Microbiological and Sensorial Quality of Beef Meat (Longissimus dorsi) Marinated with Cinnamon Extract and Stored at Various Temperatures
Abstract
1. Introduction
2. Materials and Methods
2.1. Cinnamon Extract Preparation (CE)
2.2. Anti-Pseudomonas Activity of the CE
2.2.1. Preparation of Bacterial Strains
2.2.2. Disc Diffusion Assay
2.2.3. Minimal Inhibitory and Minimal Bactericidal Concentration (MIC and MBC)
2.2.4. Time-Kill Analysis
2.2.5. Total Phenolic Content (TPC)
2.2.6. DPPH Radical Scavenging Method
2.3. CE treatment on Meat during Different Storage Temperatures
2.3.1. Meat Treatment
2.3.2. Microbiological Analysis
2.3.3. pH Determination
2.3.4. Sensory Evaluation
2.4. Statistical Analysis
3. Results and Discussion
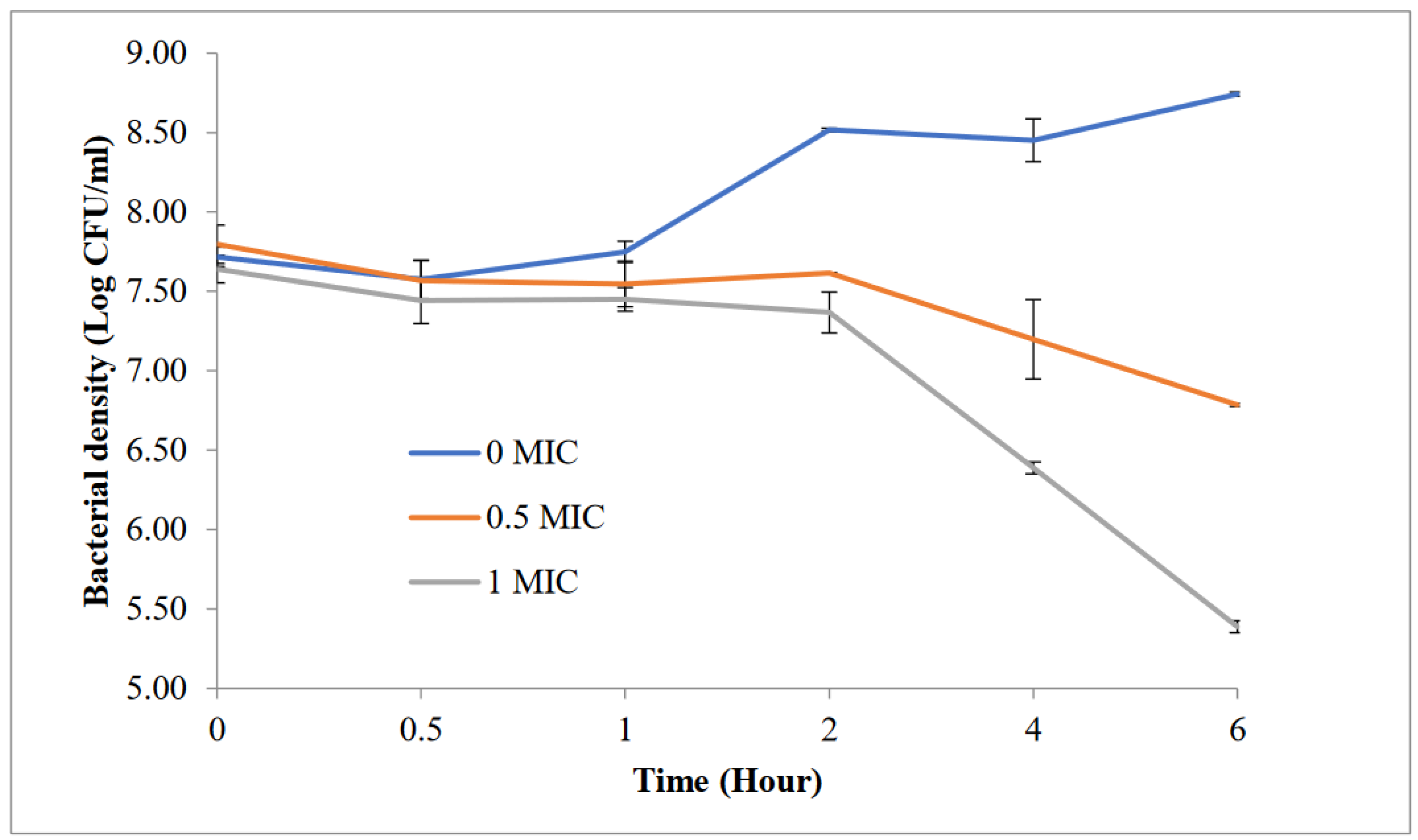
3.1. Anti-Pseudomonas Activity
3.2. Total Phenolic Content and Antioxidant Activity
3.3. Effect of CE Treatment on Meat during Different Storage Temperatures
3.3.1. Microbiological Changes
3.3.2. pH
3.3.3. Sensory Analyses
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bogataj, D.; Hudoklin, D.; Bogataj, M.; Dimovski, V.; Colnar, S. Risk mitigation in a meat supply chain with options of redirection. Sustainability 2020, 12, 8690. [Google Scholar] [CrossRef]
- Jiang, J.; Xiong, Y.L. Natural antioxidants as food and feed additives to promote health benefits and quality of meat products: A review. Meat Sci. 2016, 120, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, M.; Bertelsen, G. Colour stability and lipid oxidation of fresh beef. Development of a response surface model for predicting the effects of temperature, storage time, and modified atmosphere composition. Meat Sci. 2000, 54, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Ndraha, N.; Hsiao, H.I.; Vlajic, J.; Yang, M.F.; Lin, H.T.V. Time-temperature abuse in the food cold chain: Review of issues, challenges, and recommendations. Food Control 2018, 89, 12–21. [Google Scholar] [CrossRef]
- Koutsoumanis, K.; Stamatiou, A.; Skandamis, P.; Nychas, G.-J.E. Development of a microbial model for the combined effect of temperature and pH on spoilage of ground meat and validation of the model under dynamic temperature conditions. Appl. Environ. Microbiol. 2006, 72, 124–134. [Google Scholar] [CrossRef]
- Pennacchia, C.; Ercolini, D.; Villani, F. Spoilage-related microbiota associated with chilled beef stored in air or vakuum pack. Food Microb. 2011, 28, 84–93. [Google Scholar] [CrossRef]
- Shao, L.; Chen, S.; Wang, H.; Zhang, J.; Xu, X.; Wang, H. Advances in understanding the predominance, phenotypes, and mechanisms of bacteria related to meat spoilage. Trends Food Sci. Technol. 2021, 118, 822–832. [Google Scholar] [CrossRef]
- Wang, G.; Tang, W.; Ma, F.; Wang, H.; Xu, X.; Qiu, W. AprD is important for extracellular proteolytic activity, physicochemical properties and spoilage potential in meat-borne Pseudomonas fragi. Food Control 2021, 124, 107868. [Google Scholar] [CrossRef]
- Widders, P.R.; Coates, K.J.; Warner, S.; Beattie, J.C.; Morgan, I.R.; Hickey, M.W. Controlling microbial contamination on beef and lamb meat during processing. Aust. Vet. J. 1995, 72, 208–211. [Google Scholar] [CrossRef]
- Ercolini, D.; Russo, F.; Nasi, A.; Ferranti, P.; Villani, F. Mesophilic and psychrotrophic bacteria from meat and their spoilage potential in vitro and in beef. Appl. Environ. Microbiol. 2009, 75, 1990–2001. [Google Scholar] [CrossRef]
- Oswell, N.J.; Thippareddi, H.; Pegg, R.B. Practical use of natural antioxidants in meat products in the US: A review. Meat Sci. 2018, 145, 469–479. [Google Scholar] [CrossRef]
- Su, M.S.; Shyu, Y.T.; Chien, P.J. Antioxidant activities of citrus herbal product extracts. Food Chem. 2008, 111, 892–896. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). Current Market Situation and Medium-Term Outlook. Paper Presented at the Seventeenth Session of the Intergovernmental Group on Tea, 29 November–1 December 2006. Available online: http://www.fao.org (accessed on 28 October 2020).
- Lucera, A.; Costa, C.; Conte, A.; Del Nobile, M.A. Food applications of natural antimicrobial compounds. Front. Microbiol. 2012, 3, 287. [Google Scholar] [CrossRef]
- Das, A.K.; Anjaneyulu, A.S.R.; Biswas, S. Effect of carnosine preblending on the quality of ground buffalo meat. Food Chem. 2006, 97, 531–538. [Google Scholar] [CrossRef]
- Batiha, G.E.S.; Hussein, D.E.; Algammal, A.M.; George, T.T.; Jeandet, P.; Al-Snafi, A.E.; Tiwari, A.; Pagnossa, J.P.; Lima, C.M.; Thorat, N.D.; et al. Application of natural antimicrobials in food preservation: Recent views. Food Control 2021, 126, 108066. [Google Scholar] [CrossRef]
- Aksu, M.I.; Kaya, M. The effect of α-tocopherol and butylated hydroxyanisole on the colour properties and lipid oxidation of kavurma, a cooked meat product. Meat Sci. 2005, 71, 277–283. [Google Scholar] [CrossRef]
- Lee, R.; Balick, M.J. Sweet wood–cinnamon and its importance as a spice and medicine. Explore 2005, 1, 61–64. [Google Scholar] [CrossRef]
- Boughendjioua, H.; Djeddi, S. Study of The Organoleptic and Physicochemical Properties of Cinnamon Essential Oil (Cinnamomum zeylanicum). Am. J. Life Sci. Res. 2018, 6, 123–130. [Google Scholar]
- Singh, G.; Maurya, S.; deLampasona, M.P.; Catalan, C.A.N. A comparison of chemical, antioxidant and antimicrobial studies of cinnamon leaf and bark volatile oils, oleoresins and their constituents. Food Chem. Toxicol. 2007, 45, 1650–1661. [Google Scholar] [CrossRef]
- Lu, Z.; Jia, Q.; Wang, R.; Wu, X.; Wu, Y.; Huang, C.; Li, Y. Hypoglycemic activities of A-and B-type procyanidin oligomer-rich extracts from different Cinnamon barks. Phytomedicine 2011, 18, 298–302. [Google Scholar] [CrossRef]
- Johny, A.K.; Darre, M.J.; Hoagland, T.A.; Schreiber, D.T.; Donoghue, A.M.; Donoghue, D.J.; Venkitanarayanan, K. Antibacterial effect of trans-cinnamaldehyde on Salmonella enteritidis and Campylobacter jejuni in chicken drinking water. J. Appl. Poult. Res. 2008, 17, 490–497. [Google Scholar] [CrossRef]
- Mishra, A.; Bhatti, R.; Singh, A.; Singh Ishar, M.P. Ameliorative effect of the cinnamon oil from Cinnamomum zeylanicum upon early stage diabetic nephropathy. Planta Med. 2009, 76, 412–417. [Google Scholar] [CrossRef]
- Nassar-Abbas, S.M.; Halkman, K. Antimicrobial effect of water extract of sumac (Rhus coriaria L.) on the growth of some food borne bacteria including pathogens. Int. J. Food Microbiol. 2004, 97, 63–69. [Google Scholar] [CrossRef]
- Performance Standards for Antimicrobial Susceptibility Testing; Twenty-second Informational Supplement; Clinical and Laboratory Standards Institute (CLSI): Wayne, PA, USA, 2013.
- Ismaili, H.; Milella, L.; Fkih-Tetouani, S.; Ilidrissi, A.; Camporese, A.; Sosa, S.; Altinier, G.; Della Loggia, R.; Aquino, R. In vivo topical anti-inflammatory and in vitro antioxidant activities of two extracts of Thymus satureioides leaves. J. Ethnopharmacol. 2004, 91, 31–36. [Google Scholar] [CrossRef]
- Rukayadi, Y.; Han, S.; Yong, D.; Hwang, J.K. In vitro antibacterial activity of panduratin A against enterococci clinical isolates. Biol. Pharm. Bull. 2010, 33, 1489–1493. [Google Scholar] [CrossRef]
- MacFie, H.J.; Bratchell, N.; Greenhoff, K.; Vallis, L.V. Designs to balance the effect of order of presentation and first-order carry-over effects in hall tests. J. Sens. Stud. 1989, 4, 129–148. [Google Scholar] [CrossRef]
- ISO 8589; Sensory Analysis—General Guidance for the Design of Test Rooms. International Organization for Standardization: Geneva, Switzerland, 1988. Available online: http://www.iso.org (accessed on 15 July 2021).
- Weerakkody, N.S.; Caffin, N.; Turner, M.S.; Dykes, G.A. In vitro antimicrobial activity of less-utilized spice and herb extracts against selected food-borne bacteria. Food Control 2010, 21, 1408–1414. [Google Scholar] [CrossRef]
- Norhana, M.W.; Poole, S.E.; Deeth, H.C.; Dykes, G.A. Effects of bilimbi (Averrhoa bilimbi L.) and tamarind (Tamarindus indica L.) juice on Listeria monocytogenes Scott A and Salmonella Typhimurium ATCC 14028 and the sensory properties of raw shrimps. Int. J. Food Microbiol. 2009, 136, 88–94. [Google Scholar] [CrossRef]
- Pandey, S.; Pandey, R.; Singh, R. Phytochemical screening of selected medicinal plant cinnamon zeylanicum bark extract, area of research; uttarakhand, India. Int. J. Sci. Res. Publ. 2014, 4, 1–6. [Google Scholar]
- Hacioglu, M.; Dosler, S.; Tan, A.S.B.; Otuk, G. Antimicrobial activities of widely consumed herbal teas, alone or in combination with antibiotics: An in vitro study. PeerJ 2017, 5, e3467. [Google Scholar] [CrossRef]
- Gilani, S.; Najafpour, G. Evaluation of the extraction process parameters on bioactive compounds of cinnamon bark: A comparative study. Process Biochem. 2022, 114, 93–101. [Google Scholar] [CrossRef]
- Sana, S.; Arshad, M.U.; Farhan, S.; Ahmad, R.; Ali, I.; Tabussam, T. Nutritional characterization of cinnamon and turmeric with special reference to their antioxidant profile. Int. J. Biosci. 2019, 15, 178–187. [Google Scholar]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Madsen, H.L.; Bertelsen, G. Spices as antioxidants. Trends Food Sci. Technol. 1995, 6, 271–277. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Sharma, A.; Baek, K.H. Antibacterial mode of action of Cudrania tricuspidata fruit essential oil, affecting membrane permeability and surface characteristics of food-borne pathogens. Food Control 2013, 32, 582–590. [Google Scholar] [CrossRef]
- Gutierrez, J.; Barry-Ryan, C.; Bourke, P. The antimicrobial efficacy of plant essential oil combinations and interactions with food ingredients. Int. J. Food Microbiol. 2008, 124, 91–97. [Google Scholar] [CrossRef]
- González-Fandos, E.; Herrera, B.; Maya, N. Efficacy of citric acid against Listeria monocytogenes attached to poultry skin during refrigerated storage. Int. J. Food Sci. Technol. 2009, 44, 262–268. [Google Scholar] [CrossRef]
- Hassan, M.A.; Ibrahim, H.M.; Shawky, N.A.; Sheir, S.H. Incidence of Psychotropic bacteria in frozen chicken meat products with special reference to Pseudomonas species. Benha Vet. Med. J. 2020, 39, 165–168. [Google Scholar]
- Elbehiry, A.; Marzouk, E.; Aldubaib, M.; Moussa, I.; Abalkhail, A.; Ibrahem, M.; Hamada, M.; Sindi, W.; Alzaben, F.; Almuzaini, A.M.; et al. Pseudomonas species prevalence, protein analysis, and antibiotic resistance: An evolving public health challenge. AMB Express 2022, 12, 53. [Google Scholar] [CrossRef]
- Zhao, Y.; Wells, J.H.; McMillin, K.W. Applications of dynamic modified atmosphere packaging systems for fresh red meats: Review. J. Muscle Foods 1994, 5, 299–328. [Google Scholar] [CrossRef]
- Jamilah, M.B.; Abbas, K.A.; Rahman, R.A. A Review on Some Organic Acids Additives as Shelf Life Extenders of Fresh Beef Cuts. Am. J. Agric. Biol. Sci. 2008, 3, 556–574. [Google Scholar] [CrossRef][Green Version]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Bouhdid, S.; Abrini, J.; Amensour, M.; Zhiri, A.; Espuny, M.J.; Manresa, A. Functional and ultrastructural changes in Pseudomonas aeruginosa and Staphylococcus aureus cells induced by Cinnamomum verum essential oil. J. Appl. Microbiol. 2010, 109, 1139–1149. [Google Scholar] [CrossRef]
- Shange, N.; Makasi, T.; Gouws, P.; Hoffman, L.C. Preservation of previously frozen black wildebeest meat (Connochaetes gnou) using oregano (Oreganum vulgare) essential oil. Meat Sci. 2019, 148, 88–95. [Google Scholar] [CrossRef]
- Hussain, Z.; Li, X.; Zhang, D.; Hou, C.; Ijaz, M.; Bai, Y.; Xiao, X.; Zheng, X. Influence of adding cinnamon bark oil on meat quality of ground lamb during storage at 4 C. Meat Sci. 2021, 171, 108269. [Google Scholar] [CrossRef]
- Van Haute, S.; Raes, K.; Van Der Meeren, P.; Sampers, I. The effect of cinnamon, oregano and thyme essential oils in marinade on the microbial shelf life of fish and meat products. Food Control 2016, 68, 30–39. [Google Scholar] [CrossRef]
- Putra, A.A.; Wattanachant, S.; Wattanachant, C. Sensory-related attributes of raw and cooked meat of culled Saanen goat marinated in ginger and pineapple juices. Trop. Anim. Sci. J. 2019, 42, 59–67. [Google Scholar] [CrossRef]
- García-Casal, M.N.; Peña-Rosas, J.P.; Malavé, H. Sauces, spices, and condiments: Definitions, potential benefits, consumption patterns, and global markets. Ann. N. Y. Acad. Sci. 2016, 1379, 3–16. [Google Scholar] [CrossRef]
| Cinnamon Extract Concentration (%) | Zone of Inhibition (mm) |
|---|---|
| 20 | 7.75 ± 0.42 a |
| 40 | 8.83 ± 0.42 b |
| 60 | 9.67 ± 0.82 bc |
| 80 | 10.50 ± 0.84 cd |
| 100 | 13.50 ± 0.84 d |
| Control (Gentamicin) | 15.00 ± 0.32 e |
| Time (h) | 5 °C | 10 °C | 15 °C | 25 °C | ||||
|---|---|---|---|---|---|---|---|---|
| Marinated | Control | Marinated | Control | Marinated | Control | Marinated | Control | |
| 0 | 5.31 ± 0.51 Aa | 5.84 ± 0.06 Ba | 5.31 ± 0.51 Aa | 5.84 ± 0.06 Ba | 5.31 ± 0.51 Aa | 5.84 ± 0.06 Ba | 5.31 ± 0.51 Aa | 5.84 ± 0.06 Ba |
| 6 | 6.52 ± 1.28 Ab | 5.80 ± 0.08 Ba | 5.47 ± 0.70 Aa | 6.81 ± 0.04 Bb | 5.65 ± 0.77 Aab | 7.78 ± 0.00 Bb | 6.68 ± 0.30 Ab | 7.88 ± 0.27 Bb |
| 12 | 6.60 ± 1.00 Ab | 6.54 ± 0.06 Ab | 6.26 ± 0.27 Abc | 7.37 ± 0.06 Bc | 5.99 ± 0.65 Ab | 7.85 ± 0.03 Bb | 7.90 ± 0.17 Ac | 8.08 ± 0.12 Bbc |
| 18 | 6.29 ± 0.89 Ab | 6.28 ± 0.09 Ac | 6.09 ± 0.97 Ab | 7.34 ± 0.07 Bcd | 6.96 ± 0.27 Ac | 8.06 ± 0.06 Bbc | 8.36 ± 0.07 Ad | 8.58 ± 0.35 Ac |
| 24 | 6.74 ± 0.56 Abc | 7.22 ± 0.13 Bd | 6.22 ± 0.75 Abc | 7.52 ± 0.11 Bcd | 7.35 ± 0.22 Acd | 8.97 ± 0.04 Bc | 8.43 ± 0.30 Ad | 8.84 ± 0.03 Ad |
| 36 | 6.83 ± 0.61 Ac | 7.57 ± 0.01 Bde | 6.62 ± 0.79 Ac | 7.73 ± 0.15 Bd | 7.71 ± 0.28 Ad | 8.50 ± 0.10 Bbc | 8.62 ± 0.25 Ad | 8.87 ± 0.03 Ad |
| 48 | 6.61 ± 0.54 Ac | 7.64 ± 0.11 Be | 6.60 ± 0.64 Ac | 8.18 ± 0.01 Be | 8.23 ± 0.19 Ae | 8.94 ± 0.09 Bc | 9.17 ± 0.17 Ae | 9.19 ± 0.04 Ae |
| Time (h) | 5 °C | 10 °C | 15 °C | 25 °C | ||||
|---|---|---|---|---|---|---|---|---|
| Marinated | Control | Marinated | Control | Marinated | Control | Marinated | Control | |
| 0 | 4.24 ± 0.69 Aa | 4.96 ± 0.60 Aa | 4.24 ± 0.69 Aa | 4.96 ± 0.60 Ba | 4.24 ± 0.69 Aa | 4.96 ± 0.60 Aa | 4.24 ± 0.69 Aa | 4.96 ± 0.60 Aa |
| 6 | 3.79 ± 0.62 Aa | 5.12 ± 0.01 Ba | 4.09 ± 0.91 Aa | 5.97 ± 0.02 Bb | 4.13 ± 0.84 Aa | 4.65 ± 0.12 Aa | 4.78 ± 0.11 Aa | 5.50 ± 0.15 Ba |
| 12 | 4.38 ± 0.28 Aab | 5.07 ± 0.01 Ba | 4.82 ± 0.17 Aab | 6.92 ± 0.04 Bc | 4.66 ± 0.94 Aab | 6.73 ± 0.10 Bb | 5.94 ± 0.16 Ab | 6.52 ± 0.12 Bb |
| 18 | 4.99 ± 0.61 Abc | 5.30 ± 0.00 Aab | 4.41 ± 1.02 Aab | 7.01 ± 0.05 Bc | 5.30 ± 0.13 Ab | 6.49 ± 0.10 Bb | 7.21 ± 0.43 Ac | 6.90 ± 0.35 Ab |
| 24 | 5.37 ± 0.18 Ac | 5.28 ± 0.03 Ab | 4.87 ± 0.57 Aab | 7.20 ± 0.04 Bcd | 6.18 ± 0.22 Ac | 8.15 ± 0.05 Bcd | 7.85 ± 0.43 Acd | 7.92 ± 0.03 Ac |
| 36 | 4.89 ± 0.18 Abc | 5.79 ± 0.01 Bc | 4.14 ± 1.85 Aab | 7.21 ± 0.18 Bd | 6.66 ± 0.09 Ac | 8.03 ± 0.03 Bc | 8.35 ± 0.48 Ad | 8.11 ± 0.03 Acd |
| 48 | 5.39 ± 0.22 Ac | 5.86 ± 0.02 Bc | 5.94 ± 0.42 Ac | 7.36 ± 0.00 Bd | 7.46 ± 0.32 Ad | 8.12 ± 0.10 bd | 8.80 ± 0.25 Ad | 8.20 ± 0.04 Bd |
| Storage Time (h) | 5 °C | 10 °C | 15 °C | 25 °C |
|---|---|---|---|---|
| 0 | 6.10 ± 0.09 a | 6.10 ± 0.09 a | 6.10 ± 0.09 a | 6.10 ± 0.09 a |
| 6 | 5.98 ± 0.10 ab | 6.15 ± 0.21 a | 6.32 ± 0.40 ab | 6.06 ± 0.30 a |
| 12 | 6.10 ± 0.18 a | 6.19 ± 0.30 a | 6.20 ± 0.26 a | 5.93 ± 0.32 a |
| 18 | 6.05 ± 0.19 a | 5.95 ± 0.14 ab | 5.90 ± 0.13 a | 5.74 ± 0.53 a |
| 24 | 5.90 ± 0.07 ab | 6.06 ± 0.12 ab | 6.05 ± 0.15 a | 5.87 ± 0.61 a |
| 36 | 5.86 ± 0.08 b | 6.03 ± 0.12 ab | 5.96 ± 0.26 a | 6.02 ± 0.50 ab |
| 48 | 5.90 ± 0.12 ab | 6.11 ± 0.23 a | 6.05 ± 0.32 a | 6.48 ± 0.39 b |
| Sample | Odour | Colour | Texture | Overall Acceptance |
|---|---|---|---|---|
| Cinnamon-marinated meat | 4.617 ± 1.460 a | 4.233 ± 0.989 b | 4.183 ± 1.441 b | 4.450 ± 1.177 b |
| Non-marinated meat | 4.883 ± 1.127 a | 5.550 ± 1.117 a | 4.967 ± 1.224 a | 5.183 ± 0.966 a |
| p-value | 0.432 | 0.000 | 0.027 | 0.012 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zawani, C.J.; Nor-Khaizura, M.A.R.; Mahyudin, N.A.; Ismail-Fitry, M.R.; Nirmal, N.P. Microbiological and Sensorial Quality of Beef Meat (Longissimus dorsi) Marinated with Cinnamon Extract and Stored at Various Temperatures. Foods 2022, 11, 3971. https://doi.org/10.3390/foods11243971
Zawani CJ, Nor-Khaizura MAR, Mahyudin NA, Ismail-Fitry MR, Nirmal NP. Microbiological and Sensorial Quality of Beef Meat (Longissimus dorsi) Marinated with Cinnamon Extract and Stored at Various Temperatures. Foods. 2022; 11(24):3971. https://doi.org/10.3390/foods11243971
Chicago/Turabian StyleZawani, Che Jaafar, Mahmud Ab Rashid Nor-Khaizura, Nor Ainy Mahyudin, Mohammad Rashedi Ismail-Fitry, and Nilesh Prakash Nirmal. 2022. "Microbiological and Sensorial Quality of Beef Meat (Longissimus dorsi) Marinated with Cinnamon Extract and Stored at Various Temperatures" Foods 11, no. 24: 3971. https://doi.org/10.3390/foods11243971
APA StyleZawani, C. J., Nor-Khaizura, M. A. R., Mahyudin, N. A., Ismail-Fitry, M. R., & Nirmal, N. P. (2022). Microbiological and Sensorial Quality of Beef Meat (Longissimus dorsi) Marinated with Cinnamon Extract and Stored at Various Temperatures. Foods, 11(24), 3971. https://doi.org/10.3390/foods11243971








