Wolffia globosa-Based Nutritious Snack Formulation with High Protein and Dietary Fiber Contents
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Preparation of Snack Product
2.3. Experimental Design by Response Surface Methodology (RSM)
2.4. Determination of Physical Properties
2.5. Nutritional and Amino Acid Profiles Analysis
2.6. Sensory Evaluation
2.7. Microbiological Quality
2.8. Sample Extraction and Determination of Phytochemicals
2.9. Determination of Antioxidant Properties
2.10. Bacterial Reverse Mutation Assay (Ames Test)
2.11. Statistical Anylysis
3. Results
3.1. Effect of Different Ingredient Ratios on Snack Quality
3.1.1. Physical Properties
3.1.2. Nutritional Values
3.1.3. Sensory Evaluation
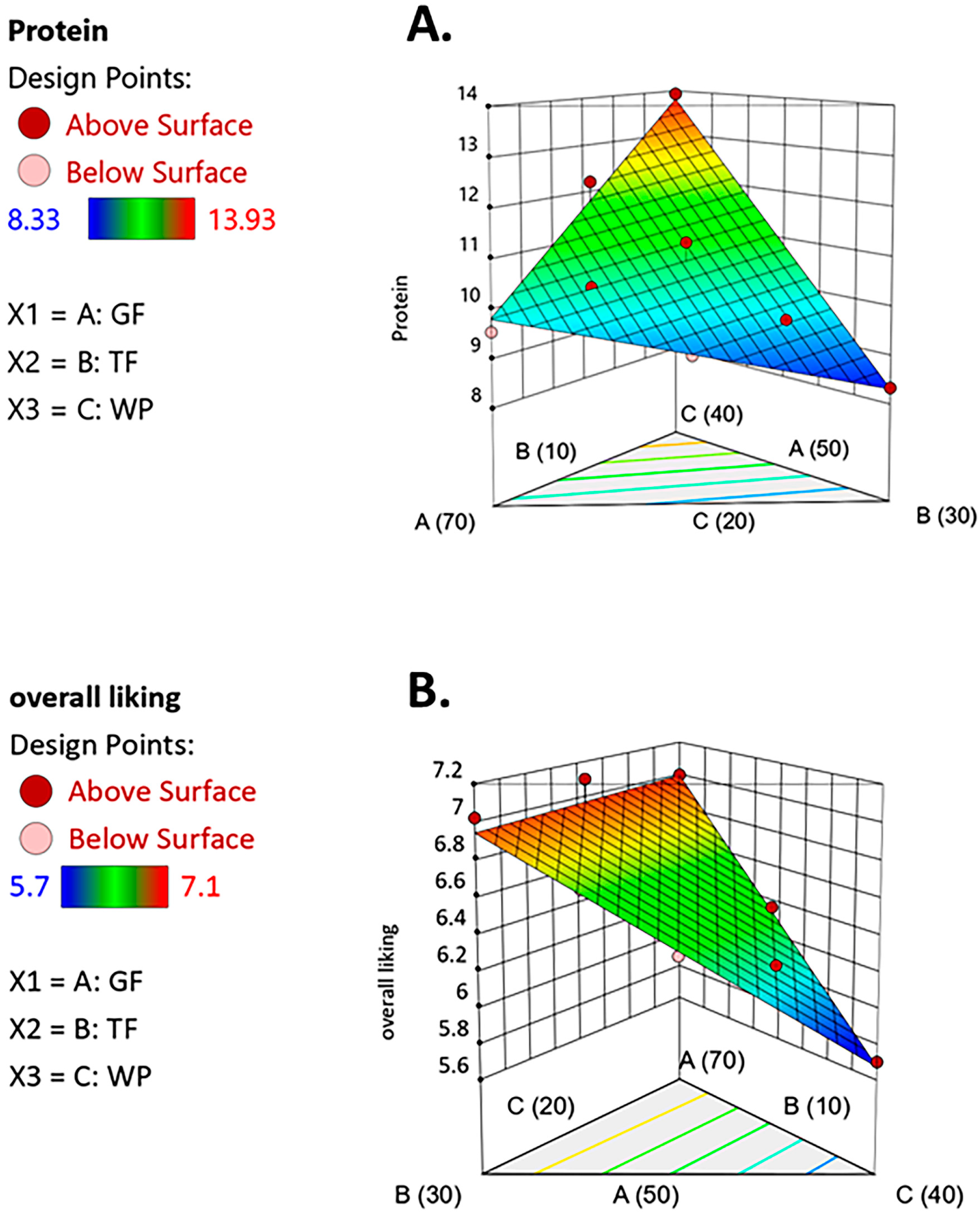
3.2. Optimization of Ingredients for W. globosa Snack Development
3.3. Comparison of the Control and Developed W. globosa Snack Quality
3.3.1. Nutritional Values and Amino Acid Profiles
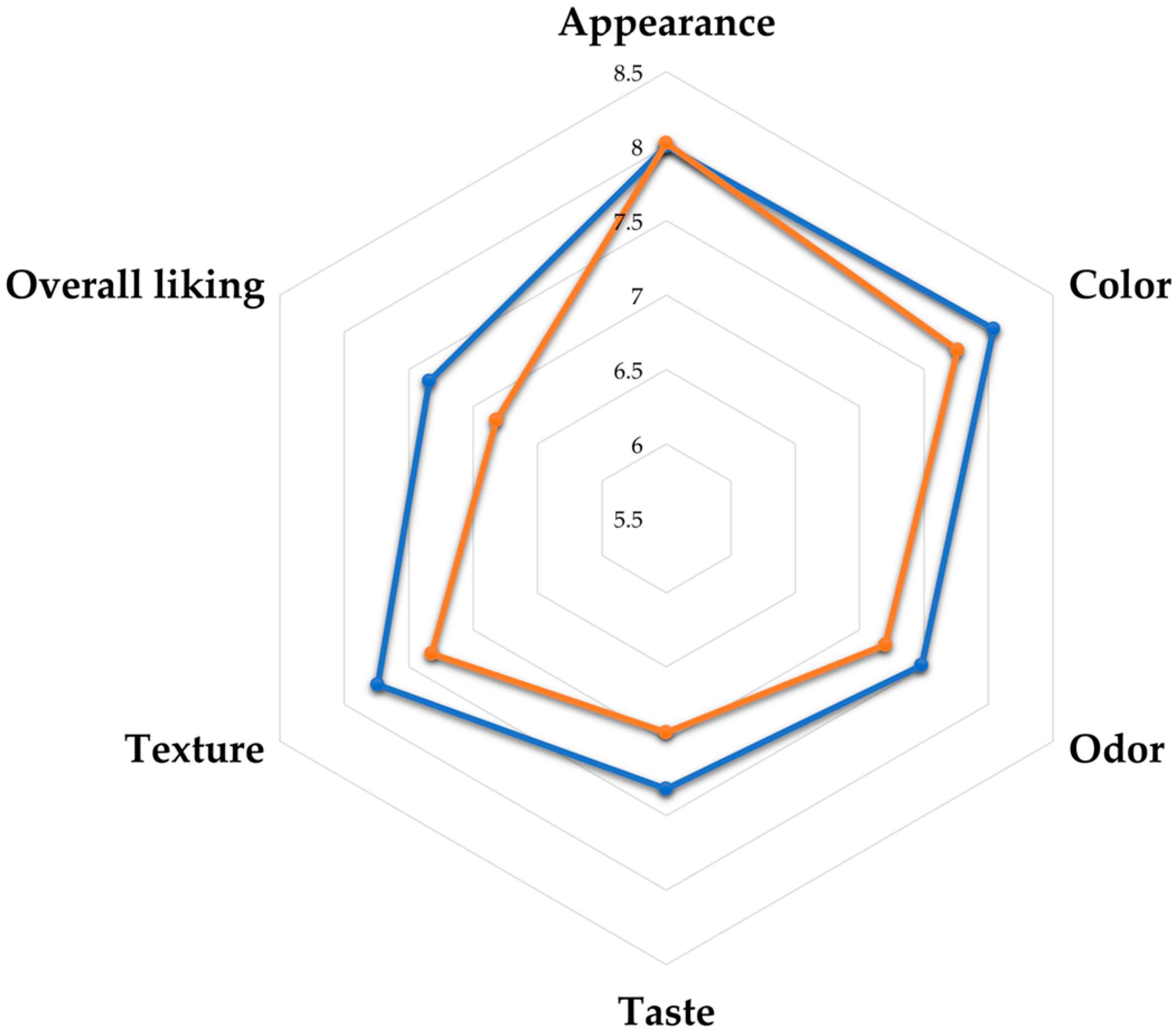
3.3.2. Sensory Evaluation and Microbiological Quality
3.4. Phytochemicals and Antioxidant Activities of W. globosa Snack
3.5. Evaluation of Mutagenicity Potential of W. globosa Powder (WP) Ethanolic Extract
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bhutta, Z.A.; Sadiq, K. Protein Deficiency. In Encyclopedia of Human Nutrition, 3rd ed.; Caballero, B., Ed.; Academic Press: Waltham, MA, USA, 2013; pp. 111–115. [Google Scholar] [CrossRef]
- Biswas, V.; Praveen, A.; Marisetti, A.L.; Sharma, A.; Kumar, V.; Sahu, S.K.; Tewari, D. A Mechanistic Overview on Impact of Dietary Fibres on Gut Microbiota and Its Association with Colon Cancer. Dietetics 2022, 1, 182–202. [Google Scholar] [CrossRef]
- World Health Organization. Fruit and Vegetables for Health: Report of the Joint FAO/WHO Workshop on Fruit and Vegetables for Health, 1–3 September 2004, Kobe, Japan; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Malnutrition. Available online: https://www.nhs.uk/conditions/malnutrition/ (accessed on 22 April 2023).
- Hess, J.M.; Jonnalagadda, S.S.; Slavin, J.L. What Is a Snack, Why Do We Snack, and How Can We Choose Better Snacks? A Review of the Definitions of Snacking, Motivations to Snack, Contributions to Dietary Intake, and Recommendations for Improvement. Adv. Nutr. 2016, 7, 466–475. [Google Scholar] [CrossRef]
- Zhang, J.; Azizullah, A. Genetic Diversity and DNA Barcoding in the Duckweed Family. In The Duckweed Genomes; Cao, X.H., Fourounjian, P., Wang, W., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 59–65. [Google Scholar]
- Appenroth, K.J.; Sree, K.S.; Bog, M.; Ecker, J.; Seeliger, C.; Böhm, V.; Lorkowski, S.; Sommer, K.; Vetter, W.; Tolzin-Banasch, K.; et al. Nutritional Value of the Duckweed Species of the Genus Wolffia (Lemnaceae) as Human Food. Front. Chem. 2018, 6, 483. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Shen, Y.; Zheng, Y.; Smith, G.; Sun, X.S.; Wang, D.; Zhao, Y.; Zhang, W.; Li, Y. Duckweed (Lemnaceae) for potentially nutritious human food: A review. Food Rev. Int. 2021, 1–15. [Google Scholar] [CrossRef]
- Baek, G.; Saeed, M.; Choi, H.K. Duckweeds: Their utilization, metabolites and cultivation. Appl. Biol. Chem. 2021, 64, 73. [Google Scholar] [CrossRef]
- Ghosh, S.; Lee, S.-M.; Jung, C.; Meyer-Rochow, V.B. Nutritional composition of five commercial edible insects in South Korea. J. Asia-Pac. Entomol. 2017, 20, 686–694. [Google Scholar] [CrossRef]
- Joint WHO/FAO/UNU Expert Consultation. Protein and amino acid requirements in human nutrition. World Health Organ Tech. Rep. Ser. 2007, 935, 1–265. [Google Scholar]
- Ruekaewma, N.; Piyatiratitivorakul, S.; Powtongsook, S. Culture system for Wolffia globosa L. (Lemnaceae) for hygiene human food. Songklanakarin J. Sci. Technol. 2015, 35, 575–580. [Google Scholar]
- Appenroth, K.-J.; Sree, K.S.; Böhm, V.; Hammann, S.; Vetter, W.; Leiterer, M.; Jahreis, G. Nutritional value of duckweeds (Lemnaceae) as human food. Food Chem. 2017, 217, 266–273. [Google Scholar] [CrossRef]
- Monthakantirat, O.; Chulikhit, Y.; Maneenet, J.; Khamphukdee, C.; Chotritthirong, Y.; Limsakul, S.; Punya, T.; Turapra, B.; Boonyarat, C.; Daodee, S. Total Active Compounds and Mineral Contents in Wolffia globosa. J. Chem. 2022, 2022, 9212872. [Google Scholar] [CrossRef]
- Trautwein, E.A.; Vermeer, M.A.; Hiemstra, H.; Ras, R.T. LDL-Cholesterol Lowering of Plant Sterols and Stanols-Which Factors Influence Their Efficacy? Nutrients 2018, 10, 1262. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Gan, R.Y.; Li, S.; Zhou, Y.; Li, A.N.; Xu, D.P.; Li, H.B. Antioxidant Phytochemicals for the Prevention and Treatment of Chronic Diseases. Molecules 2015, 20, 21138–21156. [Google Scholar] [CrossRef] [PubMed]
- Yaskolka Meir, A.; Tsaban, G.; Zelicha, H.; Rinott, E.; Kaplan, A.; Youngster, I.; Rudich, A.; Shelef, I.; Tirosh, A.; Brikner, D.; et al. A Green-Mediterranean Diet, Supplemented with Mankai Duckweed, Preserves Iron-Homeostasis in Humans and Is Efficient in Reversal of Anemia in Rats. J. Nutr. 2019, 149, 1004–1011. [Google Scholar] [CrossRef]
- Engelhaupt, E. Why Death Smells so Deadly. Available online: https://www.sciencenews.org/blog/gory-details/why-death-smells-so-deadly (accessed on 11 May 2023).
- Khemthong, C.; Chamchan, R.; Suttisansanee, U.; Charoenkiatkul, S.; Chupeerach, C.; On-Nom, N. Development of Healthy Snack from Sa-Med Mushroom (Boletus griseipurpureus Corner). Walailak J. Sci. Technol. 2020, 17, 1157–1167. [Google Scholar] [CrossRef]
- Thai Recommended Daily Intakes (Thai RDIs), Appendix No.3; Royal Thai Government Gazette: Nonthaburi, Thailand, 1998.
- MOPH Notification No. 392 B.E. 2561 Re: Nutrition Labelling (No.3); Royal Thai Government Gazette: Nonthaburi, Thailand, 2018.
- Gámbaro, A.; Giménez, A.; Ares, G.; Gilardi, V. Influence of enzymes on the texture of brown pan bread. J. Texture Stud. 2006, 37, 300–314. [Google Scholar] [CrossRef]
- Giménez, A.; Varela, P.; Salvador, A.; Ares, G.; Fiszman, S.; Garitta, L. Shelf life estimation of brown pan bread: A consumer approach. Food Qual. Prefer. 2007, 18, 196–204. [Google Scholar] [CrossRef]
- Giménez, A.; Ares, G.; Gámbaro, A. Survival analysis to estimate sensory shelf life using acceptability scores. J. Sens. Stud. 2008, 23, 571–582. [Google Scholar] [CrossRef]
- Chiu, H.-W.; Peng, J.-C.; Tsai, S.-J.; Tsay, J.-R.; Lui, W.-B. Process Optimization by Response Surface Methodology and Characteristics Investigation of Corn Extrudate Fortified with Yam (Dioscorea alata L.). Food Bioprocess Technol. 2013, 6, 1494–1504. [Google Scholar] [CrossRef]
- Keeratipibul, S.; Luangsakul, N.; Lertsatchayarn, T. The effect of Thai glutinous rice cultivars, grain length and cultivating locations on the quality of rice cracker (arare). LWT-Food Sci. Technol. 2008, 41, 1934–1943. [Google Scholar] [CrossRef]
- AOAC. Official Method of Analysis of AOAC Internation, 21st ed.; AOAC International: Arlington, VA, USA, 2019. [Google Scholar]
- Official Journal of the European Communities, L 257/16, 16 September 1983. 1983. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:1998:257:0014:0028:En:PDF (accessed on 16 January 2023).
- Meilgaard, M.; Civille, G.V.; Carr, B.T. Sensory Evaluation Techniques, 4th ed.; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- FDA. Bacteriological Analytical Manual (BAM); US Food and Drug Administration: Silver Spring, MD, USA, 2001.
- Hu, Z.; Fang, Y.; Yi, Z.; Tian, X.; Li, J.; Jin, Y.; He, K.; Liu, P.; Du, A.; Huang, Y.; et al. Determining the nutritional value and antioxidant capacity of duckweed (Wolffia arrhiza) under artificial conditions. LWT-Food Sci. Technol. 2022, 153, 112477. [Google Scholar] [CrossRef]
- Subedi, S.; Suttisansanee, U.; Kettawan, A.; Chupeerach, C.; Khemthong, C.; Thangsiri, S.; On-Nom, N. Food Fortification of Instant Pulse Porridge Powder with Improved Iron and Zinc Bioaccessibility Using Roselle Calyx. Nutrients 2022, 14, 4070. [Google Scholar] [CrossRef] [PubMed]
- Sirichai, P.; Kittibunchakul, S.; Thangsiri, S.; On-Nom, N.; Chupeerach, C.; Temviriyanukul, P.; Inthachat, W.; Nuchuchua, O.; Aursalung, A.; Sahasakul, Y. Impact of drying processes on phenolics and in vitro health-related activities of indigenous plants in Thailand. Plants 2022, 11, 294. [Google Scholar] [CrossRef] [PubMed]
- OECD. Test No. 471: Bacterial Reverse Mutation Test; Organisation for Economic Co-Operation and Development: Paris, France, 2020. [Google Scholar] [CrossRef]
- Inthachat, W.; Temviriyanukul, P.; On-Nom, N.; Kanoongon, P.; Thangsiri, S.; Chupeerach, C.; Suttisansanee, U. Optimization of Phytochemical-Rich Citrus maxima Albedo Extract Using Response Surface Methodology. Molecules 2023, 28, 4121. [Google Scholar] [CrossRef] [PubMed]
- TISI. Thai Community Product Standard, Crispy Snack; Thai Industrial Standards Institute: Bangkok, Thailand, 2011.
- Félix-Medina, J.V.; Gutiérrez-Dorado, R.; López-Valenzuela, J.A.; López-Ángulo, G.; Quintero-Soto, M.F.; Perales-Sánchez, J.X.K.; Montes-Ávila, J. Nutritional, antioxidant and phytochemical characterization of healthy ready-to-eat expanded snack produced from maize/common bean mixture by extrusion. LWT-Food Sci. Technol. 2021, 142, 111053. [Google Scholar] [CrossRef]
- Félix-Medina, J.V.; Montes-Ávila, J.; Reyes-Moreno, C.; Perales-Sánchez, J.X.K.; Gómez-Favela, M.A.; Aguilar-Palazuelos, E.; Gutiérrez-Dorado, R. Second-generation snacks with high nutritional and antioxidant value produced by an optimized extrusion process from corn/common bean flours mixtures. LWT-Food Sci. Technol. 2020, 124, 109172. [Google Scholar] [CrossRef]
- Bhanthumnavin, K.; McGarry, M.G. Wolffia arrhiza as a Possible Source of Inexpensive Protein. Nature 1971, 232, 495. [Google Scholar] [CrossRef]
- Kaplan, A.; Zelicha, H.; Tsaban, G.; Yaskolka Meir, A.; Rinott, E.; Kovsan, J.; Novack, L.; Thiery, J.; Ceglarek, U.; Burkhardt, R.; et al. Protein bioavailability of Wolffia globosa duckweed, a novel aquatic plant-A randomized controlled trial. Clin. Nutr. 2019, 38, 2576–2582. [Google Scholar] [CrossRef]
- Zelicha, H.; Kaplan, A.; Yaskolka Meir, A.; Tsaban, G.; Rinott, E.; Shelef, I.; Tirosh, A.; Brikner, D.; Pupkin, E.; Qi, L.; et al. The Effect of Wolffia globosa Mankai, a Green Aquatic Plant, on Postprandial Glycemic Response: A Randomized Crossover Controlled Trial. Diabetes Care 2019, 42, 1162–1169. [Google Scholar] [CrossRef]
- Zelicha, H.; Kloting, N.; Kaplan, A.; Yaskolka Meir, A.; Rinott, E.; Tsaban, G.; Chassidim, Y.; Bluher, M.; Ceglarek, U.; Isermann, B.; et al. The effect of high-polyphenol Mediterranean diet on visceral adiposity: The DIRECT PLUS randomized controlled trial. BMC Med. 2022, 20, 327. [Google Scholar] [CrossRef]
- Li, M.; Zhu, K.; Guo, X.; Peng, W.; Zhou, H. Effect of water activity (aw) and irradiation on the shelf-life of fresh noodles. Innov. Food Sci. Emerg. Technol. 2011, 12, 526–530. [Google Scholar] [CrossRef]
- Rocha, T.; Marty-Audouin, C.; Lebert, A. Effect of drying temperature and blanching on the degradation of chlorophyll a and b in mint (Mentha spicata Huds.) and basil (Ocimum basilicum): Analysis by high performance liquid chromatography with photodiode array detection. Chromatographia 1993, 36, 152–156. [Google Scholar] [CrossRef]
- Siripahanakul, T.; Thongsila, S.; Tanuthong, T.; Chockchaisawasdee, S. Product development of Wolffia-pork ball. Int. Food Res. J. 2013, 20, 213–217. [Google Scholar]
- Hirunyophat, P.; Chalermchaiwat, P.; On-nom, N. Optimization of ratio of silkworm pupae powder to broken rice flour and of barrel temperature to develop high protein breakfast cereal using response surface methodology. Agric. Nat. Resour. 2020, 54, 609–616. [Google Scholar]
- Nascimento, T.A.; Calado, V.; Carvalho, C.W.P. Effect of Brewer’s spent grain and temperature on physical properties of expanded extrudates from rice. LWT-Food Sci. Technol. 2017, 79, 145–151. [Google Scholar] [CrossRef]
- Parada, J.; Aguilera, J.M.; Brennan, C. Effect of guar gum content on some physical and nutritional properties of extruded products. J. Food Eng. 2011, 103, 324–332. [Google Scholar] [CrossRef]
- Duangjarus, N.; Chaiworapuek, W.; Rachtanapun, C.; Ritthiruangdej, P.; Charoensiddhi, S. Antimicrobial and Functional Properties of Duckweed (Wolffia globosa) Protein and Peptide Extracts Prepared by Ultrasound-Assisted Extraction. Foods 2022, 11, 2348. [Google Scholar] [CrossRef]
- Promsakha na Sakon Nakhon, P.; Jangchud, K.; Jangchud, A.; Charunuch, C. Optimization of pumpkin and feed moisture content to produce healthy pumpkin-germinated brown rice extruded snacks. Agric. Nat. Resour. 2018, 52, 550–556. [Google Scholar] [CrossRef]
- Ruiz-Armenta, X.A.; Zazueta-Morales, J.d.J.; Aguilar-Palazuelos, E.; Delgado-Nieblas, C.I.; López-Diaz, A.; Camacho-Hernández, I.L.; Gutiérrez-Dorado, R.; Martínez-Bustos, F. Effect of extrusion on the carotenoid content, physical and sensory properties of snacks added with bagasse of naranjita fruit: Optimization process. CYTA J. Food 2018, 16, 172–180. [Google Scholar] [CrossRef]
- Małecki, J.; Tomasevic, I.; Djekic, I.; Sołowiej, B.G. The Effect of Protein Source on the Physicochemical, Nutritional Properties and Microstructure of High-Protein Bars Intended for Physically Active People. Foods 2020, 9, 1467. [Google Scholar] [CrossRef]
- Binou, P.; Yanni, A.E.; Karathanos, V.T. Physical properties, sensory acceptance, postprandial glycemic response, and satiety of cereal based foods enriched with legume flours: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 2722–2740. [Google Scholar] [CrossRef]
- Escobedo, A.; Mojica, L. Pulse-based snacks as functional foods: Processing challenges and biological potential. Compr. Rev. Food Sci. Food Saf. 2021, 20, 4678–4702. [Google Scholar] [CrossRef] [PubMed]
- Sparvoli, F.; Giofré, S.; Cominelli, E.; Avite, E.; Giuberti, G.; Luongo, D.; Gatti, E.; Cianciabella, M.; Daniele, G.M.; Rossi, M.; et al. Sensory Characteristics and Nutritional Quality of Food Products Made with a Biofortified and Lectin Free Common Bean (Phaseolus vulgaris L.) Flour. Nutrients 2021, 13, 4517. [Google Scholar] [CrossRef] [PubMed]
- Holeček, M. Side effects of amino acid supplements. Physiol. Res. 2022, 71, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Rieu, I.; Balage, M.; Sornet, C.; Giraudet, C.; Pujos, E.; Grizard, J.; Mosoni, L.; Dardevet, D. Leucine supplementation improves muscle protein synthesis in elderly men independently of hyperaminoacidaemia. J. Physiol. 2006, 575, 305–315. [Google Scholar] [CrossRef]
- Xu, Z.R.; Tan, Z.J.; Zhang, Q.; Gui, Q.F.; Yang, Y.M. The effectiveness of leucine on muscle protein synthesis, lean body mass and leg lean mass accretion in older people: A systematic review and meta-analysis. Br. J. Nutr. 2015, 113, 25–34. [Google Scholar] [CrossRef]
- Arise, A.K.; Oriade, K.F.; Asogwa, T.N.; Nwachukwu, I. Amino acid profile, physicochemical and sensory properties of noodles produced from wheat-Bambara protein isolate. Meas. Food 2022, 5, 100020. [Google Scholar] [CrossRef]
- Arise, A.K.; Taiwo, G.O.; Malomo, S.A. Amino acid profile, pasting, and sensory properties of croissant snacks produced from wheat-fermented Bambara flour. Legume Sci. 2020, 2, e53. [Google Scholar] [CrossRef]
- Somdee, T.; Mahaweerawat, U.; Phadungkit, M.; Yangyuen, S. Antioxidant Compounds and Activities in Selected Fresh and Blanched Vegetables from Northeastern Thailand. Chiang Mai. J. Sci. 2016, 43, 834–844. [Google Scholar]
- Dewanji, A. Amino acid composition of leaf proteins extracted from some aquatic weeds. J. Agric. Food Chem. 1993, 41, 1232–1236. [Google Scholar] [CrossRef]
- Zhao, Y.; Fang, Y.; Jin, Y.; Huang, J.; Bao, S.; Fu, T.; He, Z.; Wang, F.; Wang, M.; Zhao, H. Pilot-scale comparison of four duckweed strains from different genera for potential application in nutrient recovery from wastewater and valuable biomass production. Plant Biol. 2015, 17 (Suppl. S1), 82–90. [Google Scholar] [CrossRef]
- Liang, N.; Kitts, D.D. Antioxidant property of coffee components: Assessment of methods that define mechanisms of action. Molecules 2014, 19, 19180–19208. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Nath, H.; Samtiya, M.; Dhewa, T. Beneficial attributes and adverse effects of major plant-based foods anti-nutrients on health: A review. Hum. Nutr. Metab. 2022, 28, 200147. [Google Scholar] [CrossRef]
| Run | Actual Variables | ||
|---|---|---|---|
| X1 (% w/w) | X2 (% w/w) | X3 (% w/w) | |
| 1 | 50 | 10 | 40 |
| 2 | 70 | 10 | 20 |
| 3 | 50 | 30 | 20 |
| 4 | 50 | 20 | 30 |
| 5 | 60 | 10 | 30 |
| 6 | 60 | 20 | 20 |
| 7 | 56.67 | 16.66 | 26.67 |
| 8 | 53.34 | 13.33 | 33.33 |
| 9 | 63.34 | 13.33 | 23.33 |
| 10 | 53.34 | 23.33 | 23.33 |
| Formulations | aw | Color | BD (g/mL) | Hardness (g) | ||
|---|---|---|---|---|---|---|
| L* | a* | b* | ||||
| 1 | 0.09 ± 0.00 g | 34.05 ± 0.05 h | −0.49 ± 0.00 a | 28.08 ± 0.11 c | 0.31 ± 0.00 a | 2448.54 ± 238.11 a |
| 2 | 0.23 ± 0.00 a | 38.70 ± 0.06 d | −1.57 ± 0.04 d | 28.93 ± 0.09 ab | 0.29 ± 0.00 b | 1950.84 ± 495.78 bc |
| 3 | 0.21 ± 0.01 b | 40.39 ± 0.01 b | −2.95 ± 0.02 f | 29.09 ± 0.05 a | 0.26 ± 0.01 c | 1658.63 ± 246.32 c |
| 4 | 0.19 ± 0.01 c | 39.02 ± 0.03 c | −2.69 ± 0.01 ef | 28.91 ± 0.07 bc | 0.22 ± 0.01 f | 2183.07 ± 170.56 ab |
| 5 | 0.20 ± 0.00 b | 40.62 ± 0.01 a | −2.51 ± 0.00 e | 28.59 ± 0.15 b | 0.22 ± 0.00 f | 1943.36 ± 507.53 bc |
| 6 | 0.23 ± 0.00 a | 40.27 ± 0.15 b | −1.11 ± 0.55 bc | 26.98 ± 0.60 d | 0.24 ± 0.01 d | 1950.68 ± 380.63 bc |
| 7 | 0.17 ± 0.00 e | 38.60 ± 0.00 d | −1.33 ± 0.02 cd | 27.44 ± 0.04 d | 0.23 ± 0.01 def | 1868.12 ± 472.86 bc |
| 8 | 0.10 ± 0.00 f | 37.40 ± 0.07 g | −0.85 ± 0.04 ab | 27.34 ± 0.19 d | 0.22 ± 0.00 ef | 1909.48 ± 373.28 bc |
| 9 | 0.16 ± 0.00 e | 37.89 ± 0.16 f | −0.93 ± 0.03 b | 27.00 ± 0.14 d | 0.23 ± 0.01 de | 2020.85 ± 301.14 bc |
| 10 | 0.17 ± 0.00 d | 38.40 ± 0.08 e | −0.79 ± 0.03 ab | 27.34 ± 0.11 d | 0.23 ± 0.01 de | 2010.21 ± 492.46 bc |
| Formulations | Energy (kcal) | Carbohydrate (g) | Protein (g) | Fat (g) | Dietary Fiber (g) |
|---|---|---|---|---|---|
| 1 | 428.06 ± 3.41 b | 59.34 ± 0.47 f | 13.93 ± 0.11 a | 12.95 ± 0.10 a | 12.63 ± 0.10 a |
| 2 | 420.45 ± 2.69 c | 66.50 ± 0.43 bc | 9.53 ± 0.06 g | 11.92 ± 0.08 e | 6.21 ± 0.04 hi |
| 3 | 415.11 ± 2.32 d | 66.74 ± 0.37 abc | 8.33 ± 0.05 i | 11.76 ± 0.07 f | 6.15 ± 0.03 i |
| 4 | 412.64 ± 1.35 d | 61.67 ± 0.20 e | 10.86 ± 0.04 e | 12.09 ± 0.04 d | 9.15 ± 0.03 d |
| 5 | 442.25 ± 0.70 a | 65.63 ± 0.10 d | 12.21 ± 0.02 b | 12.96 ± 0.02 a | 9.79 ± 0.02 c |
| 6 | 422.01 ± 2.91 c | 67.30 ± 0.46 a | 9.02 ± 0.06 h | 11.96 ± 0.08 e | 6.24 ± 0.04 h |
| 7 | 439.36 ± 1.84 a | 67.00 ± 0.28 ab | 11.03 ± 0.05 d | 12.73 ± 0.05 b | 8.66 ± 0.04 e |
| 8 | 404.24 ± 1.26 e | 58.84 ± 0.18 f | 11.65 ± 0.04 c | 11.97 ± 0.04 e | 9.95 ± 0.03 b |
| 9 | 429.85 ± 0.81 b | 66.78 ± 0.13 abc | 10.26 ± 0.02 f | 12.32 ± 0.02 c | 7.41 ± 0.01 f |
| 10 | 423.09 ± 2.32 c | 66.27 ± 0.36 c | 9.55 ± 0.05 g | 12.13 ± 0.07 d | 7.30 ± 0.04 g |
| Formulations | Appearance | Color ns | Odor | Taste | Texture | Overall Liking |
|---|---|---|---|---|---|---|
| 1 | 7.34 ± 0.88 abc | 7.00 ± 1.06 | 6.26 ± 1.52 c | 5.78 ± 1.63 c | 6.20 ± 1.47 d | 5.70 ± 1.72 d |
| 2 | 7.56 ± 0.98 a | 7.44 ± 1.00 | 7.16 ± 1.08 a | 7.00 ± 1.31 a | 7.26 ± 1.02 a | 7.00 ± 1.22 ab |
| 3 | 7.62 ± 0.93 a | 7.32 ± 0.97 | 7.14 ± 0.98 a | 6.90 ± 1.15 ab | 7.20 ± 1.02 ab | 7.02 ± 1.05 ab |
| 4 | 7.08 ± 0.93 c | 7.14 ± 0.98 | 6.58 ± 1.04 bc | 6.30 ± 1.14 bcd | 6.54 ± 1.17 cd | 6.28 ± 1.10 c |
| 5 | 7.12 ± 1.19 bc | 7.12 ± 1.12 | 6.50 ± 1.19 bc | 6.24 ± 1.19 cd | 6.68 ± 1.21 bcd | 6.36 ± 1.13 c |
| 6 | 7.52 ± 0.85 ab | 7.36 ± 0.79 | 7.16 ± 0.86 a | 6.84 ± 1.05 abc | 7.36 ± 0.84 a | 7.10 ± 0.88 a |
| 7 | 7.22 ± 1.03 abc | 7.18 ± 1.05 | 6.70 ± 1.24 abc | 6.44 ± 1.51 abc | 6.84 ± 1.47 abc | 6.46 ± 1.46 bc |
| 8 | 7.30 ± 1.06 abc | 7.14 ± 1.30 | 6.56 ± 1.34 bc | 6.24 ± 1.42 cd | 6.68 ± 1.25 bcd | 6.16 ± 1.57 cd |
| 9 | 7.50 ± 0.78 abc | 7.40 ± 0.80 | 6.96 ± 1.11 ab | 6.50 ± 1.45 abc | 7.12 ± 1.16 ab | 6.70 ± 1.06 abc |
| 10 | 7.48 ± 0.72 abc | 7.36 ± 0.91 | 6.68 ± 1.26 abc | 6.40 ± 1.50 abc | 7.02 ± 1.14 abc | 6.58 ± 1.39 abc |
| Response Variable | Predictive Model | R2 | p-Value |
|---|---|---|---|
| Protein | Y1 = 0.065(X1) − 0.009(X2) + 0.266(X3) | 0.97 | <0.05 |
| Overall liking | Y2 = 0.083(X1) + 0.081(X2) + 0.017(X3) | 0.96 | <0.05 |
| Ingredients (% w/w) | W. globosa Powder | Control Snack | W. globosa Snack |
|---|---|---|---|
| Glutinous rice flour | 0 | 70 | 64 |
| Tapioca flour | 0 | 30 | 10 |
| W. globosa powder | 100 | 0 | 26 |
| Nutritional Values | W. globosa Powder | Control Snack | W. globosa Snack |
|---|---|---|---|
| Energy (kcal) | 383.03 ± 0.71 b | 455.84 ± 5.51 a | 457.58 ± 5.35 a |
| Carbohydrate (g) | 52.59 ± 0.16 c | 78.49 ± 1.55 a | 71.50 ± 0.75 b |
| Fat (g) | 5.18 ± 0.01 b | 13.08 ± 0.16 a | 13.35 ± 0.08 a |
| Protein (g) | 31.50 ± 0.31 a | 6.28 ± 0.18 c | 12.86 ± 0.76 b |
| Dietary fiber (g) | 36.52 ± 0.21 a | 1.69 ± 0.01 c | 9.97 ± 0.07 b |
| Ash (g) | 10.73 ± 0.12 a | 1.72 ± 0.59 b | 2.79 ± 0.64 b |
| Amino Acid Profiles | W. globosa Powder | Control Snack | W. globosa Snack |
|---|---|---|---|
| Essential amino acids | |||
| Leucine | 2367.97 ± 2.28 a | 418.52 ± 1.39 c | 961.30 ± 1.30 b |
| Lysine | 1672.86 ± 3.39 a | 113.16 ± 0.52 c | 511.42 ± 0.08 b |
| Isoleucine | 1091.63 ± 0.92 a | 191.06 ± 0.26 c | 437.13 ± 0.40 b |
| Histidine | 539.18 ± 1.34 a | 116.97 ± 0.14 c | 256.73 ± 0.54 b |
| Tryptophan | 335.29 ± 0.74 a | 148.47 ± 0.73 c | 162.04 ± 0.22 b |
| Valine | 1681.18 ± 1.32 a | 285.52 ± 1.27 c | 691.22 ± 0.49 b |
| Methionine | 254.75 ± 1.80 a | ND | 199.02 ± 0.19 b |
| Phenylalanine | 1623.17 ± 3.15 a | 261.86 ± 0.56 c | 622.99 ± 0.30 b |
| Threonine | 1178.07 ± 2.77 a | 198.15 ± 0.43 c | 442.85 ± 0.55 b |
| TEAA | 10,743.08 | 1733.69 | 4284.68 |
| Nonessential amino acids | |||
| Tyrosine | 963.05 ± 2.79 a | 248.93 ± 1.34 c | 341.94 ± 0.10 b |
| Cystine | ND | ND | ND |
| Alanine | 2681.18 ± 2.26 a | 282.89 ± 0.96 c | 1000.18 ± 1.82 b |
| Glutamic acid | 3283.25 ± 3.06 a | 960.18 ± 1.18 c | 1668.82 ± 1.68 b |
| Glycine | 1476.53 ± 2.60 a | 207.82 ± 1.17 c | 575.78 ± 0.66 b |
| Arginine | 1692.51 ± 3.73 a | 368.75 ± 0.61 c | 754.03 ± 0.06 b |
| Aspartic acid | 3275.10 ± 1.62 a | 439.46 ± 0.80 c | 1290.45 ± 0.84 b |
| Serine | 1250.41 ± 2.76 a | 260.75 ± 1.36 c | 555.92 ± 0.32 b |
| Proline | 1267.16 ± 2.42 a | 228.41 ± 1.02 c | 478.92 ± 0.61 b |
| TNEAA | 15,889.16 | 2997.18 | 6666.02 |
| Hydrophilic amino acids | 4868.05 | 915.64 | 1916.48 |
| Hydrophobic amino acids | 10,966.02 | 1668.25 | 4390.74 |
| Acidic amino acids | 3904.55 | 1399.63 | 2959.27 |
| Basic amino acids | 6558.34 | 598.88 | 1522.18 |
| Samples | TPCs (mg of GAE/g DW) | TFCs (mg of QE/g DW) | TTCs (mg of TAE/g DW) | Antioxidant Activities (µmol of TE/g DW) | ||
|---|---|---|---|---|---|---|
| DPPH Radical Scavenging Assay | FRAP Assay | ORAC Assay | ||||
| WP extract | 11.67 ± 0.11 a | 12.51 ± 0.38 a | 32.31 ± 1.00 a | 55.01 ± 4.26 a | 63.19 ± 4.03 a | 397.52 ± 27.20 a |
| Control snack | ND | ND | 1.41 ± 0.17 c | 2.50 ± 0.21 c | ND | 0.83 ± 0.04 b |
| W. globosa snack | 2.94 ± 0.04 b | 3.11 ± 0.08 b | 6.97 ± 0.39 b | 8.95 ± 0.73 b | 15.94 ± 2.05 b | 191.80 ± 14.41 c |
| Doses (μg/Plate) | TA98 | TA100 | TA102 | TA1535 | TA1537 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Revertant Colonies | MR | Revertant Colonies | MR | Revertant Colonies | MR | Revertant Colonies | MR | Revertant Colonies | MR | |
| Neg | 82.83 ± 2.19 | 1.00 (–) | 69.83 ± 2.73 | 1.00 (–) | 353.67 ± 6.75 | 1.00 (–) | 9.83 ± 1.07 | 1.00 (–) | 11.17 ± 1.07 | 1.00 (–) |
| 10 | 84.83 ± 2.79 | 1.02 (–) | 67.17 ± 2.19 | 0.96 (–) | 356.50 ± 4.99 | 1.01 (–) | 10.00 ± 1.00 | 1.02 (–) | 10.00 ± 0.82 | 0.90 (–) |
| 100 | 81.50 ± 4.46 | 0.98 (–) | 67.50 ± 1.38 | 0.97 (–) | 357.33 ± 5.22 | 1.01 (–) | 10.67 ± 1.11 | 1.08 (–) | 10.33 ± 0.94 | 0.93 (–) |
| 500 | 82.67 ± 2.29 | 1.00 (–) | 69.50 ± 1.89 | 1.00 (–) | 362.33 ± 7.32 | 1.02 (–) | 11.00 ± 0.82 | 1.12 (–) | 9.83 ± 0.69 | 0.88 (–) |
| 1000 | 83.33 ± 3.45 | 1.01 (–) | 68.83 ± 3.02 | 0.99 (–) | 358.83 ± 6.39 | 1.01 (–) | 9.17 ± 0.69 | 0.93 (–) | 10.83 ± 0.69 | 0.97 (–) |
| 2000 | 84.67 ± 2.87 | 1.02 (–) | 68.50 ± 1.98 | 0.98 (–) | 364.33 ± 5.44 | 1.03 (–) | 10.17 ± 1.07 | 1.03 (–) | 11.00 ± 0.82 | 0.99 (–) |
| 4-NQO | 1071.33 ± 27.94 | 12.93 (+) | ||||||||
| NaN3 | 1147.33 ± 13.74 | 16.43 (+) | 260.50 ± 8.67 | 24.49 (+) | ||||||
| MMC | 1060.00 ± 24.00 | 3.00 (+) | ||||||||
| 9-AA | 742.67 ± 29.09 | 66.51 (+) | ||||||||
| Doses (μg/Plate) | TA98 | TA100 | TA102 | TA1535 | TA1537 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Revertant Colonies | MR | Revertant Colonies | MR | Revertant Colonies | MR | Revertant Colonies | MR | Revertant Colonies | MR | |
| Neg | 87.00 ± 3.56 | 1.00 (–) | 79.33 ± 3.99 | 1.00 (–) | 363.17 ± 5.24 | 1.00 (–) | 9.50 ± 0.76 | 1.00 (–) | 10.50 ± 1.26 | 1.00 (–) |
| 10 | 85.17 ± 3.13 | 0.98 (–) | 73.50 ± 2.43 | 0.93 (–) | 362.83 ± 4.52 | 1.00 (–) | 10.50 ± 1.26 | 1.11 (–) | 10.33 ± 0.94 | 0.98 (–) |
| 100 | 87.00 ± 4.00 | 1.00 (–) | 74.67 ± 3.14 | 0.94 (–) | 360.50 ± 4.96 | 0.99 (–) | 9.50 ± 0.96 | 1.00 (–) | 10.00 ± 0.82 | 0.95 (–) |
| 500 | 88.50 ± 3.95 | 1.02 (–) | 74.00 ± 3.56 | 0.93 (–) | 362.33 ± 6.18 | 1.00 (–) | 10.67 ± 0.94 | 1.12 (–) | 10.67 ± 1.11 | 1.02 (–) |
| 1000 | 90.33 ± 3.45 | 1.04 (–) | 73.83 ± 4.37 | 0.93 (–) | 360.17 ± 5.34 | 0.99 (–) | 10.33 ± 1.60 | 1.09 (–) | 10.00 ± 1.00 | 0.95 (–) |
| 2000 | 89.50 ± 2.22 | 1.03 (–) | 79.00 ± 3.96 | 1.00 (–) | 358.67 ± 5.31 | 0.99 (–) | 9.33 ± 0.47 | 0.98 (–) | 9.50 ± 0.76 | 0.90 (–) |
| 2-AA | 1125.33 ± 24.29 | 12.93 (+) | 1018.67 ± 48.20 | 11.73 (+) | 1140.00 ± 49.48 | 3.14 (+) | 370.33 ± 9.03 | 38.98 (+) | 199.33 ± 8.94 | 18.98 (+) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
On-Nom, N.; Promdang, P.; Inthachat, W.; Kanoongon, P.; Sahasakul, Y.; Chupeerach, C.; Suttisansanee, U.; Temviriyanukul, P. Wolffia globosa-Based Nutritious Snack Formulation with High Protein and Dietary Fiber Contents. Foods 2023, 12, 2647. https://doi.org/10.3390/foods12142647
On-Nom N, Promdang P, Inthachat W, Kanoongon P, Sahasakul Y, Chupeerach C, Suttisansanee U, Temviriyanukul P. Wolffia globosa-Based Nutritious Snack Formulation with High Protein and Dietary Fiber Contents. Foods. 2023; 12(14):2647. https://doi.org/10.3390/foods12142647
Chicago/Turabian StyleOn-Nom, Nattira, Prapatsorn Promdang, Woorawee Inthachat, Panyaporn Kanoongon, Yuraporn Sahasakul, Chaowanee Chupeerach, Uthaiwan Suttisansanee, and Piya Temviriyanukul. 2023. "Wolffia globosa-Based Nutritious Snack Formulation with High Protein and Dietary Fiber Contents" Foods 12, no. 14: 2647. https://doi.org/10.3390/foods12142647
APA StyleOn-Nom, N., Promdang, P., Inthachat, W., Kanoongon, P., Sahasakul, Y., Chupeerach, C., Suttisansanee, U., & Temviriyanukul, P. (2023). Wolffia globosa-Based Nutritious Snack Formulation with High Protein and Dietary Fiber Contents. Foods, 12(14), 2647. https://doi.org/10.3390/foods12142647







