Hydrogen Peroxide Effects on Natural-Sourced Polysacchrides: Free Radical Formation/Production, Degradation Process, and Reaction Mechanism—A Critical Synopsis
Abstract
1. Introduction
2. Justification of Synopsis and Objective Statement
3. Survey Methodology
4. Discussion of Findings
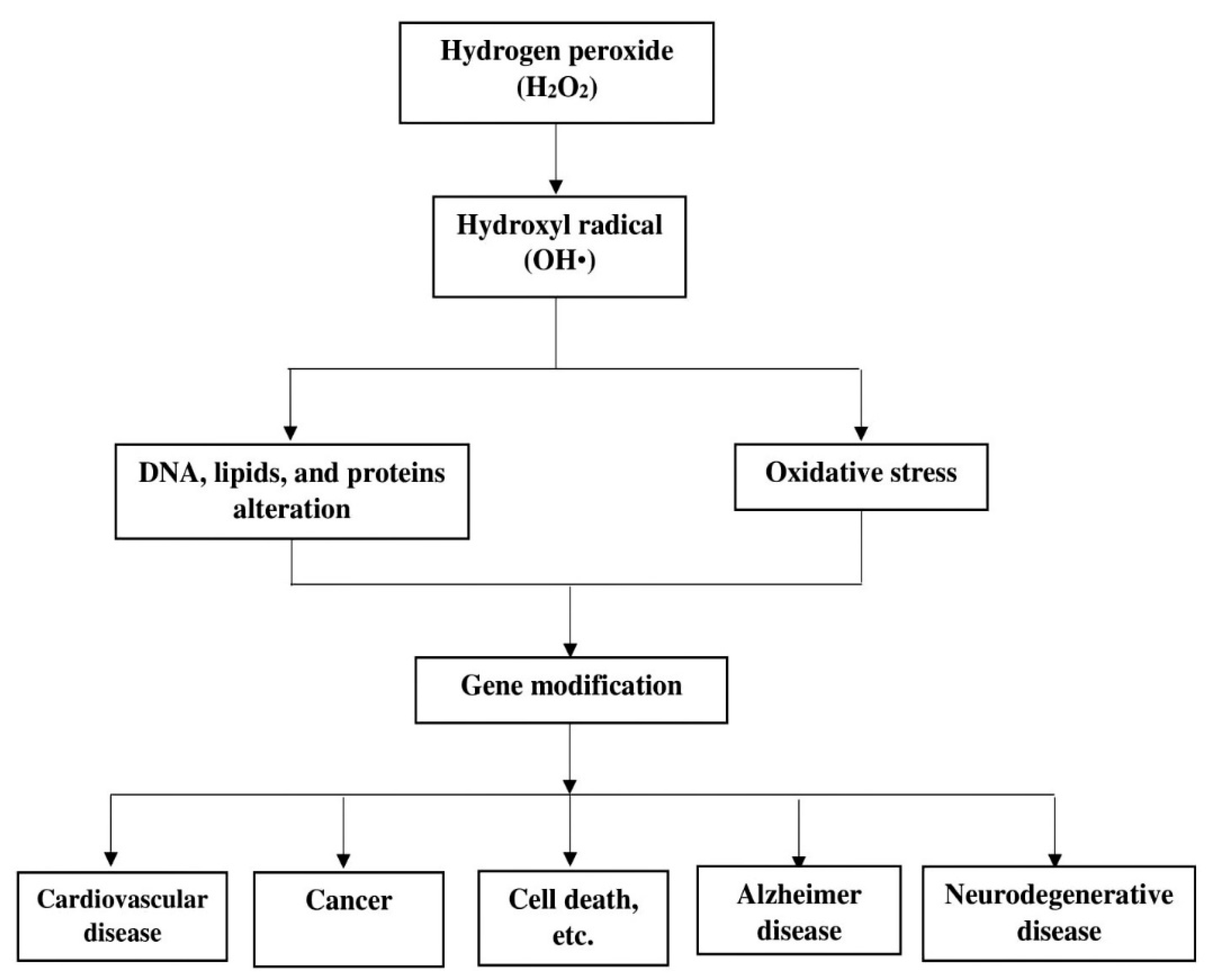
4.1. H2O2 Formation and Factors Influencing its Production
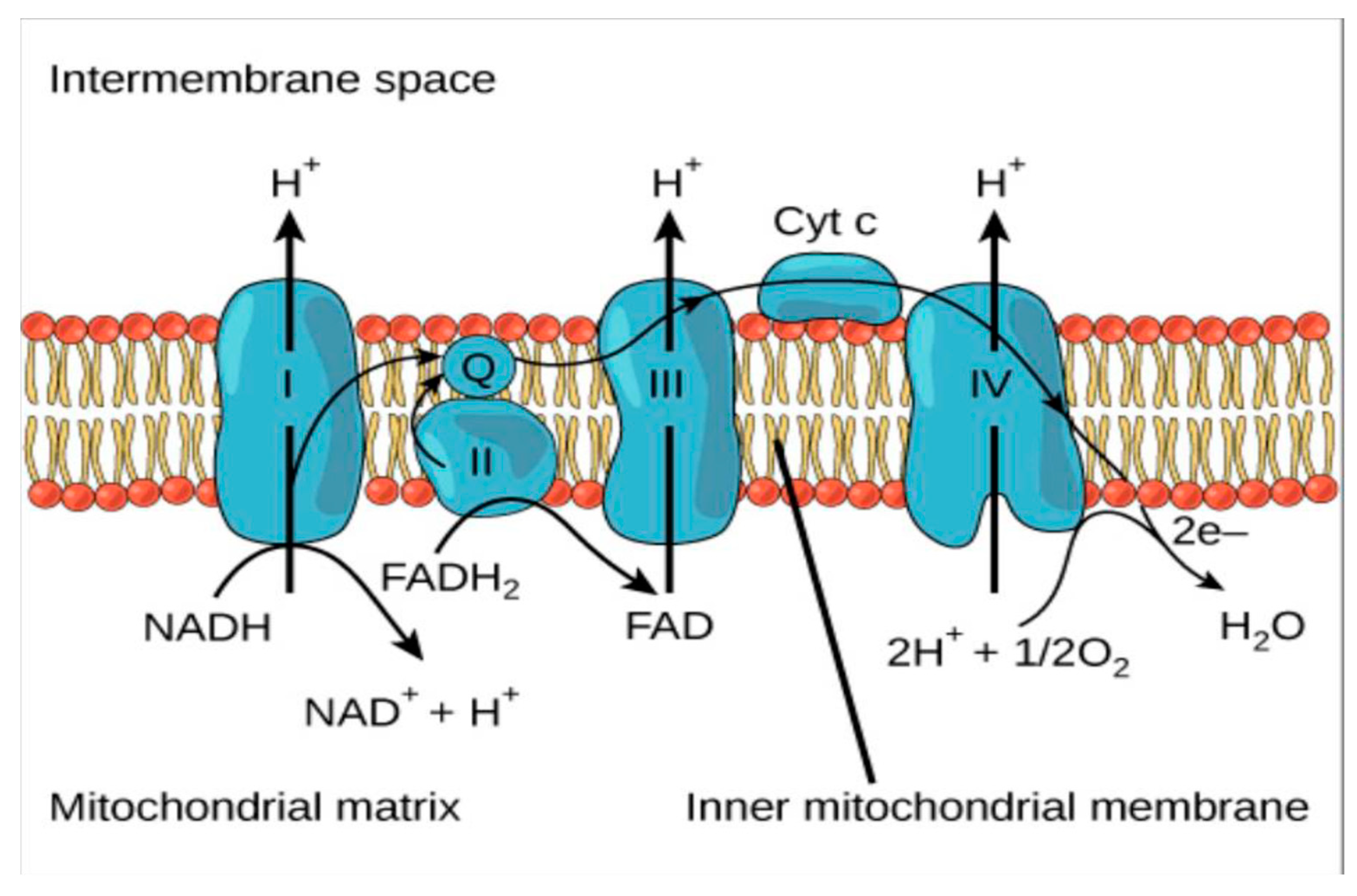
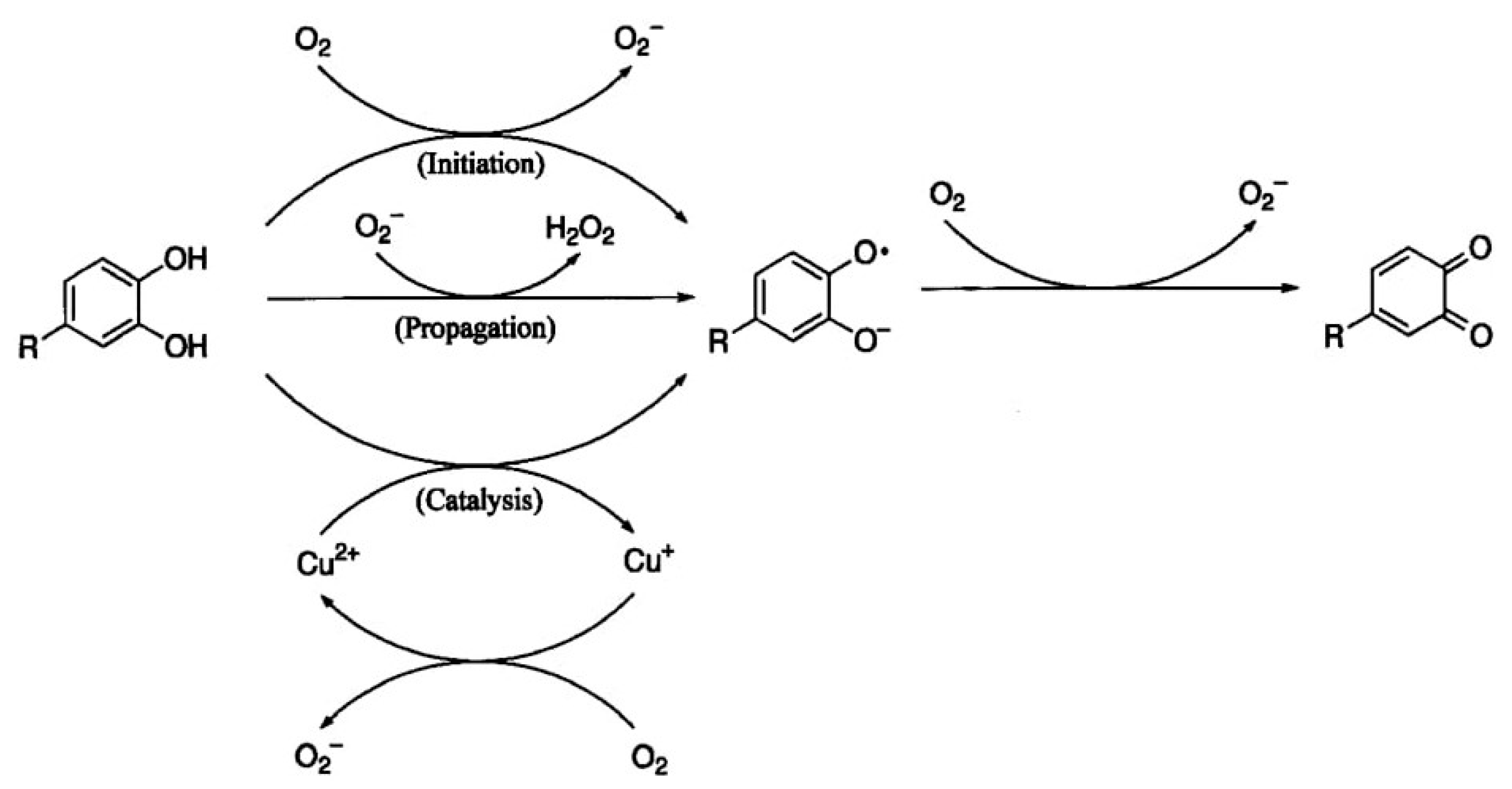
4.1.1. Formation of H2O2
- (a)
- In vitro production of H2O2
- (b)
- In vivo production of H2O2
4.1.2. Factors That Influence the Production of H2O2
- (a)
- Composition and structure of phenolic compounds
- (b)
- Oxygen and metal ions
- (c)
- pH
- (d)
- Incubation condition
- (e)
- Enzymes
4.2. Polysaccharide: The Degradation Process and Effects with H2O2
4.2.1. Polysaccharide Degradation
4.2.2. Methods of Polysaccharide Degradation
4.2.3. Effect of Polysaccharide Degradation with H2O2
- (a)
- Physicochemical Properties
- (i)
- Molecular weight
- (ii)
- Intrinsic viscosity
- (iii)
- Water solubility
- (b)
- Structural Properties
- (c)
- Bioactivities
- (i)
- Antioxidant activity
- Radical scavenging capacity
- Reducing power
- Metal ion chelating ability
4.3. The Reaction Mechanism Involving Polysaccharides’ Antioxidant Capacity on Free Radicals: A Primer
4.4. Some Examples of Antioxidative Polysaccharides and Their Sources
5. Concluding Remarks and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Morello, M.J.; Shahidi, F.; Ho, C.T. Free Radicals in Foods: Chemistry, Nutrition and Health Effects; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2002; pp. 1–9. [Google Scholar]
- Maddu, N. Diseases Related to Types of Free Radicals; Intech Open: London, UK, 2019; pp. 1–8. [Google Scholar]
- Turkan, I. ROS and RNS key signalling molecules in plants. J. Exp. Bot. 2018, 69, 3313–3315. [Google Scholar] [CrossRef]
- Messner, K.R.; Imlay, J.A. Mechanism of Superoxide and Hydrogen Peroxide Formation by Fumarate Reductase, Succinate Dehydrogenase, and Aspartate Oxidase; American Society for Biochemistry and Molecular Biology Inc.: Rockville, MD, USA, 2002. [Google Scholar]
- Gulcin, I. Antioxidants and Antioxidant Methods: An Updated Review. Archives of Toxicology; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Patel, R.; Rinker, L.; Peng, J.; Chilian, W.M. Reactive Oxygen Species: The Good and the Bad. React. Oxyg. Species (ROS) Living Cells 2018, 7, 7–20. [Google Scholar]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. [Google Scholar] [CrossRef]
- Genestra, M. Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cell. Signal. 2007, 19, 1807–1819. [Google Scholar] [CrossRef]
- Lee, J.Y.; Jang, Y.W.; Kang, H.S.; Moon, H.; Sim, S.S.; Kim, C.J. Anti-inflammatory action of phenolic compounds from Gastrodia elata root. Arch. Pharmacal Res. 2006, 29, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.W.; Albers, A.E.; Pralle, A.; Isacoff, E.Y.; Chang, C.J. Boronate-Based Fluorescent Probes for Imaging Cellular Hydrogen Peroxide. J. Am. Chem. Soc. 2005, 127, 16652–16659. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.; Hwang, O.; Yoo, D.; Khang, G.; Lee, D. Detection of hydrogen peroxide in vitro and in vivo using peroxalate chemilumiescent micelles. Bull. Korean Chem. Soc. 2011, 33, 2187–2192. [Google Scholar] [CrossRef]
- Elmarakby, A.A.; Pollock, D.M.; Imig, J.D. Renal dysfunction in hypertension and obesity. In Comprehensive Medicinal Chemistry II; Taylor, J.B., Triggle, D.J., Eds.; Elsevier Ltd.: Amsterdam, The Netherlands, 2007; Volume 6, pp. 575–595. [Google Scholar]
- Chatterjee, S. Oxidative Stress, Inflammation, and Disease. In Oxidative Stress and Biomaterials; Dziuba, T., Butterfield, A.D., Eds.; Elsevier Academic Press: Amsterdam, The Netherlands, 2016; pp. 35–58. ISBN 9780128032695. [Google Scholar]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef]
- Vamanu, E. Antioxidant properties of polysaccharides obtained by batch cultivation of Pleurotus ostreatus mycelium. Nat. Prod. Res. 2012, 27, 1115–1118. [Google Scholar] [CrossRef]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules 2019, 24, 1583. [Google Scholar] [CrossRef]
- Lennicke, C.; Rahn, J.; Lichtenfels, R.; Wessjohann, J.A.; Seliger, B. Hydrogen peroxide—production, fate and role in redox signalling of tumour cells. Cell Commun. Signal. 2015, 13, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Armogida, M.; Nisticò, R.; Mercuri, N.B. Therapeutic potential of targeting hydrogen peroxide metabolism in the treatment of brain ischaemia. Br. J. Pharmacol. 2012, 166, 1211–1224. [Google Scholar] [CrossRef]
- Weinstain, R.; Savariar, E.N.; Felsen, C.N.; Tsien, R.Y. In Vivo Targeting of Hydrogen Peroxide by Activatable Cell-Penetrating Peptides. J. Am. Chem. Soc. 2014, 136, 874–877. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Clement, M.V.; Long, L.H. Hydrogen peroxide in the human body. FEBS Lett. 2000, 486, 10–13. [Google Scholar] [CrossRef]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef] [PubMed]
- Holmström, K.M.; Finkel, T. Cellular mechanisms and physiological consequences of redox-dependent signalling. Nat. Rev. Mol. Cell Biol. 2014, 15, 411–421. [Google Scholar] [CrossRef]
- Park, H.; Kim, S.; Song, Y.; Seung, K.; Hong, D.; Khang, G.; Lee, D. Antioxidant and anti-inflammatory activities of hydroxybenzyl alcohol releasing biodegradable polyoxalate nanoparticles. Biomacromolecules 2010, 11, 2103–2108. [Google Scholar] [CrossRef] [PubMed]
- Collin, F. Chemical Basis of Reactive Oxygen Species Reactivity and Involvement in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 2407. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Lui, Y.M.; Qi, Z.M.; Wang, S.Y.; Liu, S.X.; Li, X.; Wang, H.J.; Xia, X.C. An overview on natural polysaccharides with antioxidant properties. Curr. Med. Chem. 2013, 20, 2899–2913. [Google Scholar] [CrossRef]
- Erickson, M.C. Lipid oxidation in muscle foods. In Food Lipids Chemistry, Nutrition and Biotechnology; Akoh, C.C., Min, D.B., Eds.; CRC Press: New York, NY, USA, 2003; pp. 365–411. [Google Scholar]
- Bensid, A.; El Abed, N.; Houicher, A.; Regenstein, J.M.; Özogul, F. Antioxidant and antimicrobial preservatives: Properties, mechanism of action and applications in food—A review. Crit. Rev. Food Sci. Nutr. 2020, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Jamila, V.; Vavrikova, E. Chitosan derivatives with antimicrobial, antitumor and anti-oxidant activities: A review. Curr. Pharm. Design. 2011, 17, 3596–3607. [Google Scholar]
- Yang, L.; Zhang, L.M. Chemical structural and chain conformational characterization of some bioactive polysaccharides isolated from natural sources. Carbohydr. Polym. 2009, 76, 349–361. [Google Scholar] [CrossRef]
- Wang, N.; Zhang, X.; Wang, S.; Guo, Q.; Li, Z.; Liu, H.; Wang, C. Structural characterisation and immunomodulatory activity of polysaccharides from white asparagus skin. Carbohydr. Polym. 2020, 227, 115314. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Gao, X.D.; Zhou, G.C.; Cai, L.; Yao, W.B. In vitro and in vivo antioxidant activity of aqueous extract from Choerospondias axillaris fruit. Food Chem. 2008, 106, 888–895. [Google Scholar] [CrossRef]
- Zhang, Y.; Dai, L.; Kong, X.; Chen, L. Characterization and in vitro antioxidant activities of polysaccharides from Pleurotus ostreatus. Int. J. Biol. Macromol. 2012, 51, 259–265. [Google Scholar] [CrossRef]
- Fan, L.; Ding, S.; Ai, L.; Deng, K. Antitumor and immunomodulatory activity of water-soluble polysaccharide from Inonotus obliquus. Carbohydr. Polym. 2012, 90, 870–874. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, X.; Zhao, M.; Qi, H. Free-radical degradation by Fe2+/Vc/H2O2 and antioxidant activity of polysaccharide from Tremella fuciformis. Carbohydr. Polym. 2014, 112, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Jhamandas, J.H.; Wie, M.B.; Harris, K.; MacTavish, D.; Kar, S. Fucoidaninhibits cellular and neurotoxic effects of β-amyloid (Aβ) in rat cholinergic basalforebrain neurons. Eur. J. Neurosci. 2005, 21, 2649–2659. [Google Scholar] [CrossRef]
- Zhang, N.; Chen, H.; Ma, L.; Zhang, Y. Physical modifications of polysaccharide from Inonotus obliquus and the antioxidant properties. Int. J. Biol. Macromol. 2013, 54, 209–215. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Wang, X.M.; Mo, X.F.; Qi, H.M. Degradation and the antioxidant activity of polysaccharide from Enteromorpha linza. Carbohydr. Polym. 2013, 92, 2084–2087. [Google Scholar] [CrossRef]
- Jing, L.; Sheng, J.; Jiang, J.; Wang, Y.; Shen, X.; Liu, D.; Zhang, W.; Mao, S. Chemical characteristics and cytoprotective activities of polysaccharide fractions from Athyrium Multidentatum (Doll.) Ching. Int. J. Biol. Macromol. 2020, 158, 1227–1238. [Google Scholar] [CrossRef]
- Sun, L.; Wang, L.; Li, J.; Liu, H. Characterization and antioxidant activities of degraded polysaccharides from two marine Chrysophyta. Food Chem. 2014, 160, 1–7. [Google Scholar] [CrossRef]
- Xu, Y.; Niu, X.; Liu, N.; Gao, Y.; Wang, L.; Xu, G.; Li, X.; Yang, Y. Characterisation, antioxidant and hypoglycemic activities of degraded polysaccharides from black currant (Ribes nigrum L.) fruits. Food Chem. 2017, 243, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhu, H.; Li, X.; Liu, Z.; Zheng, W.; Chen, T.; Yu, B.; Wong, K.-H. Induction of Apoptosis and cell cycle arrest in A549 Human Lung Adernocarcinoma cells by Surface-Capping Selenium Nanoparticles: An Effect enhanced by Polysachharide-Protein Complexes from Polyporus rhinoceros. J. Agric. Food Chem. 2013, 61, 9859–9866. [Google Scholar] [CrossRef]
- Shen, H.; Tang, G.; Zeng, G.; Yang, Y.; Cai, X.; Li, D.; Liu, H.; Zhou, N. Purification and characterisation of an antitumor polysaccharide from Portulaca oleracea L. Carbohydr. Polym. 2013, 93, 395–400. [Google Scholar] [CrossRef]
- Saha, S.; Galhardi, L.C.; Yamamoto, K.A.; Linhares, R.E.C.; Bandyopadhyay, S.S.; Sinha, S.; Nozawa, C.; Ray, B. Water-extracted polysaccharides from Azadirachta indica leaves: Structural features, chemical modification and anti-bovine herpesvirus type 1 (BoHV-1) activity. Int. J. Biol. Macromol. 2010, 47, 640–645. [Google Scholar] [CrossRef]
- Zhu, Z.-Y.; Dong, F.; Liu, X.; Lv, Q.; Yang, Y.; Liu, F.; Chen, L.; Wang, T.; Wang, Z.; Zhang, Y. Effects of extraction methods on the yield, chemical structure and anti-tumor activity of polysaccharides from Cordyceps gunnii mycelia. Carbohydr. Polym. 2016, 140, 461–471. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: Oxidative eustress. Redox Biol. 2017, 11, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Kasper, D.L. Oxidative depolymerisation of polysaccharides by reactive oxygen/nitrogen species. Glycobiology 2011, 21, 401–409. [Google Scholar] [CrossRef]
- Pagano, G.; Talamanca, A.A.; Castello, G.; Cordero, M.D.; Dischia, M.; Gadaleta, M.N. Oxidative stress and mitochondrial dysfunction across broad-ranging pathologies: Toward mitochondria-targeted clinical strategies. Oxidative Med. Cell. Longev. 2014, 54, 12–30. [Google Scholar] [CrossRef] [PubMed]
- Lv, W.; Booz, G.W.; Fan, F.; Wang, Y.; Roman, R.J. Oxidative stress and renal fibrosis: Recent insights for the development of novel therapeutic strategies. Front. Physiol. 2018, 9, 105. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Wang, S.; Zhong, W.; Yang, B.; Sun, L.; Zheng, Y. Oxidative stress in the trabecular meshwork (Review). Int. J. Mol. Med. 2016, 38, 995–1002. [Google Scholar] [CrossRef]
- Electron Transport Chain, Lumen, Biology for Majors. Available online: https://courses.lumenlearning.com/wm-biology1/chapter/reading-electron-transport-chain/ (accessed on 23 December 2020).
- Fenton, H.J.H. Oxidation of tartaric acid in the presence of iron. J. Chem. Soc. Trans. 1894, 65, 899–910. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 2nd ed.; Clarendon Press: Oxford, UK, 1989; pp. 1–3. [Google Scholar]
- Kehrer, J.P. The Haber-Weiss reaction and mechanisms of toxicity. Toxicology 2000, 149, 43–50. [Google Scholar] [CrossRef]
- Anraku, M.; Gebicki, J.M.; Iohara, D.; Tomida, H.; Uekama, K.; Maruyama, T.; Hirayama, F.; Otagiri, M. Antioxidant activities of chitosans and its derivatives in in vitro and in vivo studies. Carbohydr. Polym. 2018, 199, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Sindhi, V.; Gupta, V.; Sharma, K.; Bhatnagar, S.; Kumari, R.; Dhaka, N. Potential applications of antioxidants—A review. J. Pharm. Res. 2013, 7, 828–835. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J. Bot. 2012, 2012, 217037. [Google Scholar] [CrossRef]
- Nakagawa, H.; Wachi, M.; Woo, J.-T.; Kato, M.; Kasai, S.; Takahashi, F.; Lee, I.-S.; Nagai, K. Fenton Reaction Is Primarily Involved in a Mechanism of (−)-Epigallocatechin-3-gallate to Induce Osteoclastic Cell Death. Biochem. Biophys. Res. Commun. 2002, 292, 94–101. [Google Scholar] [CrossRef]
- Muller, R. The formation of hydrogen peroxide during oxidation of thiol-containing proteins. J. Inst. Brew. 1997, 103, 307–310. [Google Scholar] [CrossRef]
- Bopitiya, D.; Christensen, D.; Martin, M.; Zhang, J.; Bennett, L.E. Production of hydrogen peroxide in formulated beverages is associated with the presence of ascorbic acid combined with selected redox-active functional ingredients. Food Chem. 2021, 338, 127947. [Google Scholar] [CrossRef]
- Long, L.L.; Lan, A.N.B.; Hsuan, F.L.Y.; Halliwell, B. Generation of hydrogen peroxide by “Antioxidant” beverages and the effect of milk addition. Is cocoa the best beverage? Free Radic. Res. 1999, 31, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, T.; Ichiba, M.; Kuwabara, M.; Kujiya, K.; Kumazawa, S. Mechanisms and structural specificity of hydrogen peroxide formation during oxidation of catechins. Food Sci. Technol. Res. 2002, 8, 261–267. [Google Scholar] [CrossRef][Green Version]
- Akagawa, M.; Shigemitsu, T.; Suyama, K. Production of Hydrogen Peroxide by Polyphenols and Polyphenol-rich Beverages under Quasi-physiological Conditions. Biosci. Biotechnol. Biochem. 2003, 67, 2632–2640. [Google Scholar] [CrossRef]
- Grzesik, M.; Bartosz, G.; Stefaniuk, I.; Pichla, M.; Namieśnik, J.; Sadowska-Bartosz, I. Dietary antioxidants as a source of hydrogen peroxide. Food Chem. 2019, 278, 692–699. [Google Scholar] [CrossRef]
- Nakagawa, T.; Yokozawa, T. Direct scavenging of nitric oxide and superoxide by green tea. Food Chem. Toxicol. 2002, 40, 1745–1750. [Google Scholar] [CrossRef]
- Kim, H.-S.; Quon, M.J.; Kim, J.-A. New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate. Redox Biol. 2014, 2, 187–195. [Google Scholar] [CrossRef]
- Nakayama, T.; Enoki, Y.; Hashimoto, K. Hydrogen Peroxide Formation during Catechin Oxidation Is Inhibited by Superoxide Dismutase. Food Sci. Technol. Int. Tokyo 1995, 1, 65–69. [Google Scholar] [CrossRef][Green Version]
- Wee, L.M.; Long, L.H.; Whiteman, M.; Halliwell, B. Factors affecting the ascorbate- and phenolic-dependent generation of hydrogen peroxide in Dulbecco’s Modified Eagles Medium. Free Radic. Res. 2003, 37, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Clement, M.V.; Ramalingam, J.; Long, L.H. Hydrogen Peroxide. Ubiquitous in cell culture and in vivo? Life 2000, 50, 251–257. [Google Scholar]
- Halliwell, B.; Gutteridge, J. Free Radicals in Biology and Medicine, 3rd ed.; Oxford University Press: Oxford, UK, 1999; p. 936. [Google Scholar]
- Kwong, L.K.; Sohal, R.S. Substrate and site specificity of hydrogen peroxide generation in mouse mitochondria. Arch. Biochem. Biophys. 1998, 350, 118–126. [Google Scholar] [CrossRef]
- Braidot, E.; Petrissa, E.; Vicunello, A.; Macri, F. Hydrogen peroxide generation by higher plant mitochondria oxidizing complex I or complex II substrates. FEBS Lett. 1999, 451, 347–350. [Google Scholar] [CrossRef]
- Lambert, J.D.; Kwon, S.J.; Hong, J.; Yang, C.S. Salivary hydrogen peroxide produced by holding or chewing green tea in the oral cavity. Free Radic. Res. 2007, 41, 850–853. [Google Scholar] [CrossRef]
- Adahi, T.; Yamada, H.; Hara, H.; Futenma, A.; Kakumu, S. Increase of urinary EC-SOD level correlated with cAMP. FEBS Lett. 1999, 458, 370–374. [Google Scholar] [CrossRef]
- Spector, A.; Ma, W.; Wang, R.R. The aqueous humor is capable of generating and degrading H2O2. Investig. Ophthalmol. Vis. Sci. 1998, 39, 1188–1197. [Google Scholar]
- Chen, Y.; Mehta, G.; Vasiliou, V. Antioxidant defences in the ocular surface. Ocul. Surf. 2009, 7, 176–185. [Google Scholar] [CrossRef]
- Wielgus, A.R.; Sarna, T. Ascorbate enhances photogeneration of hydrogen peroxide mediated by the Iris melanin. Photochem. Photobiol. 2008, 84, 683–691. [Google Scholar] [CrossRef]
- Long, L.H.; Clément, M.V.; Halliwell, B. Artifacts in Cell Culture: Rapid Generation of Hydrogen Peroxide on Addition of (−)-Epigallocatechin, (−)-Epigallocatechin Gallate, (+)-Catechin, and Quercetin to Commonly Used Cell Culture Media. Biochem. Biophys. Res. Commun. 2000, 273, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Akagawa, M.; Shigemitsu, T.; Suyama, K. Oxidative deamination of Benzylamine and Lysine residue in bovine serum albumin by green tea, black tea, and coffee. J. Agric. Food Chem. 2005, 53, 8019–8024. [Google Scholar] [CrossRef]
- Hoshino, N.; Kimura, T.; Yamaji, A.; Ando, T. Damage to the cytoplasmic membrane of Escherichia coli by catechin-copper (II) complexes. Free Radic. Biol. Med. 1999, 27, 1245–1250. [Google Scholar] [CrossRef]
- Hiramoto, K.; Kida, T.; Kikugawa, K. Increased urinary hydrogen peroxide levels caused by coŠee drinking. Biol. Pharm. Bull. 2002, 25, 1467–1471. [Google Scholar] [CrossRef] [PubMed]
- Santos-Sánchez, N.F.; Salas-Coronado, R.; Villanueva-Cañongo, C.; Hernández-Carlos, B. Antioxidant Compounds and Their Antioxidant Mechanism. In Antioxidants; Shalaby, E., Ed.; IntechOpen: London, UK, 2019; pp. 1–28. [Google Scholar]
- Davies, K.J. Oxidative stress: The paradox of aerobic life. Biochem. Soc. Symp. 1995, 61, 1–31. [Google Scholar] [CrossRef]
- Mochizuki, M.; Yamazaki, S.; Kano, K.; Ikeda, T. Kinetic analysis and mechanistic aspects of autoxidation of catechins. Biochim. Biophys. Acta 2002, 1569, 35–44. [Google Scholar] [CrossRef]
- Rinaldi, A.; Porcu, M.C.G.; Curreli, N.; Rescigno, A.; Finazziagro, A.; Pedersen, J.; Rinaldi, A.; Sanjust, E. Autoxidation of 4-Methylcatechol: A Model for the Study of the Biosynthesis of Copper Amine Oxidases Quinonoid Cofactor. Biochem. Biophys. Res. Commun. 1995, 214, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Adányi, N.; Barna, T.; Emri, T.; Miseki, M.; Pocsi, I. Hydrogen peroxide producing and decomposing enzymes: Their use in biosensors and other applications. In Industrial Enzymes; Polaina, J., MacCabe, A.P., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 441–459. ISBN 978-1-4020-5376-4. [Google Scholar]
- Baveris, A.; Cadenas, E. Mitochondrial production of hydrogen peroxide regulation by nitric oxide and the role of ubisemiquinone. IUBMB Life 2000, 50, 245–250. [Google Scholar] [CrossRef]
- Wong, H.-S.; Benoit, B.; Brand, M.D. Mitochondrial and cytosolic sources of hydrogen peroxide in resting C2C12 myoblasts. Free Radic. Biol. Med. 2019, 130, 140–150. [Google Scholar] [CrossRef]
- Treberg, J.R.; Braun, K.; Selseleh, P. Mitochondria can act as energy-sensing regulators of hydrogen peroxide availability. Redox Biol. 2019, 20, 484–488. [Google Scholar] [CrossRef]
- Druck, T.; Cheung, D.G.; Park, D.; Trapasso, F.; Pichiorri, F.; Gaspari, M.; Palumbo, T.; Aqeilan, R.I.; Gaudio, E.; Okumura, H.; et al. Fhit–Fdxr interaction in the mitochondria: Modulation of reactive oxygen species generation and apoptosis in cancer cells. Cell Death Dis. 2019, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bottje, W.G. Oxidative metabolism and efficiceny; the delicate balancing act of mitochondria. Poult. Sci. 2019, 98, 4223–4230. [Google Scholar] [CrossRef]
- Fang, J.; Wong, H.-S.; Brand, M.D. Production of superoxide and hydrogen peroxide in the mitochondrial matrix is dominated by site IQ of complex I in diverse cell lines. Redox Biol. 2020, 37, 101722. [Google Scholar] [CrossRef]
- Theng, B.K.G. Polysaccharides. In Formation and Properties of Clay-Polymer Complexes; Theng, B.K.G., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2012; Volume 4, pp. 351–390. [Google Scholar]
- Udayan, A.; Arumugam, M.; Pandey, A. Nutraceuticals from algae and cynobacteria. In Algal Green Chemistry—Recent Progress in Biotechnology; Rastogi, R.P., Madamwav, D., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 65–89. [Google Scholar]
- Li, S.; Xiong, Q.; Lai, X.; Li, X.; Wan, M.; Zhang, J.; Yan, Y.; Cao, M.; Lu, L.; Guan, J.; et al. Molecular Modification of Polysaccharides and Resulting Bioactivities. Compr. Rev. Food Sci. Food Saf. 2015, 15, 237–250. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, C.A.; Alnaief, M.; Smirnova, I. Polysaccharide-based aerogels—Promising biodegradable carriers for drug delivery systems-Review Article. Carbohydr. Polym. 2011, 86, 1425–1438. [Google Scholar] [CrossRef]
- Huang, D.; Li, Y.; Cui, F.; Chen, J.; Sun, J. Purification and characterization of a novel polysaccharide-peptide complex from Clinacanthus nutans Lindau leaves. Carbohydr. Polym. 2016, 137, 701–708. [Google Scholar] [CrossRef]
- Tian, F.; Liu, Y.; Hu, K.; Zhao, B. The depolymerisation mechanism of chitosan by hydrogen peroxide. J. Mater. Sci. 2003, 38, 4709–4712. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Wang, X.M.; Liu, C.B.; Li, J.F. The degradation, antioxidant and antimutagenic activity of the mucilage polysaccharide from Dioscorea opposite. Carbohydr. Polym. 2016, 150, 227–231. [Google Scholar] [CrossRef]
- Gao, G.; Jiao, Q.; Li, X. Structural modification of pachymaran and the mechanism interaction between Azur A. Acta Laser. Biol. Sin. 2005, 14, 228–232. [Google Scholar]
- Li, S.Q.; Shah, N.P. Antioxidant and antibacterial activities of sulphated polysaccharides from Pleurotus eryngii and Streptococcus thermophilus ASCC 1275. Food Chem. 2014, 165, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Alban, S.; Franz, G. Characterization of the anticoagulant actions of a semisynthetic curdlan sulfate. Thromb. Res. 2000, 99, 377–388. [Google Scholar] [CrossRef]
- Yalpani, M. Chemistry of polysaccharide modification and degradation. In Carbohydrates: Structures, Syntheses and Dynamics; Finch, P., Ed.; Springer: Dordrecht, The Netherlands, 1999; pp. 294–318. [Google Scholar]
- Cardoso, M.J.; Caridade, S.G.; Costa, R.R.; Mano, J.F. Enzymatic degradation of polysaccharide-based layer-by-layer structures. Biomacromolecules 2016, 17, 1347–1357. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, P.; Zhao, H.; Sun, R.; Ma, F.; Zhang, B. Effect of solution plasma process with hydrogen peroxide on the degradation and antioxidant activity of polysaccharides from Auricularia acuricula. Int. J. Biol. Macromol. 2018, 117, 1299–1304. [Google Scholar] [CrossRef]
- Ren, Y.L.; Zheng, G.Q.; You, L.J.; Wen, L.R.; Li, C.; Fu, X. Structural characterization and macrophage immunomodulatory activity of a polysaccharide isolated from Gracilaria lemaneiformis. J. Funct. Foods 2017, 33, 286–296. [Google Scholar] [CrossRef]
- Chen, X.; Zhange, R.; Li, Y.; You, L.; Kulikouskaya, V.; Hileuskaya, K. Degradation of polysaccharides from Sargassum fusiforme using UV/H2O2 and its effects on structural characteristics. Carbohydr. Polym. 2019. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Liu, H.; Yang, X.; Li, L.; Qi, B.; Hu, X.; Ma, H.; Li, C.; Pan, C. Degradation of sulphated polysaccharides from Grateloupia livida and antioxidant activity of the degraded components. Int. J. Biol. Macromol. 2020, 156, 660–668. [Google Scholar] [CrossRef]
- Yang, H.; Bai, J.; Ma, C.; Wang, L.; Li, X.; Zhang, Y.; Xu, Y.; Yang, Y. Degradation models, structure, rheological properties and protective effects on erythrocyte hemolysis of the polysaccharides from Ribes nigrum L. Int. J. Biol. Macromol. 2020, 165, 738–746. [Google Scholar] [CrossRef]
- Hou, Y.; Wang, J.; Jin, W.H.; Zhang, H.; Zhang, Q.B. Degradation of Laminaria japonica fucoidan by hydrogen peroxide and antioxidant activities of the degradation products of different molecular weights. Carbohydr. Polym. 2012, 87, 153–159. [Google Scholar] [CrossRef]
- Huang, Q.Z.; Wang, S.M.; Wang, Q.S.; Huang, J.F. Study on degradation of chitosan with UV/H2O2. J. Fujian Norm. Univ. 2004, 20, 63–67. [Google Scholar]
- Qin, C.Q.; Du, Y.M.; Liu, Y.; Xiao, L. Antibacterial activity of degraded chitosan by H2O2/HAc. J. Xiaogan Univ. 2001, 21, 9–13. [Google Scholar]
- Li, M.; Ma, F.; Li, R.; Ren, G.; Yan, D.; Zhang, H.; Zhu, X.; Wu, R.; Wu, J. Degradation of Tremella fuciformis polysaccharide by a combined ultrasound and hydrogen peroxide treatment: Process paramters, structural characteristics, and antioxidant activities. Int. J. Biol. Macromol. 2020, 160, 979–990. [Google Scholar] [CrossRef]
- Ayanda, O.S.; Nelana, S.M.; Naidoo, E.B. Ultrasonic degradation of aqueous phenolsulfonphthalein (PSP) in the presence of nano-Fe/H2O2. Ultrason. Sonochem. 2018, 47, 29–35. [Google Scholar] [CrossRef]
- Carbinatto, F.M.; De Castro, A.D.; Cury, B.S.; Magalhães, A.; Evangelista, R.C. Physical properties of pectin–high amylose starch mixtures cross-linked with sodium trimetaphosphate. Int. J. Pharm. 2012, 423, 281–288. [Google Scholar] [CrossRef]
- Tuhin, M.O.; Rahman, N.; Haque, M.E.; Khan, R.A.; Dafader, N.C.; Islam, R.; Nurnabi, M.; Tonny, W. Modification of mechanical and thermal property of chitosan-starch blend films. Radiat. Phys. Chem. 2012, 81, 1659–1668. [Google Scholar] [CrossRef]
- BeMiller, J.N. Modified Starch. In Encyclopedia of Food Science and Nutrition, 2nd ed.; Caballero, B., Trugo, L.C., Finglas, P.M., Eds.; Elsevier Academic Press: Amsterdam, The Netherlands, 2003; Volume 9, pp. 5576–5579. [Google Scholar]
- Bae, I.Y.; Kim, H.Y.; Lee, S.; Lee, H.G. Effect of the degree of oxidation on the physicochemical and biological properties of Grifola frondosa polysaccharides. Carbohydr. Polym. 2011, 83, 1298–1302. [Google Scholar] [CrossRef]
- Tian, Y.; Zeng, H.; Xu, Z.; Zheng, B.; Lin, Y.; Gan, C.; Lo, Y.M. Ultrasonic-assisted extraction and antioxidant activity of polysaccharides recovered from white button mushroom (Agaricus bisporus). Carbohydr. Polym. 2012, 2, 522–529. [Google Scholar] [CrossRef]
- Zhao, Z.; Li, J.; Wu, X.; Dai, H.; Gao, X.; Liu, M.; Tu, P. Structures and immunological activities of two pectic polysaccharides from the fruits of Ziziphus jujuba Mill. cv. jinsixiaozao Hort. Food Res. Int. 2006, 39, 917–923. [Google Scholar] [CrossRef]
- Chang, S.; Hsu, B.; Chen, B. Structural characterization of polysaccharides from Zizyphus jujuba and evaluation of antioxidant activity. Int. J. Biol. Macromol. 2010, 47, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Huang, Q.; Fu, X.; Yue, X.-J.; Liu, R.H.; You, L. Characterization, antioxidant and immunomodulatory activities of polysaccharides from Prunella vulgaris Linn. Int. J. Biol. Macromol. 2015, 75, 298–305. [Google Scholar] [CrossRef]
- Hecht, F.; Pessoa, C.F.; Gentile, L.B.; Rosenthal, D.; Carvalho, D.P.; Fortunato, R.S. The role of oxidative stress on breast cancer development and therapy. Tumor Biol. 2016, 37, 4281–4291. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Z.; Zhao, M. Carboxymethylation of polysaccharides from Tremella fuciformis for antioxidant and moisture-preserving activities. Int. J. Biol. Macromol. 2015, 72, 526–530. [Google Scholar] [CrossRef]
- Lindsay, R.C. Food additives. In Food Chemistry; Fennema, O.R., Ed.; Marcel Dekker Inc.: New York, NY, USA, 1996; pp. 778–780. [Google Scholar]
- Yan, J.K.; Li, L.; Weng, Z.M.; Leung, P.H.; Wang, W.Q.; Wu, J.Y. Acidic degradation and enhanced antioxidant activities of exopolysaccharides from Cordyceps sinensis mycelial culture. Food Chem. 2009, 117, 641–646. [Google Scholar] [CrossRef]
- You, Q.H.; Yin, X.L.; Zhang, S.N.; Jiang, Z.H. Extraction, purification, and antioxidant activities of polysaccharides from Tricholoma mongolicum Imai. Carbohydr. Polym. 2014, 99, 1–10. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Chen, H.; Ju, Y.; Li, J.; Yu, M. Antioxidant activities of polysaccharides from Lentinus edodes and their significance for disease prevention. Int. J. Biol. Macromol. 2012, 50, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Chun-hui, L.; Chang-hai, W.; Xu, Z.-L.; Wang, Y. Isolation, chemical characterization and antioxidant activities of two polysaccharides from the gel and the skin of Aloe barbadensis Miller irrigated with sea water. Process Biochem. 2007, 42, 961–970. [Google Scholar] [CrossRef]
- Li, X.; Zhou, A.; Han, Y. Anti-oxidation and anti-microorganism activities of purification polysaccharide from Lygodium japonicum in vitro. Carbohydr. Polym. 2006, 66, 34–42. [Google Scholar] [CrossRef]
- He, X.; Wang, X.; Fang, J.; Chang, Y.; Ning, N.; Guo, H.; Huang, L.; Huang, X.; Zhao, Z. Polysaccharides in Grifola frondosa mushroom and their health promoting properties: A review. Int. J. Biol. Macromol. 2017, 101, 910–921. [Google Scholar] [CrossRef] [PubMed]
- Subhapradha, N.; Ramasamy, P.; Seedevi, P.; Shanmugam, V.; Srinivasan, A.; Shamugam, A. Extraction, characterization and its antioxidant efficacy of polysaccharides from Sepia aculeate (Orbigny, 1848) cuttlebone. Afr. J. Biotechnol. 2013, 13, 138–144. [Google Scholar]
- Gulcin, I. Antioxidant and antiradical activities of L-carnitine. Life Sci. 2006, 78, 803–811. [Google Scholar] [CrossRef]
- Fan, J.; Wu, Z.; Zhao, T.; Sun, Y.; Ye, H.; Xu, R.; Zeng, X. Characterization, antioxidant and hepatoprotective activities of polysaccharides from Ilex latifolia Thunb. Carbohydr. Polym. 2014, 101, 990–997. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, Q.; Zhang, Z.; Li, Z. Antioxidant activity of sulfated polysaccharide fractions extracted from Laminaria japonica. Int. J. Biol. Macromol. 2008, 42, 127–132. [Google Scholar] [CrossRef]
- Ghosh, N.; Chakraborty, T.; Mallick, S.; Mana, S.; Singha, D.; Ghosh, B.; Roy, S. Synthesis, characterization and study of antioxidant activity of quercetin–magnesium complex. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 151, 807–813. [Google Scholar] [CrossRef]
- Joly, N.; Ghemati, D.; Aliouche, D.; Martin, P. Interaction of metal ions with mono- and polysaccharides for wastewater treatment: A Review. Nat. Prod. Chem. Res. 2020, 8, 1–17. [Google Scholar]
- Li, B.; Liu, S.; Xing, R.; Li, K.; Li, R.; Qin, Y.; Wang, X.; Wei, Z.; Li, P. Degradation of sulfated polysaccharides from Enteromorpha prolifera and their antioxidant activities. Carbohydr. Polym. 2013, 92, 1991–1996. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, S.E.; Fry, S.C. Redox and wall-restructuring. In The Expanding Cell; Verbelen, J.-P., Vissenberg, K., Eds.; Springer: Berlin, Germany, 2007; pp. 159–190. [Google Scholar]
- Vreeburg, R.A.M.; Aririanah, O.B.; Fry, S.C. Fingerprinting of hydroxyl radical-attacked polysaccharides by N-isopropyl-2-aminoacridone labelling. Biochem. J. 2014, 463, 225–237. [Google Scholar] [CrossRef]
- Qiao, D.; Ke, C.; Hu, B.; Luo, J.; Ye, H.; Sun, Y.; Yan, X.; Zeng, X. Antioxidant activities of polysaccharides from Hyriopsis cumingii. Carbohydr. Polym. 2010, 78, 199–204. [Google Scholar] [CrossRef]
- Qin, C.; Huang, K.; Xu, H. Isolation and characterisation of a novel polysaccharide from the mucus of the loach, Misgurnus anguillicaudatus. Carbohydr. Polym. 2002, 49, 367–371. [Google Scholar] [CrossRef]
- Daud, N.S.; Din, A.R.J.M.; Rosli, M.A.; Azam, Z.M.; Othman, N.Z.; Sarmidi, M.R. Paenibacillus polymyxa bioactive compounds for agricultural and biotechnological applications. Biocatal. Agric. Biotechnol. 2019, 18, 101092. [Google Scholar] [CrossRef]
- Guo, S.; Mao, W.; Han, Y.; Zhang, X.; Yang, C.; Chen, Y.; Chen, Y.; Xu, J.; Li, H.; Qi, X.; et al. Structural characteristics and antioxidant activities of the extracellular polysaccharides produced by marine bacterium Edwardsiella tarda. Bioresour. Technol. 2010, 101, 4729–4732. [Google Scholar] [CrossRef]
- He, F.; Yang, Y.; Yang, G.; Yu, L. Components and antioxidant activity of the polysaccharide from Streptomyces virginia H03. Zeitschrift Naturforschung C 2008, 63, 181–188. [Google Scholar] [CrossRef][Green Version]
- Qiao, D.; Luo, J.; Ke, C.; Sun, Y.; Ye, H.; Zeng, X. Immunostimulatory activity of the polysaccharides from Hyriopsis cumingii. Int. J. Biol. Macromol. 2010, 47, 676–680. [Google Scholar] [CrossRef] [PubMed]
- Katzenellenbogen, E.; Kocharova, N.A.; Shashkov, A.S.; Górska-Frączek, S.; Bogulska, M.; Gamian, A.; Knirel, Y.A. Structure of the O-polysaccharide of Edwardsiella tarda PCM 1150 containing an amide of d-glucuronic acid with l-alanine. Carbohydr. Res. 2013, 368, 84–88. [Google Scholar] [CrossRef]
- Katzenellenbogen, E.; Kocharova, N.A.; Shashkov, A.S.; Górska-Frączek, S.; Gamian, A.; Knirel, Y.A. Structure of the O-polysaccharide of Edwardsiella tarda PCM 1156. Carbohydr. Res. 2013, 374, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Kodali, V.P.; Das, S.; Sen, R. An exopolysaccharide from a probiotic: Biosynthesis dyanmics, composition and emulsifying activity. Food Res. Int. 2009, 42, 695–699. [Google Scholar] [CrossRef]
- Ramanathan, T.; Ahmad, A.; Ahmad, A.S.; Kalimutho, M. Taxonomical identity and polysaccharide produced by Bacillus species isolated from old aged medicinal decoctions. J. Sustain. Sci. Manag. 2011, 6, 2–9. [Google Scholar]
- Raza, W.; Makeen, K.; Wang, Y.; Xu, Y.; Qirong, S. Optimization, purification, characterization and antioxidant activity of an extracellular polysaccharide produced by Paenibacillus polymyxa SQR-21. Bioresour. Technol. 2011, 102, 6095–6103. [Google Scholar] [CrossRef]
- Mian, J.A.; Percival, E. Carbohydrates of the brown seaweeds Himanthalia lorea, Bifurcaria bifurcata, and Padina pavonia: Part 1. Extraction and fractionation. Carbohydr. Res. 1973, 26, 133–146. [Google Scholar] [CrossRef]
- Rodríguez-Jasso, R.M.; Mussatto, S.I.; Pastrana, L.; Aguilar, C.N.; Teixeira, J.A. Chemical composition and antioxidant activity of sulphated polysaccharides extracted from Fucus vesiculosus using different hydrothermal processes. Chem. Pap. 2014, 68, 203–209. [Google Scholar] [CrossRef]
- Rupérez, P.; Ahrazem, O.; Leal, J.A. Potential Antioxidant Capacity of Sulfated Polysaccharides from the Edible Marine Brown SeaweedFucus vesiculosus. J. Agric. Food Chem. 2002, 50, 840–845. [Google Scholar] [CrossRef]
- Saha, S.; Navid, M.H.; Bandyopadhyay, S.S.; Schnitzler, P.; Ray, B. Sulfated polysaccharides from Laminaria angustata: Structural features and in vitro antiviral activities. Carbohydr. Polym. 2012, 87, 123–130. [Google Scholar] [CrossRef]
- Soukaina, B.; Zainab, E.A.-T.; Guillaume, P.; Halima, R.; Philippe, M.; Cherkaoui, E.M.; Cedric, D. Radical depolymerisation of Alginate extracted from Moroccan Brown Seaweed Bifiucaria bifurcate. Appl. Sci. 2020, 10, 4166. [Google Scholar] [CrossRef]
- Teruya, T.; Takeda, S.; Tamaki, Y.; Tako, M. Fucoidan Isolated from Laminaria angustata var. longissima Induced Macrophage Activation. Biosci. Biotechnol. Biochem. 2010, 74, 1960–1962. [Google Scholar] [CrossRef] [PubMed]
- Vasantharaja, R.; Abraham, L.S.; Gopinath, V.; Hariharan, D.; Smita, K.M. Attenuation of oxidative stress induced mitochondrial dysfunction and cytoxicity in fibroblast cells by sulphated polysaccharide from Padina gymnospora. Int. J. Biol. Macromol. 2019, 124, 50–59. [Google Scholar] [CrossRef]
- Wijesekara, I.; Pangestuti, R.; Kim, S.-K. Biological activities and potential health benefits of sulphated polysaccharides derived from marine algae. Carbohydr. Polym. 2011, 84, 14–21. [Google Scholar] [CrossRef]
- Xu, C.H.; Fang, Y.; Lin, H.; Chen, L.; Li, Z.J.; Deng, D.; Lu, C.X. Chemical characters and anti oxidative properties of sulphated polysaccharides from Lamina japonica. J. Appl. Phycol. 2001, 13, 67–70. [Google Scholar] [CrossRef]
- Chen, T.; Li, B.; Li, Y.; Zhao, C.; Shen, J.; Zhang, H. Catalytic synthesis and antitumor activities of sulfated polysaccharide from Gynostemma pentaphyllum Makino. Carbohydr. Polym. 2011, 83, 554–560. [Google Scholar] [CrossRef]
- Chen, L.; Huang, G. Extraction, characterization and antioxidant activities of pumpkin polysaccharide. Int. J. Biol. Macromol. 2018, 118, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Chen, L.-X.; Wu, D.-T.; Cheong, K.-L.; Chen, N.-F.; Zhao, J.; Li, S.-P. Qualitative and quantitative analysis of specific polysaccharides in Dendrobium huoshanense by using saccharide mapping and chromatographic methods. J. Pharm. Biomed. Anal. 2016, 129, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Zhang, Q.; Li, Y.; Li, L.; Lan, W.; He, J.; Li, H.; Xiong, Y.; Qin, W. Extraction, characterisation and antioxidant activities of polysaccharides of Chuanminshen violaceum. Int. J. Biol. Macromol. 2016, 86, 224–232. [Google Scholar] [CrossRef]
- Jiang, J.; Meng, F.Y.; He, Z.; Ning, Y.L.; Li, X.H.; Song, H.; Wang, J.; Zhou, R. Sulfated modification of longan polysaccharide and its immunomodulatory and antitumor activity in vitro. Int. J. Biol. Macromol. 2014, 67, 323–329. [Google Scholar] [CrossRef]
- Jing, Y.; Huang, L.; Lv, W.; Tong, H.; Song, L.; Hu, X.; Yu, R. Structural Characterization of a Novel Polysaccharide from Pulp Tissues of Litchi chinensis and Its Immunomodulatory Activity. J. Agric. Food Chem. 2014, 62, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Khaskheli, S.G.; Zheng, W.; Sheikh, S.A.; Khaskheli, A.A.; Liu, Y.; Soomro, A.H.; Feng, X.; Sauer, M.B.; Wang, Y.F.; Huang, W. Characterisation of Auricularia auricula polysaccharides and its antioxidant properties in fresh and pickled product. Int. J. Biol. Macromol. 2015, 81, 387–395. [Google Scholar] [CrossRef]
- Li, H.; Su, J.; Jiang, J.; Li, Y.; Gan, Z.; Ding, Y.; Li, Y.; Liu, J.; Wang, S.; Ke, Y. Characterization of polysaccharide from Scutellaria barbata and its antagonistic effect on the migration and invasion of HT-29 colorectal cancer cells induced by TGF-β1. Int. J. Biol. Macromol. 2019, 131, 886–895. [Google Scholar] [CrossRef]
- Liu, J.; Zhao, Z.Z.; Chen, H.-B. Review of Astragali radix. Chin. Herb. Med. 2011, 3, 90–105. [Google Scholar]
- Liu, J.; Meng, C.-G.; Yan, Y.-H.; Shan, Y.-N.; Kan, J.; Jin, C.-H. Structure, physical property and antioxidant activity of catechin grafted Tremella fuciformis polysaccharide. Int. J. Biol. Macromol. 2016, 82, 719–724. [Google Scholar] [CrossRef]
- Lu, X.; Mo, X.; Guo, H.; Zhang, Y. Sulfation modification and anticoagulant activity of the polysaccharides obtained from persimmon (Diospyros kaki L.) fruits. Int. J. Biol. Macromol. 2012, 51, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Luo, Q.; Cai, Y.; Yan, J.; Sun, M.; Corke, H. Hypoglycemic and hypolipidemic effects and antioxidant activity of fruit extracts from Lycium barbarum. Life Sci. 2004, 76, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Mckenna, D.J.; Hughes, K.; Jones, K. Astragalus. Altern. Ther. Health Med. 2002, 8, 34–40. [Google Scholar]
- Nguyen, T.L.; Chen, J.; Hu, Y.; Wang, D.; Fan, Y.; Wang, J.; Abula, S.; Zhang, J.; Qin, T.; Chen, X. In vitro antiviral activity of sulfated Auricularia auricular polysaccharides. Carbohydr. Polym. 2012, 90, 1254–1258. [Google Scholar] [CrossRef]
- Qian, X.P.; Zha, X.Q.; Xiao, J.J.; Zhang, H.L.; Pan, L.H.; Luo, J.P. Sulfated modification can enhance antiglycation abilities of polysaccharides from Dendrobium huoshanense. Carbohydr. Polym. 2014, 101, 982–989. [Google Scholar] [CrossRef]
- Song, X.; Yin, Z.; Zhao, X.; Cheng, A.; Jia, R.; Yuan, G.; Xu, J.; Fan, Q.; Dai, S.; Lu, H. Antiviral activity of sulfated Chuanmingshen violaceum polysaccharide against Newcastle disease virus. J. Gen. Virol. 2013, 94, 2164–2174. [Google Scholar] [CrossRef]
- Song, X.; Xu, J.; Yin, Z.; Jia, R.; Cheng, A.; Deng, Y.; Lu, C.; Liang, X.; Wang, Y.; Yao, X.; et al. Effects of Polysaccharide from Chuanminshen violaceum on Immune Response of Newcastle Disease Vaccine in Chicken. Acta Sci. Vet. 2013, 41, 1104. [Google Scholar]
- Song, Y.; Yang, Y.; Zhang, Y.; Duan, L.; Zhou, C.; Ni, Y.; Liao, X.; Li, Q.; Hu, X. Effect of acetylation on antioxidant and cytoprotective activity of polysaccharides isolated from pumpkin (Cucurbita pepo, lady godiva). Carbohydr. Polym. 2013, 98, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Jiang, H.; Sun, C.; Adu-Frimpong, M.; Deng, W.; Yu, J.; Xu, X. Antioxidant and hepatoprotective effects of purified Rhodiola rosea polysaccharides. Int. J. Biol. Macromol. 2018, 117, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.-K.; Wang, Y.-Y.; Ma, H.-L.; Wang, Z.-B. Ultrasonic effects on the degradation kinetics, preliminary characterization and antioxidant activities of polysaccharides from Phellinus linteus mycelia. Ultrason. Sonochem. 2015, 29, 251–257. [Google Scholar] [CrossRef]
- Yang, C.; He, N.; Ling, X.; Ye, M.; Zhang, C.; Shah, W.; Yao, C.; Wang, Z.; Li, Q. The isolation and characterisation of polysaccharides from longan pulp. Sep. Purif. Technol. 2008, 63, 226–230. [Google Scholar] [CrossRef]
- Yin, J.Y.; Chan, B.C.L.; Yu, H.; Lau, I.Y.K.; Han, X.Q.; Cheng, S.W. Separation, structure characterization, conformation and immunomodulating effect of a hyperbranched heteroglycan from Radix astragali. Carbohydr. Polym. 2012, 87, 667–675. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, L.; Ding, Q.; Chen, X. Studies on molecular weights of polysaccharides of Auricularia auricula-judae. Carbohydr. Res. 1995, 270, 1–10. [Google Scholar] [CrossRef]
- Zhang, Y.; Lu, X.; Fu, Z.; Wang, Z.; Zhang, J. Sulphated modification of a polysaccharide obtained from fresh persimmon (Diospyros kaki L.) fruit and antioxidant activities of the sulphated derivatives. Food Chem. 2011, 127, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Ayimbila, F.; Keawsompong, S. Functional composition and antioxidant property of crude polysaccharides from the fruiting bodies of Lentinus squarrosulus. 3 Biotech. 2021, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Wu, W.H.; Xu, X.J.; Zhang, L.N.; Liu, Y.; Wang, K.P. Chain conformation and anti-tumor activity of derivatives of polysaccharide from Rhizoma Panacis Japonici. Carbohydr. Polym. 2014, 105, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.Y.; Yi, Y.; Zhang, L.F.; Zhang, R.F.; Zhang, Y.; Wei, Z.C.; Tang, X.J.; Zhang, M.W. Immunomodulatory activity and partial characterization of polysaccharides from Momordica charantia. Molecules 2014, 19, 13432–13447. [Google Scholar] [CrossRef]
- Deng, C.; Fu, H.; Xu, J.; Shang, J.; Cheng, Y. Physiochemical and biological properties of phosphorylated polysaccharides from Dictyophora indusiata. Int. J. Biol. Macromol. 2015, 72, 894–899. [Google Scholar] [CrossRef]
- Jung, H.Y.; Bae, I.Y.; Lee, S.; Lee, H.G. Effect of the degree of sulfation on the physicochemical and biological properties of Pleurotus eryngii polysaccharides. Food Hydrocoll. 2011, 25, 1291–1295. [Google Scholar] [CrossRef]
- Juyi, G.; Jiantin, Z.; Yihong, H. Isolation, Purification and Characterization of Polysaccharide from the Fruit Bodies of Pleurotus cornucopiae (Paul:Pers.) Roll. 1999. Available online: https://en.cnki.com.cn/Article_en/CJFDTotal-FJSZ902.018.htm (accessed on 19 January 2021).
- Ren, D.; Jiao, Y.; Yang, X.; Yuan, L.; Guo, J.; Zhao, Y. Antioxidant and antitumor effects of polysaccharides from the fungus Pleurotus abalonus. Chem. Interact. 2015, 237, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Rout, D.; Mondal, S.; Chakraborty, I.; Islam, S.S. The structure of a polysaccharide from Fraction-II of an edible mushroom, Pleurotus florida. Carbohydr. Res. 2006, 341, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Villares, A. Polysaccharides from the edible mushroom Calocybe gambosa: Structure and chain conformation of a (1→4),(1→6)-linked glucan. Carbohydr. Res. 2013, 375, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Hao, L.; Jia, S.; Wang, Q.; Zhang, X.; Niu, S. A Review of Research on Polysaccharide from Coriolus versicolor. In Proceedings of the 2012 International Conference on Applied Biotechnology (ICAB 2012); Lecture Notes in Electrical Engineering; Zhang, T.C., Ouyang, P., Kaplan, S., Skarnes, B., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; Volume 249. [Google Scholar]
- Wu, Y.; Ye, M.; Du, Z.; Jing, L.; Surahio, M.; Yang, L. Carboxymethylation of an exopolysaccharide from Lachnum and effect of its derivatives on experimental chronic renal failure. Carbohydr. Polym. 2014, 114, 190–195. [Google Scholar] [CrossRef]
- Xin, X.; Zheng, K.; Niu, Y.; Song, M.; Kang, W. Effect of Flammulina velutipes (golden needle mushroom, eno-kitake) polysaccharides on constipation. Open Chem. 2018, 16, 155–162. [Google Scholar] [CrossRef]
- Xu, W.; Zhang, F.; Luo, Y.; Ma, L.; Kou, X.; Huang, K. Antioxidant activity of a water-soluble polysaccharide purified from Pteridium aquilinum. Carbohydr. Res. 2009, 344, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Yan, H.; Tang, J.; Chen, J.; Zhang, X. Polysaccharides inLentinus edodes: Isolation, Structure, Immunomodulating Activity and Future Prospective. Crit. Rev. Food Sci. Nutr. 2014, 54, 474–487. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Chen, W.-X.; Qiu, T.; Yuan, R.-Y.; Ye, Y.-W.; Cai, J.-M. Structural characterisation and anti-ageing activity of extracellular polysaccharide from a strain of Lachnum sp. Food Chem. 2012, 132, 338–343. [Google Scholar] [CrossRef]
- Zhang, H.; Li, W.-J.; Nie, S.-P.; Chen, Y.; Wang, Y.-X.; Xie, M.-Y. Structural characterisation of a novel bioactive polysaccharide from Ganoderma atrum. Carbohydr. Polym. 2012, 88, 1047–1054. [Google Scholar] [CrossRef]
- Zhao, L.; Dong, Y.; Chen, G.; Hu, Q. Extraction, purification, characterization and antitumor activity of polysaccharides from Ganoderma lucidum. Carbohydr. Polym. 2010, 80, 783–789. [Google Scholar] [CrossRef]
- Zhu, Z.Y.; Pang, W.; Li, Y.Y.; Ge, X.R.; Chen, L.J.; Liu, X.C.; Lv, Q.; Dong, G.L.; Liu, A.J.; Zhang, Y. Effect of ultrasonic treatment on structure and antitumor activity of mycelial polysaccharides from Cordyceps gunnii. Carbohydr. Polym. 2014, 114, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Guo, Q.; Zhang, K.; Wang, N.; Li, C.; Li, Z.; Zhang, A.; Wang, C. Polysaccharide from Pleurotus nebrodensis: Physicochemical, structural characterization and in vitro fermentation characteristics. Int. J. Biol. Macromol. 2020, 165, 1960–1969. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.B.; Pi, Z.F.; Fu, M.; Li, L.; Hou, J.; Shi, L.S.; Chen, R.R.; Jiang, Y.; Liu, F.; Yue, H.; et al. A polysaccharopeptide complex and a condensed tannin with antioxidant activity from dried rose (Rosa rugosa) flowers. J. Pharm. Pharmacol. 2006, 58, 529–534. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, M.; Xie, B. Components and antioxidant activity of polysaccharide conjugate from green tea. Food Chem. 2005, 90, 17–21. [Google Scholar] [CrossRef]
- Tannin-Spitz, T.; Bergman, M.; Van-Moppes, D.; Grossman, S.; Arad, S. (Malis) Antioxidant activity of the polysaccharide of the red microalga Porphyridium sp. Environ. Boil. Fishes 2005, 17, 215–222. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, X.; Zhao, M.; Yu, S.; Qi, H. The immunological and antioxidant activities of polysaccharides extracted from Enteromorpha linza. Int. J. Biol. Macromol. 2013, 57, 45–49. [Google Scholar] [CrossRef]
- Kodali, V.P.; Sen, R. Antioxidant and free radical scavenging activities of an exopolysaccharide from a probiotic bacterium. Biotechnol. J. 2008, 3, 245–251. [Google Scholar] [CrossRef]
- Okafor, D.C.; Onuegbu, N.C.; Odimegwu, N.E.; Ibeabuchi, J.C.; Njoku, N.E.; Agunwa, M.; Ofoedu, C.E.; Njoku, C.C. Antioxidant and Antimicrobial Activities of Oyster Mushroom. Am. J. Food Sci. Technol. 2017, 5, 64–69. [Google Scholar] [CrossRef]
- Zhu, Z.-Y.; Liu, R.-Q.; Si, C.-L.; Zhou, F.; Wang, Y.-X.; Ding, L.-N.; Jing, C.; Liu, A.-J.; Zhang, Y.-M. Structural analysis and anti-tumor activity comparison of polysaccharides from Astragalus. Carbohydr. Polym. 2011, 854, 895–902. [Google Scholar] [CrossRef]
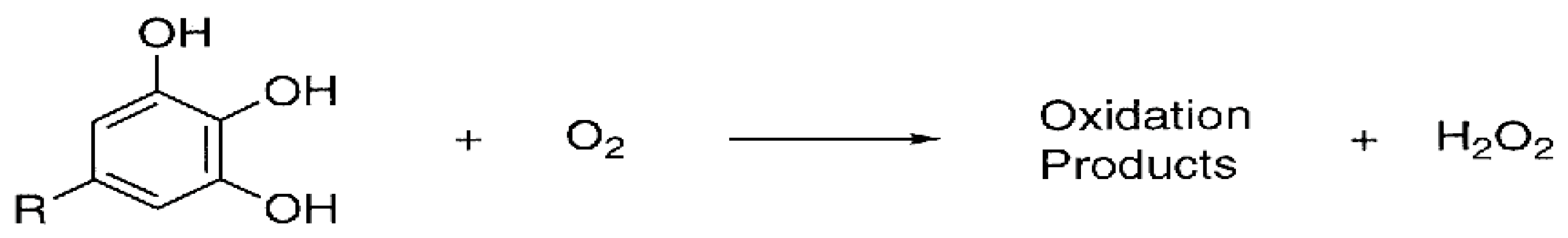
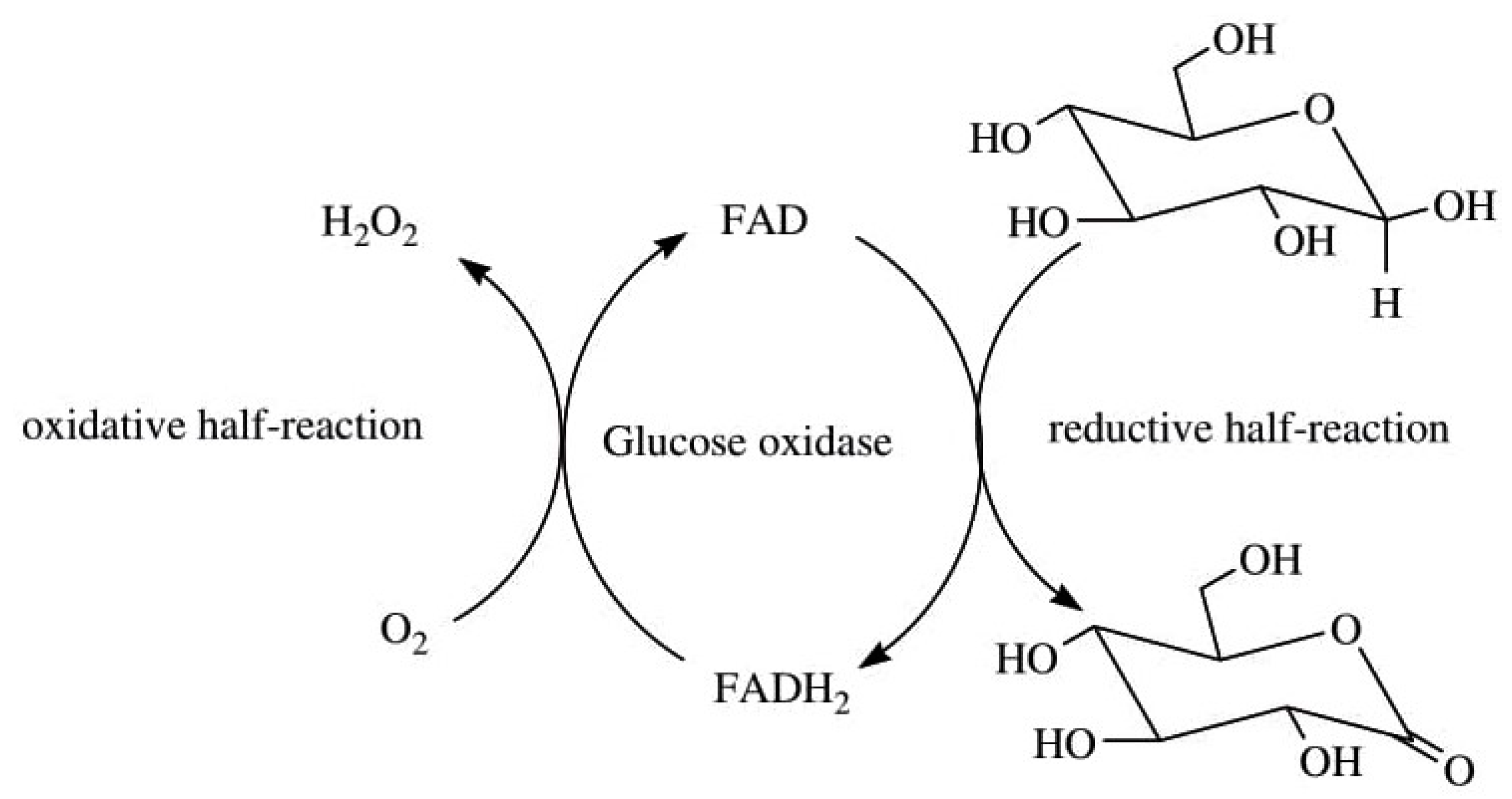
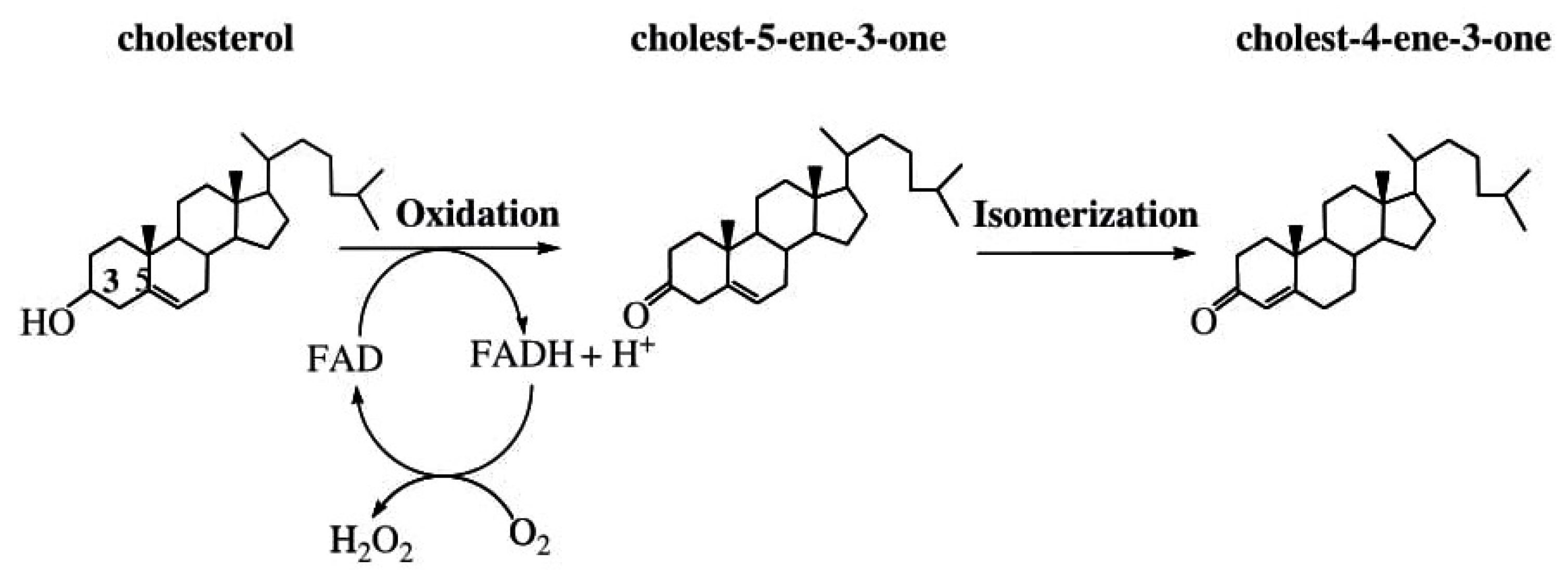

| Location | In Vitro | Reference(s) | In Vivo | Reference(s) |
|---|---|---|---|---|
| Source/site | Beverages (e.g., green tea, black tea, wine, beer, coffee) | Grzesik et al. [63] | Mitochondria | Lennicke et al. [15]; Baveris and Cadenas [86]; Wong et al. [87]; Treberg et al. [88] |
| Trigger agent | Certain phenolic compounds (e.g., tea catechins, (+)-catechin, and gallic acid) | Wee et al. [67]; Grzesik et al. [63] | Escaped electron (e−) in the electron transport chain | Phaniendra et al. [7]; Druck et al. [89]; Maddu [2] |
| Production process | Auto-oxidation | Erickson [26]; Kim et al. [65]; Bensid et al. [27] | Cellular oxidative metabolism | Patel et al. [6]; Bottje [90]; Fang et al. [91] |
| Factors responsible for H2O2 formation | Content and composition of phenolic compounds; Structure of phenolic compounds; Oxygen; Metal ions; Temperature; pH; Incubation time. | Akagawa et al. [62]; Grzesik et al. [63]; Bopitiya et al. [59] | The action of certain enzymes such as superoxide dismutase (SOD), xanthine oxidase, monoamine oxidase, and glycollate oxidase; Type of ingested beverage such as green tea, black tea, and/or coffee; Available oxygen; The joint action of in vivo dismutation of superoxide radical and in vitro auto-oxidation of phenolic compounds | Fang et al. [91]; Grzesik et al. [63]; Bopitiya et al. [59] |
| Antioxidative Polysaccharide | Sources | Polysaccharides Specifics | References |
|---|---|---|---|
| Astragali radix | Plant | From Astragali radix, isolated two glacans (AG-1, identified as as α-(1,4) and α-(1,6) glucan (5:2); and AG-2 identified as α-(1,4) glucan) and two heterosaccharides (AH-1, identified as acidic polysaccharide, composed of hexouronic acid, glucose, rhamnose, and arabinose in the ratio of 1:0.04:0.02:0.0; and AH-2, identified as glucose and arabinose in the ratio of 1:0.15) from an Astragali radix polysaccharides | Liu et al. [169]; Mckenna et al. [173] |
| Astragalus mongholicus | Plant | Polysaccharides from A. mongholicus, named as APS-I and APS-II, comprised backbone (1,3)-β-d-glucopyranosyl (Glcp) residues, with bioactivities closely related to their chemical composition, configuration, molecular weight, and physical properties | Yin et al. [182]; Zhu et al. [210] |
| Litchi chinensis Sonn. | Plant | A novel polysaccharide (LCP50W) from pulp tissues of Litchi chinensis had main chain consist of (1→3)-linked β-l-rhamnopyranosyl, (1→6)-linked α-d-glucopyranosyl, and (1→2,6)-linked α-d-glucopyranosyl residues, which branched at O-6. The three branches consisted of (1→2)-linked α-l-rhamnopyranosyl, (1→3)-linked α-d-galactopyranosyl, and (1→3)-linked α-l-mannopyranosyl residues, terminated with (1→)-linked α-l-arabinopyranosyl residues, respectively | Jing et al. [166]; Yin et al. [182] |
| Lycium barbarum | Plant | Raw and Purified Lycium barbarum polysaccharides (LBP) would contain sugars such as Rha, Gal, Glc, Ara, Man, Xyl, with molar ratio of 4.22, 2.43, 1.38, 1, 0.95, and 0.38, respectively | Luo et al. [172]; Yin et al. [182] |
| Scutellaria barbata D. Don | Plant | From the whole plant of Scutellaria barbata D. Don, polysaccharide, SBPW3, showed a molecular weight of 10.2 kDa and composed of arabinose (25.68%), galactose (27.72%), glucose (20.59%), mannose (12.56%), rhamnose (2.51%), and xylose (10.94%) | Li et al. [168]; Yin et al. [182] |
| Ribes nigrum L. | Plant | Polysaccharides from blackcurrant (Ribes nigrum L.) (BCP), showed two low-molecular-weight polysaccharides (DBCP-1, DBCP-2) with same monosaccharide units with resembling glycosidic linkage patterns | Xu et al. [179]; Yin et al. [182] |
| Tremella fuciformis | Plant | Tremella fuciformis polysaccharide (catechin-g-TPS) with molecular weight of 5.82 × 105 Da, given by polysaccharide fractions (F-1 and F-2) | Liu et al. [170]; Wang et al. [123]; Zhang et al. [34] |
| Auricularia auricula | Plant | - Fruit bodies of Auricularia auricula were comprised of Glucans A, C, and E, mainly backbone chain of Beta (1-3)-d-glucose residues, with various branched groups. Specifically, polysaccharides D and B would have residues of d-xylose, d-mannose, d-galactose, d-glucose, and d-glucuronic acid. - Two polysaccharide fractions (AAPF, AAPP) purified from the fruiting body of A. auricula, showed AAPF with five monosaccharides, including glucose, rhamnose, arabinose, mannose, and galactose with a molar ratio of 16.74:1.0:1.18:1.0:1.0; and AAPP with four monosaccharides, namely arabinose, mannose, galactose, and xylose with the molar ratio of 15.59:1.52:4.76:1.0 | Zhang et al. [183]; Khaskheli et al. [167]; Nguyen et al. [174]; Zhang et al. [34] |
| Chuanminshen violaceum | Plant | - Chuanminshen violaceum polysaccharides (CVPS) comprise d-carubinose and d-glucose, in the ratio of 1: 16.2. The average molecular and number-average molecular weight CVPS of 9.7632 × 105 Da and 5.2270 × 104 Da respectively. - Another study showed Chuanminshen violaceum polysaccharides (CVPs) with neutral polysaccharides, mainly composed of glucose and galactose, with molecular weights ranging between 233.69 and 11.02 kDa. | Dong et al. [164]; Song et al. [176,178] |
| Dendrobium huishanense | Plant | With molecular weights range 1.16 × 105 to 2.17 × 105 Da, with main monosaccharide compositions that include Man and Glc, with glycosidic linkages β-1,4-Manp and β-1,4-Glcp, and substituted with acetyl groups at O-2 and O-3 of 1,4-linked Manp | Deng et al. [163]; Qian et al. [175] |
| Persimmon | Plant | Sulphated modification, polysaccharide was obtained from fresh (Diospyros kaki L.) persimmon fruit (PFP), Three sulphated derivatives of PFP, known as PFP-SI, PFP-SII and PFP-SIII, were obtained, with average molecular weights of 53, 51 and 48 kDa, respectively, | Lu et al. [171]; Zhang et al. [184] |
| Longan | Plant | Longan polysaccharides (LPSs), extracted from (logan) pulp, showed β-type acidic heteropolysaccharides with pyran group, wherein LPS-N had glucose/xylose molar ratio of 1.9:1, with LPS-A1 having rhamnose, xylose, arabinose and galactose molar ratio of 1:1.64:4.33:2.28, and LPS-A2 having only rhamnose | Jiang et al. [165]; Yang et al. [181] |
| Azadirachta indica leaves | Plant | A native polysaccharide of Azadirachta indica (leaves) obtained an apparent molecular mass of 80 kDa, constituting (1→5)-/(1→3,5)-linked α-l-arabinosyl, (1→3)-/(1→6)-/(1→3,6)-linked β-d-galactosyl, and terminal-rhamnosyl and α-l-arabinosyl residues | Saha et al. [42] |
| Portulaca oleracea L. | Plant | A yield of 6.45% of dried raw material, P. oleracea showed water-soluble crude polysaccharides (DCPOP), 92% carbohydrate, free of proteins and with average molecular weight of 24.6 kDa | Chen et al. [161]; Shen et al. [42] |
| Pumpkin | Plant | Potentially containing acetyl groups, the pumpkin polysaccharide showed α/β-glycosidic bond linkage. It is considered a heteropolysaccharide, composed of six monosaccharides, namely arabinose, galactose, glucose, glucuronic acid, rhamnose, and xylose | Chen and Huang [162]; Song et al. [177] |
| Ganoderma lucidum | Fungi | Polysaccharides of Ganoderma lucidum consisted primarily of glucose and galactose, small amounts of mannose, rhamnose and fucose, and minor amount of glucuronic acid | Wang et al. [25]; Xu et al. [197]; Zhao et al. [201] |
| Athyrium multidentatum | Fungi | Five polysaccharide fractions (PS-1, PS-2, PS-3, PS-4, and PS-5) isolated from Athyrium multidentatum (Doll.) Ching, was heteropolysaccharides with different molecular weights and monosaccharide compositions | Jing et al. [38]; Wang et al. [25] |
| Flammulina spp | Fungi | Flammulina polysaccharide comprise glucan, which could have a mix of some other fractions, like fucosan, galactose, glycan, mannan, and xylan | Wang et al. [25]; Xin et al. [196] |
| Coriolus versicolor | Fungi | Polysaccharide of Coriolus versicolor contains extracellular polysaccharide (EPS) mainly β-1, 3/β-1, 6-linked d-glucose molecules, predominantly comprising glucose and small amounts of arabinose, galactose, mannose, and xylose | Wang et al. [194]; Wang et al. [25] |
| Pleurotus nebrodensis | Fungi | Polysaccharide (PNPS) from the fruiting body of Pleurotus nebrodensis contained mainly glucose, with a small percentage of galactose and mannose | Gao et al. [203]; Wang et al. [25] |
| Pleurotus abalunes | Fungi | Polysaccharides of Pleurotus abalonus is heteropolysaccharide, comprise d-mannose, d-ribose, l-rhamnose, d-glucuronic acid, d-glucose and d-galactose, having corresponding mole percentages 3.4%, 1.1%, 1.9%, 1.4%, 87.9% and 4.4%, respectively | Ren et al. [191]; Wang et al. [25] |
| Pleurotus eryngii | Fungi | Polysaccharides from P. eryngii comprise mainly of β-(1→3)-glucans with β-(1→6) branches | Jung et al. [189]; Wang et al. [25] |
| Pleurotus corncopiae | Fungi | Polysaccharides isolated from Pleurotus cornucopiae composed of arabinose, glucose, mannose and xylose | Juyi et al. [190]; Wang et al. [25] |
| Pleurotus florida | Fungi | P. florida consists of three different polysaccharides, which include (1→3)-, (1→6)-branched glucan, and (1→6)-α-glucan | Rout et al. [192]; Wang et al. [25] |
| Calocybe gambosa | Fungi | -Three polysaccharides were isolated from the fruiting bodies Calocybe gambosa mushroom, composed of glucose, and methylation analysis, showed the units were (1→4),(1→6)-linked with a degree of branching (DB) of 4% | Villares [193]; Wang et al. [25] |
| Lentinus edodes | Fungi | L. edodes can be divided into three layers: (1) the outside layer has heteropolysaccharide and β-(1→3)-glucan with β-(1→6) branches; (2) middle layer has mainly β-(1→6)-glucan with a small number of β-(1→3) branches; (3) inner layer being complex chitin, β-glucan, with small amount of acid polymer | Xu et al. [198]; Wang et al. [25] |
| Lentinus polyschrous Lev. | Fungi | The major monosaccharides of Lentinus polychrous include d-galactose, d-glucose, and d-mannose at different proportions, fucose, and xylose | Ayimbila and Keawsompong [185]; Wang et al. [25] |
| Pleurotus ostreatus | Fungi | Polysaccharide isolated from Pleurotus ostreatus is heteropolysaccharide, composed of galactose, glucose, mannose, rhamnose, and xylose | Vamanu [15]; Wang et al. [25]; Zhang et al. [200] |
| Cordyceps gunni | Fungi | The polysaccharide of Cordyceps gunnii was the α-polysaccharide type, with a glycosidic bond, mainly composed of d-mannose, d-glucose, and d-galactose | Zhu et al. [202]; Zhu et al. [45] |
| Grifola frondosa | Fungi | G. frondosa polysaccharides contain glucose, galactose, mannose, fucose, and ribose, with high amounts of (1→3,1→6)-β-d-glucans, which would account for 13.2% of water-soluble polysaccharides | Bae et al. [117]; He et al. [131] |
| Lachnum | Fungi | Polysaccharide of Lachnum YM261(LEPS-1) is considered homogenous with a molecular weight of 21,670 Da, with its glucan linked by β-(1→3)-d-pyran glycosidic bond | Wu et al. [195]; Ye et al. [199] |
| Dictyophora indusiata | Fungi | Polysaccharide (DIP) extracted from Dictyophora indusiata indicated a specific polysaccharide of (1→3)- β-d-glucan with (1→6)-β-glucopyranoside side chains | Deng et al. [187]; Deng et al. [188] |
| Ganoderma atrum | Fungi | Polysaccharide fraction from Ganoderma atrum composed of glucose (Glc), mannose (Man), galactose (Gal) and galacturonic acid (GalA) in molar ratio of 4.91:1:1.28:0.71 | Chen et al. [186]; Zhang et al. [200] |
| Paenibacillus polymyxa | Bacteria | - Paenibacillus polymyxa bioactive compounds like exopolysaccharides, for example, d-glucuronic acid, and polysaccharide beads - Paenibacillus polymyxa was shown to comprise mannose, galactose, and glucose in a ratio of 1.23:1.14:1 | Daud et al. [143]; Raza et al. [151]; Wang et al. [25] |
| Bacillus coagulans | Bacteria | - Bacillus coagulans polysaccharide could have with monosaccharides like galactose, glucose, lactose, mannose, raffinose, rhamnose, and xylose - Exopolysaccharide (EPS) from Bacillus coagulans, comprise galactose, mannose, fucose, glucose, and glucosamine | Kodali et al. [149]; Ramanathan et al. [150]; Wang et al. [25]; |
| Edwardsiella tarda | Bacteria | - E. tarda polysaccharide comprise glucosamine, a fucose, a mannose and a galactosamine unit; - Another E. tarda polysaccharide comprise d-glucuronic acid with l-alanine, branched hexasaccharide repeating unit. - branch points situated at C-2 and C-6 positions of the (1→3)-linked mannose residues, whereas the side chains composed of (1→2)-linked and (1→)-linked mannose residues | Guo et al. [144]; Katzenellenbogen et al. [147,148]; Wang et al. [25] |
| Streptomyces virginia | Bacteria | -A purified polysaccharide Streptomyces virginia (HO3 type) comprised mannose, glucose and galactose, in a 2:1:1 proportion, with average apparent molecular weight of 3.76 × 104 Da | He et al. [145] |
| Hyriopsis cumingii | Animal | Polysaccharides of Hyriopsis cumingii have pyranose rings, with glycosyl residues, either linked by α- and β-configuration glycosidic bonds | Qiao et al. [141]; Wang et al. [25] |
| Misgurnus anguillicaudatus | Animal | Misgurnus anguillicaudatus, polysaccharide (MAP) or misgurnan, comprise the major structural monomers identified included d-galactose, l-fucose and d-mannose, and monoses that link each other by α-1,3 bonds | Qin et al. [142]; Wang et al. [25] |
| Laminaria japonica | Algae | sulfated polysaccharide (L-A) has high galactose content but low fucose residues proportion | Wang et al. [25]; Xu et al. [160] |
| Enteromorpha linza | Algae | Enteromorpha linza polysaccharides contain large amount of sulfated polysaccharides, composed mainly of galactose, mannose, glucuronic acid, glucose, rhamnose, and xylose | Wang et al. [25]; Zhang et al. [36] |
| Bifurcaria bifurcata | Algae | Glucose and myo-inositol have been detected in Bifurcaria bifurcata | Mian and Percival [152]; Soukaina et al. [156] |
| Phaeophyceae spp | Algae | Sulfated polysaccharides (SPs) found in Phaeophyceae spp can include laminarans, which comprise repeating dimeric units of λ-carrageenan | Wang et al. [25]; Wijesekara et al. [159] |
| Fucus vesiculosus Linnaeus | Algae | - The main neutral sugars of Fucus vesiculosus include fucose, glucose, galactose, and xylose - Sulphated polysaccharides of Fucus vesiculosus has a heterogeneous structure | Rodriguez-Jasso et al. [153]; Rupérez, Ahrazem, and Antonio Leal [154]; Wang et al. [25] |
| Padina gymnospora Sonder | Algae | Sulfated polysaccharide (SP) isolated from Padina gymnospora contains 29.4 ± 0.35% of sulfate, 27 ± 0.11% of fucose, 0.05 ± 0.12% of protein | Vasantharaja et al. [158]; Wang et al. [25] |
| Laminaria angustata | Algae | Polysaccharides from Laminaria angustata obtained via fucoidan -led macrophage activation produced chemical composition ratio of l-fucose:d-galactose:d-glucose:d-xylose:uronic acid:sulfate = 1.00:0.54:0.08:0.08:0.64:0.87 | Saha et al. [155]; Teruya et al. [157] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ofoedu, C.E.; You, L.; Osuji, C.M.; Iwouno, J.O.; Kabuo, N.O.; Ojukwu, M.; Agunwah, I.M.; Chacha, J.S.; Muobike, O.P.; Agunbiade, A.O.; et al. Hydrogen Peroxide Effects on Natural-Sourced Polysacchrides: Free Radical Formation/Production, Degradation Process, and Reaction Mechanism—A Critical Synopsis. Foods 2021, 10, 699. https://doi.org/10.3390/foods10040699
Ofoedu CE, You L, Osuji CM, Iwouno JO, Kabuo NO, Ojukwu M, Agunwah IM, Chacha JS, Muobike OP, Agunbiade AO, et al. Hydrogen Peroxide Effects on Natural-Sourced Polysacchrides: Free Radical Formation/Production, Degradation Process, and Reaction Mechanism—A Critical Synopsis. Foods. 2021; 10(4):699. https://doi.org/10.3390/foods10040699
Chicago/Turabian StyleOfoedu, Chigozie E., Lijun You, Chijioke M. Osuji, Jude O. Iwouno, Ngozi O. Kabuo, Moses Ojukwu, Ijeoma M. Agunwah, James S. Chacha, Onyinye P. Muobike, Adedoyin O. Agunbiade, and et al. 2021. "Hydrogen Peroxide Effects on Natural-Sourced Polysacchrides: Free Radical Formation/Production, Degradation Process, and Reaction Mechanism—A Critical Synopsis" Foods 10, no. 4: 699. https://doi.org/10.3390/foods10040699
APA StyleOfoedu, C. E., You, L., Osuji, C. M., Iwouno, J. O., Kabuo, N. O., Ojukwu, M., Agunwah, I. M., Chacha, J. S., Muobike, O. P., Agunbiade, A. O., Sardo, G., Bono, G., Okpala, C. O. R., & Korzeniowska, M. (2021). Hydrogen Peroxide Effects on Natural-Sourced Polysacchrides: Free Radical Formation/Production, Degradation Process, and Reaction Mechanism—A Critical Synopsis. Foods, 10(4), 699. https://doi.org/10.3390/foods10040699











