Bioactive Compounds and Sensory Quality in Chips of Native Potato Clones (Solanum tuberosum spp. andigena) Grown in the High Andean Region of PERU
Abstract
1. Introduction
2. Materials and Methods
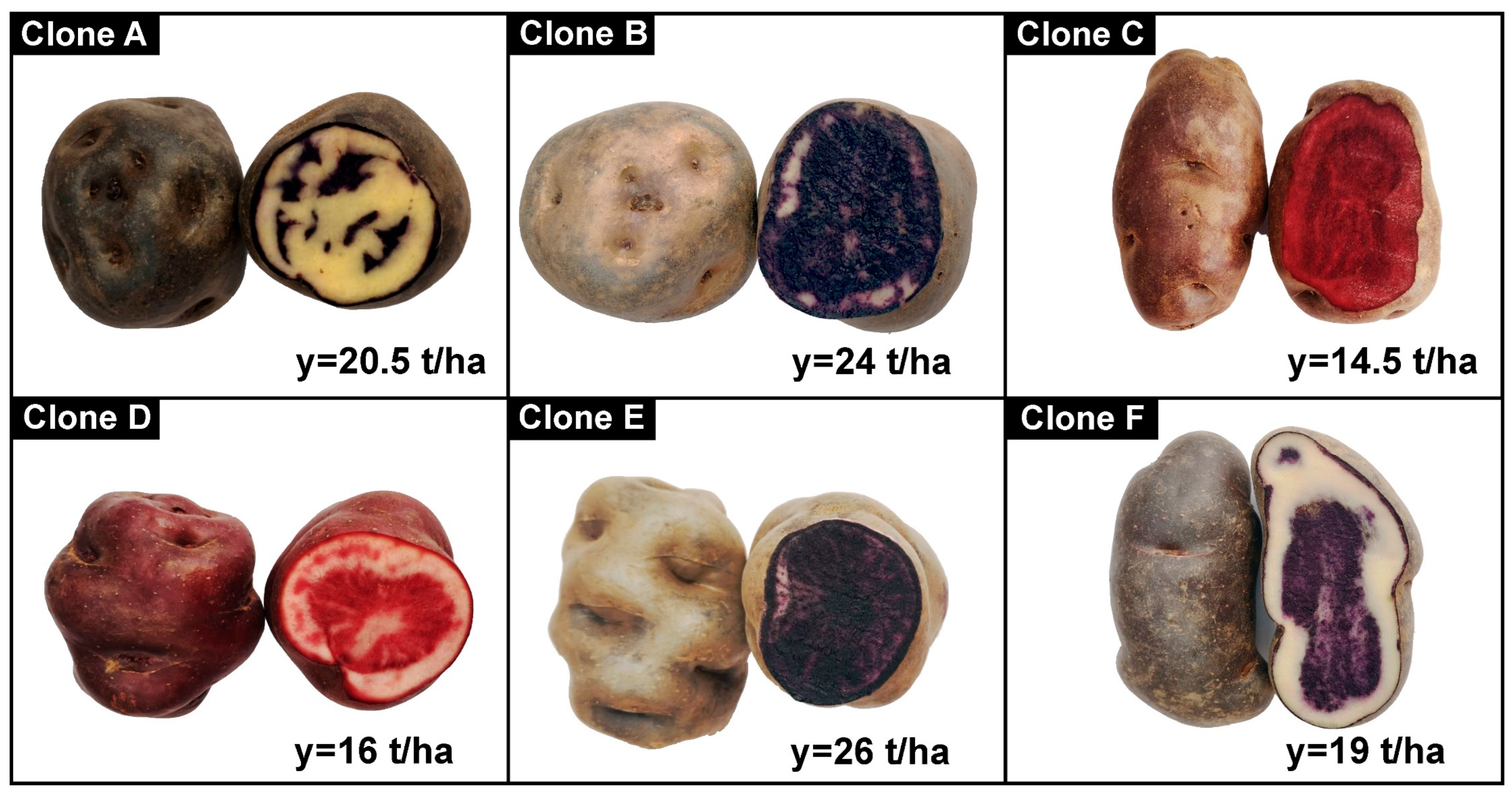
2.1. Materials
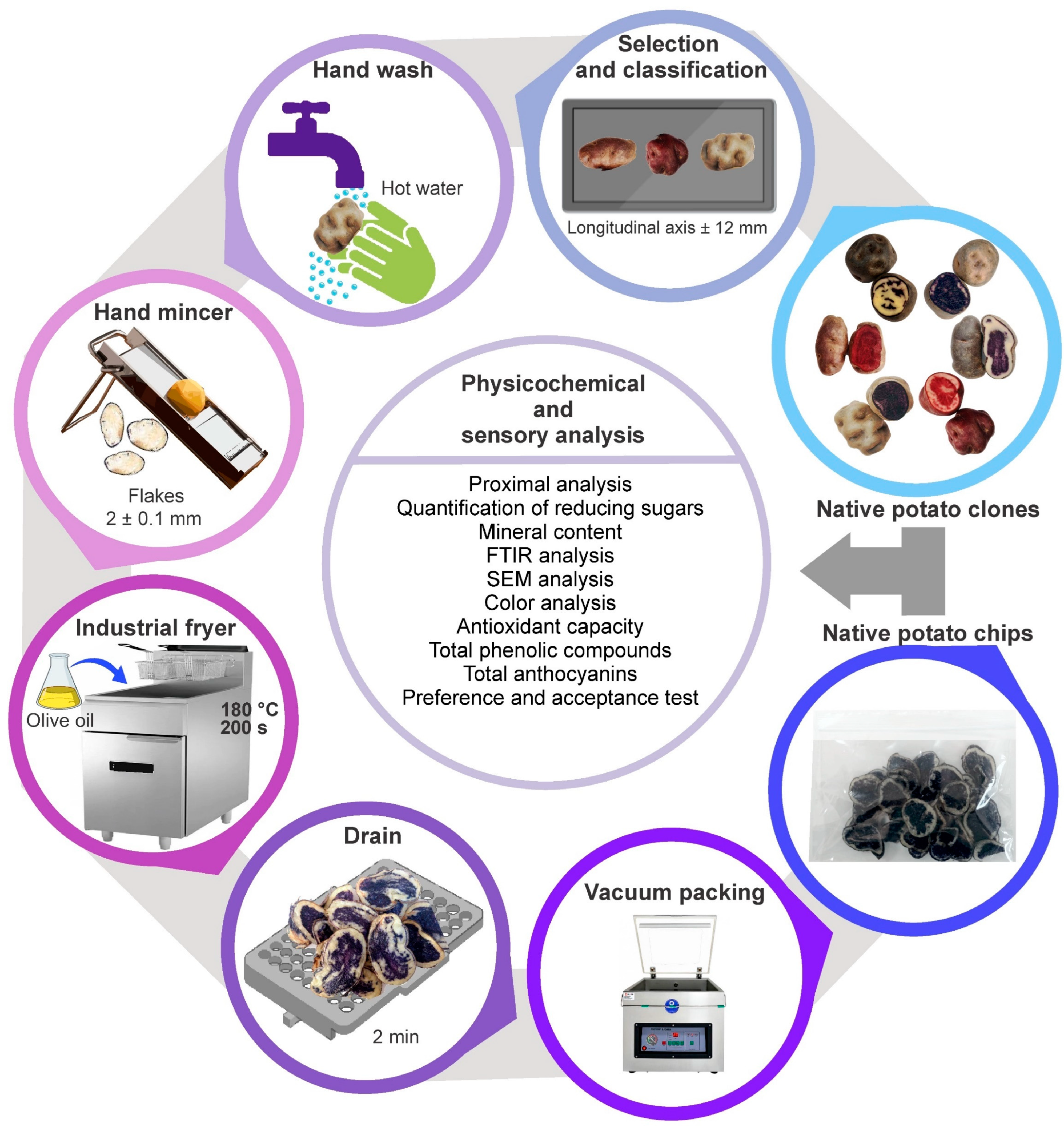
2.2. Obtaining Chips from Native Potato Clones
2.3. Proximate Analysis
2.4. Reducing Sugars
2.5. Mineral Content
2.6. Color Analysis
2.7. Analysis by Fourier Transform Infrared Spectroscopy (FTIR)
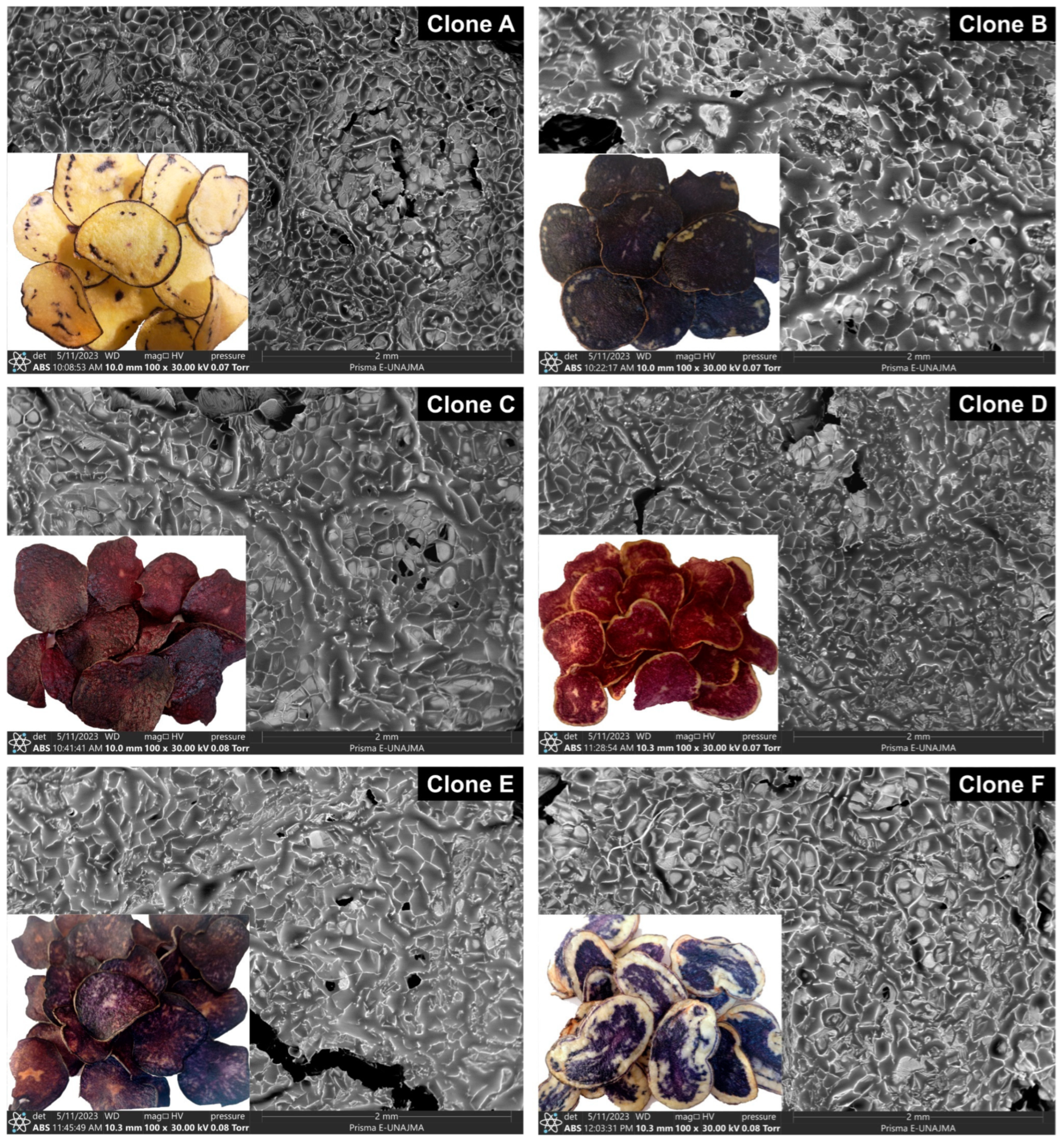
2.8. Analysis by Scanning Electron Microscopy (SEM)
2.9. Antioxidant Capacity
2.10. Total Phenolic Compounds
2.11. Total Anthocyanins
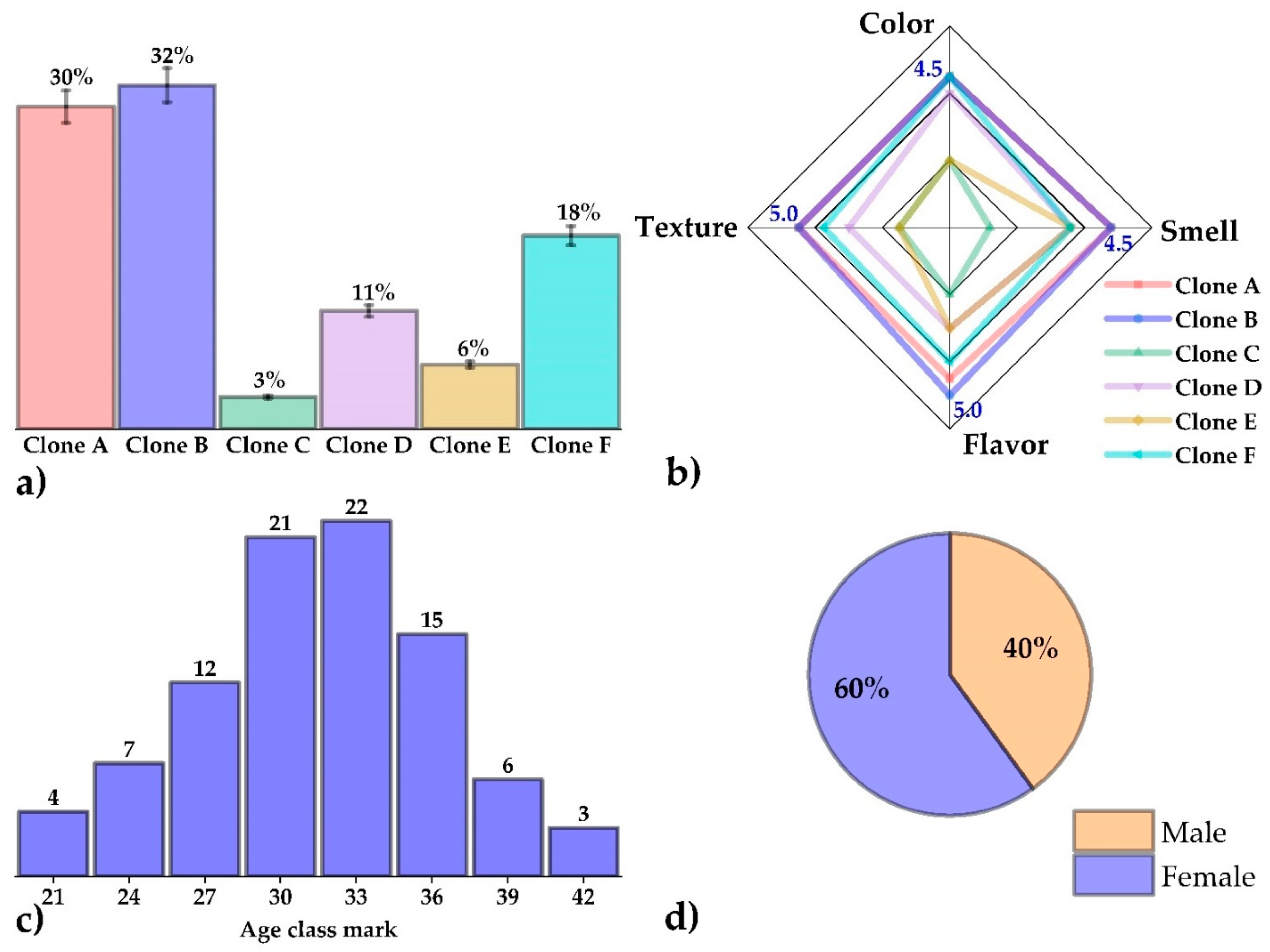
2.12. Preference and Acceptance Test
2.13. Statistical Analysis
3. Results and Discussion
3.1. Proximate Analysis
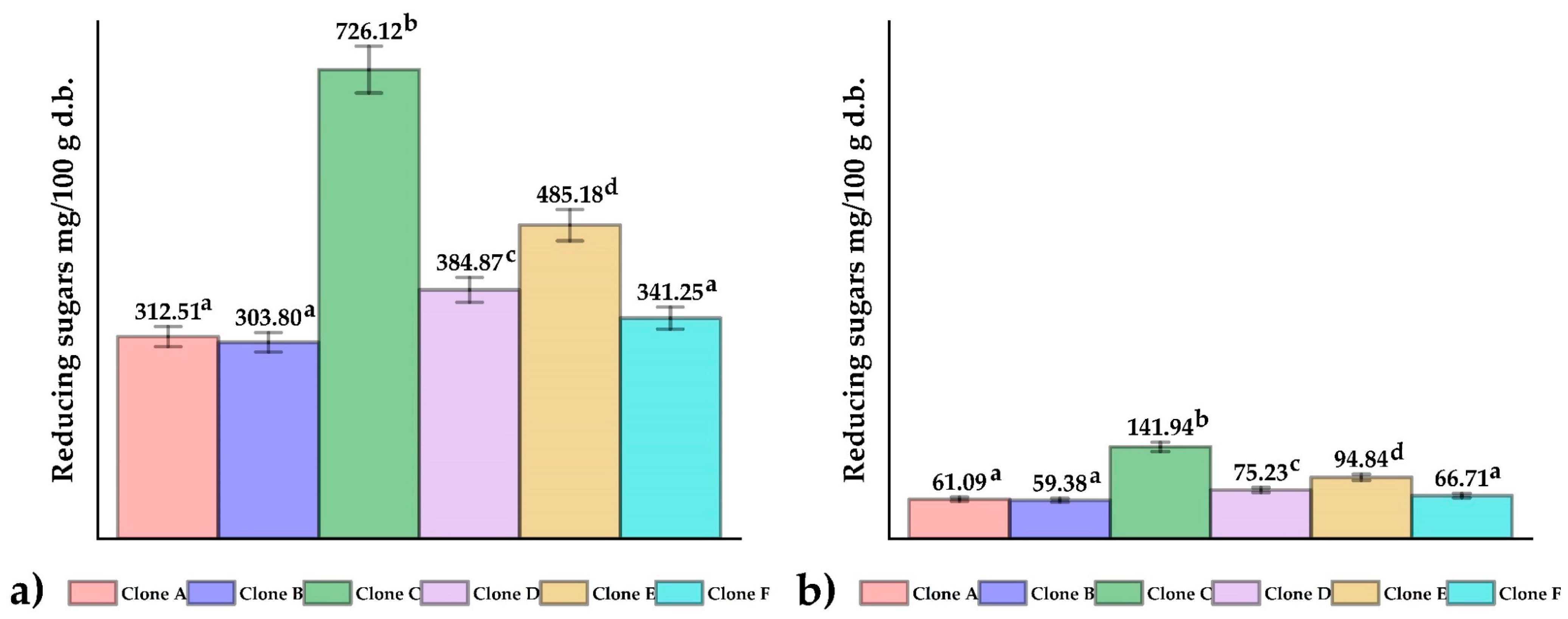
3.2. Reducing Sugars
3.3. Mineral Content
3.4. Color Analysis
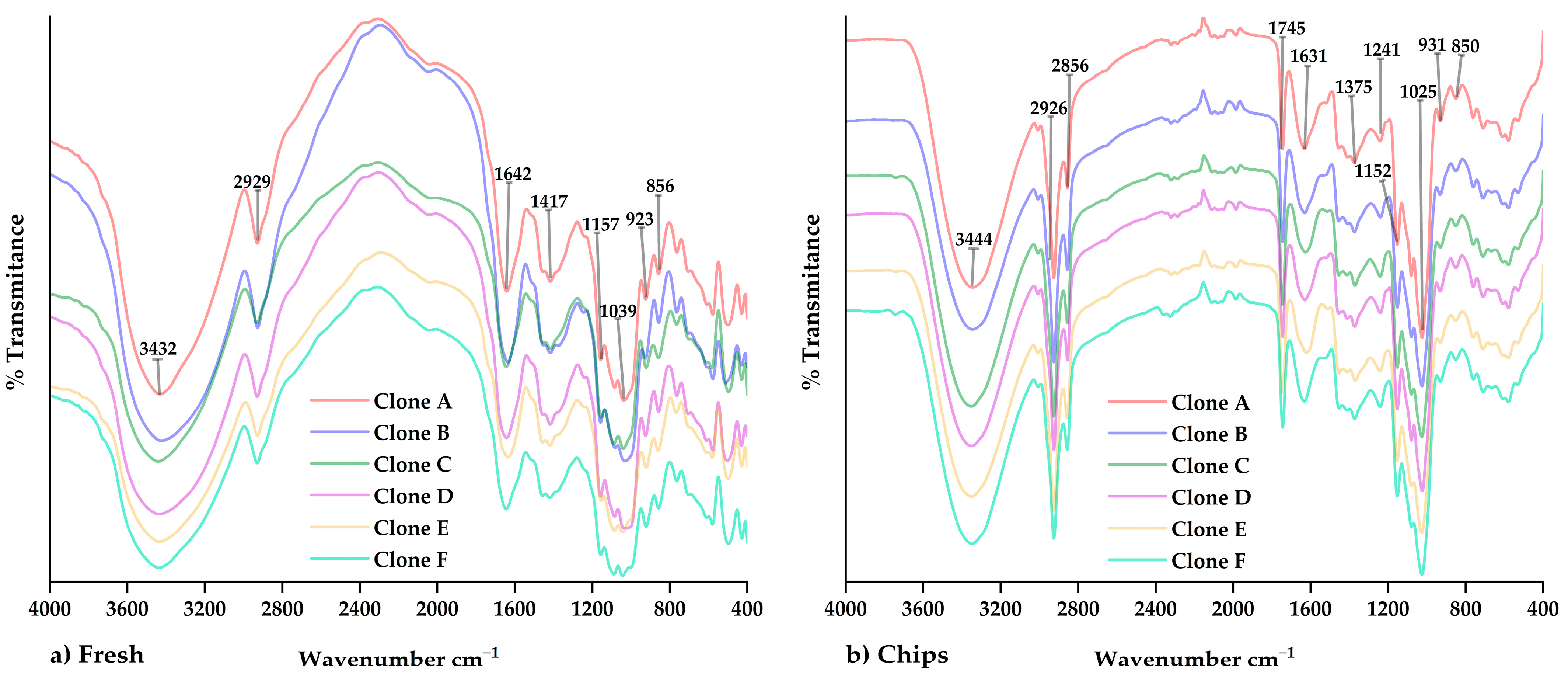
3.5. FTIR Analysis
3.6. SEM Analysis
3.7. Antioxidant Capacity, Phenolic Compounds, and Anthocyanins
3.8. Preference and Acceptance Test
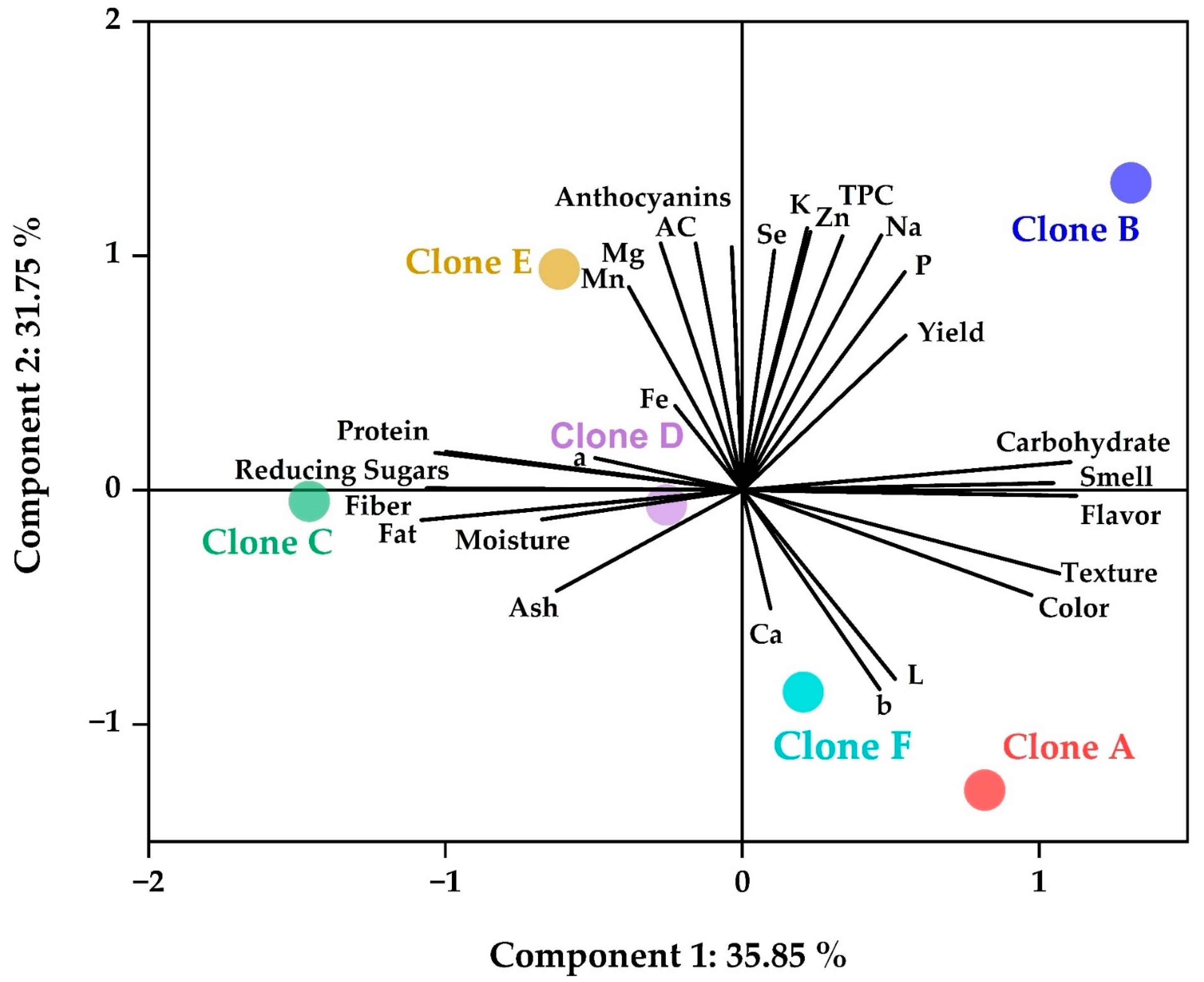
3.9. Overview of Results on Native Potato Clone Chips
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tobin, D.; Bates, R.; Brennan, M.; Gill, T. Peru potato potential: Biodiversity conservation and value chain development. Renew. Agric. Food Syst. 2018, 33, 19–32. [Google Scholar] [CrossRef]
- Ah-Hen, K.; Fuenzalida, C.; Hess, S.; Contreras, A.; Vega-Gálvez, A.; Lemus-Mondaca, R. Antioxidant Capacity and Total Phenolic Compounds of Twelve Selected Potato Landrace Clones Grown in Southern Chile. Chil. J. Agric. Res. 2012, 72, 3–9. [Google Scholar] [CrossRef]
- Ticsihua-Huamán, J.; Arteaga-Llacza, P.; Miranda-Jara, A.; Quispe-Barrantes, P.; Miranda-Chávez, H.; Quispe-Solano, M.Á.; Chuquilín-Goicochea, R. Changes in Sugars, Dry Matter, and Characteristics Sensory of Chip of Native Potato in the Chopcca Region. In Proceedings of the 7th Brazilian Technology Symposium (BTSym’21): Emerging Trends in Human Smart and Sustainable Future of Cities (Volume 1); Springer International Publishing: Cham, Switzerland, 2022; pp. 554–559. [Google Scholar]
- García-Torres, S.M.; Chire-Fajardo, G.C.; Repo-Carrasco, R.; Ureña-Peralta, M.O. Efecto de la fritura sobre los componentes bioactivos de la papa nativa (Solanum tuberosum sp.) Puka Ambrosio. Rev. Chil. De Nutr. 2022, 49, 7–16. [Google Scholar] [CrossRef]
- Natividad Bardales, Á.D.; Muñoz Garay, S.G.; Villanueva Tiburcio, J.E.; Rojas Portal, R.M.; Chamorro Gómez, R.E.; Cueto Rosales, C.R.; Bravo Romaina, J.M.; Beraun Bedoya, J.R.; Mendoza Aguilar, A. Caracterización fisicoquímica de cuatro variedades de papas nativas (Solanum tuberosum) con aptitud para fritura, cultivadas en dos zonas en Huánuco. Rev. De La Soc. Química Del Perú 2022, 88, 237–250. [Google Scholar] [CrossRef]
- Cerón-Lasso, M.; Alzate-Arbeláez, A.F.; Rojano, B.A.; Ñuztez-Lopez, C.E. Composición Fisicoquímica y Propiedades Antioxidantes de Genotipos Nativos de Papa Criolla (Solanum tuberosum Grupo Phureja). Inf. Tecnol. 2018, 29, 205–216. [Google Scholar] [CrossRef]
- Villacrés, E.; Quilca, N.; Reinoso, I.A.; Monteros, C.; Muñoz, R. Valorización Nutricional Y Funcional de Las Papas Nativas (Solanum Andígena ssp.); INIAP/CIP: Guaranda, Ecuador, 2011; pp. 33–35. [Google Scholar]
- Tirado-Lara, R.; Tirado-Malaver, R.; Mayta-Huatuco, E.; Amoros-Briones, W. Identificación de clones de papa con pulpa pigmentada de alto rendimiento comercial y mejor calidad de fritura: Estabilidad y análisis multivariado de la interacción genotipo-ambiente. Sci. Agropecu. 2020, 11, 323–334. [Google Scholar] [CrossRef]
- Molina, C.A.; Dudenhoefer, D.; Polar, V.; Scurrah, M.; Ccanto, R.C.; Heider, B. Gender Roles and Native Potato Diversity Management in Highland Communities of Peru. Sustainability 2022, 14, 3455. [Google Scholar] [CrossRef]
- Martínez, P.; Peña, F.; Bello-Pérez, L.A.; Núñez-Santiago, C.; Yee-Madeira, H.; Velezmoro, C. Physicochemical, functional and morphological characterization of starches isolated from three native potatoes of the Andean region. Food Chem. X 2019, 2, 100030. [Google Scholar] [CrossRef]
- Alva, C.; Ramírez, C.; Siche, R. Visión computacional en la evaluación del efecto de la temperatura y tiempo de fritura sobre el color de hojuelas de papa (Solanum tuberosum) nativa. Agroind. Sci. 2015, 5, 153–160. [Google Scholar] [CrossRef]
- Gomide, A.I.; Monteiro, R.L.; Carciofi, B.A.; Laurindo, J.B. The Effect of Pretreatments on the Physical Properties and Starch Structure of Potato Chips Dried by Microwaves under Vacuum. Foods 2022, 11, 2259. [Google Scholar] [CrossRef]
- Kita, A.; Bąkowska-Barczak, A.; Hamouz, K.; Kułakowska, K.; Lisińska, G. The effect of frying on anthocyanin stability and antioxidant activity of crisps from red- and purple-fleshed potatoes (Solanum tuberosum L.). J. Food Compos. Anal. 2013, 32, 169–175. [Google Scholar] [CrossRef]
- Kita, A.; Kołodziejczyk, M.; Michalska-Ciechanowska, A.; Brzezowska, J.; Wicha-Komsta, K.; Turski, W. The Effect of Thermal Treatment on Selected Properties and Content of Biologically Active Compounds in Potato Crisps. Appl. Sci. 2022, 12, 555. [Google Scholar] [CrossRef]
- Zhang, J.; Fan, L. Effects of preliminary treatment by ultrasonic and convective air drying on the properties and oil absorption of potato chips. Ultrason. Sonochem. 2021, 74, 105548. [Google Scholar] [CrossRef] [PubMed]
- Gomide, A.I.; Monteiro, R.L.; Laurindo, J.B. Impact of the power density on the physical properties, starch structure, and acceptability of oil-free potato chips dehydrated by microwave vacuum drying. LWT 2022, 155, 112917. [Google Scholar] [CrossRef]
- Elmore, J.S.; Briddon, A.; Dodson, A.T.; Muttucumaru, N.; Halford, N.G.; Mottram, D.S. Acrylamide in potato crisps prepared from 20 UK-grown varieties: Effects of variety and tuber storage time. Food Chem. 2015, 182, 1–8. [Google Scholar] [CrossRef]
- Ligarda-Samanez, C.A.; Choque-Quispe, D.; Allende-Allende, L.F.; Ramos Pacheco, B.S.; Peralta-Guevara, D.E. Calidad sensorial y proximal en conservas de mondongo de res (Bos taurus) en salsa de ají amarillo (Capsicum baccatum). Cienc. Y Tecnol. Agropecu. 2023, 24, 13. [Google Scholar] [CrossRef]
- Choque-Quispe, D.; Ligarda-Samanez, C.A.; Huamán-Rosales, E.R.; Aguirre Landa, J.P.; Agreda Cerna, H.W.; Zamalloa-Puma, M.M.; Álvarez-López, G.J.; Barboza-Palomino, G.I.; Alzamora-Flores, H.; Gamarra-Villanueva, W. Bioactive Compounds and Sensory Analysis of Freeze-Dried Prickly Pear Fruits from an Inter-Andean Valley in Peru. Molecules 2023, 28, 3862. [Google Scholar] [CrossRef]
- Suárez, S.; Ale, N.; Trabucco, J.; Sanabria, O. Polifenoles, micronutrientes minerales y potencial antioxidante de papas nativas. Rev. De La Soc. Química Del Perú 2014, 80, 108–114. [Google Scholar]
- Ignacio-Cárdenas, S.; Gonzales-Pariona, F.J.; Jacobo-Salinas, S.S. Caracterización morfológica de papas nativas (Solanum tuberosum L. Grupo Andigenum) de Huánuco. Rev. Investig. Agrar. 2022, 4, 13–25. [Google Scholar] [CrossRef]
- Horwitz, W. Official Methods of Analysis of AOAC International. Volume I, Agricultural Chemicals, Contaminants, Drugs; AOAC International: Gaithersburg, MD, USA, 2010. [Google Scholar]
- El-Shishtawy, R.M.; Al Angari, Y.M.; Alotaibi, M.M.; Almulaiky, Y.Q. Novel and Facile Colorimetric Detection of Reducing Sugars in Foods via In Situ Formed Gelatin-Capped Silver Nanoparticles. Polymers 2023, 15, 1086. [Google Scholar] [CrossRef]
- Lempiäinen, H.; Lappalainen, K.; Haverinen, J.; Tuuttila, T.; Hu, T.; Jaakkola, M.; Lassi, U. The Effect of Mechanocatalytic Pretreatment on the Structure and Depolymerization of Willow. Catalysts 2020, 10, 255. [Google Scholar] [CrossRef]
- Ligarda-Samanez, C.A.; Choque-Quispe, D.; Moscoso-Moscoso, E.; Huamán-Carrión, M.L.; Ramos-Pacheco, B.S.; Peralta-Guevara, D.E.; Cruz, G.D.; Martínez-Huamán, E.L.; Arévalo-Quijano, J.C.; Muñoz-Saenz, J.C.; et al. Obtaining and Characterizing Andean Multi-Floral Propolis Nanoencapsulates in Polymeric Matrices. Foods 2022, 11, 3153. [Google Scholar] [CrossRef]
- Ligarda-Samanez, C.A.; Moscoso-Moscoso, E.; Choque-Quispe, D.; Palomino-Rincón, H.; Martínez-Huamán, E.L.; Huamán-Carrión, M.L.; Peralta-Guevara, D.E.; Aroni-Huamán, J.; Arévalo-Quijano, J.C.; Palomino-Rincón, W.; et al. Microencapsulation of Erythrocytes Extracted from Cavia porcellus Blood in Matrices of Tara Gum and Native Potato Starch. Foods 2022, 11, 2107. [Google Scholar] [CrossRef]
- Ligarda-Samanez, C.A.; Choque-Quispe, D.; Moscoso-Moscoso, E.; Huamán-Carrión, M.L.; Ramos-Pacheco, B.S.; De la Cruz, G.; Arévalo-Quijano, J.C.; Muñoz-Saenz, J.C.; Muñoz-Melgarejo, M.; Quispe-Quezada, U.R.; et al. Microencapsulation of Propolis and Honey Using Mixtures of Maltodextrin/Tara Gum and Modified Native Potato Starch/Tara Gum. Foods 2023, 12, 1873. [Google Scholar] [CrossRef]
- Ligarda-Samanez, C.A.; Choque-Quispe, D.; Palomino-Rincón, H.; Ramos-Pacheco, B.S.; Moscoso-Moscoso, E.; Huamán-Carrión, M.L.; Peralta-Guevara, D.E.; Obregón-Yupanqui, M.E.; Aroni-Huamán, J.; Bravo-Franco, E.Y.; et al. Modified Polymeric Biosorbents from Rumex acetosella for the Removal of Heavy Metals in Wastewater. Polymers 2022, 14, 2191. [Google Scholar] [CrossRef]
- Choque-Quispe, D.; Ligarda-Samanez, C.A.; Ramos-Pacheco, B.S.; Leguía-Damiano, S.; Calla-Florez, M.; Zamalloa-Puma, L.M.; Colque-Condeña, L. Phenolic compounds, antioxidant capacity, and protein content of three varieties of germinated quinoa (Chenopodium quinoa Willd). Ing. E Investig. 2021, 41, 7. [Google Scholar] [CrossRef]
- Cruz, G.; Cruz-Tirado, J.P.; Delgado, K.; Guzman, Y.; Castro, F.; Rojas, M.L.; Linares, G. Impact of pre-drying and frying time on physical properties and sensorial acceptability of fried potato chips. J. Food Sci. Technol. 2018, 55, 138–144. [Google Scholar] [CrossRef]
- Solarte-Montúfar, J.G.; Díaz-Murangal, A.E.; Osorio-Mora, O.; Mejía-España, D.F. Propiedades Reológicas y Funcionales del Almidón. Procedente de Tres Variedades de Papa Criolla. Inf. Tecnol. 2019, 30, 35–44. [Google Scholar] [CrossRef]
- Benavides-Guerrero, R.; Revelo-Cuarán, Y.A.; Arango-Bedoya, O.; Osorio-Mora, O. Extracción asistida con ultrasonido de compuestos fenólicos de dos variedades de papas (Solanum phureja) nativas andinas y evaluación de su actividad antioxidante. Inf. Tecnol. 2020, 31, 43–50. [Google Scholar] [CrossRef]
- Silveira, A.C.; Vilaró, F.; Kvapil, M.F.; Rodríguez, S.D.C.; Zaccari, F. Physical-chemical characterization and potential for frying of genetic potato (Solanum tuberosum) materials. Rev. Chapingo. Ser. Hortic. 2020, 26, 143–157. [Google Scholar] [CrossRef]
- Ouchon, P.B.; Pyle, D.L. Studying Oil Absorption in Restructured Potato Chips. J. Food Sci. 2004, 69, FEP115–FEP122. [Google Scholar] [CrossRef]
- Liyanage, D.W.K.; Yevtushenko, D.P.; Konschuh, M.; Bizimungu, B.; Lu, Z.-X. Processing strategies to decrease acrylamide formation, reducing sugars and free asparagine content in potato chips from three commercial cultivars. Food Control 2021, 119, 107452. [Google Scholar] [CrossRef]
- Halford, N.G.; Curtis, T.Y.; Muttucumaru, N.; Postles, J.; Elmore, J.S.; Mottram, D.S. The acrylamide problem: A plant and agronomic science issue. J. Exp. Bot. 2012, 63, 2841–2851. [Google Scholar] [CrossRef]
- Muttucumaru, N.; Powers, S.J.; Elmore, J.S.; Dodson, A.; Briddon, A.; Mottram, D.S.; Halford, N.G. Acrylamide-forming potential of potatoes grown at different locations, and the ratio of free asparagine to reducing sugars at which free asparagine becomes a limiting factor for acrylamide formation. Food Chem. 2017, 220, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Rosen, C.; Sun, N.; Olsen, N.; Thornton, M.; Pavek, M.; Knowles, L.; Richard Knowles, N. Impact of Agronomic and Storage Practices on Acrylamide in Processed Potatoes. Am. J. Potato Res. 2018, 95, 319–327. [Google Scholar] [CrossRef]
- Gupta, S.K.; Crants, J. Identification and impact of stable prognostic biochemical markers for cold-induced sweetening resistance on selection efficiency in potato (Solanum tuberosum L.) breeding programs. PLoS ONE 2019, 14, e0225411. [Google Scholar] [CrossRef] [PubMed]
- Landeta, M.C.d.; Pighín, A.F.; Gómez, G.A.J.D. Contenido de minerales en papas crudas, procesadas industrialmente y cocidas por diferentes métodos. Diaeta 2017, 35, 17–24. Available online: http://www.scielo.org.ar/scielo.php?script=sci_arttext&pid=S1852-73372017000200003 (accessed on 2 May 2023).
- de Haan, S.; Burgos, G.; Arcos, J.; Ccanto, R.; Scurrah, M.; Salas, E.; Bonierbale, M. Traditional Processing of Black and White Chuño in the Peruvian Andes: Regional Variants and Effect on the Mineral Content of Native Potato Cultivars. Econ. Bot. 2010, 64, 217–234. [Google Scholar] [CrossRef]
- Diamante, L.M.; Savage, G.P.; Vanhanen, L. Optimisation of vacuum frying of gold kiwifruit slices: Application of response surface methodology. Int. J. Food Sci. Technol. 2012, 47, 518–524. [Google Scholar] [CrossRef]
- Safar, M.; Bertrand, D.; Robert, P.; Devaux, M.F.; Genot, C. Characterization of edible oils, butters and margarines by Fourier transform infrared spectroscopy with attenuated total reflectance. J. Am. Oil Chem. Soc. 1994, 71, 371–377. [Google Scholar] [CrossRef]
- Guillen, M.D.; Cabo, N. Infrared spectroscopy in the study of edible oils and fats. J. Sci. Food Agric. 1997, 75, 1–11. [Google Scholar] [CrossRef]
- Wang, S.; Wang, J.; Yu, J.; Wang, S. Effect of fatty acids on functional properties of normal wheat and waxy wheat starches: A structural basis. Food Chem. 2016, 190, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Kalogianni, E.P.; Papastergiadis, E. Crust pore characteristics and their development during frying of French-fries. J. Food Eng. 2014, 120, 175–182. [Google Scholar] [CrossRef]
- Su, Y.; Zhang, M.; Zhang, W.; Liu, C.; Adhikari, B. Ultrasonic microwave-assisted vacuum frying technique as a novel frying method for potato chips at low frying temperature. Food Bioprod. Process. 2018, 108, 95–104. [Google Scholar] [CrossRef]
- Zhang, J.; Yu, P.; Fan, L.; Sun, Y. Effects of ultrasound treatment on the starch properties and oil absorption of potato chips. Ultrason. Sonochem. 2021, 70, 105347. [Google Scholar] [CrossRef]
- Yang, D.; Wu, G.; Li, P.; Zhang, H.; Qi, X. Comparative analysis of the oil absorption behavior and microstructural changes of fresh and pre-frozen potato strips during frying via MRl, SEM, and XRD. Food Res. Int. 2019, 122, 295–302. [Google Scholar] [CrossRef]
- Anwar, N.Z.R.; Ghani, A.A. Effect of different processing methods on the physicochemical properties and sensory evaluations of sweet potatoes chips. J. Agrobiotechnol. 2019, 10, 51–63. [Google Scholar]
| Property | Clone A | Clone B | Clone C | Clone D | Clone E | Clone F | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ±SD | ±SD | ±SD | ±SD | ±SD | ±SD | |||||||
| Fresh | ||||||||||||
| Moisture (%) | 68.00 a | ±0.26 | 65.78 f | ±0.25 | 72.10 c | ±0.20 | 73.70 d | ±0.18 | 76.60 e | ±0.16 | 69.80 b | ±0.19 |
| Protein (%) | 1.32 a | ±0.21 | 1.30 a | ±0.31 | 1.43 a | ±0.17 | 1.56 a | ±0.19 | 1.24 a | ±0.23 | 1.45 a | ±0.21 |
| Fat (%) | 0.36 a | ±0.32 | 0.41 a | ±0.14 | 0.44 a | ±0.19 | 0.39 a | ±0.22 | 0.28 a | ±0.18 | 0.44 a | ±0.30 |
| Ash (%) | 0.97 ab | ±0.19 | 0.84 ab | ±0.20 | 0.75 a | ±0.13 | 0.87 ab | ±0.17 | 0.79 a | ±0.16 | 1.10 b | ±0.18 |
| Fiber (%) | 0.66 a | ±0.24 | 0.58 a | ±0.13 | 0.71 a | ±0.21 | 0.62 a | ±0.18 | 0.74 a | ±0.21 | 0.75 a | ±0.22 |
| Carbohydrates (%) | 28.70 a | ±0.22 | 31.10 b | ±0.15 | 24.60 c | ±0.18 | 22.90 d | ±0.21 | 20.30 e | ±0.22 | 26.50 f | ±0.19 |
| Chips | ||||||||||||
| Moisture (%) | 1.28 a | ±0.35 | 0.96 a | ±0.14 | 1.30 a | ±0.25 | 1.10 a | ±0.25 | 1.35 a | ±0.19 | 0.98 a | ±0.23 |
| Protein (%) | 2.38 a | ±0.28 | 2.09 a | ±0.19 | 3.36 b | ±0.18 | 3.45 b | ±0.18 | 3.36 b | ±0.21 | 2.42 a | ±0.15 |
| Fat (%) | 31.36 a | ±0.21 | 29.38 b | ±0.28 | 50.81 c | ±0.23 | 39.06 d | ±0.26 | 40.00 e | ±0.23 | 40.63 f | ±0.35 |
| Ash (%) | 1.74 ab | ±0.32 | 1.36 b | ±0.23 | 1.82 a | ±0.30 | 1.92 a | ±0.15 | 2.14 a | ±0.26 | 2.17 a | ±0.13 |
| Fiber (%) | 1.21 ac | ±0.19 | 0.93 a | ±0.17 | 1.66 b | ±0.17 | 1.41 cb | ±0.31 | 1.71 b | ±0.14 | 1.28 ac | ±0.25 |
| Carbohydrates (%) | 62.03 a | ±0.25 | 65.28 b | ±0.26 | 41.05 c | ±0.29 | 53.06 a | ±0.16 | 51.44 cd | ±0.13 | 52.52 e | ±0.18 |
| Mineral (mg/100 g db) | Wavelength (nm) | Clone A | Clone B | Clone C | Clone D | Clone E | Clone F | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ±SD | ±SD | ±SD | ±SD | ±SD | ±SD | ||||||||
| Fresh | |||||||||||||
| K | 767 | 635 a | ±3.06 | 1079 b | ±2.66 | 605 a | ±2.25 | 864 c | ±3.03 | 1069 b | ±2.35 | 455 a | ±2.07 |
| Mg | 280 | 32.06 a | ±2.68 | 53.52 b | ±2.44 | 47.02 b | ±2.24 | 35.36 a | ±3.11 | 71.46 c | ±5.78 | 27.99 a | ±1.22 |
| Ca | 185 | 13.70 a | ±0.20 | 5.53 b | ±0.06 | 7.83 c | ±0.12 | 8.20 d | ±0.10 | 6.30 e | ±0.01 | 2.53 f | ±0.06 |
| P | 252 | 0.86 a | ±0.03 | 3.84 b | ±0.02 | 0.09 c | ±0.03 | 2.87 d | ±0.06 | 2.30 e | ±0.06 | 0.54 f | ±0.03 |
| Mn | 258 | 0.28 a | ±0.01 | 0.35 b | ±0.01 | 0.35 b | ±0.01 | 0.39 c | ±0.02 | 0.47 d | ±0.01 | 0.15 e | ±0.01 |
| Na | 589 | ND | 2.68 a | ±0.01 | 0.54 b | ±0.01 | 1.02 c | ±0.03 | 1.02 c | ±0.01 | 0.43 d | ±0.03 | |
| Se | 196 | ND | 0.17 a | ±0.12 | ND | ND | 0.27 a | ±0.06 | ND | ||||
| Fe | 240 | 0.03 a | ±0.01 | 0.07 b | ±0.01 | 0.06 c | ±0.01 | 0.17 d | ±0.02 | 0.07 e | ±0.02 | 0.04 f | ±0.01 |
| Zn | 214 | ND | 0.22 a | ±0.01 | ND | 0.05 b | ±0.01 | 0.26 a | ±0.03 | ND | |||
| Chips | |||||||||||||
| K | 767 | 1440 a | ±2.28 | 1961 ab | ±4.23 | 1960 ab | ±1.95 | 2540 b | ±1.98 | 2384 b | ±2.49 | 1485 a | ±2.76 |
| Mg | 280 | 59.02 a | ±0.01 | 100 b | ±0.26 | 103 b | ±0.19 | 82.10 ab | ±0.19 | 74.01 ab | ±0.06 | 83.10 ab | ±0.19 |
| Ca | 185 | 50.15 a | ±0.15 | 107 b | ±0.58 | 65.02 a | ±0.17 | 50.11 a | ±0.20 | 58.31 a | ±0.17 | 83.57 a | ±0.46 |
| P | 252 | 2.01 a | ±0.01 | 4.21 b | ±0.01 | 1.15 a | ±0.01 | 3.05 b | ±0.01 | 3.14 b | ±0.01 | 1.03 a | ±0.01 |
| Mn | 258 | 4.04 b | ±0.01 | 1.21 a | ±0.01 | 0.81 a | ±0.01 | 0.61 a | ±0.01 | 0.62 a | ±0.01 | 0.81 a | ±0.01 |
| Na | 589 | 46.18 a | ±0.19 | 139 b | ±0.82 | 51.11 a | ±0.04 | 41.12 a | ±0.02 | 45.05 a | ±0.04 | 41.10 a | ±0.15 |
| Se | 196 | ND | 0.31 a | ±0.01 | ND | ND | 0.33 a | ±0.01 | ND | ||||
| Fe | 240 | 2.12 a | ±0.01 | 3.08 a | ±0.02 | 3.06 a | ±0.02 | 2.10 a | ±0.01 | 2.16 a | ±0.01 | 4.11 a | ±0.03 |
| Zn | 214 | 5.03 a | ±0.03 | 8.11 b | ±0.05 | 3.08 a | ±0.01 | 5.12 a | ±0.04 | 7.12 b | ±0.05 | 4.02 a | ±0.03 |
| Parameters | Clone A | Clone B | Clone C | Clone D | Clone E | Clone F | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ±SD | ±SD | ±SD | ±SD | ±SD | ±SD | |||||||
| Fresh | ||||||||||||
| L* | 67.48 a | ±0.06 | 21.91 b | ±0.15 | 26.63 c | ±0.25 | 36.65 d | ±0.46 | 21.13 e | ±0.31 | 24.24 f | ±0.58 |
| a* | 1.54 a | ±0.06 | 0.62 b | ±0.10 | 13.11 c | ±0.24 | 14.31 d | ±0.26 | 2.37 e | ±0.19 | 2.98 f | ±0.58 |
| b* | 36.16 a | ±0.16 | −4.35 b | ±0.03 | 3.62 c | ±0.15 | 4.94 d | ±0.07 | −3.16 e | ±0.12 | −3.92 f | ±0.28 |
| Chips | ||||||||||||
| L* | 57.58 a | ±0.21 | 24.43 b | ±0.66 | 19.53 c | ±0.07 | 31.06 d | ±0.27 | 20.70 e | ±0.12 | 40.09 f | ±0.11 |
| a* | 0.25 a | ±0.21 | 0.82 b | ±0.13 | 4.95 c | ±0.10 | 15.73 d | ±0.13 | 4.79 c | ±0.10 | 1.00 b | ±0.08 |
| b* | 25.12 a | ±0.26 | 1.11 b | ±0.68 | 0.26 c | ±0.01 | 6.99 d | ±0.13 | 1.82 e | ±0.08 | 2.86 f | ±0.27 |
| ΔE*ab | 14.86 a | ±0.15 | 6.02 b | 0.80 | 11.33 c | 0.26 | 6.12 b | 0.16 | 5.53 b | 0.10 | 17.36 d | 0.38 |
| Clones | Clone A | Clone B | Clone C | Clone D | Clone E | Clone F | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ±SD | ±SD | ±SD | ±SD | ±SD | ±SD | |||||||
| Fresh | ||||||||||||
| AC µmol ET/g db | 867 a | ±2.28 | 929 b | ±2.83 | 902 b | ±1.78 | 879 c | ±2.48 | 887 d | ±2.51 | 857 a | ±8.27 |
| TPC mg GAE/g db | 20.37 a | ±1.91 | 27.38 b | ±2.56 | 23.19 a | ±2.23 | 22.00 a | ±1.74 | 22.82 a | ±2.14 | 21.44 a | ±0.08 |
| Antocyanins mg C3G/g db | 0.43 a | ±0.02 | 2.60 b | ±0.01 | 1.98 c | ±0.02 | 1.43 d | ±0.03 | 1.58 e | ±0.04 | 1.44 d | ±0.02 |
| Chips | ||||||||||||
| AC µmol ET/g db | 884 a | ±2.37 | 938 b | ±3.74 | 929 b | ±2.74 | 897 c | ±3.41 | 914 d | ±1.51 | 883 a | ±8.27 |
| TPC mg GAE/g db | 28.93 a | ±2.62 | 39.15 b | ±3.04 | 32.70 a | ±2.08 | 31.46 a | ±2.30 | 32.40 a | ±1.88 | 30.01 a | ±0.08 |
| Antocyanins mg C3G/g db | 0.20 a | ±0.01 | 1.17 b | ±0.02 | 0.87 c | ±0.03 | 0.67 d | ±0.02 | 0.71 e | ±0.01 | 0.65 d | ±0.02 |
| Attribute | Clone A | Clone B | Clone C | Clone D | Clone E | Clone F | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Descriptor | Descriptor | Descriptor | Descriptor | Descriptor | Descriptor | |||||||
| Color | I like it very much | 4.5 a | I like it very much | 4.5 a | I dislike it slightly | 2.0 b | I like it slightly | 4.0 c | I dislike it slightly | 2.0 b | I like it very much | 4.5 a |
| Smell | I like it very much | 4.5 a | I like it very much | 4.5 a | Neither like nor dislike | 3.0 b | I like it slightly | 4.0 c | I like it slightly | 4.0 c | I dislike it slightly | 4.0 c |
| Flavor | I like it very much | 4.5 a | I like it very much | 5.0 a | I dislike it slightly | 2.0 b | I like it slightly | 3.0 c | Neither like nor dislike | 3.0 c | I like it very much | 4.0 d |
| Texture | I like it very much | 5.0 a | I like it very much | 5.0 a | Neither like nor dislike | 3.0 b | I like it slightly | 4.0 c | Neither like nor dislike | 3.0 b | I like it very much | 4.5 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ligarda-Samanez, C.A.; Palomino-Rincón, H.; Choque-Quispe, D.; Moscoso-Moscoso, E.; Arévalo-Quijano, J.C.; Huamán-Carrión, M.L.; Quispe-Quezada, U.R.; Muñoz-Saenz, J.C.; Gutiérrez-Gómez, E.; Cabel-Moscoso, D.J.; et al. Bioactive Compounds and Sensory Quality in Chips of Native Potato Clones (Solanum tuberosum spp. andigena) Grown in the High Andean Region of PERU. Foods 2023, 12, 2511. https://doi.org/10.3390/foods12132511
Ligarda-Samanez CA, Palomino-Rincón H, Choque-Quispe D, Moscoso-Moscoso E, Arévalo-Quijano JC, Huamán-Carrión ML, Quispe-Quezada UR, Muñoz-Saenz JC, Gutiérrez-Gómez E, Cabel-Moscoso DJ, et al. Bioactive Compounds and Sensory Quality in Chips of Native Potato Clones (Solanum tuberosum spp. andigena) Grown in the High Andean Region of PERU. Foods. 2023; 12(13):2511. https://doi.org/10.3390/foods12132511
Chicago/Turabian StyleLigarda-Samanez, Carlos A., Henry Palomino-Rincón, David Choque-Quispe, Elibet Moscoso-Moscoso, José C. Arévalo-Quijano, Mary L. Huamán-Carrión, Uriel R. Quispe-Quezada, Jenny C. Muñoz-Saenz, Edgar Gutiérrez-Gómez, Domingo J. Cabel-Moscoso, and et al. 2023. "Bioactive Compounds and Sensory Quality in Chips of Native Potato Clones (Solanum tuberosum spp. andigena) Grown in the High Andean Region of PERU" Foods 12, no. 13: 2511. https://doi.org/10.3390/foods12132511
APA StyleLigarda-Samanez, C. A., Palomino-Rincón, H., Choque-Quispe, D., Moscoso-Moscoso, E., Arévalo-Quijano, J. C., Huamán-Carrión, M. L., Quispe-Quezada, U. R., Muñoz-Saenz, J. C., Gutiérrez-Gómez, E., Cabel-Moscoso, D. J., Sucari-León, R., Aroquipa-Durán, Y., & García-Espinoza, A. J. (2023). Bioactive Compounds and Sensory Quality in Chips of Native Potato Clones (Solanum tuberosum spp. andigena) Grown in the High Andean Region of PERU. Foods, 12(13), 2511. https://doi.org/10.3390/foods12132511















