Essential Oils of Mentha arvensis and Cinnamomum cassia Exhibit Distinct Antibacterial Activity at Different Temperatures In Vitro and on Chicken Skin
Abstract
1. Introduction
2. Materials and Methods
2.1. Essential Oils
2.2. EO Chemical Composition Analysis Using GC-MS
2.3. Bacterial Cultures and Media
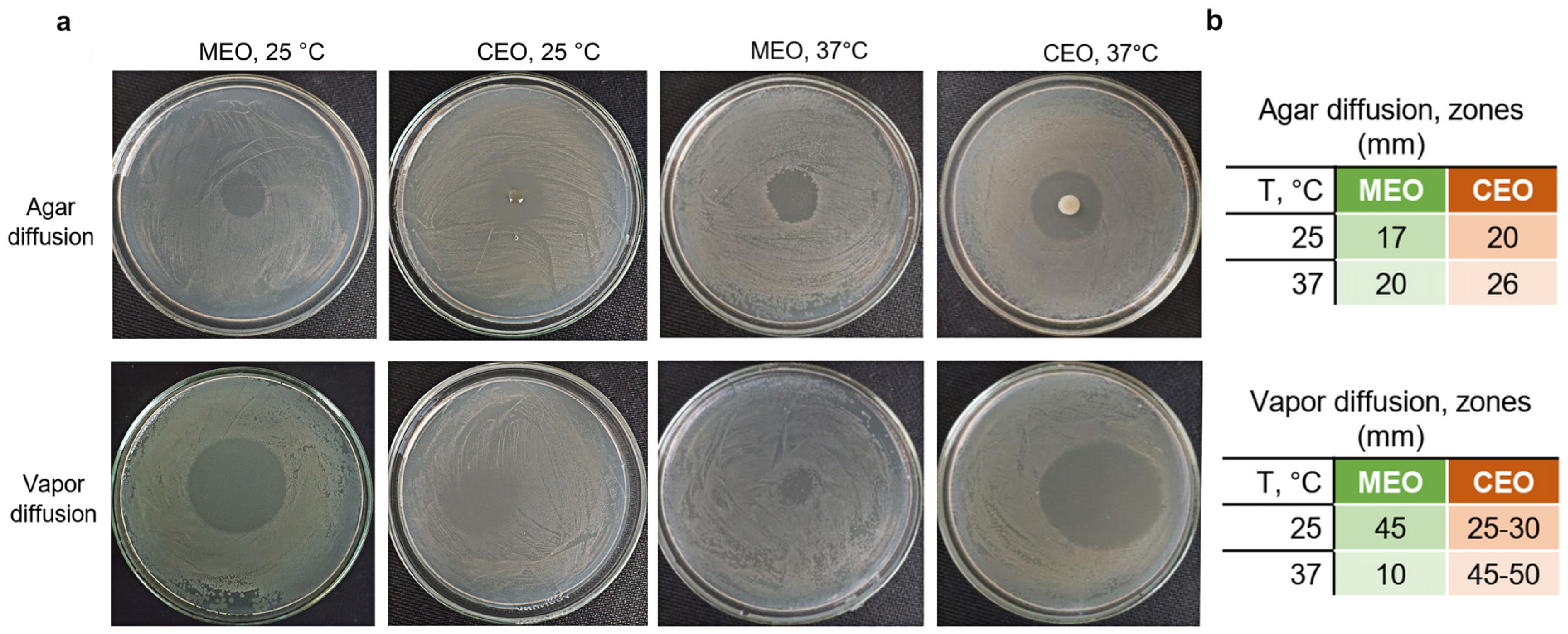
2.4. Antibacterial Activity Testing in Agar Plates
2.5. Determination of MIC and MBC
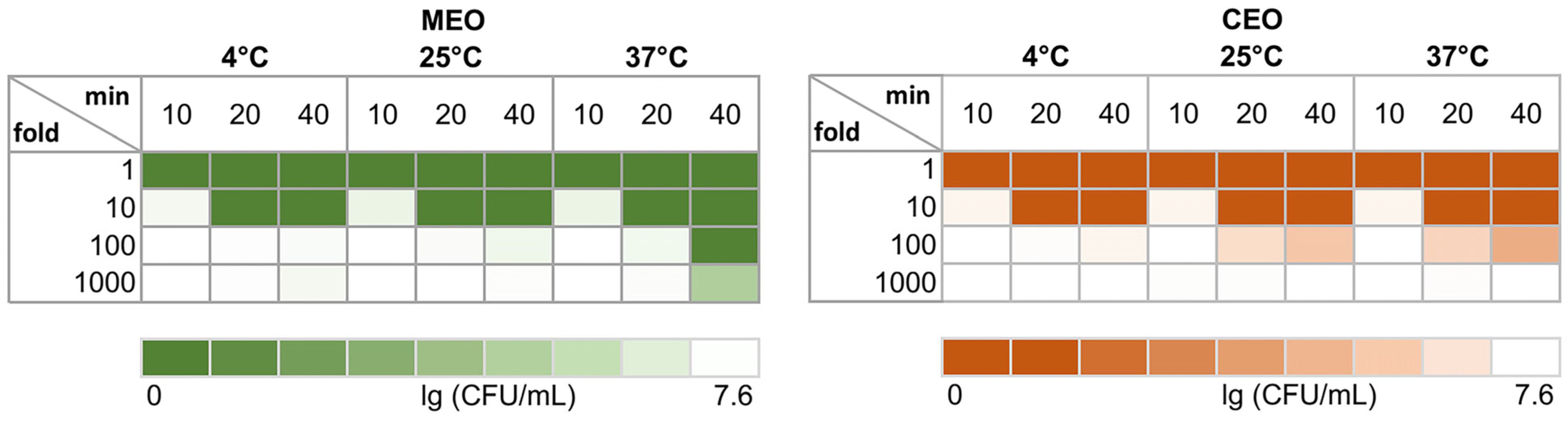
2.6. S. typhimurium Killing upon Direct Contact with EO In Vitro
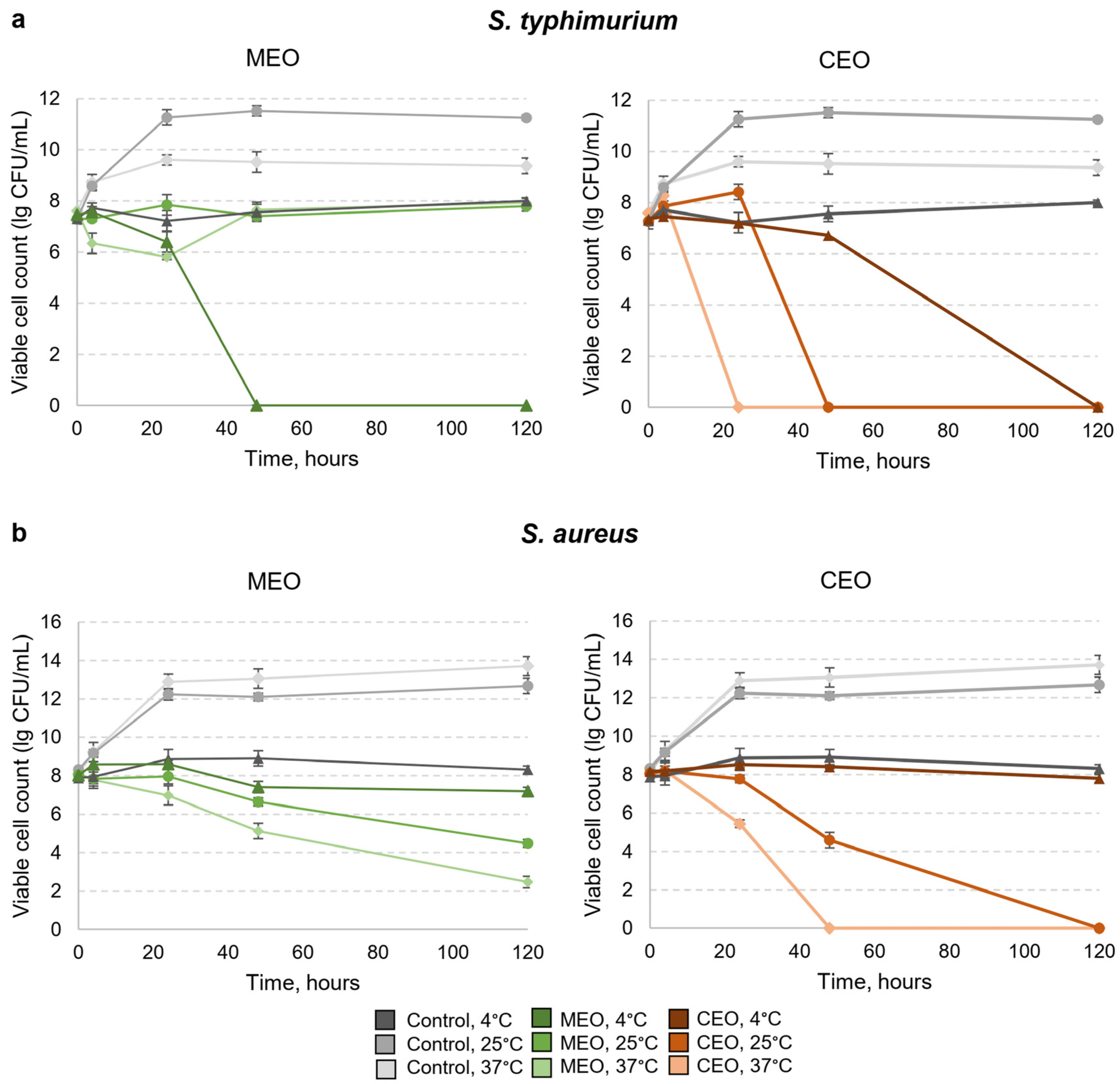
2.7. Antibacterial Activity of EO Vapor In Vitro
2.8. Antibacterial Activity of EO Vapor on Chicken Skin
2.9. Statistical Analysis
3. Results and Discussion
3.1. Chemical Compositions of C. cassia and M. arvensis EOs
3.2. CEO and MEO Antibacterial Activity against S. typhimurium in Agar Plates
3.3. S. typhimurium Cells Killed during Direct Contact with MEO and CEO In Vitro
3.4. Effects of MEO and CEO Vapors on the Viability of S. typhimurium and S. aureus
3.5. Effects of CEO and MEO Vapors on S. typhimurium-Infected Chicken Skin
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pandey, A.K.; Kumar, P.; Singh, P.; Tripathi, N.N.; Bajpai, V.K. Essential Oils: Sources of Antimicrobials and Food Preservatives. Front. Microbiol. 2016, 7, 2161. [Google Scholar] [CrossRef] [PubMed]
- Yaghoubi, M.; Alirezalu, K.; Hesari, J.; Peighambardoust, S.H.; Marcinkowska-Lesiak, M.; Barzegar, Y.; Hoseinian-Khosrowshahi, S.R.; Marszałek, K.; Mousavi Khaneghah, A. Application of Oleaster Leaves (Elaeagnus Angustifolia L.) Essential Oil and Natural Nanoparticle Preservatives in Frankfurter-Type Sausages: An Assessment of Quality Attributes and Stability during Refrigerated Storage. Meat Sci. 2023, 198, 109097. [Google Scholar] [CrossRef] [PubMed]
- Pateiro, M.; Munekata, P.E.S.; Sant’Ana, A.S.; Domínguez, R.; Rodríguez-Lázaro, D.; Lorenzo, J.M. Application of Essential Oils as Antimicrobial Agents against Spoilage and Pathogenic Microorganisms in Meat Products. Int. J. Food Microbiol. 2021, 337, 108966. [Google Scholar] [CrossRef] [PubMed]
- Burt, S. Essential Oils: Their Antibacterial Properties and Potential Applications in Foods-a Review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Eng, S.-K.; Pusparajah, P.; Ab Mutalib, N.-S.; Ser, H.-L.; Chan, K.-G.; Lee, L.-H. Salmonella: A Review on Pathogenesis, Epidemiology and Antibiotic Resistance. Front. Life Sci. 2015, 8, 284–293. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Baek, K.-H.; Kang, S.C. Control of Salmonella in Foods by Using Essential Oils: A Review. Food Res. Int. 2012, 45, 722–734. [Google Scholar] [CrossRef]
- Carocho, M.; Barreiro, M.F.; Morales, P.; Ferreira, I.C.F.R. Adding Molecules to Food, Pros and Cons: A Review on Synthetic and Natural Food Additives. Compr. Rev. Food Sci. Food Saf. 2014, 13, 377–399. [Google Scholar] [CrossRef]
- Lucera, A.; Costa, C.; Conte, A.; Del Nobile, M.A. Food Applications of Natural Antimicrobial Compounds. Front. Microbiol. 2012, 3, 287. [Google Scholar] [CrossRef]
- Franco, C.M.; Vázquez, B.I. Natural Compounds as Antimicrobial Agents. Antibiotics 2020, 9, 217. [Google Scholar] [CrossRef]
- Gurtler, J.B.; Garner, C.M. A Review of Essential Oils as Antimicrobials in Foods with Special Emphasis on Fresh Produce. J. Food Prot. 2022, 85, 1300–1319. [Google Scholar] [CrossRef]
- Garcia-Oliveira, P.; Carreira-Casais, A.; Pereira, E.; Dias, M.I.; Pereira, C.; Calhelha, R.C.; Stojković, D.; Sokovic, M.; Simal-Gandara, J.; Prieto, M.A.; et al. From Tradition to Health: Chemical and Bioactive Characterization of Five Traditional Plants. Molecules 2022, 27, 6495. [Google Scholar] [CrossRef] [PubMed]
- Feyaerts, A.F.; Mathé, L.; Luyten, W.; Tournu, H.; Van Dyck, K.; Broekx, L.; Van Dijck, P. Assay and Recommendations for the Detection of Vapour-Phase-Mediated Antimicrobial Activities. Flavour. Fragr. J. 2017, 32, 347–353. [Google Scholar] [CrossRef]
- Feyaerts, A.F.; Mathé, L.; Luyten, W.; De Graeve, S.; Van Dyck, K.; Broekx, L.; Van Dijck, P. Essential Oils and Their Components Are a Class of Antifungals with Potent Vapour-Phase-Mediated Anti-Candida Activity. Sci. Rep. 2018, 8, 3958. [Google Scholar] [CrossRef] [PubMed]
- Tassou, C.C.; Drosinos, E.H.; Nychas, G.J. Effects of Essential Oil from Mint (Mentha Piperita) on Salmonella Enteritidis and Listeria Monocytogenes in Model Food Systems at 4 Degrees and 10 Degrees C. J. Appl. Bacteriol. 1995, 78, 593–600. [Google Scholar] [CrossRef]
- Al-Nabulsi, A.A.; Osaili, T.M.; Olaimat, A.N.; Almasri, W.E.; Ayyash, M.; Al-Holy, M.A.; Jaradat, Z.W.; Obaid, R.S.; Holley, R.A. Inactivation of Salmonella Spp. in Tahini Using Plant Essential Oil Extracts. Food Microbiol. 2020, 86, 103338. [Google Scholar] [CrossRef]
- Hąc-Wydro, K.; Flasiński, M.; Romańczuk, K. Essential Oils as Food Eco-Preservatives: Model System Studies on the Effect of Temperature on Limonene Antibacterial Activity. Food Chem. 2017, 235, 127–135. [Google Scholar] [CrossRef]
- Chen, L.; Zhao, X.; Li, R.; Yang, H. Integrated Metabolomics and Transcriptomics Reveal the Adaptive Responses of Salmonella Enterica Serovar Typhimurium to Thyme and Cinnamon Oils. Food Res. Int. 2022, 157, 111241. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, C.; Zhang, N.; Peng, Y.; Ma, Y.; Gu, K.; Liu, X.; Liu, X.; Liu, X.; Liu, Y.; et al. Menthone Exerts Its Antimicrobial Activity Against Methicillin Resistant Staphylococcus Aureus by Affecting Cell Membrane Properties and Lipid Profile. Drug Des. Devel. Ther. 2023, 17, 219–236. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, Y.; Chen, X.; Li, W.; Li, W.; Du, J.; Wang, L. Effects of Cinnamon Essential Oil on Oxidative Damage and Outer Membrane Protein Genes of Salmonella Enteritidis Cells. Foods 2022, 11, 2234. [Google Scholar] [CrossRef]
- Yin, L.; Dai, Y.; Chen, H.; He, X.; Ouyang, P.; Huang, X.; Sun, X.; Ai, Y.; Lai, S.; Zhu, L.; et al. Cinnamaldehyde Resist Salmonella Typhimurium Adhesion by Inhibiting Type I Fimbriae. Molecules 2022, 27, 7753. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, Y.; Chen, X.; Li, W.; Wang, L.; Li, W.; Du, J.; Zhang, S. Effects of Cinnamon Essential Oil on the Physiological Metabolism of Salmonella Enteritidis. Front. Microbiol. 2022, 13, 1035894. [Google Scholar] [CrossRef]
- de Sousa Guedes, J.P.; da Costa Medeiros, J.A.; de Souza, E.; Silva, R.S.; de Sousa, J.M.B.; da Conceição, M.L.; de Souza, E.L. The Efficacy of Mentha Arvensis L. and M. Piperita L. Essential Oils in Reducing Pathogenic Bacteria and Maintaining Quality Characteristics in Cashew, Guava, Mango, and Pineapple Juices. Int. J. Food Microbiol. 2016, 238, 183–192. [Google Scholar] [CrossRef]
- Clemente, I.; Aznar, M.; Nerín, C. Synergistic Properties of Mustard and Cinnamon Essential Oils for the Inactivation of Foodborne Moulds in vitro and on Spanish Bread. Int. J. Food Microbiol. 2019, 298, 44–50. [Google Scholar] [CrossRef]
- Zhao, H.; Ren, S.; Yang, H.; Tang, S.; Guo, C.; Liu, M.; Tao, Q.; Ming, T.; Xu, H. Peppermint Essential Oil: Its Phytochemistry, Biological Activity, Pharmacological Effect and Application. Biomed. Pharmacother. 2022, 154, 113559. [Google Scholar] [CrossRef]
- Salehi, B.; Stojanović-Radić, Z.; Matejić, J.; Sharopov, F.; Antolak, H.; Kręgiel, D.; Sen, S.; Sharifi-Rad, M.; Acharya, K.; Sharifi-Rad, R.; et al. Plants of Genus Mentha: From Farm to Food Factory. Plants 2018, 7, 70. [Google Scholar] [CrossRef]
- Işcan, G.; Kirimer, N.; Kürkcüoğlu, M.; Başer, K.H.C.; Demirci, F. Antimicrobial Screening of Mentha Piperita Essential Oils. J. Agric. Food Chem. 2002, 50, 3943–3946. [Google Scholar] [CrossRef]
- Silva, W.M.F.; Bona, N.P.; Pedra, N.S.; Da Cunha, K.F.; Fiorentini, A.M.; Stefanello, F.M.; Zavareze, E.R.; Dias, A.R.G. Risk Assessment of in vitro Cytotoxicity, Antioxidant and Antimicrobial Activities of Mentha Piperita L. Essential Oil. J. Toxicol. Environ. Health. A 2022, 85, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, A.K.; Gottardi, D.; Malik, A.; Guerzoni, M.E. Anti-Yeast Activity of Mentha Oil and Vapours through in vitro and in Vivo (Real Fruit Juices) Assays. Food Chem. 2013, 137, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Camele, I.; Gruľová, D.; Elshafie, H.S. Chemical Composition and Antimicrobial Properties of Mentha × Piperita Cv. “Kristinka” Essential Oil. Plants 2021, 10, 1567. [Google Scholar] [CrossRef]
- Erler, F. Fumigant Activity of Six Monoterpenoids from Aromatic Plants in Turkey against the Two Stored-Product Pests Confused Flour Beetle, Tribolium Confusum, and Mediterranean Flour Moth, Ephestia Kuehniella. J. Plant Dis. Prot. 2005, 112, 602–611. [Google Scholar] [CrossRef]
- Graf, M.; Stappen, I. Beyond the Bark: An Overview of the Chemistry and Biological Activities of Selected Bark Essential Oils. Molecules 2022, 27, 7295. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Dey, A.; Koirala, N.; Shaheen, S.; El Omari, N.; Salehi, B.; Goloshvili, T.; Cirone Silva, N.C.; Bouyahya, A.; Vitalini, S.; et al. Cinnamomum Species: Bridging Phytochemistry Knowledge, Pharmacological Properties and Toxicological Safety for Health Benefits. Front. Pharmacol. 2021, 12, 600139. [Google Scholar] [CrossRef] [PubMed]
- Helander, I.M.; Alakomi, H.-L.; Latva-Kala, K.; Mattila-Sandholm, T.; Pol, I.; Smid, E.J.; Gorris, L.G.M.; von Wright, A. Characterization of the Action of Selected Essential Oil Components on Gram-Negative Bacteria. J. Agric. Food Chem. 1998, 46, 3590–3595. [Google Scholar] [CrossRef]
- Unlu, M.; Ergene, E.; Unlu, G.V.; Zeytinoglu, H.S.; Vural, N. Composition, Antimicrobial Activity and in vitro Cytotoxicity of Essential Oil from Cinnamomum Zeylanicum Blume (Lauraceae). Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2010, 48, 3274–3280. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Quadrupole Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Puplishing Corp.: Carol Stream, IL, USA, 2007. [Google Scholar]
- Ribeiro-Santos, R.; Andrade, M.; de Melo, N.R.; Sanches-Silva, A. Use of Essential Oils in Active Food Packaging: Recent Advances and Future Trends. Trends Food Sci. Technol. 2017, 61, 132–140. [Google Scholar] [CrossRef]
- Pang, X.; Feng, Y.-X.; Qi, X.-J.; Wang, Y.; Almaz, B.; Xi, C.; Du, S.-S. Toxicity and Repellent Activity of Essential Oil from Mentha Piperita Linn. Leaves and Its Major Monoterpenoids against Three Stored Product Insects. Environ. Sci. Pollut. Res. Int. 2020, 27, 7618–7627. [Google Scholar] [CrossRef]
- Zore, G.; Thakre, A.; Abdulghani, M.; Bhosle, K.; Shelar, A.; Patil, R.; Kharat, K.; Karuppayil, S. Menthol Inhibits Candida Albicans Growth by Affecting the Membrane Integrity Followed by Apoptosis. Evid. Based. Complement. Alternat. Med. 2022, 2022, 1297888. [Google Scholar] [CrossRef]
- Masumoto, N.; Ito, M. Genetic Identification of the Original Plant Species for Mentha Herb Listed in the Japanese Pharmacopoeia and Analyses of Their Essential Oil Composition. J. Nat. Med. 2023, 77, 489–495. [Google Scholar] [CrossRef]
- Darby, J.; Sheorey, H. Searching for Salmonella. Aust. Fam. Physician 2008, 37, 806–810. [Google Scholar]
- Ebani, V.V.; Nardoni, S.; Bertelloni, F.; Tosi, G.; Massi, P.; Pistelli, L.; Mancianti, F. In Vitro Antimicrobial Activity of Essential Oils Against Salmonella Enterica Serotypes Enteritidis and Typhimurium Strains Isolated from Poultry. Molecules 2019, 24, 900. [Google Scholar] [CrossRef]
- Soković, M.; Glamočlija, J.; Marin, P.D.; Brkić, D.; van Griensven, L.J.L.D. Antibacterial Effects of the Essential Oils of Commonly Consumed Medicinal Herbs Using an in vitro Model. Molecules 2010, 15, 7532–7546. [Google Scholar] [CrossRef]
- Chahbi, A.; Nassik, S.; El Amri, H.; Douaik, A.; El Maadoudi, E.H.; Boukharta, M.; El Hadrami, E.M. Chemical Composition and Antimicrobial Activity of the Essential Oils of Two Aromatic Plants Cultivated in Morocco (Cinnamomum Cassia and Origanum Compactum). J. Chem. 2020, 2020, 1628710. [Google Scholar] [CrossRef]
- Jirovetz, L.; Buchbauer, G.; Bail, S.; Denkova, Z.; Slavchev, A.; Stoyanova, A.; Schmidt, E.; Geissler, M. Antimicrobial Activities of Essential Oils of Mint and Peppermint as Well as Some of Their Main Compounds. J. Essent. Oil Res. 2009, 21, 363–366. [Google Scholar] [CrossRef]
- Campana, R.; Casettari, L.; Fagioli, L.; Cespi, M.; Bonacucina, G.; Baffone, W. Activity of Essential Oil-Based Microemulsions against Staphylococcus Aureus Biofilms Developed on Stainless Steel Surface in Different Culture Media and Growth Conditions. Int. J. Food Microbiol. 2017, 241, 132–140. [Google Scholar] [CrossRef]
- Chang, S.T.; Chen, P.F.; Chang, S.C. Antibacterial Activity of Leaf Essential Oils and Their Constituents from Cinnamomum Osmophloeum. J. Ethnopharmacol. 2001, 77, 123–127. [Google Scholar] [CrossRef]
- Bhatia, R.; Shreaz, S.; Khan, N.; Muralidhar, S.; Basir, S.F.; Manzoor, N.; Khan, L.A. Proton Pumping ATPase Mediated Fungicidal Activity of Two Essential Oil Components. J. Basic Microbiol. 2012, 52, 504–512. [Google Scholar] [CrossRef]
- Smadi, H.; Sargeant, J.M.; Shannon, H.S.; Raina, P. Growth and Inactivation of Salmonella at Low Refrigerated Storage Temperatures and Thermal Inactivation on Raw Chicken Meat and Laboratory Media: Mixed Effect Meta-Analysis. J. Epidemiol. Glob. Health 2012, 2, 165–179. [Google Scholar] [CrossRef][Green Version]
- Duan, X.; Qin, D.; Li, H.; Zhang, T.; Han, Y.; Huang, Y.Q.; He, D.; Wu, K.; Chai, X.; Chen, C. Study of Antimicrobial Activity and Mechanism of Vapor-Phase Cinnamaldehyde for Killing Escherichia Coli Based on Fumigation Method. Front. Nutr. 2022, 9, 1040152. [Google Scholar] [CrossRef]
- Wesche, A.M.; Gurtler, J.B.; Marks, B.P.; Ryser, E.T. Stress, Sublethal Injury, Resuscitation, and Virulence of Bacterial Foodborne Pathogens. J. Food Prot. 2009, 72, 1121–1138. [Google Scholar] [CrossRef]
- Kadariya, J.; Smith, T.C.; Thapaliya, D. Staphylococcus Aureus and Staphylococcal Food-Borne Disease: An Ongoing Challenge in Public Health. Biomed Res. Int. 2014, 2014, 827965. [Google Scholar] [CrossRef]
- Yun, J.; Fan, X.; Li, X. Inactivation of Salmonella Enterica Serovar Typhimurium and Quality Maintenance of Cherry Tomatoes Treated with Gaseous Essential Oils. J. Food Sci. 2013, 78, M458–M464. [Google Scholar] [CrossRef]
- Yin, Y.; Lee, S.-Y. Current View of Ligand and Lipid Recognition by the Menthol Receptor TRPM8. Trends Biochem. Sci. 2020, 45, 806–819. [Google Scholar] [CrossRef]
- Schelz, Z.; Molnar, J.; Hohmann, J. Antimicrobial and Antiplasmid Activities of Essential Oils. Fitoterapia 2006, 77, 279–285. [Google Scholar] [CrossRef]
- Hettmann, K.; Monnard, F.W.; Melo Rodriguez, G.; Hilty, F.M.; Yildirim, S.; Schoelkopf, J. Porous Coatings to Control Release Rates of Essential Oils to Generate an Atmosphere with Botanical Actives. Materials 2022, 15, 2155. [Google Scholar] [CrossRef]
- Oscar, T.P. Predictive Models for Growth of Salmonella Typhimurium DT104 from Low and High Initial Density on Ground Chicken with a Natural Microflora. Food Microbiol. 2007, 24, 640–651. [Google Scholar] [CrossRef]
- Fratianni, F.; De Martino, L.; Melone, A.; De Feo, V.; Coppola, R.; Nazzaro, F. Preservation of Chicken Breast Meat Treated with Thyme and Balm Essential Oils. J. Food Sci. 2010, 75, M528–M535. [Google Scholar] [CrossRef]
- Thanissery, R.; Smith, D.P. Marinade with Thyme and Orange Oils Reduces Salmonella Enteritidis and Campylobacter Coli on Inoculated Broiler Breast Fillets and Whole Wings. Poult. Sci. 2014, 93, 1258–1262. [Google Scholar] [CrossRef]
- Barbosa, L.N.; Probst, I.S.; Murbach Teles Andrade, B.F.; Bérgamo Alves, F.C.; Albano, M.; Mores Rall, V.L.; Júnior, A.F. Essential Oils from Herbs against Foodborne Pathogens in Chicken Sausage. J. Oleo Sci. 2015, 64, 117–124. [Google Scholar] [CrossRef]
- Porter, J.A.; Morey, A.; Monu, E.A. Antimicrobial Efficacy of White Mustard Essential Oil and Carvacrol against Salmonella in Refrigerated Ground Chicken. Poult. Sci. 2020, 99, 5091–5095. [Google Scholar] [CrossRef]
- Dewi, G.; Nair, D.V.T.; Peichel, C.; Johnson, T.J.; Noll, S.; Kollanoor Johny, A. Effect of Lemongrass Essential Oil against Multidrug-Resistant Salmonella Heidelberg and Its Attachment to Chicken Skin and Meat. Poult. Sci. 2021, 100, 101116. [Google Scholar] [CrossRef]
- Das, J.K.; Chatterjee, N.; Pal, S.; Nanda, P.K.; Das, A.; Das, L.; Dhar, P.; Das, A.K. Effect of Bamboo Essential Oil on the Oxidative Stability, Microbial Attributes and Sensory Quality of Chicken Meatballs. Foods 2023, 12, 218. [Google Scholar] [CrossRef] [PubMed]
- Azizah, F.; Nursakti, H.; Ningrum, A. Supriyadi Development of Edible Composite Film from Fish Gelatin-Pectin Incorporated with Lemongrass Essential Oil and Its Application in Chicken Meat. Polymers 2023, 15, 2075. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.S.; Haq, M.; Roy, V.C.; Ho, T.C.; Park, J.-S.; Han, J.-M.; Chun, B.-S. Development of Fish Gelatin/Carrageenan/Zein Bio-Nanocomposite Active-Films Incorporated with Turmeric Essential Oil and Their Application in Chicken Meat Preservation. Colloids Surf. B Biointerfaces 2023, 226, 113320. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.; Arfat, Y.A.; Bher, A.; Mulla, M.; Jacob, H.; Auras, R. Active Chicken Meat Packaging Based on Polylactide Films and Bimetallic Ag-Cu Nanoparticles and Essential Oil. J. Food Sci. 2018, 83, 1299–1310. [Google Scholar] [CrossRef] [PubMed]

| Wild Mint EO (MEO) | Chinese Cinnamon Bark EO (CEO) | |
|---|---|---|
| Botanical name | Mentha arvensis | Cinnamomum cassia |
| Batch number | LOT: MA02-06-18 | P244801 |
| Country of origin | Nepal | Australia |
| Best before | June 2023 | February 2026 |
| Compound Name | RILit/RIExp | MEO, % | CEO, % |
|---|---|---|---|
| α-pinene | 939/939 | 1.07 | 0.10 |
| β-pinene | 979/978 | 1.06 | 0.04 |
| Limonene | 1029/1030 | 2.73 | 0.07 |
| Menthone | 1152/1152 | 7.9 | |
| Isomenthone | 1162/1162 | 6.42 | |
| Menthol | 1171/1172 | 68.61 | |
| Geraniol | 1252/1258 | 2.01 | |
| (E)-cinnamaldehyde | 1270/1271 | 83.32 | |
| Menthyl acetate | 1295/1295 | 2.1 | |
| (E)-cinnamyl acetate | 1446/1445 | 4.69 | |
| (E)-methoxy-cinnamaldehyde | 1528/1529 | 7.62 | |
| Total | 91.90 | 95.86 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vepštaitė-Monstavičė, I.; Ravoitytė, B.; Būdienė, J.; Valys, A.; Lukša, J.; Servienė, E. Essential Oils of Mentha arvensis and Cinnamomum cassia Exhibit Distinct Antibacterial Activity at Different Temperatures In Vitro and on Chicken Skin. Foods 2023, 12, 3938. https://doi.org/10.3390/foods12213938
Vepštaitė-Monstavičė I, Ravoitytė B, Būdienė J, Valys A, Lukša J, Servienė E. Essential Oils of Mentha arvensis and Cinnamomum cassia Exhibit Distinct Antibacterial Activity at Different Temperatures In Vitro and on Chicken Skin. Foods. 2023; 12(21):3938. https://doi.org/10.3390/foods12213938
Chicago/Turabian StyleVepštaitė-Monstavičė, Iglė, Bazilė Ravoitytė, Jurga Būdienė, Algirdas Valys, Juliana Lukša, and Elena Servienė. 2023. "Essential Oils of Mentha arvensis and Cinnamomum cassia Exhibit Distinct Antibacterial Activity at Different Temperatures In Vitro and on Chicken Skin" Foods 12, no. 21: 3938. https://doi.org/10.3390/foods12213938
APA StyleVepštaitė-Monstavičė, I., Ravoitytė, B., Būdienė, J., Valys, A., Lukša, J., & Servienė, E. (2023). Essential Oils of Mentha arvensis and Cinnamomum cassia Exhibit Distinct Antibacterial Activity at Different Temperatures In Vitro and on Chicken Skin. Foods, 12(21), 3938. https://doi.org/10.3390/foods12213938







