Inactivation of Escherichia coli in an Orange Juice Beverage by Combined Ultrasonic and Microwave Treatment
Abstract
1. Introduction
2. Results and Discussion
2.1. Box–Behnken Analysis of E. coli Inactivation
2.2. Performance and Fit of the RSM Model
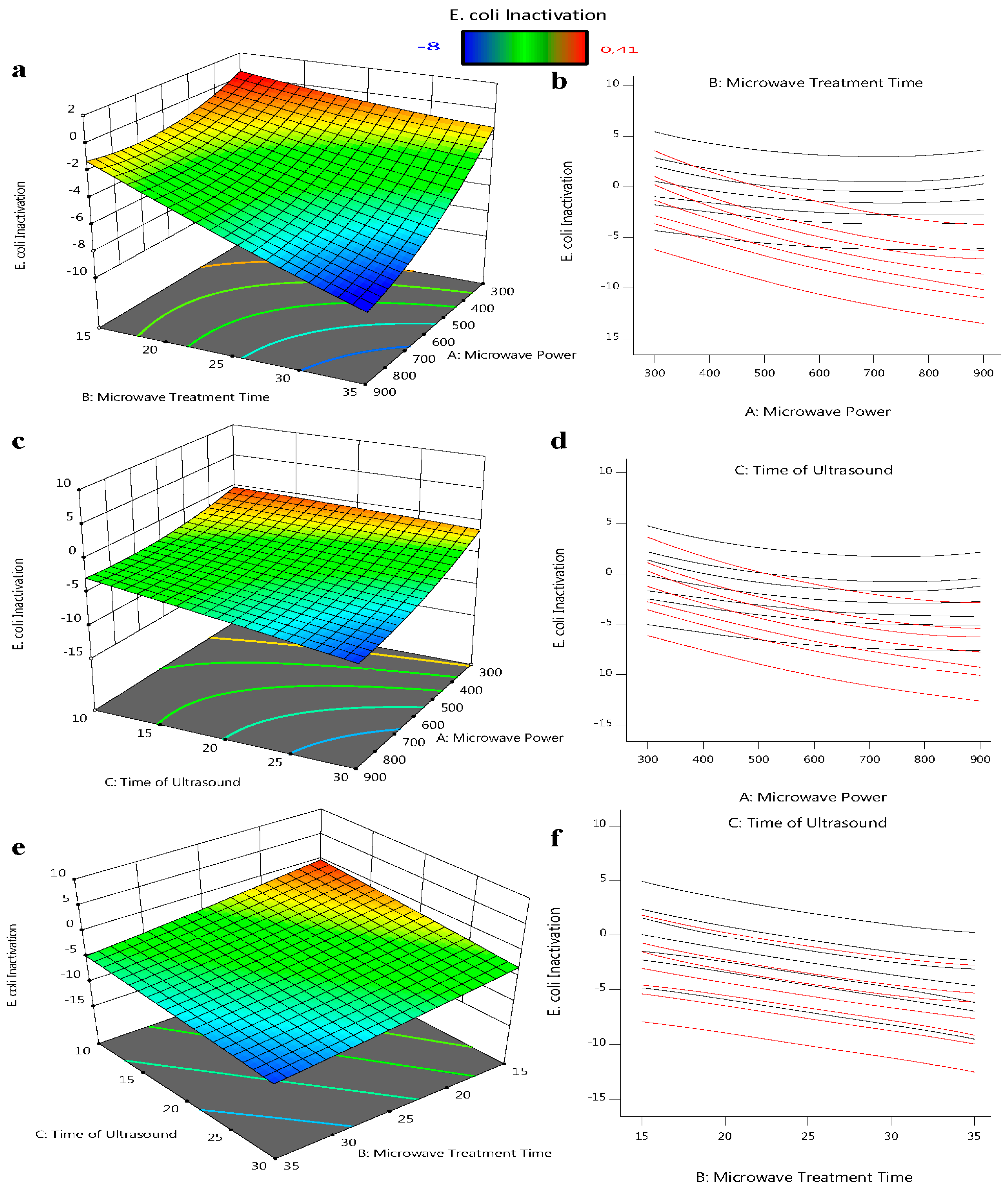
2.3. Analysis of Interactions between Influencing Factors
2.4. Validation of the Model
3. Materials and Methods
3.1. Conditions for Culture and Bacterial Strain
3.2. Inoculation of Orange Juice
3.3. Procedure of Treatment by Microwave and Ultrasound
3.4. Enumeration of Survival Cells
3.5. Optimal Inactivation of E. coli Using the RSM Method and the Box–Behnken Design Enumeration of Survival Cells
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lima Tribst, A.A.; de Souza Sant’Ana, A.; de Massaguer, P.R. Microbiological quality and safety of fruit juices—Past, present and future perspectives. Crit. Rev. Microbiol. 2009, 35, 310–339. [Google Scholar] [CrossRef]
- Standard, G. Microbiological Criteria for Food Stuffs-Part 1; GCC: Riyadh, Saudi Arabia, 2000; pp. 7–20. [Google Scholar]
- Yap, M.; Ercolini, D.; Álvarez-Ordóñez, A.; O’Toole, P.W.; O’Sullivan, O.; Cotter, P.D. Next-generation food research: Use of meta-omic approaches for characterizing microbial communities along the food chain. Annu. Rev. Food Sci. Technol. 2022, 13, 361–384. [Google Scholar] [CrossRef]
- Badenhorst, A.B. The Risk of Pathogenic Microbiological Contamination of South African Fresh Fruit for the Export and Local Market. Ph.D. Thesis, Stellenbosch University, Stellenbsoch, South Africa, 2020. [Google Scholar]
- Foster, J.W. Acid stress responses of Salmonella and E. coli: Survival mechanisms, regulation, and implications for pathogenesis. J. Microbiol. 2001, 39, 89–94. [Google Scholar]
- Pal Roy, M.; Mazumdar, D.; Dutta, S.; Saha, S.P.; Ghosh, S. Cloning and expression of phytase appA gene from Shigella sp. CD2 in Pichia pastoris and comparison of properties with recombinant enzyme expressed in E. coli. PLoS ONE 2016, 11, e0145745. [Google Scholar] [CrossRef]
- Wang, Z.; Fang, Y.; Zhi, S.; Simpson, D.J.; Gill, A.; McMullen, L.M.; Neumann, N.F.; Gänzle, M.G. The locus of heat resistance confers resistance to chlorine and other oxidizing chemicals in Escherichia coli. Appl. Environ. Microbiol. 2020, 86, e02123-19. [Google Scholar] [CrossRef]
- Toepfl, S.; Heinz, V.; Knorr, D. High intensity pulsed electric fields applied for food preservation. Chem. Eng. Process. Process Intensif. 2007, 46, 537–546. [Google Scholar] [CrossRef]
- Patil, S.; Bourke, P.; Frias, J.M.; Tiwari, B.; Cullen, P. Inactivation of Escherichia coli in orange juice using ozone. Innov. Food Sci. Emerg. Technol. 2009, 10, 551–557. [Google Scholar] [CrossRef]
- Tahiri, I.; Makhlouf, J.; Paquin, P.; Fliss, I. Inactivation of food spoilage bacteria and Escherichia coli O157: H7 in phosphate buffer and orange juice using dynamic high pressure. Food Res. Int. 2006, 39, 98–105. [Google Scholar] [CrossRef]
- Torres, E.; González-M, G.; Klotz, B.; Rodrigo, D. Effects of high hydrostatic pressure and temperature increase on Escherichia coli spp. and pectin methyl esterase inactivation in orange juice. Food Sci. Technol. Int. 2016, 22, 173–180. [Google Scholar] [CrossRef]
- Anvarian, A.H.; Smith, M.P.; Overton, T.W. The effects of orange juice clarification on the physiology of Escherichia coli; growth-based and flow cytometric analysis. Int. J. Food Microbiol. 2016, 219, 38–43. [Google Scholar] [CrossRef]
- Dasan, B.G.; Boyaci, I.H. Effect of cold atmospheric plasma on inactivation of Escherichia coli and physicochemical properties of apple, orange, tomato juices, and sour cherry nectar. Food Bioprocess Technol. 2018, 11, 334–343. [Google Scholar] [CrossRef]
- Rifna, E.; Singh, S.K.; Chakraborty, S.; Dwivedi, M. Effect of thermal and non-thermal techniques for microbial safety in food powder: Recent advances. Food Res. Int. 2019, 126, 108654. [Google Scholar] [CrossRef]
- Mohideen, F.W.; Solval, K.M.; Li, J.; Zhang, J.; Chouljenko, A.; Chotiko, A.; Prudente, A.D.; Bankston, J.D.; Sathivel, S. Effect of continuous ultra-sonication on microbial counts and physico-chemical properties of blueberry (Vaccinium corymbosum) juice. LWT 2015, 60, 563–570. [Google Scholar] [CrossRef]
- Tiwari, B.; Muthukumarappan, K.; O’Donnell, C.; Cullen, P. Effects of sonication on the kinetics of orange juice quality parameters. J. Agric. Food Chem. 2008, 56, 2423–2428. [Google Scholar] [CrossRef]
- Ferrario, M.; Alzamora, S.M.; Guerrero, S. Study of the inactivation of spoilage microorganisms in apple juice by pulsed light and ultrasound. Food Microbiol. 2015, 46, 635–642. [Google Scholar] [CrossRef]
- Huang, Y.; Sheng, J.; Yang, F.; Hu, Q. Effect of enzyme inactivation by microwave and oven heating on preservation quality of green tea. J. Food Eng. 2007, 78, 687–692. [Google Scholar] [CrossRef]
- Kernou, O.N.; Belbahi, A.; Amir, A.; Bedjaoui, K.; Kerdouche, K.; Dairi, S.; Aoun, O.; Madani, K. Effect of sonication on microwave inactivation of Escherichia coli in an orange juice beverage. Food Process Eng. 2021, 44, e13664. [Google Scholar] [CrossRef]
- Rostami, S.; Behruzian, M.; Samani, B.H.; Lorigooini, Z.; Hosseinabadi, T.; Zareiforoush, H.; Behruzian, A. Study of combined ultrasound-microwave effect on chemical compositions and E. Coli count of rose aromatic water. Iran. J. Pharm. Res. 2018, 17, 146. [Google Scholar]
- Demirok, N.T.; Yıkmış, S. Combined Effect of Ultrasound and Microwave Power in Tangerine Juice Processing: Bioactive Compounds, Amino Acids, Minerals, and Pathogens. Processes 2022, 10, 2100. [Google Scholar] [CrossRef]
- Das, M.J.; Das, A.J.; Chakraborty, S.; Baishya, P.; Ramteke, A.; Deka, S.C. Effects of microwave combined with ultrasound treatment on the pasteurization and nutritional properties of bottle gourd (Lagenaria siceraria) juice. J. Food Process. Preserv. 2020, 44, e14904. [Google Scholar] [CrossRef]
- Bhat, R.; Kamaruddin, N.S.B.C.; Min-Tze, L.; Karim, A. Sonication improves kasturi lime (Citrus microcarpa) juice quality. Ultrason. Sonochem. 2011, 18, 1295–1300. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef]
- Baş, D.; Boyacı, I.H. Modeling and optimization I: Usability of response surface methodology. J. Food Eng. 2007, 78, 836–845. [Google Scholar] [CrossRef]
- Omar, W.; Nordin, N.; Mohamed, M.; Amin, N. A two-step biodiesel production from waste cooking oil: Optimization of pre-treatment step. Appl. Sci. 2009, 9, 3098–3103. [Google Scholar] [CrossRef]
- Shojaei, S.; Shojaei, S. Experimental design and modeling of removal of Acid Green 25 dye by nanoscale zero-valent iron. EuroMediterr. J. Environ. Integr. 2017, 2, 15. [Google Scholar] [CrossRef]
- Hosseinzadeh Samani, B.; Khoshtaghaza, M.; Minaee, S. Modeling the simultaneous effects of microwave and ultrasound treatments on sour cherry juice using response surface methodology. J. Agric. Sci. Technol. 2018, 17, 837–846. [Google Scholar]
- Banu, A.; Ali, M.Y.; Rahman, M.A.; Konneh, M. Stability of micro dry wire EDM: OFAT and DOE method. Int. J. Adv. Manuf. Technol. 2020, 106, 4247–4261. [Google Scholar] [CrossRef]
- Montgomery, D.C. Design and Analysis of Experiments, 9th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Tajchakavit, S.; Ramaswamy, H.; Fustier, P. Enhanced destruction of spoilage microorganisms in apple juice during continuous flow microwave heating. Food Res. Int. 1998, 31, 713–722. [Google Scholar] [CrossRef]
- Wu, J.; Gamage, T.; Vilkhu, K.; Simons, L.; Mawson, R. Effect of thermosonication on quality improvement of tomato juice. Innov. Food Sci. Emerg. Technol. 2008, 9, 186–195. [Google Scholar] [CrossRef]
- Samani, B.H.; Khoshtaghaza, M.H.; Lorigooini, Z.; Minaei, S.; Zareiforoush, H. Analysis of the combinative effect of ultrasound and microwave power on Saccharomyces cerevisiae in orange juice processing. Innov. Food Sci. Emerg. Technol. 2015, 32, 110–115. [Google Scholar] [CrossRef]
- Raso, J.; Pagan, R.; Condon, S.; Sala, F. Influence of temperature and pressure on the lethality of ultrasound. Appl. Environ. Microbiol. 1998, 64, 465–471. [Google Scholar] [CrossRef]
- Ugarte-Romero, E.; Feng, H.; Martin, S.E. Inactivation of Shigella boydii 18 IDPH and Listeria monocytogenes Scott A with power ultrasound at different acoustic energy densities and temperatures. J. Food Sci. Technol. 2007, 72, M103–M107. [Google Scholar] [CrossRef]
- Trujillo-Mayol, I.; Céspedes-Acuña, C.; Silva, F.L.; Alarcón-Enos, J. Improvement of the polyphenol extraction from avocado peel by assisted ultrasound and microwaves. J. Food Process Eng. 2019, 42, e13197. [Google Scholar] [CrossRef]
- Jafarpour, D.; Hashemi, S.M.B.; Mousavifard, M. Inactivation kinetics of pathogenic bacteria in persimmon using the combination of thermosonication and formic acid. Food Sci. Technol. Int. 2022, 10820132221095718. [Google Scholar] [CrossRef]
- Anaya-Esparza, L.M.; Méndez-Robles, M.D.; Sayago-Ayerdi, S.G.; García-Magaña, M.d.L.; Ramírez-Mares, M.V.; Sánchez-Burgos, J.A.; Montalvo-González, E. Effect of thermosonication on pathogenic bacteria, quality attributes and stability of soursop nectar during cold storage. CyTA-J. Food 2017, 15, 592–600. [Google Scholar] [CrossRef]
- Parreiras, P.M.; Nogueira, J.A.V.; da Cunha, L.R.; Passos, M.C.; Gomes, N.R.; Breguez, G.S.; Falco, T.S.; Bearzoti, E.; Menezes, C.C. Effect of thermosonication on microorganisms, the antioxidant activity and the retinol level of human milk. Food Control 2020, 113, 107172. [Google Scholar] [CrossRef]
- Russell, A. Lethal effects of heat on bacterial physiology and structure. Sci. Prog. 2003, 86, 115–137. [Google Scholar] [CrossRef]
- Cabassi, C.S.; Falanga, G.; Romani, A. Disinfectant and Antimicrobial Compositions, in Particular for the Veterinary Field. U.S. Patent Application 15/318,570, 11 May 2017. [Google Scholar]
- Xie, Y.; Hu, P.; Zhu, N.; Lei, F.; Xing, L.; Xu, L. Collaborative optimization of ground source heat pump-radiant ceiling air conditioning system based on response surface method and NSGA-II. Renew. Energy 2020, 147, 249–264. [Google Scholar] [CrossRef]
- Srivastava, A.; Singh, V.; Haque, S.; Pandey, S.; Mishra, M.; Jawed, A.; Shukla, P.; Singh, P.; Tripathi, C. Response surface methodology-genetic algorithm based medium optimization, purification, and characterization of cholesterol oxidase from Streptomyces rimosus. Sci. Rep. 2018, 8, 10913. [Google Scholar] [CrossRef]

| Independent Variable | Level | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| Microwave power (Watt, A) | 300 | 600 | 900 |
| Microwave treatment time (second, B) | 15 | 25 | 35 |
| Time of ultrasound (minute, C) | 10 | 20 | 30 |
| Factor A | Factor B | Factor C | Response | |
|---|---|---|---|---|
| Run | Microwave Power | Microwave Treatment Time | Time of Ultrasound | E. coli Inactivation |
| W | s | min | Log (N/N0) | |
| 1 | 600 | 35 | 30 | −8 ± 0 |
| 2 | 300 | 35 | 20 | −0.037 ± 0.3 |
| 3 | 300 | 25 | 30 | −0.370 ± 0 |
| 4 | 600 | 25 | 20 | −3.94 ± 0 |
| 5 | 600 | 15 | 10 | 0.11 ± 0 |
| 6 | 300 | 25 | 10 | 0.07 ± 0 |
| 7 | 900 | 35 | 20 | −8 ± 0 |
| 8 | 600 | 35 | 10 | −4.47 ± 0 |
| 9 | 900 | 25 | 30 | −8 ± 0 |
| 10 | 900 | 15 | 20 | −1.8931 ± 0 |
| 11 | 600 | 25 | 20 | −3.49 ± 0 |
| 12 | 600 | 15 | 30 | −3.33 ± 0 |
| 13 | 300 | 15 | 20 | 0.48 ± 0 |
| 14 | 600 | 25 | 20 | −2.83 ± 0 |
| 15 | 900 | 25 | 10 | −3.8 ± 0 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 124.44 | 9 | 13.83 | 29.63 | 0.0008 | Significant |
| A-microwave power | 59.60 | 1 | 59.60 | 127.71 | <0.0001 | |
| B-microwave treatment time | 31.50 | 1 | 31.50 | 67.49 | 0.0004 | |
| C-time of ultrasound | 16.85 | 1 | 16.85 | 36.10 | 0.0018 | |
| AB | 7.81 | 1 | 7.81 | 16.74 | 0.0094 | |
| AC | 3.53 | 1 | 3.53 | 7.57 | 0.0402 | |
| BC | 0.0020 | 1 | 0.0020 | 0.0043 | 0.9500 | |
| A2 | 3.53 | 1 | 3.53 | 7.56 | 0.0403 | |
| B2 | 0.0236 | 1 | 0.0236 | 0.0506 | 0.8309 | |
| C2 | 1.25 | 1 | 1.25 | 2.68 | 0.1623 | |
| Residual | 2.33 | 5 | 0.4667 | |||
| Lack of Fit | 1.71 | 3 | 0.5700 | 1.83 | 0.3726 | Not significant |
| Pure Error | 0.6234 | 2 | 0.3117 | |||
| Cor Total R2 Adjusted R2 AdeqPrecision C.V. % | 126.77 | 14 | 0.9816 0.9485 16.9018 21.57 |
| Solution Number | Microwave | Microwave Treatment Time | Time of Ultrasound | E. coli Inactivation Actual | E. coli Inactivation Predicted | Std. Err | Desirability |
|---|---|---|---|---|---|---|---|
| W | S | min | Log (N/N0) | Log (N/N0) | |||
| 1 | 600 | 35 | 30 | −8.000 | −8.000 | 0.553 | 1 |
| 2 | 900 | 33 | 20 | −8.000 | −8.000 | 0.487 | 1 |
| Study Type | Response Surface | Subtype | Randomized | |||
|---|---|---|---|---|---|---|
| Design type | Box–Behnken | Runs 15 | ||||
| Design mode | Quadratic | No blocks | ||||
| Factor | Name | Units | Type | Minimum | Maximum | Mean |
| A | Microwave | Watt | Numeric | 300 | 900 | 600 |
| B | Microwave treatment time | Second | Numeric | 15 | 35 | 25 |
| C | Time of ultrasound | Minute | Numeric | 10 | 30 | 20 |
| Response | Name | Units | Obs | Analysis | ||
| R | Inactivation | - | 15 | Polynomial | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kernou, O.-N.; Azzouz, Z.; Belbahi, A.; Kerdouche, K.; Kaanin-Boudraa, G.; Amir, A.; Madani, K.; Rijo, P. Inactivation of Escherichia coli in an Orange Juice Beverage by Combined Ultrasonic and Microwave Treatment. Foods 2023, 12, 666. https://doi.org/10.3390/foods12030666
Kernou O-N, Azzouz Z, Belbahi A, Kerdouche K, Kaanin-Boudraa G, Amir A, Madani K, Rijo P. Inactivation of Escherichia coli in an Orange Juice Beverage by Combined Ultrasonic and Microwave Treatment. Foods. 2023; 12(3):666. https://doi.org/10.3390/foods12030666
Chicago/Turabian StyleKernou, Ourdia-Nouara, Zahra Azzouz, Amine Belbahi, Kamelia Kerdouche, Ghania Kaanin-Boudraa, Akila Amir, Khodir Madani, and Patricia Rijo. 2023. "Inactivation of Escherichia coli in an Orange Juice Beverage by Combined Ultrasonic and Microwave Treatment" Foods 12, no. 3: 666. https://doi.org/10.3390/foods12030666
APA StyleKernou, O.-N., Azzouz, Z., Belbahi, A., Kerdouche, K., Kaanin-Boudraa, G., Amir, A., Madani, K., & Rijo, P. (2023). Inactivation of Escherichia coli in an Orange Juice Beverage by Combined Ultrasonic and Microwave Treatment. Foods, 12(3), 666. https://doi.org/10.3390/foods12030666







