Abstract
The micro- and nanoencapsulation of bioactive compounds has resulted in a large improvement in the food, nutraceutical, pharmaceutical, and agriculture industries. These technologies serve, on one side, to protect, among others, vitamins, minerals, essential fatty acids, polyphenols, flavours, antimicrobials, colorants, and antioxidants, and, on the other hand, to control the release and assure the delivery of the bioactive compounds, targeting them to specific cells, tissues, or organs in the human body by improving their absorption/penetration through the gastrointestinal tract. The food industry has been applying nanotechnology in several ways to improve food texture, flavour, taste, nutrient bioavailability, and shelf life using nanostructures. The use of micro- and nanocapsules in food is an actual trend used mainly in the cereal, bakery, dairy, and beverage industries, as well as packaging and coating. The elaboration of bio capsules with high-value compounds from agro-industrial by-products is sustainable for the natural ecosystem and economically interesting from a circular economy perspective. This critical review presents the principal methodologies for performing micro- and nanoencapsulation, classifies them (top-down and/or bottom-up), and discusses the differences and advantages among them; the principal types of encapsulation systems; the natural plant sources, including agro-industrial by-products, of bioactive compounds with interest for the food industry to be encapsulated; the bioavailability of encapsulates; and the main techniques used to analyse micro- and nanocapsules. Research work on the use of encapsulated bioactive compounds, such as lycopene, hydroxytyrosol, and resveratrol, from agro-industrial by-products must be further reinforced, and it plays an important role, as it presents a high potential for the use of their antioxidant and/or antimicrobial activities in food applications and, therefore, in the food industry. The incorporation of these bioactive compounds in food is a challenge and must be evaluated, not only for their nutritional aspect, but also for the chemical safety of the ingredients. The potential use of these products is an available economical alternative towards a circular economy and, as a consequence, sustainability.
1. Introduction
In recent years, awareness has led to healthy options when buying food, and consumers, in general, prefer foods that do not only satisfy their primary needs, but that also include health effect promoters besides the nutrients, and that can replace synthetic additives by natural bioactive compounds, which may prevent illnesses. However, natural bioactive compounds, although safe, sometimes present an unpleasant taste or odour and instability. The direct incorporation of these compounds in food is often difficult, so nanoencapsulation constitutes an alternative to solve this issue. The use of nano- or microcapsules in food is a trend used in the cereal, bakery, dairy, and beverage industries, as well as packaging and coating [1]. Encapsulated compounds in foods include vitamins, essential fatty acids, minerals, flavours, antimicrobial agents, colorants, antioxidants, and polyphenols [2]. The incorporation of nutraceuticals [3] in food presents an issue, as both the nutritional purpose and the chemical safety of ingredients must be considered and evaluated [1].
The nanoencapsulation of bioactive compounds has brought large improvements to the food, nutraceutical, pharmaceutical, and agriculture industries. This technology allows the protection of bioactive compounds applied to different matrices and the control of their release. The elaboration of bio capsules with natural bioactive compounds is a challenge nowadays. The use of bioactive compounds extracted from by-products is economically interesting in the scope of a circular economy approach, and is more sustainable for the natural ecosystem [4,5].
Nanoencapsulation serves at the same time to target cells, tissues, or organs and to control the release and delivery of bioactive molecules. It also serves to protect the bioactive compound from several reactions that can modify the molecule or alter its bioactivity. Factors such as temperature, acidity, hydrolysis, and oxidation due to light exposure are some of the hazards for bioactivity. The advantages of this technology include (i) allowing targeting all the bioactive beneficial characteristics of the compounds to specific targets, (ii) controlling the release of the bioactive components, (iii) allowing the concentration of the compound to be decreased, thus preserving it in hostile mediums and maintaining its natural characteristics, (iv) preventing interactions between the bioactive molecules and other food ingredients, and (v) presenting a high solubility and dispersibility [1].
Nanoencapsulation deals with several types of materials on the atomic, molecular, and supramolecular scale, normally from 1 to 100 nm; nevertheless, some nanocapsules may present sizes up to 600 nm [6]. Nano refers to the size of 10−9 m, and the British standardization body defines nanotechnology as the conceptualisation, elaboration, characterization, and application of the products (nanocapsules) and systems [7].
In the last years, the food industry has applied nanotechnology in diverse forms to improve food texture, flavour, taste, consistency, nutrient bioavailability, and shelf life using nanostructures in stable emulsions. In addition, nanotechnology also presents well-known benefits in the fields of nutraceuticals, food microbiology, and nutrient delivery, by improving nutrient absorption in the gastrointestinal tract [8]. In food packaging, nanoencapsulation has improved materials by enhancing their mechanical strength and by producing antimicrobial films and nanosensors to detect pathogens or specific compounds, e.g., indicators of disease conditions, which are important tools to enlighten consumers about food safety issues [9].
Actually, not only value-added components of foods are in focus. It is also possible to remove undesirable components from modified functional foods, such as sugars, salt, and fats to obtain more healthy products.
It is important to distinguish microencapsulation from nanoencapsulation, with the principal difference being the size, but with the two also having different functions. The dimensions that distinguish microparticles from nanoparticles is still under debate. In microencapsulation, the scale varies between 1 and 8000 µm (Table 1). Other authors, such as Rossi et al. [5], have stated that nanocapsules and microcapsules are 10–1000 nm and 3–800 µm in diameter, respectively. Shishir et al. [10] have claimed that microcapsules stay in the range of 1 to 1000 µm, sub-microcapsules range from several hundred nanometres to less than 1 µm, and nanocapsules range from one to several hundred nanometres. Some authors have proposed a diameter inferior or equal to 100 nm for nanoparticles [11] and the range for colloid associations as 5 to 100 nm to be considered a nanoparticle [12]. The functionalities of microencapsulation are (i) the protection of bioactive compounds; (ii) the control of the release profile; (iii) the masking of undesirable flavours; (iv) the improvement of flow properties; (v) an increase in the shelf life; and (vi) product enrichment with specific nutrients. Very similarly, the functions of nanoencapsulation are (i) to reduce the practical size and create a restricted distribution of particles; (ii) to increase the surface area; (iii) to improve the delivery of bioactive molecules; (iv) to increase the bioavailability of encapsulated compounds; (v) to increase the physical stability and extend the shelf life; (vi) to increase precision targeting and to promote passage through fenestrated cells; (vii) to strengthen the barrier between bioactive and other compounds; and (viii) to improve intracellular absorption [10].

Table 1.
Micro- and nanoencapsulation methodologies.
This review aims to perform a critical analysis of (i) the different methodologies to carry out micro- and nanoencapsulation for food applications, classifying these methodologies according to top-down (TD) and/or bottom-up (BU); (ii) the principal types of encapsulation systems; (iii) the natural plant sources of bioactive compounds of interest for the food industry to be encapsulated, enhancing the agro-industrial by-products; (iv) the bioavailability of the micro- and nanocapsules, presenting the various mechanisms for the release of the bioactive compound; and (v) the main techniques used to analyse these micro- and nanocapsules, and their respective advantages.
2. Micro- and Nanoencapsulation Methodologies
Nanoencapsulation is a process by which an active agent is coated by carrier material in order to form capsules on the nanometric scale. The coated materials are also designated as the core, fill, or internal phase, while the carrier materials are known as the wall material, membrane, capsule, shell, matrix, or external phase [10].
In nanoencapsulation, two aspects are normally relevant; one is the selection of the encapsulating material and the other is the specific encapsulation technique. For food applications, the encapsulating materials must be “generally recognized as safe” (GRAS). Nevertheless, it is of relevance as well to consider the encapsulated compound concentration, the stability of the capsule, its function in the food application, the target aimed for, and the efficiency of delivery [10]. The nanocapsule can be formed by biopolymers, such as proteins, carbohydrates, fats, and other organic and inorganic materials [1]. Emulsification, coacervation, inclusion complexation, emulsification solvent evaporation, extraction, nano-precipitation, electro-spraying (spray drying), and electro-spinning are the most common encapsulation techniques used for bioactive compounds [8]. The chosen technique varies according to the type of bioactive compound that is encapsulated, for example, whether it is volatile or not, the size of the nano- or microcapsule, the type of shell material, and other parameters. Nanoencapsulation is an area in constant innovation, and many recent studies have proposed new formulations or new encapsulation systems for the preservation of bioactive compounds with improved functionalities [10].
There is no standard technique to perform nanoencapsulation, and to choose a method, the type and characteristics of the active compound, which will be the core of the particle, must be taken into account; regarding the carrier material, its molecular weight, polarity, solubility, structure, and encapsulation efficiency need to be considered, and the particle size is also an important factor [5,10]. The principal techniques used for micro- and nanoencapsulation are listed in Table 1. Sometimes, two or more methods can be combined, such as emulsions used with spray drying or freeze drying.
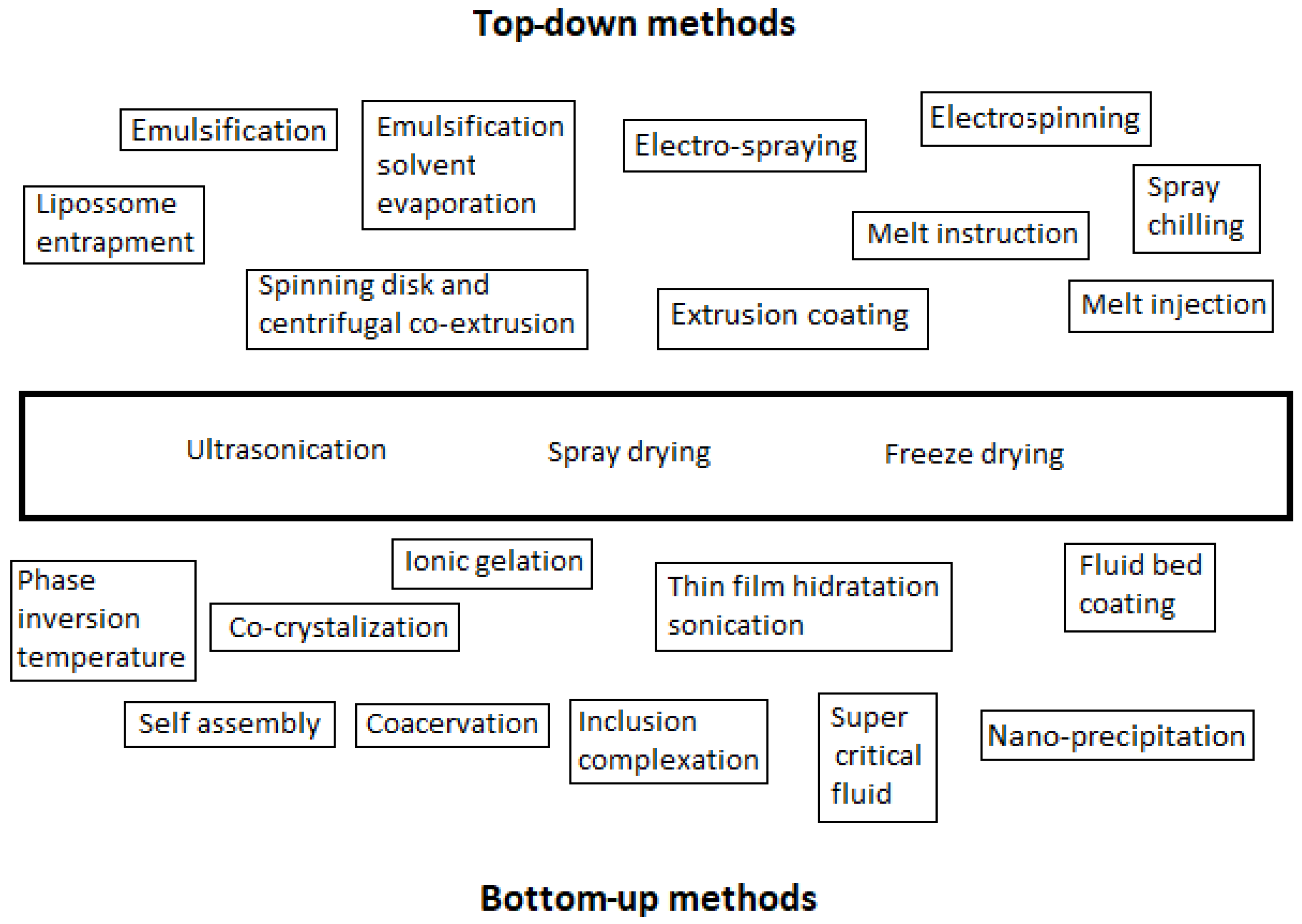
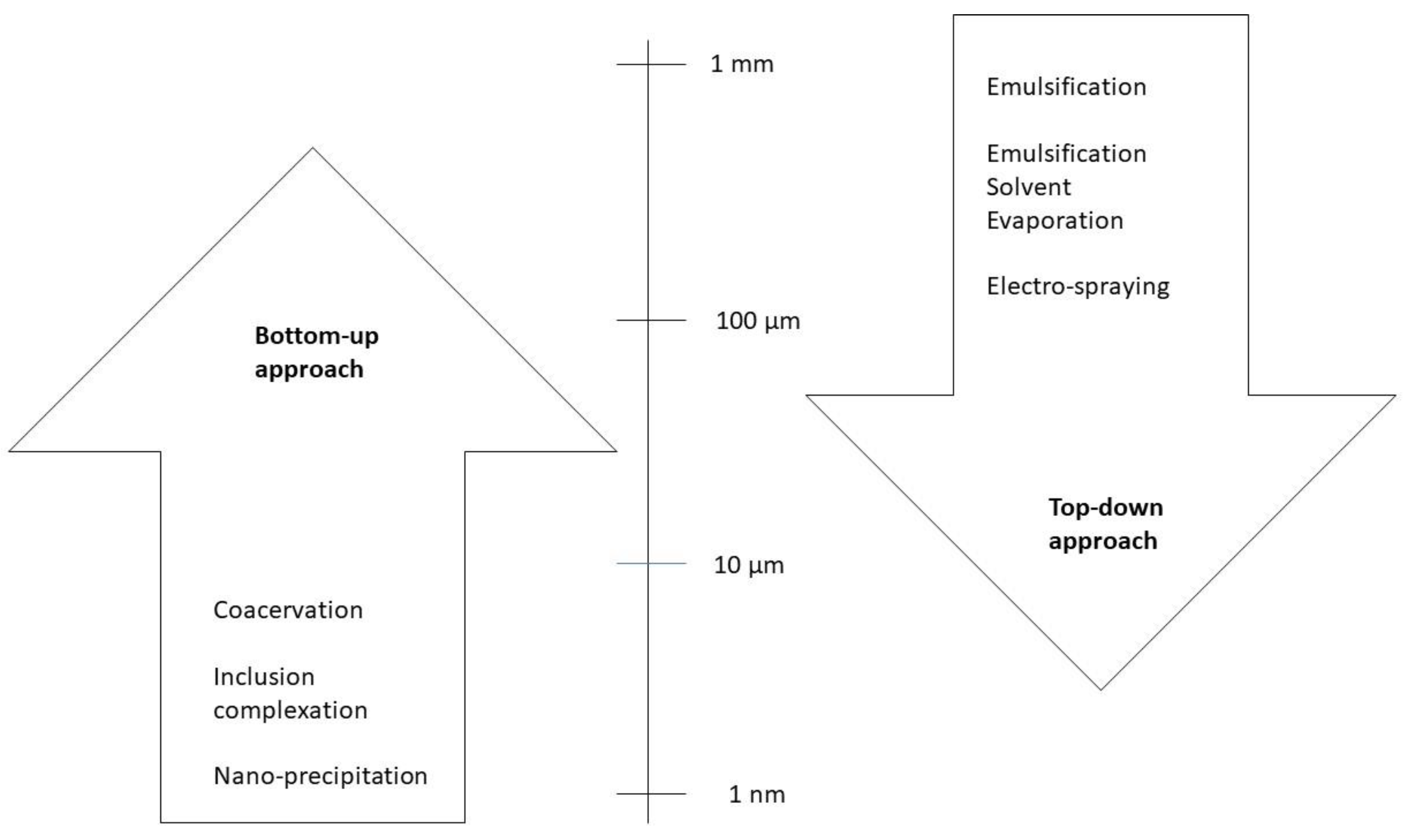
Two approaches, the top-down and the bottom-up approaches, can be used to produce micro- and nanocapsules. Figure 1 presents several technologies used to produce micro- and nanocapsules according to these approaches. Figure 2 illustrates the difference between the two approaches relative to the size of the particles: the bottom-up one consists of the elaboration of capsules by the self-building or self-organization of molecules from small particles (nm) to big encapsulated aggregates (µm); the top-down approach consists of the opposite, involving the disintegration of bulk solids, liquids, or large particles (1 mm), in small particles (from 1 µm down to, for example, 200 to 450 nm of gelatine hydrogel particles [11]).

Figure 1.
Techniques to produce micro- and nanocapsules, according to the top-down and bottom-up methodologies.

Figure 2.
Size of the micro- and nanocapsules (adapted with permission from [39]. Copyright 2017, Elsevier).
The top-down (TD) approach goes from a large-structure material to a small-structured one (Figure 2) through a size reduction and shaping of the structure through mechanical ways, such as milling, shredding, and grinding. It may use three types of forces to disrupt the particles: shear, impact, and compression. TD methodologies include emulsification, solvent evaporation, and extrusion. The latter, for example, creates small biopolymer particles in a solution by forcing it to pass through a nozzle into a gelling environment, with the size of the particles depending on the diameter of the needle, the flow rate, and the viscosity of the solution. Ultrasounds can be used to break up the polymer solution stream. Another process used is homogenization, which involves forming an emulsion by joining two immiscible liquids. This emulsion can be gelled by temperature management or by adding gelling agents, with the possibility of being an oil-in-water emulsion (normally, for proteins or polysaccharides) or a water-in-oil emulsion (hydrogels of 200–450 nm), but it can also be a water-in-water-in-oil or a water-in-oil-in-water emulsion [11]. Hydrophilic or hydrophobic compounds can be encapsulated by this TD methodology. However, it requires special tools and exerts low control over the particle size and structure. It may be applied only for some types of matrices. Furthermore, grinding and milling are not recommended for high-value and/or sensitive bioactive compounds because of the pressure and mechanical stress, which may induce damage [10,11].
The bottom-up (BU) methodology consists of the association of small particles (Figure 2) through self-building and self-organization. These can be affected by pH, temperature, the concentration of the encapsulated compound, and the ionic strengths. This approach includes spray drying, supercritical fluid expansion, inclusion complexation, coacervation, and nano-precipitation. These techniques consume less energy than top-down methods and allow control over the particle size, distribution, and structural morphology [10,11]. Coacervation is the process by which two oppositely charged biopolymers interact through electrostatic attraction, but hydrophobic interactions and hydrogen bonds may also occur [11]. Inclusion complexation is the molecular encapsulation of a bioactive molecule in the cavity of a host molecule. Drying is another technique, and it can be spray drying or freeze drying. Spray drying consists of atomizing a solution of biopolymers and bioactive compounds into fine droplets, forming microspheres or microgels with a size of 1 to 10 µm. Freeze drying includes freezing, sublimation, and desorption, requiring capsules with a high porosity, which affects the stability of the enclosed bioactive component and its retention efficiency. In a fluid gel formation, which includes thermal and ionic gelation, capsules are formed by applying shear forces on a biopolymer solution [11].
From a critical analysis of the various techniques to carry out micro- and nanoencapsulation (Table 1), it is possible to conclude that most methodologies begin by producing an emulsion through the junction of immiscible liquids or the solubilisation of solids, and then a specific technique is applied. For example, techniques such as nanoemulsion, ultrasonication, and freeze drying [40]; ionic gelation, ultrasonication, and freeze drying [41]; or ionic gelation and freeze drying [42] may be simultaneously used.
The literature underline that some techniques are used in both top-down and bottom-up methodologies, such as ultrasonication, spray drying, and freeze drying. However, there are others, such as ionic gelation and phase temperature inversion (some authors call it “fluid gel formation”, e.g., Joye et al. [11]), for which the classification as a TD or BU approach is non-consensual. Based on the principle of the nature, which consists of a powder solubilizing in a solvent, and taking into consideration that these methods use temperature inversion or ionic strength to form a gelling substance, they were classified as BU methodology in the present review work.
The TD methodology involves the use of precise tools and specified equipment, which allow size reduction and structure shaping. However, it is a more expensive technique due to the costs of the equipment and its maintenance. Another issue is the elaboration of particles with a well-defined structure. Furthermore, grinding and milling may not be suitable for sensitive bioactive ingredients. While using a BU approach, the aggregates are built by the self-organization of the molecules, and this is influenced by several factors. This approach allows the production of very fine particles, thus controlling the size, morphology, and physical state. In addition, the risk of contamination is significantly reduced compared with the TD approach, and BU methods require higher energy than TD ones [11].
Another important issue is how to choose the technique to use, and here it may be suggested that the final function of the encapsulated product, such as gelling or in the form of a powder or a solution for spraying or dipping the food product, is most likely what dictates this selection.
Either technique of micro/nanocapsulation must allow a high loading capacity, a high encapsulation efficiency, and the stability of the encapsulated system, while providing a long shelf life, biocompatibility, and the desired release characteristics of the encapsulated active compound [10].
3. Nanoencapsulation Systems
Encapsulation can be affected by the size, shape, and internal structure of the capsules, their physicochemical stability, and their entrapment and release behaviours, with their biological activity being important as well. It is possible to associate the nanocapsule to the methodology that is used to make the aggregates.
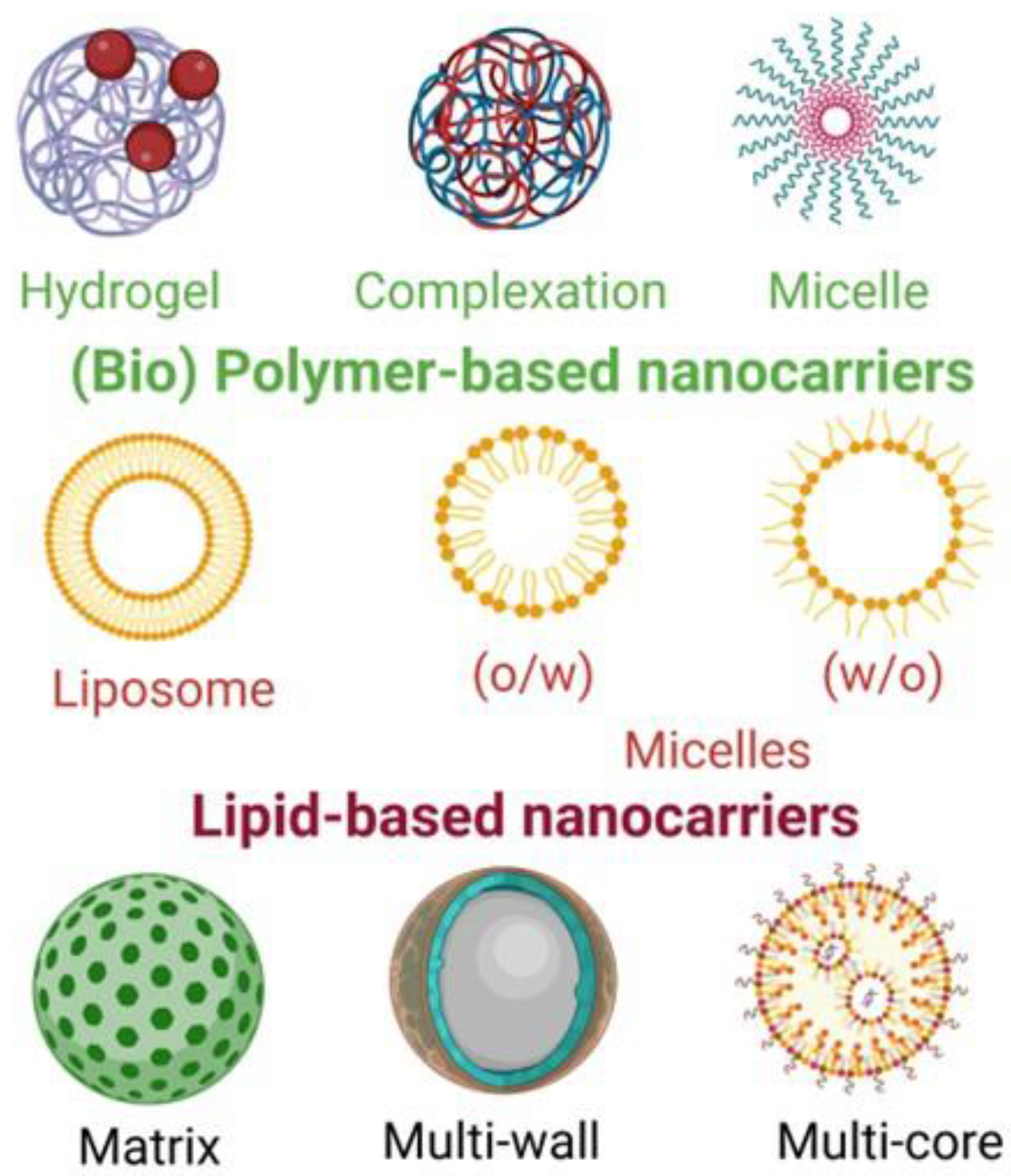
Nowadays, several techniques are available for producing nanocapsules, as well as several materials to use as the shell of these aggregates, allowing various types of nanocapsules. The principal types are mentioned in the following text and are illustrated in Figure 3 and Figure 4. The characteristics of the compound to be encapsulated and the objective for its use condition the choice of the encapsulation system, e.g., the polarity of the compound and its hydrophilicity or lipophilicity. There is a high variety of carriers in nanoencapsulation, natural compounds or synthetic polymers, with principal characteristics in the text below and illustrated in the next figures.

Figure 3.
Some important nanocarriers for bioactive compounds used in food applications (created with BioRender.com, accessed on 4 December 2022).

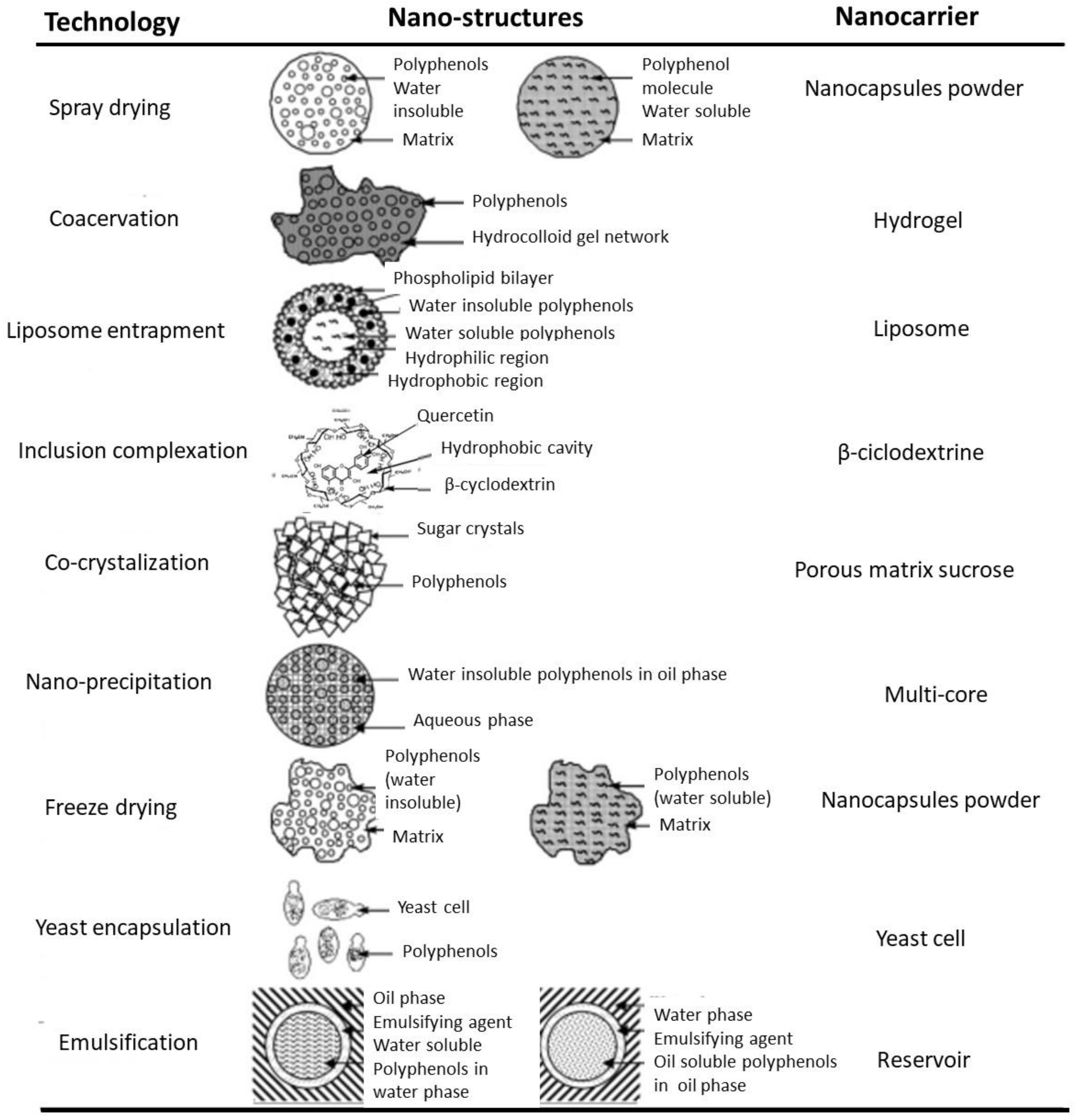
Figure 4.
Nanostructures and nanocarriers for some nanoencapsulated bioactive compounds (adapted with permission from [25]. Copyright 2010, Elsevier).
There are several types of nanoencapsulation systems:
Reservoir and matrix (Figure 3): the active compound, surrounded by a polymeric membrane, is in a single hollow chamber in the reservoir system, which is called the capsule, single core, mono core, or core shell. The active compound is distributed in the encapsulated material, but it can also exist on the surface of the matrix system, called the sphere or particle. Both the reservoir and matrix may be combined, resulting in a multilayer form or coated matrix [5,10,43].
Emulsion (Figure 4): there are two immiscible phases in an emulsion: the disperse phase and the continuous one. The disperse phase droplets can entrap the bioactive compound and the continuous phase protects the loaded droplets from the environment. Two types of this emulsion are the water-in-oil and oil-in-water types. The disadvantages of these are their thermodynamic instability and susceptibility to destabilization. Multilayer emulsions, multiple emulsions, and nano emulsions can be formed [5,10,44].
Lipid nano-particles (Figure 3): in preparation, this system is similar to the emulsion systems, with lipophilic active compounds being spread in a mixture of solid and liquid forms of lipids with emulsifiers. Nanoparticles can be composed of solid lipids and carriers can be nanostructured lipids. To produce these particles, high-energy consumption may be used, such as high-pressure homogenization, micro-fluidization, or sonication methods; low-energy consumption methods, such as the phase inversion temperature, microemulsion, solvent diffusion or injection, and supercritical fluid technology, may also be used [10,44].
Lipid vesicular carriers (Figure 3 and Figure 4): among the diverse vesicular carriers, such as niosomes, bilosomes, transferosomes, ethosomes, and phytosomes, liposomes are the most used in food. Liposomes contain a hydrophilic head (polar) and a hydrophobic tail (non-polar), acting as a semi-permeable membrane, which separates the inner aqueous phase from the external water phase. The methods used to produce liposomes are high-pressure homogenization, micro-fluidization, electro-spraying, supercritical carbon dioxide technology, and ethanol injection [10,44].
Hydrogel particles (Figure 4): this system is a three-dimensional and cross-linked polymeric network that is able to quickly absorb water and to hold it in less favourable conditions (heat, pressure). Natural polymers have attracted more interest than synthetic ones. Polysaccharide-based and protein-based polymers are the most used to prepare hydrogel particles through gelation. These techniques allow the encapsulation of both hydrophilic and hydrophobic bioactive compounds [10,44].
Biopolymer-based systems (Figure 3): this system consists of polyelectrolyte complexes formed by electrostatic interactions among oppositely charged polymers, such as carrageenan and protamine, solubilizing the nutraceutical compounds in either a positively or negatively charged biopolymer. Biopolymer substances, such as amylose, starch, pectin carrageenan, and chitosan, can be used.
Protein carbohydrates: protein carbohydrates are self-built structures composed of anionic polysaccharide and cationic protein surface groups, which may be produced by thermal denaturation or aggregation [44].
Molecular inclusion complexes (Figure 3 and Figure 4): molecular inclusion complexes are other encapsulated forms that are less used, such as cyclodextrins, but there are also nanofibres, nanotubes, and micelles [10].
The stability of a nanoencapsulation system may be inferred through the potential zeta, which is a physical property of particles in colloidal dispersions, influenced by the nanocapsule composition and the medium surrounding them [45].
4. Micro- and Nanocapsules from Natural Products
The use of “generally recognized as safe” (GRAS) materials is important for producing safe nanocapsules for food applications, along with the nutritional quality and stability. Polysaccharide-based carrier agents are natural carbohydrate polymers composed of various monosaccharides with glycosidic bonds and can be found in plants, animals, algae, and microorganisms. Some examples are starches from plants and the dextrins, maltodextrins, and cyclodextrins used to encapsulate hydrophobic bioactive compounds. Another example is cellulose, but it presents the limitation of being poorly soluble in water. Other compounds are pectin, chitosan, alginate, and gums. Upadhyay et al. [46] studied Cananga odorata essential oil nanoencapsulated with chitosan, forming a nano-emulsion. They concluded that it inhibited the growth of Apergilus flavus and improved the antioxidant activity. These results demonstrated that this formulation improved the shelf life of stored food against Apergilus flavus, which produces aflatoxin B1, and lipid peroxidation, turning natural nanocapsules into good candidates for edible coating formulations.
Protein-based carriers, which have a high nutritional value, may be GRAS materials. An example is whey protein, which has emulsifier proprieties; the caseins present in milk can constitute an excellent emulsifier. Good sources of edible proteins are gelatines, soy proteins, cereal (wheat, barley, and corn) proteins, potato protein, and pulse proteins (peas, chickpeas, lentils, beans, and lupins), and they present a high nutritional value. Another group of compounds with interest is the lipid-based carriers, fats, and oils. They may be polar lipids, such as monoglycerides and phospholipids, or nonpolar lipids, such as triacylglycerol and cholesterol. They are good emulsifiers that may be used in the encapsulation of bioactive compounds [10,45,47].
Natural resources are widely used as raw materials in industry. Nowadays, the reuse of natural bioactive compounds from by-products from the agro-food industry has increased, and under a circular bioeconomy perspective, these products are not wastes, but a source of economic-value compounds with a variety of applications. In some cases, by-products with low economic interest, but that are abundant, may also be used. The rational use of agro-industrial by-products in the nanobiotechnology field aims for the development of novel products and high-value-added processes. These agro-industrial by-products include fruit peels and seeds, pomaces, and straws, and they may be used as a source of high-value compounds, such as flavonoids, lycopene, and polyphenols. The main advantages of agro-industrial by-products are their abundance, low cost, biocompatibility, biodegradability, and, often, bioactivity. In this way, bioactive compound delivery systems may be obtained with relatively low production costs and environmentally sustainable management, due to the reuse of by-products [48].
Actually, around 1.3 billion tonnes of food are lost or wasted globally, costing USD 900 billion along the whole supply chain [49]. For instance, according to the World Bank, a 1% decrease in post-harvest losses might result in a USD 40 million reduction in this loss [50]. A more efficient use of agro-industrial by-products, considered residues, would most certainly contribute as well to reduce this loss, contributing at the same time to a circular economy by the conservation of the product value, materials, and resources in the economy for a long period with reduced waste generation, according to the Circular Economy Action Plan [49]. The extraction from these residues and the further micro- and nanoencapsulation of bioactive compounds with economic value for the food industry would represent a valorisation and a contribution to sustainable agro-food residue management.
4.1. Micro- and Nanoencapsulation for Functional Ingredient Delivery in Food Applications
Wrong lifestyle choices often contribute to the development of obesity, type 2 diabetes, and cardiovascular diseases. An adequate diet through functional ingredients could manage prevention at an early stage and before a therapeutic intervention. However, some of these ingredients cannot permeate into the small intestine in a sufficient concentration for efficacy without an efficient oral delivery system. Nowadays, there are various solutions to respond to these problems, such as using low-permeable hydrophilic peptides and macromolecules (nanoparticles), or intestinal permeation enhancers, for these mucolytics appear as a solution and with potential application. Gleeson et al. [44] found that there is an opportunity for the nutraceutical industry to explore the progress of the pharmaceutical industry in drug delivery systems. Excipients or substances already tested in humans have a high potential to be used in formulations in a delivery approach. This might improve the solubility, stability, or permeability of those molecules. In diet, the bioavailability of bioactive compounds is a challenge, mainly for hydrophobic compounds. Nanoencapsulation presents itself as a solution for isolating a bioactive compound from its natural environment, e.g., a fruit or vegetable, and incorporating it into a suitable delivery system [51]. The technological use of edible nanocapsules might serve to preserve bioactive compounds that are adequate for food applications, protecting the antioxidant and antimicrobial characteristics of these compounds (Table 2).

Table 2.
Micro- and nanoencapsulated bioactive compounds, plant sources, and functionalities for food applications.
From a critical analysis of the different micro- and nanoencapsulated bioactive compounds and related functionalities (Table 2), it is possible to conclude that the main purpose of using these compounds is to extend the shelf life of food products, due to their antimicrobial (mainly antifungal) and antioxidant activities. The principal technique of micro/nanoencapsulation is ionic gelation, probably because it is easy to apply in antimicrobial in vitro tests. Therefore, further studies are required on the application of encapsulates to the final food products. The studies reported in the literature are essentially in vitro studies on the effects of nano-aggregates on microorganisms; only a few studies apply to real food products.
As already mentioned in this review, there are nanocapsules that carry natural compounds, mainly essential oils, but also extracts from plants and fruits, for food applications, as antimicrobials and antioxidants. However, there are only few studies on nanocapsules carrying natural compounds from agricultural by-products (Table 3).

Table 3.
Micro- and nanoencapsulated bioactive compounds from agro-industrial by-products for the shelf-life extension of food products.
In addition, other types of compounds from by-products can be used in the shell composition of the nanocapsule. As mentioned above, soy, cereal, potato, and pulse proteins, polysaccharides, and lipid-based carriers are some examples of such compounds that may be used to encapsulate bioactive compounds. Table 3 shows some examples of shell materials, which can be obtained from agri-food by-products.
It would be of the uttermost importance to carry out more research work on the extraction and preservation by micro- and nanoencapsulation of identified bioactive compounds from agro-industrial by-products. As previously mentioned, these agro-industrial by-products are actually under-used (e.g., as animal feed), often abundant, of low cost, and contain high-value compounds with antimicrobial and antioxidant properties, presenting a high potential for further re-use in the food industry. On the other hand, the economic features of agro-industrial by-product re-use have not yet been fully explored. Therefore, all these research studies would contribute to advances towards a circular economy and sustainability.
4.2. Nanoparticles with Natural Compounds from Agriculture By-Products
Searching for the use of by-products in nanotechnology brings along another issue that should be considered in this review: the use of nanoparticles from different sources, such as gold, silver, platinum, zinc, or iron, with these particles being coated with natural compounds from agricultural wastes [90,91]. The use of these by-products is usually related to antioxidant activity, while reinforcing the antimicrobial activity. Normally, the production of these nanoparticles is studied at variable conditions (temperature, pH) in order to optimize their antimicrobial and/or antioxidant activities [90,92].
The production of nanoparticles coated with natural bioactive compounds is very interesting and has been explored by several authors. For example, authors have investigated the use of silver antimicrobial nanoparticles in combination with grape seeds and pomace extracts [91,93], orange peel [91,92], persimmon seeds, peel and calyx extracts [94], Persea americana peel, Beta vulgaris peel and Arachis hypogaea shell [90], Cocos nucifera L. shell [95], sugar cane bagasse [96], banana peels extract [97], and mango peel extract [98].
Although most examples of nanoparticles are from inorganic materials, some may also be lipid-, protein-, or polysaccharide-based (see Section 3, nanoencapsulation systems, above). For example, Angourani et al. [99] used zein (corn protein) nanoparticles to entrap rosemary essential oil (entrapment efficiency ca. 71%) in order to prevent evaporation and preserve its bioactivity. Bilal et al. [100] suggested the use of starch, cellulose, and pectin as the main types of polysaccharide-based nanoparticles for food applications.
5. Micro- and Nanocapsule Bioavailability
Recently, research has focused on the development of new nanocarriers with potential applications in the formulation of functional foods. However, only a few products have been commercialized. Among all the nanocarriers studied, only five were approved by the Food Safety European Authority [101]; these included inorganic materials (iron, silver, calcium, magnesium, selenium, and titanium oxide) for nanoparticles, and organic nanomaterials (lipid-, protein-, and polysaccharide-based). This is because a major portion of the tests were in vitro and only a few were in vivo and, therefore, more of the latter studies were required to demonstrate the efficiency and safety of such nanoparticles/capsules [101].
The rule of five [101] outlines that 90% of the oral drugs in the pharmaceutical area must obey three out of four of the following guidelines, which allows compounds to be obtained that have a good oral bioavailability:
- Molecular weight (MW) ≤ 500 Da;
- Logarithm of its partition coefficient (log(P)) ≤ 5;
- Hydrogen bond acceptors (HBA) ≤ 10;
- Hydrogen bond donors (HBD) ≤ 5.
Some authors also add a polar surface area (PSA) ≤ 140 Å2 and a number of rotatable bonds (NRotB) ≤ 10. These guidelines were constructed based on the results of oral bioavailability [101,102].
The absorption of bioactive nanocapsules is conditioned by the properties of the bioactive compounds, the nanocapsule used, and the environmental conditions. Table 4 summarizes the principal steps of the absorption of nanoencapsulated compounds.

Table 4.
Nanocapsule absorption in the human body ([103,104]).
There are various mechanisms for the release of bioactive compounds from nanocapsules: diffusion, dissolution, erosion, swelling, osmosis, degradation, and fragmentation [103,105]:
Diffusion: the diffusion of a bioactive compound from the interior to the exterior of an encapsulation system depends on the solubility in the encapsulated system and its permeability through the capsule material. The release rate of bioactive compounds can be affected by several factors, such as the bioactive compound characteristics (molecular weight, polarity, and vapour pressure); it depends also on the encapsulation system itself (polarity, physical state, interactions, and rheology).
Dissolution: two types of dissolution can be defined, with the first one being the encapsulation–dissolution-controlled system, in which bioactive compounds are encapsulated in dissolving materials and the dissolution rate is controlled by the solubility of the bioactive compounds and the physico-chemical proprieties of the carrier. In the second type, the matrix–dissolution-controlled system, the bioactive compounds are distributed uniformly through the particle, having an influence on the dissolution rate.
Erosion: this release mechanism happens when the encapsulated system faces a specific environmental condition, the chemical degradation of the particle matrix, causing the release of the bioactive compound. It may be caused by various factors, such as physical (high temperatures), chemical (strong acids or bases), or enzymatic. There are two types of erosion: bulk erosion, where the degradation occurs throughout the entire particle, and surface erosion, where the degradation only occurs on the surface of the encapsulated compound.
Swelling: the release of bioactive compounds happens when the capsule swells because of the solvent absorption; this release mechanism may be controlled by the selection of the polymeric matrix and the environmental conditions, such as temperature and pH.
Osmosis: the release begins when an osmotic pressure is created by water absorption, triggering the release of bioactive compounds. Ultimately, this type of release mechanism is comparable with a solvent-activated release (swelling mechanism) because the particles absorb the solvent until rupture and then release the bioactive compounds.
Degradation: degradation is the disruption of biomaterials by biological systems (microorganisms). The core compounds dispersed in the polymer matrix are released after the biodegradation of the polymer.
Fragmentation: the bioactive compounds are released from the encapsulation system when this is ruptured, due to environmental factors, such as pressure, pH, or enzymatic.
The release of nanoencapsulated bioactive compounds depends on the type of the bioactive compound and on the encapsulated system. The properties of bioactive compounds, such as the solubility, diffusivity, interior–exterior concentration gradient across the particle, interactions (repulsion forces between the bioactive compound and the encapsulation system), entrapment type of the bioactive compound inside the carrier, and size of the particle, will affect the release profile. The encapsulate properties can also condition the release of the bioactive compounds: the size, shape, structure, porosity, and composition can affect the release. Small nanoparticles tend to create an initial crack release followed by a slower one; however, big nanoparticles are degraded more slowly and display a slower diffusion of the bioactive compounds. The hydrophilicity or the hydrophobicity of a polymer may also cause strong interactions between the capsule and the bioactive compound, reducing the release rate. The oil phase of emulsion-based delivery systems controls the oil droplet polarity and increases the polymer molecular weight, with a high molecular weight displaying a reduced decomposition rate and causing an extended constant release of the bioactive compound over time. Some encapsulated bioactive compounds are soluble in either the oil or water phases and, as a consequence, they can leave the oil droplets when the emulsion is diluted in water. The environmental conditions, such as temperature, agitation, pH changes, and the presence of ions, can control the profile release of the bioactive compounds. For example, hydrogels are temperature-sensitive and pH-responsive. Other factors that can control the bioactive compound release are ultrasounds, light, oxidation, a reduction in the potential, and enzymes [103].
The effectiveness of nanocapsules requires evaluation, since they cannot be controlled with precision due to the complex processes of absorption, distribution, metabolism, and excretion [106].
6. Analysis of the Micro- and Nanocapsules
Nowadays, several techniques are available to analyse the structure of micro- and nanoencapsulated aggregates. Table 5 describes some of them, as well as their advantages in comparison to others, and what is possible to analyse by each technique.

Table 5.
Techniques to analyse the nanocapsules.
7. Conclusions
The absorption of nutraceuticals in the human body increases with the use of micro- and nanocapsules and the protection of bioactive compounds, with a better control release being the major advantages of these systems. An attempt towards the optimization of the use of micro- and nanocapsules in food applications has been carried out over time. This review comprises a critical analysis of the different methodologies for performing micro- and nanoencapsulation for food applications, classifying them according to top-down (TD) and/or bottom-up (BU); the principal types of encapsulation systems; the natural plant sources, including agro-industrial by-products, of bioactive compounds of interest for the food industry to be encapsulated; the bioavailability of the micro- and nanocapsules; and the main techniques used to analyse them. It was possible to conclude that most methodologies of encapsulation begin by producing an emulsion, and then another technique is applied. Some techniques may be used in both TD and BU approaches, but others are not consensually classified. It is suggested that the final function of the encapsulated product is likely to dictate the selection of the micro/nanoencapsulation technique to be used. It was possible to conclude, as well, that among different food applications, the main focus of micro- and nanocapsuled bioactive compounds from plant sources, including agro-industrial by-products, is to extend the shelf life of food products, given their antimicrobial and antioxidant activities. Most studies have been performed in vitro, with the principal micro/nanoencapsulation technique applied being ionic gelation. Therefore, more studies on real food products and in vivo studies need to be carried out. In addition, research work on the use of encapsulated natural bioactive compounds obtained from agro-industrial by-products must be further reinforced, as it presents a high potential in food applications and in the food industry, and it may be an available economical alternative towards a circular economy, with sustainability for the natural ecosystem. Finally, the release of micro/nanoencapsulated bioactive compounds depends on several factors and the effectiveness of the nanocapsule requires evaluation, as it cannot be precisely controlled given the complex processes involved.
Author Contributions
Conceptualization, R.M.S.C.M., A.M.M.B.M.; initial draft preparation, A.M.M.B.M., V.F.R.M.; writing, V.F.R.M.; writing—review and editing, A.M.M.B.M.; review, R.M.S.C.M., M.E.P.; supervision, A.M.M.B.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
This work was supported by National Funds from FCT - Fundação para a Ciência e a Tecnologia through Project UIDB/50016/2020. The first author thanks FCT for the PhD research grant, reference UI/BD/152825/2022.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Nejatian, M.; Darabzadeh, N.; Bodbodak., S.; Saberian., H.; Rafiee, Z.; Kharazmi., S.; Jafari., S.M. Practical applications of nanoencapsulated nutraceuticals in real food products; a systematic review. Adv. Colloid Interface Sci. 2022, 305, 102690. [Google Scholar] [CrossRef]
- Nallamuthu, I.; Khanum, F.; Fathima, S.J.; Patil, M.M.; Anand, T. Enhanced nutrient delivery through nanoencapsulation techniques: The current trend in food industry. In Nutrient Delivery; Academic Press: London, UK, 2017; pp. 619–651. [Google Scholar]
- Gulati, O.P.; Ottaway, P.B. Legislation relating to nutraceuticals in the European Union with a particular focus on botanical-sourced products. Toxicology 2006, 221, 75–87. [Google Scholar] [CrossRef]
- Garcia, J.L.G.; Rodriguez, A.V.C.; Valdés, M.H.R.; Godina, F.R.; Olivo, A.R.; Osuna, H.T.G. Micro and nanoencapsulation of bioactive compounds for agri-food applications: A review. Ind. Crop. Prod. 2022, 186, 115198. [Google Scholar] [CrossRef]
- Rossi, Y.E.; Braber, N.L.V.; Vergara, L.I.D.; Montenegro, M.A. Bioactive ingredients Obtained from Agro-industrial By-products: Recent Advances and Innovation in Micro- and Nanoencapsulation. J. Agric. Food Chem. 2021, 69, 15066–15075. [Google Scholar] [CrossRef]
- Pateiro, M.; Gomez, B.; Munekata, P.E.S.; Barba, F.J.; Putnik, P.; Danijela, B.K.; Lorenzo, J.M. Nanoencapsulation of promising bioactive compounds to improve their absorption, stability, functionality, and the appearance of the final food products. Molecules 2021, 26, 1547. [Google Scholar] [CrossRef]
- Ezhilarasi, P.N.; Karthik, P.; Chhanwal, N.; Anandharamakrishnan, C. Nanoencapsulation Techniques for Food Bioactive Components: A Review. Food Bioprocess Technol. 2013, 6, 628–647. [Google Scholar] [CrossRef]
- Awuchi, C.G.; Morya, S.; Dendegh, T.A.; Okpala, C.O.R.; Korzeniowska, M. Nanoencapsulation of food bioactive constituents and its associated processes: A revisit. Bioresour. Technol. Rep. 2022, 19, 101088. [Google Scholar] [CrossRef]
- Zehra, A.; Rai, A.; Singh, S.K.; Aamir, M.; Ansari, W.A.; Ram, S.U. An overview of nanotechnology in plant disease management, food safety, and sustainable agriculture. In Food Security and Plant Disease Management; Woodhead Publishing: Sawston, Cambridge, UK, 2021; pp. 193–219. [Google Scholar]
- Shishir, M.R.I.; Xie, L.; Sun, C.; Zheng, X.; Chen, W. Advances in micro and nano-encapsulation of bioactive compounds using biopolymer and lipid-based transporters. Trends Food Sci. Technol. 2018, 78, 34–60. [Google Scholar] [CrossRef]
- Joye, I.J.; McClements, D.J. Biopolymer based nanoparticles and microparticles: Fabrication, characterization and application. Curr. Opin. Colloid Interface Sci. 2014, 19, 417–427. [Google Scholar] [CrossRef]
- Weiss, J.; Takhistov, P.; McClements, D.J. Functional Materials in Food Nanotechnology. J. Food Sci. 2006, 71, R107–R116. [Google Scholar] [CrossRef]
- Wen, P.; Zong, M.H.; Linhardt, R.J.; Feng, K.; Wu, H. Electrospinning: A novel nano-encapsulation approach for bioactive compounds. Trends Food Sci. Technol. 2017, 70, 56–68. [Google Scholar] [CrossRef]
- Stoleru, E.; Munteanu, S.B.; Dumitriu, R.P.; Coroaba, A.; Drobota, M.; Zemljic, L.F.; Pricope, G.M.; Vasile, C. Polyethylene materials with multifunctional surface properties by electrospraying chitosan/vitamin E formulation destined to biomedical and food packaging applications. Iran Polym. J. 2016, 25, 295–307. [Google Scholar] [CrossRef]
- Tapia-Hernández, J.A.; Torres-Chávez, P.I.; Remirez-Wong, B.; Rascón-Chu, A.; Plascencia-Jatomea, M.; Barreras-Urbina, C.G.; Rangel-Vasquez, N.A.; Rodriguez-Felix, F. Micro- and Nanoparticles by Electrospray: Advances and Applications in Food. J. Agric. Food Chem. 2015, 63, 4699–4707. [Google Scholar] [CrossRef]
- Wang, J.; Jansen, J.A.; Yang, F. Electrospraying: Possibilities and Challenges of Engineering Carriers for Biomedical Applications—A Mini Review. Front. Chem. 2019, 7, 258. [Google Scholar] [CrossRef]
- Jafari, S.M. An overview of nanoencapsulation techniques and their classification. In Nanoencapsulation Technologies for the Food and Nutraceutical Industries; Jafari, S.M., Levy, N., Eds.; Gorgan University of Agriculture Science and Natural Resources: Gorgan, Iran, 2017; pp. 5–8. [Google Scholar]
- Fei, T.; Wan, Z.; Wang, T. Dispersing insoluble yolk low-density lipoprotein (LDL) recovered by complexing with carboxymethylcellulose (CMC) for the nanoencapsulation of hemp cannabidiol (CBD) through emulsification at neutral pH. Food Hydrocolloids. 2021, 116, 106656. [Google Scholar] [CrossRef]
- Gibbs, B.F.; Kermasha, S.; Alli, I.; Mulligan, C.N. Encapsulation in the food industry: A review. Int. J. Food Sci. Nutr. 1999, 50, 213–224. [Google Scholar]
- Jeyakumari, K.; Zynudheen, A.A.; Parvathy, U. Microencapsulation of Bioactive Food Ingredients and Controlled Release—A Review. MOJ Food Process. Technol. 2016, 2, 214–224. [Google Scholar]
- Pimentel-Moral, S.; Verardo, V.; Robert, P.; Segura-Carretero, A.; Martinez-Ferez, A. Nanoencapsulation strategies applied to maximize target delivery of intact polyphenols. In Nanotechnology in the Agri-Food Industry; Grumezescu, A.M., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 559–595. [Google Scholar]
- Castro, N.; Durrieu, V.; Raynaud, C.; Rouilly, A.; Rigal, L.; Quellet, C. Melt Extrusion Encapsulation of Flavors: A Review. Polym. Rev. 2016, 56, 137–186. [Google Scholar] [CrossRef][Green Version]
- Bamidele, O.P.; Emmambux, M.N. Encapsulation of bioactive compounds by “Extrusion” technologies: A review. Crit. Rev. Food Sci. Nutr. 2021, 61, 3100–3118. [Google Scholar] [CrossRef]
- Bampi, G.B.; Backes, G.T.; Cansian, R.L.; Matos, F.E., Jr.; Ansolin, I.M.A.; Poleto, B.C.; Corezzolla, L.R.; Trindade, C.S.F. Spray Chilling Microencapsulation of Lactobacillus acidophilus and Bifidobacterium animalis subsp. Lactis and Its Use in the Preparation of Savory Probiotic Bars. Food Bioprocess Technol. 2016, 9, 1422–1428. [Google Scholar]
- Fang, Z.; Bhandari, B. Encapsulation of polyphenols—A review. Trends Food Sci. Technol. 2010, 21, 510–523. [Google Scholar] [CrossRef]
- Klettenhammer, S.; Ferrentino, G.; Morozova, K.; Scampicchio, M. Novel Technologies Based on Supercritical Fluids for the Encapsulation of Food Grade Bioactive Compounds. Foods 2020, 9, 1395. [Google Scholar] [CrossRef]
- Guignon, B.; Duquenoy, A.; Dumoulin, E.D. Fluid Bed Encapsulation of Particles: Principles and Practice. Dry. Technol. 2002, 20, 419–447. [Google Scholar] [CrossRef]
- Mohammadi, A.; Hashemi, M.; Hosseini, S.M. Nanoencapsulation of Zataria multiflora essential oil preparation and characterization with enhanced antifungal activity for controlling Botrytis cinera, the causal agent of gray mould disease. Innov. Food Sci. Emerg. Technol. 2015, 28, 73–80. [Google Scholar] [CrossRef]
- Han, H.J.; Lee, J.S.; Park, S.A.; Ahn, J.B.; Lee, H.G. Extraction optimization and nanoencapsulation of jujube pulp and seed for enhancing antioxidant activity. Colloids Surf. B Biointerfaces 2015, 130, 93–100. [Google Scholar] [CrossRef]
- Bedoya-Serna, C.M.; Dacanal, G.C.; Fernandes, A.; Pinho, S.C. Antifungal activity of nanoemulsions encapsulating orégano (Origanum vulgare) essential oil: In vitro study and application in Minas Padrão chesse. Food Microbiol. 2018, 49, 929–935. [Google Scholar] [CrossRef]
- Peng, H.; Chen, S.; Luo, M.; Ning, F.; Zhu, X.; Xiong, H. Preparation and Self-Assembly Mechanism of Bovine Serum Albumin-Citrus Peel Pectin Conjugated Hydrogel: A Potential Delivery System for Vitamin C. J. Agric. Food Chem. 2016, 64, 7377–7384. [Google Scholar] [CrossRef]
- Pezeshky, A.; Ghanbarzadeh, B.; Hamishehkar, H.; Moghadam, M.; Babazadeh, A. Vitamin A palmitate-bearing nanoliposomes: Preparation and characterization. Food Biosci. 2016, 13, 49–55. [Google Scholar] [CrossRef]
- Vahidmoghadam, F.; Pourahmad, R.; Mortazavi, A.; Davoodi, D.; Azizinezhad, R. Characteristics of freeze-dried nanoencapsulated fish oil with whey protein concentrate and gum Arabic as wall materials. Food Sci. Technol. 2019, 39, 475–481. [Google Scholar] [CrossRef]
- Arpagaus, C.; Collenberg, A.; Rutti, D.; Assadpour, E.; Jafari, S.M. Nano spray drying for encapsulation of pharmaceuticals. Int. J. Pharm. 2018, 546, 194–214. [Google Scholar] [CrossRef]
- Rigon, R.T.; Zapata Noreña, C.P. Microencapsulation by spray-drying of bioactive compounds extracted from blackberry (Rubus fruticosus). J. Food Sci. Technol. 2016, 53, 1515–1524. [Google Scholar] [CrossRef]
- Kujur, A.; Kumar, A.; Yadav, A.; Prakash, B. Antifungal and aflatoxin B1 inhibitory efficacy of nanoencapsulated Pelargonium graveolens L. essential oil and its mode of action. LWT Food Sci.Technol. 2020, 130, 109619. [Google Scholar] [CrossRef]
- Shi, G.; Rao, L.; Yu, H.; Xiang, H.; Pen, G.; Long, S. Yeast cell-based microencapsulation of chlorogenic acid as a water-soluble antioxidant. J. Food Eng. 2007, 80, 1060–1067. [Google Scholar] [CrossRef]
- Gladbach, C.E.B. Encapsulation of Flavour in Yeast Using High Pressure Spraying. Ph.D. Thesis, Faculty of Mechanical Engineering, University of Ruhr, Bochum, Germany, 2021. [Google Scholar]
- Tontul, I.; Eroglu, E.; Topuz, A. Nanoencapsulation of Fish Oil and Essential Fatty Acids. In Nanoencapsulation of Food Bioactive Ingredients; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2017; Chapter 3; pp. 103–144. [Google Scholar]
- Kumar, A.; Singh, P.P.; Gupta, V.; Prakash, B. Assessing the antifungal and Aflatoxin B1 inhibitory efficacy of nanoencapsulated antifungal formulation based on combination of Ocimum spp. Essential oils. Int. J. Food Microbiol. 2020, 330, 108766. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, P.P.; Kumar, M.; Prakash, B. Nanoencapsulated plant-based antifungal formulation against the Aspergillus flavus and aflatoxin B1 contamination: Unraveling the biochemical and molecular mechanism of action. Int. J. Food Microbiol. 2022, 372, 109681. [Google Scholar] [CrossRef]
- Tiwari, S.; Dubey, N.K. Nanoencapsulated essential oils as novel green preservatives against fungal and mycotoxin contamination of food commodities. Curr. Opin. Food Sci. 2022, 45, 100831. [Google Scholar] [CrossRef]
- Jafari, S.M.; Assadpoor, E.; He, Y.; Bhandari, B. Encapsulation Efficiency of Food Flavors and Oils during Spray Drying. Dry. Technol. 2008, 26, 816–835. [Google Scholar] [CrossRef]
- Gleeson, J.P.; Ryan, S.M.; Brayden, D.J. Oral delivery strategies for nutraceuticals: Delivery vehicles and absorption enhancers. Trends Food Sci. Technol. 2016, 53, 90–101. [Google Scholar] [CrossRef]
- Santos, P.P.; Flôres, S.H.; Rios, A.O.; Chisté, R.C. Biodegradable polymers as wall materials to the synthesis of bioactive compound nanocapsules. Trends Food Sci. Technol. 2016, 53, 23–33. [Google Scholar] [CrossRef]
- Upadhyay, N.; Singh, V.K.; Dwived, A.K.; Chaudhari, A.K.; Dubey, N.K. Assessment of nanoencapsulated Cananga odarata essential oil in chitosan nanopolymer as a green approach to boost the antifungal, antioxidant and in situ efficacy. Int. J. Biol. Macromol. 2021, 171, 480–490. [Google Scholar] [CrossRef]
- Singh, H.; Ye, A.; Thompson, A. Nanoencapsulation Systems Based on Milk Proteins and Phospholipids. In Micro/Nanoencapsulation of Active Food Ingredients; Huang, Q., Given, P., Qian, M., Eds.; ABS Books Department, American Chemical Society: Washington, DC, USA, 2009; Chapter 8; pp. 131–142. [Google Scholar]
- Graziella, J.A.; Luciano, P.S. The emerging potential of by-products as platforms for drug delivery systems. Curr. Drug Targets 2014, 15, 478–485. [Google Scholar]
- Mak, T.M.; Xiong, X.; Tsang, D.C.; Iris, K.M.; Poon, C.S. Sustainable food waste management towards circular bioeconomy: Policy review, limitations and opportunities. Bioresour. Technol. 2020, 297, 122497. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, B. What’s Food Loss and Waste Got to Do with Sustainable Development? A Lot, Actually. 2015. World Resources Institute. Available online: https://www.wri.org/insights/whats-food-loss-and-waste-got-do-sustainable-development-lot-actually (accessed on 12 December 2022).
- Varzakas, T.; Zakynthinos, G.; Verpoort, F. Plant Food Residues as a Source of Nutraceuticals and Functional Foods. Foods 2016, 5, 88. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.R.; Andrade, M.; Silva, A.S. Application of encapsulated essential oils as antimicrobial agents in food packaging. Curr. Opin. Food Sci. 2017, 14, 78–84. [Google Scholar] [CrossRef]
- Paranagama, P.A.; Dayananda, K.R.; Hewage, J.W. Chemistry and Bioactive Compounds of Cinnamomum zeylanicum Blume. In Cinnamon; Senaratne, R., Pathirana, R., Eds.; Springer: Cham, Switzerland, 2020; pp. 251–271. [Google Scholar]
- Linares, I.B.; Stojanovic, Z.; Piné, R.Q.; Román, D.A.; Gajic, J.S.; Gutiérrez, A.F.; Carretero, A.S. Rosmarinus Officinalis Leaves as a Natural Source of Bioactive Compounds. Int. J. Mol. Sci. 2014, 15, 20585–20606. [Google Scholar] [CrossRef]
- Pezzani, R.; Vitalini, S.; Marcello, I. Bioactivities of origanum vulgare L.: An update. Phytochem. Rev. 2017, 16, 1253–1268. [Google Scholar] [CrossRef]
- Badgujar, S.B.; Patel, V.V.; Bandivdekar, A.H. Foeniculum vulgare Mill: A Review of Its Botany, Phytochemistry, Pharmacology, Contemporary Application, and Toxicology. Biomed. Res. Int. 2014, 2014, 842674. [Google Scholar] [CrossRef]
- Ghayempour, S.; Montazer, M.; Rad, M.M. Tragacanth gum as a natural polymeric Wall for producing antimicrobial nanocapsules loaded with plant extract. Int. J. Biol. Macromol. 2015, 81, 514–520. [Google Scholar] [CrossRef]
- Das, S.; Singh, V.K.; Dwivedy, A.K.; Chaudhari, A.K.; Dubey, N.K. Anethum graveolens Essential Oil Encapsulation in Chitosan Nanomatrix: Investigations on in vitro release behavior, Organoleptic Attributes, and Efficacy as Potential Delivery Vehicles against Biodeterioration of rice (Oryza sativa L.). Food Bioprocess Technol. 2021, 14, 831–853. [Google Scholar] [CrossRef]
- Das, S.; Singh, V.K.; Dwivedy, A.K.; Chaudhari, A.K.; Deeppika; Dubey, N.K. Nanostructured Pimpinella anisum essential oil as novel green food preservative against fungal infestation, aflatoxin B1 contamination and deterioration of nutritional qualities. Food Chem. 2021, 344, 128574. [Google Scholar] [CrossRef]
- Kapustova, M.; Granata, G.; Napoli, E.; Puskárová, A.; Bucková, M.; Pangallo, D.; Geraci, C. Nanoencapsulated Essential Oils with Enhanced Antifungal Activity for Potential Applications on Agri-food, Material and Environmental Fields. J. Antibiot. 2021, 10, 31. [Google Scholar] [CrossRef] [PubMed]
- Osanloo, M.; Abdollahi, A.; Valizadeh, A.; Abedinpour, N. Antibacterial potential od essential oils of Zataria Multiflora and Mentha piperita, micro and nano formulated forms. Iran J. Microbiol. 2020, 12, 43–51. [Google Scholar] [PubMed]
- Das, S.; Singh, V.K.; Dwivedy, A.K.; Chaudhari, A.K.; Upadhyay, N.; Singh, A.; Deepika; Dubey, N.K. Fabrication, characterization and practical efficacy of Myristica fragrans essential oil nanoemulsion delivery system against postharvest biodeterioration. Ecotoxicol. Environ. Saf. 2020, 189, 110000. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Singh, V.K.; Dwivedy, A.K.; Chaudhari, A.K.; Deepika; Dubey, N.K. Eugenol loaded chitosan nanoemulsion for food protection and inhibition of Aflatoxin B1 synthesizing genes based on molecular docking. Carbohydr. Polym. 2021, 255, 117339. [Google Scholar] [CrossRef]
- Singh, V.K.; Das, S.; Dwivedy, A.K.; Rathore, R.; Dubey, N.K. Assessment of chemical characterized nanoencapsulated Ocimum sanctum essential oil against aflatoxigenic fungi contaminating herbal raw materials and its novel mode of action as methyglyoxal inhibitor. Postharvest Biol. Technol. 2019, 153, 87–95. [Google Scholar] [CrossRef]
- Khan, A.; Ahmad, A.; Akhtar, F.; Yousuf, S.; Xess, I.; Khan, L.A.; Manzoor, N. Ocimum sanctum essential oil and its active principles exert their antifungal activity by disrupting ergosterol biosynthesis and membrane integrity. Res. Microbiol. 2010, 161, 816–823. [Google Scholar] [CrossRef]
- Li, J.; Xu, J.; Yang, X.; Ren, L.; Wang, Y.; Ma, D.; Fan, P.; Wang, H.; Liu, L.; Dong, B.; et al. Effects of phenanthrene on the essential oil composition and leaf metabolome in peppermint plants (Mentha piperita L.). Ind. Crop. Prod. 2022, 187, 115383. [Google Scholar] [CrossRef]
- Beyki, M.; Zhaveh, S.; Khalili, S.T.; Rahmani-Cherati, T.; Abollahi, A.; Bayat, M.; Tabatabaei, M.; Mohsenifar, A. Encapsulation of Mentha piperita essential oils in chitosan-cinnamic acid nanogel with enhanced antimicrobial activity against Aspergillus flavus. Ind. Crop. Prod. 2014, 54, 310–319. [Google Scholar] [CrossRef]
- López-Menezes, A.K.; Plascencia-Jatomea, M.; Lizardi-Mendoza, J.; Fernández-Quiroz, D.; Rodriguez-Félix, F.; Mouriño-Perez, R.R.; Cortez-Rocha, M.O. Schinus molle L. essential oil-loaded chitosan nanoparticles: Preparation, characterization, antifungal and anti-aflatoxigenic properties. LWT Food Sci. Technol. 2018, 96, 597–603. [Google Scholar] [CrossRef]
- Singh, A.; Deepika; Chaudhari, A.K.; Das, S.; Singh, V.K.; Dwivedy, A.K.; Shivalingam, R.K.; Dubey, N.K. Assessment of preservative potential of Bunium persicum (Boiss) essential oil against fungal and aflatoxin contamination of stored masticatories and improvement in efficacy through encapsulation into chitosan nanomatrix. Environ. Sci. Pollut. Res. 2020, 27, 27635–27650. [Google Scholar] [CrossRef]
- Kumar, A.; Kujur, A.; Singh, P.P.; Prakash, B. Nanoencapsulated plant based bioactive formulation against food-borne molds and aflatoxin B1 contamination: Preparation, characterization, and stability evaluation in the food system. Food Chem. 2019, 287, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Dwivedy, A.K.; Singh, V.K.; Prakash, B.; Dubey, N.K. Nanoencapsulated Illicium verum Hook.f. essential oil as an effective novel plant-based preservative against aflatoxin B1 production and free radical generation. Food Chem. Toxicol. 2018, 111, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Dasgupta, N.; Singh, V.; Mishra, N.C.; Singh, H.; Purohit, S.D.; Srivastava, N.; Ranjan, S.; Yadav, P.; Mishra, N. Inhibitory effect of clove oil nanoemulsion on fumonisin isolated from maize kernels. LWT Food Sci. Technol. 2020, 134, 110237. [Google Scholar] [CrossRef]
- Deepika; Chaudhary, A.K.; Singh, A.; Das, S.; Dubey, N.K. Nanoencasulated Pretoselinum crispum essential oil: Characterization and practical efficacy against fungal and aflatoxin contamination of stored chia seeds. Food Biosci. 2021, 42, 101117. [Google Scholar] [CrossRef]
- Pandey, S.; Vindya, H.A.; Rani, N.D.K.; Kumar, K.A.; Rao, P.J. Curcumin loaded core-shell biopolymers colloid and its incorporation in Indian Basmati rice: An enhanced stability, anti-oxidant activity and sensory attributes of fortified rice. Food Chem. 2022, 387, 132860. [Google Scholar] [CrossRef]
- Teixeira, V.M.C.; Silva, R.F.G.; Gonçalves, O.H.; Pereira, C.; Barros, L.; Ferreira, I.C.F.R.; Bona, E.; Leimann, F.V. Chemometric approaches to evaluate the substitution of synthetic food dyes by natural compounds: The case of nanoencapsulated curcumin, spirulina and hibiscus extracts. LWT Food Sci. Technol. 2022, 154, 112786. [Google Scholar] [CrossRef]
- Silva, L.M.; Hill, L.E.; Figueiredo, E.; Gomes, L.C. Delivery of phytochemicals of tropical fruit by-products using poly (DL-lactide-co-glycolide) (PLGA) nanoparticles: Synthesis, characterization, and antimicrobial activity. Food Chem. 2014, 165, 362–370. [Google Scholar] [CrossRef]
- Rashid, R.; Wani, S.M.; Manzoor, S.; Masoodi, F.A.; Dar, M.M. Improving oxidative stability of edible oils with nanoencapsulated orange peel extract powder during accelerated shelf life storage. Food Biosci. 2022, 49, 101917. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Y.; Wu, X.; Liu, J.; Zhang, Y.; Shi, Q.; Fang, Z. Ultrasonic-assisted extraction, calcium alginate encapsulation and storage stability of mulberry pomace phenolics. J. Food Meas. Charact. 2021, 15, 4517–4529. [Google Scholar] [CrossRef]
- Luca, A.; Cilek, B.; Hasirci, V.; Sahin, S.; Sumnu, G. Effect of Degritting of Phenolic Extract from Sour Cherry Pomace on Encapsulation Efficiency Production of Nano-suspension. Food Bioprocess Technol. 2013, 6, 2494–2502. [Google Scholar] [CrossRef]
- Mahmoud, K.F.; Ibrahim, M.A.; Mervat, E.D.; Shaaban, H.A.; Kamil, M.M.; Hegazy, N.A. Nano-encapsulation efficiency of Lemon and Orange Peels Extracts on Cake Shelf Life. Am. J. Food Technol. 2016, 11, 63–75. [Google Scholar] [CrossRef]
- Kaur, G.; Sharma, N.; Singh, A.; Kapoor, S.; Khatkar, S.K. Ultrasound-assisted microemulsions of anthocyanins extracted from black carrot pomace and its utilisation as functional component in kulfi: Implication on in vitro release, bio-functional components and rheological characteristics. Int. J. Food Sci. Technol. 2022. [Google Scholar] [CrossRef]
- Rashid, R.; Masoodi, F.A.; Wani, S.M.; Manzoor, S.; Gull, A. Ultrasound assisted extraction of bioactive compounds from pomegranate peel, their nanoencapsulation and application for improvement in shelf life extension of edible oils. Food Chem. 2022, 385, 132608. [Google Scholar] [CrossRef] [PubMed]
- Correa Filho, L.C.; Lourenco, S.C.; Duarte, D.F.; Moldão-Martins, M.; Alves, V.D. Microencapsulation of Tomato (Solanum lycopersicum L.) Pomace Ethanolic Extract by Spray Drying: Optimization of Process Conditions. Appl. Sci. 2019, 9, 612. [Google Scholar] [CrossRef]
- Hadidi, M.; Rostamabadi, H.; Moreno, A.; Jafari, S.M. Nanoencapsulation of essential oils from industrial hemp (Cannabis sativa L.) by-products into alfalfa protein nanoparticles. Food Chem. 2022, 386, 132765. [Google Scholar] [CrossRef] [PubMed]
- Caballero, S.; Li, Y.O.; McClements, D.J.; Davidov-Pardo, G. Encapsulation and delivery of bioactive citrus pomace polyphenols: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 8028–8044. [Google Scholar] [CrossRef] [PubMed]
- Bamba, B.S.B.; Shi, J.; Tranchant, C.C.; Xue, S.J.; Forney, C.F.; Lim, L.T.; Xu, W.; Xu, G. Coencapsulation of Polyphenols and Anthocyanins from Blueberry Pomace by Double Emulsion Stabilized by Whey Proteins: Effect of Homogenization Parameters. Molecules 2018, 23, 2525. [Google Scholar] [CrossRef] [PubMed]
- Saffarzadeh-Matin, S.; Shahbazi, M. Modeling and Optimization of process conditions in nanoencapsulation of the polyphenolic extract of industrial apple pomace. Innov. Food Technol. 2016, 3, 1–13. [Google Scholar]
- Tsali, A.; Goula, A.M. Valorization of grape pomace: Encapsulation and storage stability of its phenolic extract. Powder Technol. 2018, 340, 194–207. [Google Scholar] [CrossRef]
- Sylla, N.; Bouyahya, A.; Taha, D.; Dakka, N.; Elhajji, H. Study of the antioxidant and antidiabetic activity in vitro of free and encapsulated phenolic compounds of olive pomace. Biocatal. Agric. Biotechnol. 2021, 36, 102126. [Google Scholar] [CrossRef]
- Das, G.; Shin, H.S.; Patra, J.K. Comparative Assessment of Antioxidant, Anti-Diabetic and Cytotoxic Effects of Tree Peel/Shell food waste extract-Mediated Silver Nanoparticles. Int. J. Nanomedicine 2020, 15, 9075–9088. [Google Scholar] [CrossRef]
- Soto, K.M.; Crevantes, C.T.Q.; Iturriaga, M.H.; Barcenas, G.L.; Duhalt, R.V.; Mendoza, S. Fruit peels waste for the green synthesis of silver nanoparticles with antimicrobial activity against food borne pathogens. LWT Food Sci. Technol. 2019, 103, 293–300. [Google Scholar] [CrossRef]
- Konwarh, R.; Gogoi, B.; Philip, R.; Laskar, M.A.; Karak, N. Biomimetic preparation of polymer-supported free radical acavenging, cytocompatible and antimicrobial “green” silver nanoparticles using aqueous extract of Citrus sinensis peel. Colloids Surf. B Biointerfaces 2011, 84, 338–345. [Google Scholar] [CrossRef]
- Yao, P.; Zhang, J.; Xing, T.; Chen, G.; Tao, R.; Choo, K.H. Green synthesis of silver nanoparticles using grape seed extract and their applications for reductive catalysis of Direct Orange 26. J. Ind. Eng. Chem. 2018, 58, 74–79. [Google Scholar]
- Ramachandraiah, K.; Gnoc, N.T.B.; Chin, K.B. Biosynthesis of Silver Nanoparticles from Persimmon By-products and Incorporation in Biodegradable Sodium Alginate Thin Film. J. Food Sci. 2017, 82, 2329–2336. [Google Scholar] [CrossRef]
- Sinsinwar, S.; Sarkar, M.K.; Suriya, K.R.; Nithyanand, P.; Valdivel, V. Use of agricultural waste (coconut shell) for the synthesis of silver nanoparticles and evaluation of their antibacterial activity against selected human pathogens. Microb. Pathog. 2018, 124, 30–37. [Google Scholar] [CrossRef]
- Aguilar, N.M.; Cardona, F.A.; Estevez, J.O.; González, N.R.S.; Serrano, J.C.B.; Kuri, U.S. Controlled biosynthesis of silver nanoparticles using sugar industry waste, and its antimicrobial activity. J. Environ. Chem. Eng. 2018, 6, 6275–6281. [Google Scholar] [CrossRef]
- Ibrahim, H.M.M. Green synthesis and characterization of silver nanoparticles using banana peel extract and their antimicrobial activity against representative microorganisms. J. Radiat. Res. Appl. Sci. 2015, 8, 265–275. [Google Scholar] [CrossRef]
- Yang, N.; Li, W.H. Mango peel extract mediated novel route for synthesis of silver nanoparticles and antibacterial application of silver nanoparticles loaded onto-woven fabrics. Ind. Crop. Prod. 2013, 48, 81–88. [Google Scholar] [CrossRef]
- Angourani, H.R.; Heydari, M.; Yousefi, A.R.; Pashaei, B.; Mastinu, A. Nanoparticles Based-Plant Protein Containing Rosmarinus officinalis Essential Oil; Fabrication, Characterization, and Evaluation. Appl. Sci. 2022, 12, 9968. [Google Scholar] [CrossRef]
- Bilal, M.; Gul, I.; Basharat, A.; Qamar, S.A. Polysaccharides-based bio-nanostructures and their potential food applications. Int. J. Biol. Macromol. 2021, 176, 540–557. [Google Scholar] [CrossRef]
- Dima, C.; Assadpour, E.; Dima, S.; Jafari, S.M. Bioactive-loaded nanocariers for functional foods: From designing to bioavailability. Curr. Opin. Food Sci. 2020, 33, 21–29. [Google Scholar] [CrossRef]
- Matsson, P.; Doak, B.C.; Over, B.; Kihlberg, J. Cell permeability beyond the rule of five. Adv. Drug Deliv. Rev. 2016, 11, 42–61. [Google Scholar] [CrossRef]
- Boostani, S.; Jafari, S.M. Controller release of nanoencapsulated food ingredients. In Nanoencapsulation in the Food Industry, Release and Bioavailability of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; Chapter 2; Volume 5, pp. 27–78. [Google Scholar]
- Peanparkdee, M.; Iwamoto, S. Encapsulation for Improving in Vitro Gastrointestinal Digestion of Plant Polyphenols and Their Application in Food Products. Food Rev. Int. 2022, 38, 335–353. [Google Scholar] [CrossRef]
- Boostani, S.; Jafari, S.M. A comprehensive review on the controlled release of encapsulated food ingredients; fundamental concepts to design and applications. Tends Food Sci. Technol. 2021, 109, 303–321. [Google Scholar] [CrossRef]
- Zhang, A.; Meng, K.; Liu, Y.; Pan, Y.; Qu, W.; Chen, D.; Xie, S. Absorption, distribution, metabolism, and excretion of nanocarriers in vivo and their influences. Adv. Colloid Interface Sci. 2020, 284, 102261. [Google Scholar] [CrossRef]
- Tarhan, O.; Jafari, S.M. Spectroscopic and chromatographic analyses of nanoencapsulated food ingredients. In Characterization of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; Chapter 17; Volume 4, pp. 585–615. [Google Scholar]
- Kochani, R.; Jafari, S.M.; Van de Ven, T.G.M. Going deep inside bioactive-loaded nanocarriers through Nuclear Magnetic Resonance (NMR) spectroscopy. Trends Food Sci. Technol. 2020, 101, 198–212. [Google Scholar]
- Kochani, R.; Jafari, S.M. Nuclear magnetic resonance (NMR) spectroscopy of nanoencapsulated food ingredients. In Characterization of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Academic press: Cambridge, MA, USA, 2020; Chapter 12; Volume 4, pp. 411–458. [Google Scholar]
- Tosi, M.M.; Ramos, A.P.; Esposto, B.S.; Jafari, S.M. Dynamic light scattering (DLS) of nanoencapsulated food ingredients. In Characterization of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; Chapter 6; Volume 4, pp. 191–211. [Google Scholar]
- Sarabandi, K.; Gharehbeglou, P.; Jafari, S.M. Scanning electron microscopy (SEM) of nanoencapsulated food ingredients. In Characterization of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; Chapter 3; Volume 4, pp. 83–130. [Google Scholar]
- Sarabandi, K.; Mahoonak, A.S.; Hamishikar, H.; Ghorbani, M.; Jafari, S.M. Microencapsulation of casein hydrolysates: Physicochemical, antioxidant and microstructure properties. J. Food Eng. 2018, 237, 86–95. [Google Scholar] [CrossRef]
- Djomeh, Z.E.; Pure, A.E.; Pure, M.E. Atomic force microscopy (AFM) of nanoencapsulated food ingredients. In Characterization of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; Chapter 5; Volume 4, pp. 159–188. [Google Scholar]
- Rafiee, Z.; Nejatian, M.; Bodbodak, S.; Jafari, S.M. Surface composition of nanoencapsulated food ingredients by X-ray photoelectron spectroscopy (XPS). In Characterization of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; Chapter 8; Volume 4, pp. 243–268. [Google Scholar]
- Sharif, L.; Nia, S.K.; Jafari, S.M. Confocal laser scanning microscopy (CLSM) of nanoencapsulated food ingredients. In Characterization of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; Chapter 4; Volume 4, pp. 131–158. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).