Application of White-Wine-Pomace-Derived Ingredients in Extending Storage Stability of Fresh Pork Burgers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material
2.1.1. Valorization Process of the White Wine Pomace
2.1.2. Burgers Manufacture and Experimental Design
2.2. Methods
2.2.1. Polyphenoloxidase (PPO) Enzyme Activity in Pomace
2.2.2. Profile of Individual Phenolic Compounds of VP
2.2.3. Physicochemical Composition of Valorized Pomace and Burgers
2.2.4. Microbiological Analysis of Valorized Pomace and Burgers
2.2.5. Instrumental Color of Burgers
2.2.6. Lipid and Protein Oxidation of Burgers
2.2.7. Total Phenolic Compounds Content in Pomace and Burgers
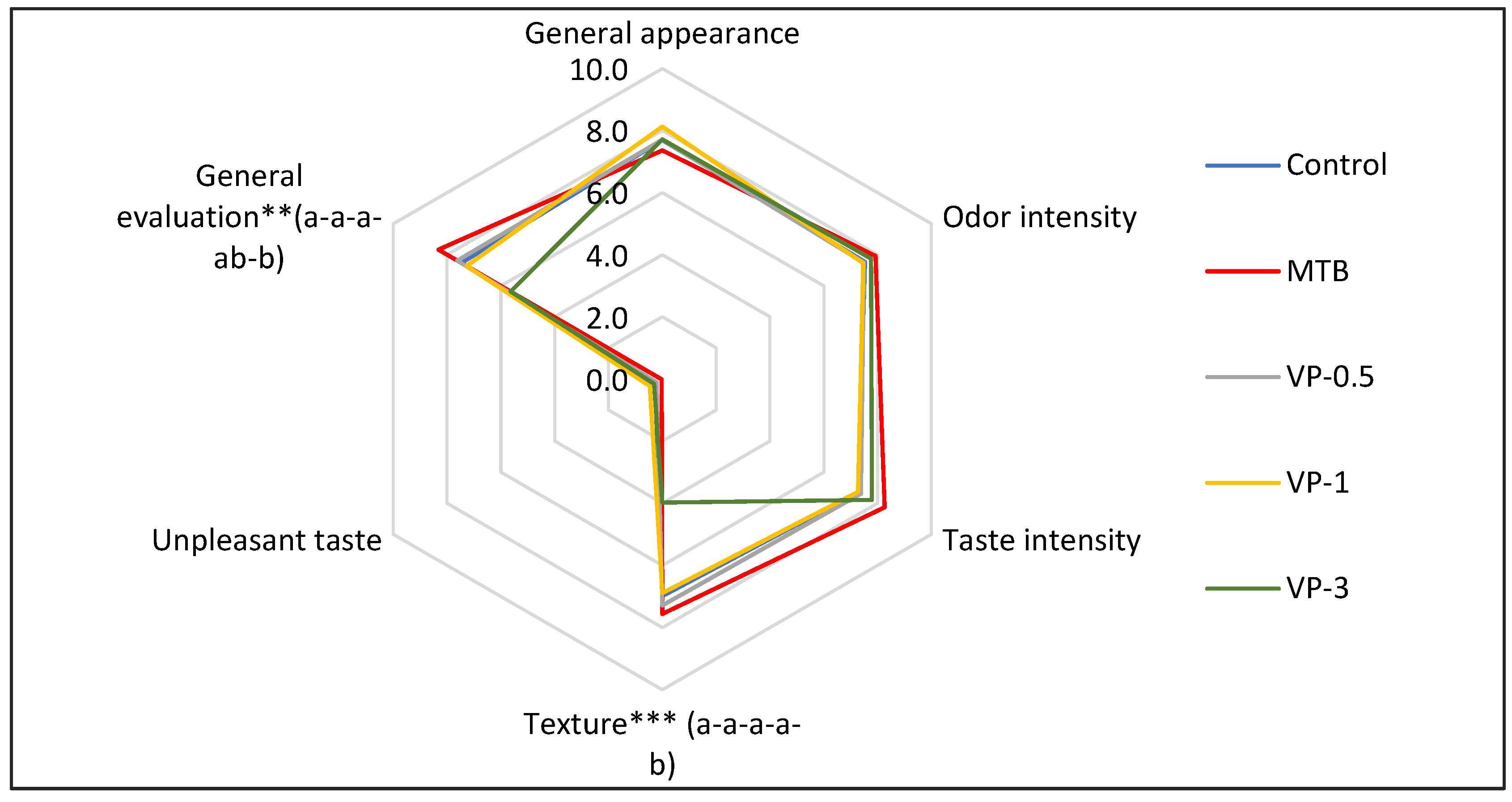
2.2.8. Sensory Analysis of Burgers
2.3. Statistical Analysis
3. Results and Discussion
3.1. Manufacture Process of the Ingredient of White Wine Pomace (VP) and Characterization
3.2. Effect of Valorized Pomace for the Preservation of Pork Burgers
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de Alencar, M.G.; de Quadros, C.P.; Luna, A.L.L.P.; Neto, A.F.; da Costa, M.M.; Queiroz, M.A.Á.; de Carvalho, F.A.L.; da Silva Araújo, D.H.; Gois, G.C.; dos Anjos Santos, V.L.; et al. Grape Skin Flour Obtained from Wine Processing as an Antioxidant in Beef Burgers. Meat Sci. 2022, 194, 108963. [Google Scholar] [CrossRef]
- Serrano, R.; Bañón, S. Reducing SO2 in Fresh Pork Burgers by Adding Chitosan. Meat Sci. 2012, 92, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Bellés, M.; Alonso, V.; Roncalés, P.; Beltrán, J.A. Sulfite-Free Lamb Burger Meat: Antimicrobial and Antioxidant Properties of Green Tea and Carvacrol. J. Sci. Food Agric. 2019, 99, 464–472. [Google Scholar] [CrossRef]
- Fernandes, I.; Rodrigues, J.A.; Almeida, C.M.M. Sulfites in Fresh Meat and Meat Preparations Commercialized in Portugal. Food Anal. Methods 2022, 15, 172–184. [Google Scholar] [CrossRef]
- D’Amore, T.; Di Taranto, A.; Berardi, G.; Vita, V.; Marchesani, G.; Chiaravalle, A.E.; Iammarino, M. Sulfites in Meat: Occurrence, Activity, Toxicity, Regulation, and Detection. A Comprehensive Review. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2701–2720. [Google Scholar] [CrossRef]
- Awad, A.M.; Kumar, P.; Ismail-Fitry, M.R.; Jusoh, S.; Ab Aziz, M.F.; Sazili, A.Q. Overview of Plant Extracts as Natural Preservatives in Meat. J. Food Process. Preserv. 2022, 46, e16796. [Google Scholar] [CrossRef]
- Beres, C.; Costa, G.N.S.; Cabezudo, I.; da Silva-James, N.K.; Teles, A.S.C.; Cruz, A.P.G.; Mellinger-Silva, C.; Tonon, R.V.; Cabral, L.M.C.; Freitas, S.P. Towards Integral Utilization of Grape Pomace from Winemaking Process: A Review. Waste Manag. 2017, 68, 581–594. [Google Scholar] [CrossRef] [PubMed]
- García-Lomillo, J.; González-SanJosé, M.L. Applications of Wine Pomace in the Food Industry: Approaches and Functions. Compr. Rev. Food Sci. Food Saf. 2017, 16, 3–22. [Google Scholar] [CrossRef]
- Argade, A.; Bishnoi, S.; Ahlawat, S.S. Utilization of Wine Industry Waste (Grape Pomace) in processed Meat Products: A Review. Pharma Innov. J. 2017, 6, 297–301. [Google Scholar]
- Nardoia, M.; Ruiz-Capillas, C.; Casamassima, D.; Herrero, A.M.; Pintado, T.; Jiménez-Colmenero, F.; Chamorro, S.; Brenes, A. Effect of Polyphenols Dietary Grape By-Products on Chicken Patties. Eur. Food Res. Technol. 2018, 244, 367–377. [Google Scholar] [CrossRef]
- Andrés, A.I.; Petrón, M.J.; Adámez, J.D.; López, M.; Timón, M.L. Food By-Products as Potential Antioxidant and Antimicrobial Additives in Chill Stored Raw Lamb Patties. Meat Sci. 2017, 129, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Skibsted, L.H.; Mikkelsen, A.; Bertelsen, G. Lipid-Derived Off-Flavours in Meat. In Flavour of Meat, Meat Products and Seafoods; F. Shahidi.: London, UK, 1998. [Google Scholar]
- D’Arrigo, M.; Delgado-Adámez, J.; Rocha-Pimienta, J.; Valdés-Sánchez, M.E.; Ramírez-Bernabé, M.R. Integral Use of Red Wine Pomace after Hydrostatic High Pressure: Application of Two Consecutive Cycles of Treatment. Food Chem. X 2023. submitted. [Google Scholar]
- Ramírez, R.; Delgado, J.; Rocha-Pimienta, J.; Valdés, M.E.; Martín-Mateos, M.J.; Ayuso-Yuste, M.C. Preservation of White Wine Pomace by Hydrostatic High Pressure. Heliyon 2023, 9, e21199. [Google Scholar] [CrossRef] [PubMed]
- González-Cebrino, F.; Durán, R.; Delgado-Adámez, J.; Contador, R.; Ramírez, R. Changes after High-Pressure Processing on Physicochemical Parameters, Bioactive Compounds, and Polyphenol Oxidase Activity of Red Flesh and Peel Plum Purée. Innov. Food Sci. Emerg. Technol. 2013, 20, 34–41. [Google Scholar] [CrossRef]
- Portu, J.; López, R.; Baroja, E.; Santamaría, P.; Garde-Cerdán, T. Improvement of Grape and Wine Phenolic Content by Foliar Application to Grapevine of Three Different Elicitors: Methyl Jasmonate, Chitosan, and Yeast Extract. Food Chem. 2016, 201, 213–221. [Google Scholar] [CrossRef]
- Díaz-Fernández, Á.; Díaz-Losada, E.; Moreno, D.; Esperanza Valdés Sánchez, M. Anthocyanin Profile of Galician Endangered Varieties. A Tool for Varietal Selection. Food Res. Int. 2022, 154. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Muñoz, N.; Fernández-González, M.; Gómez-Alonso, S.; García-Romero, E.; Hermosín-Gutiérrez, I. Red-Color Related Phenolic Composition of Garnacha Tintorera (Vitis vinifera L.) Grapes and Red Wines. J. Agric. Food Chem. 2009, 57, 7883–7891. [Google Scholar] [CrossRef]
- AOAC International; Latimer, G.W. Official Methods of Analysis of AOAC International, 20th ed.; AOAC International: Rockville, MD, USA, 2016. [Google Scholar]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A Simple Method for the Isolation and Purification of Total Lipides from Animal Tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, M.J.; Barragán, R. Determinación Cuantitativa de la Fracción Hidrocarbonada en Alimentos. Anal. Bromatol. XXXVII 1985, 37, 61–77. [Google Scholar]
- UNE EN ISO 6579-1; Microbiología de La Cadena Alimentaria. Método Horizontal Para la Detección, Enumeración y Serotipado de Salmonella. Parte 1: Detección de Salmonella Spp. (ISO 6579-1:2017). ISO: Geneva, Switzerland, 2017.
- UNE-EN ISO 11290-1; Microbiología de La Cadena Alimentaria. Método Horizontal Para la Detección y el Recuento de Listeria Monocytogenes y de Listeria Spp. Parte 1: Método de Detección (ISO 11290-1:2017). ISO: Geneva, Switzerland, 2018.
- Sørensen, G.; Storgaard Jørgensen, S. A Critical Examination of Some Experimental Variables in the 2-Thiobarbituric Acid (TBA) Test for Lipid Oxidation in Meat Products. Eur. Food Res. Technol. 1996, 202, 205–210. [Google Scholar] [CrossRef]
- Oliver, C.N.; Ahn, B.W.; Moerman, E.J.; Goldstein, S.; Stadtman, E.R. Age-Related Changes in Oxidized Proteins. J. Biol. Chem. 1987, 262, 5488–5491. [Google Scholar] [CrossRef] [PubMed]
- Lima, V.L.A.G.; Mélo, E.A.; Maciel, M.I.S.; Prazeres, F.G.; Musser, R.S.; Lima, D.E.S. Total Phenolic and Carotenoid Contents in Acerola Genotypes Harvested at Three Ripening Stages. Food Chem. 2005, 4, 565–568. [Google Scholar] [CrossRef]
- Xiao, H.W.; Pan, Z.; Deng, L.Z.; El-Mashad, H.M.; Yang, X.H.; Mujumdar, A.S.; Gao, Z.J.; Zhang, Q. Recent Developments and Trends in Thermal Blanching—A Comprehensive Review. Inf. Process. Agric. 2017, 4, 101–127. [Google Scholar] [CrossRef]
- Lambri, M.; Torchio, F.; Colangelo, D.; Río Segade, S.; Giacosa, S.; De Faveri, D.M.; Gerbi, V.; Rolle, L. Influence of Different Berry Thermal Treatment Conditions, Grape Anthocyanin Profile, and Skin Hardness on the Extraction of Anthocyanin Compounds in the Colored Grape Juice Production. Food Res. Int. 2015, 77, 584–590. [Google Scholar] [CrossRef]
- Río Segade, S.; Torchio, F.; Giacosa, S.; Ricauda Aimonino, D.; Gay, P.; Lambri, M.; Dordoni, R.; Gerbi, V.; Rolle, L. Impact of Several Pre-Treatments on the Extraction of Phenolic Compounds in Winegrape Varieties with Different Anthocyanin Profiles and Skin Mechanical Properties. J. Agric. Food Chem. 2014, 62, 8437–8451. [Google Scholar] [CrossRef] [PubMed]
- Urquiaga, I.; Troncoso, D.; Mackenna, M.J.; Urzúa, C.; Pérez, D.; Dicenta, S.; de la Cerda, P.M.; Amigo, L.; Carreño, J.C.; Echeverría, G.; et al. The Consumption of Beef Burgers Prepared with Wine Grape Pomace Flour Improves Fasting Glucose, Plasma Antioxidant Levels, and Oxidative Damage Markers in Humans: A Controlled Trial. Nutrients 2018, 10, 1388. [Google Scholar] [CrossRef] [PubMed]
- Selani, M.M.; Contreras-Castillo, C.J.; Shirahigue, L.D.; Gallo, C.R.; Plata-Oviedo, M.; Montes-Villanueva, N.D. Wine Industry Residues Extracts as Natural Antioxidants in Raw and Cooked Chicken Meat during Frozen Storage. Meat Sci. 2011, 88, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Abdelhakam, O.S.; Elsebaie, E.M.; Ghazi, A.K.; Gouda, M.S. Quality Characteristics of Beef Hamburger Enriched with Red Grape Pomace Powder during Freezing Storage. Slov. Vet. Res. 2019, 56, 333–340. [Google Scholar] [CrossRef]
- Antonić, B.; Jančíková, S.; Dordević, D.; Tremlová, B. Grape Pomace Valorization: A Systematic Review and Meta-Analysis. Foods 2020, 9, 1627. [Google Scholar] [CrossRef]
- Javier, H.; Siles, J.Á.; Aida, G.; del Carmen, G.M.; de los Ángeles, M.M. Revalorization of Grape Marc Waste from Liqueur Wine: Biomethanization. J. Chem. Technol. Biotechnol. 2019, 94, 1499–1508. [Google Scholar] [CrossRef]
- Pereira, A.; Lee, H.C.; Lammert, R.; Wolberg, C.; Ma, D.; Immoos, C.; Casassa, F.; Kang, I. Effects of Red-Wine Grape Pomace on the Quality and Sensory Attributes of Beef Hamburger Patty. Int. J. Food Sci. Technol. 2022, 57, 1814–1823. [Google Scholar] [CrossRef]
- Nogales-Bueno, J.; Baca-Bocanegra, B.; Jara-Palacios, M.J.; Hernández-Hierro, J.M.; Heredia, F.J. Evaluation of the Influence of White Grape Seed Extracts as Copigment Sources on the Anthocyanin Extraction from Grape Skins Previously Classified by near Infrared Hyperspectral Tools. Food Chem. 2017, 221, 1685–1690. [Google Scholar] [CrossRef] [PubMed]
- Kammerer, D.; Claus, A.; Carle, R.; Schieber, A. Polyphenol Screening of Pomace from Red and White Grape Varieties (Vitis vinifera L.) by HPLC-DAD-MS/MS. J. Agric. Food Chem. 2004, 52, 4360–4367. [Google Scholar] [CrossRef] [PubMed]
- Jara-Palacios, M.J.; Hernanz, D.; Cifuentes-Gomez, T.; Escudero-Gilete, M.L.; Heredia, F.J.; Spencer, J.P.E. Assessment of White Grape Pomace from Winemaking as Source of Bioactive Compounds, and Its Antiproliferative Activity. Food Chem. 2015, 183, 78–82. [Google Scholar] [CrossRef]
- Mildner-Szkudlarz, S.; Bajerska, J.; Zawirska-Wojtasiak, R.; Górecka, D. White Grape Pomace as a Source of Dietary Fibre and Polyphenols and Its Effect on Physical and Nutraceutical Characteristics of Wheat Biscuits. J. Sci. Food Agric. 2013, 93, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Brazinha, C.; Cadima, M.; Crespo, J.G. Optimization of Extraction of Bioactive Compounds from Different Types of Grape Pomace Produced at Wineries and Distilleries. J. Food Sci. 2014, 79, E1142–E1149. [Google Scholar] [CrossRef] [PubMed]
- Lucas-González, R.; Roldán-Verdu, A.; Sayas-Barberá, E.; Fernández-López, J.; Pérez-Álvarez, J.A.; Viuda-Martos, M. Assessment of Emulsion Gels Formulated with Chestnut (Castanea sativa M.) Flour and Chia (Salvia hispanica L.) Oil as Partial Fat Replacers in Pork Burger Formulation. J. Sci. Food Agric. 2020, 100, 1265–1273. [Google Scholar] [CrossRef]
- Diario Oficial de la Unión Europea, num. 404, Reglamento CE No 1924 2006 Del Parlamento Europeo y Del Consejo de 20 de Diciembre 2006, Relativo a Las Declaraciones Nutricionales y de Propiedades Saludables En Los Alim. 2006, 9-25. Available online: https://www.boe.es/doue/2006/404/L00009-00025.pdf (accessed on 3 October 2023).
- García-Lomillo, J.; González-SanJosé, M.a.L.; Del Pino-García, R.; Rivero-Pérez, M.a.D.; Muñiz-Rodríguez, P. Alternative Natural Seasoning to Improve the Microbial Stability of Low-Salt Beef Patties. Food Chem. 2017, 227, 122–128. [Google Scholar] [CrossRef]
- Hayes, J.E.; Stepanyan, V.; Allen, P.; O’Grady, M.N.; Kerry, J.P. Effect of Lutein, Sesamol, Ellagic Acid and Olive Leaf Extract on the Quality and Shelf-Life Stability of Packaged Raw Minced Beef Patties. Meat Sci. 2010, 84, 613–620. [Google Scholar] [CrossRef]
- Kalogianni, A.I.; Lazou, T.; Bossis, I.; Gelasakis, A.I. Natural Phenolic Compounds for the Control of Oxidation, Bacterial Spoilage, and Foodborne Pathogens in Meat. Foods 2020, 9, 794. [Google Scholar] [CrossRef]
- Commission Regulation (EC) No 2073/2005 of 15 November 2005 on Microbiological Criteria for Foodstuffs (Text with EEA Relevance). 2005. [WWW Document]. Available online: https://eur-lex.europa.eu/eli/reg/2005/2073/oj (accessed on 3 October 2023).
- Martínez, L.; Djenane, D.; Cilla, I.; Beltrán, J.A.; Roncalés, P. Effect of Varying Oxygen Concentrations on the Shelf-Life of Fresh Pork Sausages Packaged in Modified Atmosphere. Food Chem. 2006, 94, 219–225. [Google Scholar] [CrossRef]
- Adams, J.B. Food Additive-Additive Interactions Involving Sulphur Dioxide and Ascorbic and Nitrous Acids: A Review. Food Chem. 1997, 59, 401–409. [Google Scholar] [CrossRef]
- Garrido, M.D.; Auqui, M.; Martí, N.; Linares, M.B. Effect of Two Different Red Grape Pomace Extracts Obtained under Different Extraction Systems on Meat Quality of Pork Burgers. LWT 2011, 44, 2238–2243. [Google Scholar] [CrossRef]
- Kumar, Y.; Yadav, D.N.; Ahmad, T.; Narsaiah, K. Recent Trends in the Use of Natural Antioxidants for Meat and Meat Products. Compr. Rev. Food Sci. Food Saf. 2015, 14, 796–812. [Google Scholar] [CrossRef]
- Sagdic, O.; Ozturk, I.; Yilmaz, M.T.; Yetim, H. Effect of Grape Pomace Extracts Obtained from Different Grape Varieties on Microbial Quality of Beef Patty. J. Food Sci. 2011, 76, M515–M521. [Google Scholar] [CrossRef]
- Reddy, G.V.B.; Sen, A.R.; Nair, P.N.; Reddy, K.S.; Reddy, K.K.; Kondaiah, N. Effects of Grape Seed Extract on the Oxidative and Microbial Stability of Restructured Mutton Slices. Meat Sci. 2013, 95, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Han, M.; Bu, Y.; Li, X.; Yi, S.; Xu, Y.; Li, J. Plant Polyphenols Regulating Myoglobin Oxidation and Color Stability in Red Meat and Certain Fish: A Review. Crit. Rev. Food Sci. Nutr. 2022, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Sabow, A.B.; Sazili, A.Q.; Aghwan, Z.A.; Zulkifli, I.; Goh, Y.M.; Ab Kadir, M.Z.A.; Nakyinsige, K.; Kaka, U.; Adeyemi, K.D. Changes of Microbial Spoilage, Lipid-Protein Oxidation and Physicochemical Properties during Post Mortem Refrigerated Storage of Goat Meat. Anim. Sci. J. 2016, 87, 816–826. [Google Scholar] [CrossRef]
- Li, S.; Zamaratskaia, G.; Roos, S.; Båth, K.; Meijer, J.; Borch, E.; Johansson, M. Inter-Relationships between the Metrics of Instrumental Meat Color and Microbial Growth during Aerobic Storage of Beef at 4 °C. Acta Agric. Scand. A Anim. Sci. 2015, 65, 97–106. [Google Scholar] [CrossRef]
- Mancini, R.A.; Hunt, M.C. Current Research in Meat Color. Meat Sci. 2005, 71, 100–121. [Google Scholar] [CrossRef]
- de Paula Paseto Fernandes, R.; Trindade, M.A.; de Melo, M.P. Natural Antioxidants and Food Applications: Healthy Perspectives. Altern. Replace. Foods 2018, 17, 31–64. [Google Scholar] [CrossRef]
- Estévez, M. Protein Carbonyls in Meat Systems: A Review. Meat Sci. 2011, 89, 259–279. [Google Scholar] [CrossRef]
- Jongberg, S.; Skov, S.H.; Tørngren, M.A.; Skibsted, L.H.; Lund, M.N. Effect of White Grape Extract and Modified Atmosphere Packaging on Lipid and Protein Oxidation in Chill Stored Beef Patties. Food Chem. 2011, 128, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, I.A.M.; Babiker, E.E.; Al-Juhaimi, F.Y.; Bekhit, A.E.D.A. Clove Polyphenolic Compounds Improve the Microbiological Status, Lipid Stability, and Sensory Attributes of Beef Burgers during Cold Storage. Antioxidants 2022, 11, 1354. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. In Methods in Enzymology; Academic Press: Cambridge, UK, 1999; Volume 299. [Google Scholar]
- De Gil, D.M.A.; Rebelo, M.J.F. Metabisulfite Interference in Biosensing and Folin-Ciocalteu Analysis of Polyphenols. Microchim. Acta 2009, 167, 253–258. [Google Scholar] [CrossRef]
- Goñi, I.; Brenes, A.; Centeno, C.; Viveros, A.; Saura-Calixto, F.; Rebolé, A.; Arija, I.; Estevez, R. Effect of Dietary Grape Pomace and Vitamin E on Growth Performance, Nutrient Digestibility, and Susceptibility to Meat Lipid Oxidation in Chickens. Poult. Sci. 2007, 86, 508–516. [Google Scholar] [CrossRef]
- Awad, E.; Mnayer, D.; Joubrane, K. The Effect of Citric Acid on the Physicochemical and Microbiological Parameters of Processed Burger Meat. J. Food Process. Technol. 2020, 11, 35248. [Google Scholar] [CrossRef]
- Kryževičūtė, N.; Jaime, I.; Diez, A.M.; Rovira, J.; Venskutonis, P.R. Effect of Raspberry Pomace Extracts Isolated by High Pressure Extraction on the Quality and Shelf-Life of Beef Burgers. Int. J. Food Sci. Technol. 2017, 52, 1852–1861. [Google Scholar] [CrossRef]
| Initial-WWP | TB | TB+HHP | p-Value Initial-TB | p-Value TB-HHP | p-Value Initial-HHP | |
|---|---|---|---|---|---|---|
| PPO (%) | 100.0a ± 1.6 | 0.9b ± 0.0 | 1.3b ± 0.0 | *** | ns | *** |
| Phenolic compounds | 457.0b ± 24.6 | 758.2a ± 30.2 | 766.7a ± 15.6 | ** | ns | *** |
| Mesophilic counts | 5.8a ± 0.1 | 4.5ab ± 1.2 | 3.3b ± 0.2 | ns | ns | ** |
| Molds and Yeasts | 4.6a ± 0.1 | 2.7b ± 0.3 | 1.4c ± 0.5 | *** | ** | *** |
| Enterobacteria | 2.2 ± 1.1 | <1 | <1 | ns | ns | ns |
| Valorized Pomace | |
|---|---|
| pH | 3.96 ± 0.03 |
| Aw | 0.971 ± 0.013 |
| Proximate composition (g 100 g−1) | |
| Moisture | 69.2 ± 1.5 |
| Fiber | 18.0 ± 0.9 |
| Protein | 2.3 ± 0.2 |
| Fat | 1.7 ± 0.1 |
| Fatty acids profile (%) | |
| C12:0 | 0.0 ± 0.0 |
| C14:0 | 0.1 ± 0.0 |
| C16:0 | 9.6 ± 0.1 |
| C16:1 | 0.2 ± 0.0 |
| C17:1 | 0.1 ± 0.0 |
| C17:1 | 0.0 ± 0.0 |
| C18:0 | 4.8 ± 0.1 |
| C18:1 | 19.7 ± 0.1 |
| C18:2 | 63.2 ± 0.3 |
| C18:3 | 1.8 ± 0.0 |
| C20:0 | 0.3 ± 0.0 |
| C20:1 | 0.2 ± 0.0 |
| Individual phenolic compounds content (%) | |
| Flavanols | |
| PB1+PB3 | 31.62 ± 1.10 |
| PB2 | 5.59 ± 0.14 |
| Total procyanidins | 37.21 ± 1.24 |
| (+)-Catechin | 15.70 ± 1.24 |
| (-)-Epicatechin | 8.27 ± 0.69 |
| Epigallocatechin | 1.31 ± 0.20 |
| Total Catechin | 25.27 ± 2.13 |
| Total Flavanols | 62.48 ± 3.37 |
| Flavonols | |
| Myricetin-3-galactoside | 0.05 ± 0.01 |
| Myricetin-3-glucoside | 0.10 ± 0.01 |
| Quercetin-3-glucuronide | 18.60 ± 0.60 |
| Quercetin-3-glucoside | 9.93 ± 0.25 |
| Kaempferol- 3-glucoside+3-rutinoside | 4.12 ± 0.11 |
| Isorhamnetin-3- glucoside+-3-rutinoside | 0.25 ± 0.02 |
| Total Flavonols | 33.05 ± 0.99 |
| Acids and derivatives | |
| Gallic | 0.79 ± 0.05 |
| Syringic | 1.87 ± 0.58 |
| Coutaric | 0.70 ± 0.03 |
| Caffeic | 0.07 ± 0.00 |
| Ferouyl-tartaric | 0.83 ± 0.02 |
| Coumaric | 0.08 ± 0.00 |
| Ferulic | 0.11 ± 0.70 |
| Total Acids and derivatives | 4.46 ± 0.70 |
| Stilbenes | |
| trans -Resveratrol | 0.01 ± 0.00 |
| Storage (Days) | Control | MTB | VP-0.5 | VP-1 | VP-3 | p-Value | |
|---|---|---|---|---|---|---|---|
| pH | 5.86a ± 0.01 | 5.84b ± 0.00 | 5.84b ± 0.01 | 5.83b ± 0.01 | 5.80c ± 0.02 | *** | |
| Mesophilic counts | 1 | 6.5 ± 0.0 | 6.3 ± 0.1 | 6.2 ± 0.4 | 6.4 ± 0.2 | 6.6 ± 0.1 | ns |
| 7 | 8.1 ± 0.5 | 7.8 ± 1.7 | 8.1 ± 0.3 | 7.8 ± 0.2 | 8.2 ± 0.4 | ns | |
| P-storage | *** | ns | *** | *** | *** | ||
| Psychrophilic counts | 1 | 6.6a ± 0.1 | 6.0b ± 0.3 | 6.7a ± 0.5 | 6.5ab ± 0.4 | 6.6a ± 0.1 | * |
| 7 | 8.0 ± 0.6 | 8.0 ± 0.5 | 7.9 ± 0.4 | 7.7 ± 0.5 | 8.5 ± 0.5 | ns | |
| P-storage | ** | *** | ** | ** | *** | ||
| Molds and Yeasts | 1 | 4.5 ± 0.2 | 4.4 ± 0.3 | 4.2 ± 0.3 | 4.4 ± 0.2 | 4.5 ± 0.2 | ns |
| 7 | 4.8b ± 0.2 | 5.4a ± 0.3 | 4.4b ± 0.4 | 4.6b ± 0.2 | 4.5b ± 0.3 | *** | |
| P-storage | ns | *** | ns | ns | ns | ||
| S. aureus | 1 | 2.1 ± 0.2 | 2.3 ± 0.3 | 2.0 ± 0.1 | 2.1 ± 0.3 | 2.2 ± 0.3 | ns |
| 7 | 2.1b ± 0.2 | 2.6a ± 0.1 | 2.3b ± 0.3 | 2.1b ± 0.3 | 2.0b ± 0.2 | ** | |
| P-storage | ns | * | * | ns | ns | ||
| Sulfite-reducing bacteria | 1 | <1 | <1 | <1 | <1 | <1 | ns |
| 7 | <1 | <1 | <1 | <1 | <1 | ns | |
| P-storage | ns | ns | ns | ns | ns | ||
| Total coliforms | 1 | 3.7 ± 0.1 | 3.9 ± 0.3 | 3.9 ± 0.1 | 3.7 ± 0.2 | 3.9 ± 0.1 | ns |
| 7 | 5.1a ± 0.1 | 4.3b ± 0.3 | 4.7b ± 0.2 | 4.6b ± 0.3 | 4.7b ± 0.3 | ** | |
| P-storage | *** | ns | *** | ** | *** | ||
| E. coli | 1 | <1 | <1 | <1 | <1 | <1 | ns |
| 7 | <1 | <1 | <1 | <1 | <1 | ns | |
| P-storage | ns | ns | ns | ns | ns |
| Storage (Days) | Control | MTB | VP-0.5 | VP-1 | VP-3 | p-Value | |
|---|---|---|---|---|---|---|---|
| CIE L* | 1 | 54.4 ± 1.7 | 54.3 ± 1.1 | 53.8 ± 1.7 | 53.0 ± 1.4 | 52.3 ± 0.9 | ns |
| 7 | 54.6 ± 1.3 | 54.7 ± 1.9 | 54.4 ± 1.1 | 54.3 ± 0.5 | 55.7 ± 1.0 | ns | |
| P-storage | ns | ns | ns | ns | *** | ||
| CIE a* | 1 | 11.1 ± 0.7 | 11.9 ± 1.8 | 10.9 ± 0.9 | 10.9 ± 0.8 | 10.0 ± 0.4 | ns |
| 7 | 4.1c ± 1.6 | 10.1a ± 1.9 | 6.6bc ± 1.2 | 7.2b ± 1.4 | 5.8bc ± 0.8 | *** | |
| P-storage | *** | ns | *** | ** | *** | ||
| CIE b* | 1 | 13.1 ± 0.7 | 12.9 ± 1.4 | 12.7 ± 0.4 | 13.5 ± 0.4 | 14.0 ± 0.6 | ns |
| 7 | 10.4b ± 0.8 | 12.9a ± 1.6 | 11.4ab ± 0.4 | 11.9ab ± 0.9 | 12.6a ± 0.4 | ** | |
| P-storage | *** | ns | ** | ** | ** | ||
| Chroma | 1 | 17.2 ± 0.9 | 17.5 ± 2.1 | 16.8 ± 0.7 | 17.4 ± 0.7 | 17.2 ± 0.6 | ns |
| 7 | 11.3c ± 0.5 | 16.4a ± 2.4 | 13.3b ± 0.8 | 14.0b ± 1.0 | 13.9b ± 0.7 | *** | |
| P-storage | *** | ns | *** | *** | *** | ||
| Hue | 1 | 49.7b ± 1.7 | 47.6b ± 2.4 | 49.4b ± 2.0 | 51.0b ± 1.9 | 54.3a ± 1.5 | *** |
| 7 | 68.8a ± 9.0 | 52.2c ± 1.9 | 60.9abc ± 3.8 | 58.9bc ± 5.5 | 65.5ab ± 2.3 | ** | |
| P-storage | ** | ** | *** | * | *** |
| Storage (Days) | Control | MTB | VP-0.5 | VP-1 | VP-3 | p-Value | |
|---|---|---|---|---|---|---|---|
| Lipid oxidation | 1 | 0.282a ± 0.008 | 0.265a ± 0.072 | 0.124b ± 0.009 | 0.100b ± 0.022 | 0.105b ± 0.006 | *** |
| 7 | 0.193b ± 0.029 | 0.261a ± 0.011 | 0.108c ± 0.009 | 0.097c ± 0.003 | 0.110c ± 0.004 | *** | |
| P-storage | *** | ns | * | ns | ns | ||
| Protein oxidation | 1 | 2.231a ± 0.447 | 1.909ab ± 0.189 | 1.575b ± 0.465 | 2.033ab ± 0.171 | 1.832ab ± 0.141 | * |
| 7 | 1.896 ± 0.230 | 1.603 ± 0.290 | 1.429 ± 0.299 | 1.347 ± 0.305 | 1.371 ± 0.754 | ns | |
| P-storage | ns | ns | ns | ** | ns | ||
| Phenolic compounds | 1 | 59.86c ± 3.06 | 72.71a ± 3.11 | 65.01bc ± 3.88 | 64.89bc ± 3.11 | 67.67b ± 2.00 | *** |
| 7 | 64.48 ± 2.39 | 77.23 ± 18.02 | 63.53 ± 3.39 | 59.94 ± 7.07 | 64.90 ± 10.57 | ns | |
| P-storage | * | ns | ns | ns | ns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín-Mateos, M.J.; Delgado-Adámez, J.; Moreno-Cardona, D.; Valdés-Sánchez, M.E.; Ramírez-Bernabé, M.R. Application of White-Wine-Pomace-Derived Ingredients in Extending Storage Stability of Fresh Pork Burgers. Foods 2023, 12, 4468. https://doi.org/10.3390/foods12244468
Martín-Mateos MJ, Delgado-Adámez J, Moreno-Cardona D, Valdés-Sánchez ME, Ramírez-Bernabé MR. Application of White-Wine-Pomace-Derived Ingredients in Extending Storage Stability of Fresh Pork Burgers. Foods. 2023; 12(24):4468. https://doi.org/10.3390/foods12244468
Chicago/Turabian StyleMartín-Mateos, María Jesús, Jonathan Delgado-Adámez, Daniel Moreno-Cardona, M. Esperanza Valdés-Sánchez, and M. Rosario Ramírez-Bernabé. 2023. "Application of White-Wine-Pomace-Derived Ingredients in Extending Storage Stability of Fresh Pork Burgers" Foods 12, no. 24: 4468. https://doi.org/10.3390/foods12244468
APA StyleMartín-Mateos, M. J., Delgado-Adámez, J., Moreno-Cardona, D., Valdés-Sánchez, M. E., & Ramírez-Bernabé, M. R. (2023). Application of White-Wine-Pomace-Derived Ingredients in Extending Storage Stability of Fresh Pork Burgers. Foods, 12(24), 4468. https://doi.org/10.3390/foods12244468








