Morus alba L. Plant: Bioactive Compounds and Potential as a Functional Food Ingredient
Abstract
1. Introduction
2. Nutrients and Phytochemical Compositions of Morus alba
2.1. Nutrient Composition of Morus alba
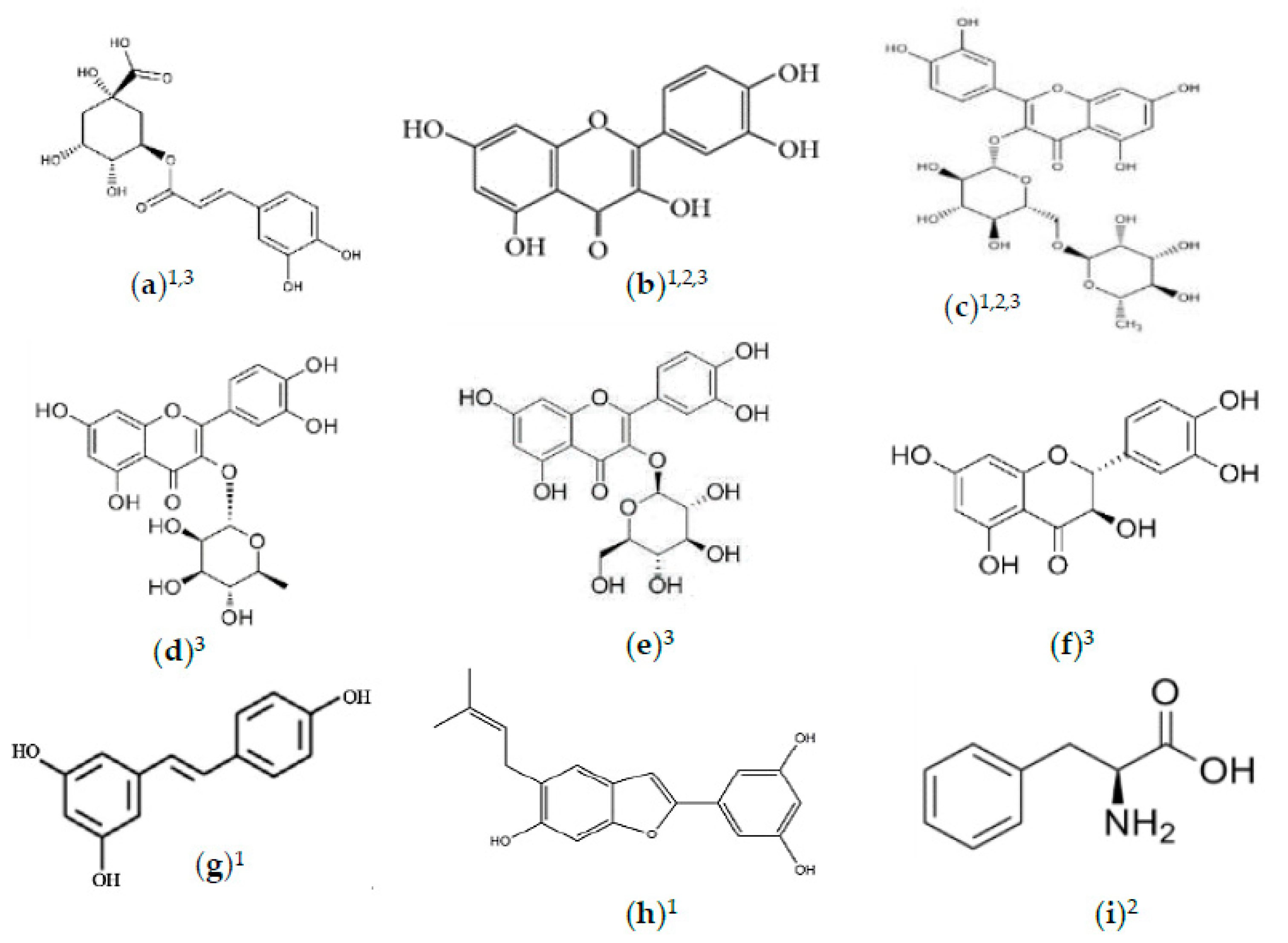
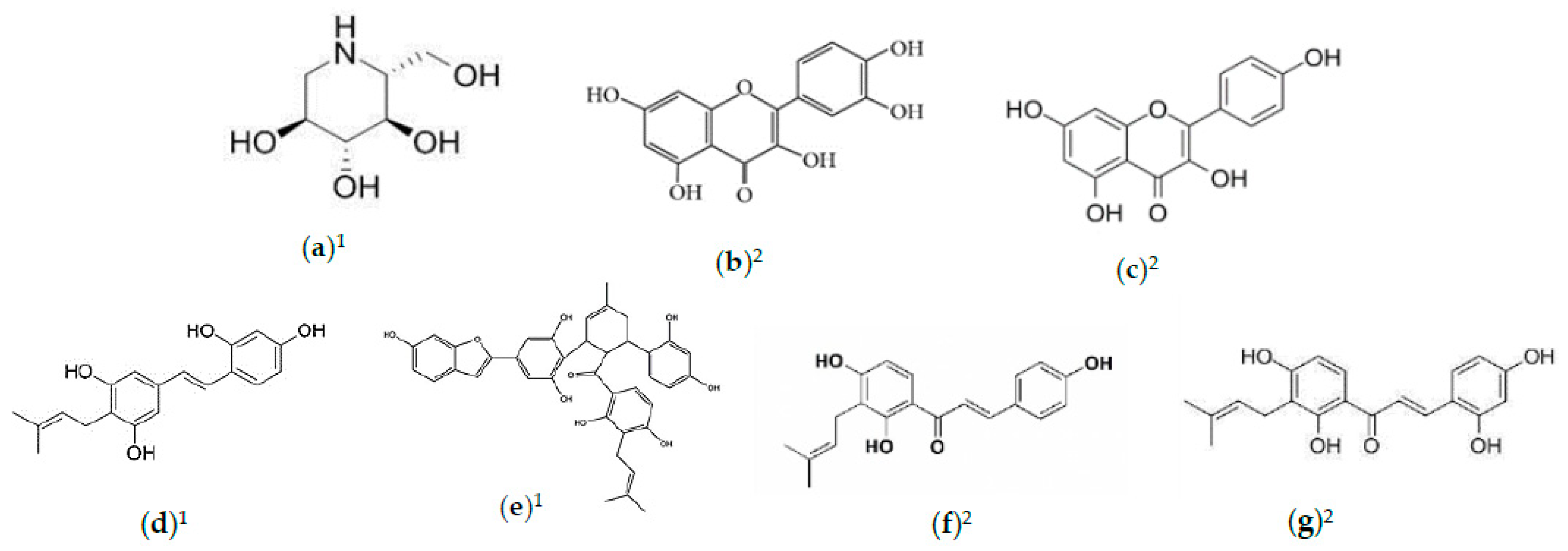
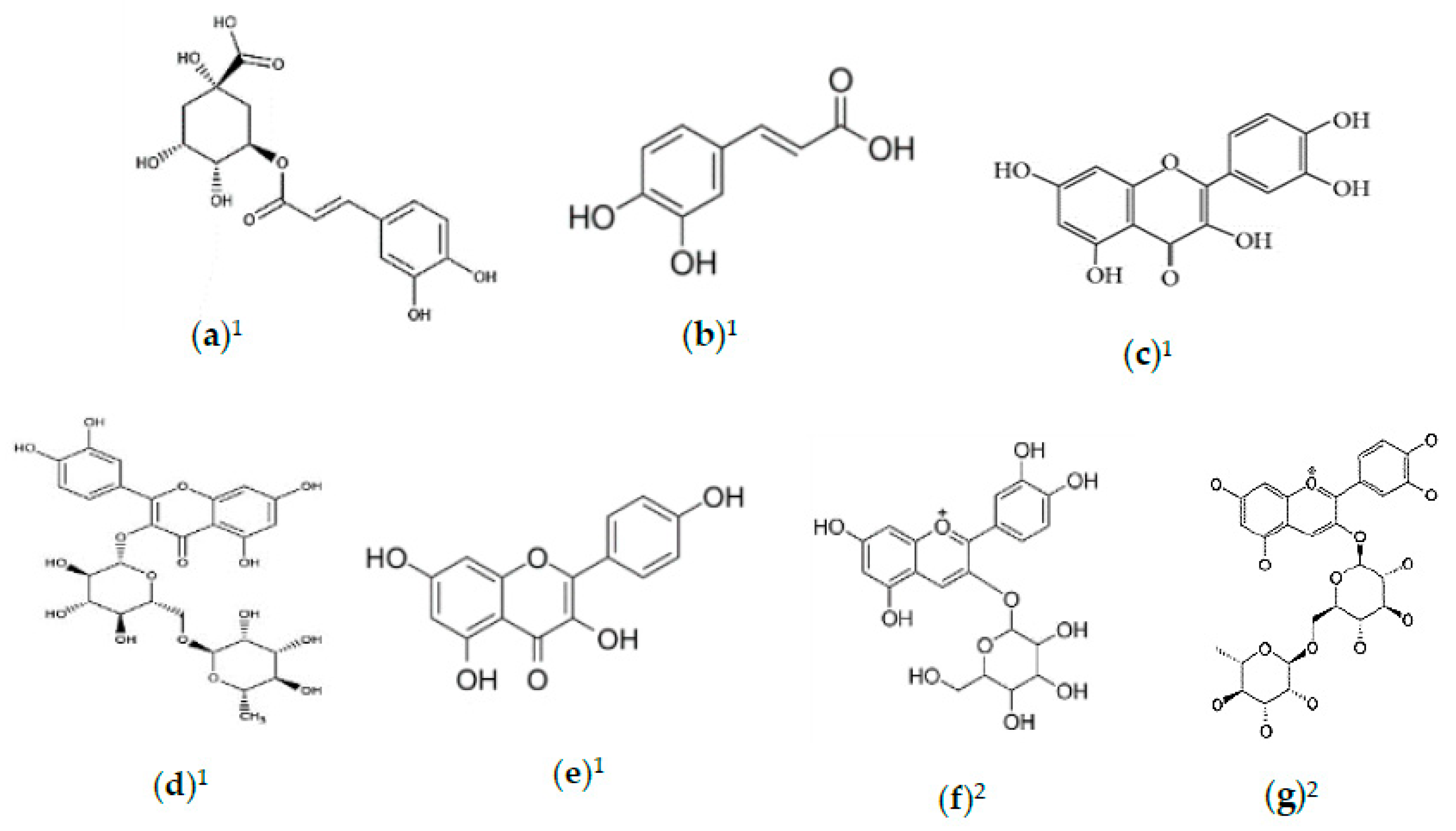
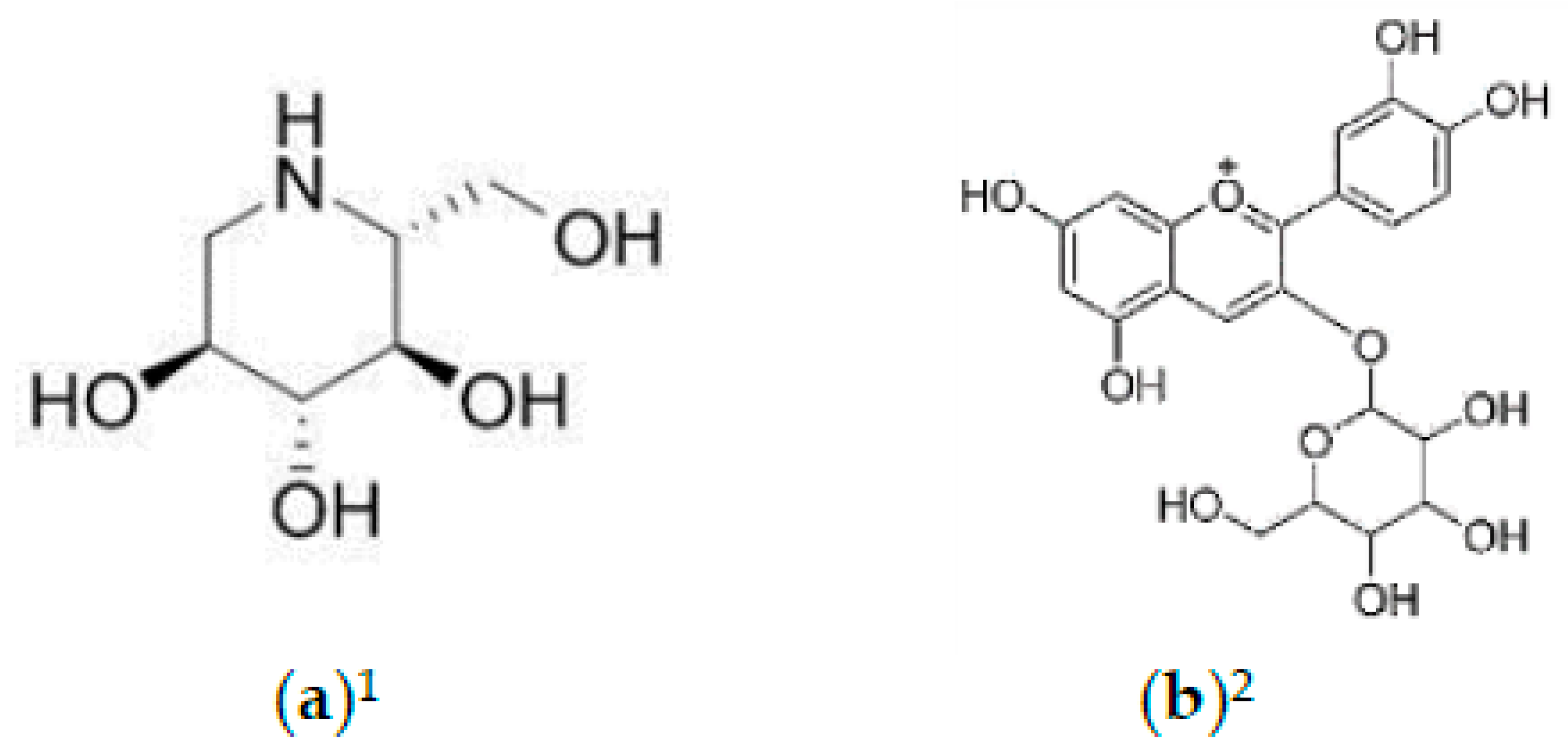
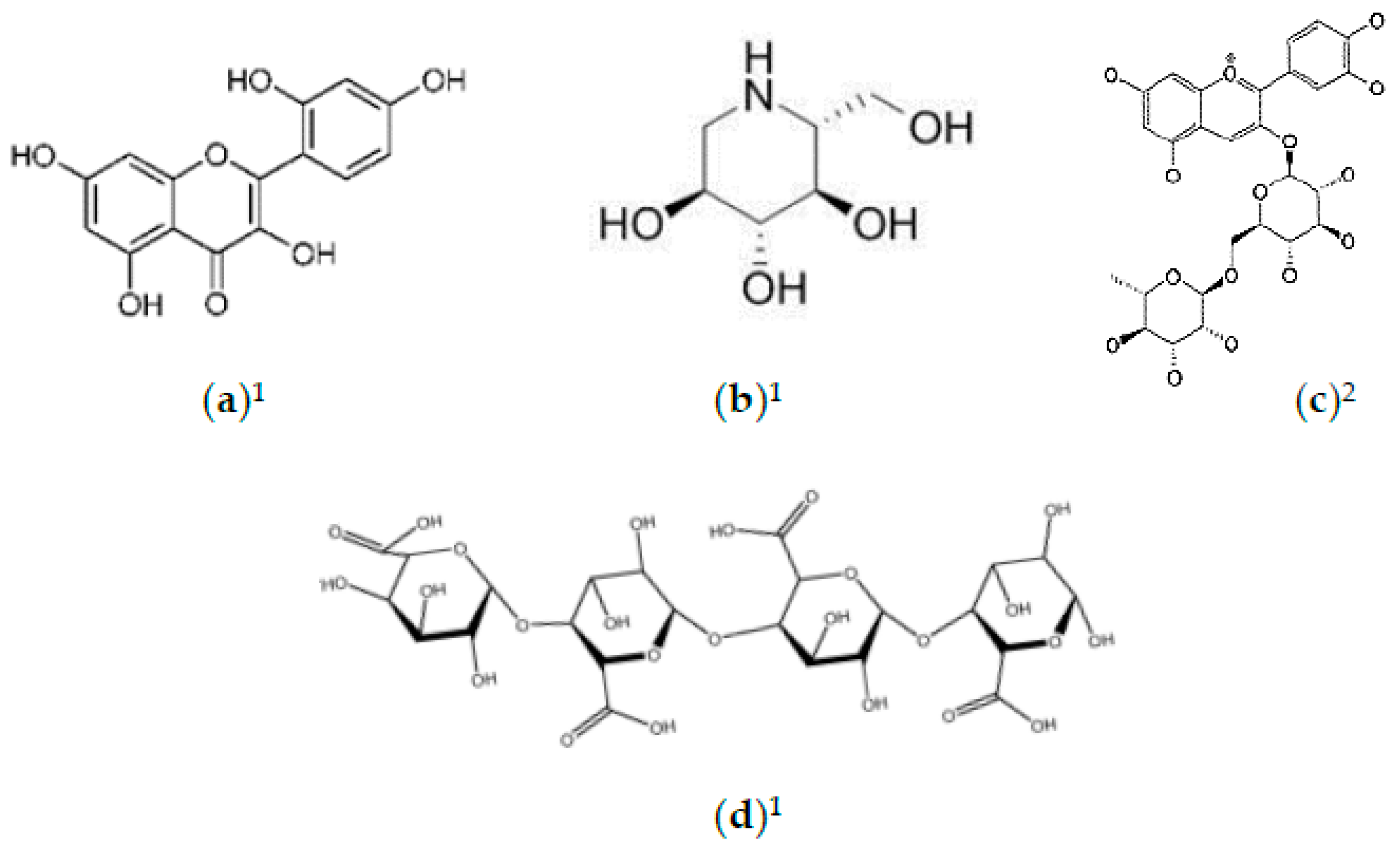
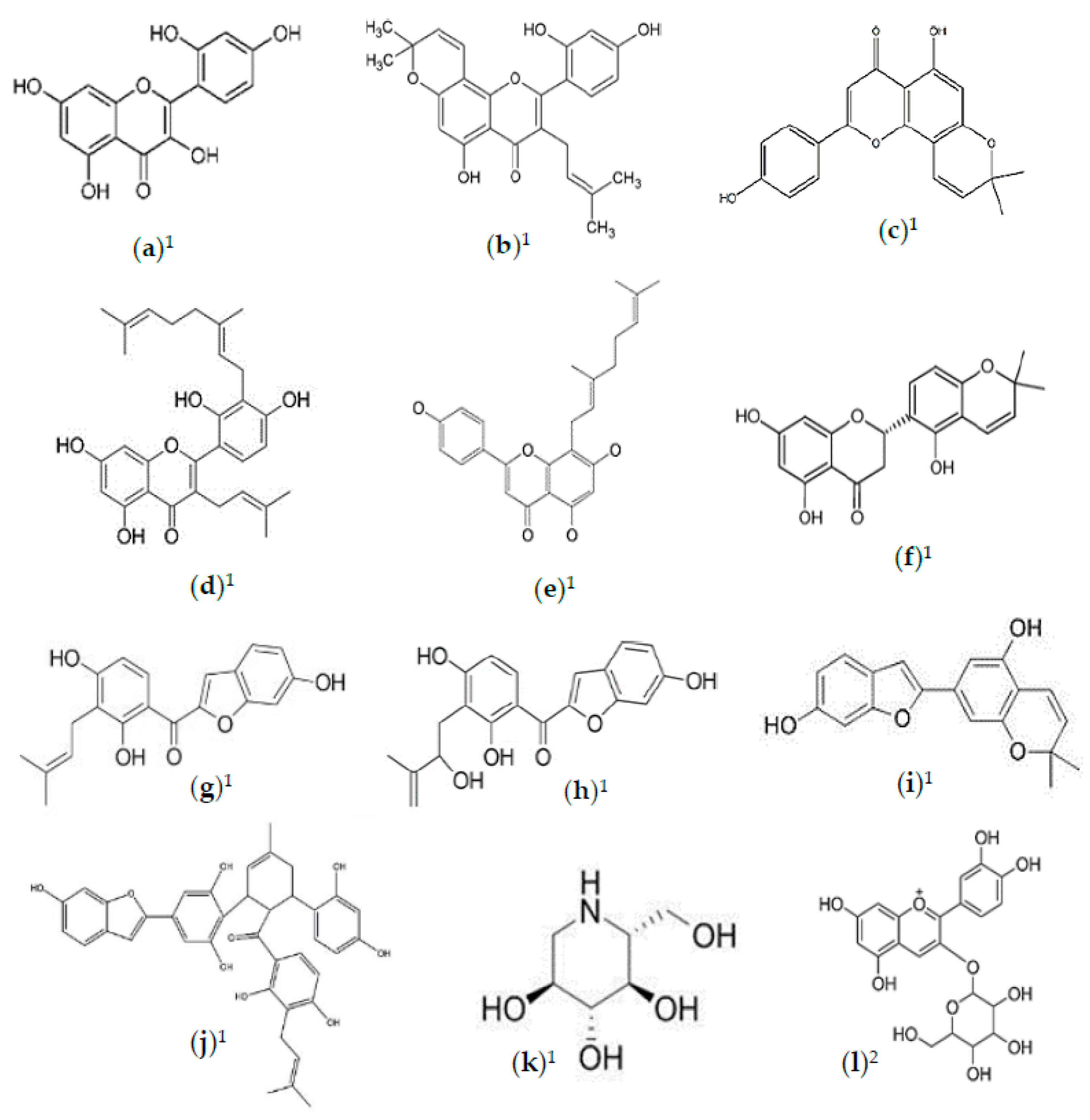
2.2. Phytochemicals of Morus alba Leaves
2.3. Phytochemicals of Morus alba Fruit
2.4. Phytochemicals of Morus alba Seeds
3. Nutraceutical Properties of Morus alba
3.1. Antioxidative Activity
3.2. Antidiabetic Property
3.3. Antihyperlipidaemia and Antiobesity Activity
3.4. Neuroprotective Ability
3.5. Antimicrobial and Antiviral Activity
3.6. Cytotoxicity and Anticancer Activities
4. Toxicity Study of Morus alba
5. Functional Ingredients in Food Applications
Microencapsulation of Morus alba
6. Industrial Scale-Up
7. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ercisli, S.; Orhan, E. Chemical composition of white (Morus alba), red (Morus rubra) and black (Morus nigra) mulberry fruits. Food Chem. 2007, 103, 1380–1384. [Google Scholar] [CrossRef]
- Dat, N.T.; Binh, P.T.X.; Van Minh, C.; Huong, H.T.; Lee, J.J. Cytotoxic prenylated flavonoids from Morus alba. Fitoterapia 2010, 81, 1224–1227. [Google Scholar] [CrossRef]
- Sánchez, M.D. (Ed.) World distribution and utilization of mulberry, potential for animal feeding. In Proceedings of FAO Electronic Conference Mulberry Animal Production (Morus1-L); Food and Agriculture Organization: Rome, Italy, 2000. [Google Scholar]
- Xie, H.-H.; Wei, J.-G.; Liu, F.; Pan, X.-H.; Yang, X.-B. First report of mulberry root rot caused by Lasiodiplodia theobromae in China. Plant Dis. 2014, 98, 1581. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Salcedo, E.M.; Mena, P.; García-Viguera, C.; Hernández, F.; Martínez, J.J. (Poly) phenolic compounds and antioxidant activity of white (Morus alba) and black (Morus nigra) mulberry leaves: Their potential for new products rich in phytochemicals. J. Funct. Foods 2015, 18, 1039–1046. [Google Scholar] [CrossRef]
- Sánchez-Salcedo, E.M.; Mena, P.; García-Viguera, C.; Martínez, J.J.; Hernández, F. Phytochemical evaluation of white (Morus alba L.) and black (Morus nigra L.) mulberry fruits, a starting point for the assessment of their beneficial properties. J. Funct. Foods 2015, 12, 399–408. [Google Scholar] [CrossRef]
- Hussain, F.; Rana, Z.; Shafique, H.; Malik, A.; Hussain, Z. Phytopharmacological potential of different species of Morus alba and their bioactive phytochemicals: A review. Asian Pac. J. Trop. Biomed. 2017, 7, 950–956. [Google Scholar] [CrossRef]
- Wen, P.; Hu, T.-G.; Linhardt, R.J.; Liao, S.-T.; Wu, H.; Zou, Y.-X. Mulberry: A review of bioactive compounds and advanced processing technology. Trends Food Sci. Technol. 2019, 83, 138–158. [Google Scholar] [CrossRef]
- Ludwig, I.A.; Mena, P.; Calani, L.; Cid, C.; Del Rio, D.; Lean, M.E.; Crozier, A. Variations in caffeine and chlorogenic acid contents of coffees: What are we drinking? Food Funct. 2014, 5, 1718–1726. [Google Scholar] [CrossRef]
- Ruiz-Cruz, S.; Chaparro-Hernández, S.; Hernández-Ruiz, K.L.; Cira-Chávez, L.A.; Estrada-Alvarado, M.I.; Gassos Ortega, L.; Ornelas-Paz, J.d.J.; Lopez Mata, M. Flavonoids: Important biocompounds in food. In Flavonoids: From Biosynthesis to Human Health; Justino, G., Ed.; Intechopen: London, UK, 2017; pp. 353–370. [Google Scholar] [CrossRef]
- Jiang, Y.; Nie, W.-J. Chemical properties in fruits of mulberry species from the Xinjiang province of China. Food Chem. 2015, 174, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Salcedo, E.M.; Amorós, A.; Hernández, F.; Martínez, J.J. Physicochemical properties of white (Morus alba) and black (Morus nigra) mulberry leaves, a new food supplement. J. Food Nutr. Res. 2017, 5, 253–261. [Google Scholar] [CrossRef]
- Srivastava, S.; Kapoor, R.; Thathola, A.; Srivastava, R.P. Nutritional quality of leaves of some genotypes of mulberry (Morus alba). Int. J. Food Sci. Nutr. 2006, 57, 305–313. [Google Scholar] [CrossRef]
- Bozhüyük, M.R.; Pehluvan, M.; Tuncay, K.; Doğru, B. Organic acid composition of selected mulberry genotypes from Aras Valley. Atatürk Üniversitesi Ziraat Fakültesi Dergisi 2015, 46, 69–74. [Google Scholar]
- Owon, M.A.; Gafar, A.; Saleh, S.M.; Shaheen, M. Identification of bioactive compounds from egyptian mulberry fruits and their uses in improvement the quality of some foods. J. Agric. Res. Kafr El-Sheikh Univ. 2016, 42, 33–52. [Google Scholar]
- Lonnie, M.; Hooker, E.; Brunstrom, J.M.; Corfe, B.M.; Green, M.A.; Watson, A.W.; Williams, E.A.; Stevenson, E.J.; Penson, S.; Johnstone, A.M. Protein for life: Review of optimal protein intake, sustainable dietary sources and the effect on appetite in ageing adults. Nutrients 2018, 10, 360. [Google Scholar] [CrossRef]
- Lamiaa, M. Nutritive value and Hypolipidrmic effects of mulberry leaves powder. Indian J. Appl. Res. 2014, 4, 7–12. [Google Scholar]
- Hassan, F.-U.; Arshad, M.A.; Li, M.; Rehman, M.S.-U.; Loor, J.J.; Huang, J. Potential of Mulberry Leaf Biomass and Its Flavonoids to Improve Production and Health in Ruminants: Mechanistic Insights and Prospects. Animals 2020, 10, 2076. [Google Scholar] [CrossRef]
- Munir, A.; Khera, R.A.; Rehman, R.; Nisar, S. Multipurpose White mulberry: A Review. Int. J. Biol. Chem. Sci. 2018, 13, 31–35. [Google Scholar]
- Imran, M.; Khan, H.; Shah, M.; Khan, R.; Khan, F. Chemical composition and antioxidant activity of certain Morus species. J. Zhejiang Univ. Sci. B 2010, 11, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Nooruldin, S.; Kamili, S.; Mir, M.; Wani, A.; Malik, G.; Raja, A.; Bilal, S. Seasonal variation in macro nutrient contents of mulberry (Morus alba) leaves under temperate climatic conditions of Kashmir. Int. J. Agric. Innov. Res. 2015, 4, 147–152. [Google Scholar]
- Levickienė, D.; Vaitkevičienė, N.; Jarienė, E.; Mažeika, R. The content of macroelements in white mulberry (Morus alba L.) leaves. Žemės Ūkio Mokslai 2018, 25, 177–183. [Google Scholar] [CrossRef]
- Liang, L.; Wu, X.; Zhu, M.; Zhao, W.; Li, F.; Zou, Y.; Yang, L. Chemical composition, nutritional value, and antioxidant activities of eight mulberry cultivars from China. Pharmacogn. Mag. 2012, 8, 215. [Google Scholar] [CrossRef]
- Thabti, I.; Elfalleh, W.; Hannachi, H.; Ferchichi, A.; Campos, M.D.G. Identification and quantification of phenolic acids and flavonol glycosides in Tunisian Morus species by HPLC-DAD and HPLC–MS. J. Funct. Foods 2012, 4, 367–374. [Google Scholar] [CrossRef]
- Memon, A.A.; Memon, N.; Luthria, D.L.; Bhanger, M.I.; Pitafi, A.A. Phenolic acids profiling and antioxidant potential of mulberry (Morus laevigata W., Morus nigra L., Morus alba L.) leaves and fruits grown in Pakistan. Pol. J. Food Nutr. Sci. 2010, 60, 25–32. [Google Scholar]
- Zhang, H.-R.; Li, M.; Wang, M.-M.; Wang, X.-N.; Shen, T.; Wang, S.-Q.; Ren, D.-M. Antioxidant flavan derivatives from the leaves of Morus alba. Phytochem. Lett. 2019, 29, 84–90. [Google Scholar] [CrossRef]
- Wu, X.; Li, M.; Wang, X.; Shen, T.; Wang, S.; Ren, D. Two new 2-arylbenzofurnan derivatives from the leaves of Morus alba. Nat. Prod. Res. 2019, 33, 204–211. [Google Scholar] [CrossRef]
- Wulandari, Y.R.E.; Prasasty, V.D.; Rio, A.; Geniola, C. Determination of 1-Deoxynojirimycin Content and Phytochemical Profiles from Young and Mature Mulberry Leaves of Morus Spp. OnLine J. Biol. Sci. 2019, 19, 124–131. [Google Scholar] [CrossRef][Green Version]
- Jia, C.; Li, Y.; Zhang, S.; Fei, T.; Pang, S. Thermogravimetric analysis, kinetic study, and pyrolysis–GC/MS analysis of 1, 1′-azobis-1, 2, 3-triazole and 4, 4′-azobis-1, 2, 4-triazole. Chem. Cent. J. 2018, 12, 22. [Google Scholar] [CrossRef] [PubMed]
- Polumackanycz, M.; Sledzinski, T.; Goyke, E.; Wesolowski, M.; Viapiana, A. A Comparative Study on the Phenolic Composition and Biological Activities of Morus alba L. Commercial Samples. Molecules 2019, 24, 3082. [Google Scholar] [CrossRef]
- Ali, M.; Rathaur, B.; Nishad, U. Phytochemical Screening, Physicochemical and Antioxidant Activity, TLC & Finger Print of HPTLC, Morus Alba Ethanol Extraction. Int. J. Innov. Sci. Res. Technol. 2020, 5, 1–14. [Google Scholar]
- Chan, E.W.C.; Wong, S.K.; Tangah, J.; Inoue, T.; Chan, H.T. Phenolic constituents and anticancer properties of Morus alba (white mulberry) leaves. J. Integr. Med. 2020, 18, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Li, H.; Zhang, B.; Wang, J.; Shi, X.; Huang, J.; Yang, J.; Zhang, Y.; Deng, Z. Nutritional and functional components of mulberry leaves from different varieties: Evaluation of their potential as food materials. Int. J. Food Prop. 2018, 21, 1495–1507. [Google Scholar] [CrossRef]
- Raman, S.T.; Ganeshan, A.K.P.G.; Chen, C.; Jin, C.; Li, S.-H.; Chen, H.-J.; Gui, Z. In vitro and in vivo antioxidant activity of flavonoid extracted from mulberry fruit (Morus alba L.). Pharmacogn. Mag. 2016, 12, 128. [Google Scholar] [CrossRef]
- Hao, J.-Y.; Wan, Y.; Yao, X.-H.; Zhao, W.-G.; Hu, R.-Z.; Chen, C.; Li, L.; Zhang, D.-Y.; Wu, G.-H. Effect of different planting areas on the chemical compositions and hypoglycemic and antioxidant activities of mulberry leaf extracts in Southern China. PLoS ONE 2018, 13, e0198072. [Google Scholar] [CrossRef] [PubMed]
- Asano, N.; Yamashita, T.; Yasuda, K.; Ikeda, K.; Kizu, H.; Kameda, Y.; Kato, A.; Nash, R.J.; Lee, H.S.; Ryu, K.S. Polyhydroxylated alkaloids isolated from mulberry trees (Morus alba L.) and silkworms (Bombyx mori L.). J. Agric. Food Chem. 2001, 49, 4208–4213. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-S.; Ha, T.-Y.; Ahn, J.-Y.; Kim, H.-K.; Kim, S.-A. Composition and quantitative analysis of stilbenoids in mulberry (Morus alba L.) leaves and fruits with DAD/UV HPLC. J. Korean Soc. Food Sci. Nutr. 2008, 37, 124–128. [Google Scholar] [CrossRef]
- Chen, W.; Li, Y.; Bao, T.; Gowd, V. Mulberry fruit extract affords protection against ethyl carbamate-induced cytotoxicity and oxidative stress. Oxidative. Med. Cell. Longev. 2017, 2017, 1594963. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xiang, L.; Wang, C.; Tang, C.; He, X. Antidiabetic and antioxidant effects and phytochemicals of mulberry fruit (Morus alba L.) polyphenol enhanced extract. PLoS ONE 2013, 8, e71144. [Google Scholar] [CrossRef] [PubMed]
- Calín-Sánchez, Á.; Martínez-Nicolás, J.J.; Munera-Picazo, S.; Carbonell-Barrachina, Á.A.; Legua, P.; Hernández, F. Bioactive compounds and sensory quality of black and white mulberries grown in Spain. Plant Foods Hum. Nutr. 2013, 68, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Chu, Q.; Lin, M.; Tian, X.; Ye, J. Study on capillary electrophoresis–amperometric detection profiles of different parts of Morus alba L. J. Chromatogr. A 2006, 1116, 286–290. [Google Scholar] [CrossRef]
- Xu, X.; Huang, Y.; Xu, J.; He, X.; Wang, Y. Anti-neuroinflammatory and antioxidant phenols from mulberry fruit (Morus alba L.). J. Funct. Foods 2020, 68, 103914. [Google Scholar] [CrossRef]
- Zhang, H.; Ma, Z.F.; Luo, X.; Li, X. Effects of mulberry fruit (Morus alba L.) consumption on health outcomes: A mini-review. Antioxidants 2018, 7, 69. [Google Scholar] [CrossRef] [PubMed]
- Aramwit, P.; Bang, N.; Srichana, T. The properties and stability of anthocyanins in mulberry fruits. Food Res. Int. 2010, 43, 1093–1097. [Google Scholar] [CrossRef]
- El-Baz, F.K.; Hassan, A.Z.; Abd-Alla, H.I.; Aly, H.F.; Mahmoud, K. Phytochemical analysis, assessment of antiproliferative and free radical scavenging activity of Morus alba and Morus rubra fruits. Asian J. Pharm. Clin. Res. 2017, 10, 189. [Google Scholar] [CrossRef]
- Kobus-Cisowska, J.; Szczepaniak, O.; Szymanowska-Powałowska, D.; Piechocka, J.; Szulc, P.; Dziedziński, M. Antioxidant potential of various solvent extract from Morus alba fruits and its major polyphenols composition. Ciênc. Rural 2020, 50, 50. [Google Scholar] [CrossRef]
- Kim, I.; Lee, J. Variations in Anthocyanin Profiles and Antioxidant Activity of 12 Genotypes of Mulberry (Morus spp.) Fruits and Their Changes during Processing. Antioxidants 2020, 9, 242. [Google Scholar] [CrossRef]
- Lee, Y.-J.; Kim, E.-O.; Choi, S.-W. Isolation and identification of antioxidant polyphenolic compounds in mulberry (Morus alba L.) seeds. J. Korean Soc. Food Sci. Nutr. 2011, 40, 517–524. [Google Scholar] [CrossRef]
- Oh, M.; Bae, S.Y.; Chung, M.S. Mulberry (Morus alba) seed extract and its polyphenol compounds for control of foodborne viral surrogates. J. Korean Soc. Appl. Biol. Chem. 2013, 56, 655–660. [Google Scholar] [CrossRef]
- Argon, Z.Ü.; İlhan, N.; Gökyer, A.; Öztürk, S.B.; Koparal, B. Phytochemical Evaluation of Morus alba Seeds and Cold Pressed Oil. J. Turk. Chem. Soc. Sect. A Chem. 2019, 6, 41–50. [Google Scholar] [CrossRef]
- Sbihi, H.M.; Nehdi, I.A.; Mokbli, S.; Romdhani-Younes, M.; Al-Resayes, S.I. Hexane and ethanol extracted seed oils and leaf essential compositions from two castor plant (Ricinus communis L.) varieties. Ind. Crops Prod. 2018, 122, 174–181. [Google Scholar] [CrossRef]
- Trigui, I.; Zarai, Z.; Chevance, S.; Cheikh-Rouhou, S.; Attia, H.; Ayadi, M. Physicochemical properties, antioxidant activity and in vitro gastrointestinal digestion of purified proteins from black cumin seeds. Int. J. Biol. Macromol. 2019, 126, 454–465. [Google Scholar] [CrossRef] [PubMed]
- Yılmaz, M.A.; Durmaz, G. Mulberry seed oil: A rich source of δ-tocopherol. J. Am. Oil Chem. Soc. 2015, 92, 553–559. [Google Scholar] [CrossRef]
- Yao, X.-H.; Shen, Y.-S.; Hu, R.-Z.; Xu, M.; Huang, J.-X.; He, C.-X.; Cao, F.-L.; Fu, Y.-J.; Zhang, D.-Y.; Zhao, W.-G. The antioxidant activity and composition of the seed oil of mulberry cultivars. Food Biosci. 2020, 37, 100709. [Google Scholar] [CrossRef]
- Rahman, M.M.; Akther, A.; Moinuddin, M.; Yeasmin, M.S.; Rahman, M.M.; Rahman, M.S.; Ferdousi, S.A.; Sayeed, M.A. Investigation some physicochemical properties, lipids, glycerides and fatty acid composition of mulberry (Morus alba L.) seed oil of three different regions of Bangladesh. Am. J. Appl. Chem. 2014, 2, 38–41. [Google Scholar] [CrossRef]
- Cui, H.; Lu, T.; Wang, M.; Zou, X.; Zhang, Y.; Yang, X.; Dong, Y.; Zhou, H. Flavonoids from Morus alba L. leaves: Optimization of extraction by response surface methodology and comprehensive evaluation of their antioxidant, antimicrobial, and inhibition of α-amylase activities through analytical hierarchy process. Molecules 2019, 24, 2398. [Google Scholar] [CrossRef]
- Swapana, N.; Jotinkumar, T.; Devi, C.B.; Singh, M.S.; Singh, S.B.; Singh, C. Total Phenolic, Total Flavonoid Contents And Antioxidant Activity Of A Fe w Indigenous Fruits Grown In Manipur. Bioscan 2012, 7, 73–76. [Google Scholar]
- Jha, S.; Gupta, S.K.; Bhattacharyya, P.; Ghosh, A.; Mandal, P. In vitro antioxidant and antidiabetic activity of oligopeptides derived from different mulberry (Morus alba L.) cultivars. Pharmacogn. Res. 2018, 10, 361. [Google Scholar] [CrossRef]
- D’urso, G.; Mes, J.J.; Montoro, P.; Hall, R.D.; de Vos, R.C. Identification of bioactive phytochemicals in mulberries. Metabolites 2020, 10, 7. [Google Scholar] [CrossRef]
- Przeor, M.; Flaczyk, E.; Beszterda, M.; Szymandera-Buszka, K.E.; Piechocka, J.; Kmiecik, D.; Szczepaniak, O.; Kobus-Cisowska, J.; Jarzębski, M.; Tylewicz, U. Air-drying temperature changes the content of the phenolic acids and flavonols in white mulberry (Morus alba L.) leaves. Ciênc. Rural 2019, 49, 1–4. [Google Scholar] [CrossRef]
- Tu, J.; Shi, D.; Wen, L.; Jiang, Y.; Zhao, Y.; Yang, J.; Liu, H.; Liu, G.; Yang, B. Identification of moracin N in mulberry leaf and evaluation of antioxidant activity. Food Chem. Toxicol. 2019, 132, 110730. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Chen, C.; Fu, X. The effect of geographic variation on chemical composition, antioxidant and hypoglycemic activities of Morus alba L. polysaccharides. J. Food Process. Preserv. 2019, 43, e14206. [Google Scholar] [CrossRef]
- Malviya, N.; Jain, S.; Malviya, S. Antidiabetic potential of medicinal plants. Acta Pol. Pharm. 2010, 67, 113–118. [Google Scholar]
- Ahn, E.; Choi, S.-W.; Kim, E. Anti-diabetic effect of sericultural product in high fat diet-fed mice. J. Korean Soc. Food Sci. Nutr. 2017, 46, 289–297. [Google Scholar] [CrossRef]
- Eruygur, N.; Dural, E. Determination of 1-deoxynojirimycin by a developed and validated HPLC-FLD method and assessment of in-vitro antioxidant, α-amylase and α-glucosidase inhibitory activity in mulberry varieties from Turkey. Phytomedicine 2019, 53, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Vichasilp, C.; Nakagawa, K.; Sookwong, P.; Higuchi, O.; Luemunkong, S.; Miyazawa, T. Development of high 1-deoxynojirimycin (DNJ) content mulberry tea and use of response surface methodology to optimize tea-making conditions for highest DNJ extraction. LWT Food Sci. Technol. 2012, 45, 226–232. [Google Scholar] [CrossRef]
- Jeon, Y.-H.; Choi, S.-W. Isolation, Identification, and Quantification of Tyrosinase and α-Glucosidase Inhibitors from UVC-Irradiated Mulberry (Morus alba L.) Leaves. Prev. Nutr. Food Sci. 2019, 24, 84. [Google Scholar] [CrossRef]
- Choi, K.H.; Lee, H.A.; Park, M.H.; Han, J.-S. Mulberry (Morus alba L.) fruit extract containing anthocyanins improves glycemic control and insulin sensitivity via activation of AMP-activated protein kinase in diabetic C57BL/Ksj-db/db mice. J. Med. Food 2016, 19, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Khyade, V.B. Utilization of mulberry methanolic leaf extractive (MMLE) for treating hypercholesterolemia in rat, rattus norvegicus (l). Int. J. Acad. Res. 2019, 1, 404–418. [Google Scholar]
- Metwally, F.M.; Rashad, H.; Mahmoud, A.A. Morus alba L. Diminishes visceral adiposity, insulin resistance, behavioral alterations via regulation of gene expression of leptin, resistin and adiponectin in rats fed a high-cholesterol diet. Physiol. Behav. 2019, 201, 1–11. [Google Scholar] [CrossRef]
- Li, H.X.; Jo, E.; Myung, C.-S.; Kim, Y.H.; Yang, S.Y. Lipolytic effect of compounds isolated from leaves of mulberry (Morus alba L.) in 3T3-L1 adipocytes. Nat. Prod. Res. 2018, 32, 1963–1966. [Google Scholar] [CrossRef]
- Lupo, M.G.; Macchi, C.; Marchianò, S.; Cristofani, R.; Greco, M.F.; Dall’Acqua, S.; Chen, H.; Sirtori, C.R.; Corsini, A.; Ruscica, M. Differential effects of red yeast rice, Berberis aristata and Morus alba extracts on PCSK9 and LDL uptake. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 1245–1253. [Google Scholar] [CrossRef]
- Yang, X.; Yang, L.; Zheng, H. Hypolipidemic and antioxidant effects of mulberry (Morus alba L.) fruit in hyperlipidaemia rats. Food Chem. Toxicol. 2010, 48, 2374–2379. [Google Scholar] [CrossRef] [PubMed]
- El-Baz, F.K.; Aly, H.F.; Abd-Alla, H.I.; Fayed, D.B. Therapeutic impact of berries (Morus alba and Morus rubra) fruit extract in the regression of high-fat diet-induced cardiac dysfunction in rats. Asian J. Pharm. Clin. Res. 2018, 11, 314–320. [Google Scholar] [CrossRef]
- Gupta, G.; Afzal, M.; David, S.R.; Verma, R.; Candaswamy, M.; Anwar, F. Anticonvulsant activity of Morus alba and its effect on brain gamma-aminobutyric acid level in rats. Pharmacogn. Res. 2014, 6, 188. [Google Scholar] [CrossRef] [PubMed]
- Rayam, S.; Kudagi, B.; Sufyan, U.; Buchineni, M.; Pathapati, R.M. Assessment of Morus alba (Mulberry) leaves extract for anti convulsant property in rats. Int. J. Basic Clin. Pharmacol. 2019, 8, 520–523. [Google Scholar] [CrossRef]
- Diniz, T.C.; Silva, J.C.; Lima-Saraiva, S.R.; Ribeiro, F.P.; Pacheco, A.G.; de Freitas, R.M.; Quintans-Júnior, L.J.; Quintans, J.D.; Mendes, R.L.; Almeida, J.R. The role of flavonoids on oxidative stress in epilepsy. Oxid. Med. Cell. Longev. 2015, 2015, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tamtaji, O.R.; Mohammadifar, M.; Behnam, M.; Taghizadeh, M.; Talaei, S.A. Investigation of the effect of alcoholic Morus alba leave extract on scopolamine-induced spatial memory impairment in rats. Sci. J. Kurd. Univ. Med. Sci. 2016, 21, 30–39. [Google Scholar]
- Chen, W.; Liang, T.; Zuo, W.; Wu, X.; Shen, Z.; Wang, F.; Li, C.; Zheng, Y.; Peng, G. Neuroprotective effect of 1-Deoxynojirimycin on cognitive impairment, β-amyloid deposition, and neuroinflammation in the SAMP8 mice. Biomed. Pharmacother. 2018, 106, 92–97. [Google Scholar] [CrossRef]
- Liu, D.; Du, D. Mulberry Fruit Extract Alleviates Cognitive Impairment by Promoting the Clearance of Amyloid-β and Inhibiting Neuroinflammation in Alzheimer’s Disease Mice. Neurochem. Res. 2020, 45, 2009–2019. [Google Scholar] [CrossRef]
- El-Baz, F.K.; Aly, H.F.; Abd-Alla, H.I.; Ali, S.A. Neurorestorative mulberries potential of alzheimer’s disease in animal model. Asian J. Pharm. Clin. Res. 2018, 11, 318–324. [Google Scholar] [CrossRef]
- Rebai, O.; Belkhir, M.; Boujelben, A.; Fattouch, S.; Amri, M. Morus alba leaf extract mediates neuroprotection against glyphosate-induced toxicity and biochemical alterations in the brain. Environ. Sci. Pollut. Res. 2017, 24, 9605–9613. [Google Scholar] [CrossRef]
- Gu, P.S.; Moon, M.; Choi, J.G.; Oh, M.S. Mulberry fruit ameliorates Parkinson’s-disease-related pathology by reducing α-synuclein and ubiquitin levels in a 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine/probenecid model. J. Nutr. Biochem. 2017, 39, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, M.I.H.; Kim, H.-B.; Kim, S.Y.; Cho, K.-O. The neuroprotective potential of cyanidin-3-glucoside fraction extracted from mulberry following oxygen-glucose deprivation. Korean J. Physiol. Pharmacol. 2011, 15, 353. [Google Scholar] [CrossRef]
- Suwansri, S.; Khaoprasert, S.; Ratanatriwong, P.; Promboon, A. Natural preservative from Thai mulberry: The antioxidant and antibacterial properties of leaf extracts from different cultivars. Acta Hortic. 2008, 786, 115–124. [Google Scholar] [CrossRef]
- Thabti, I.; Elfalleh, W.; Tlili, N.; Ziadi, M.; Campos, M.G.; Ferchichi, A. Phenols, flavonoids, and antioxidant and antibacterial activity of leaves and stem bark of Morus species. Int. J. Food Prop. 2014, 17, 842–854. [Google Scholar] [CrossRef]
- De Oliveira, A.M.; Mesquita, M.d.S.; da Silva, G.C.; de Oliveira Lima, E.; de Medeiros, P.L.; Paiva, P.M.G.; Souza, I.A.d.; Napoleão, T.H. Evaluation of toxicity and antimicrobial activity of an ethanolic extract from leaves of Morus alba L. (Moraceae). Evid. Based Complementary Altern. Med. 2015, 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-Y.; Lee, H.-S. Evaluation of antioxidant and antibacterial activities of morin isolated from mulberry fruits (Morus alba L.). J. Korean Soc. Appl. Biol. Chem. 2012, 55, 485–489. [Google Scholar] [CrossRef]
- Kumar, R.V.; Srivastava, D.; Singh, V.; Kumar, U.; Vishvakarma, V.K.; Singh, P.; Kumar, D.; Kumar, R. Characterization, biological evaluation and molecular docking of mulberry fruit pectin. Sci. Rep. 2020, 10, 1–16. [Google Scholar] [CrossRef]
- Kostić, D.S.D.D.A.; Mitić, G.S.S.S.-S.; Đorđević, M.N.M.A.S. Phenolic composition, antioxidant activity, mineral content and antimicrobial activity of fresh fruit extracts of Morus alba L. J. Food Nutr. Res. 2014, 53, 22–30. [Google Scholar]
- Genovese, C.; Scarpaci, K.; D’angeli, F.; Caserta, G.; Nicolosi, D. Antibacterial activity of Morus alba L. leaf extracts. Ann. Clin. Case Stud. 2020, 2, 1017. [Google Scholar]
- Kim, B.S.; Kim, H.; Kang, S.-S. In vitro anti-bacterial and anti-inflammatory activities of lactic acid bacteria-biotransformed mulberry (Morus alba Linnaeus) fruit extract against Salmonella Typhimurium. Food Control 2019, 106, 106758. [Google Scholar] [CrossRef]
- Jacob, J.R.; Mansfield, K.; You, J.-E.; Tennant, B.C.; Kim, Y.-H. Natural iminosugar derivatives of 1-deoxynojirimycin inhibit glycosylation of hepatitis viral envelope proteins. J. Microbiol. 2007, 45, 431–440. [Google Scholar] [PubMed]
- Soltanian, S.; Sheikhbahaei, M.; Mohamadi, N. Cytotoxicity evaluation of methanol extracts of some medicinal plants on P19 embryonal carcinoma cells. J. Appl. Pharm. Sci. 2017, 7, 142–149. [Google Scholar]
- Fathy, S.A.; Singab, A.N.B.; Agwa, S.A.; El Hamid, D.M.A.; Zahra, F.A.; El Moneim, S.M.A. The antiproliferative effect of mulberry (Morus alba L.) plant on hepatocarcinoma cell line HepG2. Egypt. J. Med. Hum. Genet. 2013, 14, 375–382. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, F.; Thakur, K.; Wang, J.; Wang, H.; Hu, F.; Zhang, J.-G.; Wei, Z.-J. Molecular mechanism of anti-cancerous potential of Morin extracted from mulberry in Hela cells. Food Chem. Toxicol. 2018, 112, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Fallah, S.; Karimi, A.; Panahi, G.; Nejad, S.G.; Fadaei, R.; Seifi, M. Human colon cancer HT-29 cell death responses to doxorubicin and Morus alba leaves flavonoid extract. Cell. Mol. Biol. 2016, 62, 72–77. [Google Scholar] [PubMed]
- Jyoti, A.; Anand, K.; Sunanda, P. Synergistic action of phytochemicals augments their antioxidative efficacy: An in vitro comparative study. Asian J. Pharm. Clin. Res. 2013, 6, 121–126. [Google Scholar]
- Cho, E.; Y Chung, E.; Jang, H.-Y.; Hong, O.-Y.; S Chae, H.; Jeong, Y.-J.; Kim, S.-Y.; Kim, B.-S.; J Yoo, D.; Kim, J.-S. Anti-cancer effect of cyanidin-3-glucoside from mulberry via caspase-3 cleavage and DNA fragmentation in vitro and in vivo. Anticancer Agents Med. Chem. 2017, 17, 1519–1525. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.S.; Lee, D.; Lee, S.R.; Lee, J.W.; Choi, C.-I.; Jang, T.S.; Kang, K.S.; Kim, K.H. Chemical characterization of cytotoxic indole acetic acid derivative from mulberry fruit (Morus alba L.) against human cervical cancer. Bioorg. Chem. 2018, 76, 28–36. [Google Scholar] [CrossRef]
- Ramis, E.S.; Hammoud, G.M.; ElSawy, K.M. In vitro and In vivo Studies on Mulberry Extracts: Evaluation of Chemical and Anticancer Activities and Attenuation of Lead Toxicity. Asian J. Res. Biochem. 2018, 1–14. [Google Scholar] [CrossRef]
- Mitsuya, M. Four-week oral toxicity studies of the leaf powder of mulberry (Morus alba L.) in rats. Pharmacometrics 2001, 61, 169–179. [Google Scholar]
- Phornchirasilp, S.; Luanratana, O.; Sirikulchayanonta, V.; Tiengda, C. A Toxicity Study of Morus alba L. Leaf Extract. Thai J. Pharm. Sci. 2004, 26, 70. [Google Scholar]
- de Oliveira, A.M.; do Nascimento, M.F.; Ferreira, M.R.A.; de Moura, D.F.; dos Santos Souza, T.G.; da Silva, G.C.; da Silva Ramos, E.H.; Paiva, P.M.G.; de Medeiros, P.L.; da Silva, T.G. Evaluation of acute toxicity, genotoxicity and inhibitory effect on acute inflammation of an ethanol extract of Morus alba L. (Moraceae) in mice. J. Ethnopharmacol. 2016, 194, 162–168. [Google Scholar] [CrossRef]
- Jiao, Y.; Wang, X.; Jiang, X.; Kong, F.; Wang, S.; Yan, C. Antidiabetic effects of Morus alba fruit polysaccharides on high-fat diet-and streptozotocin-induced type 2 diabetes in rats. J. Ethnopharmacol. 2017, 199, 119–127. [Google Scholar] [CrossRef]
- Chang, B.Y.; Kim, S.B.; Lee, M.K.; Park, H.; Kim, S.Y. Nonclinical Safety Assessment of Morus alba L. Fruits: Study of 90-D Toxicity in Sprague Dawley Rats and Genotoxicity in Salmonella. J. Food Sci. 2016, 81, T1328–T1335. [Google Scholar] [CrossRef]
- Paul, A.; Shakya, A.; Zaman, M.K. Assessment of acute and sub-chronic neurotoxicity of Morus alba L. fruits in rodents. Future J. Pharm. Sci. 2020, 6, 1–11. [Google Scholar] [CrossRef]
- Ruengdech, A.; Siripatrawan, U.; Sangnark, A.; Benedetti, S.; Buratti, S. Rapid evaluation of phenolic compounds and antioxidant activity of mulberry leaf tea during storage using electronic tongue coupled with chemometrics. J. Berry Res. 2019, 9, 563–574. [Google Scholar] [CrossRef]
- Bhosale, S.C.; Khyade, V.B.; Patil, V.S. Preparation of functional and healthy food: The paratha (cooked dough) through the use of leaves of mulberry, Morus alba (L) from the moriculture unit of agricultural development Trust, Baramati (India). Int. J. Recent Acad. Res. 2019, 1, 506–518. [Google Scholar]
- Lim, H.S.; Park, S.H.; Ghafoor, K.; Hwang, S.Y.; Park, J. Quality and antioxidant properties of bread containing turmeric (Curcuma longa L.) cultivated in South Korea. Food Chem. 2011, 124, 1577–1582. [Google Scholar] [CrossRef]
- Jomova, K.; Valko, M. Advances in metal-induced oxidative stress and human disease. Toxicology 2011, 283, 65–87. [Google Scholar] [CrossRef] [PubMed]
- Alfeo, V.; Bravi, E.; Ceccaroni, D.; Perretti, G.; Marconi, O.; Sileoni, V. Effect of Baking Time and Temperature on Nutrients and Phenolic Compounds Content of Fresh Sprouts Breadlike Product. Foods 2020, 9, 1447. [Google Scholar] [CrossRef]
- Yang, J.; Li, T.; Cao, H.; Bu, H.; Hou, X.; Lu, C. Effects of dietary forage mulberry on the growth performance and meat quality of finishing pigs. Chin. J. Anim. Sci. 2014, 50, 52–56. [Google Scholar] [CrossRef]
- Zhu, Z.; Jiang, J.-J.; Jie, Y.; Mao, X.-B.; Bing, Y.; Chen, D.-W. Effect of dietary supplementation with mulberry (Morus alba L.) leaves on the growth performance, meat quality and antioxidative capacity of finishing pigs. J. Integr. Agric. 2019, 18, 143–151. [Google Scholar]
- Ouyang, J.; Hou, Q.; Wang, M.; Zhao, W.; Feng, D.; Pi, Y.; Sun, X.-Z. Effects of dietary mulberry leaf powder on growth performance, blood metabolites, meat quality and antioxidant enzyme related gene expression of fattening Hu lambs. Can. J. Anim. Sci. 2020, 100, 510–521. [Google Scholar] [CrossRef]
- Zhang, X.; Li, D.; Meng, Q.; He, C.; Ren, L. Effect of mulberry leaf extracts on color, lipid oxidation, antioxidant enzyme activities and oxidative breakdown products of raw ground beef during refrigerated storage. J. Food Qual. 2016, 39, 159–170. [Google Scholar] [CrossRef]
- Butkhup, L.; Samappito, W.; Samappito, S. Phenolic composition and antioxidant activity of white mulberry (Morus alba L.) fruits. Int. J. Food Sci. Technol. 2013, 48, 934–940. [Google Scholar] [CrossRef]
- Vanitha, T.; Khan, M. Role of pectin in food processing and food packaging. In Pectins-Extraction, Purification, Characterization and Applications; Books on Demand: Norderstedt, Germany, 2019. [Google Scholar]
- Natić, M.M.; Dabić, D.Č.; Papetti, A.; Akšić, M.M.F.; Ognjanov, V.; Ljubojević, M.; Tešić, Ž.L. Analysis and characterisation of phytochemicals in mulberry (Morus alba L.) fruits grown in Vojvodina, North Serbia. Food Chem. 2015, 171, 128–136. [Google Scholar] [CrossRef]
- ve Depolama, A.K.Ü.Ç. Phenolic Composition, Colour and Anthocyanin Stability of Black Mulberry and White Mulberry Jam as Affected by Cultivar and Storage Temperature. J. Biol. Chem. 2016, 44, 395–399. [Google Scholar] [CrossRef]
- Olofsson, P.; Hultqvist, M.; Hellgren, L.I.; Holmdahl, R. Phytol: A chlorophyll component with anti-inflammatory and metabolic properties. In Recent Advances in Redox Active Plant and Microbial Products; Claus, J., Gilbert, K., Alan, S., Paul, G.W., Torsten, B., Eds.; Springer: Dordrecht, Netherland, 2014; pp. 345–359. [Google Scholar]
- Xu, L.; Zhu, M.-J.; Liu, X.-M.; Cheng, J.-R. Inhibitory effect of mulberry (Morus alba) polyphenol on the lipid and protein oxidation of dried minced pork slices during heat processing and storage. LWT 2018, 91, 222–228. [Google Scholar] [CrossRef]
- Xiang, R.; Cheng, J.; Zhu, M.; Liu, X. Effect of mulberry (Morus alba) polyphenols as antioxidant on physiochemical properties, oxidation and bio-safety in Cantonese sausages. LWT 2019, 116, 108504. [Google Scholar] [CrossRef]
- Tomczyk, M.; Miłek, M.; Sidor, E.; Kapusta, I.; Litwińczuk, W.; Puchalski, C.; Dżugan, M. The Effect of Adding the Leaves and Fruits of Morus alba to Rape Honey on Its Antioxidant Properties, Polyphenolic Profile, and Amylase Activity. Molecules 2020, 25, 84. [Google Scholar] [CrossRef]
- Kobus-Cisowska, J.; Dziedziński, M.; Szymanowska, D.; Szczepaniak, O.; Byczkiewicz, S.; Telichowska, A.; Szulc, P. The Effects of Morus alba L. Fortification on the Quality, Functional Properties and Sensory Attributes of Bread Stored under Refrigerated Conditions. Sustainability 2020, 12, 6691. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Andrade, M.; de Melo, N.R.; Sanches-Silva, A. Use of essential oils in active food packaging: Recent advances and future trends. Trends Food Sci. Technol. 2017, 61, 132–140. [Google Scholar] [CrossRef]
- Zhi-ming, L.; Hai-ying, W.; Shan-shan, L.; Nai-xiang, J. Volatile Components of Essential Oil from Mulberry Variety “Longsang 1” Leaves. Nat. Prod. Res. 2011, 23, 1069–1084. [Google Scholar]
- Radulović, N.S.; Miljković, V.M.; Mladenović, M.Z.; Nikolić, G.S. Essential oils of Morus alba and M. nigra leaves: Effect of drying on the chemical composition. Nat. Prod. Commun. 2017, 12, 1934578X1701200133. [Google Scholar] [CrossRef]
- Santos, C.C.D.M.P.; Salvadori, M.S.; Mota, V.G.; Costa, L.M.; de Almeida, A.A.C.; de Oliveira, G.A.L.; Costa, J.P.; de Sousa, D.P.; de Freitas, R.M.; de Almeida, R.N. Antinociceptive and antioxidant activities of phytol in vivo and in vitro models. Neurosci. J. 2013, 2013, 94945. [Google Scholar] [CrossRef]
- Aparna, V.; Dileep, K.V.; Mandal, P.K.; Karthe, P.; Sadasivan, C.; Haridas, M. Anti-inflammatory property of n-hexadecanoic acid: Structural evidence and kinetic assessment. Chem. Biol. Drug Des. 2012, 80, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Ravi, L.; Krishnan, K. Research Article Cytotoxic Potential of N-hexadecanoic Acid Extracted from Kigelia pinnata Leaves. Asian J. Cell Biol. 2017, 12, 20–27. [Google Scholar] [CrossRef]
- Yeo, S.K.; Ali, A.Y.; Hayward, O.A.; Turnham, D.; Jackson, T.; Bowen, I.D.; Clarkson, R. β-Bisabolene, a sesquiterpene from the essential oil extract of opoponax (Commiphora guidottii), exhibits cytotoxicity in breast cancer cell lines. Phytother. Res. 2016, 30, 418–425. [Google Scholar] [CrossRef]
- Dong, X.; Gao, X.; Liu, L.; Chen, L.; Liu, Q.; Zhang, D. Function-specific volatiles and volatilization characteristics of Dendrobium officinale. J. King Saud Univ. Sci. 2020, 32, 2020–2028. [Google Scholar] [CrossRef]
- Caputi, L.; Aprea, E. Use of terpenoids as natural flavouring compounds in food industry. Recent Pat. Food Nutr. Agric. 2011, 3, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Mezzomo, N.; Ferreira, S.R. Carotenoids functionality, sources, and processing by supercritical technology: A review. J. Chem. 2016, 2016, 3164312. [Google Scholar] [CrossRef]
- Özcan, M.M.; Lemiasheuski, V.; Özcan, M.M. Fatty acid compositions of white and black mulberry fruit oils. J. Agroaliment. Process. Technol. 2019, 25, 179–181. [Google Scholar]
- Mimica-Dukić, N.; Božin, B.; Soković, M.; Mihajlović, B.; Matavulj, M. Antimicrobial and antioxidant activities of three Mentha species essential oils. Planta Med. 2003, 69, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, D.J.; Stratford, M.; Narbad, A. Analysis of the inhibition of food spoilage yeasts by vanillin. Int. J. Food Microbiol. 2003, 86, 113–122. [Google Scholar] [CrossRef]
- Oussalah, M.; Caillet, S.; Saucier, L.; Lacroix, M. Inhibitory effects of selected plant essential oils on the growth of four pathogenic bacteria: E. coli O157: H7, Salmonella typhimurium, Staphylococcus aureus and Listeria monocytogenes. Food Control 2007, 18, 414–420. [Google Scholar] [CrossRef]
- Pandey, A.K.; Kumar, P.; Singh, P.; Tripathi, N.N.; Bajpai, V.K. Essential oils: Sources of antimicrobials and food preservatives. Front. Microbiol. 2017, 7, 2161. [Google Scholar] [CrossRef]
- Chang, J.; Kang, X.; Yuan, J.-L. Enhancing emulsification and antioxidant ability of egg albumin by moderately acid hydrolysis: Modulating an emulsion-based system for mulberry seed oil. Food Res. Int. 2018, 109, 334–342. [Google Scholar] [CrossRef]
- Li, Y.-Q.; Kong, D.-X.; Huang, R.-S.; Liang, H.-L.; Xu, C.-G.; Wu, H. Variations in essential oil yields and compositions of Cinnamomum cassia leaves at different developmental stages. Ind. Crops Prod. 2013, 47, 92–101. [Google Scholar] [CrossRef]
- Negi, P.S. Plant extracts for the control of bacterial growth: Efficacy, stability and safety issues for food application. Int. J. Food Microbiol. 2012, 156, 7–17. [Google Scholar] [CrossRef]
- Li, D.; Zhu, M.; Liu, X.; Wang, Y.; Cheng, J. Insight into the effect of microcapsule technology on the processing stability of mulberry polyphenols. LWT 2020, 126, 109144. [Google Scholar] [CrossRef]
- Xu, L.; Cheng, J.-R.; Liu, X.-M.; Zhu, M.-J. Effect of microencapsulated process on stability of mulberry polyphenol and oxidation property of dried minced pork slices during heat processing and storage. LWT 2019, 100, 62–68. [Google Scholar] [CrossRef]
- Cheng, J.-R.; Xiang, R.; Liu, X.-M.; Zhu, M.-J. The effects of thermal processing and β-cyclodextrin on extractable polyphenols in mulberry juice-enriched dried minced pork slices. LWT 2019, 116, 108503. [Google Scholar] [CrossRef]
- Butstraen, C.; Salaün, F. Preparation of microcapsules by complex coacervation of gum Arabic and chitosan. Carbohydr. Polym. 2014, 99, 608–616. [Google Scholar] [CrossRef]
- Efara, É.; Marquis, F.; Tremblay, A. Scaling Up Biotechnological Chemical Processes: A Better Alternative to the Traditional Develop-Then-Scale Model. Ind. Biotechnol. 2019, 15, 157–161. [Google Scholar] [CrossRef]
- Flaczyk, E.; Kobus-Cisowska, J.; Przeor, M.; Korczak, J.; Remiszewski, M.; Korbas, E.; Buchowski, M. Chemical characterization and antioxidative properties of Polish variety of Morus alba L. leaf aqueous extracts from the laboratory and pilot-scale processes. J. Agric. Sci. 2013, 4, 141. [Google Scholar] [CrossRef]
- Gang, T.; Xin-Fu, Y.; Xiao-Ming, P.; Cui-Qing, L.; Teng, W.; Qi, Y. Response Surface Methodology-Optimized Pilot-Plant Extraction of Polysaccharides from Mulberry Leaves. In Proceedings of the Joint International Conference on Social Science and Environment Science (SSES) and International Conference on Food Science and Engneering (ICFSE), Guangzhou, China, 15–16 October 2016; DEStech: Lancaster, PA, USA, 2016; pp. 1–13. [Google Scholar]
- Tian, G.; Yin, X.; Peng, X.; Li, C.; Wang, T.; Feng, J.; Ju, R. Response Surface Methodology Optimized Pilot Plant Extraction Process of 1-Deoxynojirimycin from Mulberry Leaves. J. Biobased Mater. Bioenergy 2019, 13, 207–213. [Google Scholar] [CrossRef]
- Komaikul, J.; Kitisripanya, T.; Inyai, C.; Likhitwitayawuid, K.; Sritularak, B.; Tanaka, H.; Putalun, W. Phytostilbenoid production in white mulberry (Morus alba L.) cell culture using bioreactors and simple deglycosylation by endogenous enzymatic hydrolysis. In Vitro Cell. Dev. Biol. Plant 2019, 55, 199–208. [Google Scholar] [CrossRef]
| Nutrients | Carbohydrate | Protein | Fats | Fibre | Ascorbic Acid | β-Carotene | References | |
|---|---|---|---|---|---|---|---|---|
| Sample | ||||||||
| M. alba leaves | 28.8% | 27.1% | 1.9% | 26.5% | - | - | [17] | |
| 9.7–29.6% | 15.3–30.9% | 2.1–4.9% | 27.6–36.7% | 100–200 mg/100 g | 8.4–13.1 mg/100 g | [13] | ||
| 9.7–39.7% | 14.0–34.2% | 3.5–8.1% | 5.4–38.4% | - | - | [18] | ||
| M. alba fruit | - | - | 1.10% | - | 22.4 mg/100 mL | - | [1] | |
| - | 10.2– 13.3% | - | - | - | - | [6] | ||
| - | 12.98% | - | 8.32% | 351 mg/g | 13.7 mg/100 g | [15] | ||
| 71% | 13–15% | 2–3.5% | 12–14% | - | - | [19] | ||
| - | 1.55 g/100 g | 0.48 g/100 g | 1.47 g/100 g | 15.2 g/100 g | - | [20] | ||
| Minerals | Ca | Zn | Fe | Mg | P | K | Mn | Na | References | |
|---|---|---|---|---|---|---|---|---|---|---|
| Sample | ||||||||||
| M. alba leaves | 14.9 mg/g | 2.2 mg/100 g | 27.1 mg/100 g | 5.3 mg/g | 3.7 mg/g | 12.4 mg/g | - | 58.62 mg/100 g | [17] | |
| 1.0–1.6% | - | - | 0.4–0.7% | 0.2–0.3% | 1.3–1.9% | - | - | [21] | ||
| 11.2–27.6 mg/g | - | - | 2.2–3.4 mg/g | 3.8–7.02 mg/g | 16.1–18.6 mg/100 g | - | - | [22] | ||
| M. alba fruit | 1.52 mg/g | 2.8 mg/100 g | 4.2 mg/100 g | 1.06 mg/g | 2.47 mg/g | 16.68 mg/g | 3.8 mg/100 g | 60 mg/100 g | [1] | |
| 0.2–0.4 g/100 g | 14.9–19.6 mg/kg | 28.2–46.7 mg/kg | 0.1–0.2 g/100 g | 0.2–0.3 g/100 g | 1.6–2.1 g/100 g | 12.3–19.4 mg/kg | 0.01 g/100 g | [6] | ||
| 2.73 mg/g | 1.8 mg/100 g | 6.3 mg/100 g | 1.82 mg/g | 1.98 mg/g | 9.07 mg/g | 0.8 mg/100 g | 82.8 mg/100 g | [23] | ||
| 5.76 mg/g | 50.20 mg/100 g | 73 mg/100 g | 2.4 mg/g | - | 17.31 mg/g | - | 2.8 mg/g | [20] | ||
| Phytochemical Class | Amount in Leaves | References |
|---|---|---|
| Flavonoids | 57.8% | [32] |
| 21.36–56.41 mg RE/g DW | [33] | |
| 20.4–187.23 mg QUE/g | [34] | |
| 3.66–6.11 mg/g DW | [5] | |
| Benzofurans | 17.9% | [32] |
| Phenolic acids | 10.7% | [32] |
| 0.84–1.07 mg/g DW | [25] | |
| 6.78–8.48 mg/g DW | [5] | |
| Alkaloids | 6.4% | [32] |
| 0.680–6.909 mg/g | [35] | |
| 1359 mg/kg | [36] | |
| Coumarins | 3.6% | [32] |
| Chalcones | 2.9% | [32] |
| Stilbenes | 0.7% | [32] |
| 188.57 mg/100 g DW | [37] |
| Phytochemical Class | Amount in Fruit | References |
|---|---|---|
| Phenolic acids | 0.013–0.57 mg CAE/g DW | [30] |
| 0.90–2.18 mg/g DW | [46] | |
| 1.17–3.62 mg/g DW | [6] | |
| 0.62–0.84 mg/g DW | [25] | |
| Flavonoids | 0.026–0.607 mg QE/g | [30] |
| 0.553–2.83 mg/g DW | [46] | |
| 3.66–6.11 mg/g DW | [6] | |
| Alkaloids | 660 mg/100 FW | [20] |
| 1047 mg/kg | [36] | |
| Anthocyanins | 0.95–28.61 mg/g DW | [47] |
| 6.52 mg/100 g DW | [24] | |
| Sterol | 28.07% | [45] |
| Stilbenes | 609.15 mg/100 g DW | [37] |
| M. alba Materials | Extraction Solvent | Assay | Result | References |
|---|---|---|---|---|
| Leaves | Ethanol, Petroleum ether, Ethyl acetate, and n-butanol. | DPPH | IC50 = 145–2070 mg/mL | [56] |
| ABTS | IC50 = 32.73–642.33 mg/mL | |||
| FRAP | EC50 = 868.67–2429.33 mg/mL | |||
| Leaves | Methanol | DPPH | 33.22–56.37 μmol TE/g DW | [33] |
| FRAP | 91.62–149.15 μmol AAE/g DW | |||
| ABTS | 51.28–70.84 μmol TE/g DW | |||
| Leaf oligopeptides | Liquid nitrogen | DPPH | 322.7–876.9 μg/mL | [58] |
| ABTS | 141.3–259.6 μg/mL | |||
| Nitric oxide scavenging | 5.11–176.4 μg/mL | |||
| Metal chelation | 169.6–328.6 μg/mL | |||
| Anti-lipid peroxidation | 202.3–315.5 μg/mL | |||
| Leaf Moracin N, Resveratrol, and Quercetin | Ethanol | Oxygen radical absorption capacity | 1.55–10.86 μmol TE/μmol | [61] |
| DPPH | IC50 = 40.00–285.54 μM | |||
| Cellular antioxidant activity | No PBS wash: 6.68–22.51 μmol QE/100 μmol | |||
| Fruit | Ethanol | DPPH | IC50 = 0.518 mg/mL | [34] |
| FRAP | 0.522–0.685 | |||
| Ferrous ion chelation | 72.6% | |||
| Lipid peroxidation | 39–45.51% | |||
| Fruit | Ethanol, Hexane, Chloroform, Ethyl acetate and n-Butanol | DPPH | EC50 = 71.12–623.86 mg/L | [39] |
| Superoxide anion radical-scavenging | EC50 = 82.37–921.83 mg/L | |||
| Fruit | Ethanol, N-hexane, Dichloromethane, and Ethyl acetate | DPPH | 35.43–66.6% | [45] |
| Fruit polysaccharides | Ethanol | Oxygen radical absorption capacity | 1117.3–2159.8 μmol TE/g | [62] |
| Rapid peroxyl radical scavenging capacity | 75.23–461.32 μmol Vit C/g | |||
| Cellular antioxidant activity | No PBS wash: 19.64–66.41 μmol QE/100 g | |||
| PBS wash: 3.13–7.66 μmol QE/10 g | ||||
| Seed polyphenols | Methanol | DPPH | IC50 = 20.2–48.2 μM | [48] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.; Mohamad Razali, U.H.; Saikim, F.H.; Mahyudin, A.; Mohd Noor, N.Q.I. Morus alba L. Plant: Bioactive Compounds and Potential as a Functional Food Ingredient. Foods 2021, 10, 689. https://doi.org/10.3390/foods10030689
Chen C, Mohamad Razali UH, Saikim FH, Mahyudin A, Mohd Noor NQI. Morus alba L. Plant: Bioactive Compounds and Potential as a Functional Food Ingredient. Foods. 2021; 10(3):689. https://doi.org/10.3390/foods10030689
Chicago/Turabian StyleChen, Centhyea, Umi Hartina Mohamad Razali, Fiffy Hanisdah Saikim, Azniza Mahyudin, and Nor Qhairul Izzreen Mohd Noor. 2021. "Morus alba L. Plant: Bioactive Compounds and Potential as a Functional Food Ingredient" Foods 10, no. 3: 689. https://doi.org/10.3390/foods10030689
APA StyleChen, C., Mohamad Razali, U. H., Saikim, F. H., Mahyudin, A., & Mohd Noor, N. Q. I. (2021). Morus alba L. Plant: Bioactive Compounds and Potential as a Functional Food Ingredient. Foods, 10(3), 689. https://doi.org/10.3390/foods10030689








