Effects of Cross-Linking on Physicochemical and Film Properties of Lotus (Nelumbo nucifera G.) Seed Starch
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Starch Isolation
2.3. Cross-Linking of Lotus Seed Starch
2.4. Properties of Native and Cross-Linked Lotus Seed Starch
2.4.1. Degree of Cross-Linking
2.4.2. Amylose Content
2.4.3. Swelling Power and Solubility
2.4.4. Transmittance (%)
2.4.5. Crystalline Structure (XRD)
2.4.6. Pasting Properties
2.4.7. Dynamic Rheological Behavior
2.5. Preparation of LS Films
2.6. Properties of LS Films
2.6.1. Moisture Content and Thickness
2.6.2. Water Vapor Permeability (WVP) of LS Film
2.6.3. Water Solubility
2.6.4. Mechanical Properties
2.6.5. Opacity of LS Films
2.6.6. FTIR Spectral Analysis
2.6.7. Granular Morphology (SEM)
2.6.8. Thermal Properties (DSC)
2.7. Statistical Analysis
3. Results and Discussion
3.1. Native and Cross-Linked Lotus Seed Starch Properties
3.1.1. Determination of Amylose Content (AC) and Degree of Cross-Linking (DC) of LS
3.1.2. Solubility (S) and Swelling Power (SP) of LS
3.1.3. Light Transmittance
3.1.4. Crystalline Structure
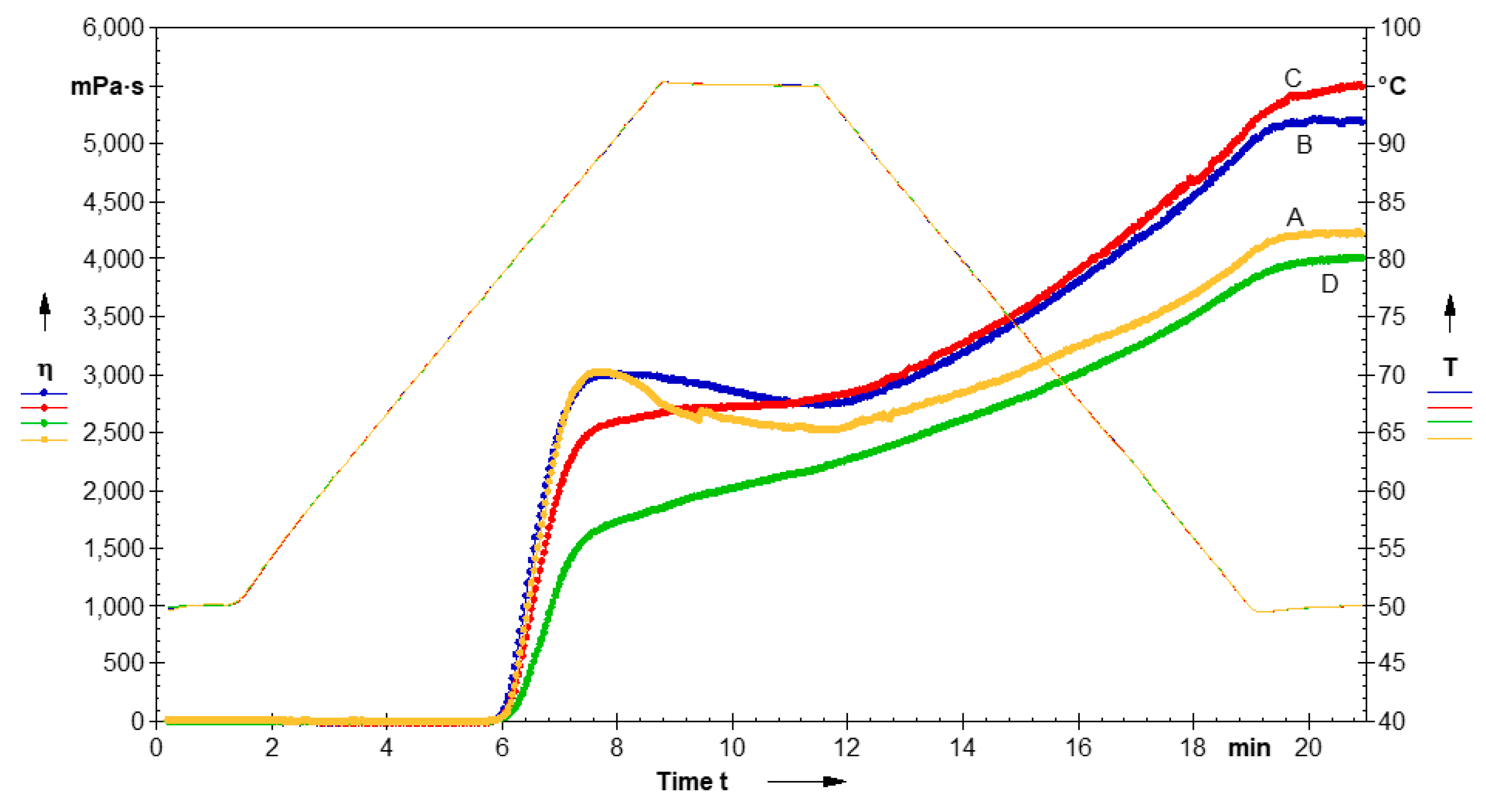
3.1.5. Pasting Parameters
3.1.6. Dynamic Rheological Behavior
3.2. Properties of Lotus Seed Starch Films
3.2.1. Moisture Content (MC), Thickness, Water Solubility, and Transparency of LS Films
3.2.2. Water Vapor Permeability
3.2.3. Mechanical Properties
3.2.4. FTIR Spectral Analysis
3.2.5. Morphological Characteristics
3.2.6. Thermal Properties of LS Films (DSC)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bangar, S.P.; Whiteside, W.S.; Dunno, K.D.; Cavender, G.A.; Dawson, P. Pearl millet starch-based nanocomposite films reinforced with Kudzu cellulose nanocrystals and essential oil: Effect on functionality and biodegradability. Food Res. Int. 2022, 157, 111384. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef]
- Zheng, J.; Suh, S. Strategies to reduce the global carbon footprint of plastics. Nat. Clim. Change 2019, 9, 374–378. [Google Scholar] [CrossRef]
- Emadian, S.M.; Onay, T.T.; Demirel, B. Biodegradation of bioplastics in natural environments. Waste Manag. 2017, 59, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Dhull, S.B.; Bangar, S.P.; Deswal, R.; Dhandhi, P.; Kumar, M.; Trif, M.; Rusu, A. Development and Characterization of Active Native and Cross-Linked Pearl Millet Starch-Based Film Loaded with Fenugreek Oil. Foods 2021, 10, 3097. [Google Scholar] [CrossRef] [PubMed]
- Tarahi, M.; Shahidi, F.; Hedayati, S. A novel starch from bitter vetch (Vicia ervilia) seeds: A comparison of its physicochemical, structural, thermal, rheological, and pasting properties with conventional starches. Int. J. Food Sci. Technol. 2022, 57, 6833–6842. [Google Scholar] [CrossRef]
- Li, M.; Tian, X.; Jin, R.; Li, D. Preparation and characterization of nanocomposite films containing starch and cellulose nanofibers. Ind. Crop. Prod. 2018, 123, 654–660. [Google Scholar] [CrossRef]
- Dhull, S.B.; Chandak, A.; Collins, M.N.; Bangar, S.P.; Chawla, P.; Singh, A. Lotus Seed Starch: A Novel Functional Ingredient with Promising Properties and Applications in Food—A Review. Starch-Stärke 2022, 74, 2200064. [Google Scholar] [CrossRef]
- Bhat, R.; Sridhar, K.R. Nutritional quality evaluation of electron beam-irradiated lotus (Nelumbo nucifera) seeds. Food Chem. 2008, 107, 174–184. [Google Scholar] [CrossRef]
- Kim, M.-J.; Shin, H.-S. Antioxidative effect of lotus seed and seedpod extracts. Food Sci. Biotechnol. 2012, 21, 1761–1766. [Google Scholar] [CrossRef]
- Punia, S.; Dhull, S.B.; Kunner, P.; Rohilla, S. Effect of γ-radiation on physico-chemical, morphological and thermal characteristics of lotus seed (Nelumbo nucifera) starch. Int. J. Biol. Macromol. 2020, 157, 584–590. [Google Scholar] [CrossRef]
- Guo, Z.; Zeng, S.; Lu, X.; Zhou, M.; Zheng, M.; Zheng, B. Structural and physicochemical properties of lotus seed starch treated with ultra-high pressure. Food Chem. 2015, 186, 223–230. [Google Scholar] [CrossRef]
- Man, J.; Cai, J.; Cai, C.; Xu, B.; Huai, H.; Wei, C. Comparison of physicochemical properties of starches from seed and rhizome of lotus. Carbohydr. Polym. 2012, 88, 676–683. [Google Scholar] [CrossRef]
- Guo, Z.B.; Liu, W.T.; Zeng, S.X.; Zheng, B.D. Effect of ultra high pressure processing onthe particle characteristics of lo-tus seed starch. Chin. J. Struct. Chem. 2013, 32, 525–532. [Google Scholar]
- Heo, H.; Lee, Y.-K.; Chang, Y.H. Rheaological, pasting, and structural properties of potato starch by cross-linking. Int. J. Food Prop. 2017, 20, 2138–2150. [Google Scholar] [CrossRef]
- Wang, X.; Huang, L.; Zhang, C.; Deng, Y.; Xie, P.; Liu, L.; Cheng, J. Research advances in chemical modifications of starch for hydrophobicity and its applications: A review. Carbohydr. Polym. 2020, 240, 116292. [Google Scholar] [CrossRef]
- Gui-Jie, M.; Peng, W.; Xiang-Sheng, M.; Xing, Z.; Tong, Z. Crosslinking of corn starch with sodium trimetaphosphate in solid state by microwave irradiation. J. Appl. Polym. Sci. 2006, 102, 5854–5860. [Google Scholar] [CrossRef]
- Lewicka, K.; Siemion, P.; Kurcok, P. Chemical Modifications of Starch: Microwave Effect. Int. J. Polym. Sci. 2015, 5854–5860. [Google Scholar] [CrossRef]
- Atta, O.; Manan, S.; Ahmed, A.; Awad, M.; Ul-Islam, M.; Subhan, F.; Ullah, M.; Yang, G. Development and Characterization of Yeast-Incorporated Antimicrobial Cellulose Biofilms for Edible Food Packaging Application. Polymers 2021, 13, 2310. [Google Scholar] [CrossRef]
- Atta, O.M.; Manan, S.; Ul-Islam, M.; Ahmed, A.A.Q.; Ullah, M.W.; Yang, G. Development and characterization of plant oil-incorporated carboxymethyl cellulose/bacterial cellulose/glycerol-based antimicrobial edible films for food packaging applications. Adv. Compos. Hybrid Mater. 2022, 5, 973–990. [Google Scholar] [CrossRef]
- Garavand, F.; Rouhi, M.; Razavi, S.H.; Cacciotti, I.; Mohammadi, R. Improving the integrity of natural biopolymer films used in food packaging by crosslinking approach: A review. Int. J. Biol. Macromol. 2017, 104, 687–707. [Google Scholar] [CrossRef] [PubMed]
- Woo, K.; Seib, P.A. Cross-linking of wheat starch and hydroxypropylated wheat starch in alkaline slurry with sodium trimetaphosphate. Carbohydr. Polym. 1997, 33, 263–271. [Google Scholar] [CrossRef]
- Kaur, L.; Singh, J.; Singh, N. Effect of cross-linking on some properties of potato (Solanum tuberosum L.) starches. J. Sci. Food Agric. 2006, 86, 1945–1954. [Google Scholar] [CrossRef]
- Williams, P.C.; Kuzina, F.D.; Hlynka, I. Rapid colorimetric procedure for estimating the amylose content of starches and flours. Cereal Chem. 1970, 411–420. [Google Scholar]
- Leach, H.W. Structure of starch granules. I. Swelling and solubility patterns of various starches. Cereal Chem. 1959, 36, 534–544. [Google Scholar]
- Perera, C.; Hoover, R. Influence of hydroxypropylation on retrogradation properties of native, defatted and heat-moisture treated potato starches. Food Chem. 1999, 64, 361–375. [Google Scholar] [CrossRef]
- Kaur, M.; Singh, S. Physicochemical, Morphological, Pasting, and Rheological Properties of Tamarind (Tamarindus indica L.) Kernel Starch. Int. J. Food Prop. 2016, 19, 2432–2442. [Google Scholar] [CrossRef]
- ASTM. E 96/E96M-15; Standard Test Methods for Water Vapor Transmission of 479 Materials. ASTM: Philadelphia, PA, USA, 2015; pp. 1–13.
- Sukhija, S.; Singh, S.; Riar, C.S. Development and characterization of biodegradable films from whey protein concentrate, psyllium husk and oxidized, crosslinked, dual-modified lotus rhizome starch composite. J. Sci. Food Agric. 2019, 99, 3398–3409. [Google Scholar] [CrossRef]
- Romero-Bastida, C.A.; Bello-Pérez, L.A.; García, M.A.; Martino, M.N.; Solorza-Feria, J.; Zaritzky, N.E. Physicochemical and microstructural characterization of films prepared by thermal and cold gelatinization from non-conventional sources of starches. Carbohydr. Polym. 2005, 60, 235–244. [Google Scholar] [CrossRef]
- ASTM D882-12; Standard Test Method for Tensile Properties of Thin Plastic Sheeting. ASTM: Philadelphia, PA, USA, 2012; pp. 1–11.
- Han, J.H.; Floros, J.D. Casting Antimicrobial Packaging Films and Measuring Their Physical Properties and Antimicrobial Activity. J. Plast. Film Sheeting 1997, 13, 287–298. [Google Scholar] [CrossRef]
- Sharma, V.; Kaur, M.; Sandhu, K.S.; Godara, S.K. Effect of cross-linking on physico-chemical, thermal, pasting, in vitro digestibility and film forming properties of Faba bean (Vicia faba L.) starch. Int. J. Biol. Macromol. 2020, 159, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Hazarika, B.J.; Sit, N. Effect of dual modification with hydroxypropylation and cross-linking on physicochemical properties of taro starch. Carbohydr. Polym. 2016, 140, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, H.; Shad, M.A.; Saleem, S.; Khan, M.U.A.; Nishan, U.; Rasheed, T.; Bilal, M.; Iqbal, H.M. Characteristics of starch isolated from microwave heat treated lotus (Nelumbo nucifera) seed flour. Int. J. Biol. Macromol. 2018, 113, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Punia, S.; Kumar, M.; Siroha, A.K.; Kennedy, J.F.; Dhull, S.B.; Whiteside, W.S. Pearl millet grain as an emerging source of starch: A review on its structure, physicochemical properties, functionalization, and industrial applications. Carbohydr. Polym. 2021, 260, 117776. [Google Scholar] [CrossRef] [PubMed]
- Jyothi, A.N.; Moorthy, S.N.; Rajasekharan, K.N. Effect of Cross-linking with Epichlorohydrin on the Properties of Cassava (Manihot esculenta Crantz) Starch. Starch/Stärke 2006, 58, 292–299. [Google Scholar] [CrossRef]
- Liu, J.; Wang, B.; Lin, L.; Zhang, J.; Liu, W.; Xie, J.; Ding, Y. Functional, physicochemical properties and structure of cross-linked oxidized maize starch. Food Hydrocoll. 2014, 36, 45–52. [Google Scholar] [CrossRef]
- Li, L.; He, S.; Lin, Y.; Zheng, B.; Zhang, Y.; Zeng, H. A novel lotus seed cross-linked resistant starch: Structural, physicochemical and digestive properties. Front. Nutr. 2022, 9, 989042. [Google Scholar] [CrossRef]
- Hoover, R.; Sosulski, F. Effect of Cross-Linking on Functional Properties of Legume Starches. Starch-Stärke 1986, 38, 149–155. [Google Scholar] [CrossRef]
- Chen, B.; Dang, L.; Zhang, X.; Fang, W.; Hou, M.; Liu, T.; Wang, Z. Physicochemical properties and micro-structural characteristics in starch from kudzu root as affected by cross-linking. Food Chem. 2017, 219, 93–101. [Google Scholar] [CrossRef]
- Wongsagonsup, R.; Pujchakarn, T.; Jitrakbumrung, S.; Chaiwat, W.; Fuongfuchat, A.; Varavinit, S.; Dangtip, S.; Suphantharika, M. Effect of cross-linking on physicochemical properties of tapioca starch and its application in soup product. Carbohydr. Polym. 2014, 101, 656–665. [Google Scholar] [CrossRef]
- Muhammad, K.; Hussin, F.; Man, Y.; Ghazali, H.; Kennedy, J. Effect of pH on phosphorylation of sago starch. Carbohydr. Polym. 2000, 42, 85–90. [Google Scholar] [CrossRef]
- Clerici, M.T.P.S.; Sampaio, U.M.; Schmiele, M. Identification and Analysis of Starch. In Starches for Food Application; Academic Press: Cambridge, MA, USA, 2018; pp. 23–69. [Google Scholar] [CrossRef]
- Kim, B.-Y.; Yoo, B. Effects of cross-linking on the rheological and thermal properties of sweet potato starch. Starch/Stärke 2010, 62, 577–583. [Google Scholar] [CrossRef]
- Gutiérrez, T.J.; Tapia, M.S.; Pérez, E.; Famá, L. Structural and mechanical properties of edible films made from native and modified cush-cush yam and cassava starch. Food Hydrocoll. 2015, 45, 211–217. [Google Scholar] [CrossRef]
- El Halal, S.L.M.; Colussi, R.; Biduski, B.; Evangelho, J.A.D.; Bruni, G.P.; Antunes, M.D.; Dias, A.R.G.; Zavareze, E.D.R. Morphological, mechanical, barrier and properties of films based on acetylated starch and cellulose from barley. J. Sci. Food Agric. 2016, 97, 411–419. [Google Scholar] [CrossRef]
- Garcia, P.S.; Grossmann, M.V.E.; Shirai, M.A.; Lazaretti, M.M.; Yamashita, F.; Muller, C.M.O.; Mali, S. Improving action of citric acid as compatibiliser in starch/polyester blown films. Indus. Crops Pdts. 2014, 52, 305–312. [Google Scholar] [CrossRef]
- Li, J.; Ye, F.; Liu, J.; Zhao, G. Effects of octenylsuccination on physical, mechanical and moisture-proof properties of stretchable sweet potato starch film. Food Hydrocoll. 2015, 46, 226–232. [Google Scholar] [CrossRef]
- Mali, S.; Sakanaka, L.; Yamashita, F.; Grossmann, M. Water sorption and mechanical properties of cassava starch films and their relation to plasticizing effect. Carbohydr. Polym. 2005, 60, 283–289. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Citric acid cross-linking of starch films. Food Chem. 2010, 118, 702–711. [Google Scholar] [CrossRef]
- Liu, Y.F.; Laohasongkram, K.; Chaiwanichsiri, S. Effects of Heat-Moisture Treatment on Molecular Interactions and Physicochemical Properties of Tapioca Starch. MOJ Food Process. Technol. 2016, 3, 304–311. [Google Scholar] [CrossRef]
- Detduangchan, N.; Sridach, W.; Wittaya, T. Enhancement of the properties of biodegradable rice starch films by using chemical crosslinking agents. Int. Food Res. J. 2014, 21, 1225–1235. [Google Scholar]
- Bruni, G.P.; De Oliveira, J.P.; El Halal, S.L.M.; Flores, W.H.; Gundel, A.; De Miranda, M.Z.; Dias, A.R.G.; Zavareze, E.D.R. Phosphorylated and Cross-Linked Wheat Starches in the Presence of Polyethylene Oxide and Their Application in Biocomposite Films. Starch-Stärke. 2018, 70, 1700192. [Google Scholar] [CrossRef]
- Sukhija, S.; Singh, S.; Riar, C.S. Physical, Mechanical, Morphological, and Barrier Properties of Elephant Foot Yam Starch, Whey Protein Concentrate and psyllium Husk Based Composite Biodegradable Films. Polym. Compos. 2017, 39, E407–E415. [Google Scholar] [CrossRef]
- Mathew, S.; Brahmakumar, M.; Abraham, T.E. Microstructural imaging and characterization of the mechanical, chemical, thermal, and swelling properties of starch–chitosan blend films. Biopolymers 2006, 82, 176–187. [Google Scholar] [CrossRef]
- Wu, J.; Zhong, F.; Li, Y.; Shoemaker, C.; Xia, W. Preparation and characterization of pullulan–chitosan and pullulan–carboxymethyl chitosan blended films. Food Hydrocoll. 2012, 30, 82–91. [Google Scholar] [CrossRef]
- Ramos, L.; Reinas, I.; Silva, S.I.; Fernandes, J.C.; Cerqueira, M.A.; Pereira, R.N.; Vicente, A.A.; Poças, M.F.; Pintado, M.E.; Malcata, F.X. Effect of whey protein purity and glycerol content upon physical properties of edible films manufactured therefrom. Food Hydrocoll. 2012, 30, 110–122. [Google Scholar] [CrossRef]
- Liang, J.; Wang, R.; Chen, R. The Impact of Cross-linking Mode on the Physical and Antimicrobial Properties of a Chitosan/Bacterial Cellulose Composite. Polymers 2019, 11, 491. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Canisag, H.; Mu, B.; Yang, Y. Robust and Flexible Films from 100% Starch Cross-Linked by Biobased Disaccharide Derivative. ACS Sustain. Chem. Eng. 2015, 3, 2631–2639. [Google Scholar] [CrossRef]

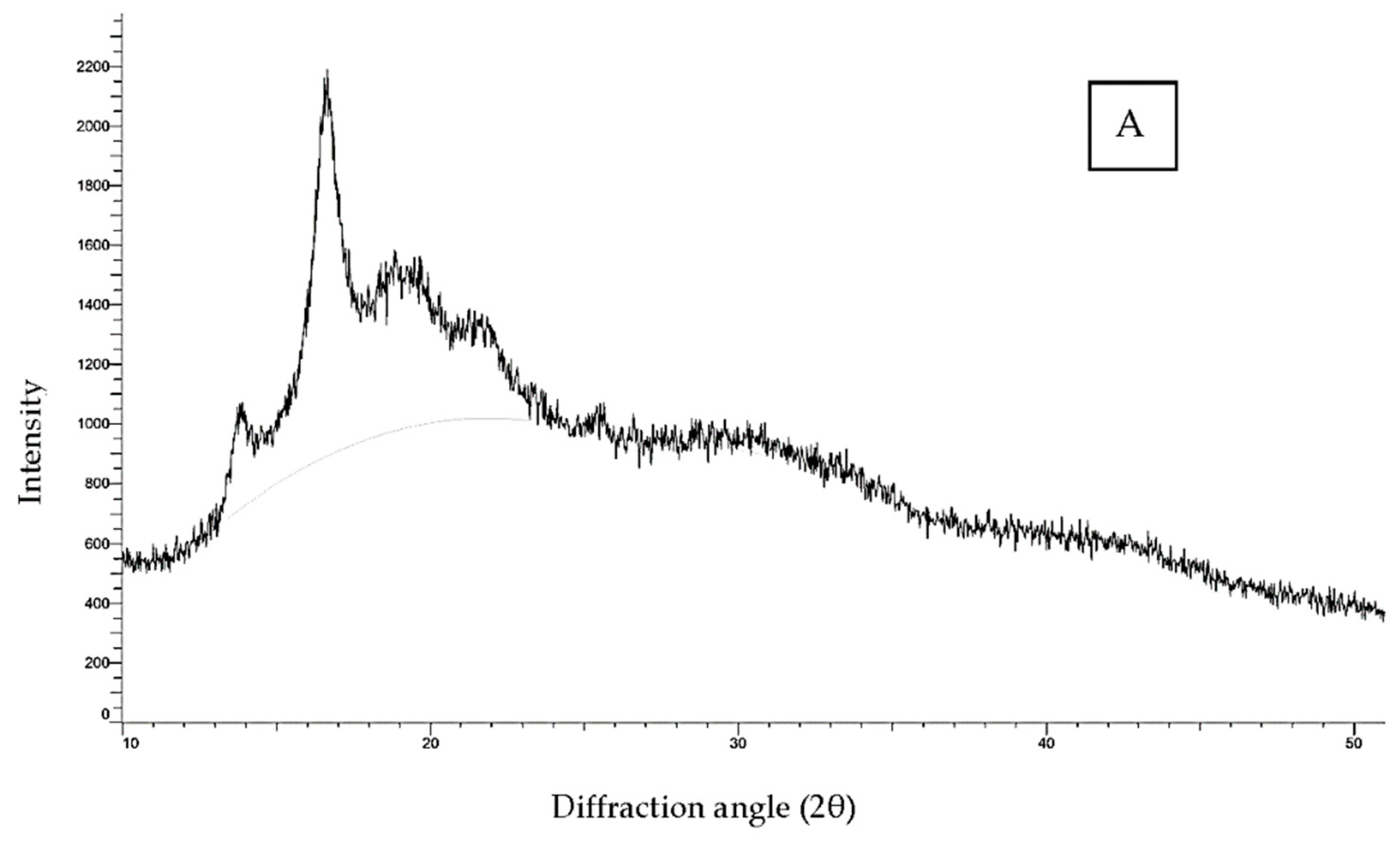
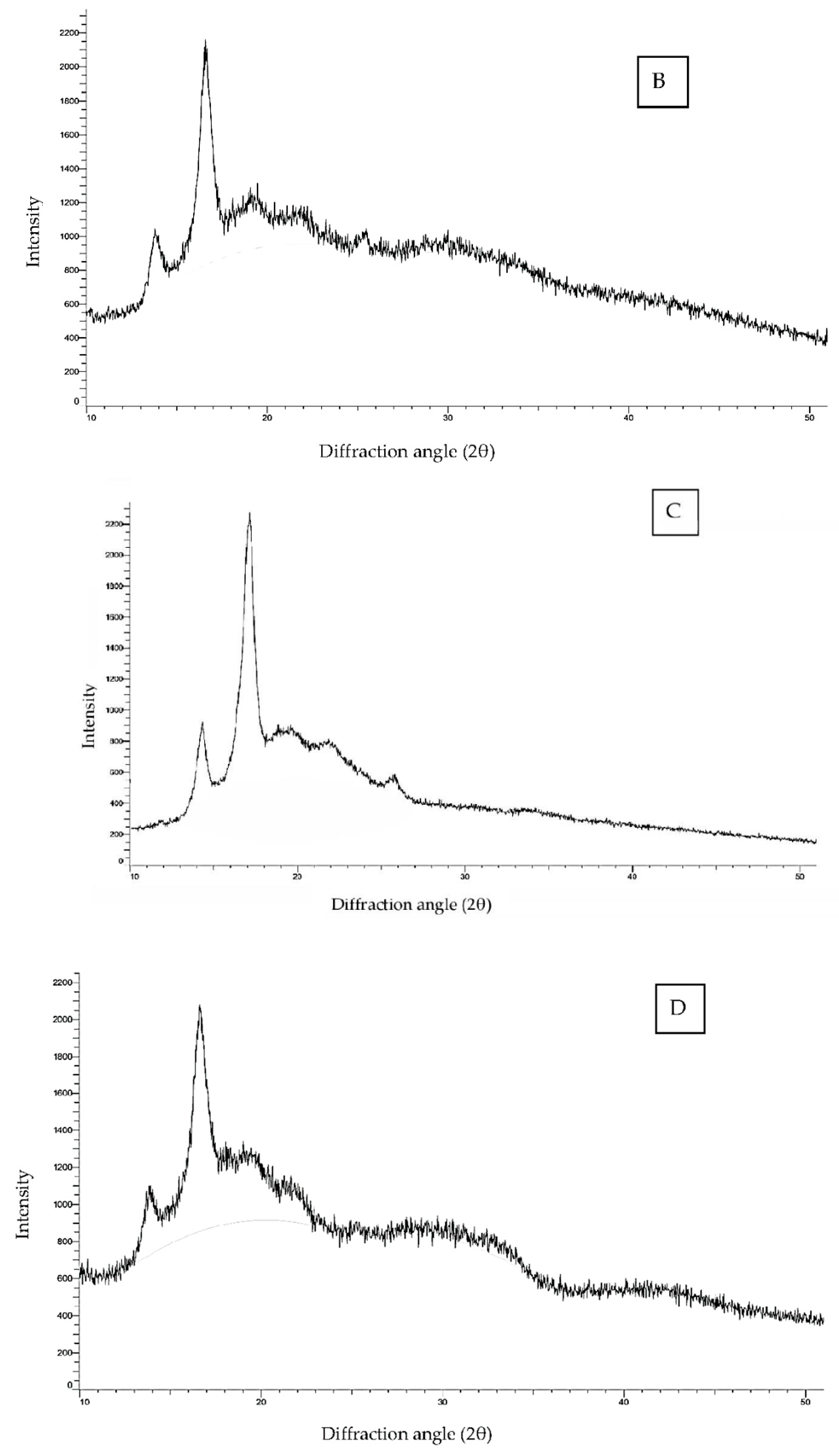

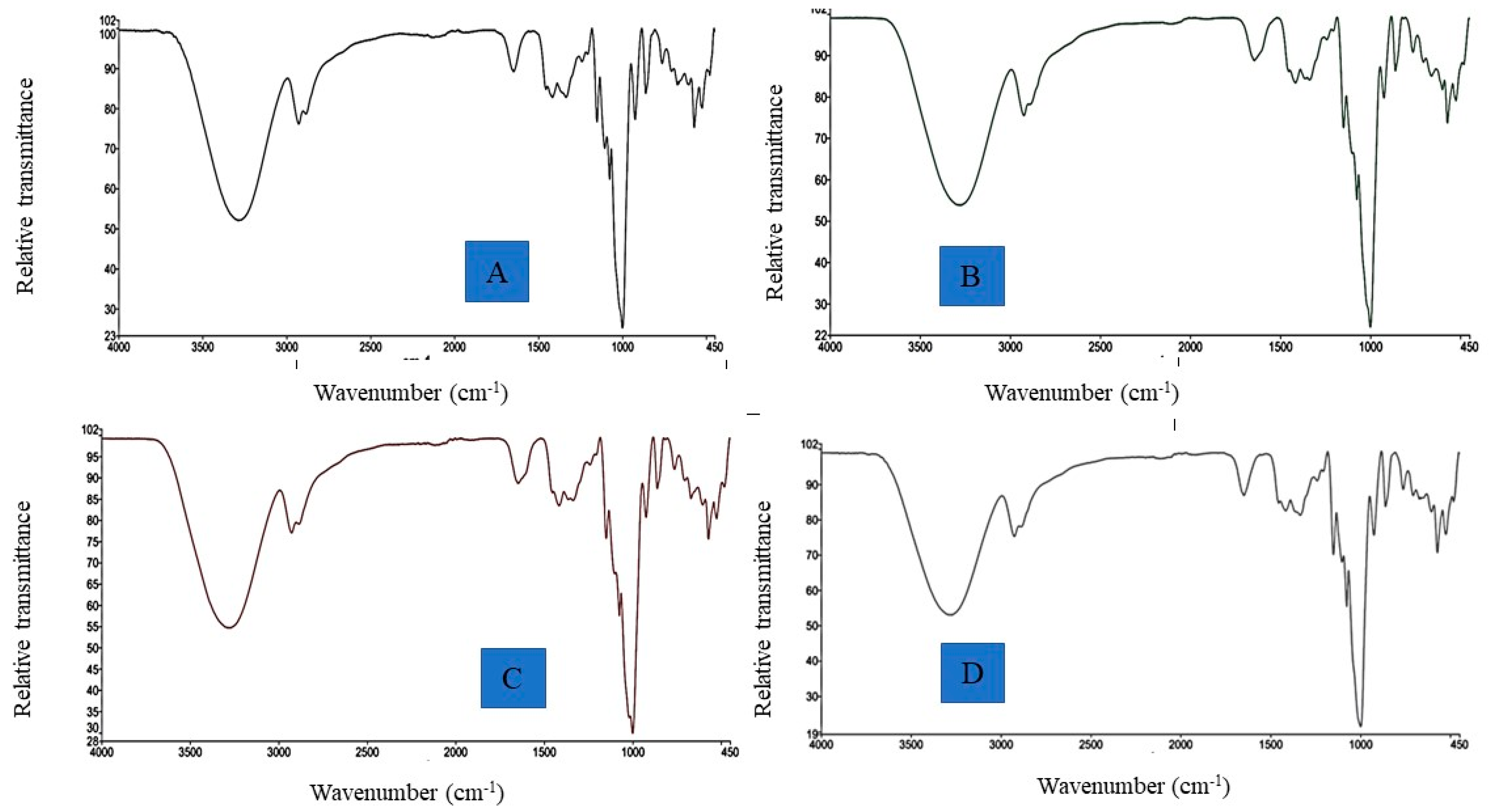

| Starch Sample | Degree of Cross-Linking (%) | Amylose Content (%) | Swelling Power (90 °C) (g/g) | Solubility (90 °C) (%) | Relative Crystallinity (%) |
|---|---|---|---|---|---|
| Native LS | - | 27.98 ± 0.10 a | 15.02 ± 0.21 a | 16.00 ± 0.13 a | 30.27 ± 0.40 a |
| CL-LS-1 | 2.58 ± 0.03 c | 25.16 ± 0.12 b | 14.83 ± 0.19 b | 15.44 ± 0.12 b | 29.21 ± 0.45 b |
| CL-LS-3 | 15.77 ± 0.16 b | 19.36 ± 0.13 c | 12.44 ± 0.11 c | 15.35 ± 0.14 bc | 29.15 ± 0.55 b |
| CL-LS-5 | 45.84 ± 0.21 a | 13.45 ± 0.15 d | 9.05 ± 0.23 d | 15.13 ± 0.15 c | 29.01 ± 0.42 b |
| Sample | PV (MPa·s) | TV (MPa·s) | BDV (MPa·s) | SBV (MPa·s) | FV (MPa·s) | PT (°C) |
|---|---|---|---|---|---|---|
| Native LS | 3028.51 ± 34 a | 2666.27 ± 35 b | 362.24 ± 17 a | 2085.57 ± 31 b | 4751.84 ± 40 c | 79.13 ± 0.03 c |
| CL-LS-1 | 2950.43 ± 33 b | 2737 ± 21 a | 213.43 ± 12 b | 2458 ± 38 a | 5195 ± 41 b | 79.7 ± 0.07 a |
| CL-LS-3 | 2550.80 ± 28 c | - | - | - | 5489.24 ± 37 a | 79.7 ± 0.06 a |
| CL-LS-5 | 1640.22 ± 19 d | - | - | - | 4007.86 ± 39 d | 79.52 ± 0.06 b |
| Sample | Storage Modulus G′ (Pa) | Loss Modulus G″ (Pa) | tanδ (G″/G′) |
|---|---|---|---|
| Native LS | 1784 ± 14 c | 144.63 ± 3 a | 0.08 |
| CL-LS-1 | 1900 ± 18 a | 140.92 ± 2 a | 0.07 |
| CL-LS-3 | 1830 ± 15 b | 136.31 ± 3 b | 0.07 |
| CL-LS-5 | 1796 ± 15 c | 136.52 ± 2 b | 0.07 |
| Film Sample | Moisture Content (%) | Thickness (mm) | Water Solubility (%) | Opacity (%) | WVP (g·m/Pa·s·m2) | Tensile Strength (MPa) | Elongation at Break (%) |
|---|---|---|---|---|---|---|---|
| Native LSF | 25.12 ± 0.16 a | 0.10 ± 0.00 c | 48.12 ± 0.21 a | 1.57 ± 0.01 d | 1.58 ± 0.01 a | 6.12 ± 0.12 d | 37.52 ± 1.34 a |
| CL-LSF-1 | 23.34 ± 0.20 b | 0.13 ± 0.01 b | 35.65 ± 0.19 b | 2.59 ± 0.02 a | 1.48 ± 0.03 b | 10.45 ± 0.15 c | 35.54 ± 1.52 a |
| CL-LSF-3 | 22.22 ± 0.14 c | 0.14 ± 0.02 ab | 33.45 ± 0.16 c | 2.27 ± 0.01 b | 1.41 ± 0.05 c | 11.09 ± 0.13 b | 30.91 ± 0.88 b |
| CL-LSF-5 | 20.13 ± 0.22 d | 0.15 ± 0.01 a | 29.23 ± 0.11 d | 2.07 ± 0.02 c | 1.45 ± 0.02 bc | 12.52 ± 0.14 a | 26.11 ± 0.52 c |
| Sample | To (°C) | Tp (°C) | Tc (°C) | ΔHgel (J/g) |
|---|---|---|---|---|
| Native LSF | 30.08 ± 0.9 b | 67.30 ± 0.11 d | 104.82 ± 0.17 d | 152.70 ± 0.18 d |
| CL-LSF-1 | 32.04 ± 0.3 a | 74.49 ± 0.21 c | 122.57 ± 0.14 c | 177.91 ± 0.22 c |
| CL-LSF-3 | 30.19 ± 0.1 b | 76.78 ± 0.19 b | 123.06 ± 0.16 b | 187.47 ± 0.13 b |
| CL-LSF-5 | 32.47 ± 0.5 a | 83.58 ± 0.18 a | 127.34 ± 0.15 a | 214.16 ± 0.27 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chandak, A.; Dhull, S.B.; Punia Bangar, S.; Rusu, A.V. Effects of Cross-Linking on Physicochemical and Film Properties of Lotus (Nelumbo nucifera G.) Seed Starch. Foods 2022, 11, 3069. https://doi.org/10.3390/foods11193069
Chandak A, Dhull SB, Punia Bangar S, Rusu AV. Effects of Cross-Linking on Physicochemical and Film Properties of Lotus (Nelumbo nucifera G.) Seed Starch. Foods. 2022; 11(19):3069. https://doi.org/10.3390/foods11193069
Chicago/Turabian StyleChandak, Ankita, Sanju Bala Dhull, Sneh Punia Bangar, and Alexandru Vasile Rusu. 2022. "Effects of Cross-Linking on Physicochemical and Film Properties of Lotus (Nelumbo nucifera G.) Seed Starch" Foods 11, no. 19: 3069. https://doi.org/10.3390/foods11193069
APA StyleChandak, A., Dhull, S. B., Punia Bangar, S., & Rusu, A. V. (2022). Effects of Cross-Linking on Physicochemical and Film Properties of Lotus (Nelumbo nucifera G.) Seed Starch. Foods, 11(19), 3069. https://doi.org/10.3390/foods11193069








