Impact of Green Extraction on Curcuminoid Content, Antioxidant Activities and Anti-Cancer Efficiency (In Vitro) from Turmeric Rhizomes (Curcuma longa L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Extraction Methods
2.3. Physical and Chemical Analysis and Yield of Extraction
2.4. Antioxidant Activities: Total Phenolic Content (TPC), DPPH and FRAP
2.5. Analysis of Curcuminoid Content Using High Pressure Liquid Chromatograph (HPLC)
2.6. Human Cell Culture
2.7. Cell Viability Assay
2.8. Statistical Analysis
3. Results
3.1. Proximate Composition of Dried Turmeric
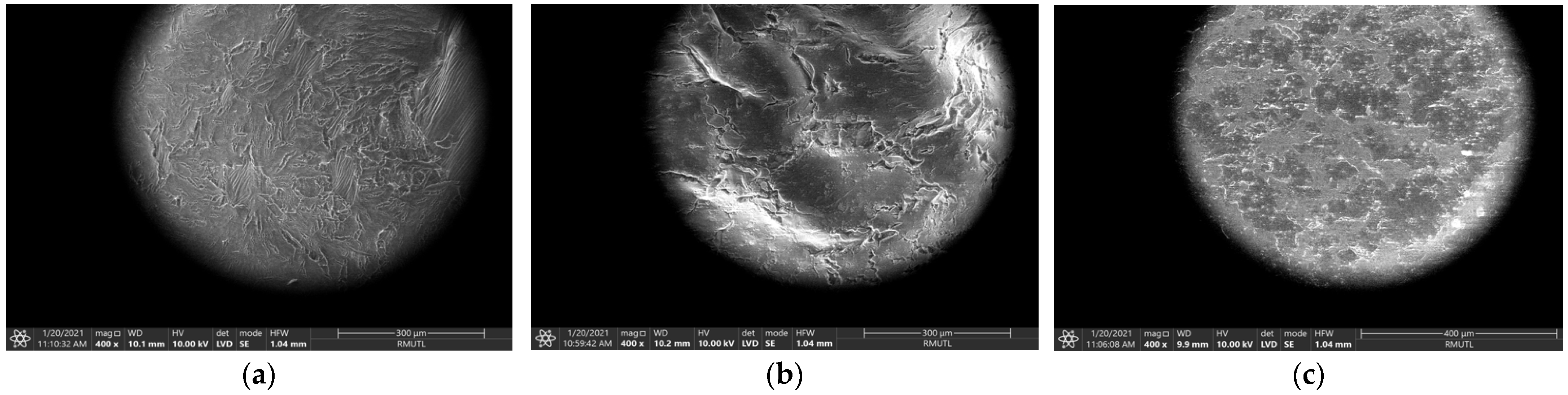
3.2. Effect of Ultrasonic-Assisted Extraction (UAE)
3.3. Effect of Microwave-Assisted Extraction (MAE)
3.4. Effect of Traditional Extraction (TDE)
3.5. Comparison of UAE, MAE and TDE
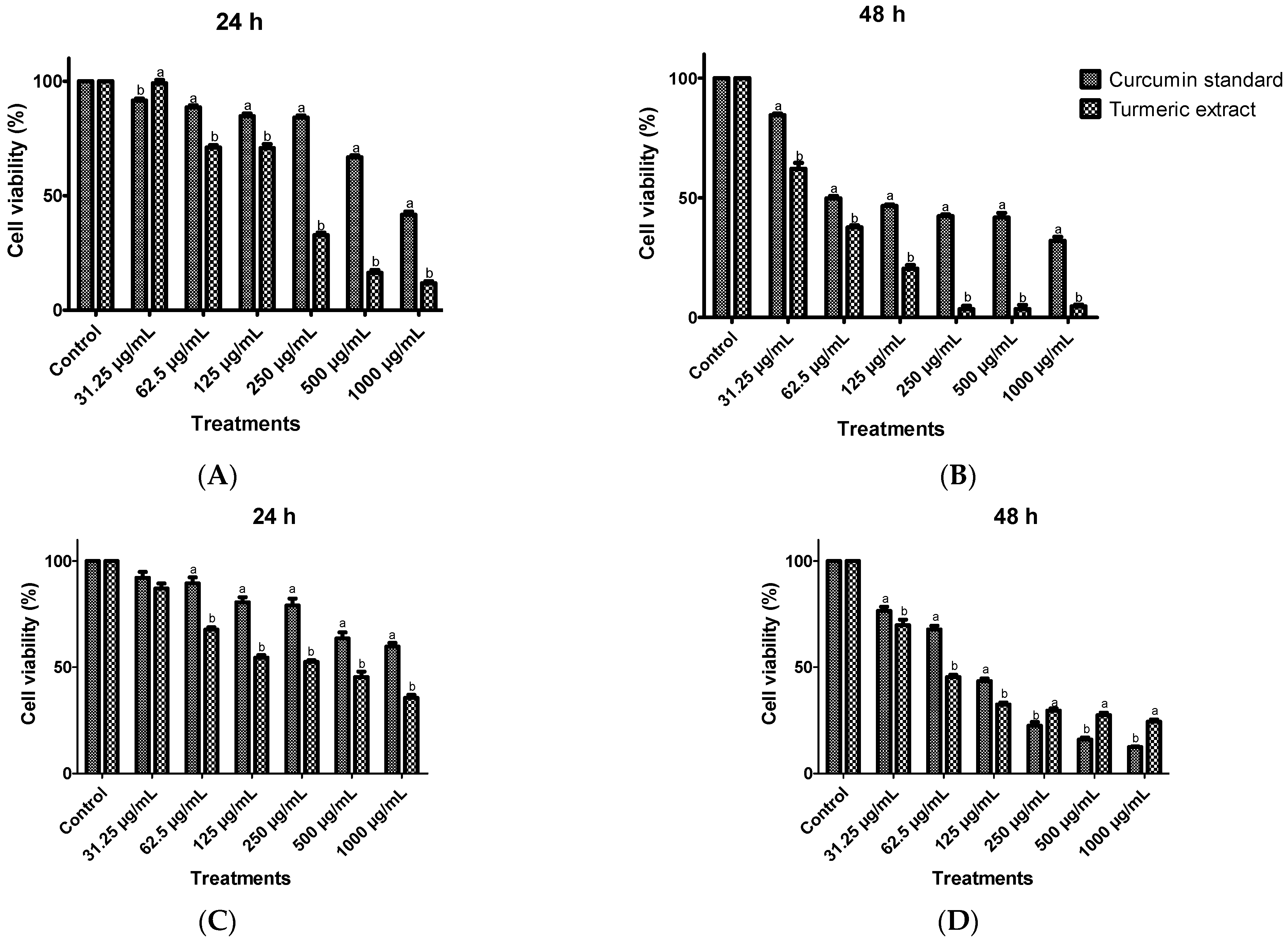
3.6. Anticancer Activity from Turmeric Extracts on Huh7 Cells and HCT116 Cells Using Cell Viability Assay
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Deserti, M.; Tavolari, S.; Naldi, M.; Frega, G.; Brandi, G. Antitumoral Efficacy of Two Turmeric Extracts According to Different Extraction Methods in Hepatocellular Carcinoma Cell Lines. J. Med. Aromat. 2018, 7, 2167. [Google Scholar] [CrossRef]
- De Oliveira, A.C.; Miyagawa, L.M.; Monteiro, K.M.; Dias, A.L.; Longato, G.B.; Spindola, H.; Vendramini-Costa, D.B.; Quetin-Leclercq, J.; Carvalho, J.E.; Rogez, H. Phenolic Composition, Antiproliferative and Antiulcerogenic Activities of a Polyphenol-Rich Purified Extract from Acai (Euterpe oleracea) Fruits. Int. J. Food Sci. technol. 2021, 56, 6626–6634. [Google Scholar] [CrossRef]
- Garra, A.; Alkalai-Tuvia, S.; Telerman, A.; Paran, I.; Fallik, E.; Elmann, A. Anti-proliferative Activities, Phytochemical Levels and Fruit Quality of Pepper (Capsicum spp.) Following Prolonged Storage. Int. J. Food Sci. Technol. 2020, 55, 3574–3584. [Google Scholar] [CrossRef]
- Thumbrain, D.; Dwarka, D.; Gerrano, A.S.; Mellem, J.J. Antioxidant and Apoptotic Potential of Protein Isolates Derived from Vigna unguiculata (L.) Walp. Int. J. Food Sci. Technol. 2020, 55, 2813–2823. [Google Scholar] [CrossRef]
- Hemmati, V.; Garavand, F.; Goudarzi, M.; Sarlak, Z.; Cacciotti, I.; Tiwari, B.K. Cold Atmospheric-Pressure Plasma Treatment of Turmeric Powder: Microbial Load, Essential Oil Profile, Bioactivity and Microstructure Analyses. Int. J. Food Sci. Technol. 2020, 56, 2224–2232. [Google Scholar] [CrossRef]
- Ravindran, J.; Prasad, S.; Aggarwal, B.B. Curcumin and Cancer Cells: How Many Ways Can Curry Kill Tumor Cells Selectively? AAPS J. 2009, 11, 495–510. [Google Scholar] [CrossRef]
- Golchinfar, Z.; Tabibiazar, M.; Abdi, F.; Taghvimi, A.; Roufegarinejad, L. Effect of Resveratrol and Curcumin on Formation of N-Carboxymethyl Lysine and Its Intracellular Oxidative Stress. Int. J. Food Sci. Technol. 2022, 57, 6903–6912. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, J.; Yu, J.; Chen, X.; Zhang, S.; Cai, Y.; Li, L. Curcumin As a Mild Natural α-Glucosidase Inhibitor: A Study on Its Mechanism In Vitro. Int. J. Food Sci. Technol. 2022, 57, 2689–2700. [Google Scholar] [CrossRef]
- Ahmad, R.S.; Hussain, M.B.; Sultan, M.T.; Arshad, M.S.; Waheed, M.; Shariati, M.A.; Plygun, S.; Hashempur, M.M. Biochemistry, Safety, Pharmacological Activities, and Clinical Applications of Turmeric: Mechanistic Review. Hindawi 2020, 2020, 7656919. [Google Scholar] [CrossRef]
- Wakte, P.S.; Sachin, B.S.; Patil, A.A.; Mohato, D.M.; Band, T.H.; Shinde, D.B. Optimization of Microwave, Ultrasonic and Supercritical Carbondioxide Assisted Extraction Techniques for Curcumin from Curcuma longa. Sep. Purif. Technol. 2011, 46, 50–55. [Google Scholar] [CrossRef]
- Vetal, M.D.; Lade, V.G.; Rathod, V.K. Extraction of Ursolic Acid from Ocimum Sanctum by Ultrasound: Process Intensification and Kinetic Studies. Chem. Eng. Process. 2013, 69, 24–30. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, M.; Lin, L. Effects of Extraction Methods on Structural Characteristics and Bile Acid-Binding Capacities of Moringa Oleifera Leaf Polysaccharide Fractions. Int. J. Food Sci. Technol. 2020, 55, 1539–1546. [Google Scholar] [CrossRef]
- Zhao, Y.; Tran, K.; Brennan, M.; Brennan, C. Kinetics of Ultrasonic Extraction of Polyphenols, Anthocyanins and Tannins from Five Different New Zealand Grape Pomaces. Int. J. Food Sci. Technol. 2021, 56, 2687–2695. [Google Scholar] [CrossRef]
- Machado, A.P.D.F.; Sumere, B.R.; Mekaru, C.; Martinez, J.; Bezerra, R.M.N.; Rostagno, M.A. Extraction of Polyphenols and Antioxidants from Pomegranate Peel Using Ultrasound: Influence of Temperature, Frequency and Operation Mode. Int. J. Food Sci. Technol. 2019, 54, 2792–2801. [Google Scholar] [CrossRef]
- Cravotto, G.; Mariatti, F.; Gunjevic, V.; Secondo, M.; Villa, M.; Parolin, J.; Cavaglià, G. Pilot Scale Cavitational Reactors and Other Enabling Technologies to Design the Industrial Recovery of Polyphenols from Agro-Food By-Products, a Technical and Economical Overview. Foods 2018, 7, 130. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Ngadi, M.O.; Ma, Y. Optimisation of Pulsed Ultrasonic and Microwave-Assisted Extraction for Curcuminoids by Response Surface Methodology and Kinetic Study. Food Chem. 2014, 165, 29–34. [Google Scholar] [CrossRef]
- Chumroenphata, T.; Somboonwatthanakula, I.; Saensoukb, S.; Siriamornpunc, S. Changes in Curcuminoids and Chemical Components of Turmeric (Curcuma longa L.) under Freeze-drying and Low-temperature Drying Methods. Food Chem. 2021, 339, 128121. [Google Scholar]
- Shirsath, S.R.; Sable, S.S.; Sonawane, S.H.; Saini, D.R.; Gogate, P.R. Intensification of Extraction of Curcumin from Curcuma amanda using Ultrasound Assisted Approach: Effect of Different Operating Parameters. Ultrason. Sonochem. 2017, 38, 437–445. [Google Scholar] [CrossRef]
- Singh, K.; Srichairatanakool, S.; Chewonarin, T.; Brennan, C.S.; Brennan, M.A.; Klangpetch, W.; Utama-ang, N. Manipulation of the Phenolic Quality of Assam Green Tea through Thermal Regulation and Utilization of Microwave and Ultrasonic Extraction Techniques. Horticulturae 2022, 8, 338. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis; Association of Official Analytical Chemists: Washington, DC, USA, 1975. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Singh, K.; Simapaisan, P.; Utama-ang, N. Effect of Microwave-Assisted Extraction on Curcuminoid from Turmeric and Application in Germinate-Coated Rice. Food Appl. Biosci. 2017, 5, 11–22. [Google Scholar]
- Jaganathan, S.K.; Supriyanto, E.; Mandal, M. Events Associated with Apoptotic Effect of P-Coumaric Acid in HCT-15 Colon Cancer Cells. World J. Gastroenterol. 2013, 19, 7726. [Google Scholar] [CrossRef] [PubMed]
- Sahne, F.; Mohammadi, M.; Najafpour, G.D.; Moghadamnia, A.A. Extraction of Bioactive Compound Curcumin from Turmeric (Curcuma longa L.) Via Different Routes: A Comparative Study. Pakistan J. Biotechnol. 2016, 13, 173–180. [Google Scholar]
- Yadav, P.; Jadhav, S.E.; Kumar, V.; Kaul, K.K.; Pant, S.C.; Flora, S.J. Protective Efficacy of 2-PAMCl, Atropine and Curcumin Against Dichlorvos Induced Toxicity in Rats. Interdiscip. Toxicol. 2012, 5, 1–8. [Google Scholar] [CrossRef]
- Patil, S.S.; Bhasarkar, S.; Rathod, V.K. Extraction of Curcuminoids from Curcuma Longa: Comparative Study Between Batch Extraction and Novel Three Phase Partitioning. Prep. Biochem. Biotechnol. 2019, 49, 407–418. [Google Scholar] [CrossRef]
- Kulkarni, S.K.; Bhutani, M.K.; Bishnoi, M. Antidepressant Activity of Curcumin: Involvement of Serotonin and Dopamine System. Psychopharmacology 2008, 201, 435–442. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, H.; Zhang, M.; Wang, Z. Improved Extraction of Oil from Chickpea under Ultrasound in a Dynamic System. J. Food Eng. 2010, 98, 13–18. [Google Scholar] [CrossRef]
- Swamy, K.M.; Narayana, K.L. Intensification of Leaching Process by Dual-Frequency Ultrasound. Ultrason. Sonochem. 2001, 8, 341–346. [Google Scholar] [CrossRef]
- Dey, S.; Rathod, V.K. Ultrasound Assisted Extraction of β-Carotene from Spirulina platensis. Ultrason. Sonochem. 2013, 20, 271–276. [Google Scholar] [CrossRef]
- Laokuldilok, N.; Kopermsub, P.; Thakeow, P.; Utama-ang, N. Microwave Assisted Extraction of Bioactive Compounds from Turmeric (Curcuma longa). Int. J. Agric. Technol. 2015, 11, 1185–1196. [Google Scholar]
- Dandekar, D.V.; Gaikar, V. Microwave Assisted Extraction of Curcuminoids from Curcuma Longa. Sep. Sci. Technol. 2002, 37, 2669–2690. [Google Scholar] [CrossRef]
- Chhouk, K.; Quitain, A.T.; Pag-asa, D.G.; Maridable, J.B.; Sasaki, M.; Shimoyama, Y.; Goto, M. Supercritical Carbon Dioxide-Mediated Hydrothermal Extraction of Bioactive Compounds from Garcinia Mangostana pericarp. J. Supercrit. Fluids 2016, 110, 167–175. [Google Scholar] [CrossRef]
- Yeong, Y.L.; Pang, S.F.; Chong, S.Y.; Gimbun, J. Comparison of Microwave and Ultrasonic Assisted Extraction of Kaempferol from Cassia Alata. Int. J. Eng. Sci. 2018, 7, 84–89. [Google Scholar]
- Mandal, V.; Mohan, Y.; Hemalatha, S. Microwave Assisted Extraction of Curcumin by Sample-Solvent Dual Heating Mechanism Using Taguchi L9 Orthogonal Design. J. Pharm. Biomed. Anal. 2008, 46, 322–327. [Google Scholar] [CrossRef]
- Subramani, A.P.; Panati, K.; Lebaka, V.R.; Reddy, R.R.; Narala, V.R. Nanostructures for Curcumin Delivery Posibilities and Challenges. J. Nanobiotechnol. 2017, 21, 393–418. [Google Scholar]
- Chuang, C.E.; Cheng, C.L.; Lin, J.K.; Kua, M.L. Inhibition by Curcumin of Diethylnitrosamine-Induced Hepatic Hyperplasia, InFlammatation, Cellular Gene Products and Cell-Cycle-Related Proteins in Rats. Food Chem Toxicol. 2000, 38, 991–995. [Google Scholar] [CrossRef]
- Jager, R.; Lowery, P.; Calvanese, A.V.; Joy, J.M.; Purpuna, M.; Wilson, J. Comparative Absorption of Curcumin Formulations. Nutr. J. 2014, 13, 11. [Google Scholar] [CrossRef]


| Extraction Technique | Extraction Yield (%, DW) | TPC (mgGAE/g DW) | DPPH (mgGAE/g DW) | FRAP (mg Fe (II)/g DW) |
|---|---|---|---|---|
| Turmeric powder | - | 41.93 ± 4.28 d | 17.77 ± 3.90 d | 25.34 ± 5.43 d |
| MAE | 17.89 ± 1.43 a | 178.36 ± 4.76 a | 72.54 ± 4.70 a | 82.11 ± 6.09 a |
| UAE | 11.34 ± 2.48 b | 112.50 ± 8.46 b | 64.32 ± 3.64 b | 77.82 ± 4.58 b |
| TDE | 5.54 ± 0.98 c | 97.14 ± 10.04 c | 60.07 ± 4.87 b | 72.72 ± 5.45 b |
| Extraction Technique | C (mg/g DW) | DMC (mg/g DW) | BDMC (mg/g DW) |
|---|---|---|---|
| Turmeric powder | 50.43 ± 5.51 d | 26.03 ± 1.56 d | 11.76 ± 2.88 d |
| MAE | 186.64 ± 9.73 a | 95.12 ± 1.45 a | 45.03 ± 1.02 a |
| UAE | 117.44 ± 5.94 b | 83.54 ± 3.64 b | 40.19 ± 8.73 b |
| TDE | 95.57 ± 6.65 c | 81.70 ± 2.40 b | 38.56 ± 6.91 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, K.; Srichairatanakool, S.; Chewonarin, T.; Prommaban, A.; Samakradhamrongthai, R.S.; Brennan, M.A.; Brennan, C.S.; Utama-ang, N. Impact of Green Extraction on Curcuminoid Content, Antioxidant Activities and Anti-Cancer Efficiency (In Vitro) from Turmeric Rhizomes (Curcuma longa L.). Foods 2022, 11, 3633. https://doi.org/10.3390/foods11223633
Singh K, Srichairatanakool S, Chewonarin T, Prommaban A, Samakradhamrongthai RS, Brennan MA, Brennan CS, Utama-ang N. Impact of Green Extraction on Curcuminoid Content, Antioxidant Activities and Anti-Cancer Efficiency (In Vitro) from Turmeric Rhizomes (Curcuma longa L.). Foods. 2022; 11(22):3633. https://doi.org/10.3390/foods11223633
Chicago/Turabian StyleSingh, Kanjana, Somdet Srichairatanakool, Teera Chewonarin, Adchara Prommaban, Rajnibhas Sukeaw Samakradhamrongthai, Margaret Anne Brennan, Charles Stephen Brennan, and Niramon Utama-ang. 2022. "Impact of Green Extraction on Curcuminoid Content, Antioxidant Activities and Anti-Cancer Efficiency (In Vitro) from Turmeric Rhizomes (Curcuma longa L.)" Foods 11, no. 22: 3633. https://doi.org/10.3390/foods11223633
APA StyleSingh, K., Srichairatanakool, S., Chewonarin, T., Prommaban, A., Samakradhamrongthai, R. S., Brennan, M. A., Brennan, C. S., & Utama-ang, N. (2022). Impact of Green Extraction on Curcuminoid Content, Antioxidant Activities and Anti-Cancer Efficiency (In Vitro) from Turmeric Rhizomes (Curcuma longa L.). Foods, 11(22), 3633. https://doi.org/10.3390/foods11223633








