Application of Aqueous Saline Process to Extract Silkworm Pupae Oil (Bombyx mori): Process Optimization and Composition Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Materials and Chemicals
2.2. Extraction of the Silkworm Pupae Oil
2.3. Determination of the Peroxide Value (PV)
2.4. Determination of the Acid Value (AV)
2.5. Analysis of the Chemical Composition of the Silkworm Pupae and Oil
2.6. Determination of 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Free Radical Scavenging Activity
2.7. Experimental Design and Statistical Analysis
3. Results and Discussion
3.1. Chemical Composition of Silkworm Pupae
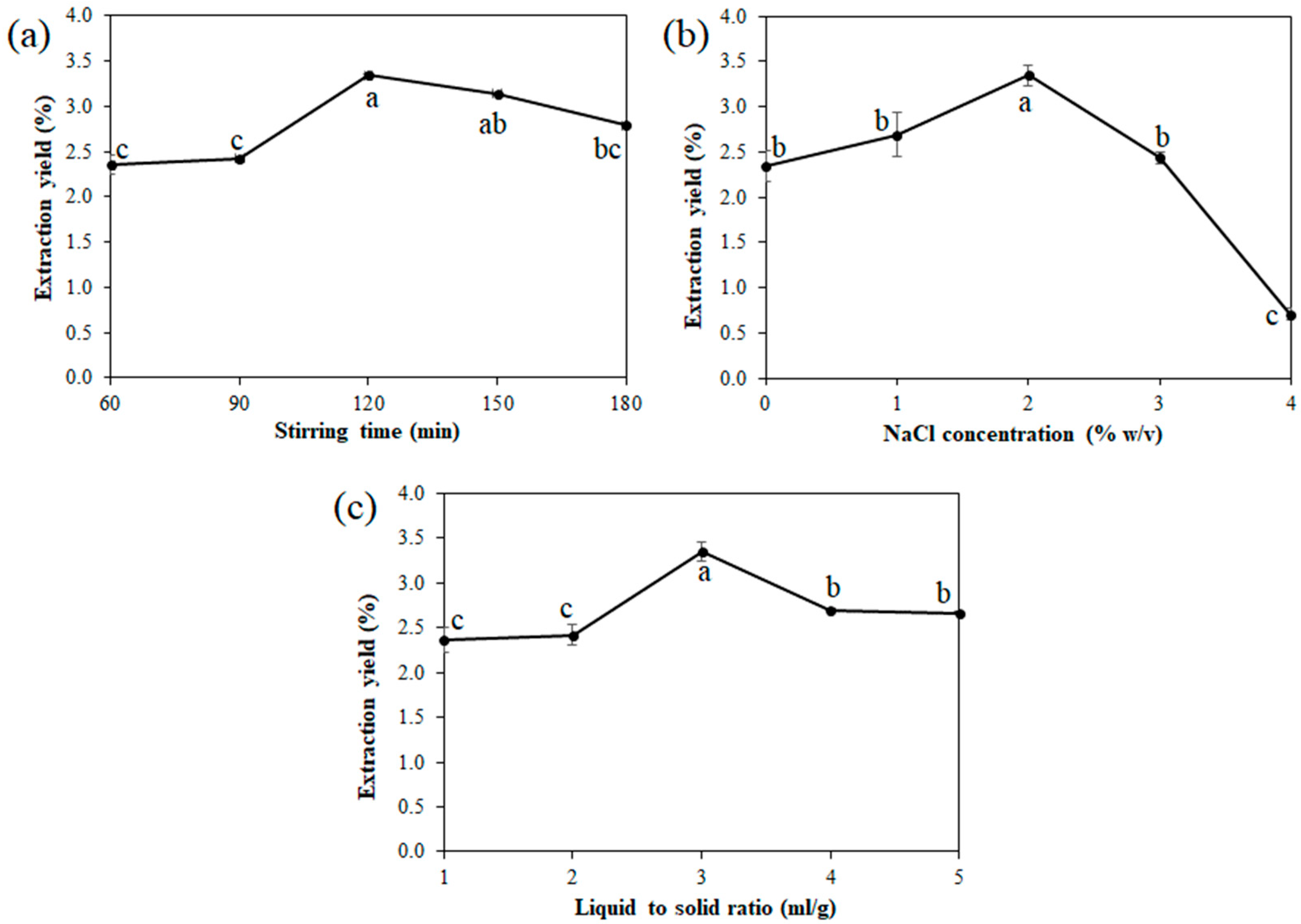
3.2. Effect of a Single Factor on the Extraction Yield
3.2.1. Effect of Stirring Time
3.2.2. Effect of the Concentration of NaCl
3.2.3. Effect of the Liquid-to-Solid Ratio
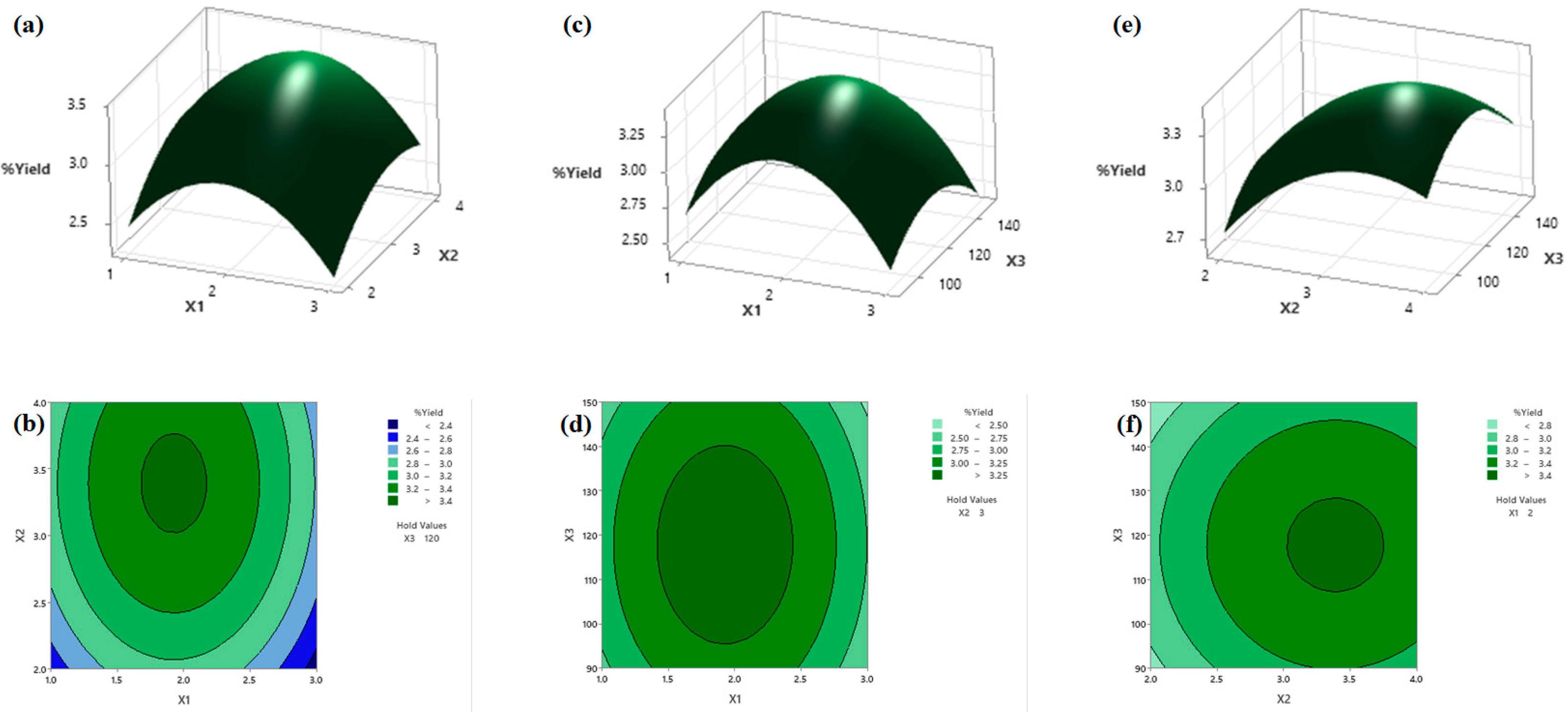
3.3. Effect of Independent Processing Parameters on Response Variables from the BBD Experiment
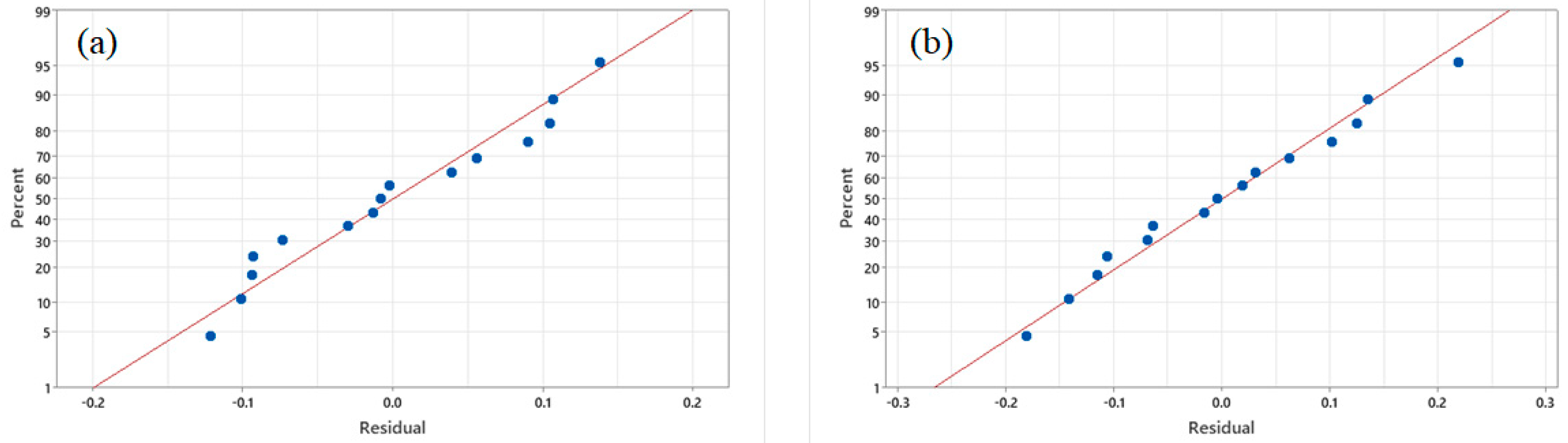
3.4. Model Fitting
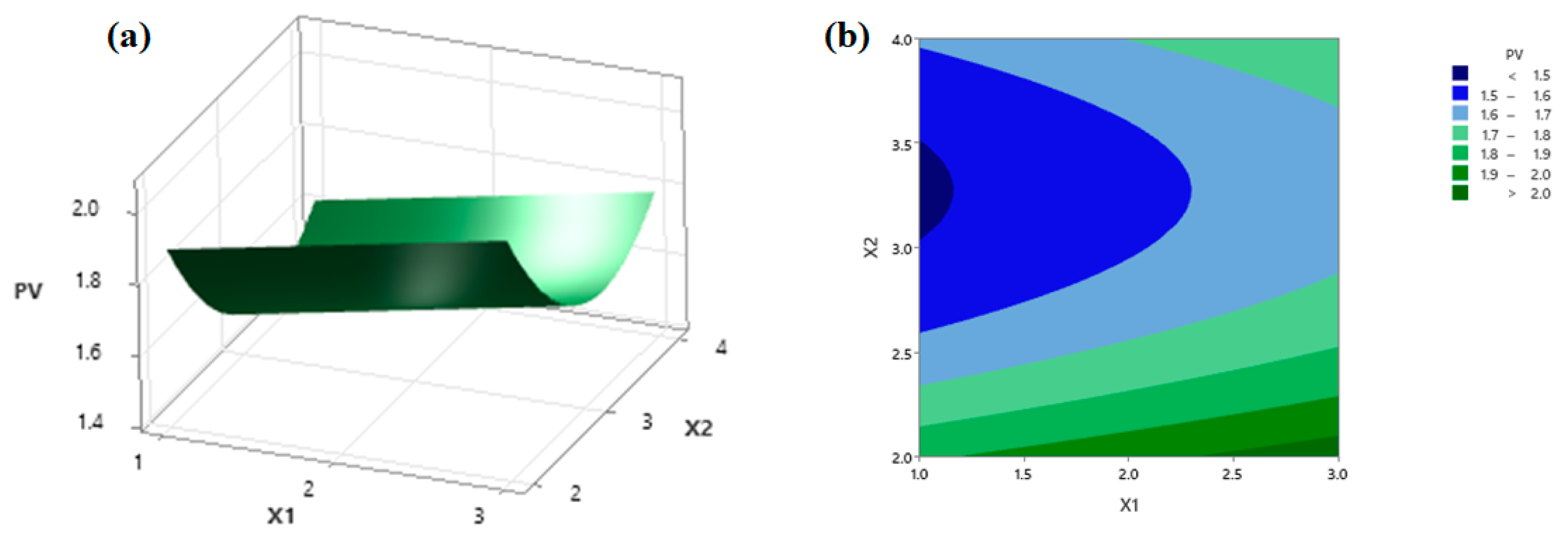
3.5. Analysis of the Response Surface
3.6. Optimization and Validation of the Regression Model
3.7. Chemical Compositions of Silkworm Pupae Oil Extracted by Aqueous Saline
3.7.1. Fatty Acid Composition
3.7.2. Minor Components
3.8. DPPH Free Radical Scavenging Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Manjunath, R.N.; Kumar, A.; Kumar, K.P.A. Utilisation of Sericulture Waste by Employing Possible Approaches. In Contaminants in Agriculture Sources, Impacts and Management; Naeem, M., Ansari, A.A., Gill, S.S., Eds.; Springer: Cham, Switzerland, 2020; pp. 385–399. [Google Scholar]
- Hu, B.; Li, C.; Zhang, Z.; Zhao, Q.; Zhu, Y.; Su, Z.; Chen, Y. Microwave-assisted extraction of silkworm pupal oil and evaluation of its fatty acid composition, physicochemical properties and antioxidant activities. Food Chem. 2017, 231, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Sonjui, T.; Noomhorm, C.; Promboon, A. Sericin recovery from silk cocoon degumming wastewater by a membrane process. Kasetsart J. (Nat. Sci.) 2009, 43, 538–549. [Google Scholar]
- Rao, P.; Rathod, V. Valorization of Food and Agricultural Waste: A Step towards Greener Future. Chem Rec. 2019, 19, 1858–1871. [Google Scholar] [CrossRef]
- Hirunyophat, P.; Chalermchaiwat, P.; On-nom, N.; Prinyawiwatkul, W. Selected nutritional quality and physicochemical properties of silkworm pupae (frozen or powdered) from two species. Int. J. Food Sci. 2021, 56, 3578–3587. [Google Scholar] [CrossRef]
- Longvah, T.; Manghtya, K.; Qadri, S.S. Eri silkworm: A source of edible oil with a high content of α-linolenic acid and of significant nutritional value. J. Sci. Food Agric. 2012, 92, 1988–1993. [Google Scholar] [CrossRef] [PubMed]
- Pereira, N.R.; Ferrarese-Filho, O.; Matsushita, M.; de Souza, N.E. Proximate composition and fatty acid profile of Bombyx mori L. chrysalis toast. J. Food Compost Anal. 2003, 16, 451–457. [Google Scholar] [CrossRef]
- Rao, P.U. Chemical composition and nutritional evaluation of spent silk worm pupae. J. Agric. Food Chem. 1994, 42, 2201–2203. [Google Scholar] [CrossRef]
- Ray, M.; Gangopadhyay, D. Effect of maturation stage and sex on proximate, fatty acid and mineral composition of eri silkworm (Samia ricini) from India. J. Food Compost. Anal. 2021, 100, 103898. [Google Scholar] [CrossRef]
- Saviane, A.; Tassoni, L.; Naviglio, D.; Lupi, D.; Savoldelli, S.; Bianchi, G.; Cortellino, G.; Bondioli, P.; Folegatti, L.; Casartelli, M. Mechanical processing of Hermetia illucens larvae and Bombyx mori pupae produces oils with antimicrobial activity. Animals 2021, 11, 783. [Google Scholar] [CrossRef] [PubMed]
- Kotake-Nara, E.; Yamamoto, K.; Nozawa, M.; Miyashita, K.; Murakami, T. Lipid profiles and oxidative stability of silkworm pupal oil. J. Oleo Sci. 2002, 51, 681–690. [Google Scholar] [CrossRef] [Green Version]
- Pan, W.J.; Liao, A.M.; Zhang, J.G.; Dong, Z.; Wei, Z.J. Supercritical carbon dioxide extraction of the oak silkworm (Antheraea pernyi) pupal oil: Process optimization and composition determination. Int. J. Mol. Sci. 2012, 13, 2354–2367. [Google Scholar] [CrossRef] [Green Version]
- Srinivas, G.; Nidoni, U.; Ramachandra, C.T.; Ramappa, K.T.; Ashoka, J. Supercritical fluid extraction of pupae oil from mulberry silkworm (Bombyx mori L.). J. Pharmacogn Phytochem. 2019, 8, 4507–4513. [Google Scholar]
- Wei, Z.-J.; Liao, A.-M.; Zhang, H.-X.; Liu, J.; Jiang, S.-T. Optimization of supercritical carbon dioxide extraction of silkworm pupal oil applying the response surface methodology. Bioresour. Technol. 2009, 100, 4214–4219. [Google Scholar] [CrossRef]
- Aitta, E.; Marsol-Vall, A.; Damerau, A.; Yang, B. Enzyme-assisted extraction of fish oil from whole fish and by-products of Baltic herring (Clupea harengus membras). Foods 2021, 10, 1811. [Google Scholar] [CrossRef]
- Li, X.; Cao, J.; Bai, X.; Zhang, F. Chemical composition and thermal properties of tilapia oil extracted by different methods. Int. J. Food Prop. 2018, 21, 1575–1585. [Google Scholar] [CrossRef] [Green Version]
- Okada, T.; Morrissey, M.T. Recovery and characterization of sardine oil extracted by pH adjustment. J. Agric. Food Chem. 2007, 55, 1808–1813. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, V.V.; Ghaly, A.E.; Brooks, M.S.; Budge, S.M. Extraction of oil from mackerel fish processing waste using alcalase enzyme. Enzym. Eng. 2013, 2, 1–10. [Google Scholar]
- Tzompa-Sosa, D.A.; Yi, L.; van Valenberg, H.J.F.; Lakemond, C.M.M. Four insect oils as food ingredient: Physical and chemical characterisation of insect oils obtained by an aqueous oil extraction. J. Insects Food Feed. 2019, 5, 279–292. [Google Scholar] [CrossRef]
- Saha, B.C.; Hayashi, K. Debittering of protein hydrolyzates. Biotechnol. Adv. 2001, 19, 355–370. [Google Scholar] [CrossRef]
- Yoon, S.; Wong, N.A.K.; Chae, M.; Auh, J.-H. Comparative characterization of protein hydrolysates from three edible insects: Mealworm larvae, adult crickets, and silkworm pupae. Foods 2019, 8, 563. [Google Scholar] [CrossRef] [Green Version]
- Sui, Y.; Huang, W.-C.; Wu, Y.; Qi, X.; Mao, X. Lipid extraction from Greenland halibut (Reinhardtius hippoglossoides) by-product in low-voltage DC electric field and its mechanism. J. Clean. Prod. 2021, 283, 124673. [Google Scholar] [CrossRef]
- Cruz, S.; Yousfi, K.; Pérez, A.G.; Mariscal, C.; Garcia, J.M. Salt improves physical extraction of olive oil. Eur. Food Res. Technol. 2007, 225, 359–365. [Google Scholar] [CrossRef]
- Kadioglu, S.I.; Phan, T.T.; Sabatini, D.A. Surfactant-based oil extraction of corn germ. J. Am. Oil Chem. Soc. 2011, 88, 863–869. [Google Scholar] [CrossRef]
- Prichapan, N.; McClements, D.; Klinkesorn, U. Encapsulation of iron within W1/O/W2 emulsions formulated using a natural hydrophilic surfactant (saponin): Impact of surfactant level and oil phase crystallization. Food Biophysics. 2020, 15, 346–354. [Google Scholar] [CrossRef]
- AOCS. Official and Recommended Practices of the AOCS; AOCS Press Publication: Champaign, IL, USA, 1997. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International, 21st ed.; AOAC International: Rockville, MD, USA, 2019. [Google Scholar]
- Speek, A.J.; Schrijver, J.; Schreurs, W.H.P. Vitamin E composition of some seed oils as determined by high-performance liquid chromatography with fluorometric detection. J. Food Sci. 1985, 50, 121–124. [Google Scholar] [CrossRef]
- Speek, A.J.; Temalilwa, C.R.; Schrijver, J. Determination of β-carotene content and vitamin A activity of vegetables by high-performance liquid chromatography and spectrophotometry. Food Chem. 1986, 19, 65–74. [Google Scholar] [CrossRef]
- Singh, R.P.; Chidambara Murthy, K.N.; Jayaprakasha, G.K. Studies on the antioxidant activity of pomegranate (Punica granatum) peel and seed extracts using in vitro models. J. Agric. Food Chem. 2002, 50, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Yolmeh, M.; Jafari, S.M. Applications of response surface methodology in the food industry processes. Food Bioprocess Technol. 2017, 10, 413–433. [Google Scholar] [CrossRef]
- Pennino, M.; Dierenfeld, E.; Behler, J. Retinol, α-tocopherol and proximate nutrient composition of invertebrates used as feed. Int. Zoo Yearbook. 2007, 30, 143–149. [Google Scholar] [CrossRef]
- Longvah, T.; Mangthya, K.; Ramulu, P. Nutrient composition and protein quality evaluation of eri silkworm (Samia ricinii) prepupae and pupae. Food Chem. 2011, 128, 400–403. [Google Scholar] [CrossRef]
- White, D.; Kendall, K. Grizzly bears and army cutworm moths in the alpine of Glacier National Park, Montana. Int. Bear News. 1993, 2, 2–3. [Google Scholar]
- FAO; WHO. Food Labelling; FAO: Rome, Italy, 2007. [Google Scholar]
- Rumpold, B.A.; Schlüter, O.K. Nutritional composition and safety aspects of edible insects. Mol. Nutr. Food Res. 2013, 57, 802–823. [Google Scholar] [CrossRef]
- Zhang, R.; Rao, Z.; Li, Y.; Li, H.; Fei, L.; Lei, S.; Wang, Y. Silkworm excrement derived in-situ co-doped nanoporous carbon as confining sulfur host for lithium sulfur batteries. Chem. Select. 2019, 4, 5678–5685. [Google Scholar] [CrossRef]
- Xu, X.-H.; Liu, Z.-X.; Shi, X.-Y.; Miao, C.; Sheng, S.; Xu, Y.; Wu, F.-A.; Wang, J. Fed-batch fermentation of Yarrowia Lipolytica using defatted silkworm pupae hydrolysate: A dynamic model-based approach for high yield of lipid production. Waste Biomass Valoriz. 2018, 9, 2399–2411. [Google Scholar] [CrossRef]
- Wang, W.; Wang, N.; Zhou, Y.; Zhang, Y.; Xu, L.; Xu, J.; Feng, F.; He, G. Isolation of a novel peptide from silkworm pupae protein components and interaction characteristics to angiotensin I-converting enzyme. Eur. Food Res. Technol. 2011, 232, 29–38. [Google Scholar] [CrossRef]
- Bhavsar, P.; Fontana, G.D.; Tonin, C.; Patrucco, A.; Zoccola, M. Superheated water hydrolyses of waste silkworm pupae protein hydrolysate: A novel application for natural dyeing of silk fabric. Dyes Pigm. 2020, 183, 108678. [Google Scholar] [CrossRef]
- Tomotake, H.; Katagiri, M.; Yamato, M. Silkworm pupae (Bombyx mori) are new sources of high quality protein and lipid. J. Nutr. Sci. Vitaminol. (Tokyo) 2010, 56, 446–448. [Google Scholar] [CrossRef] [Green Version]
- Matsakidou, A.; Mantzouridou, F.T.; Kiosseoglou, V. Optimization of water extraction of naturally emulsified oil from maize germ. LWT-Food Sci. Tech. 2015, 63, 206–213. [Google Scholar] [CrossRef]
- Rosenthal, A.; Pyle, D.L.; Niranjan, K. Aqueous and enzymatic processes for edible oil extraction. Enzyme Microb. Technol. 1996, 19, 402–420. [Google Scholar] [CrossRef]
- Huang, Y.; Yin, Z.; Guo, J.; Wang, F.; Zhang, J. Oil extraction and evaluation from yellow horn using a microwave-assisted aqueous saline process. Molecules 2019, 24, 2598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maseer Nejres, A. Salting Out. 2020. Available online: https://www.researchgate.net/publication/347936827_salting_out (accessed on 3 December 2021).
- Yi, L.; Van Boekel, M.A.J.S.; Lakemond, C.M.M. Extracting Tenebrio molitor protein while preventing browning: Effect of pH and NaCl on protein yield. J. Insects as Food Feed. 2017, 3, 21–31. [Google Scholar] [CrossRef]
- Hashemi, S.M.B.; Kamani, M.H.; Amani, H.; Mousavi Khaneghah, A. Voltage and NaCl concentration on extraction of essential oil from Vitex pseudonegundo using ohmic-hydrodistillation. Ind. Crops Prod. 2019, 141, 111734. [Google Scholar] [CrossRef]
- Li, W.; Zhou, R.; Mu, Y. Salting effects on protein components in aqueous NaCl and urea solutions: Toward understanding of urea-induced protein denaturation. J. Phys. Chem. B 2012, 116, 1446–1451. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, M. Optimization of deep eutectic solvent-based ultrasound-assisted extraction of polysaccharides from Dioscorea opposita Thunb. Int. J. Biol. Macromol. 2017, 95, 675–681. [Google Scholar] [CrossRef]
- Williams, J.P.; Williams, J.R.; Kirabo, A.; Chester, D.; Peterson, M. Nutrient content and health benefits of insects. In Insects as Sustainable Food Ingredients; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 61–84. [Google Scholar]
- Zou, Y.-X.; Hu, T.-G.; Shi, Y.; Liu, J.; Mu, L.-X.; Xiao, Y.; Liao, S.-T. Establishment of a model to evaluate the nutritional quality of Bombyx mori Linnaeus (Lepidoptera, Bombycidae) pupae lipid based on principal components. J. Asia Pac. Entomol. 2017, 20, 1364–1371. [Google Scholar] [CrossRef]
- Shanker, K.S.; Shireesha, K.; Kanjilal, S.; Kumar, S.V.; Srinivas, C.; Rao, J.V.; Prasad, R.B. Isolation and characterization of neutral lipids of desilked eri silkworm pupae grown on castor and tapioca leaves. J. Agric. Food Chem. 2006, 54, 3305–3309. [Google Scholar] [CrossRef]
- Alimentarius, C. Codex Standard for edible fats and oils not covered by individual standards. Codex Stan 1981, 19, 1–6. [Google Scholar]
- Granato, D.; de Araújo Calado, V.M.; Jarvis, B. Observations on the use of statistical methods in food science and technology. Food Res. Int. 2014, 55, 137–149. [Google Scholar] [CrossRef]
- Hippolyte, M.; Augustin, M.; Tchougang, H.; Robert, N.; Somashekar, D. Application of response surface methodology to improve the production of antimicrobial biosurfactants by Lactobacillus paracasei subsp. tolerans N2 using sugar cane molasses as substrate. Bioresour. Bioprocess. 2018, 5, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Baş, D.; Boyacı, İ.H. Modeling and optimization I: Usability of response surface methodology. J. Food Eng. 2007, 78, 836–845. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhu, Y.; Zheng, Y.; Liu, Z.; Zhong, Y.; Deng, Y.; Zhao, Y. Effects of salting-in/out-assisted extractions on structural, physicochemical and functional properties of Tenebrio molitor larvae protein isolates. Food Chem. 2021, 338, 128158. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Xu, Z.; Zheng, B.; Lo, Y.M. Optimization of ultrasonic-assisted extraction of pomegranate (Punica granatum L.) seed oil. Ultrason. Sonochem. 2013, 20, 202–208. [Google Scholar] [CrossRef]
- Zhao, B.; Zhou, H.; Zhang, S.; Pan, X.; Li, S.; Zhu, N.; Wu, Q.; Wang, S.; Qiao, X.; Chen, W. Changes of protein oxidation, lipid oxidation and lipolysis in Chinese dry sausage with different sodium chloride curing salt content. Food Sci. Hum. Wellness. 2020, 9, 328–337. [Google Scholar] [CrossRef]
- Mi, E. Effect of some preservatives on the peroxide value and carbohydrate content of dry melon seeds. J. Chem. Pharm. 2017, 9, 139–142. [Google Scholar]
- Farmani, J.; Rostammiri, L. Characterization of chicken waste fat for application in food technology. J. Food Meas. Charact. 2015, 9, 143–150. [Google Scholar] [CrossRef]
- Sabir, S.M.; Hayat, I.; Gardezi, S.D.A. Estimation of sterols in edible fats and oils. Pak. J. Nutr. 2003, 2, 178–181. [Google Scholar]
- Chee, K.M.; Gong, J.X.; Rees, D.M.G.; Meydanl, M.; Ausman, L.; Johnson, J.; Siguel, E.N.; Schaefer, E.J. Fatty acid content of marine oil capsules. Lipids 1990, 25, 523–528. [Google Scholar] [CrossRef]
- Precht, D. Cholesterol content in European bovine milk fats. Die Nahrung. 2001, 45, 2–8. [Google Scholar] [CrossRef]
- Belluco, S.; Losasso, C.; Maggioletti, M.; Alonzi, C.C.; Paoletti, M.G.; Ricci, A. Edible insects in a food safety and nutritional perspective: A critical review. Compr. Rev. Food Sci. Food Saf. 2013, 12, 296–313. [Google Scholar] [CrossRef]
- Winitchai, S.; Manosroi, J.; Abe, M.; Boonpisuttinant, K.; Manosroi, A. Free radical scavenging activity, tyrosinase inhibition activity and fatty acids composition of oils from pupae of native Thai silkworm (Bombyx mori L.). Agric. Nat. Resour. 2011, 45, 404–412. [Google Scholar]
- Ekpo, K.; Onigbinde, A.; Asia, I.O. Pharmaceutical potentials of the oils of some popular insects consumed in southern Nigeria. Afr. J. Pharm. Pharmacol. 2009, 3, 51–57. [Google Scholar]
- Nagendran, B.; Unnithan, U.; Choo, Y.; Sundram, K. Characteristics of red palm oil, a carotene-and vitamin E–rich refined oil for food uses. Food Nutr. Bull. 2000, 21, 189–194. [Google Scholar] [CrossRef]
- Kreps, F.; Vrbiková, L.; Schmidt, Š. Influence of industrial physical refining on tocopherol, chlorophyll and beta-carotene content in sunflower and rapeseed oil. Eur. J. Lipid Sci. Technol. 2014, 116, 1572–1582. [Google Scholar] [CrossRef]
- Teh, S.-S.; Birch, E. Physicochemical and quality characteristics of cold-pressed hemp, flax and canola seed oils. J. Food Compost Anal. 2013, 30, 26–31. [Google Scholar] [CrossRef]
- Grilo, E.C.; Costa, P.N.; Gurgel, C.S.S.; Beserra, A.F.D.L.; Almeida, F.N.D.S.; Dimenstein, R. Alpha-tocopherol and gamma-tocopherol concentration in vegetable oils. Food Sci. Technol. 2014, 34, 379–385. [Google Scholar] [CrossRef] [Green Version]
- Vidrih, R.; Vidakovič, S.; Abramovič, H. Biochemical parameters and oxidative resistance to thermal treatment of refined and unrefined vegetable edible oils. Czech J. Food Sci. 2010, 28, 376–384. [Google Scholar] [CrossRef] [Green Version]
- Ravinder, T.; Kaki, S.S.; Kunduru, K.R.; Kanjilal, S.; Rao, B.; Swain, S.; Prasad, R.B.N. Physico-chemical characterization and oxidative stability studies of eri silkworm oils. Int. J. Modern Chem. Appl. Sci. 2016, 3, 293–300. [Google Scholar]
- Samaram, S.; Mirhosseini, H.; Tan, C.P.; Ghazali, H.M.; Bordbar, S.; Serjouie, A. Optimisation of ultrasound-assisted extraction of oil from papaya seed by response surface methodology: Oil recovery, radical scavenging antioxidant activity, and oxidation stability. Food Chem. 2015, 172, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.T.; Wu, C.Y.; Weng, Y.M.; Tseng, C.Y. Ultrasound-assisted extraction methodology as a tool to improve the antioxidant properties of herbal drug Xiao-chia-hu-tang. J. Ethnopharmacol. 2005, 99, 293–300. [Google Scholar] [CrossRef]
- Deori, M.; Boruah, D.C.; Devi, D.; Devi, R. Antioxidant and antigenotoxic effects of pupae of the muga silkworm Antheraea assamensis. Food Biosci. 2014, 5, 108–114. [Google Scholar] [CrossRef]

| Nutrient | Content (per 100 g) | |
|---|---|---|
| Fresh Pupae | Dry Basis | |
| Moisture (g) | 77.43 ± 0.08 | - |
| Energy (kcal) | 117.55 ± 0.49 | 520.69 ± 3.96 |
| Protein (g) | 11.72 ± 0.01 | 51.89 ± 0.15 |
| Total fat (g) | 6.07 ± 0.15 | 26.87 ± 0.75 |
| Total carbohydrate (g) | 4.03 ± 0.21 | 17.83 ± 0.85 |
| Dietary fiber (g) | 1.07 ± 0.06 | 4.72 ± 0.30 |
| Ash (g) | 0.77 ± 0.01 | 3.41 ± 0.05 |
| Run | NaCl Concentration (% w/v, X1) | Liquid-to-Solid Ratio (mL/g, X2) | Stirring Time (min, X3) | Extraction Yield (%, Y1) | PV (mM, Y2) | AV (mg KOH/g, Y3) | ||
|---|---|---|---|---|---|---|---|---|
| Exp | Pred | Exp | Pred | |||||
| 1 | 2 (0) | 4 (1) | 90 (−1) | 3.25 ± 0.02 c | 3.10 | 1.64 ± 0.03 cd | 1.70 | 0.61 ± 0.09 a |
| 2 | 2 (0) | 4 (1) | 150 (1) | 3.09 ± 0.05 d | 2.98 | 1.64 ± 0.05 cd | 1.70 | 0.56 ± 0.14 a |
| 3 | 3 (1) | 3 (0) | 90 (−1) | 2.51 ± 0.02 gh | 2.51 | 1.66 ± 0.03 cd | 1.68 | 0.72 ± 0.10 a |
| 4 | 1 (−1) | 4 (1) | 120 (0) | 2.78 ± 0.01 e | 2.83 | 1.72 ± 0.19 cd | 1.61 | 0.64 ± 0.15 a |
| 5 | 3 (1) | 4 (1) | 120 (0) | 2.57 ± 0.03 fg | 2.67 | 1.82 ± 0.14 bc | 1.78 | 0.75 ± 0.18 a |
| 6 | 3 (1) | 3 (0) | 150 (1) | 2.48 ± 0.04 gh | 2.33 | 1.74 ± 0.12 c | 1.68 | 0.71 ± 0.11 a |
| 7 | 1 (−1) | 3 (0) | 150 (1) | 2.60 ± 0.06 fg | 2.55 | 1.40 ± 0.03 e | 1.50 | 0.64 ± 0.15 a |
| 8 | 1 (−1) | 3 (0) | 90 (−1) | 2.65 ± 0.01 f | 2.67 | 1.72 ± 0.24 cd | 1.50 | 0.53 ± 0.15 a |
| 9 | 1 (−1) | 2 (−1) | 120 (0) | 2.57 ± 0.03 fg | 2.44 | 1.74 ± 0.15 c | 1.88 | 0.64 ± 0.15 a |
| 10 | 2 (0) | 3 (0) | 120 (0) | 3.30 ± 0.04 bc | 3.37 | 1.41 ± 0.03 e | 1.59 | 0.68 ± 0.08 a |
| 11 | 2 (0) | 2 (−1) | 90 (−1) | 2.62 ± 0.11 fg | 2.71 | 1.99 ± 0.05 ab | 1.97 | 0.64 ± 0.15 a |
| 12 | 2 (0) | 3 (0) | 120 (0) | 3.50 ± 0.09 a | 3.37 | 1.73 ± 0.08 cd | 1.59 | 0.72 ± 0.10 a |
| 13 | 2 (0) | 2 (−1) | 150 (1) | 2.55 ± 0.08 gh | 2.59 | 2.10 ± 0.10 a | 1.97 | 0.55 ± 0.06 a |
| 14 | 3 (1) | 2 (−1) | 120 (0) | 2.39 ± 0.02 h | 2.27 | 2.06 ± 0.10 a | 2.06 | 0.63 ± 0.09 a |
| 15 | 2 (0) | 3 (0) | 120 (0) | 3.38 ± 0.04 ab | 3.37 | 1.48 ± 0.22 de | 1.59 | 0.62 ± 0.08 a |
| Regression Terms a | Extraction Yield (%, Y1) | PV (mM, Y2) | ||||
|---|---|---|---|---|---|---|
| Coefficient | F-Value | p-Value b | Coefficient | F-Value | p-Value b | |
| Model | 24.17 | 0.000 | 8.71 | 0.003 | ||
| Constant (β0) | −5.650 | 4.048 | ||||
| X1 (β1) | 2.186 | 4.04 | 0.079 | 0.088 | 3.71 | 0.080 |
| X2 (β2) | 1.691 | 23.57 | 0.001 | 1.620 | 8.75 | 0.013 |
| X3 (β3) | 0.070 | 0.93 | 0.363 | |||
| X1X1 (β11) | −0.567 | 91.80 | 0.000 | |||
| X2X2 (β22) | −0.249 | 17.76 | 0.003 | 0.247 | 13.66 | 0.004 |
| X3X3 (β33) | −0.0003 | 20.30 | 0.002 | |||
| R2 | 0.948 | 0.704 | ||||
| Adjusted R2 | 0.908 | 0.623 | ||||
| Lack of fit | 1.37 | 0.480 | 0.51 | 0.802 | ||
| AAD (%) | 2.760 | 5.581 | ||||
| Response Variables | Experimental Value | Predicted Value | p-Value a |
|---|---|---|---|
| Extraction yield (wt%) | 3.32 ± 0.03 | 3.38 | 0.154 |
| PV (mM) | 1.55 ± 0.02 | 1.54 | 0.147 |
| Criteria | Content |
|---|---|
| Fatty acid composition (% Total fatty acids) | |
| Saturated fatty acids | 33.10 ± 0.05 |
| Caprylic acid (C8:0) | 0.12 ± 0.01 |
| Lauric acid (C12:0) | 0.05 ± 0.00 |
| Myristic acid (C14:0) | 0.16 ± 0.00 |
| Palmitic acid (C16:0) | 26.0 ± 0.05 |
| Stearic acid (C18:0) | 6.78 ± 0.01 |
| Monounsaturated fatty acids (MUFA) | 37.82 ± 0.03 |
| Palmitoleic acid (C16:1) | 0.98 ± 0.00 |
| Oleic acid (C18:1) | 36.84 ± 0.03 |
| Polyunsaturated fatty acids (PUFA) | 29.09 ± 0.07 |
| Linoleic acid (18:2, n-6) | 4.25 ± 0.01 |
| Linolenic acid (18:3, n-3) | 24.85 ± 0.06 |
| Minor compounds | |
| Cholesterol (mg/100 g) | 108.66 ± 0.09 |
| β-Carotene (μg/100 g) | 784.89 ± 12.17 |
| α-Tocopherol (μg/100 g) | 9434.39 ± 367.95 |
| DPPH free radical scavenging activity (%) | 57.59 ± 0.93 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tangsanthatkun, J.; Peanparkdee, M.; Katekhong, W.; Harnsilawat, T.; Tan, C.P.; Klinkesorn, U. Application of Aqueous Saline Process to Extract Silkworm Pupae Oil (Bombyx mori): Process Optimization and Composition Analysis. Foods 2022, 11, 291. https://doi.org/10.3390/foods11030291
Tangsanthatkun J, Peanparkdee M, Katekhong W, Harnsilawat T, Tan CP, Klinkesorn U. Application of Aqueous Saline Process to Extract Silkworm Pupae Oil (Bombyx mori): Process Optimization and Composition Analysis. Foods. 2022; 11(3):291. https://doi.org/10.3390/foods11030291
Chicago/Turabian StyleTangsanthatkun, Janjira, Methavee Peanparkdee, Wattinee Katekhong, Thepkunya Harnsilawat, Chin Ping Tan, and Utai Klinkesorn. 2022. "Application of Aqueous Saline Process to Extract Silkworm Pupae Oil (Bombyx mori): Process Optimization and Composition Analysis" Foods 11, no. 3: 291. https://doi.org/10.3390/foods11030291
APA StyleTangsanthatkun, J., Peanparkdee, M., Katekhong, W., Harnsilawat, T., Tan, C. P., & Klinkesorn, U. (2022). Application of Aqueous Saline Process to Extract Silkworm Pupae Oil (Bombyx mori): Process Optimization and Composition Analysis. Foods, 11(3), 291. https://doi.org/10.3390/foods11030291







