Role of Postbiotics in Diabetes Mellitus: Current Knowledge and Future Perspectives
Abstract
1. Introduction
1.1. Postbiotics
1.1.1. Characteristics of Postbiotics
1.1.2. Classification
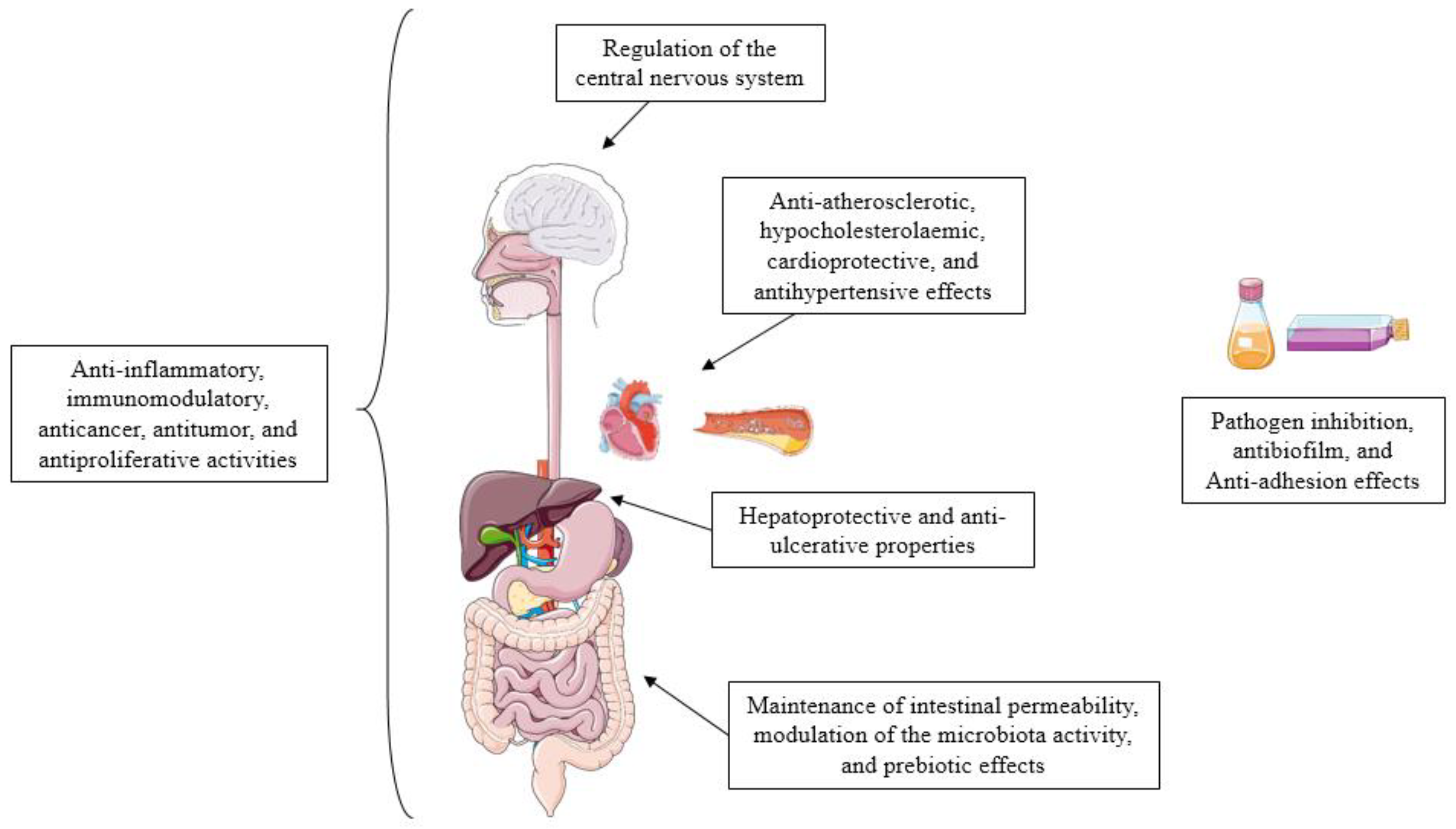
1.1.3. Health Benefits of Postbiotics
1.2. Lactic Acid Bacteria
1.3. Microbiotherapy for Diabetes Mellitus Management
2. Objectives and Search Strategy
3. Results
3.1. Exopolysaccharides (EPSs)
3.2. GABA
3.3. Extracellular Vesicles (EVs)
3.4. Supernatants, Extracts, and Surfactants
| Type of Research | Component | Bioactive Molecule(s) | Source/Origin | Model System (If Apply) | Findings | Study |
|---|---|---|---|---|---|---|
| Animal | Supernatant | Supernatant | Lactobacillus rhamnosus GG | C57BL/6J induced metabolic dysfunction with HFFD and intermittent hypoxia | ↓ FBG (vs. baseline values), ↑insulin sensitivity, ↑ energy expenditure, improved body composition (fat and muscle mass), ↓ TC and TG, ↑ NEFAs, ↑ total faecal SFCAs, ↓ proinflammatory cytokines expression, downregulated lipogenesis, upregulated lipid oxidation | Liu et al., 2020 [103] |
| Extract | Fermented soybean extracts (served dried with corn starch), low and high doses | Bacillus subtilis MORI/Isolated from Chungkookjang | Wistar rats induced T1D with STZ (55 mg/kg) | Low and high doses (vs. diabetic controls): ↑ BW, attenuated rise in FBG, ↓ food and water intake, ↓ MDA serum levels, ↑ CAT and GSH-Px activity, improved vascular function High doses (vs. diabetic control): ↑serum insulin levels and SOD levels | Lim et al., 2012 [102] | |
| Biosurfactant | Biosurfactant (served as crude lipopeptide preparation) | Bacillus subtilis SPB1/Isolated from Tunisian soil | Wistar rats induced T1D with alloxan (150 mg/kg) | ↓ FBG and α-amylase activity in the plasma, ↓ TC, TG, and LDL levels, ↑ HDL levels, protected tissues (pancreatic b cells, liver, intestine, and kidney) | Zouari et al., 2015 [104] | |
| Wistar rats induced T2D with HFFD | ↓FBG, improved glucose tolerance (OGTT), normalised serum α-amylase activity | Zouari et al., 2017 [105] | ||||
| In vitro | Supernatant | CFS | Five Lactobacillus plantarum strains: CCFM0236, CCFM 12, CCFM 10, CCFM0311, and CCFM 23 | - | α-glucosidase inhibitory activity (%): from 14.5 to 32.2 | Li et al., 2016 [107] |
| CFE and CFS | 8 LAB isolates (K1, K8, K16, K19, K29, K35, K45, K96, and LGG)/Isolated from commercial water kefir grains | - | α-glucosidase inhibitory activity (%): from 5.2 to 39.4 in CFS, from 2.3 to 15.5 in CFE | Koh et al., 2018 [108] | ||
| CFE, CFS and CFES | 14 Lactobacillus spp. strains/Isolated from traditional fermented products | - | DPP-IV inhibitory activity (%): from 0 to 55.4 in CFE, from 0 to 7.13 in CFES/reducing activity (mmol of cysteine): from 73.3 to 189.7 in CFS, from 53.0 to 159.7 in CFE/DPPH free radical-scavenging activity (%): from 36.8 to 62.1 in CFS, from 12.9 to 34.5 in CFE/hydroxyl radical scavenging activity: from 13.7 to 68.6 in CFS; from 15.9 to 38.8 in CFE/superoxide anion radical scavenging activity (%): from 2.6 to 16.2 in CFS; from 12.2 to 43.3 in CFE/lipid peroxidation inhibiting capacity (%): from 1.5 to 18.5 in CFS; from 5.9 to 31.4 in CFE. | Yan et al., 2020 [109] |
3.5. Inanimate Microorganisms
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Şanlier, N.; Gökcen, B.B.; Sezgin, A.C. Health benefits of fermented foods. Crit. Rev. Food Sci. Nutr. 2019, 59, 506–527. [Google Scholar] [CrossRef]
- Kranich, J.; Maslowski, K.M.; Mackay, C.R. Commensal flora and the regulation of inflammatory and autoimmune responses. Semin. Immunol. 2011, 23, 139–145. [Google Scholar] [CrossRef]
- Macia, L.; Thorburn, A.N.; Binge, L.C.; Marino, E.; Rogers, K.E.; Maslowski, K.M.; Vieira, A.T.; Kranich, J.; Mackay, C.R. Microbial influences on epithelial integrity and immune function as a basis for inflammatory diseases. Immunol. Rev. 2012, 245, 164–176. [Google Scholar] [CrossRef]
- Clarke, G.; Stilling, R.M.; Kennedy, P.J.; Stanton, C.; Cryan, J.F.; Dinan, T.G. Minireview: Gut Microbiota: The Neglected Endocrine Organ. Mol. Endocrinol. 2014, 28, 1221–1238. [Google Scholar] [CrossRef]
- Qiu, Q.; Lin, Y.; Ma, Y.; Li, X.; Liang, J.; Chen, Z.; Liu, K.; Huang, Y.; Luo, H.; Huang, R.; et al. Exploring the Emerging Role of the Gut Microbiota and Tumor Microenvironment in Cancer Immunotherapy. Front. Immunol. 2021, 11, 1–15. [Google Scholar] [CrossRef]
- Sekirov, I.; Russel, S.L.; Antunes, L.C.M.; Finlay, B.B. Gut Microbiota in Health and Disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef] [PubMed]
- Tlaskalová-Hogenová, H.; Tpánková, R.; Kozáková, H.; Hudcovic, T.; Vannucci, L.; Tuková, L.; Rossmann, P.; Hrní, T.; Kverka, M.; Zákostelská, Z.; et al. The role of gut microbiota (commensal bacteria) and the mucosal barrier in the pathogenesis of inflammatory and autoimmune diseases and cancer: Contribution of germ-free and gnotobiotic animal models of human diseases. Cell. Mol. Immunol. 2011, 8, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xu, X.; Xia, Y. Further analysis reveals new gut microbiome markers of type 2 diabetes mellitus. Antonie Van Leeuwenhoek 2017, 110, 445–453. [Google Scholar] [CrossRef]
- Chaluvadi, S.; Hotchkiss, A.T.; Yam, K.L. Gut Microbiota: Impact of Probiotics, Prebiotics, Synbiotics, Pharmabiotics, and Postbiotics on Human Health. In Probiotics, Prebiotics, and Synbiotics: Bioactive Foods in Health Promotion; Elsevier Inc.: Hoboken, NJ, USA, 2015; pp. 515–523. ISBN 9780128023716. [Google Scholar]
- Vyas, U.; Ranganathan, N. Probiotics, Prebiotics, and Synbiotics: Gut and Beyond. Gastroenterol. Res. Pract. 2012, 11. [Google Scholar] [CrossRef]
- Ojeda, P.; Bobe, A.; Dolan, K.; Leone, V.; Martinez, K. Nutritional modulation of gut microbiota—The impact on metabolic disease pathophysiology. J. Nutr. Biochem. 2016, 28, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Bermudez-Brito, M.; Plaza-Díaz, J.; Muñoz-Quezada, S.; Gómez-Llorente, C.; Gil, A. Probiotic mechanisms of action. Ann. Nutr. Metab. 2012, 61, 160–174. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Suez, J.; Zmora, N.; Segal, E.; Elinav, E. The pros, cons, and many unknowns of probiotics. Nat. Med. 2019, 25, 716–729. [Google Scholar] [CrossRef]
- Martín, R.; Langella, P. Emerging health concepts in the probiotics field: Streamlining the definitions. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Sanders, M.E.; Guarner, F.; Mills, D.; Pot, B.; Rafter, J.; Reid, G.; Ringel, Y. Selected Topics in Probiotics and Prebiotics: Meeting Report for the 2004 International Scientific Association for Probiotics and Prebiotics. Curr. Issues Intestig. Microbiol. 2005, 6, 55–68. [Google Scholar]
- Roberfroid, M.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.; Rowland, I.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr. 2010, 104, 72. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Tsilingiri, K.; Barbosa, T.; Penna, G.; Caprioli, F.; Sonzogni, A.; Viale, G.; Rescigno, M. Probiotic and postbiotic activity in health and disease: Comparison on a novel polarised ex-vivo organ culture model. Gut 2012, 61, 1007–1015. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat. Rev. Endocrinol. 2021. [Google Scholar] [CrossRef]
- Cuevas-González, P.F.; Liceaga, A.M.; Aguilar-Toalá, J.E. Postbiotics and paraprobiotics: From concepts to applications. Food Res. Int. 2020, 136. [Google Scholar] [CrossRef]
- Maria Carmen, C.; Seppo, S.; Vinderola, G. Chapter 11-Postbiotics: Defining the impact of inactivated microbes and their metabolites on promotion of health. In The Human Microbiome in Early Life; Koren, O., Rautava, S., Eds.; Academic Press: Cambridge, MA, USA, 2021; pp. 257–268. ISBN 9780128180976. [Google Scholar]
- Wegh, C.A.M.; Geerlings, S.Y.; Knol, J.; Roeselers, G.; Belzer, C. Postbiotics and Their Potential Applications in Early Life Nutrition and Beyond. Int. J. Mol. Med. 2019, 20, 4673. [Google Scholar] [CrossRef]
- Aguilar-Toalá, J.E.; Garcia-Varela, R.; Garcia, H.S.; Mata-Haro, V.; González-Córdova, A.F.; Vallejo-Cordoba, B.; Hernández-Mendoza, A. Postbiotics: An evolving term within the functional foods field. Trends Food Sci. Technol. 2018, 75, 105–114. [Google Scholar] [CrossRef]
- Li, X.; Xu, Q.; Jiang, T.; Fang, S.; Wang, G.; Zhao, J.; Zhang, H.; Chen, W. A comparative study of the antidiabetic effects exerted by live and dead multi-strain probiotics in the type 2 diabetes model of mice. Food Funct. 2016, 7, 4851–4860. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Lu, Z.; Huang, M.; Wang, Y.; Wang, Y. Effects of Se-enriched polysaccharides produced by Enterobacter cloacae Z0206 on alloxan-induced diabetic mice. Int. J. Biol. Macromol. 2012, 50, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Tsilingiri, K.; Rescigno, M. Postbiotics: What else? Benef. Microbes 2013, 4, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Taverniti, V.; Guglielmetti, S. The immunomodulatory properties of probiotic microorganisms beyond their viability (ghost probiotics: Proposal of paraprobiotic concept). Genes Nutr. 2011, 6, 261–274. [Google Scholar] [CrossRef]
- Wang, S.; Ahmadi, S.; Nagpal, R.; Jain, S.; Mishra, S.P.; Kavanagh, K.; Zhu, X.; Wang, Z.; McClain, D.A.; Kritchevsky, S.B.; et al. Lipoteichoic acid from the cell wall of a heat killed Lactobacillus paracasei D3-5 ameliorates aging-related leaky gut, inflammation and improves physical and cognitive functions: From C. elegans to mice. GeroScience 2020, 42, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Toscano, M.; De Grandi, R.; Pastorelli, L.; Vecchi, M.; Drago, L. A consumer’s guide for probiotics: 10 golden rules for a correct use. Dig. Liver Dis. 2017, 49, 1177–1184. [Google Scholar] [CrossRef] [PubMed]
- Żółkiewicz, J.; Marzec, A.; Ruszczyński, M.; Feleszko, W. Postbiotics—A step beyond pre-and probiotics. Nutrients 2020, 12, 2189. [Google Scholar] [CrossRef]
- Nataraj, B.H.; Ali, S.A.; Behare, P.V.; Yadav, H. Postbiotics-parabiotics: The new horizons in microbial biotherapy and functional foods. Microb. Cell Fact. 2020, 19, 1–22. [Google Scholar] [CrossRef]
- Patel, A.; Prajapat, J.B. Food and Health Applications of Exopolysaccharides produced by Lactic acid Bacteria. Adv. Dairy Res. 2013, 1, 1–8. [Google Scholar] [CrossRef]
- Zacharof, M.P.; Lovitt, R.W. Bacteriocins Produced by Lactic Acid Bacteria A Review Article. Apcbee Procedia 2012, 2, 50–56. [Google Scholar] [CrossRef]
- Piqué, N.; Berlanga, M.; Miñana-Galbis, D. Health Benefits of Heat-Killed (Tyndallized ) Probiotics: An Overview. Int. J. Mol. Sci. 2019, 20, 2534. [Google Scholar] [CrossRef]
- Patel, S.; Majumder, A.; Goyal, A. Potentials of Exopolysaccharides from Lactic Acid Bacteria. Indian J. Microbiol. 2012, 52, 3–12. [Google Scholar] [CrossRef] [PubMed]
- de Almada, C.N.; Almada, C.N.; Martinez, R.C.R.; Sant’Ana, A.S. Paraprobiotics: Evidences on their ability to modify biological responses, inactivation methods and perspectives on their application in foods. Trends Food Sci. Technol. 2016, 58, 96–114. [Google Scholar] [CrossRef]
- Almada, C.N.; Almada-Érix, C.N.; Roquetto, A.R.; Santos-Junior, V.A.; Cabral, L.; Noronha, M.F.; Gonçalves, A.E.S.S.; dos Santos, P.; dos Santos, A.; Martinez, J.; et al. Paraprobiotics obtained by six different inactivation processes: Impacts on the biochemical parameters and intestinal microbiota of Wistar male rats. Int. J. Food Sci. Nutr. 2021, 1–14. [Google Scholar] [CrossRef]
- Anderson, R.C. Are Postbiotics the Long Sought-After Solution for a Leaky Gut? J. Nutr. 2019, 149, 1873–1874. [Google Scholar] [CrossRef] [PubMed]
- Chelakkot, C.; Choi, Y.; Kim, D.K.; Park, H.T.; Ghim, J.; Kwon, Y.; Jeon, J.; Kim, M.S.; Jee, Y.K.; Gho, Y.S.; et al. Akkermansia muciniphila-derived extracellular vesicles influence gut permeability through the regulation of tight junctions. Exp. Mol. Med. 2018, 50, e450. [Google Scholar] [CrossRef]
- Plaza-Diaz, J.; Ruiz-Ojeda, F.J.; Gil-Campos, M.; Gil, A. Mechanisms of Action of Probiotics. Adv. Nutr. 2019, 10, S49–S66. [Google Scholar] [CrossRef]
- Hayes, M.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Putting microbes to work: Diary fermentation, cell factories and bioactive peptides. Part I: Overview. Biotechnol. J. 2007, 2, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Peluzio, M.D.C.G.; Martinez, J.A.; Milagro, F.I. Postbiotics: Metabolites and mechanisms involved in microbiota-host interactions. Trends Food Sci. Technol. 2021, 108, 11–26. [Google Scholar] [CrossRef]
- Haas-neill, S.; Forsythe, P. A budding relationship: Bacterial extracellular vesicles in the microbiota–Gut–brain axis. Int. J. Mol. Sci. 2020, 21, 8899. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Hase, K. Gut microbiota-generated metabolites in animal health and disease. Nat. Chem. Biol. 2014, 10, 416–424. [Google Scholar] [CrossRef]
- Lee, B.H.; Wu, S.C.; Shen, T.L.; Hsu, Y.Y.; Chen, C.H.; Hsu, W.H. The applications of Lactobacillus plantarum-derived extracellular vesicles as a novel natural antibacterial agent for improving quality and safety in tuna fish. Food Chem. 2021, 340, 128104. [Google Scholar] [CrossRef] [PubMed]
- Mora-Sánchez, B.; Balcázar, J.L.; Pérez-Sánchez, T. Effect of a novel postbiotic containing lactic acid bacteria on the intestinal microbiota and disease resistance of rainbow trout (Oncorhynchus mykiss). Biotechnol. Lett. 2020, 42, 1957–1962. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.R.; Hoffman, M.W.; Zelicha, H.; Gepner, Y.; Willoughby, D.S.; Feinstein, U.; Ostfeld, I. The Effect of 2 Weeks of Inactivated Probiotic Bacillus coagulans on Endocrine, Inflammatory, and Performance Responses During Self-Defense Training in Soldiers. J. Strength Cond. Res. 2019, 33, 2330–2337. [Google Scholar] [CrossRef]
- Fontana, L.; Bermudez-Brito, M.; Plaza-Diaz, J.; Muñoz-Quesada, S.; Gil, A. Sources, isolation, characterisation and evaluation of probiotics. Br. J. Nutr. 2013, 109, S30–S50. [Google Scholar] [CrossRef]
- Evivie, S.E.; Huo, G.-C.; Igene, J.O.; Bian, X. Some current applications, limitations and future perspectives of lactic acid bacteria as probiotics. Food Nutr. Res. 2017, 61, 1318034. [Google Scholar] [CrossRef]
- Leroy, F.; De Vuyst, L. Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends Food Sci. Technol. 2004, 15, 67–78. [Google Scholar] [CrossRef]
- Zannini, E.; Waters, D.M.; Coffey, A.; Arendt, E.K. Production, properties, and industrial food application of lactic acid bacteria-derived exopolysaccharides. Appl. Microbiol. Biotechnol. 2016, 100, 1121–1135. [Google Scholar] [CrossRef]
- García-Cano, I.; Rocha-Mendoza, D.; Ortega-Anaya, J.; Wang, K.; Kosmerl, E.; Jiménez-Flores, R. Lactic acid bacteria isolated from dairy products as potential producers of lipolytic, proteolytic and antibacterial proteins. Appl. Microbiol. Biotechnol. 2019, 103, 5243–5257. [Google Scholar] [CrossRef] [PubMed]
- Thakur, K.; Tomar, S.K.; De, S. Lactic acid bacteria as a cell factory for riboflavin production. Microb. Biotechnol. 2016, 9, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Ménard, S.; Candalh, C.; Bambou, J.C.; Terpend, K.; Cerf-Bensussan, N.; Heyman, M. Lactic acid bacteria secrete metabolites retaining anti-inflammatory properties after intestinal transport. Gut 2004, 53, 821–828. [Google Scholar] [CrossRef] [PubMed]
- ADA. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2020. Diabetes Care 2020, 43, S14–S31. [Google Scholar] [CrossRef] [PubMed]
- Glovaci, D.; Fan, W.; Wong, N.D. Epidemiology of Diabetes Mellitus and Cardiovascular Disease. Curr. Cardiol. Rep. 2019, 21. [Google Scholar] [CrossRef]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef]
- Tilg, H.; Moschen, A.R. Microbiota and Diabetes: An Evolving Relationship. Gut 2014, 63, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Cabello-Olmo, M.; Araña, M.; Radichev, I.; Smith, P.; Huarte, E.; Barajas, M. New Insights into Immunotherapy Strategies for Treating Autoimmune Diabetes. Int. J. Mol. Sci. 2019, 20, 4789. [Google Scholar] [CrossRef]
- Villanueva-Millan, M.J.; Perez-Matute, P.; Oteo, J.A. Gut microbiota: A key player in health and disease. A review focused on obesity. J. Physiol. Biochem. 2015, 71, 509–525. [Google Scholar] [CrossRef]
- Magno, C.; Bertelli, S.; De Castro, R.; De Souza, G.T.; Rossato, C.; Carlos, F.; Anete, M.; Valente, S.; Vitor, J.; Rettore, P.; et al. Intestinal Microbiota as Modulators of the Immune System and Neuroimmune System: Impact on the Host Health and Homeostasis. J. Immunol. Res. 2015, 2015, 1–15. [Google Scholar] [CrossRef]
- Round, J.L.; Mazmanian, S.K. The gut microbiome shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 2009, 9, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.Y.; Wong, J.L.M.; Sim, Y.J.; Wong, S.S.; Elhassan, S.A.M.; Tan, S.H.; Lim, G.P.L.; Tay, N.W.R.; Annan, N.C.; Bhattamisra, S.K.; et al. Type 1 and 2 diabetes mellitus: A review on current treatment approach and gene therapy as potential intervention. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Blandino, G.; Inturri, R.; Lazzara, F.; Di Rosa, M.; Malaguarnera, L. Impact of gut microbiota on diabetes mellitus. Diabetes Metab. 2016, 42, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.T.; Nieuwdorp, M.; Bäckhed, F. Microbial modulation of insulin sensitivity. Cell Metab. 2014, 20, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Delzenne, N.M.; Cani, P.D.; Everard, A.; Neyrinck, A.M.; Bindels, L.B. Gut microorganisms as promising targets for the management of type 2 diabetes. Diabetologia 2015, 58, 2206–2217. [Google Scholar] [CrossRef]
- Ejtahed, H.S.; Hoseini-Tavassol, Z.; Khatami, S.; Zangeneh, M.; Behrouzi, A.; Ahmadi Badi, S.; Moshiri, A.; Hasani-Ranjbar, S.; Soroush, A.R.; Vaziri, F.; et al. Main gut bacterial composition differs between patients with type 1 and type 2 diabetes and non-diabetic adults. J. Diabetes Metab. Disord. 2020, 19, 265–271. [Google Scholar] [CrossRef]
- Adachi, K.; Sugiyama, T.; Yamaguchi, Y.; Tamura, Y.; Izawa, S.; Hijikata, Y.; Ebi, M.; Funaki, Y.; Ogasawara, N.; Goto, C.; et al. Gut microbiota disorders cause type 2 diabetes mellitus and homeostatic disturbances in gut related metabolism in Japanese subjects. J. Clin. Biochem. Nutr. 2019, 64, 231–238. [Google Scholar] [CrossRef]
- Piper, M.S.; Saad, R.J. Diabetes Mellitus and the Colon. Curr. Treat. Options Gastroenterol. 2017, 15, 460–474. [Google Scholar] [CrossRef] [PubMed]
- Vaarala, O.; Atkinson, M.A.; Neu, J. The “Perfect Storm” for Type 1 Diabetes: The Complex Interplay Between Intestinal Microbiota, Gut Permeability, and Mucosal Immunity. Diabetes 2008, 57, 2555–2562. [Google Scholar] [CrossRef]
- Gurung, M.; Li, Z.; You, H.; Rodrigues, R.; Jump, D.B.; Morgun, A.; Shulzhenko, N. Role of gut microbiota in type 2 diabetes pathophysiology. EBioMedicine 2020, 51, 102590. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic Endotoxemia Initiates Obesity and Insulin Resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef]
- Sato, J.; Kanazawa, A.; Watada, H. Type 2 diabetes and bacteremia. Ann. Nutr. Metab. 2017, 71, 17–22. [Google Scholar] [CrossRef]
- He, C.; Shan, Y.; Song, W. Targeting gut microbiota as a possible therapy for diabetes. Nutr. Res. 2015, 35, 361–367. [Google Scholar] [CrossRef]
- Kootte, R.S.; Vrieze, A.; Holleman, F.; Dallinga-Thie, G.M.; Zoetendal, E.G.; de Vos, W.M.; Groen, A.K.; Hoekstra, J.B.L.; Stroes, E.S.; Nieuwdorp, M. The therapeutic potential of manipulating gut microbiota in obesity and type 2 diabetes mellitus. Diabetes Obes. Metab. 2012, 14, 112–120. [Google Scholar] [CrossRef]
- Hampe, C.S.; Roth, C.L. Probiotic strains and mechanistic insights for the treatment of type 2 diabetes. Endocrine 2017, 58, 207–227. [Google Scholar] [CrossRef]
- Sato, J.; Kanazawa, A.; Azuma, K.; Ikeda, F.; Goto, H.; Komiya, K.; Kanno, R.; Tamura, Y.; Asahara, T.; Takahashi, T.; et al. Probiotic reduces bacterial translocation in type 2 diabetes mellitus: A randomised controlled study. Sci. Rep. 2017, 7, 12115. [Google Scholar] [CrossRef]
- Hansen, C.H.F.; Larsen, C.S.; Petersson, H.O.; Zachariassen, L.F.; Vegge, A.; Lauridsen, C.; Kot, W.; Krych, Ł.; Nielsen, D.S.; Hansen, A.K. Targeting gut microbiota and barrier function with prebiotics to alleviate autoimmune manifestations in NOD mice. Diabetologia 2019, 62, 1689–1700. [Google Scholar] [CrossRef]
- Gille, D.; Schmid, A.; Walther, B.; Vergères, G. Fermented Food and Non-Communicable Chronic Diseases: A Review. Nutrients 2018, 10, 448. [Google Scholar] [CrossRef] [PubMed]
- Cabello-Olmo, M.; Oneca, M.; Torre, P.; Sainz, N.; Moreno-aliaga, M.J.; Guruceaga, E.; Díaz, J.V.; Encio, I.J.; Barajas, M.; Araña, M. A Fermented Food Product Containing Lactic Acid Bacteria Protects ZDF Rats from the Development of Type 2 Diabetes. Nutrients 2019, 11, 2530. [Google Scholar] [CrossRef] [PubMed]
- Sivamaruthi, B.S.; Kesika, P.; Prasanth, M.I.; Chaiyasut, C. A Mini Review on Antidiabetic Properties of Fermented Foods. Nutrients 2018, 10, 1973. [Google Scholar] [CrossRef] [PubMed]
- Dahech, I.; Belghith, K.S.; Hamden, K.; Feki, A.; Belghith, H.; Mejdoub, H. Antidiabetic activity of levan polysaccharide in alloxan-induced diabetic rats. Int. J. Biol. Macromol. 2011, 49, 742–746. [Google Scholar] [CrossRef]
- Yeap, S.K.; Mohd Ali, N.; Mohd Yusof, H.; Alitheen, N.B.; Beh, B.K.; Ho, W.Y.; Koh, S.P.; Long, K. Antihyperglycemic effects of fermented and nonfermented mung bean extracts on alloxan-induced-diabetic mice. J. Biomed. Biotechnol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Barros, C.P.; Grom, L.C.; Guimarães, J.T.; Balthazar, C.F.; Rocha, R.S.; Silva, R.; Almada, C.N.; Pimentel, T.C.; Venâncio, E.L.; Collopy Junior, I.; et al. Paraprobiotic obtained by ohmic heating added in whey-grape juice drink is effective to control postprandial glycemia in healthy adults. Food Res. Int. 2021, 140. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, A.A.; Poulsen, V.K.; Janzen, T.; Buldo, P.; Derkx, P.M.F.; Øregaard, G.; Neves, A.R. Polysaccharide production by lactic acid bacteria: From genes to industrial applications. FEMS Microbiol. Rev. 2017, 41, S168–S200. [Google Scholar] [CrossRef] [PubMed]
- Gowd, V.; Baoa, T.; Wangc, L.; Huangc, Y.; Chenc, S.; Zhenga, X.; Cuib, S.; Chena, W. Antioxidant-and-antidiabetic-activity-of-blackberry-after-gastrointestinal-digestion-and-human-gut-microbiota-fermentationFood-Chemistry.pdf. Food Chem. 2018, 269, 618–627. [Google Scholar] [CrossRef] [PubMed]
- Melo, F.C.B.C.D.; Zaia, C.T.B.V.; Celligoi, M.A.P.C. Levan from Bacillus Subtilis Natto: Its Effects in Normal and in Streptozotocin-Diabetic Rats. Braz. J. Microbiol. 2012, 43, 1613–1619. [Google Scholar] [CrossRef]
- Ghoneim, M.A.M.; Hassan, A.I.; Mahmoud, M.G.; Asker, M.S. Effect of polysaccharide from Bacillus subtilis sp. on cardiovascular diseases and atherogenic indices in diabetic rats. BMC Complement. Altern. Med. 2016, 16, 112. [Google Scholar] [CrossRef]
- Huang, Z.; Lin, F.; Zhu, X.; Zhang, C.; Jiang, M.; Lu, Z. An exopolysaccharide from Lactobacillus plantarum H31 in pickled cabbage inhibits pancreas α-amylase and regulating metabolic markers in HepG2 cells by AMPK/PI3K/Akt pathway. Int. J. Biol. Macromol. 2020, 143, 775–784. [Google Scholar] [CrossRef]
- Ding, X.; Zhang, J.; Jiang, P.; Xu, X.; Liu, Z. Structural features and hypoglycaemie activity of an exopolysaccharide produced by Sorangium cellulosum. Lett. Appl. Microbiol. 2004, 38, 223–228. [Google Scholar] [CrossRef]
- Patterson, E.E.; Ryan, P.M.; Cryan, J.F.; Dinan, T.G.; Paul Ross, R.; Fitzgerald, G.F.; Stanton, C.E. Gut microbiota, obesity and diabetes. Postgrad. Med. J. 2016, 92, 286–300. [Google Scholar] [CrossRef]
- Abdelazez, A.; Abdelmotaal, H.; Evivie, S.E.; Melak, S.; Jia, F.F.; Khoso, M.H.; Zhu, Z.T.; Zhang, L.J.; Sami, R.; Meng, X.C. Screening potential probiotic characteristics of lactobacillus brevis strains in vitro and intervention effect on type i diabetes in vivo. Biomed. Res. Int. 2018, 2018. [Google Scholar] [CrossRef]
- Marques, T.M.; Patterson, E.; Wall, R.; O’Sullivan, O.; Fitzgerald, G.F.; Cotter, P.D.; Dinan, T.G.; Cryan, J.F.; Ross, R.P.; Stanton, C. Influence of GABA and GABA-producing Lactobacillus brevis DPC 6108 on the development of diabetes in a streptozotocin rat model. Benef. Microbes 2016, 7, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, L.; Zhu, X.; Lu, Z.; Lu, Y. Effect of γ-aminobutyric acid-rich yogurt on insulin sensitivity in a mouse model of type 2 diabetes mellitus. J. Dairy Sci. 2020, 103, 7719–7729. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Dang, H.N.; Yong, J.; Chui, W.S.; Dizon, M.P.G.; Yaw, C.K.Y.; Kaufman, D.L. Oral treatment with γ-aminobutyric acid improves glucose tolerance and insulin sensitivity by inhibiting inflammation in high fat diet-fed mice. PLoS ONE 2011, 6, e25338. [Google Scholar] [CrossRef]
- Soltani, N.; Qiu, H.; Aleksic, M.; Glinka, Y.; Zhao, F.; Liu, R.; Li, Y.; Zhang, N.; Chakrabarti, R.; Ng, T.; et al. GABA exerts protective and regenerative effects on islet beta cells and reverses diabetes. Proc. Natl. Acad. Sci. USA 2011, 108, 11692–11697. [Google Scholar] [CrossRef] [PubMed]
- Taneera, J.; Jin, Z.; Jin, Y.; Muhammed, S.J.; Zhang, E.; Lang, S.; Salehi, A.; Korsgren, O.; Renström, E.; Groop, L.; et al. γ-Aminobutyric acid (GABA) signalling in human pancreatic islets is altered in type 2 diabetes. Diabetologia 2012, 55, 1985–1994. [Google Scholar] [CrossRef] [PubMed]
- Chronopoulos, A.; Kalluri, R. Emerging role of bacterial extracellular vesicles in cancer. Oncogene 2020, 39, 6951–6960. [Google Scholar] [CrossRef] [PubMed]
- Lakhter, A.J.; Sims, E.K. Minireview: Emerging roles for extracellular vesicles in diabetes and related metabolic disorders. Mol. Endocrinol. 2015, 29, 1535–1548. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Zheng, L.; Zou, X.; Wang, J.; Zhong, J.; Zhong, T. Extracellular vesicles in type 2 diabetes mellitus: Key roles in pathogenesis, complications, and therapy. J. Extracell. Vesicles 2019, 8, 1625677. [Google Scholar] [CrossRef]
- Lim, K.H.; Han, J.H.; Lee, J.Y.; Park, Y.S.; Cho, Y.S.; Kang, K.D.; Yuk, W.J.; Hwang, K.Y.; Seong, S.I.; Kim, B.; et al. Assessment of antidiabetogenic potential of fermented soybean extracts in streptozotocin-induced diabetic rat. Food Chem. Toxicol. 2012, 50, 3941–3948. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, Y.; Li, F.; Gu, Z.; Liu, M.; Shao, T.; Zhang, L.; Zhou, G.; Pan, C.; He, L.; et al. Probiotic culture supernatant improves metabolic function through FGF21-adiponectin pathway in mice. J. Nutr. Biochem. 2020, 75, 108256. [Google Scholar] [CrossRef] [PubMed]
- Zouari, R.; Ben Abdallah-Kolsi, R.; Hamden, K.; El Feki, A.; Chaabouni, K.; Makni-Ayadi, F.; Sallemi, F.; Ellouze-Chaabouni, S.; Ghribi-Aydi, D. Assessment of the antidiabetic and antilipidemic properties of bacillus subtilis spb1 biosurfactant in alloxan-induced diabetic rats. Biopolymers 2015, 104, 764–774. [Google Scholar] [CrossRef]
- Zouari, R.; Hamden, K.; El Feki, A.; Chaabouni, K.; Makni-Ayadi, F.; Sallemi, F.; Ellouze-Chaabouni, S.; Ghribi-Aydi, D. Evaluation of Bacillus subtilis SPB1 biosurfactant effects on hyperglycemia, angiotensin I-converting enzyme (ACE) activity and kidney function in rats fed on high-fat–high-fructose diet. Arch. Physiol. Biochem. 2017, 123, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Hossain, U.; Das, A.K.; Ghosh, S.; Sil, P.C. An overview on the role of bioactive α-glucosidase inhibitors in ameliorating diabetic complications. Food Chem. Toxicol. 2020, 145, 111738. [Google Scholar] [CrossRef]
- Li, X.; Wang, N.; Yin, B.; Fang, D.; Jiang, T.; Fang, S.; Zhao, J.; Zhang, H.; Wang, G.; Chen, W. Effects of Lactobacillus plantarum CCFM0236 on hyperglycaemia and insulin resistance in high-fat and streptozotocin-induced type 2 diabetic mice. J. Appl. Microbiol. 2016, 121, 1727–1736. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.Y.; Utra, U.; Ahmad, R.; Rather, I.A.; Park, Y.H. Evaluation of probiotic potential and anti-hyperglycemic properties of a novel Lactobacillus strain isolated from water kefir grains. Food Sci. Biotechnol. 2018, 27, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Li, N.; Yue, Y.; Wang, C.; Zhao, L.; Evivie, S.E.; Li, B.; Huo, G. Screening for Potential Novel Probiotics With Dipeptidyl Peptidase IV-Inhibiting Activity for Type 2 Diabetes Attenuation in vitro and in vivo. Front. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, E.E. Dipeptidyl peptidase inhibitor therapy in type 2 diabetes: Control of the incretin axis and regulation of postprandial glucose and lipid metabolism. Peptides 2018, 100, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Twigg, M.S.; Baccile, N.; Banat, I.M.; Déziel, E.; Marchant, R.; Roelants, S.; Van Bogaert, I.N.A. Microbial biosurfactant research: Time to improve the rigour in the reporting of synthesis, functional characterization and process development. Microb. Biotechnol. 2021, 14, 147–170. [Google Scholar] [CrossRef]
- Adetunji, A.I.; Olaniran, A.O. Production and potential biotechnological applications of microbial surfactants: An overview. Saudi J. Biol. Sci. 2021, 28, 669–679. [Google Scholar] [CrossRef]
- Cavallari, J.F.; Fullerton, M.D.; Duggan, B.M.; Foley, K.P.; Denou, E.; Smith, B.K.; Desjardins, E.M.; Henriksbo, B.D.; Kim, K.J.; Tuinema, B.R.; et al. Muramyl Dipeptide-Based Postbiotics Mitigate Obesity-Induced Insulin Resistance via IRF4. Cell Metab. 2017, 25, 1063–1074.e3. [Google Scholar] [CrossRef]
- Shahmirzadi, F.E.; Ghavamzadeh, S.; Zamani, T. The Effect of Conjugated Linoleic Acid Supplementation on Body Composition, Serum Insulin and Leptin in Obese Adults. Arch. Iran. Med. 2019, 22, 255–261. [Google Scholar]
- Saha, S.S.; Ghosh, M. Antioxidant and anti-inflammatory effect of conjugated linolenic acid isomers against streptozotocin-induced diabetes. Br. J. Nutr. 2012, 108, 974–983. [Google Scholar] [CrossRef]
- Gao, Z.; Yin, J.; Zhang, J.; Ward, R.E.; Martin, R.J.; Lefevre, M.; Cefalu, W.T.; Ye, J. Butyrate Improves Insulin Sensitivity and Increases Energy Expenditure in Mice. Diabetes 2009, 58, 1509–1517. [Google Scholar] [CrossRef]
- Matheus, V.; Monteiro, L.; Oliveira, R.; Maschio, D.; Collares-Buzato, C. Butyrate reduces high-fat diet-induced metabolic alterations, hepatic steatosis and pancreatic beta cell and intestinal barrier dysfunctions in prediabetic mice. Exp. Biol. Med. 2017, 242, 1214–1226. [Google Scholar] [CrossRef] [PubMed]
- Traisaeng, S.; Batsukh, A.; Chuang, T.H.; Herr, D.R.; Huang, Y.F.; Chimeddorj, B.; Huang, C.M. Leuconostoc mesenteroides fermentation produces butyric acid and mediates Ffar2 to regulate blood glucose and insulin in type 1 diabetic mice. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Barros, C.P.; Guimarães, J.T.; Esmerino, E.A.; Duarte, M.C.K.; Silva, M.C.; Silva, R.; Ferreira, B.M.; Sant’Ana, A.S.; Freitas, M.Q.; Cruz, A.G. Paraprobiotics and postbiotics: Concepts and potential applications in dairy products. Curr. Opin. Food Sci. 2020, 32, 1–8. [Google Scholar] [CrossRef]
- Almada, C.N.; Almada-Érix, C.N.; Costa, W.K.A.; Silva, J.; Cabral, L.; Noronha, M.F.; Gonçalves, A.E.S.S.; dos Santos, A.; Lollo, P.C.; Magnani, M.; et al. Wheat-durum pasta added of inactivated Bifidobacterium animalis decreases glucose and total cholesterol levels and modulates gut microbiota in healthy rats. Int. J. Food Sci. Nutr. 2021, 1–13. [Google Scholar] [CrossRef]
- Ben Othman, M.; Sakamoto, K. Effect of inactivated Bifidobacterium longum intake on obese diabetes model mice (TSOD). Food Res. Int. 2020, 129. [Google Scholar] [CrossRef] [PubMed]
- Plovier, H.; Everard, A.; Druart, C.; Depommier, C.; Van Hul, M.; Geurts, L.; Chilloux, J.; Ottman, N.; Duparc, T.; Lichtenstein, L.; et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 2017, 23, 107–113. [Google Scholar] [CrossRef]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; Van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: A proof-of-concept exploratory study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef]
- Humblot, C.; Turpin, W.; Chevalier, F.; Picq, C.; Rochette, I.; Guyot, J.P. Determination of expression and activity of genes involved in starch metabolism in Lactobacillus plantarum A6 during fermentation of a cereal-based gruel. Int. J. Food Microbiol. 2014, 185, 103–111. [Google Scholar] [CrossRef]
- Feng, L.; Xie, Y.; Peng, C.; Liu, Y.; Wang, H. A novel antidiabetic food produced via solid-state fermentation of Tartary buckwheat by L. plantarum TK9 and L. paracasei TK1501. Food Technol. Biotechnol. 2018, 56, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Shigemori, S.; Oshiro, K.; Wang, P.; Yamamoto, Y.; Wang, Y.; Sato, T.; Uyeno, Y.; Shimosato, T. Generation of Dipeptidyl Peptidase-IV-Inhibiting Peptides from β -Lactoglobulin Secreted by Lactococcus lactis. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Guo, L.; Zhang, Y.; Walzem, R.L.; Pendergast, J.S.; Printz, R.L.; Morris, L.C.; Matafonova, E.; Stien, X.; Kang, L.; et al. Incorporation of therapeutically modified bacteria into gut microbiota inhibits obesity. J. Clin. Investig. 2014, 124, 3391–3406. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Khatri, P.; Billack, B.; Low, W.-K.; Shao, J. Oral Delivery of Glucagon Like Peptide-1 by a Recombinant Lactococcus lactis. Pharm. Res. 2014, 31, 3404–3414. [Google Scholar] [CrossRef] [PubMed]
- Tomaro-Duchesneau, C.; LeValley, S.L.; Roeth, D.; Sun, L.; Horrigan, F.T.; Kalkum, M.; Hyser, J.M.; Britton, R.A. Discovery of a bacterial peptide as a modulator of GLP-1 and metabolic disease. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Doyle, M.E.; Egan, J.M. Mechanisms of action of glucagon-like peptide 1 in the pancreas. Pharmacol. Ther. 2007, 113, 546–593. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, A.; Allahyar, A.; Greiner, T.U.; Plovier, H.; Gunnel, O.; Larsson, T.; Drucker, D.J.; Delzenne, N.M.; Cani, P.D.; Ba, F. Microbial Modulation of Energy Availability in the Colon Regulates Intestinal Transit. Cell Host Microbe 2013, 14, 582–590. [Google Scholar] [CrossRef]
- Yoon, H.S.; Cho, C.H.; Yun, M.S.; Jang, S.J.; You, H.J.; Kim, J.H.; Han, D.; Cha, K.H.; Moon, S.H.; Lee, K.; et al. Akkermansia muciniphila secretes a glucagon-like peptide-1-inducing protein that improves glucose homeostasis and ameliorates metabolic disease in mice. Nat. Microbiol. 2021, 6, 563–573. [Google Scholar] [CrossRef]
- Tlaskalová-Hogenová, H.; Tučková, L.; Lodinová-Žádniková, R.; Štěpánková, R.; Cukrowska, B.; Funda, D.P.; Střiž, I.; Kozáková, H.; Trebichavský, I.; Sokol, D.; et al. Mucosal immunity: Its role in defense and allergy. Int. Arch. Allergy Immunol. 2002, 128, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D. Human gut microbiome: Hopes, threats and promises. Gut 2018, 67, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Aoki, R.; Kamikado, K.; Suda, W.; Takii, H.; Mikami, Y.; Suganuma, N.; Hattori, M.; Koga, Y. A proliferative probiotic Bifidobacterium strain in the gut ameliorates progression of metabolic disorders via microbiota modulation and acetate elevation. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Li, C.; Ding, Q.; Nie, S.P.; Zhang, Y.S.; Xiong, T.; Xie, M.Y. Carrot Juice Fermented with Lactobacillus plantarum NCU116 Ameliorates. J. Agric. Food Chem. 2014, 62, 11884–11891. [Google Scholar] [CrossRef] [PubMed]
- Dai, S.; Pan, M.; El-Nezami, H.S.; Wan, J.M.F.; Wang, M.F.; Habimana, O.; Lee, J.C.Y.; Louie, J.C.Y.; Shah, N.P. Effects of Lactic Acid Bacteria-Fermented Soymilk on Isoflavone Metabolites and Short-Chain Fatty Acids Excretion and Their Modulating Effects on Gut Microbiota. J. Food Sci. 2019, 84, 1854–1863. [Google Scholar] [CrossRef]
- Alvarez-Silva, C.; Kashani, A.; Hansen, T.H.; Pinna, N.K.; Anjana, R.M.; Dutta, A.; Saxena, S.; Støy, J.; Kampmann, U.; Nielsen, T.; et al. Trans-ethnic gut microbiota signatures of type 2 diabetes in Denmark and India. Genome Med. 2021, 13, 1–13. [Google Scholar] [CrossRef]
- Pinna, N.K.; Anjana, R.M.; Saxena, S.; Dutta, A.; Gnanaprakash, V.; Rameshkumar, G.; Aswath, S.; Raghavan, S.; Rani, C.S.S.; Radha, V.; et al. Trans-ethnic gut microbial signatures of prediabetic subjects from India and Denmark. Genome Med. 2021, 13, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Bolnick, D.I.; Snowberg, L.K.; Hirsch, P.E.; Lauber, C.L.; Org, E.; Parks, B.; Lusis, A.J.; Knight, R.; Caporaso, J.G.; Svanbäck, R. Individual diet has sex-dependent effects on vertebrate gut microbiota. Nat. Commun. 2014, 5, 1–13. [Google Scholar] [CrossRef]
- Zhang, Z.; Tian, T.; Chen, Z.; Liu, L.; Luo, T.; Dai, J. Characteristics of the gut microbiome in patients with prediabetes and type 2 diabetes. PeerJ 2021, 9. [Google Scholar] [CrossRef]
- Duarte, A.M.; Guarino, M.P.; Barroso, S.; Gil, M.M. Phytopharmacological strategies in the management of type 2 diabetes mellitus. Foods 2020, 9, 271. [Google Scholar] [CrossRef]
- Abdelli, I.; Benariba, N.; Adjdir, S.; Fekhikher, Z.; Daoud, I.; Terki, M.; Benramdane, H.; Ghalem, S. In silico evaluation of phenolic compounds as inhibitors of A-amylase and A-glucosidase. J. Biomol. Struct. Dyn. 2021, 39, 816–822. [Google Scholar] [CrossRef]
- Santos, R.; Ruza, D.; Cunha, E.; Tavares, L.; Oliveira, M. Diabetic foot infections: Application of a nisin-biogel to complement the activity of conventional antibiotics and antiseptics against Staphylococcus aureus biofilms. PLoS ONE 2019, 14, e0220000. [Google Scholar] [CrossRef] [PubMed]
- Smits, L.P.; Bouter, K.E.C.; De Vos, W.M.; Borody, T.J.; Nieuwdorp, M. Therapeutic potential of fecal microbiota transplantation. Gastroenterology 2013, 145, 946–953. [Google Scholar] [CrossRef] [PubMed]
- Kootte, R.S.; Levin, E.; Salojärvi, J.; Smits, L.P.; Hartstra, A.V.; Udayappan, S.D.; Hermes, G.; Bouter, K.E.; Koopen, A.M.; Holst, J.J.; et al. Improvement of Insulin Sensitivity after Lean Donor Feces in Metabolic Syndrome Is Driven by Baseline Intestinal Microbiota Composition. Cell Metab. 2017, 26, 611–619. [Google Scholar] [CrossRef]
- Vrieze, A.; Van Nood, E.; Holleman, F.; Salojärvi, J.; Kootte, R.S.; Bartelsman, J.F.W.M.; Dallinga-Thie, G.M.; Ackermans, M.T.; Serlie, M.J.; Oozeer, R.; et al. Transfer of Intestinal Microbiota From Lean Donors Increases Insulin Sensitivity in Individuals With Metabolic Syndrome. Gastroenterology 2012, 143, 913–916. [Google Scholar] [CrossRef] [PubMed]


| Type of Research | Bioactive Component | Source/Origin | Model System | Main Findings | Study |
|---|---|---|---|---|---|
| Animal | Levan | Bacillus licheniformis | Wistar rats induced T1D with alloxan (150 mg/kg BW) | ↓Glycaemia, ↑glycogen level, ↓AST, ALT, bilirubin, creatinine, and urea levels | Dahech et al., 2011 [83] |
| Levan | Bacillus subtilis (Natto) | Wistar rats induced T1D with STZ (65 mg/kg BW) | No hypoglycaemic effect. No improvement of diabetes symptoms | Bazani et al., 2012 [88] | |
| Exopolysaccharide (unspecified) | Bacillus subtilis | Sprague-Dawley rats induced T1D with STZ (65 mg/kg BW) | ↓FBG, ↑serum insulin levels, ↓TC, LDL, VLDL and TG, ↑HDL in treated vs. control rats | Ghoneim et al., 2016 [89] | |
| Selenium-enriched exopolysaccharide | Enterobacter cloacaceae Z0206 | Female ICR mice induced T1D with alloxan (190 mg/kg) | ↓FBG, ↑serum insulin level, ↓glycosylated serum protein, ↑BW, ↓TC and TG in treated vs. control mice | Jin et al., 2012 [26] | |
| Exopolysaccharide (unspecified) | Sorangium cellulosum NUST06 | Mice (Kunming strain) induced T1D with alloxan (250 mL/kg BW) | ↓FBG in both healthy and alloxan-induced diabetic mice | Ding et al., 2004 [91] | |
| Cell line | Exopolysaccharide (unspecified) | Lactobacillus plantarum H31-2 | In vitro, insulin-resistant HepG2 cells | ↓Supernatant glucose concentration of insulin-resistant HepG2 cells, inhibition of pancreas α-amylase, upregulation of the expression of GLUT-4, Akt-2, and AMPK | Huang et al., 2020 [90] |
| Compound | Source/Origin | Model System | Main Findings | Study |
|---|---|---|---|---|
| GABA | GABA-containing fermented mung bean extract with Mardi rhizopus 5351 inoculums, low and high doses | T1D Balb/c induced T1D with high doses of alloxan (100 mg/kg BW) and glucose-induced hyperglycaemic Balb/c mice | Glucose-induced mice: ↓FBG Alloxan-induced mice: ↓ Reduced FBG, ↑insulin serum levels, ↓ TC and TG serum levels (vs. nonfermented), restored antioxidant status (↓MDA and NO) | Yeap et al., 2012 [84] |
| Yogurt fermented with Streptococcus thermophiles fmb5, low and high doses | C57BL/6 mice induced T2D with low doses of STZ (100 mg/kg BW) | Low and high doses: No hypoglycaemic effect, ↑HOMA-β, improved glucose tolerance and insulin resistance, normalised fat, kidney and liver coefficients, ↓serum urea nitrogen, no effect on HbA1c nor BW. High doses: Normalised pancreatic histology, preserved islet cells function, ↓ TC and LDL, HDL | Li et al., 2020 [95] | |
| EVs | Akkermansia muciniphila ATCC BAA-835 | C57BL/6 mice induced T2D with HFD | In vivo: Reduced BW, attenuated intestinal damage following HFD, increase expression of tight junction proteins, improved glucose tolerance (OGTT) In vitro: Enhanced barrier function in Caco-2 cells | Chelakkot et al., 2018 [40] |
| Type of Research | Microorganism | Inactivation Method | Model System | Main Findings | Study |
|---|---|---|---|---|---|
| Animal | Lactobacillus casei CCFM419, L. plantarum X1, L. rhamnosus Y37, L. brevis CCFM648, and L. plantarum CCFM36 | Heat treatment (80 °C for 30 min) | C57BL/6J mice induced T2D with STZ (100 mg/kg BW) | Inanimate probiotic: ↑ Serum IL-6 levels and faecal acetic levels Live probiotic: Improved insulin tolerance, normalised serum IL-10, TNF-α and IL-6 levels, ↑faecal acetic and butyrate levels, ↑faecal Lactobacillus, Akkermansia, and Bifidobacterium genera, ↑faecal actinobacteria (%) Both: ↓ FBG, normalisation of HbA1c and leptin levels, ↑ileum L cell levels, ↑faecal Firmicutes/Bacteroidetes ratio Both with stronger effect with live probiotics: Improved glucose tolerance, protection of pancreatic histological characteristics | Li et al., 2016 [25] |
| Bifidobacterium longum BR-108; low, medium, and high doses | Heat and pressure treatment (autoclaved at 105 °C for 20 min) | Tsumura Suzuki obese diabetes (TSOD) mice (genetically obese mice) | All the doses: ↓BG gain (vs. control mice), ↓adipose tissue accumulation, no differences in food consumption, ↓serum creatinine levels Medium and high doses: ↓FBG, ↓NEFAs, ↓creatinine urine levels Medium dose: ↓TC High doses: Improved glucose tolerance (OGTT), ↓TG | Ben Othman et al., 2019 [121] | |
| Bifidobacterium animalis subsp. lactis Bb-12, incorporated in wheat pasta | Irradiation (gamma-irradiation on ice, at 2.5 Kilogray) | Healthy Wistar rats | Control and paraprobiotic pasta: ↓FBG and TC (vs. control diet), no differences in food consumption and BW, TG, HDL, AST, ALT, and microbiota alpha-diversity indexes Paraprobiotic pasta: Differential microbiota composition (vs. control diet and control pasta) | Almada et al., 2021 [120] | |
| Akkermansia muciniphila | Pasteurised (70 °C for 30 min) | C57BL/6J mice (normal chow or high-fat diet) | ↓ IR index, ↑ faecal caloric content, ↑ goblet cell density, normalisation of adipocyte diameter and ↓leptin levels (vs. live microorganism) | Plovier et al., 2017 [122] | |
| Human | Lacticaseibacillus casei 01; in whey-grape juice drink | Ohmic heating (8 V/cm, 95 °C/7 min, 60 Hz) | In vitro experiments; healthy volunteers (n = 15) | Preliminary in vitro experiments: Live and inanimate probiotic had α-glucosidase and α-amylase inhibitory activities Postprandial glucose levels in healthy volunteers: Accelerated increase in PBG with the probiotic and inanimate probiotic drinks (vs. control) due to differences in sugar content with the control (water), no differences in AUC values, inanimate probiotic had a similar effect to control. | Barros et al., 2021 [85] |
| Akkermansia muciniphila | Pasteurised | Volunteers with excess body weight (overweight or obese), insulin resistance and a metabolic syndrome | ↓ insulin levels, improved insulin sensitivity index, fasting glycaemia and HbA1c were not modified (vs. placebo group) | Depommier et al., 2019 [123] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabello-Olmo, M.; Araña, M.; Urtasun, R.; Encio, I.J.; Barajas, M. Role of Postbiotics in Diabetes Mellitus: Current Knowledge and Future Perspectives. Foods 2021, 10, 1590. https://doi.org/10.3390/foods10071590
Cabello-Olmo M, Araña M, Urtasun R, Encio IJ, Barajas M. Role of Postbiotics in Diabetes Mellitus: Current Knowledge and Future Perspectives. Foods. 2021; 10(7):1590. https://doi.org/10.3390/foods10071590
Chicago/Turabian StyleCabello-Olmo, Miriam, Miriam Araña, Raquel Urtasun, Ignacio J. Encio, and Miguel Barajas. 2021. "Role of Postbiotics in Diabetes Mellitus: Current Knowledge and Future Perspectives" Foods 10, no. 7: 1590. https://doi.org/10.3390/foods10071590
APA StyleCabello-Olmo, M., Araña, M., Urtasun, R., Encio, I. J., & Barajas, M. (2021). Role of Postbiotics in Diabetes Mellitus: Current Knowledge and Future Perspectives. Foods, 10(7), 1590. https://doi.org/10.3390/foods10071590






