Savory and Peppermint Essential Oils-Loaded Emulsions and Nanoemulsions Effects on Enterococcus faecium Isolated from Vacuum-Packed Cured Sausage
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples Collection
2.2. Isolation of LAB Strains
2.2.1. Biochemical Characterization of Isolated Strains
2.2.2. DNA Extraction of Isolated Strains
2.2.3. Molecular Identification
2.3. Essential Oil Extraction
GC-FID Analysis of EOs
2.4. Emulsion and Nanoemulsion Preparation
2.4.1. Droplet Size and Zeta Potential
2.4.2. Storage Stability
2.4.3. Gravitational Stability
2.4.4. Antimicrobial Activity
Agar Diffusion Method
MIC and MBC
Antibacterial Dynamics
2.4.5. Transmission Electron Microscopic (TEM)
2.4.6. Scanning Electron Microscope
2.5. Statistical Analysis
3. Results and Discussion
3.1. Bacterial Identification
3.2. Yield and Chemical Composition of EO
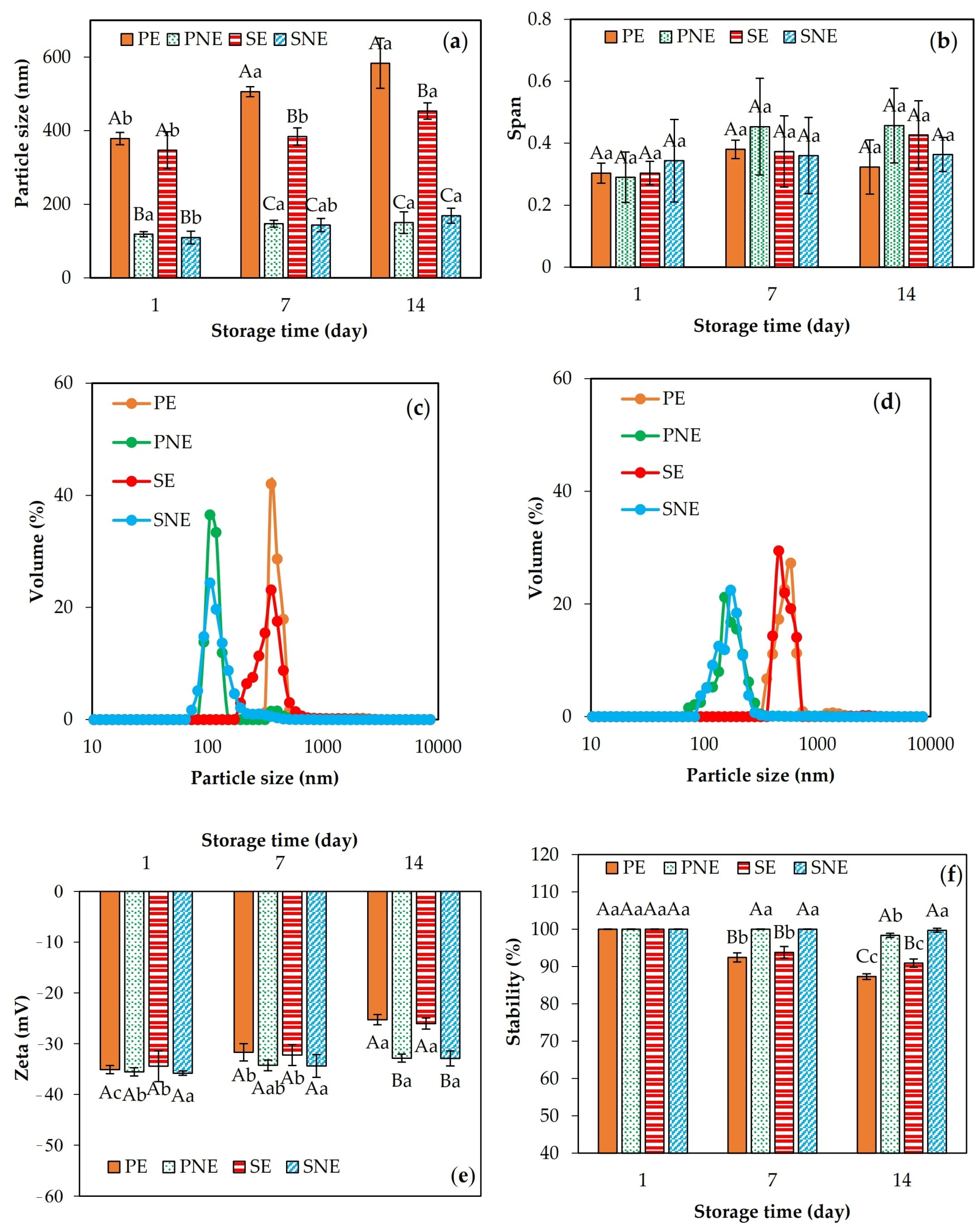
3.3. Emulsion Characterization
3.4. Antibacterial Assay
3.4.1. Agar Diffusion Method, Minimum Inhibitory Concentration, and Minimum Bactericidal Concentration
3.4.2. Antibacterial Dynamics
3.4.3. Transmission Electron Microscopy
3.4.4. Scanning Electron Microscopy
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khorsandi, A.; Eskandari, M.H.; Aminlari, M.; Shekarforoush, S.S.; Golmakani, M.T. Shelf-life extension of vacuum packed emulsion-type sausage using combination of natural antimicrobials. Food Control 2019, 104, 139–146. [Google Scholar] [CrossRef]
- Khorsandi, A.; Ziaee, E.; Shad, E.; Razmjooei, M.; Eskandari, M.H.; Aminlari, M. Antibacterial Effect of Essential Oils against Spoilage Bacteria from Vacuum-Packed Cooked Cured Sausages. J. Food Prot. 2018, 81, 1386–1393. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Jia, J.; Peng, H.; Qian, Q.; Pan, Z.; Liu, D. Nitrite and nitrate in meat processing: Functions and alternatives. Curr. Res. Food Sci. 2023, 24, 100470. [Google Scholar] [CrossRef]
- Kröckel, L. The role of lactic acid bacteria in safety and flavour development of meat and meat products. In Lactic Acid Bacteria—R & D for Food, Health and Livestock Purposes; IntechOpen: London, UK, 2013. [Google Scholar]
- Dodds, K.L.; Collins-Thompson, D.L. Nitrite tolerance and nitrite reduction in lactic acid bacteria associated with cured meat products. Int. J. Food Microbiol. 1984, 1, 163–170. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Asl, R.M.Z.; Niakousari, M.; Gahruie, H.H.; Saharkhiz, M.J.; Khaneghah, A.M. Study of two-stage ohmic hydro-extraction of essential oil from Artemisia aucheri Boiss.: Antioxidant and antimicrobial characteristics. Food Res. Int. 2018, 107, 462–469. [Google Scholar]
- Hashemi Gahruie, H.; Ziaee, E.; Eskandari, M.H.; Hosseini, S.M.H. Characterization of basil seed gum-based edible films incorporated with Zataria multiflora essential oil nanoemulsion. Carbohyd. Pol. 2017, 166, 93–103. [Google Scholar] [CrossRef]
- Ghiasi, F.; Eskandari, M.H.; Golmakani, M.-T.; Hosseini, S.M.H. Development of highly stable colloidal dispersions of gelled-oil nanoparticles loaded with cuminaldehyde. J. Colloid Interface Sci. 2019, 541, 65–74. [Google Scholar] [CrossRef]
- Radünz, M.; Hackbart, H.C.D.S.; Camargo, T.M.; Nunes, C.F.P.; de Barros, F.A.P.; Magro, J.D.; Filho, P.J.S.; Gandra, E.A.; Radünz, A.L.; Zavareze, E.D.R. Antimicrobial potential of spray drying encapsulated thyme (Thymus vulgaris) essential oil on the conservation of hamburger-like meat products. Int. J. Food Microbiol. 2020, 330, 108696. [Google Scholar] [CrossRef] [PubMed]
- Ghiasi, F.; Eskandari, M.H.; Golmakani, M.T.; Gahruie, H.H.; Zarei, R.; Naghibalhossaini, F.; Hosseini, S.M.H. A novel promising delivery system for cuminaldehyde using gelled lipid nanoparticles: Characterization and anticancer, antioxidant, and antibacterial activities. Int. J. Pharm. 2021, 610, 121274. [Google Scholar] [CrossRef]
- Niakousari, M.; Damyeh, M.S.; Gahruie, H.H.; Bekhit, A.E.D.A.; Greiner, R.; Roohinejad, S. Conventional Emulsions. In Emulsion-Based Systems for Delivery of Food Active Compounds: Formation, Application, Health and Safety; John Wiley & Sons Ltd.: Croydon, UK, 2018; pp. 1–27. [Google Scholar]
- Tastan, Ö.; Ferrari, G.; Baysal, T.; Donsì, F. Understanding the effect of formulation on functionality of modified chitosan films containing carvacrol nanoemulsions. Food Hydrocoll. 2016, 61, 756–771. [Google Scholar] [CrossRef]
- Chen, H.; Hu, X.; Chen, E.; Wu, S.; McClements, D.J.; Liu, S.; Li, B.; Li, Y. Preparation, characterization, and properties of chitosan films with cinnamaldehyde nanoemulsions. Food Hydrocoll. 2016, 61, 662–671. [Google Scholar] [CrossRef]
- Liew, S.N.; Utra, U.; Alias, A.K.; Tan, T.B.; Tan, C.P.; Yussof, N.S. Physical, morphological and antibacterial properties of lime essential oil nanoemulsions prepared via spontaneous emulsification method. LWT 2020, 128, 109388. [Google Scholar] [CrossRef]
- Acevedo-Fani, A.; Salvia-Trujillo, L.; Rojas-Graü, M.A.; Martín-Belloso, O. Edible films from essential-oil-loaded nanoemulsions: Physicochemical characterization and antimicrobial properties. Food Hydrocoll. 2015, 47, 168–177. [Google Scholar] [CrossRef]
- Shahbazi, H.; Hashemi Gahruie, H.; Golmakani, M.T.; Eskandari, M.H.; Movahedi, M. Effect of medicinal plant type and concentration on physicochemical, antioxidant, antimicrobial, and sensorial properties of kombucha. Food Sci. Nutr. 2018, 6, 2568–2577. [Google Scholar] [CrossRef]
- Diao, W.R.; Hu, Q.P.; Feng, S.S.; Li, W.Q.; Xu, J.G. Chemical composition and antibacterial activity of the essential oil from green huajiao (Zanthoxylum schinifolium) against selected foodborne pathogens. J. Agric. Food Chem. 2013, 61, 6044–6049. [Google Scholar] [CrossRef]
- Hu, P.; Zhou, G.; Xu, X.; Li, C.; Han, Y. Characterization of the predominant spoilage bacteria in sliced vacuum-packed cooked ham based on 16S rDNA-DGGE. Food Control 2009, 20, 99–104. [Google Scholar] [CrossRef]
- Qian, K.; Zhou, T. Identification of spoilage bacteria isolated from sausage. Sci. Technol. Food Ind. 2008, 4, 124–130. [Google Scholar]
- Memarzadeh, S.M.; Ghasemi Pirbalouti, A.; AdibNejad, M. Chemical composition and yield of essential oils from Bakhtiari savory (Satureja bachtiarica Bunge.) under different extraction methods. Ind. Crop. Prod. 2015, 76, 809–816. [Google Scholar] [CrossRef]
- Šojić, B.; Pavlić, B.; Tomović, V.; Ikonić, P.; Zeković, Z.; Kocić-Tanackov, S.; Đurović, S.; Škaljac, S.; Jokanović, M.; Ivić, M. Essential oil versus supercritical fluid extracts of winter savory (Satureja montana L.)—Assessment of the oxidative, microbiological and sensory quality of fresh pork sausages. Food Chem. 2019, 287, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Khalvandi, M.; Amerian, M.; Pirdashti, H.; Keramati, S.; Hosseini, J. Essential oil of peppermint in symbiotic relationship with Piriformospora indica and methyl jasmonate application under saline condition. Ind. Crop. Prod. 2019, 127, 195–202. [Google Scholar] [CrossRef]
- Kang, J.; Jin, W.; Wang, J.; Sun, Y.; Wu, X.; Liu, L. Antibacterial and anti-biofilm activities of peppermint essential oil against Staphylococcus aureus. LWT 2019, 101, 639–645. [Google Scholar] [CrossRef]
- Hashtjin, A.M.; Abbasi, S. Optimization of ultrasonic emulsification conditions for the production of orange peel essential oil nanoemulsions. J. Food Sci. Technol. 2015, 52, 2679–2689. [Google Scholar] [CrossRef]
- Taha, A.; Ahmed, E.; Ismaiel, A.; Ashokkumar, M.; Xu, X.; Pan, S.; Hu, H. Ultrasonic emulsification: An overview on the preparation of different emulsifiers-stabilized emulsions. Trends Food Sci. Technol. 2020, 105, 363–377. [Google Scholar] [CrossRef]
- Jafari, S.; He, Y.; Bhandari, B. Nano-emulsion production by sonication and microfluidization—A comparison. Int. J. Food Prop. 2006, 9, 475–485. [Google Scholar] [CrossRef]
- Hashtjin, A.M.; Abbasi, S. Nano-emulsification of orange peel essential oil using sonication and native gums. Food Hydrocoll. 2015, 44, 40–48. [Google Scholar] [CrossRef]
- Elaissi, A.; Rouis, Z.; Mabrouk, S.; Salah, K.B.H.; Aouni, M.; Khouja, M.L.; Farhat, F.; Chemli, R.; Harzallah-Skhiri, F. Correlation between chemical composition and antibacterial activity of essential oils from fifteen Eucalyptus species growing in the Korbous and Jbel Abderrahman Arboreta (North East Tunisia). Molecules 2012, 17, 3044–3057. [Google Scholar] [CrossRef] [PubMed]
- Abdollahi, M.; Damirchi, S.; Shafafi, M.; Rezaei, M.; Ariaii, P. Carboxymethyl cellulose-agar biocomposite film activated with summer savory essential oil as an antimicrobial agent. Int. J. Biol. Macromol. 2019, 126, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Atef, M.; Rezaei, M.; Behrooz, R. Characterization of physical, mechanical, and antibacterial properties of agar-cellulose bionanocomposite films incorporated with savory essential oil. Food Hydrocoll. 2015, 45, 150–157. [Google Scholar] [CrossRef]
- Feyzioglu, G.C.; Tornuk, F. Development of chitosan nanoparticles loaded with summer savory (Satureja hortensis L.) essential oil for antimicrobial and antioxidant delivery applications. LWT 2016, 70, 104–110. [Google Scholar] [CrossRef]
- Miladi, H.; Zmantar, T.; Kouidhi, B.; Al Qurashi, Y.M.A.; Bakhrouf, A.; Chaabouni, Y.; Mahdouani, K.; Chaieb, K. Synergistic effect of eugenol, carvacrol, thymol, p-cymene and γ-terpinene on inhibition of drug resistance and biofilm formation of oral bacteria. Microb. Pathog. 2017, 112, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Yoshitomi, K.; Taniguchi, S.; Tanaka, K.; Uji, Y.; Akimitsu, K.; Gomi, K. Rice terpene synthase 24 (OsTPS24) encodes a jasmonate-responsive monoterpene synthase that produces an antibacterial γ-terpinene against rice pathogen. J. Plant Physiol. 2016, 191, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Dhar, P.; Chan, P.; Cohen, D.T.; Khawam, F.; Gibbons, S.; Snyder-Leiby, T.; Dickstein, E.; Rai, P.K.; Watal, G. Synthesis, antimicrobial evaluation, and structure–activity relationship of α-pinene derivatives. J. Agric. Food Chem. 2014, 62, 3548–3552. [Google Scholar] [CrossRef]
- Merghni, A.; Haddaji, N.; Bouali, N.; Alabbosh, K.F.; Adnan, M.; Snoussi, M.; Noumi, E. Comparative study of antibacterial, antibiofilm, antiswarming and antiquorum sensing activities of Origanum vulgare essential oil and terpinene-4-ol against pathogenic bacteria. Life 2022, 12, 1616. [Google Scholar] [CrossRef]
- Niza, E.; Božik, M.; Bravo, I.; Clemente-Casares, P.; Lara-Sanchez, A.; Juan, A.; Klouček, P.; Alonso-Moreno, C. PEI-coated PLA nanoparticles to enhance the antimicrobial activity of carvacrol. Food Chem. 2020, 328, 127131. [Google Scholar] [CrossRef]
- Farhanghi, A.; Aliakbarlu, J.; Tajik, H.; Mortazavi, N.; Manafi, L.; Jalilzadeh-Amin, G. Antibacterial interactions of pulegone and 1, 8-cineole with monolaurin ornisin against Staphylococcus aureus. Food Sci. Nutr. 2022, 10, 2659–2666. [Google Scholar] [CrossRef]
- Ghazal, T.S.A.; Schelz, Z.; Vidács, L.; Szemerédi, N.; Veres, K.; Spengler, G.; Hohmann, J. Antimicrobial, Multidrug Resistance Reversal and Biofilm Formation Inhibitory Effect of Origanum majorana Extracts, Essential Oil and Monoterpenes. Plants 2022, 11, 1432. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, C.; Zhang, N.; Peng, Y.; Ma, Y.; Gu, K.; Liu, X.; Liu, X.; Liu, X.; Liu, Y. Menthone Exerts its Antimicrobial Activity Against Methicillin Resistant Staphylococcus aureus by Affecting Cell Membrane Properties and Lipid Profile. Drug Des. Dev. Ther. 2023, 17, 219–236. [Google Scholar] [CrossRef]
- Tutar, U.; Çelik, C.; Karaman, İ.; Ataş, M.; Hepokur, C. Anti-biofilm and antimicrobial activity of Mentha pulegium L essential oil against multidrug-resistant Acinetobacter baumannii. Trop. J. Pharm. Res. 2016, 15, 1039–1046. [Google Scholar] [CrossRef]
- Jahdkaran, E.; Hosseini, S.E.; Mohammadi Nafchi, A.; Nouri, L. The effects of methylcellulose coating containing carvacrol or menthol on the physicochemical, mechanical, and antimicrobial activity of polyethylene films. Food Sci. Nutr. 2021, 9, 2768–2778. [Google Scholar] [CrossRef] [PubMed]
- Angane, M.; Swift, S.; Huang, K.; Butts, C.A.; Quek, S.Y. Essential oils and their major components: An updated review on antimicrobial activities, mechanism of action and their potential application in the food industry. Foods 2022, 4, 464. [Google Scholar] [CrossRef]
- Osanloo, M.; Abdollahi, A.; Valizadeh, A.; Abedinpour, N. Antibacterial potential of essential oils of Zataria multiflora and Mentha piperita, micro-and nano-formulated forms. Iran. J. Microbiol. 2020, 12, 43. [Google Scholar] [CrossRef]
- Ziaee, E.; Razmjooei, M.; Shad, E.; Eskandari, M.H. Antibacterial mechanisms of Zataria multiflora Boiss. essential oil against Lactobacillus curvatus. LWT 2018, 87, 406–412. [Google Scholar] [CrossRef]
- Chen, Z.; He, B.; Zhou, J.; He, D.; Deng, J.; Zeng, R. Chemical compositions and antibacterial activities of essential oils extracted from Alpinia guilinensis against selected foodborne pathogens. Ind. Crop. Prod. 2016, 83, 607–613. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Wang, Y.; Jiang, P.; Quek, S. Antibacterial activity and mechanism of cinnamon essential oil against Escherichia coli and Staphylococcus aureus. Food Control 2016, 59, 282–289. [Google Scholar] [CrossRef]
- Chauhan, A.K.; Kang, S.C. Thymol disrupts the membrane integrity of Salmonella ser. typhimurium in vitro and recovers infected macrophages from oxidative stress in an ex vivo model. Res. Microbiol. 2014, 165, 559–565. [Google Scholar] [CrossRef]


| Molecular Analysis | Morphological Analysis a | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Group No. | Bacteria | ID% | Strain Code | Accession No. | Pentose Fermentation | Gas Production | Catalase | Oxidase | Gram Reaction | Spore |
| 1 | Enterococcus faecium | 97 | HHG6801 | Mk757986 | - | - | - | - | + | - |
| 2 | Lactobacillus Dextrinicus | 98 | HHG1396 | MK757985 | - | + | - | - | + | - |
| 3 | Enterococcus faecium | 99 | HHG1368 | MK447744 | - | - | - | - | + | - |
| Savory EO | Peppermint EO | ||||
|---|---|---|---|---|---|
| RT (min) | % | RT (min) | % | ||
| α-Thujene | 6.439 | 0.3379 | 1,8-Cineole | 3.044 | 17.64 |
| α-Pinene | 8.404 | 1.065 | γ-Terpinene | 3.607 | 0.9653 |
| β-Pinene | 11.494 | 0.09548 | cis-Sabinene hydrate | 4.629 | 2.388 |
| β-Myrcene | 12.676 | 0.2385 | Linalol | 4.847 | 0.6009 |
| α-Terpinene | 14.999 | 0.8001 | Menthone | 6.402 | 23.73 |
| p-Cymene | 16.418 | 0.1942 | Menthofuran | 8.769 | 1.729 |
| γ-Terpinene | 20.887 | 82.98 | neo-Menthol | 10.919 | 1.093 |
| Terpinene-4-ol | 21.767 | 2.404 | Isopulegone | 11.377 | 1.359 |
| Thymol | 23.226 | 0.2229 | Isomenthone | 12.964 | 12.04 |
| Carvacrol | 27.038 | 7.558 | Isomenthol | 13.804 | 2.74 |
| Carvacryl acetate | 28.768 | 0.07459 | Menthol | 16.001 | 30.09 |
| β-Caryophyllene | 32.618 | 0.6798 | Terpinen-4-ol | 16.611 | 0.2562 |
| Aromadendrene | 37.799 | 0.5519 | Pulegone | 17.227 | 0.3644 |
| α-Humulen | 42.783 | 0.5318 | Piperitone | 18.031 | 0.2808 |
| β-Bisabolene | 47.584 | 0.4583 | p-Menth-1-en-9-ol | 19.349 | 1.25 |
| cis-α-Bisabolene | 52.127 | 0.9014 | β-Bourbonene | 19.925 | 0.5337 |
| Spathulenol | 56.935 | 0.9019 | (E)-Caryophyllene | 21.575 | 1.283 |
| (E)-β-Farnesene | 22.997 | 0.601 | |||
| Germacrene D | 25.834 | 0.2366 | |||
| Elixene | 28.544 | 0.1623 | |||
| Viridiflorol | 31.012 | 0.6576 | |||
| Treat | MIC (µL/mL) | MBC (µL/mL) | DIZ (mm) | ||
|---|---|---|---|---|---|
| Emulsion | EO | Emulsion | EO | ||
| PNE | 15.00± 3.06 B | 900 | 30.00± 6.12 A | 1800 | 6.30 ± 0.40 C |
| PE | 30.00 ± 6.12 A | 1800 | 30.00 ± 6.12 A | 1800 | 5.10 ± 0.30 D |
| SNE | 1.88 ± 0.39 D | 112.8 | 3.75 ± 0.77 C | 225 | 12.50± 0.70 A |
| SE | 3.75 ± 0.97 C | 225 | 7.50 ± 1.92 B | 450 | 10.30 ± 0.30 B |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hashemi, H.; Shad, E.; Ghiasi, F.; Eskandari, M.H. Savory and Peppermint Essential Oils-Loaded Emulsions and Nanoemulsions Effects on Enterococcus faecium Isolated from Vacuum-Packed Cured Sausage. Foods 2024, 13, 341. https://doi.org/10.3390/foods13020341
Hashemi H, Shad E, Ghiasi F, Eskandari MH. Savory and Peppermint Essential Oils-Loaded Emulsions and Nanoemulsions Effects on Enterococcus faecium Isolated from Vacuum-Packed Cured Sausage. Foods. 2024; 13(2):341. https://doi.org/10.3390/foods13020341
Chicago/Turabian StyleHashemi, Hadi, Ehsan Shad, Fatemeh Ghiasi, and Mohammad Hadi Eskandari. 2024. "Savory and Peppermint Essential Oils-Loaded Emulsions and Nanoemulsions Effects on Enterococcus faecium Isolated from Vacuum-Packed Cured Sausage" Foods 13, no. 2: 341. https://doi.org/10.3390/foods13020341
APA StyleHashemi, H., Shad, E., Ghiasi, F., & Eskandari, M. H. (2024). Savory and Peppermint Essential Oils-Loaded Emulsions and Nanoemulsions Effects on Enterococcus faecium Isolated from Vacuum-Packed Cured Sausage. Foods, 13(2), 341. https://doi.org/10.3390/foods13020341







