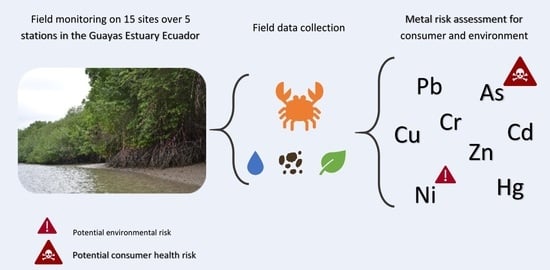
From Mangrove to Fork: Metal Presence in the Guayas Estuary (Ecuador) and Commercial Mangrove Crabs
Abstract
:1. Introduction
2. Materials and Methods
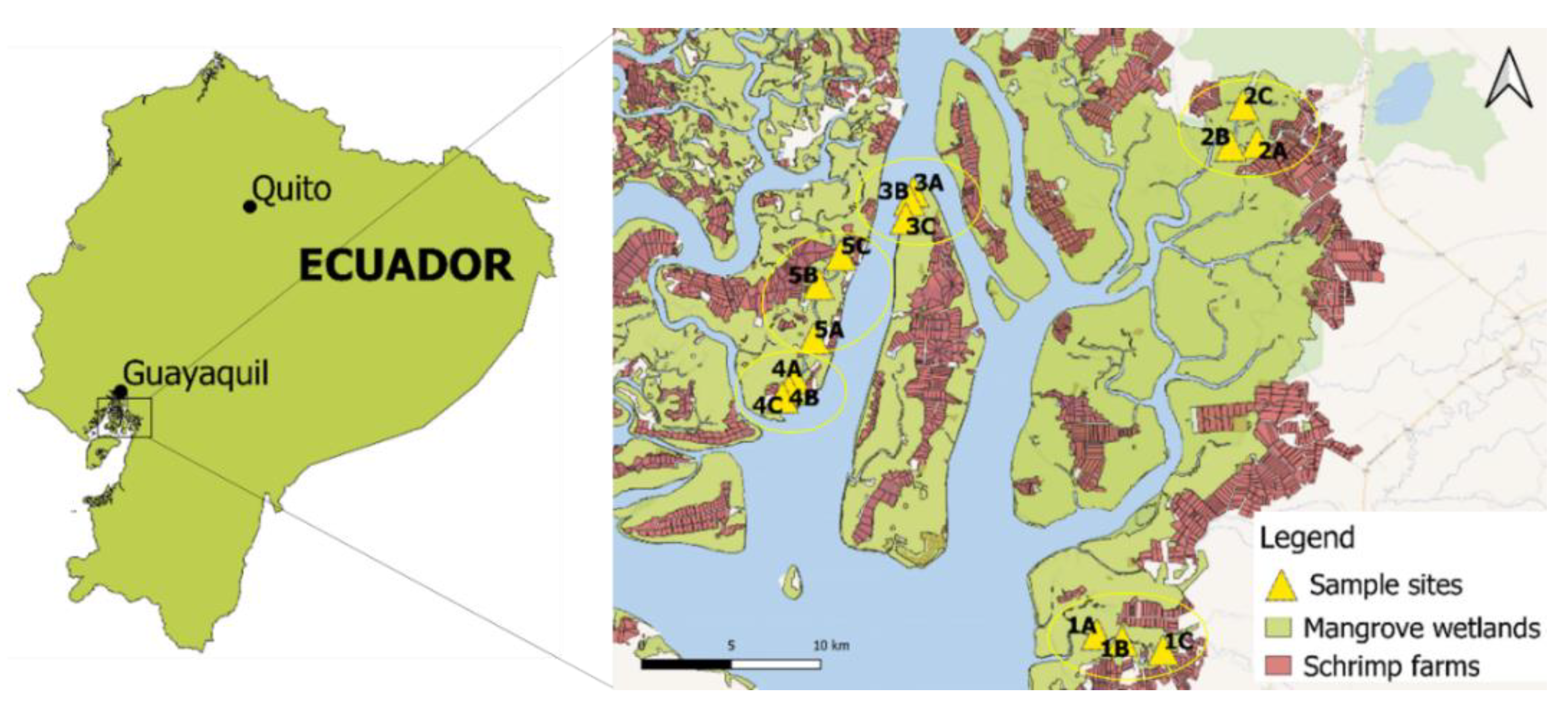
2.1. Study Area
2.2. Sampling Methodology
2.3. Sample Preparation
2.4. Metal Analysis
2.5. Environmental Evaluation Based on National and International Indices
2.6. Consumer Health Risk Assessment
2.6.1. Exposure Assessment
2.6.2. Risk Characterization
2.7. Data Analysis
3. Results
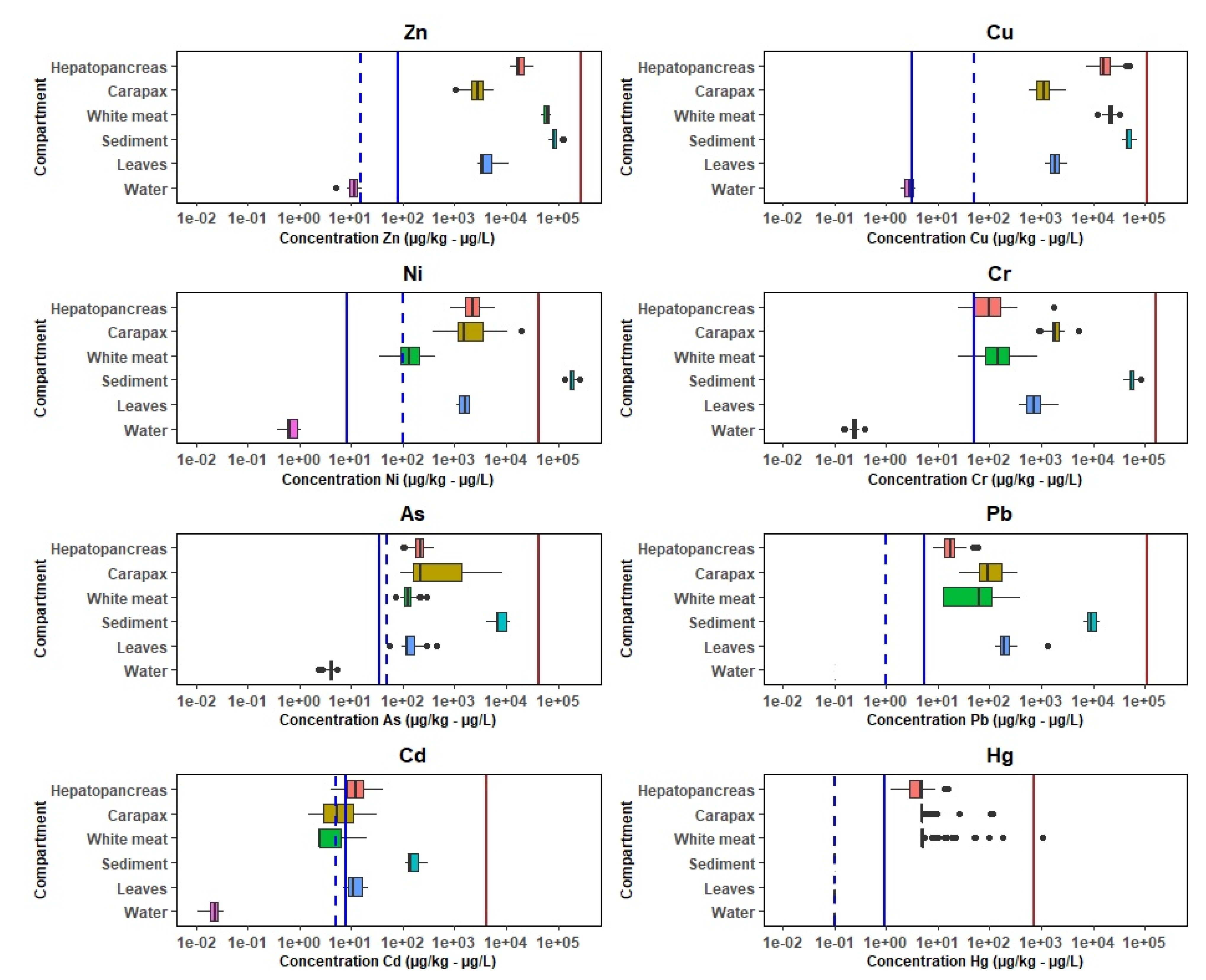
3.1. Metals in Environmental and Crab Matrix
3.1.1. Environmental Components
3.1.2. Crab Matrix
3.2. Spatial Distribution
3.3. Metal Pollution in the Mangrove Wetland
3.4. Consumer Health Risk Assessment
4. Discussion
4.1. Metal Distribution in the Mangrove Wetlands
4.2. Spatial Distribution
4.3. Health Risk Assessment
4.4. Sustainable Development in the Estuarine Basin
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- UN-Water, U. United Nations World Water Development Report 2020: Water and Climate Change; UNESCO: Paris, France, 2020. [Google Scholar]
- Bayen, S. Occurrence, bioavailability and toxic effects of trace metals and organic contaminants in mangrove ecosystems: A review. Environ. Int. 2012, 48, 84–101. [Google Scholar] [CrossRef]
- Worthington, T.A.; Zu Ermgassen, P.S.; Friess, D.A.; Krauss, K.W.; Lovelock, C.E.; Thorley, J.; Tingey, R.; Woodroffe, C.D.; Bunting, P.; Cormier, N. A global biophysical typology of mangroves and its relevance for ecosystem structure and deforestation. Sci. Rep. 2020, 10, 14652. [Google Scholar] [CrossRef]
- Menéndez, P.; Losada, I.J.; Torres-Ortega, S.; Narayan, S.; Beck, M.W. The global flood protection benefits of mangroves. Sci. Rep. 2020, 10, 4404. [Google Scholar] [CrossRef]
- Fernández-Cadena, J.C.; Andrade, S.; Silva-Coello, C.; De la Iglesia, R. Heavy metal concentration in mangrove surface sediments from the north-west coast of South America. Mar. Pollut. Bull. 2014, 82, 221–226. [Google Scholar] [CrossRef]
- Bouillon, S. Storage beneath mangroves. Nat. Geosci. 2011, 4, 282–283. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Fang, Z.; Li, J.; Sui, T.; Lin, L.; Xu, X. Copper, zinc, manganese, cadmium and chromium in crabs from the mangrove wetlands in Qi’ao Island, South China: Levels, bioaccumulation and dietary exposure. Watershed Ecol. Environ. 2019, 1, 26–32. [Google Scholar] [CrossRef]
- Qadir, M.; Wichelns, D.; Raschid-Sally, L.; McCornick, P.G.; Drechsel, P.; Bahri, A.; Minhas, P. The challenges of wastewater irrigation in developing countries. Agric. Water Manag. 2010, 97, 561–568. [Google Scholar] [CrossRef] [Green Version]
- Forio, M.A.E.; Goethals, P.L. An Integrated Approach of Multi-Community Monitoring and Assessment of Aquatic Ecosystems to Support Sustainable Development. Sustainability 2020, 12, 5603. [Google Scholar] [CrossRef]
- Deknock, A.; De Troyer, N.; Houbraken, M.; Dominguez-Granda, L.; Nolivos, I.; Van Echelpoel, W.; Forio, M.A.E.; Spanoghe, P.; Goethals, P. Distribution of agricultural pesticides in the freshwater environment of the Guayas river basin (Ecuador). Sci. Total. Environ. 2019, 646, 996–1008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barraza, F.; Maurice, L.; Uzu, G.; Becerra, S.; López, F.; Ochoa-Herrera, V.; Ruales, J.; Schreck, E. Distribution, contents and health risk assessment of metal (loid) s in small-scale farms in the Ecuadorian Amazon: An insight into impacts of oil activities. Sci. Total. Environ. 2018, 622, 106–120. [Google Scholar] [CrossRef]
- Capparelli, M.V.; Moulatlet, G.M.; de Souza Abessa, D.M.; Lucas-Solis, O.; Rosero, B.; Galarza, E.; Tuba, D.; Carpintero, N.; Ochoa-Herrera, V.; Cipriani-Avila, I. An integrative approach to identify the impacts of multiple metal contamination sources on the Eastern Andean foothills of the Ecuadorian Amazonia. Sci. Total. Environ. 2020, 709, 136088. [Google Scholar] [CrossRef]
- Ochoa, M.; Tierra, W.; Tupuna-Yerovi, D.S.; Guanoluisa, D.; Otero, X.L.; Ruales, J. Assessment of cadmium and lead contamination in rice farming soils and rice (Oryza sativa L.) from Guayas province in Ecuador. Environ. Pollut. 2020, 260, 114050. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.W.; Xu, X.R.; Sun, Y.X.; Yu, S.; Chen, Y.S.; Peng, J.X. Heavy metal and organic contaminants in mangrove ecosystems of China: A review. Environ. Sci. Pollut. Res. 2014, 21, 11938–11950. [Google Scholar] [CrossRef]
- Navarrete-Forero, G.; Baren, L.M.; Dominguez-Granda, L.; Cevallos, J.P.; Jarrín, J.R.M. Heavy metals contamination in the gulf of Guayaquil: Even limited data reflects environmental impacts from athropogenic activity Rev. Int. Contam. Ambient. 2019, 35, 731–755. [Google Scholar] [CrossRef]
- Storelli, M. Potential human health risks from metals (Hg, Cd, and Pb) and polychlorinated biphenyls (PCBs) via seafood consumption: Estimation of target hazard quotients (THQs) and toxic equivalents (TEQs). Food Chem. Toxicol. 2008, 46, 2782–2788. [Google Scholar] [CrossRef] [PubMed]
- Sandilyan, S.; Kathiresan, K. Decline of mangroves—A threat of heavy metal poisoning in Asia. Ocean. Coast. Manag. 2014, 102, 161–168. [Google Scholar] [CrossRef]
- Ali, H.; Khan, E.; Ilahi, I. Environmental chemistry and ecotoxicology of hazardous heavy metals: Environmental persistence, toxicity, and bioaccumulation. J. Chem. 2019, 2019. [Google Scholar] [CrossRef] [Green Version]
- Fatima, S.; Muzammal, M.; Rehman, A.; Rustam, S.A.; Shehzadi, Z.; Mehmood, A.; Waqar, M. Water pollution on heavy metals and its effects on fishes. Int. J. Fish. Aquat. Stud. 2020, 8, 6–14. [Google Scholar]
- Mamtani, R.; Stern, P.; Dawood, I.; Cheema, S. Metals and disease: A global primary health care perspective. J. Toxicol. 2011, 2011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lebelo, K.; Malebo, N.; Mochane, M.J.; Masinde, M. Chemical Contamination Pathways and the Food Safety Implications along the Various Stages of Food Production: A Review. Int. J. Environ. Res. Public Health 2021, 18, 5795. [Google Scholar] [CrossRef]
- Hou, D.; O’Connor, D.; Igalavithana, A.D.; Alessi, D.S.; Luo, J.; Tsang, D.C.; Sparks, D.L.; Yamauchi, Y.; Rinklebe, J.; Ok, Y.S. Metal contamination and bioremediation of agricultural soils for food safety and sustainability. Nat. Rev. Earth Environ. 2020, 7, 1–16. [Google Scholar] [CrossRef]
- Singh, J.; Kalamdhad, A.S. Effects of heavy metals on soil, plants, human health and aquatic life. Int. J. Res. Chem. Environ. 2011, 1, 15–21. [Google Scholar]
- Jan, A.T.; Azam, M.; Siddiqui, K.; Ali, A.; Choi, I.; Haq, Q.M. Heavy metals and human health: Mechanistic insight into toxicity and counter defense system of antioxidants. Int. J. Mol. Sci. 2015, 16, 29592–29630. [Google Scholar] [CrossRef] [Green Version]
- Caito, S.; Aschner, M. Neurotoxicity of metals. Handb. Clin. Neurol. 2015, 131, 169–189. [Google Scholar]
- Beltrame, M.O.; De Marco, S.G.; Marcovecchio, J.E. Influences of sex, habitat, and seasonality on heavy-metal concentrations in the burrowing crab (Neohelice granulata) from a coastal lagoon in Argentina. Arch. Environ. Contam. Toxicol. 2010, 58, 746–756. [Google Scholar] [CrossRef]
- Forio, M.A.E.; Villa-Cox, G.; Van Echelpoel, W.; Ryckebusch, H.; Lock, K.; Spanoghe, P.; Deknock, A.; De Troyer, N.; Nolivos-Alvarez, I.; Dominguez-Granda, L. Bayesian Belief Network models as trade-off tools of ecosystem services in the Guayas River Basin in Ecuador. Ecosyst. Serv. 2020, 44, 101124. [Google Scholar] [CrossRef]
- Solano, F. Aspectos Pesqueros, Biológicos y Socioeconómicos de la Captura de Cangrejo Rojo (Ucides occidentalis) en los Manglares del Ecuador; Informe Técnico; Instituto Nacional de Pesca: Guayaquil, Ecuador, 2006; Volume 20, pp. 1–15. [Google Scholar]
- Flores, J. Cadena de Valor del Cangrejo Rojo en el Golfo de Guayaquil; Reporte Técnico, Quito: Pichincha, Ecuador, 2012. [Google Scholar]
- IWMI. Recycling Realities: Managing Health Risks to Make Wastewater an Asset; Briefing, W.P., Ed.; International Water Management Institute: Colombo, Sri Lanka, 2006. [Google Scholar]
- WHO. WHO Guidelines for the Safe Use of Wastewater, Excreta and Grey Water; World Health Organization Press: Geneva, Switzerland, 2006. [Google Scholar]
- Bakan, G.; Özkoç, H.B. An ecological risk assessment of the impact of heavy metals in surface sediments on biota from the mid-Black Sea coast of Turkey. Int. J. Environ. Stud. 2007, 64, 45–57. [Google Scholar] [CrossRef]
- Nguyen, H.L.; Leermakers, M.; Osán, J.; Török, S.; Baeyens, W. Heavy metals in Lake Balaton: Water column, suspended matter, sediment and biota. Sci. Total. Environ. 2005, 340, 213–230. [Google Scholar] [CrossRef]
- Emenike, P.C.; Tenebe, I.T.; Neris, J.B.; Omole, D.O.; Afolayan, O.; Okeke, C.; Emenike, I.K. An integrated assessment of land-use change impact, seasonal variation of pollution indices and human health risk of selected toxic elements in sediments of River Atuwara, Nigeria. Environ. Pollut. 2020, 265 Pt B, 114795. [Google Scholar] [CrossRef]
- Costa, R.G.; Araújo, C.F.D.S.; Ferreol Bah, A.H.; Junior, E.A.G.; Rodrigues, Y.J.M.; Menezes-Filho, J.A. Lead in mangrove root crab (Goniopsis cruentata) and risk assessment due to exposure for estuarine villagers. Food Addit. Contam. Part B 2018, 11, 293–301. [Google Scholar] [CrossRef]
- Gutiérrez-Ravelo, A.; Gutiérrez, Á.J.; Paz, S.; Carrascosa-Iruzubieta, C.; González-Weller, D.; Caballero, J.M.; Revert, C.; Rubio, C.; Hardisson, A. Toxic metals (Al, cd, pb) and trace element (b, ba, co, cu, cr, fe, li, mn, mo, ni, sr, v, zn) levels in sarpa salpa from the north-eastern atlantic ocean region. Int. J. Environ. Res. Public Health 2020, 17, 7212. [Google Scholar] [CrossRef]
- Zambrano, R.; Meiners, C. Notas sobre taxonomía, biología y pesquería de Ucides occidentalis (Brachyura: Ocypodidae) con énfasis en el Golfo de Guayaquil, Ecuador. Rev. Peru. Biol. 2018, 25, 55–66. [Google Scholar] [CrossRef] [Green Version]
- Pontón-Cevallos, J.; Jarrín, J.R.M.; MishellRosado-Moncayo, A.; JoséBonifaz, M.; del MarQuiroga, M.; EstherEspinoza, M.; JuliaBorbor-Córdova, M.; Pozo-Cajas, M.; Goethals, P.L.M.; Domínguez-Granda, L.E. Spatio-temporal variability of Brachyura larval assemblages in mangroves of the Gulf of Guayaquil’s inner estuary. Reg. Stud. Mar. Sci. 2021, 41, 101601. [Google Scholar] [CrossRef]
- Monserrate, L. Estudio de Condiciones Físicas, Químicas y Biológicas en la Zona Intermareal de dos Sectores del Estero Salado con Diferente Desarrollo Urbano. Ph.D. Thesis, Escuela Superior Politécnica del Litoral, Guayaquil, Ecuador, February 2011. [Google Scholar]
- Alvarez-Mieles, G.; Irvine, K.; Griensven, A.; Arias-Hidalgo, M.; Torres, A.; Mynett, A.E. Relationships between aquatic biotic communities and water quality in a tropical river–wetland system (Ecuador). Environ. Sci. Policy 2013, 34, 115–127. [Google Scholar] [CrossRef]
- Liu, D.; Yang, J.; Li, Y.; Zhang, M.; Wang, L. Cd-induced apoptosis through the mitochondrial pathway in the hepatopancreas of the freshwater crab Sinopotamon henanense. PLoS ONE 2013, 8, e68770. [Google Scholar] [CrossRef] [Green Version]
- Sun, M.; Li, Y.T.; Liu, Y.; Lee, S.C.; Wang, L. Transcriptome assembly and expression profiling of molecular responses to cadmium toxicity in hepatopancreas of the freshwater crab Sinopotamon henanense. Sci. Rep. 2016, 6, 19405. [Google Scholar] [CrossRef] [Green Version]
- Bergey, L.L.; Weis, J.S. Molting as a mechanism of depuration of metals in the fiddler crab, Uca pugnax. Mar. Environ. Res. 2007, 64, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Yogesh, P.; Samantha, M.; James, L.; Lang, H. Sample Preparation Method for Mercury Analysis in Reagent Chemicals by ICP OES. Spectroscopy 2015, 31, 8–17. [Google Scholar]
- Ure, A. Methods of analysis of heavy metals in soils. In Heavy Metals in Soils; Blackie & Son Ltd.: London, UK, 1990; pp. 40–80. [Google Scholar]
- Souri, A.; Niyogi, S.; Naji, A. Distribution, source apportionment, bioavailability and ecological risks of metals in reef sediments and corals of the Persian Gulf (Iran): Khark Island, Chirouyeh, and Hendorabi Island. Mar. Pollut. Bull. 2019, 149, 110654. [Google Scholar] [CrossRef]
- Muller, G. Schwermetalle in den sedimenten des Rheins-Veranderungen seit. Umschav 1979, 79, 133–149. [Google Scholar]
- CCME. Canadian sediment quality guidelines for the protection of aquatic life: Summary tables. In Updated. Can. Environ. Qual. Guidel. 1999; Canadian Council of Ministers of the Environmen: Toronto, Canada, 2001. [Google Scholar]
- Dimitrakakis, E.; Hahladakis, J.; Gidarakos, E. The “Sea Diamond” shipwreck: Environmental impact assessment in the water column and sediments of the wreck area. Int. J. Environ. Sci. Technol. 2014, 11, 1421–1432. [Google Scholar] [CrossRef] [Green Version]
- USEPA. National Recommended Water Quality Criteria: Aquatic Life Criteria Table; United States Environmental Protection Agency: Washington, DC, USA, 2016. [Google Scholar]
- Ministerio de ambiente Ecuador. Anexo 1 del Libro vi del Texto Unificado de Legislacion Secundaria del Ministerio del Ambiente: Norma de Calidad Ambiental y de Descarga de Efluentes al Recurso Agua. 2011. Available online: http://extwprlegs1.fao.org/docs/pdf/ecu155128.pdf (accessed on 26 March 2021).
- Jacxsens, L.; Uyttendaele, M.; De Meulenaer, B. Challenges in risk assessment: Quantitative risk assessment. In Proceedings of the International Conference of Sabaragamuwa University of Sri Lanka 2015 (ICSUSL 2015): Challenges beyond Food Security, Sri Lanka, 19–20 November 2015; pp. 23–30. [Google Scholar]
- New Jersey Department of Environmental Protection. Estimate of cancer risk to consumers of crabs caught in the area of the Diamond alkali site and other areas of the Newark Bay complex from 2, 3, 7, 8-TCDD and 2, 3, 7, 8-TCDD equivalents; Technical report; New Jersey Department of Environmental Protection: Trenton, NJ, USA, 2002. [Google Scholar]
- Pflugh, K.K.; Stern, A.H.; Nesposudny, L.; Lurig, L.; Ruppel, B.; Buchanan, G.A. Consumption patterns and risk assessment of crab consumers from the Newark Bay Complex, New Jersey, USA. Sci. Total. Environ. 2011, 409, 4536–4544. [Google Scholar] [CrossRef]
- Palisade. @RISK: Risk Analysis Software using Monte Carlo Simulation for Excel; Palisade: Ithaca, NY, USA, 2021. [Google Scholar]
- Kamala, A.; Kimanya, M.; Lachat, C.; Jacxsens, L.; Haesaert, G.; Kolsteren, P.; Ortiz, J.; Tiisekwa, B.; De Meulenaer, B. Risk of exposure to multiple mycotoxins from maize-based complementary foods in Tanzania. J. Agric. Food Chem. 2017, 65, 7106–7114. [Google Scholar] [CrossRef]
- Babaahmadifooladi, M.; Jacxsens, L.; Van de Wiele, T.; Du Laing, G. Gap analysis of nickel bioaccessibility and bioavailability in different food matrices and its impact on the nickel exposure assessment. Food Res. Int. 2020, 129, 108866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- EFSA. Scientific Opinion on lead in food. EFSA J. 2010, 8, 1570. [Google Scholar]
- EFSA. Scientific Opinion on arsenic in food. EFSA J. 2009, 7, 1351. [Google Scholar] [CrossRef]
- Benford, D.; Bolger, P.M.; Carthew, P.; Coulet, M.; DiNovi, M.; Leblanc, J.-C.; Renwick, A.G.; Setzer, W.; Schlatter, J.; Smith, B. Application of the Margin of Exposure (MOE) approach to substances in food that are genotoxic and carcinogenic. Food Chem. Toxicol. 2010, 48, S2–S24. [Google Scholar] [CrossRef] [PubMed]
- WHO. Principles for Modelling Dose-Response for the Risk Assessment of Chemicals; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Bosch, A.C.; O’Neill, B.; Sigge, G.O.; Kerwath, S.E.; Hoffman, L.C. Heavy metals in marine fish meat and consumer health: A review. J. Sci. Food Agric. 2016, 96, 32–48. [Google Scholar] [CrossRef]
- Feldmann, J.; Krupp, E.M. Critical review or scientific opinion paper: Arsenosugars—A class of benign arsenic species or justification for developing partly speciated arsenic fractionation in foodstuffs? Anal. Bioanal. Chem. 2011, 399, 1735–1741. [Google Scholar] [CrossRef]
- Li, W.; Wei, C.; Zhang, C.; Van Hulle, M.; Cornelis, R.; Zhang, X. A survey of arsenic species in Chinese seafood. Food Chem. Toxicol. 2003, 41, 1103–1110. [Google Scholar] [CrossRef]
- Liu, X.; Wang, Q.; He, J.; Zhao, M.; Qiu, L.; Fan, L.; Meng, S.; Yang, G.; Li, T.; Li, Q. Occurrence, speciation analysis and health risk assessment of arsenic in Chinese mitten crabs (Eriocheir sinensis) collected from China. J. Food Compos. Anal. 2020, 94, 103647. [Google Scholar] [CrossRef]
- Zhang, W.; Guo, Z.; Song, D.; Du, S.; Zhang, L. Arsenic speciation in wild marine organisms and a health risk assessment in a subtropical bay of China. Sci. Total. Environ. 2018, 626, 621–629. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Statement on the applicability of the Margin of Exposure approach for the safety assessment of impurities which are both genotoxic and carcinogenic in substances added to food/feed. EFSA J. 2012, 10, 2578. [Google Scholar]
- WHO. Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 1993. [Google Scholar]
- EFSA. Update of the risk assessment of nickel in food and drinking water. EFSA J. 2020, 18, e06268. [Google Scholar]
- EFSA. Scientific Opinion on the risks to public health related to the presence of chromium in food and drinking water. EFSA J. 2014, 12, 3595. [Google Scholar]
- EFSA. Statement on tolerable weekly intake for cadmium. EFSA J. 2011, 9, 1975. [Google Scholar]
- EFSA. Statement on the benefits of fish/seafood consumption compared to the risks of methylmercury in fish/seafood. EFSA J. 2015, 13, 3982.
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Samara, F.; Solovieva, N.; Ghalayini, T.; Nasrallah, Z.A.; Saburova, M. Assessment of the Environmental Status of the Mangrove Ecosystem in the United Arab Emirates. Water 2020, 12, 1623. [Google Scholar] [CrossRef]
- Cuong, D.T.; Bayen, S.; Wurl, O.; Subramanian, K.; Wong, K.K.S.; Sivasothi, N.; Obbard, J.P. Heavy metal contamination in mangrove habitats of Singapore. Mar. Pollut. Bull. 2005, 50, 1732–1738. [Google Scholar] [CrossRef]
- Defew, L.H.; Mair, J.M.; Guzman, H.M. An assessment of metal contamination in mangrove sediments and leaves from Punta Mala Bay, Pacific Panama. Mar. Pollut. Bull. 2005, 50, 547–552. [Google Scholar] [CrossRef]
- El-Kady, A.A.; Abdel-Wahhab, M.A. Occurrence of trace metals in foodstuffs and their health impact. Trends Food Sci. Technol. 2018, 75, 36–45. [Google Scholar] [CrossRef]
- European Comission. Information Note: Consumption of Brown Crabmeat. 2011. Available online: http://ec.europa.eu/food/food/chemicalsafety/contaminants/information_note_cons_brown_crab_en.pdf (accessed on 25 March 2021).
- EFSA. Cadmium in food—Scientific opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2009, 7, 980. [Google Scholar]
- EFSA. Scientific Opinion on the risk for public health related to the presence of mercury and methylmercury in food. EFSA J. 2012, 10, 2985. [Google Scholar]
- European Commission. Commission Regulation (EC) No 78/2005 of 19 January 2005 amending Regulation (EC) No 466/2001 as regards heavy metals. Off. J. Eur. Union 2005, 16, 43–45. [Google Scholar]
- Brix, K.V.; Schlekat, C.E.; Garman, E.R. The mechanisms of nickel toxicity in aquatic environments: An adverse outcome pathway analysis. Environ. Toxicol. Chem. 2017, 36, 1128–1137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hashim, J.H.; Radzi, R.S.M.; Aljunid, S.M.; Nur, A.M.; Ismail, A.; Baguma, D.; Sthiannopkao, S.; Phan, K.; Wong, M.H.; Sao, V. Hair arsenic levels and prevalence of arsenicosis in three Cambodian provinces. Sci. Total. Environ. 2013, 463, 1210–1216. [Google Scholar] [CrossRef]
- Hojdová, M.; Rohovec, J.; Chrastný, V.; Penížek, V.; Navrátil, T. The influence of sample drying procedures on mercury concentrations analyzed in soils. Bull. Environ. Contam. Toxicol. 2015, 94, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Appleton, J.; Williams, T.; Orbea, H.; Carrasco, M. Fluvial contamination associated with artisanal gold mining in the Ponce Enriquez, Portovelo-Zaruma and Nambija areas, Ecuador. Water Air Soil Pollut. 2001, 131, 19–39. [Google Scholar] [CrossRef]
- Twilley, R.; Cárdenas, W.; Rivera-Monroy, V.; Espinoza, J.; Suescum, R.; Armijos, M.; Solórzano, L. The Gulf of Guayaquil and the Guayas river estuary, Ecuador. In Coastal Marine Ecosystems of Latin America; Springer: Berlin/Heidelberg, Germany, 2001; pp. 245–263. [Google Scholar]
- Chavez, E.; He, Z.; Stoffella, P.; Mylavarapu, R.; Li, Y.; Moyano, B.; Baligar, V. Concentration of cadmium in cacao beans and its relationship with soil cadmium in southern Ecuador. Sci. Total. Environ. 2015, 533, 205–214. [Google Scholar] [CrossRef]
- Bryan, G.; Langston, W. Bioavailability, accumulation and effects of heavy metals in sediments with special reference to United Kingdom estuaries: A review. Environ. Pollut. 1992, 76, 89–131. [Google Scholar] [CrossRef]
- Tarras-Wahlberg, N.; Flachier, A.; Lane, S.; Sangfors, O. Environmental impacts and metal exposure of aquatic ecosystems in rivers contaminated by small scale gold mining: The Puyango River basin, southern Ecuador. Sci. Total. Environ. 2001, 278, 239–261. [Google Scholar] [CrossRef]
- Blewett, T.A.; Glover, C.N.; Fehsenfeld, S.; Lawrence, M.J.; Niyogi, S.; Goss, G.G.; Wood, C.M. Making sense of nickel accumulation and sub-lethal toxic effects in saline waters: Fate and effects of nickel in the green crab, Carcinus maenas. Aquat. Toxicol. 2015, 164, 23–33. [Google Scholar] [CrossRef]
- Eisler, R. Nickel Hazards to Fish, Wildlife, and Invertebrates: A Synoptic Review; US Department of the Interior, US Geological Survey, Patuxent Wildlife Research Center: Prince George’s County, MD, USA, 1998. [Google Scholar]
- Rai, P.K.; Lee, S.S.; Zhang, M.; Tsang, Y.F.; Kim, K.-H. Heavy metals in food crops: Health risks, fate, mechanisms, and management. Environ. Int. 2019, 125, 365–385. [Google Scholar] [CrossRef]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef] [Green Version]
- Vijayavel, K.; Gopalakrishnan, S.; Thiagarajan, R.; Thilagam, H. Immunotoxic effects of nickel in the mud crab Scylla serrata. Fish. Shellfish. Immunol. 2009, 26, 133–139. [Google Scholar] [CrossRef]
- Bundschuh, J.; Schneider, J.; Alam, M.A.; Niazi, N.K.; Herath, I.; Parvez, F.; Tomaszewska, B.; Guilherme, L.R.G.; Maity, J.P.; López, D.L. Seven potential sources of arsenic pollution in Latin America and their environmental and health impacts. Sci. Total. Environ. 2021, 146274. [Google Scholar] [CrossRef]
- Twilley, R. Impacts of shrimp mariculture practices on the ecology of coastal ecosystems in Ecuador. A Sustainable Shrimp Mariculture Industry for Ecuador; Technical Report Series TR-E-6. International Coastal Resources Management Project. The University of Rhode Island Coastal Resources Center: Narragansett, RI, USA, 1989; pp. 91–120. [Google Scholar]
- Van Ael, E.; Blust, R.; Bervoets, L. Metals in the Scheldt estuary: From environmental concentrations to bioaccumulation. Environ. Pollut. 2017, 228, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Essien, J.; Antai, S.; Olajire, A. Distribution, seasonal variations and ecotoxicological significance of heavy metals in sediments of cross river estuary mangrove swamp. Water Air Soil Pollut. 2009, 197, 91–105. [Google Scholar] [CrossRef]
- Tam, N.F.; Wong, Y.S. Mangrove soils as sinks for wastewater-borne pollutants. Hydrobiologia 1995, 295, 231–241. [Google Scholar] [CrossRef]
- Fırat, Ö.; Cogun, H.Y.; Yüzereroğlu, T.A.; Gök, G.; Fırat, Ö.; Kargin, F.; Kötemen, Y. A comparative study on the effects of a pesticide (cypermethrin) and two metals (copper, lead) to serum biochemistry of Nile tilapia, Oreochromis niloticus. Fish Physiol. Biochem. 2011, 37, 657–666. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wright, P.; Mason, C. Spatial and seasonal variation in heavy metals in the sediments and biota of two adjacent estuaries, the Orwell and the Stour, in eastern England. Sci. Total. Environ. 1999, 226, 139–156. [Google Scholar] [CrossRef]
- Praveena, S.M.; Aris, A.Z.; Radojevic, M. Heavy metals dyanamics and source in intertidal mangrove sediment of Sabah, Borneo Island. Environ. Asia 2010, 3, 79–83. [Google Scholar]
- IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; World Health Organization: Geneva, Switzerland, 2006; Volume 86. [Google Scholar]
- Otero, X.; Tierra, W.; Atiaga, O.; Guanoluisa, D.; Nunes, L.M.; Ferreira, T.; Ruales, J. Arsenic in rice agrosystems (water, soil and rice plants) in Guayas and Los Ríos provinces, Ecuador. Sci. Total. Environ. 2016, 573, 778–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wiech, M.; Frantzen, S.; Duinker, A.; Rasinger, J.D.; Maage, A. Cadmium in brown crab Cancer pagurus. Effects of location, season, cooking and multiple physiological factors and consequences for food safety. Sci. Total. Environ. 2020, 703, 134922. [Google Scholar] [CrossRef]
- Abd-Elghany, S.M.; Zaher, H.A.; Elgazzar, M.M.; Sallam, K.I. Effect of boiling and grilling on some heavy metal residues in crabs and shrimps from the Mediterranean Coast at Damietta region with their probabilistic health risk assessment. J. Food Compos. Anal. 2020, 93, 103606. [Google Scholar] [CrossRef]
- Lahiri, S.; Bhattacharyya, S.B.; Prosenjit, P.; Ricardo, G.; Sufia, Z.; Abhijit, M. Do cooking process influences the metal contents in crab and prawn? Parana J. Sci. Educ. 2019, 5, 8–14. [Google Scholar]
- Wiener, J.G.; Sandheinrich, M.B.; Bhavsar, S.P.; Bohr, J.R.; Evers, D.C.; Monson, B.A.; Schrank, C.S. Toxicological significance of mercury in yellow perch in the Laurentian Great Lakes region. Environ. Pollut. 2012, 161, 350–357. [Google Scholar] [CrossRef]
- Lepak, R.F.; Hoffman, J.C.; Janssen, S.E.; Krabbenhoft, D.P.; Ogorek, J.M.; DeWild, J.F.; Tate, M.T.; Babiarz, C.L.; Yin, R.; Murphy, E.W. Mercury source changes and food web shifts alter contamination signatures of predatory fish from Lake Michigan. Proc. Natl. Acad. Sci. USA 2019, 116, 23600–23608. [Google Scholar] [CrossRef] [Green Version]
- Sampantamit, T.; Ho, L.; Lachat, C.; Hanley-Cook, G.; Goethals, P. The Contribution of Thai Fisheries to Sustainable Seafood Consumption: National Trends and Future Projections. Foods 2021, 10, 880. [Google Scholar] [CrossRef]
- United Nations. Sustainable Development Goals. 2015. Available online: https://sdgs.un.org/goals (accessed on 25 March 2021).
| Metal | Toxicological Value from Reference | Toxicological Value Tested Against | |||
|---|---|---|---|---|---|
| Type | Value | Reference | Type | Value | |
| Zn | PMTDI | 0.3–1 mg/kg·bw/day | [68] | TDI | 300 µg/kg·bw/day |
| Cu | PMTDI | 0.5 mg/kg·bw/day | [68] | TDI | 500 µg/kg·bw/day |
| Ni | TDI | 2.8 μg/kg·bw/day | [69] | TDI | 13 µg/kg·bw/day |
| Cr | TDI | 0.3 mg/kg·bw /day | [70] | TDI | 300 µg/kg·bw/day |
| In-As | BMDL01 | 0.3 μg/kg·bw/day | [59] | MOE | 10,000 |
| Pb | BMDL01 | 12 µg/kg·bw/day | [58] | MOE | 10 |
| Cd | PTMI | 25 μg/kg·bw/month | [71] | TDI | 0.81 µg/kg·bw/day |
| Hg | PTWI | 4 µg/kg·bw/week | [72] | TDI | 0.57 µg/kg·bw/day |
| Zn | Cu | Ni | Cr | As | Pb | Cd | Hg | |
|---|---|---|---|---|---|---|---|---|
| Hepatopancreas | S2 > S5 S4 > S5 | S2 > S1 | S2 > S1 | S2 > S3 S2 > S4 S5 > S3 | S3 > S4 S5 > S4 | S3 > S5 | ||
| Carapax | S1 > S5 S2 > S5 S3 > S5 | S1 > S4 S2 > S4 S2 > S5 | S3 > S1 S3 > S2 S3 > S4 S3 > S5 | S3 > S1 S3 > S2 S5 > S2 | S1 > S2 S1 > S4 S1 > S5 S3 > S2 S3 > S4 S3 > S5 | S3 > S2 S3 > S4 S3 > S5 | S1 > S3 S1 > S5 | |
| White meat | S1 > S3 | S1 > S2 S1 > S3 S1 > S4 S1 > S5 | ||||||
| Sediment | S2 > S4 | |||||||
| Leaves | ||||||||
| Water | S1 > S3 |
 = Significant difference between certain stations (p < 0.025).
= Significant difference between certain stations (p < 0.025).| Index | Matrix | Zn | Cu | Ni | As | Cd | Pb | Cr |
|---|---|---|---|---|---|---|---|---|
| HQs | Sediment | 0.3 | 0.5 | 4.6 | 0.2 | 0.04 | 0.1 | 0.4 |
| HQw | Water | 0.0001 | 0.0009 | 0.00009 | 0.0001 | 0.000003 | - | 0.000005 |
| Igeo | Sediment | −0.3 | −0.7 | 0.8 | 1.5 | −0.9 | −1.0 | −1.4 |
| BSAF | Crab/sediment | 0.9 | 0.1 | 0.2 | 0.2 | 0.2 | 0.02 | 0.04 |
| Metal | Mean | SD | P50 | P75 | P95 | Population Exceeding TDI (%) | % TDI | MOE at Mean Exposure |
|---|---|---|---|---|---|---|---|---|
| Zn | 6.97 | 8.19 | 3.96 | 9.60 | 27.1 | 0 | 2.32 | - |
| Cu | 3.26 | 4.03 | 1.74 | 4.25 | 12.2 | 0 | 0.65 | - |
| Ni | 0.15 | 0.20 | 0.07 | 0.19 | 0.56 | 0 | 1.15 | - |
| Cr | 0.022 | 0.036 | 0.010 | 0.027 | 0.100 | 0 | 0.01 | - |
| In-As | 0.0005 | 0.001 | 0.0002 | 0.0005 | 0.002 | - | - | 1423 |
| Pb | 0.009 | 0.015 | 0.003 | 0.011 | 0.035 | - | - | 55.8 |
| Cd | 0.001 | 0.002 | 0.001 | 0.001 | 0.004 | 0 | 0.15 | - |
| Hg | 0.002 | 0.005 | 0.0004 | 0.001 | 0.007 | 0 | 0.30 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Cock, A.; De Troyer, N.; Eurie, M.A.F.; Garcia Arevalo, I.; Van Echelpoel, W.; Jacxsens, L.; Luca, S.; Du Laing, G.; Tack, F.; Dominguez Granda, L.; et al. From Mangrove to Fork: Metal Presence in the Guayas Estuary (Ecuador) and Commercial Mangrove Crabs. Foods 2021, 10, 1880. https://doi.org/10.3390/foods10081880
De Cock A, De Troyer N, Eurie MAF, Garcia Arevalo I, Van Echelpoel W, Jacxsens L, Luca S, Du Laing G, Tack F, Dominguez Granda L, et al. From Mangrove to Fork: Metal Presence in the Guayas Estuary (Ecuador) and Commercial Mangrove Crabs. Foods. 2021; 10(8):1880. https://doi.org/10.3390/foods10081880
Chicago/Turabian StyleDe Cock, Andrée, Niels De Troyer, Marie Anne Forio Eurie, Isabel Garcia Arevalo, Wout Van Echelpoel, Liesbeth Jacxsens, Stijn Luca, Gijs Du Laing, Filip Tack, Luis Dominguez Granda, and et al. 2021. "From Mangrove to Fork: Metal Presence in the Guayas Estuary (Ecuador) and Commercial Mangrove Crabs" Foods 10, no. 8: 1880. https://doi.org/10.3390/foods10081880
APA StyleDe Cock, A., De Troyer, N., Eurie, M. A. F., Garcia Arevalo, I., Van Echelpoel, W., Jacxsens, L., Luca, S., Du Laing, G., Tack, F., Dominguez Granda, L., & Goethals, P. L. M. (2021). From Mangrove to Fork: Metal Presence in the Guayas Estuary (Ecuador) and Commercial Mangrove Crabs. Foods, 10(8), 1880. https://doi.org/10.3390/foods10081880