Twin-Screw Extrusion as Hydrothermal Technology for the Development of Gluten-Free Teff Flours: Effect on Antioxidant, Glycaemic Index and Techno-Functional Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Material
2.3. Experimental Design
2.4. Extrusion Process
2.5. Proximal Analysis
2.6. Colorimetric Study
2.7. Techno-Functional Properties
2.7.1. Hydration Properties
2.7.2. Bulk Density
2.7.3. Pasting Properties
2.8. Scanning Electron Microscopy (SEM)
2.9. Extract Preparation
2.10. Total Phenol (TP)
2.11. Antioxidant Activity (AA)
2.11.1. DPPH• Radical Scavenging Activity and Q-DPPH• Radical Scavenging Activity
2.11.2. Oxygen Radical Absorbance Capacity (ORAC)
2.11.3. ABTS•+ Radical Cation Scavenging Activity and Q-ABTS•+ Radical Cation Scavenging Activity
2.11.4. Ferric Reducing Antioxidant Power (FRAP)
2.12. Glycaemic Index (GI)
2.13. Statistical Analysis
3. Results and discussion
3.1. Proximal Analysis
3.2. Colorimetric Analysis
3.3. Functional Properties
3.3.1. Hydration Properties
3.3.2. Bulk Density
3.3.3. Pasting Properties
3.3.4. Microstructure
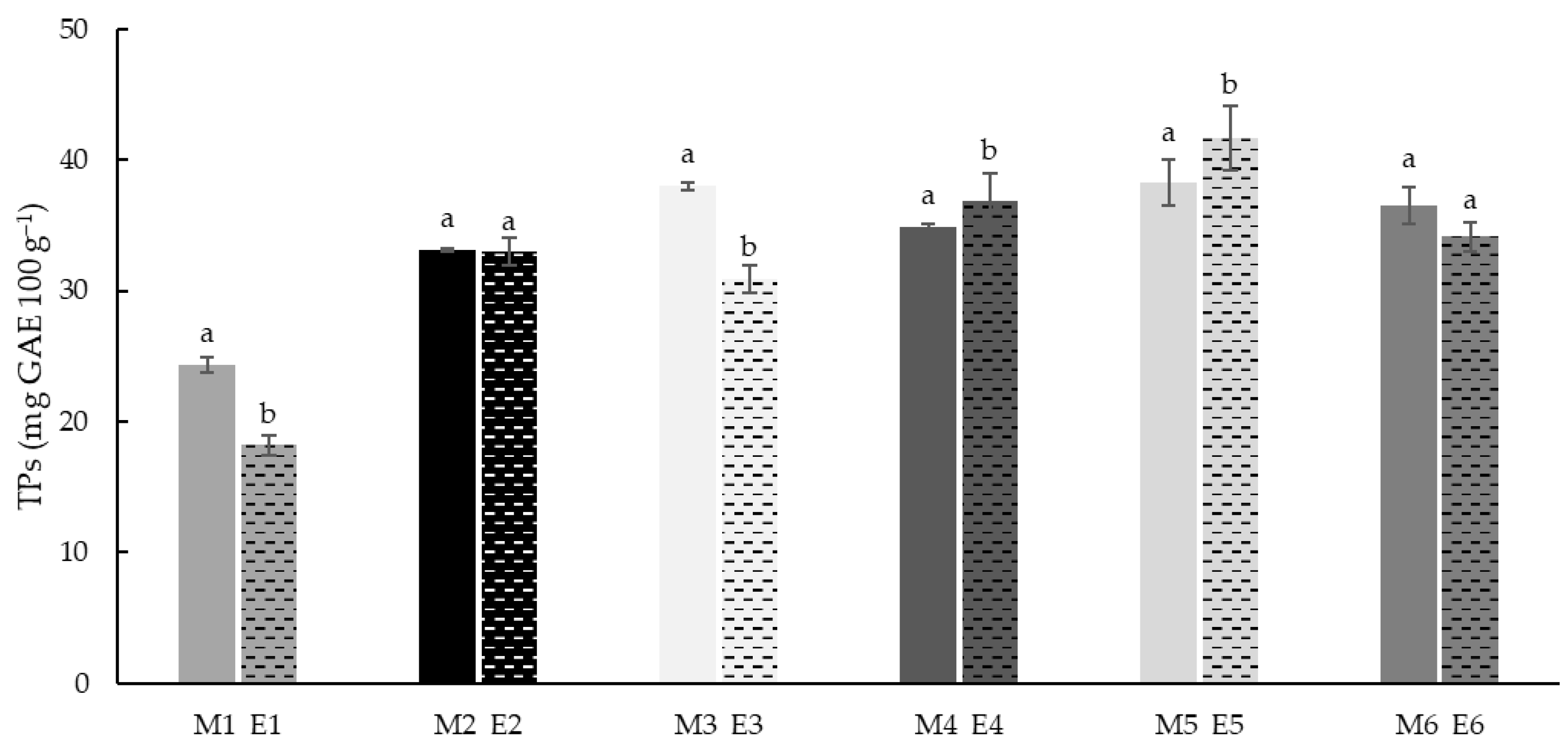
3.4. Total Phenols (TPs)
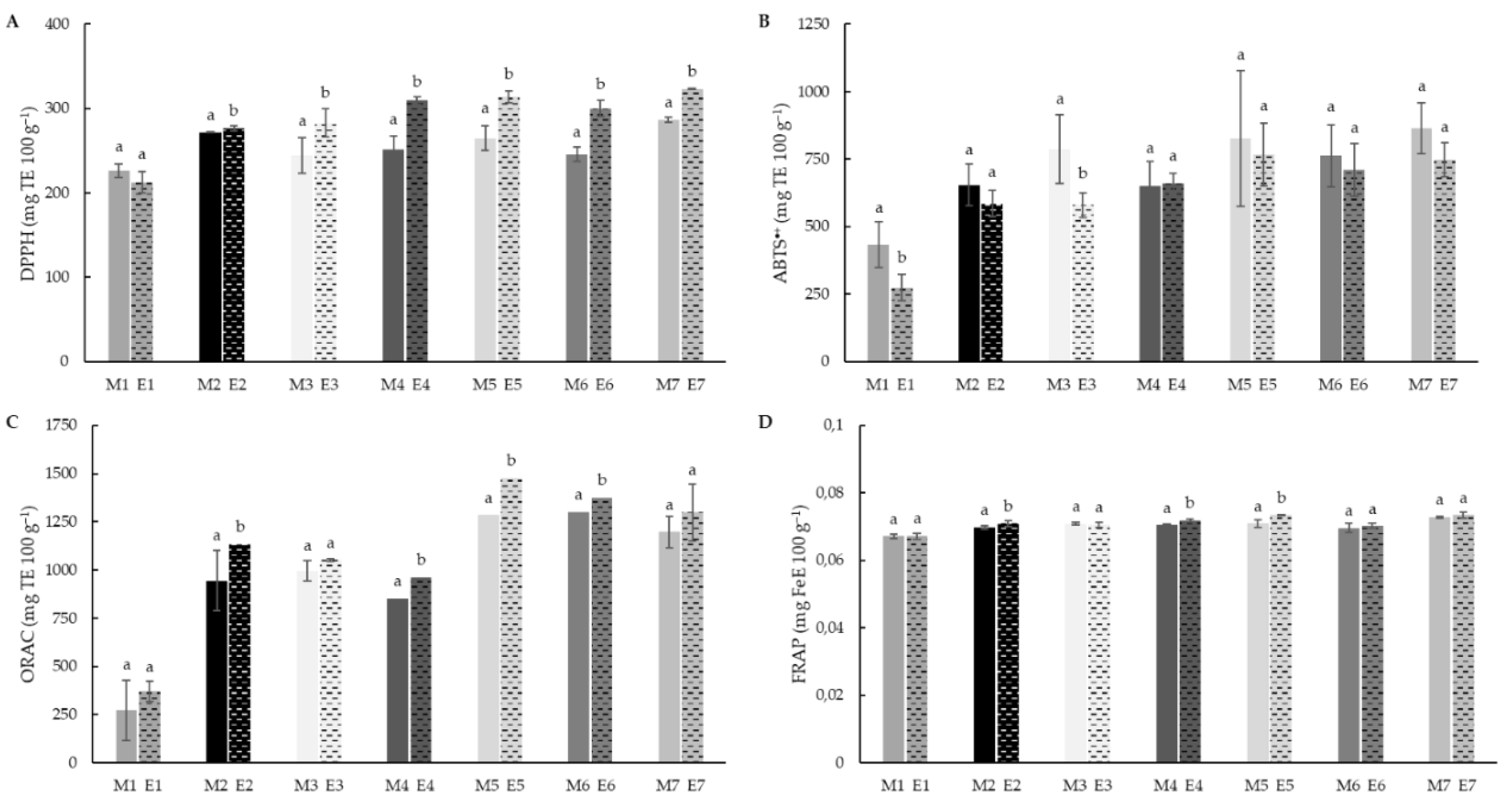
3.5. Total Antioxidant Capacity (TAC)
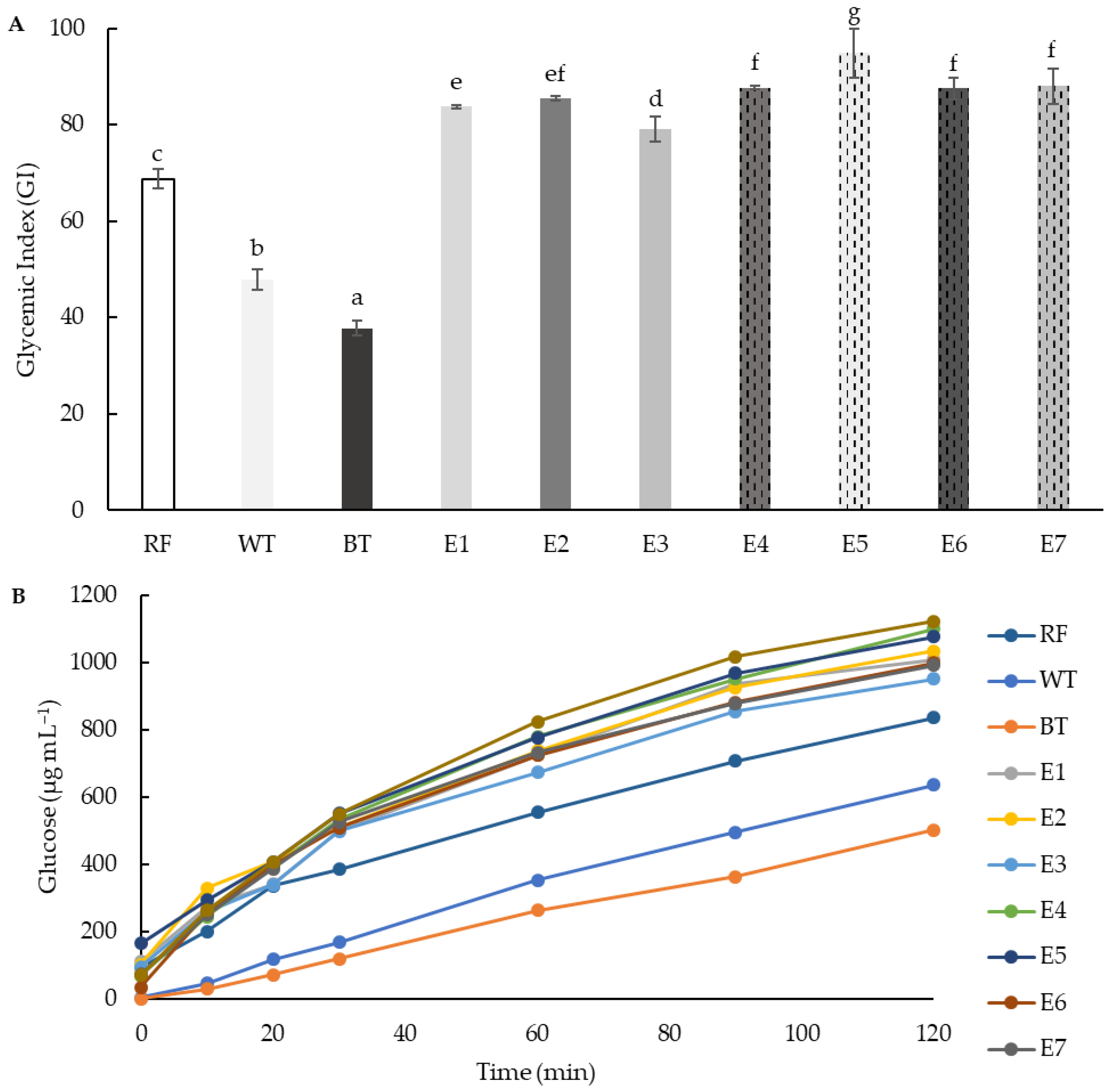
3.6. Gyceamic Index (GI)
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yu, J.M.; Lee, J.H.; Park, J.D.; Choi, Y.S.; Sung, J.M.; Jang, H.W. Analyzing Gluten Content in Various Food Products Using Different Types of ELISA Test Kits. Foods 2021, 10, 108. [Google Scholar] [CrossRef] [PubMed]
- Howdle, P.D. Gliadin, glutenin or both? The search for the Holy Grail in coeliac disease. Eur. J. Gastroenterol. Hepatol. 2006, 18, 703–706. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Tatham, A.S. The prolamin storage proteins of cereal seeds: Structure and evolution. Biochem. J. 1990, 267, 1–12. [Google Scholar] [PubMed]
- Thompson, T. Do oats belong in a gluten-free diet? J. Am. Dietetic Assoc. 1997, 97, 1415–1416. [Google Scholar]
- Janatuinen, E.K.; Pikkanainen, P.H.; Kemppainen, T.A.; Kosma, V.M.; Järvinen, R.M.; Uusitupa, M.I.; Julkunen, R.J. A comparison of diets with and without oats in adults with celiac disease. N. Engl. J. Med. 1995, 333, 1033–1037. [Google Scholar] [CrossRef]
- Caio, G.; Volta, U.; Sapone, A.; Leffler, D.A.; De Giorgio, R.; Catassi, C.; Fasano, A. Celiac disease: A comprehensive current review. BMC Med. 2019, 17, 142. [Google Scholar]
- Prada, M.; Godinho, C.; Rodrigues, D.; Lopes, C.; Garrido, M. The Impact of Gluten-Free Claim on the Perceived Healthfulness, Calories, Level of Processing and Expected Taste of Food Products. Food Qual. Prefer. 2019, 73, 284–287. [Google Scholar]
- Stoven, S.; Murray, J.A.; Marietta, E. Celiac disease: Advances in treatment via gluten modification. Clin. Gastroenterol. Hepatol. 2012, 10, 859–862. [Google Scholar]
- Saturni, L.; Ferretti, G.; Bacchetti, T. The gluten-free diet: Safety and nutritional quality. Nutrients 2010, 2, 16–34. [Google Scholar] [CrossRef]
- Niland, B.; Cash, B.D. Health Benefits and Adverse Effects of a Gluten-Free Diet in Non-Celiac Disease Patients. Gastroenterol. Hepatol. 2018, 14, 82–91. [Google Scholar]
- Cappelli, A.; Oliva, N.; Cini, E. A systematic review of gluten-free dough and bread: Dough rheology, bread characteristics, and improvement strategies. Appl. Sci. 2020, 10, 6559. [Google Scholar]
- Culetu, A.; Susman, I.E.; Duta, D.E.; Belc, N. Nutritional and Functional Properties of Gluten-Free Flours. Appl. Sci. 2021, 11, 6283. [Google Scholar]
- Gabrovská, D.; Hálová, I.; Chrpová, D.; Ouhrabková, J.; Sluková, M.; Vavreinová, S.; Faměra, O.; Kohout, P.; Pánek, J.; Skřivan, P. Cereals in Human Nutrition, (Obiloviny v Lidské Výživě), 1st ed.; Federation of the Food and Drink Industries of the Czech Republic: Prague, Czech Republic, 2015; pp. 44–49. [Google Scholar]
- Bultosa, G.; Taylor, J.R.N. Chemical and physical characterisation of grain tef (Eragrostis tef (Zucc.) Trotter) starch granule composition. Starch/Stärke 2003, 56, 304–312. [Google Scholar] [CrossRef]
- Bultosa, G. Teff: Overview. In Encyclopedia of Food Grains, 2nd ed.; Wrigley, C., Corke, H., Seetharaman, K., Faubion, J., Eds.; Elsevier: Oxford, UK, 2016; Volume 1, pp. 209–220. [Google Scholar]
- Ketema, S. Tef. Eragrostis tef (Zucc.) Trotter. Promoting the Conservation and Use of Underutilized and Neglected Crops. 12; Institute of Plant Genetics and Crop Plant Research, Gatersleben/International Plant Genetic Resources Institute: Rome, Italy, 1997. [Google Scholar]
- Rico, D.; Ronda, F.; Villanueva, M.; Perez Montero, C.; Martin-Diana, A.B. Development of healthy gluten-free crackers from white and brown tef (Eragrostis tef Zucc.) flours. Heliyon 2019, 5, e02598. [Google Scholar]
- Mengesha, M.H.; Pickett, R.C.; Davis, R.L. Genetic Variability and Interrelationship of Characters in Teff, Eragrostis tef (Zucc.) Trotter. Crop Sci. 1965, 5, 155–157. [Google Scholar] [CrossRef]
- Ronda, F.; Abebe, W.; Pérez-Quire, S.; Collar, C. Suitability of tef varieties in mixed wheat flour bread matrices: A physico-chemical and nutritional approach. J. Cereal Sci. 2015, 64, 139–146. [Google Scholar] [CrossRef]
- Abebe, W.; Collar, C.; Ronda, F. Impact of variety type and particle size distribution on starch enzymatic hydrolysis and functional properties of tef flours. Carbohydr. Polym. 2015, 115, 260–268. [Google Scholar] [CrossRef]
- Zhu, F. Chemical composition and food uses of teff (Eragrostis tef). Food Chem. 2018, 239, 402–415. [Google Scholar]
- Wolter, A.; Hager, A.S.; Zannini, E.; Arendt, E.K. In vitro starch digestibility and predicted glycaemic indexes of buckwheat, oat, quinoa, sorghum, teff and commercial gluten-free bread. J. Cereal Sci. 2013, 58, 431–436. [Google Scholar]
- Gómez, M.; Martinez, M. Changing flour functionality through physical treatments for the production of gluten-free baking goods. J. Cereal Sci. 2016, 67, 68–74. [Google Scholar]
- Stojceska, V.; Ainsworth, P.; Plunkett, A.; İbanoğlu, S. The advantage of using extrusion processing for increasing dietary fibre level in gluten-free products. Food Chem. 2010, 121, 156–164. [Google Scholar]
- Jafari, M.; Koocheki, A.; Milani, E. Functional effects of xanthan gum on quality attributes and microstructure of extruded sorghum-wheat composite dough and bread. LWT—Food Sci. Technol. 2017, 89, 551–558. [Google Scholar]
- Jing, Y.; Chi, Y. Effects of twin-screw extrusion on soluble dietary fibre and physicochemical properties of soybean residue. Food Chem. 2013, 138, 884–889. [Google Scholar] [CrossRef] [PubMed]
- Cheftel, J. Nutritional effects of extrusion-cooking. Food Chem. 1986, 20, 263–283. [Google Scholar]
- Dalbhagat, C.G.; Mahato, D.K.; Mishra, H.N. Effect of extrusion processing on physicochemical, functional and nutritional characteristics of rice and rice-based products: A review. Trends Food Sci. Technol. 2019, 85, 226–240. [Google Scholar]
- Kristiawan, M.; Micard, V.; Maladira, P.; Alchamieh, C.; Maigret, J.E.; Réguerre, A.L.; Emin, M.A.; Della Valle, G. Multi-scale structural changes of starch and proteins during pea flour extrusion. Food Res. Int. 2018, 108, 203–215. [Google Scholar] [CrossRef]
- Nikmaram, N.; Leong, S.; Koubaa, M.; Zhu, Z.; Barba, F.; Greiner, R.; Oey, I.; Roohinejad, S. Effect of extrusion on the anti-nutritional factors of food products: An overview. Food Control 2017, 79, 62–73. [Google Scholar] [CrossRef]
- Robin, F.; Dubois, C.; Pineau, N.; Labat, E.; Théoduloz, C.; Curti, D. Process, structure and texture of extruded whole wheat. J. Cereal Sci. 2012, 56, 358–366. [Google Scholar]
- Robin, F.; Théoduloz, C.; Srichuwong, S. Properties of extruded whole grain cereals and pseudocereals flours. Int. J. Food Sci. Technol. 2015, 50, 2152–2159. [Google Scholar] [CrossRef]
- Diaz, J.M.R.; Kirjoranta, S.; Tenitz, S.; Penttilä, P.A.; Serimaa, R.; Lampi, A.M.; Jouppila, K. Use of Amaranth, Quinoa and Kañiwa in extruded corn-based snacks. J. Cereal Sci. 2013, 58, 59–67. [Google Scholar]
- Sundarrajan, L. Effect of Extrusion Cooking on the Nutritional Properties of Amaranth, Quinoa, Kañiwa and Lupine. Master’s Thesis, University of Helsinki, Helsinki, Finland, 2014. [Google Scholar]
- Nadeesha Dilrukshi, H.N.; Torrico, D.D.; Brennan, M.A.; Brennan, C.S. Effects of extrusion processing on the bioactive constituents, in vitro digestibility, amino acid composition, and antioxidant potential of novel gluten-free extruded snacks fortified with cowpea and whey protein concentrate. Food Chem. 2022, 389, 133107. [Google Scholar] [PubMed]
- AOAC. Methods 990.03, 2003.05, 985.29 & 923.03. In Official Methods of Analysis of AOAC International, 18th ed.; AOAC: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Nelson, A.L. Properties of high-fibre ingredients. Cereal. Foods World 2001, 46, 93–97. [Google Scholar]
- Beuchat, L.R. Functional and electrophoretic characteristics of succinylated peanut flour protein. J. Agric. Food Chem. 1977, 25, 258–261. [Google Scholar]
- Kaushal, P.; Kumar, V.; Sharma, H.K. Comparative study of physicochemical, functional, antinutritional and pasting properties of taro (Colocasia esculenta), rice (Oryza sativa) flour, pigeon pea (Cajanus cajan) flour and their blends. LWT—Food Sci. Tech. 2012, 48, 59–68. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V.L. Total phenol analyses: Automation and comparison with manual methods. Am. J. Enol. Viticult. 1977, 28, 49–55. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT—Food Sci. Tech. 1995, 28, 25–30. [Google Scholar]
- Serpen, A.; Capuano, E.; Fogliano, V.; Gökmen, V. A new procedure to measure the antioxidant activity of insoluble food components. J. Agric. Food Chem. 2007, 55, 7676–7681. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and Validation of an Improved Oxygen Radical Absorbance Capacity Assay Using Fluorescein as the Fluorescent Probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar]
- Miller, N.J.; Rice-Evans, C.A. Factors influencing the antioxidant activity determined by the ABTS•+ radical cation assay. Free Radic. Res. 1997, 26, 195–199. [Google Scholar] [CrossRef]
- Martin-Diana, A.B.; Izquierdo, N.; Albertos, I.; Sanchez, M.S.; Herrero, A.; Sanz, M.A.; Rico, D. Valorization of Carob’s germ and seed peel as natural antioxidant ingredients in gluten-free crackers. J. Food Process. Preserv. 2017, 41, e12770. [Google Scholar] [CrossRef]
- Pereira, J.A.; Oliveira, I.; Sousa, A.; Ferreira, C.F.R.; Bento, A.; Estevinho, L. Bioactive properties and chemical composition of six walnut (Juglans regia L.) cultivars. Food Chem. Toxicol. 2008, 46, 2103–2111. [Google Scholar] [CrossRef] [PubMed]
- Gularte, M.; Rosell, C. Physicochemical properties and enzymatic hydrolysis of different starches in the presence of hydrocolloids. Carbohydr. Polym. 2011, 85, 237–244. [Google Scholar]
- Granfeldt, Y. Foods Factors Affecting Metabolic Responses to Cereal Products. Ph.D. Thesis, University of Lund, Lund, Sweden, 1994. [Google Scholar]
- Clerici, M.T.P.S.; Airoldi, C.; El-Dash, A.A. Production of acidic extruded rice flour and its influence on the qualities of gluten-free bread. LWT—Food Sci. Technol. 2009, 42, 618–623. [Google Scholar]
- Sivaramakrishnan, H.P.; Senge, B.; Chattopadhyay, P.K. Rheological properties of rice dough for making rice bread. J. Food Eng. 2004, 62, 37–45. [Google Scholar] [CrossRef]
- Hager, A.S.; Bormans, G.M.; Delcour, A. Physical and Molecular Changes during the Storage of Gluten-Free Rice and Oat Bread. J. Agric. Food Chem. 2014, 62, 5682–5689. [Google Scholar]
- Oppong, D.; Panpipat, W.; Chaijan, M. Chemical, physical, and functional properties of Thai indigenous brown rice flours. PLoS ONE 2021, 16, e0255694. [Google Scholar]
- Ethiopian Health and Nutrition Research Institute (EHNRI). Food Composition Table for Use in Ethiopia; Ethiopian Health and Nutrition Research Institute (EHNRI): Addis Ababa, Ethiopia, 1998. [Google Scholar]
- Camire, M.E.; Camire, A.L.; Krumhar, K. Chemical and nutritional changes. Crit. Rev. Food Sci. Nutr. 1990, 29, 35–57. [Google Scholar] [CrossRef]
- Singh, S.; Gamlath, S.; Wakeling, L. Nutritional aspects of food extrusion: A review. Int. J. Food Sci. Technol. 2007, 42, 916–929. [Google Scholar] [CrossRef]
- Rico, D.; Cano, A.B.; Martín-Diana, A.B. Pulse-Cereal Blend Extrusion for Improving the Antioxidant Properties of a Gluten-Free Flour. Molecules 2021, 26, 5578. [Google Scholar] [CrossRef]
- Ramírez, J.; Wanderlei, C.; Meléndez, A.; Lima, O.; Penteado, M. Caracterización Físico-Química de Pellets Extruídos de Torta de Higuerilla (Ricinus comunis L.) Visando su uso en Alimentos Balanceados; Empresa Brasileira de Pesquisa Agropecuária: Canoinhas, Brazil, 2013. [Google Scholar]
- Youssef, M.K.E.; Nassar, A.G.; EL–Fishawy, F.A.; Mostafa, M.A. Assessment of proximate chemical composition and nutritional status of wheat biscuits fortified with oat powder. Assiut. J. Agric. Sci. 2016, 47, 83–94. [Google Scholar]
- Frame, N.D. Operational Characteristics of the Co Rotating Twin Screw Extruder. In Technology of Extrusion Cooking; Blackie Academic and Professional: New York, NY, USA, 1994; pp. 1–50. [Google Scholar]
- De Pilli, T.; Giuliani, R.; Carbone, B.F.; Derossi, A.; Severini, C. Study on different emulsifiers to retain fatty fraction during extrusion of fatty flours. Cereal. Chem. 2005, 82, 494–498. [Google Scholar]
- Zhong, L.; Fang, Z.; Wahlqvist, M.L.; Hodgson, J.M.; Johnson, S.K. Multi-response surface optimisation of extrusion cooking to increase soluble dietary fibre and polyphenols in lupin seed coat. LWT 2021, 140, 110767. [Google Scholar] [CrossRef]
- Ye, G.; Wu, Y.; Wang, L.; Tan, B.; Shen, W.; Li, X.; Liu, Y.; Tian, X.; Zhang, D. Comparison of six modification methods on the chemical composition, functional properties and antioxidant capacity of wheat bran. LWT 2021, 149, 111996. [Google Scholar] [CrossRef]
- Pérez-Navarrete, C.; González, R.; Chel-Guerrero, L.; Betancur-Ancona, D. Effect of extrusion on nutritional quality of maize and Lima bean flour blends. J. Sci. Food Agric. 2006, 86, 2477–2484. [Google Scholar]
- Wu, N.-N.; Ma, Z.-Q.; Li, H.-H.; Tian, X.-H.; Fang, Y.; Tan, B. Nutritional and cooking quality improvement of brown rice noodles prepared with extruded rice bran. Cereal Chem. 2021, 98, 346–354. [Google Scholar]
- Bhatnagar, S.; Hanna, M.A. Amylose–lipid complex formation during single-screw extrusion of various corn starches. Cereal Chem. 1994, 71, 582–587. [Google Scholar]
- Nascimento, K.D.O.D.; Paes, S.D.N.D.; Oliveira, I.R.D.; Reis, I.P.; Augusta, I.M. Teff: Suitability for Different Food Applications and as a Raw Material of Gluten-free, a Literature Review. J. Food Nutr. Res. 2018, 6, 74–81. [Google Scholar] [CrossRef]
- Bhatnagar, S.; Hanna, M.A. Extrusion processing conditions for amylose-lipid complexing. Cereal Chem. 1985, 71, 587–593. [Google Scholar]
- Bekabil, F.; Befekadu, B.; Simons, R.; Tareke, B. Strengthening the Teff Value Chain in Ethiopia. In Table off Arm Level Profitability Analysis; Ethiopian ATA, Ethiopian Agricultural Transformation Agency: Addis Ababa, Ethiopia, 2011. [Google Scholar]
- Kuchin, N.; Sizova, Y.; Kuleshova, L. The effect of extrusion on the nutrient content of barley as a feed material. IOP Conf. Ser. Earth Environ. Sci. 2021, 857, 012014. [Google Scholar]
- Ismagilov, R.; Ayupov, D.; Nurlygayanov, R.; Ahiyarova, L.; Abdulloev, V. Ways to reduce anti-nutritional substances in winter rye grain. Physiol. Mol. Biol. Plants 2020, 26, 1067–1073. [Google Scholar] [CrossRef]
- Leonel, M.; Freitas, T.; Mischan, M. Physical characteristics of extruded cassava starch. Sci. Agric. 2009, 66, 486–493. [Google Scholar]
- Bhattacharya, S.; Sivakumar, V.; Chakraborty, D. Changes in CIE Lab colour parameters due to extrusion of rice-green gram blend: A response surface approach. J. Food Eng. 1997, 32, 125–131. [Google Scholar] [CrossRef]
- Rosentrater, K.A.; Muthukumarappan, K.; Kannadhason, S. Effects of Ingredients and Extrusion Parameters on Aquafeeds Containing DDGS and Potato Starch. J. Aqua. Feed Sci. Nutr. 2009, 1, 22–387. [Google Scholar]
- Paes, M.C.D.; Maga, J. Effect of extrusion on essential amino acids profile and color of whole-grain flours of Quality Protein Maize (QPM) and Normal Maize Cultivars. Rev. Bras. De Milho E Sorgo 2004, 3, 10–20. [Google Scholar] [CrossRef]
- Jozinović, A.; Šubarić, D.; Ačkar, D.; Babić, J.; Miličević, B. Influence of spelt flour addition on properties of extruded products based on corn grits. J. Food Eng. 2016, 172, 31–37. [Google Scholar]
- Wang, Q.; Li, L.; Zheng, X. A review of milling damaged starch: Generation, measurement, functionality and its effect on starch-based food systems. Food Chem. 2020, 315, 126267. [Google Scholar]
- Salamoni, F.; da Costa, E.; Soares, M.; Souza, J.A.; Vânia, A. Physical and functional evaluation of extruded flours obtained from different rice genotypes. Ciência E Agrotecnologia 2014, 38, 367–374. [Google Scholar]
- Riaz, M.N. Introduction to Extruders and Their Principles. In Extruders in Food Applications; CRC Press: Boca Raton, FL, USA, 2000; pp. 1–23. [Google Scholar]
- Ding, Q.; Ainsworth, P.; Tucker, G.; Marson, H. The effect of extrusion conditions on the physicochemical properties and sensory characteristics of rice-based expanded snack. J. Food Eng. 2005, 66, 83–289. [Google Scholar]
- de la Hera, E.; Gomez, M.; Rosell, C.M. Particle size distribution of rice flour affecting the starch enzymatic hydrolysis and hydration properties. Carbohydr Polym. 2013, 98, 421–427. [Google Scholar] [CrossRef]
- Meuser, F.; Wiedmann, W. Extrusion plant design. In Extrusion Cooking; Mercier, C., Linko, P., Harper, J.M., Eds.; American Association of Cereal Chemists: St. Paul, MN, USA, 1989; pp. 91–155. [Google Scholar]
- Veronica, A.O.; Olusola, O.O.; Adebowale, E.A. Qualities of extruded puffed snacks from maize/soybean Food Bioprocess Technol mixture. J. Food Proc. Eng. 2006, 29, 149–161. [Google Scholar] [CrossRef]
- Onwulata, C.I.; Konstance, R.P.; Smith, P.W.; Holsinger, V.H. Co-extrusion of Dietary Fiber and Milk Proteins in Expanded Corn Products. LWT—Food Sci. Technol. 2001, 34, 424–429. [Google Scholar]
- Bultosa, G.; Hall, A.N.; Taylor, J.R.N. Physico-chemical Characterization of Grain Tef [Eragrostis tef (Zucc.) Trotter] Starch. Starch-Stärke 2002, 55, 304–312. [Google Scholar]
- Corke, H. Grain: Morphology of Internal Structure. In Encyclopedia of Food Grains, 2nd ed.; Wrigley, C., Corke, H., Seetharaman, K., Faubion, J., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 41–50. [Google Scholar]
- Kumar, L.; Brennan, M.; Brennan, C.; Zheng, H. Influence of whey protein isolate on pasting, thermal, and structural characteristics of oat starch. J. Dairy Sci. 2022, 105, 56–71. [Google Scholar] [PubMed]
- Sayar, S.; Koksel, H.; Turhan, M. The Effects of Protein-Rich Fraction and Defatting on Pasting Behavior of Chickpea Starch. Starch-Stärke 2005, 57, 599–604. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, H.; Cheng, L.; Gu, Z.; Hua, D.; Qi, X.; Qian, H.; Wang, L. Effect of Extrusion on the Hydrophilic Antioxidant Capacity of Four Whole Grains. J. Food Nutr. Res. 2014, 2, 80–87. [Google Scholar] [CrossRef][Green Version]
- Forsido, S.F.; Rupasinghe, H.P.V.; Astatkie, T. Antioxidant capacity, total phenolics and nutritional content in selected ethiopian staple food ingredients. Int. J. Food Sci. Nutr. 2013, 64, 915–920. [Google Scholar] [CrossRef]
- Shumoy, H.; Raes, K. Tef: The Rising Ancient Cereal: What do we know about its Nutritional and Health Benefits? Plant Foods Hum. Nutr. 2017, 72, 335–344. [Google Scholar]
- Hu, Z.; Tang, X.; Zhang, M.; Hu, X.; Yu, C.; Zhu, Z.; Shao, Y. Effects of different extrusion temperatures on extrusion behavior, phenolic acids, antioxidant activity, anthocyanins and phytosterols of black rice. RSC Adv. 2018, 8, 7123–7132. [Google Scholar] [CrossRef]
- Rochín-Medina, J.J.; Gutiérrez-Dorado, R.; Mora-Rochín, S.; Medina-Godoy, S.; Valdez-Ortiz, Á.; López-Valenzuela, J.; Delgado-Vargas, F.; Milán-Carrillo, J.; Reyes-Moreno, C. Nutraceutical beverage from a high antioxidant activity mixture of extruded whole maize and chickpea flours. Eur. Int. J. Sci. Technol. 2012, 1, 1–14. [Google Scholar]
- Lindenmeier, M.; Faist, V.; Hofmann, T. Structural and functional characterization of pronyl-lysine, a novel protein modification in bread crust melanoidins showing in vitro antioxidative and phase I/II enzyme modulating activity. J. Agric. Food Chem. 2002, 50, 6997–7006. [Google Scholar] [CrossRef]
- Martysiak-Żurowska, D.; Wenta, W. A comparison of ABTS and DPPH methods for assessing the total antioxidant capacity of human milk. Acta Sci. Pol. Technol. Aliment. 2012, 11, 83–89. [Google Scholar] [PubMed]
- Liu, K. Comparison of Lipid Content and Fatty Acid Composition and Their Distribution within Seeds of 5 Small Grain Species. J. Food Sci. 2011, 76, C334–C342. [Google Scholar] [CrossRef] [PubMed]
- Ryan, E.; Galvin, K.; O’Connor, T.P.; Maguire, A.R. Phytosterol, Squalene, Tocopherol Content and Fatty Acid Profile of Selected Seeds, Grains, and Legumes. Plant Foods Hum. Nutr. 2007, 62, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Martens, B.M.J.; Gerrits, W.J.J.; Bruininx, E.M.A.M.; Schols, H.A. Amylopectin structure and crystallinity explains variation in digestion kinetics of starches across botanic sources in an in vitro pig model. J. Anim. Sci. Biotechnol. 2018, 9, 91. [Google Scholar]
- Callejo, M.J.; Benavente, E.; Ezpeleta, J.I.; Laguna, M.J.; Carrillo, J.M.; Rodríguez-Quijano, M. Influence of teff variety and wheat flour strength on breadmaking properties of healthier teff-based breads. J. Cereal Sci. 2016, 68, 38–45. [Google Scholar]
- Oh, S.H.; Kalyani, R.R.; Dobs, A.S. Diabetes Mellitus: Dietary Management. In Encyclopedia of Human Nutrition, 3rd ed.; Allen, L., Prentice, A., Eds.; Elsevier: Oxford, UK, 2013; Volume 2, pp. 25–39. [Google Scholar]
- Onwulata, C.I.; Thomas, A.E.; Cooke, P.H.; Phillips, J.H.; Carvalho, C.W.P.; Ascheri, J.L.R.; Tomasula, P.M. Glycemic potential of extruded barley, cassava, corn, and quinoa enriched with whey proteins and cashew pulp. Int. J. Food Prop. 2010, 13, 338–359. [Google Scholar] [CrossRef]
- Cabrera-Chavez, F.; Barca, A.M.C.; Islas-Rubio, A.R.; Marti, A.; Marengo, M.; Pagani, M.A.; Iamett, S. Molecular rearrangements in extrusion processes for the production of amaranth-enriched, gluten-free rice pasta. LWT—Food Sci. Technol. 2012, 47, 421–426. [Google Scholar] [CrossRef]
- Liu, H.; Guo, X.; Li, W.; Wang, X.; Manman, I.; Peng, Q.; Wang, M. Changes in physicochemical properties and in vitro digestibility of common buckwheat starch by heat-moisture treatment and annealing. Carbohydr. Polym. 2015, 132, 237–244. [Google Scholar]
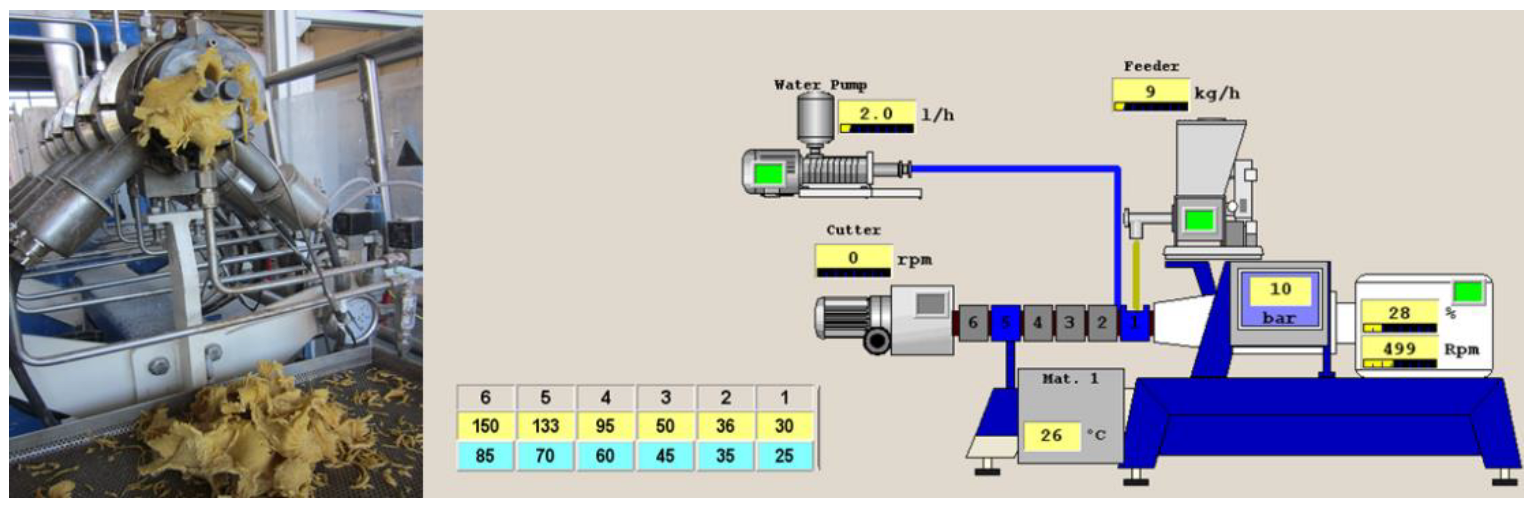



| Blend Fours | WTF | BTF | RF |
|---|---|---|---|
| 1 | 0 | 0 | 100 |
| 2 | 12.5 | 12.5 | 75 |
| 3 | 25 | 0 | 75 |
| 4 | 0 | 25 | 75 |
| 5 | 25 | 25 | 50 |
| 6 | 50 | 0 | 50 |
| 7 | 0 | 50 | 50 |
| Blends (g 100 g−1) | Ash (g 100 g−1) | Fat (g 100 g−1) | TDF (g 100 g−1) | Moisture (g 100 g−1) | Proteins (g 100 g−1) | Carbohydrates (g 100 g−1) |
|---|---|---|---|---|---|---|
| RF | 1.50 ± 0.00 c | 1.19 ± 0.02 a | 1.65 ± 0.01 a | 10.01 ± 0.12 d | 8.48 ± 0.01 a | 88.83 ± 0.03 e |
| WTF | 3.44 ± 0.03 f | 2.76 ± 0.08 e | 4.54 ± 0.15 c | 11.13 ± 0.13 e | 12.13 ± 0.03 d | 81.67 ± 0.07 cd |
| BTF | 2.50 ± 0.02 e | 2,24 ± 0.20 d | 4.45 ± 0.76 c | 11.74 ± 0.56 e | 13.28 ± 0.13 e | 81.98 ± 0.35 d |
| E1 (100 RF) | 0.82 ± 0.05 a | 1.28 ± 0.07 a | 1.85 ± 0.23 a | 7.90 ± 0.45 c | 8.35 ± 0.47 a | 79.80 ± 1.27 bcd |
| E2 (12.5 WTF/12.5 BTF/75 RF) | 1.16 ± 0.07 b | 1.79 ± 0.10 bc | 4.08 ± 0.52 bc | 6.85 ± 0.39 b | 9.26 ± 0.52 ab | 76.85 ± 1.60 ab |
| E3 (25 WTF/75 RF) | 1.14 ± 0.06 b | 1.65 ± 0.09 b | 3.50 ± 0.45 b | 5.82 ± 0.33 a | 9.16 ± 0.52 ab | 78.72 ± 1.45 bc |
| E4 (25 BTF/75 RF) | 1.09 ± 0.06 b | 1.72 ± 0.10 b | 3.93 ± 0.50 b | 5.81 ± 0.33 a | 9.49 ± 0.54 bc | 77.96 ± 1.52 b |
| E5 (25 WTF/25 BTF/50 RF) | 1.54 ± 0.09 cd | 2.02 ± 0.11 cd | 5.76 ± 0.73 d | 6.17 ± 0.35 ab | 10.06 ± 0.57 bc | 74.44 ± 1.85 a |
| E6 (50 WTF/50 RF) | 1.67 ± 0.09 d | 1.60 ± 0.09 b | 6.22 ± 0.79 d | 6.73 ± 0.38 b | 9.59 ± 0.54 bc | 74.20 ± 1.90 a |
| E7 (50 BTF/50 RF) | 1.63 ± 0.09 cd | 2.15 ± 0.12 d | 5.52 ± 0.70 d | 5.75 ± 0.33 a | 10.34 ± 0.59 c | 74.61 ± 1.83 a |
| L* | a* | b* | ||||
|---|---|---|---|---|---|---|
| Blends (g 100 g−1) | M | E | M | E | M | E |
| 1 (100RF) | 119.34 ± 0.27 b | 114.55 ± 0.64 a | −0.20 ± 0.11 a | −0.16 ± 0.08 a | 8.58 ± 0.17 a | 12.44 ± 0.13 b |
| 2 (12.5WTF/12.5 BTF/75RF) | 105.80 ± 0.12 b | 100.62 ± 0.38 a | 2.50 ± 0.03 a | 2.48 ± 0.09 a | 11.00 ± 0.09 a | 15.13 ± 0.39 b |
| 3 (25WTF/75RF) | 109.70 ± 0.08 b | 102.35 ± 0.44 a | 1.30 ±0.05 a | 1.63 ± 0.05 b | 11.50 ± 0.07 a | 17.31 ± 0.09 b |
| 4 (25BTF/75RF) | 102.40 ± 0.18 b | 98.94 ± 0.42 a | 3.50 ± 0.05 b | 3.08 ± 0.05 a | 10.90 ± 0.12 a | 13.33 ± 0.41 b |
| 5 (25WTF/25 BTF/50RF) | 100.30 ± 0.15 b | 81.35 ± 2.70 a | 3.30 ± 0.08 a | 3.22 ± 0.39 a | 13.40 ± 0.18 a | 15.19 ± 0.50 b |
| 6 (50WTF/50RF) | 107.50 ± 0.09 b | 87.01 ± 1.08 a | 1.20 ± 0.03 b | 0.73 ± 0.21 a | 15.30 ± 0.16 a | 15.98 ± 0.27 b |
| 7 (50BTF/50RF) | 94.80 ± 0.29 b | 72.17 ± 1.79 a | 5.00 ± 0.08 a | 5.06 ± 0.38 a | 13.10 ± 0.17 a | 14.31 ± 0.69 b |
| . | WAI (g/g) | WSI (%) | SP (mL/g) | |||
|---|---|---|---|---|---|---|
| Blends (g 100 g−1) | M | E | M | E | M | E |
| 1 (100RF) | 6.46 ± 0.56 a | 7.64 ± 0.06 a | 1.55 ± 1.07 a | 8.47 ± 0.49 b | 6.60 ± 0.55 a | 8.29 ± 0.03 b |
| 2 (12.5WTF/12.5BTF/75 RF) | 5.43 ± 0.12 a | 6.31 ± 0.11 b | 3.36 ± 0.29 a | 11.35 ± 1.88 b | 5.59 ± 0.13 a | 7.08 ± 0.41 b |
| 3 (25WTF/75RF) | 5.23 ± 0.23 a | 6.77 ± 0.14 b | 3.41 ± 1.05 a | 14.70 ± 0.41 b | 5.40 ± 0.20 a | 7.88 ± 0.19 b |
| 4 (25BTF/75RF) | 5.59 ± 0.07 a | 6.23 ± 0.14 b | 2.22 ± 0.60 a | 16.59 ± 0.55 b | 5.71 ± 0.07 a | 7.41 ± 0.18 b |
| 5 (25WTF/25BTF/50RF) | 5.17 ± 0.21 a | 6.84 ± 0.29 b | 2.52 ± 0.16 a | 21.02 ± 6.03 b | 5.29 ± 0.22 a | 8.58 ± 0.78 b |
| 6 (50WTF/50RF) | 5.43 ± 0.12 a | 6.85 ± 0.24 b | 3.09 ± 0.25 a | 10.57 ± 0.77 b | 5.58 ± 0.13 a | 7.61 ± 0.21 b |
| 7 (50BTF/50RF) | 5.29 ± 0.19 a | 7.37 ± 0.20 b | 2.82 ± 0.55 a | 25.11 ± 1.91 b | 5.43 ± 0.17 a | 9.69 ± 0.04 b |
| WHC (%) | SV (g/g) | WAC (g/g) | OAC (g/g) | |||||
|---|---|---|---|---|---|---|---|---|
| Blends (g 100 g−1) | M | E | M | E | M | E | M | E |
| 1 (100RF) | 2.11 ± 0.82 a | 7.88 ± 0.16 b | 2.93 ± 0.16 a | 10.81 ± 0.50 b | 2.64 ± 0.14 a | 6.80 ± 0.23 b | 1.10 ± 0.03 a | 2.05 ± 0.08 b |
| 2 (12.5WTF/12.5BTF/75 RF) | 1.80 ± 0.03 a | 7.36 ± 0.64 b | 3.00 ± 0.16 a | 8.68 ± 0.25 b | 2.67 ± 0.07 a | 6.68 ± 0.09 b | 1.23 ± 0.15 a | 1.58 ± 0.20 a |
| 3 (25WTF/75RF) | 1.75 ± 0.03 a | 6.60 ± 0.95 b | 3.06 ± 0.01 a | 8.13 ± 0.58 b | 2.55 ± 0.21 a | 6.60 ± 0.07 b | 1.50 ± 0.12 a | 1.58 ± 0.08 a |
| 4 (25BTF/75RF) | 1.66 ± 0.03 a | 6.30 ± 1.25 b | 2.91 ± 0.16 a | 9.19 ±0.52 b | 2.60 ± 0.02 a | 6.87 ± 0.08 b | 1.63 ± 0.19 a | 1.70 ± 0.10 a |
| 5 (25WTF/25BTF/50 RF) | 1.75 ± 0.03 a | 6.09 ± 0.39 b | 3.14 ± 0.16 a | 8.42 ± 0.01 b | 2.56 ± 0.04 a | 6.83 ± 0.42 b | 1.91 ± 0.11 a | 1.83 ± 0.08 a |
| 6 (50WTF/50RF) | 1.77 ± 0.03 a | 7.54 ± 0.09 b | 3.16 ± 0.16 a | 9.43 ± 0.15 b | 2.52 ± 0.07 a | 6.59 ± 0.16 b | 1.68 ± 0.06 a | 1.67 ± 0.11 a |
| 7 (50BTF/50RF) | 1.78 ± 0.02 a | 5.91 ± 0.83 b | 3.07 ± 0.01 a | 8.21 ±0.73 b | 2.62 ± 0.04 a | 6.93 ± 0.21 b | 1.56 ± 0.14 a | 1.64 ± 0.08 a |
| Blends (g 100 g−1) | M | E |
|---|---|---|
| 1 (100RF) | 812.7 ± 0.02 a | 691.6 ± 0.01 b |
| 2 (12.5WTF/12.5 BTF/75RF) | 837.0 ± 0.02 a | 724.8 ± 0.01 b |
| 3 (25WTF/75RF) | 842.0 ± 0.01 a | 710.4 ± 0.01 b |
| 4 (25BTF/75RF) | 820.0 ± 0.00 a | 695.5 ± 0.01 b |
| 5 (25WTF/25 BTF/50RF) | 817.4 ± 0.02 a | 715.6 ± 0.01 b |
| 6 (50WTF/50RF) | 821.7 ± 0.00 a | 754.5 ± 0.01 b |
| 7 (50BTF/50RF) | 819.9 ± 0.02 a | 691.6 ± 0.01 b |
| Blends (g 100 g−1) | Pasting Viscosity (cp) | Through Viscosity (cp) | Breakdown Viscosity (cp) | Final Viscosity (cp) | Setback Viscosity (cp) | Peak Time (min) | Pasting Temperature (°C) |
|---|---|---|---|---|---|---|---|
| 1 (100RF) | 3010 ± 72.06 e | 1504 ± 109.01 d | 1506 ± 105.47 c | 4049 ± 92.57 e | 2544 ± 49.52 d | 5.6 ± 0.07 a | 78.3 ± 0.08 a |
| 2 (12.5WTF/12.5BTF/75 RF) | 2141 ± 16.17 cd | 1247 ± 24.79 c | 893 ± 25.50 b | 3154 ± 37.64 cd | 1907 ± 26.03 c | 5.7 ± 0.03 abc | 78.4 ± 0.05 a |
| 3 (25WTF/75RF) | 2050 ± 16.00 c | 1237 ± 44.16 c | 813 ± 57.81 b | 3088 ± 24.34 c | 1851 ± 32.42 c | 5.6 ± 0.04 ab | 78.8 ± 0.45 a |
| 4 (25BTF/75RF) | 2213 ± 40.04 d | 1325 ± 82.42 c | 888 ± 66.84 b | 3237 ± 44.38 d | 1912 ± 38.21 c | 5.7 ± 0.00 bc | 78.3 ± 0.08 a |
| 5 (25WTF/25BTF/50RF) | 1558 ± 16.00 a | 1079 ± 17.09 ab | 479 ± 2.00 a | 2622 ± 10.26 b | 1543 ± 8.08 b | 5.8 ± 0.04 cd | 80.0 ± 0.09 a |
| 6 (50WTF/50RF) | 1717 ± 14.22 b | 1210 ± 3.00 bc | 507 ± 11.93 a | 3124 ± 23.52 cd | 1914 ± 20.52 c | 5.9 ± 0.00 d | 85.1 ± 4.48 b |
| 7 (50BTF/50RF) | 1523 ± 9.29 a | 1048 ± 5.51 a | 474 ± 7.09 a | 2353 ± 27.50 a | 1305 ± 27.50 a | 5.7 ± 0.00 bc | 79.7 ± 0.51 a |
| Blends (g 100 g−1) | Cold Peak (cp) | Raw Peak (cp) | Hold (cp) | Breakdown Viscosity (cp) | Final Viscosity (cp) | Setback Viscosity (cp) | Peak Time (min) | Cold Peak Area |
|---|---|---|---|---|---|---|---|---|
| 1 (100RF) | 851 ± 170.41 d | 896 ± 160.04 cd | 61 ± 11.15 a | 835 ± 148.94 cd | 302 ± 44.64 a | 241 ± 33.50 b | 2.1 ± 0.00 a | 1830.3 ± 436.56 c |
| 2 (12.5WTF/12.5 BTF/75RF) | 641 ± 43.02 abc | 642 ± 43.59 abc | 75 ± 40.71 ab | 567 ± 84.18 abc | 263 ± 42.16 a | 188 ± 2.08 a | 2.2 ± 0.15 ab | 1063.2 ± 75.15 ab |
| 3 (25WTF/75RF) | 982 ± 67.73 cd | 987 ± 64.13 d | 111 ± 64.13 b | 876 ± 54.53 d | 298 ± 10.82 a | 187 ± 6.24 a | 2.2 ± 0.10 ab | 1543.2 ± 83.87 bc |
| 4 (25BTF/75RF) | 554 ± 104.20 ab | 504 ± 184.92 a | 77 ± 5.13 ab | 428 ± 180.94 ab | 244 ± 28.73 a | 167 ± 24.27 a | 2.2 ± 0.17 ab | 849.3 ± 200.67 a |
| 5 (25WTF/25 BTF/50RF) | 693 ± 59.03 abcd | 726 ± 56.05 abcd | 109 ± 9.50 ab | 617 ± 61.88 bcd | 317 ± 13.08 a | 208 ± 6.03 ab | 2.5 ± 0.19 bc | 1001.8 ± 73.83 ab |
| 6 (50WTF/50RF) | 372 ± 4.93 a | 537 ± 7.55 ab | 213 ± 8.50 c | 324 ± 2.08 a | 517 ± 6.51 b | 304 ± 3.00 c | 6.2 ± 0.04 d | 642.2 ± 10.34 a |
| 7 (50BTF/50RF) | 762 ± 35.17 bcd | 810 ± 28.69 bcd | 114 ± 11.50 b | 696 ± 32.08 bcd | 306 ± 16.50 a | 193 ± 5.03 a | 2.8 ± 0.14 c | 1115.2 ± 124.89 ab |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín-Diana, A.B.; Blanco Espeso, B.; Jimenez Pulido, I.J.; Acebes Martínez, P.J.; Rico, D. Twin-Screw Extrusion as Hydrothermal Technology for the Development of Gluten-Free Teff Flours: Effect on Antioxidant, Glycaemic Index and Techno-Functional Properties. Foods 2022, 11, 3610. https://doi.org/10.3390/foods11223610
Martín-Diana AB, Blanco Espeso B, Jimenez Pulido IJ, Acebes Martínez PJ, Rico D. Twin-Screw Extrusion as Hydrothermal Technology for the Development of Gluten-Free Teff Flours: Effect on Antioxidant, Glycaemic Index and Techno-Functional Properties. Foods. 2022; 11(22):3610. https://doi.org/10.3390/foods11223610
Chicago/Turabian StyleMartín-Diana, Ana Belén, Belén Blanco Espeso, Ivan Jesus Jimenez Pulido, Pedro J. Acebes Martínez, and Daniel Rico. 2022. "Twin-Screw Extrusion as Hydrothermal Technology for the Development of Gluten-Free Teff Flours: Effect on Antioxidant, Glycaemic Index and Techno-Functional Properties" Foods 11, no. 22: 3610. https://doi.org/10.3390/foods11223610
APA StyleMartín-Diana, A. B., Blanco Espeso, B., Jimenez Pulido, I. J., Acebes Martínez, P. J., & Rico, D. (2022). Twin-Screw Extrusion as Hydrothermal Technology for the Development of Gluten-Free Teff Flours: Effect on Antioxidant, Glycaemic Index and Techno-Functional Properties. Foods, 11(22), 3610. https://doi.org/10.3390/foods11223610







