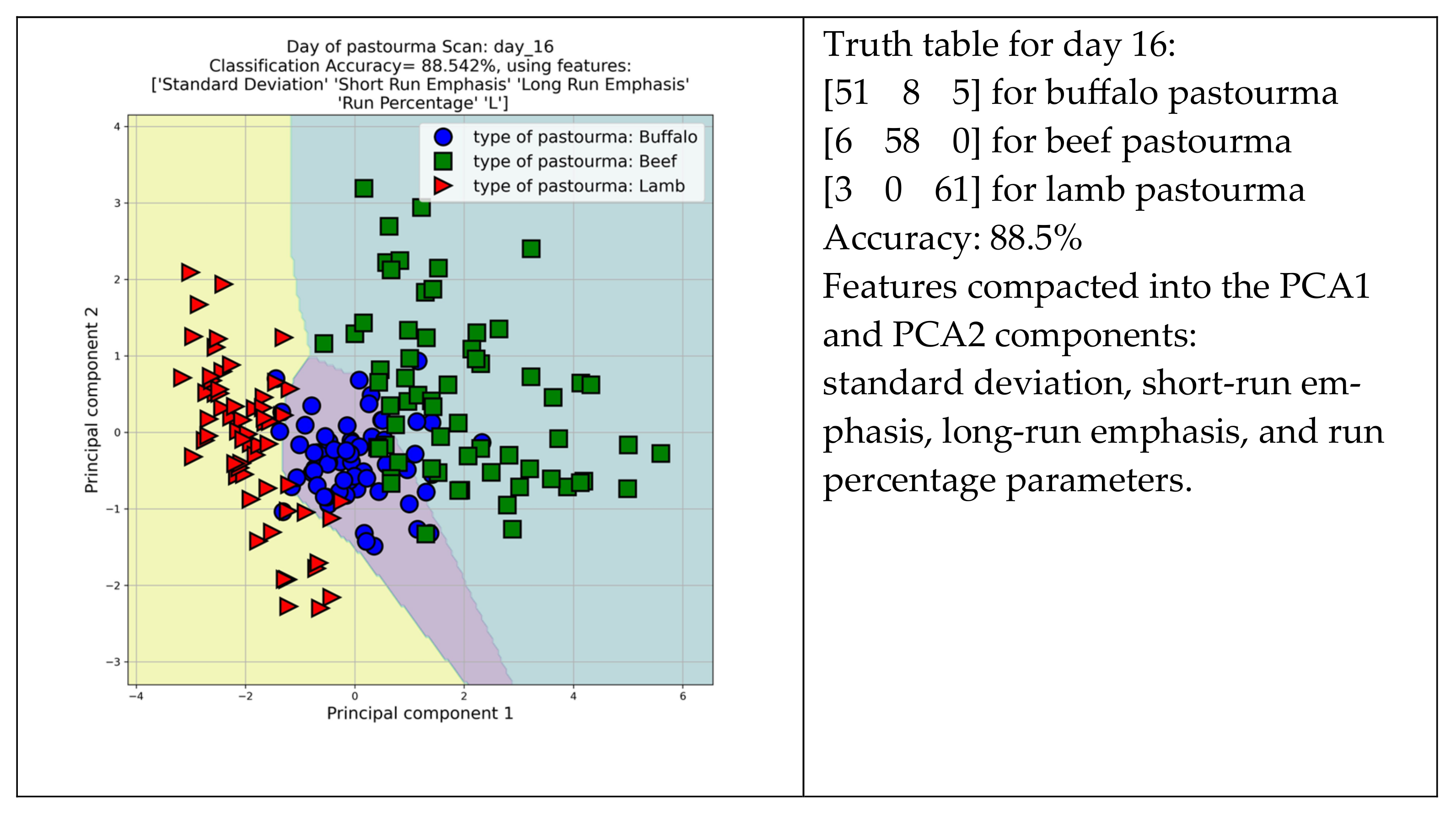





| Days of Storage | Day 1 | Day 4 | Day 8 | Day 11 | Day 14 | Day 16 |
|---|---|---|---|---|---|---|
| pastourma produced from lamb meat | ||||||
| Moisture content (%) | 45.66 ± 2.86 aA | 45.08 ± 3.35 aAΒ | 45.54 ± 2.47 aA | 37.07 ± 2.58 bA | 35.15 ± 3.80 bA | 34.93 ± 2.51 bA |
| Hue angle (h) | 20.45 ± 1.36 abA | 20.66 ± 0.65 aA | 20.29 ± 1.52 abA | 17.33 ± 2.18 bcA | 15.84 ± 1.52 cA | 14.80 ± 2.02 cA |
| Firmness (N) | 5.07 ± 0.62 aA | 5.42 ± 0.51 aA | 8.56 ± 0.25 bA | 9.25 ± 1.21 bcA | 11.07 ± 2.35 cA | 11.62 ± 1.31 cA |
| TBARS (mg malonaldehyde kg−1) | 5.10 ± 0.08 aA | 5.33 ± 0.22 aA | 4.90 ± 0.17 aA | 5.31 ± 0.22 aA | 5.00 ± 0.15 aA | 6.68 ± 0.16 bA |
| pastourma produced from beef meat | ||||||
| Moisture content (%) | 51.62 ± 2.61 aΒ | 50.77 ± 3.80 aA | 42.18 ± 0.75 bA | 42.15 ± 2.80 bB | 41.52 ± 2.87 bB | 40.37 ± 1.75 bB |
| Hue angle (h) | 26.42 ± 1.15 abB | 27.57 ± 1.56 aB | 27.90 ± 1.25 aB | 25.83 ± 2.83 abB | 25.35 ± 1.79 abB | 24.59 ± 1.55 bB |
| Firmness (N) | 3.70 ± 0.32 aB | 4.12 ± 0.36 abB | 5.06 ± 0.61 bB | 4.95 ± 0.47 bB | 5.19 ± 0.78 bB | 5.24 ± 0.59 bB |
| TBARS (mg malonaldehyde kg−1) | 0.57 ± 0.03 aB | 0.66 ± 0.04 bB | 0.84 ± 0.04 cB | 0.84 ± 0.03 cB | 0.86 ± 0.02 cB | 0.94 ± 0.03 dB |
| pastourma produced from buffalo meat | ||||||
| Moisture content (%) | 44.23 ± 3.75 aA | 39.50 ± 2.53 abB | 37.64 ± 2.44 bcB | 34.55 ± 2.62 cA | 34.18 ± 1.92 cA | 35.11 ± 1.93 cA |
| Hue angle (h) | 23.12 ± 0.99 aC | 22.05 ± 2.52 aA | 23.99 ± 2.05 aA | 22.90 ± 2.14 aB | 25.82 ± 3.75 aB | 25.16 ± 1.30 aB |
| Firmness (N) | 4.48 ± 0.19 aA | 5.46 ± 0.24 bA | 5.75 ± 0.50 bB | 8.87 ± 1.04 cA | 9.15 ± 1.80 cA | 9.08 ± 1.08 cC |
| TBARS (mg malonaldehyde kg−1) | 0.97 ± 0.04 aC | 1.23 ± 0.05 bC | 1.29 ± 0.06 bC | 1.47 ± 0.04 cC | 1.62 ± 0.05 dD | 1.65 ± 0.06 dE |
| Days of Storage | Day 1 | Day 4 | Day 8 | Day 11 | Day 14 | Day 16 |
|---|---|---|---|---|---|---|
| Regions | pastourma produced from lamb meat | |||||
| 3300 to 3500 | 0.005 ± 0.001 aA | 0.005 ± 0.001 a | 0.004 ± 0.002 a | 0.005 ± 0.001 a | 0.005 ± 0.002 a | 0.007 ± 0.001 a |
| 3287–3290 | 0.218 ± 0.008 aA | 0.207 ± 0.007 a | 0.199 ± 0.007 a | 0.192 ± 0.006 a | 0.114 ± 0.006 a | 0.088 ± 0.004 b |
| 2950–2960 | 0.031 ± 0.001 aA | 0.027 ± 0.001 a | 0.028 ± 0.003 a | 0.027 ± 0.002 a | 0.028 ± 0.006 a | 0.029 ± 0.001 a |
| 2922 | 0.129 ± 0.008 aA | 0.124 ± 0.007 a | 0.133 ± 0.011 ab | 0.145 ± 0.007 bc | 0.159 ± 0.009 c | 0.192 ± 0.011 d |
| 2870–2877 | 0.005 ± 0.001 aA | 0.008 ± 0.001 b | 0.007 ± 0.001 b | 0.008 ± 0.001 b | 0.006 ± 0.002 ab | 0.008 ± 0.002 b |
| 2854 | 0.060 ± 0.005 aA | 0.036 ± 0.003 b | 0.046 ± 0.004 c | 0.039 ± 0.002 b | 0.073 ± 0.008 d | 0.105 ± 0.007 e |
| 1743 | 0.006 ± 0.001 abA | 0.004 ± 0.001 a | 0.007 ± 0.001 b | 0.012 ± 0.000 c | 0.042 ± 0.001 d | 0.050 ± 0.001 e |
| 1728 | - | - | - | - | 0.016 ± 0.005 a | 0.031 ± 0.002 b |
| 1627–1630 | 0.452 ± 0.016 aA | 0.442 ± 0.011 a | 0.457 ± 0.030 a | 0.443 ± 0.013 a | 0.432 ± 0.016 a | 0.433 ± 0.022 a |
| 1541–1544 | 0.243 ± 0.014 aA | 0.261 ± 0.015 a | 0.258 ± 0.013 a | 0.257 ± 0.009 a | 0.247 ± 0.020 a | 0.238 ± 0.021 a |
| 1450–1452 | 0.039 ± 0.002 abA | 0.037 ± 0.001 a | 0.045 ± 0.004 b | 0.054 ± 0.003 c | 0.036 ± 0.002 a | 0.032 ± 0.002 d |
| 1392–1400 | 0.060 ± 0.003 aA | 0.059 ± 0.002 a | 0.057 ± 0.005 a | 0.057 ± 0.002 a | 0.037 ± 0.003 b | 0.031 ± 0.002 c |
| 1314 | 0.008 ± 0.001 aA | 0.008 ± 0.000 a | 0.008 ± 0.001 a | 0.009 ± 0.001 a | 0.006 ± 0.001 b | 0.007 ± 0.001 ab |
| 1238–1242 | 0.033 ± 0.003 aA | 0.030 ± 0.001 a | 0.032 ± 0.004 a | 0.032 ± 0.001 a | 0.034 ± 0.002 a | 0.030 ± 0.003 a |
| 1157–1174 | 0.011 ± 0.002 aA | 0.009 ± 0.001 ab | 0.008 ± 0.001 b | 0.008 ± 0.001 b | 0.029 ± 0.002 c | 0.025 ± 0.003 c |
| 1078–1083 | 0.023 ± 0.003 aA | 0.024 ± 0.002 a | 0.025 ± 0.003 ab | 0.029 ± 0.003 bc | 0.031 ± 0.002 c | 0.037 ± 0.003 d |
| 1060 | - | - | 0.002 ± 0.000 a | 0.010 ± 0.001 b | 0.005 ± 0.001 c | 0.001 ± 0.000 d |
| Regions | pastourma produced from beef meat | |||||
| 3300 to 3500 | 0.011 ± 0.001 aB | 0.006 ± 0.001 b | 0.006 ± 0.001 b | 0.004 ± 0.001 b | 0.006 ± 0.001 b | 0.005 ± 0.001 b |
| 3287–3290 | 0.170 ± 0.008 aB | 0.170 ± 0.007 a | 0.191 ± 0.011 b | 0.190 ± 0.012 b | 0.160 ± 0.006 ab | 0.150 ± 0.006 b |
| 2950–2960 | 0.030 ± 0.003 aA | 0.028 ± 0.001 a | 0.027 ± 0.001 a | 0.027 ± 0.001 a | 0.026 ± 0.003 a | 0.027 ± 0.001 a |
| 2922 | 0.053 ± 0.004 aB | 0.058 ± 0.002 a | 0.066 ± 0.003 b | 0.068 ± 0.002 b | 0.079 ± 0.004 c | 0.086 ± 0.003 d |
| 2870–2877 | 0.008 ± 0.001 aB | 0.008 ± 0.001 a | 0.007 ± 0.001 a | 0.007 ± 0.001 a | 0.007 ± 0.002 a | 0.007 ± 0.001 a |
| 2854 | 0.022 ± 0.003 aB | 0.015 ± 0.001 b | 0.010 ± 0.002 c | 0.031 ± 0.001 d | 0.028 ± 0.002 d | 0.018 ± 0.001 e |
| 1743 | 0.024 ± 0.002 aB | 0.012 ± 0.001 b | 0.006 ± 0.001 c | 0.003 ± 0.001 d | 0.003 ± 0.001 d | 0.004 ± 0.001 d |
| 1728 | 0.015 ± 0.001 A | - | - | - | - | - |
| 1627–1630 | 0.487 ± 0.012 aB | 0.442 ± 0.011 b | 0.444 ± 0.015 b | 0.442 ± 0.009 b | 0.443 ± 0.017 b | 0.440 ± 0.012 b |
| 1541–1544 | 0.261 ± 0.007 aA | 0.256 ± 0.012 ab | 0.256 ± 0.009 ab | 0.243 ± 0.011 bc | 0.241 ± 0.007 c | 0.279 ± 0.009 d |
| 1450–1452 | 0.019 ± 0.002 aB | 0.028 ± 0.003 b | 0.036 ± 0.002 cd | 0.034 ± 0.002 cd | 0.032 ± 0.002 c | 0.037 ± 0.002 d |
| 1392–1400 | 0.053 ± 0.004 aA | 0.056 ± 0.006 a | 0.056 ± 0.002 a | 0.055 ± 0.003 a | 0.051 ± 0.002 a | 0.056 ± 0.003 a |
| 1314 | 0.008 ± 0.001 aA | 0.009 ± 0.001 a | 0.009 ± 0.001 a | 0.009 ± 0.002 a | 0.008 ± 0.001 a | 0.009 ± 0.001 a |
| 1238–1242 | 0.028 ± 0.002 aA | 0.033 ± 0.003 b | 0.031 ± 0.002 ab | 0.030 ± 0.001 ab | 0.030 ± 0.003 ab | 0.031 ± 0.002 ab |
| 1157–1174 | 0.009 ± 0.002 abA | 0.007 ± 0.001 a | 0.009 ± 0.001 ab | 0.011 ± 0.002 b | 0.009 ± 0.001 ab | 0.011 ± 0.001 b |
| 1078–1083 | 0.027 ± 0.003 aA | 0.025 ± 0.002 a | 0.018 ± 0.003 b | 0.019 ± 0.002 b | 0.019 ± 0.003 b | 0.012 ± 0.001 c |
| 1060 | - | - | - | 0.002 ± 0.000 a | 0.003 ± 0.000 b | 0.008 ± 0.001 c |
| Regions | pastourma produced from buffalo | |||||
| 3300 to 3500 | 0.004 ± 0.001 aA | 0.004 ± 0.001 a | 0.005 ± 0.001 a | 0.005 ± 0.001 a | 0.004 ± 0.001 a | 0.004 ± 0.001 a |
| 3287–3290 | 0.122 ± 0.005 aC | 0.115 ± 0.004 a | 0.105 ± 0.005 b | 0.105 ± 0.004 b | 0.104 ± 0.006 b | 0.052 ± 0.004 c |
| 2950–2960 | 0.026 ± 0.002 aB | 0.027 ± 0.001 a | 0.025 ± 0.003 a | 0.027 ± 0.001 a | 0.024 ± 0.002 a | 0.025 ± 0.002 a |
| 2922 | 0.052 ± 0.003 aB | 0.059 ± 0.002 b | 0.070 ± 0.005 c | 0.071 ± 0.007 c | 0.076 ± 0.003 c | 0.051 ± 0.005 a |
| 2870–2877 | 0.017 ± 0.001 aC | 0.017 ± 0.001 a | 0.014 ± 0.001 b | 0.010 ± 0.001 c | 0.007 ± 0.001 d | 0.007 ± 0.000 d |
| 2854 | 0.019 ± 0.001 aB | 0.023 ± 0.002 b | 0.026 ± 0.002 b | 0.031 ± 0.002 c | 0.032 ± 0.001 c | 0.033 ± 0.001 c |
| 1743 | 0.020 ± 0.002 aB | 0.007 ± 0.001 b | 0.006 ± 0.001 b | 0.005 ± 0.001 b | 0.003 ± 0.001 c | 0.002 ± 0.000 c |
| 1728 | - | - | - | - | - | - |
| 1627–1630 | 0.469 ± 0.024 aAB | 0.463 ± 0.012 a | 0.441 ± 0.014 a | 0.449 ± 0.017 a | 0.459 ± 0.015 a | 0.444 ± 0.018 a |
| 1541–1544 | 0.257 ± 0.011 aA | 0.268 ± 0.011 a | 0.257 ± 0.009 a | 0.253 ± 0.012 a | 0.226 ± 0.009 b | 0.232 ± 0.008 b |
| 1450–1452 | 0.041 ± 0.004 aA | 0.034 ± 0.002 b | 0.032 ± 0.001 b | 0.031 ± 0.002 b | 0.033 ± 0.002 b | 0.025 ± 0.003 c |
| 1392–1400 | 0.060 ± 0.004 aA | 0.062 ± 0.004 a | 0.058 ± 0.002 a | 0.060 ± 0.003 a | 0.056 ± 0.002 a | 0.058 ± 0.003 a |
| 1314 | 0.008 ± 0.001 aA | 0.009 ± 0.001 a | 0.008 ± 0.001 a | 0.009 ± 0.001 a | 0.008 ± 0.002 a | 0.009 ± 0.001 a |
| 1238–1242 | 0.039 ± 0.002 aB | 0.031 ± 0.002 b | 0.029 ± 0.001 b | 0.030 ± 0.002 b | 0.028 ± 0.002 b | 0.029 ± 0.002 b |
| 1157–1174 | 0.019 ± 0.001 aB | 0.005 ± 0.001 b | 0.005 ± 0.001 b | 0.007 ± 0.001 b | 0.006 ± 0.001 b | 0.005 ± 0.001 b |
| 1078–1083 | 0.026 ± 0.003 aA | 0.022 ± 0.003 a | 0.014 ± 0.003 b | 0.018 ± 0.002 b | 0.015 ± 0.002 b | 0.026 ± 0.004 a |
| 1060 | 0.002 ± 0.000 a | 0.004 ± 0.001 b | 0.007 ± 0.001 c | 0.003 ± 0.001 b | 0.005 ± 0.001 bc | 0.004 ± 0.001 b |
| Protein Molecules Secondary Structure (%) | β-Parallel Sheet (%) | Random Coil (%) | α-Helix (%) | β-Turn (%) | β-Antiparallel Sheet (%) |
|---|---|---|---|---|---|
| pastourma produced from lamb meat | |||||
| Day 1 | 9.42 ± 0.50 a | 18.64 ± 0.35 a | 10.16 ± 0.61 a | 38.70 ± 1.31 a | 23.08 ± 1.08 a |
| Day 16 | 6.82 ± 0.27 b | 13.96 ± 0.34 b | 7.05 ± 0.23 b | 39.55 ± 1.24 a | 32.62 ± 1.39 b |
| pastourma produced from beef meat | |||||
| Day 1 | 7.38 ± 0.82 b | 17.68 ± 0.46 c | 5.97 ± 0.31 c | 31.91 ± 1.12 ce | 37.07 ± 1.25 c |
| Day 16 | 7.03 ± 0.56 b | 16.82 ± 0.63 c | 8.40 ± 0.56 d | 37.89 ± 1.42 a | 29.85 ± 1.11 d |
| pastourma produced from buffalo meat | |||||
| Day 1 | 7.06 ± 0.55 b | 14.58 ± 0.63 bd | 6.57 ± 0.46 cb | 29.48 ± 1.09 d | 42.30 ± 1.05 e |
| Day 16 | 6.39 ± 0.69 b | 15.59 ± 0.55 d | 11.25 ± 0.53 a | 33.50 ± 1.49 e | 33.26 ± 0.91 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kritsi, E.; Ladika, G.; Stavropoulou, N.A.; Oikonomakou, M.; Ioannou, A.-G.; Christodoulou, P.; Konteles, S.J.; Cavouras, D.; Sinanoglou, V.J. Evaluation of the Quality Changes in Three Commercial Pastourma Samples during Refrigerated Storage Using Physicochemical, Microbiological, and Image Analyses Combined with Chemometrics. Foods 2024, 13, 1017. https://doi.org/10.3390/foods13071017
Kritsi E, Ladika G, Stavropoulou NA, Oikonomakou M, Ioannou A-G, Christodoulou P, Konteles SJ, Cavouras D, Sinanoglou VJ. Evaluation of the Quality Changes in Three Commercial Pastourma Samples during Refrigerated Storage Using Physicochemical, Microbiological, and Image Analyses Combined with Chemometrics. Foods. 2024; 13(7):1017. https://doi.org/10.3390/foods13071017
Chicago/Turabian StyleKritsi, Eftichia, Georgia Ladika, Natalia A. Stavropoulou, Marianna Oikonomakou, Alexandros-George Ioannou, Paris Christodoulou, Spyridon J. Konteles, Dionisis Cavouras, and Vassilia J. Sinanoglou. 2024. "Evaluation of the Quality Changes in Three Commercial Pastourma Samples during Refrigerated Storage Using Physicochemical, Microbiological, and Image Analyses Combined with Chemometrics" Foods 13, no. 7: 1017. https://doi.org/10.3390/foods13071017
APA StyleKritsi, E., Ladika, G., Stavropoulou, N. A., Oikonomakou, M., Ioannou, A.-G., Christodoulou, P., Konteles, S. J., Cavouras, D., & Sinanoglou, V. J. (2024). Evaluation of the Quality Changes in Three Commercial Pastourma Samples during Refrigerated Storage Using Physicochemical, Microbiological, and Image Analyses Combined with Chemometrics. Foods, 13(7), 1017. https://doi.org/10.3390/foods13071017






