Probiotics as a Possible Strategy for the Prevention and Treatment of Allergies. A Narrative Review
Abstract
1. Introduction
2. Methodology
3. Results and Discussion
3.1. Characteristics of Allergic Diseases and Most Common Allergic Diseases
3.2. The Immune System and Probiotics
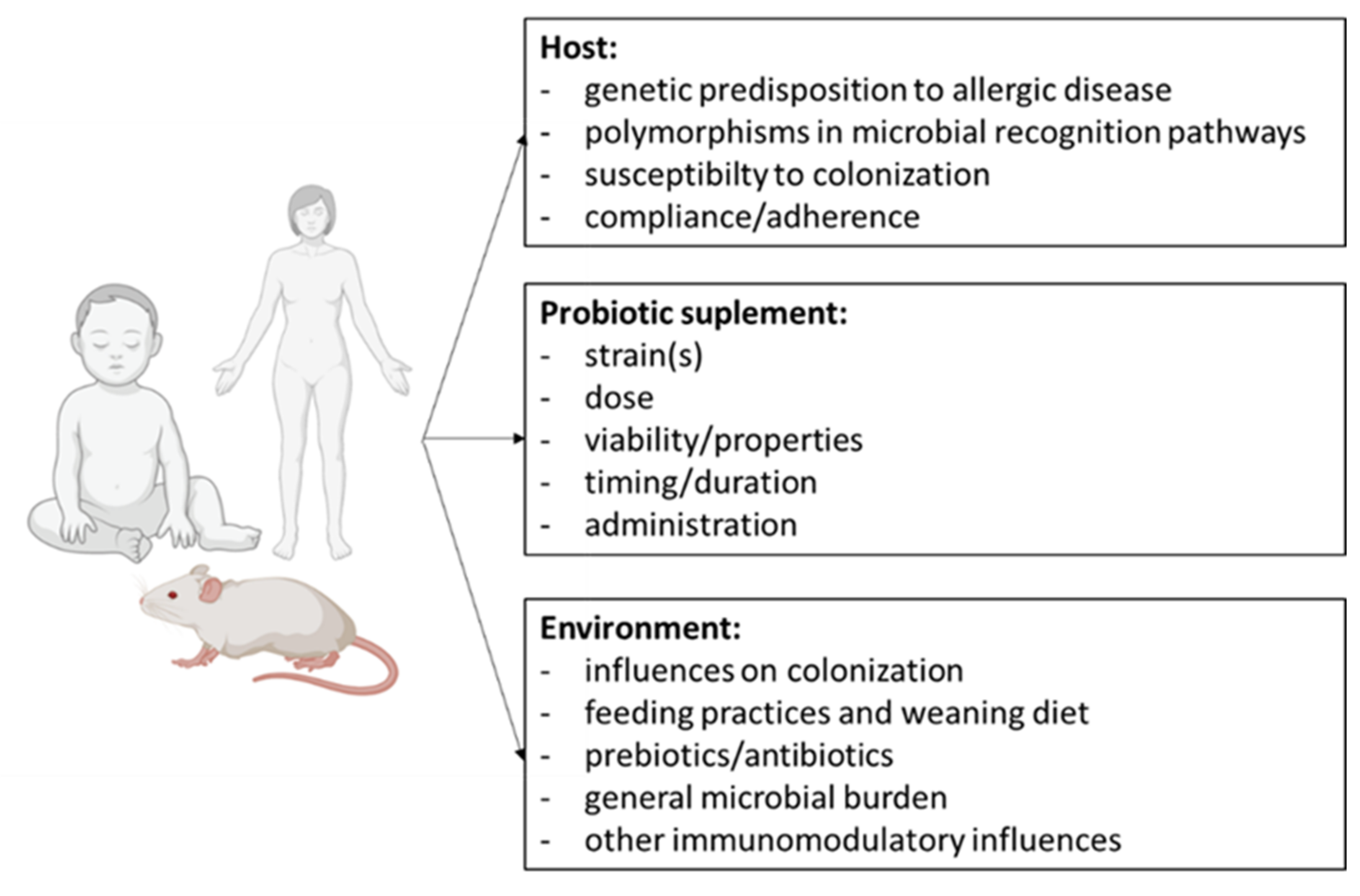
3.3. Use of Probiotics in Allergic Diseases
4. Limitations, Conclusions, and Future Trends
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Spacova, I.; Ceuppens, J.L.; Seys, S.F.; Petrova, M.I.; Lebeer, S. Probiotics against airway allergy: Host factors to consider. Dis. Models Mech. 2018, 11. [Google Scholar] [CrossRef]
- Ring, J. What Is Allergy. In Global Atlas of Allergy; Akdis, C.A., Agache, I., Eds.; European Academy of Allergy and Clinical Immunology: Zurich, Switzerland, 2014; pp. 2–3. [Google Scholar]
- Akdis, C.A.; Agache, I. (Eds.) Global Atlas of Allergy; European Academy of Allergy and Clinical Immunology: Zurich, Switzerland, 2014. [Google Scholar]
- Pawankar, R.; Canonica, G.W.; Holgate, S.T.; Lockey, R.F. WAO White Book on Allergy; World Allergy Organization: Milwaukee, WI, USA, 2011. [Google Scholar]
- Galdeano, C.M.; Cazorla, S.I.; Dumit, J.M.L.; Vélez, E.; Perdigón, G. Beneficial Effects of Probiotic Consumption on the Immune System. Ann. Nutr. Metab. 2019, 74, 115–124. [Google Scholar] [CrossRef]
- Liu, M.-Y.; Yang, Z.-Y.; Dai, W.-K.; Huang, J.-Q.; Li, Y.-H.; Zhang, J.; Qiu, C.-Z.; Wei, C.; Zhou, Q.; Sun, X.; et al. Protective Effect of Bifidobacterium infantis CGMCC313-2 on Ovalbumin-Induced Airway Asthma and β-Lactoglobulin-Induced Intestinal Food Allergy Mouse Models. World J. Gastroenterol. 2017, 23, 2149–2158. [Google Scholar] [CrossRef]
- Strachan, D.P. Hay Fever, Hygiene, and Household Size. BMJ 1989, 299, 1259–1260. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.; Gerhold, K.; Zimmermann, K.; Ockeloen, C.W.; Rossberg, S.; Wagner, P.; Sulser, C.; Bunikowski, R.; Witt, I.; Wauer, J.; et al. Oral Application of Bacterial Lysate in Infancy Decreases the Risk of Atopic Dermatitis in Children with 1 Atopic Parent in a Randomized, Placebo-Controlled Trial. J. Allergy Clin. Immunol. 2012, 129, 1040–1047. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, S.F.; Stanwell-Smith, R.; Crevel, R.W.R.; Pickup, J. Too Clean, or Not Too Clean: The Hygiene Hypothesis and Home Hygiene. Clin. Exp. Allergy 2006, 36, 402–425. [Google Scholar] [CrossRef]
- Del Giudice, M.M.; Leonardi, S.; Maiello, N.; Brunese, F.P. Food Allergy and Probiotics in Childhood. J. Clin. Gastroenterol. 2010, 44, S22–S25. [Google Scholar] [CrossRef] [PubMed]
- Leavy, O. Old McDonald Had a Farm (but No Asthma)! Nat. Rev. Immunol. 2016, 16, 535. [Google Scholar] [CrossRef]
- Schuijs, M.J.; Willart, M.A.; Vergote, K.; Gras, D.; Deswarte, K.; Ege, M.J.; Madeira, F.B.; Beyaert, R.; Van Loo, G.; Bracher, F.; et al. Farm dust and endotoxin protect against allergy through A20 induction in lung epithelial cells. Science 2015, 349(6252), 1106–1110. [Google Scholar] [CrossRef] [PubMed]
- Ojwang, V.; Nwaru, B.I.; Takkinen, H.M.; Kaila, M.; Niemelä, O.; Haapala, A.M.; Ilonen, J.; Toppari, J.; Hyöty, H.; Knip, M.; et al. Early exposure to cats, dogs and farm animals and the risk of childhood asthma and allergy. Pediatr. Allergy Immunol. 2020, 31(3), 265–272. [Google Scholar] [CrossRef] [PubMed]
- Brick, T.; Hettinga, K.; Kirchner, B.; Pfaffl, M.W.; Ege, M.J. The beneficial effect of farm milk consumption on asthma, allergies, and infections: From meta-analysis of evidence to clinical trial. J. Allergy Clin. Immunol. Pract. 2020, 8(3), 878–889.e3. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.E.; Miller, R.L. Got Milk? Understanding the Farm Milk Effect in Allergy and Asthma Prevention. J. Allergy Clin. Immunol. 2016, 137, 1707–1708. [Google Scholar] [CrossRef] [PubMed]
- Lunjani, N.; Satitsuksanoa, P.; Lukasik, Z.; Sokolowska, M.; Eiwegger, T.; O’Mahony, L. Recent Developments and Highlights in Mechanisms of Allergic Diseases: Microbiome. Allergy 2018, 73, 2314–2327. [Google Scholar] [CrossRef] [PubMed]
- Cionci, N.B.; Baffoni, L.; Gaggìa, F.; Di Gioia, D. Therapeutic Microbiology: The Role of Bifidobacterium breve as Food Supplement for the Prevention/Treatment of Paediatric Diseases. Nutrients 2018, 10, 1723. [Google Scholar] [CrossRef] [PubMed]
- Aitoro, R.; Paparo, L.; Amoroso, A.; Di Costanzo, M.; Cosenza, L.; Granata, V.; Di Scala, C.; Nocerino, R.; Trinchese, G.; Montella, M.; et al. Gut Microbiota as a Target for Preventive and Therapeutic Intervention against Food Allergy. Nutrients 2017, 9, 672. [Google Scholar] [CrossRef]
- Chernikova, D.; Yuan, I.; Shaker, M. Prevention of Allergy with Diverse and Healthy Microbiota. Curr. Opin. Pediatr. 2019, 31, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Rachid, R.; Chatila, T.A. The Role of the Gut Microbiota in Food Allergy. Curr. Opin. Pediatr. 2016, 28, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.V.; Mohite, B.V.; Patil, V.S. Probiotics for Allergic Airway Infection and Inflammations. In Probiotic Research in Therapeutics; Metzler, J.B., Ed.; Springer: Berlin/Heidelberg, Germany, 2021; pp. 295–313. [Google Scholar]
- Wang, X.; Yang, Y.; Huycke, M.M. Risks Associated with Enterococci as Probiotics. Food Res. Int. 2020, 129, 108788. [Google Scholar] [CrossRef]
- Eslami, M.; Bahar, A.; Keikha, M.; Karbalaei, M.; Kobyliak, N.; Yousefi, B. Probiotics Function and Modulation of the Immune System in Allergic Diseases. Allergol. Immunopathol. 2020, 48, 771–788. [Google Scholar] [CrossRef]
- Pawankar, R.; Canonica, G.W.; Holgate, S.T.; Lockey, R.F. Allergic Diseases and Asthma. Curr. Opin. Allergy Clin. Immunol. 2012, 12, 39–41. [Google Scholar] [CrossRef]
- Raftis, E.J.; Delday, M.I.; Cowie, P.; McCluskey, S.M.; Singh, M.D.; Ettorre, A.; Mulder, I.E. Bifidobacterium Breve MRx0004 Protects against Airway Inflammation in a Severe Asthma Model by Suppressing Both Neutrophil and Eosinophil Lung Infiltration. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Raposo, A.; Pérez, E.; Tinoco de Faria, C.; Carrascosa, C. Allergen Management as a Key Issue in Food Safety. In Food Safety and Protection; Ravishankar Rai, V., Bai, J.A., Eds.; CRC Press: Boca Ratón, FL, USA, 2017; pp. 195–242. [Google Scholar]
- Rad, A.H.; Maleki, L.A.; Kafil, H.S.; Abbasi, A. Postbiotics: A Novel Strategy in Food Allergy Treatment. Crit. Rev. Food Sci. Nutr. 2021, 61, 492–499. [Google Scholar] [CrossRef]
- Prince, B.T.; Mandel, M.J.; Nadeau, K.; Singh, A.M. Gut Microbiome and the Development of Food Allergy and Allergic Disease. Pediatr. Clin. North Am. 2015, 62, 1479–1492. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.S. Gender-Specific Asthma Treatment. Allergy Asthma Immunol. Res. 2011, 3, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Leynaert, B.; Sunyer, J.; Garcia-Esteban, R.; Svanes, C.; Jarvis, D.; Cerveri, I.; Dratva, J.; Gislason, T.; Heinrich, J.; Janson, C.; et al. Gender Differences in Prevalence, Diagnosis and Incidence of Allergic and Non-allergic Asthma: A Population-Based Cohort. Thorax 2012, 67, 625–631. [Google Scholar] [CrossRef]
- Triebner, K.; Johannessen, A.; Puggini, L.; Benediktsdóttir, B.; Bertelsen, R.J.; Bifulco, E.; Dharmage, S.C.; Dratva, J.; Franklin, K.A.; Gíslason, T.; et al. Menopause as a Predictor of New-Onset Asthma: A Longitudinal Northern European Population Study. J. Allergy Clin. Immunol. 2016, 137, 50–57. [Google Scholar] [CrossRef]
- Mendes, E.; Acetturi, B.G.; Thomas, A.M.; Martins, F.D.S.; Crisma, A.R.; Murata, G.; Braga, T.T.; Camâra, N.O.S.; Franco, A.L.D.S.; Setubal, J.C.; et al. Prophylactic Supplementation of Bifidobacterium longum 51A Protects Mice from Ovariectomy-Induced Exacerbated Allergic Airway Inflammation and Airway Hyperresponsiveness. Front. Microbiol. 2017, 8, 1732. [Google Scholar] [CrossRef]
- Greiner, A.N.; Hellings, P.W.; Rotiroti, G.; Scadding, G.K. Allergic Rhinitis. Lancet 2011, 378, 2112–2122. [Google Scholar] [CrossRef]
- Bousquet, J.; van Cauwenberge, P.; Khaltaev, N. Allergic Rhinitis and Its Impact on Asthma. J. Allergy Clin. Immunol. 2001, 108, S147–S334. [Google Scholar] [CrossRef] [PubMed]
- Bjermer, L. Time for a Paradigm Shift in Asthma Treatment: From Relieving Bronchospasm to Controlling Systemic Inflammation. J. Allergy Clin. Immunol. 2007, 120, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.K.W.; Beasley, R.; Crane, J.; Foliaki, S.; Shah, J.; Weiland, S.; the ISAAC Phase Three Study Group. Global variation in the prevalence and severity of asthma symptoms: Phase Three of the International Study of Asthma and Allergies in Childhood (ISAAC). Thorax 2009, 64, 476–483. [Google Scholar] [CrossRef]
- Church, M.K.; Weller, K.; Stock, P.; Maurer, M. Chronic spontaneous urticaria in children: Itching for insight. Pediatr. Allergy Immunol. 2011, 22, 1–8. [Google Scholar] [CrossRef]
- Zuberbier, T.; Asero, R.; Bindslev-Jensen, C.; Canonica, G.W.; Church, M.K.; Giménez-Arnau, A.; Grattan, C.E.H.; Kapp, A.; Merk, H.F.; Rogala, B.; et al. EAACI/GA2LEN/EDF/WAO Guideline: Definition, Classification and Diagnosis of Urticaria. Allergy 2009, 64, 1417–1426. [Google Scholar] [CrossRef]
- Avena-Woods, C. Overview of Atopic Dermatitis. Am. J. Manag. Care 2017, 23, S115–S123. [Google Scholar] [PubMed]
- Langan, S.M.; Irvine, A.D.; Weidinger, S. Atopic Dermatitis. Lancet 2020, 396, 345–360. [Google Scholar] [CrossRef]
- Yang, H.-J.; Min, T.K.; Lee, H.W.; Pyun, B.Y. Efficacy of Probiotic Therapy on Atopic Dermatitis in Children: A Randomized, Double-blind, Placebo-controlled Trial. Allergy Asthma Immunol. Res. 2014, 6, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Mortz, C.G.; Andersen, K.E. New Aspects in Allergic Contact Dermatitis. Curr. Opin. Allergy Clin. Immunol. 2008, 8, 428–432. [Google Scholar] [CrossRef]
- Nwaru, I.B.; Hickstein, L.; Panesar, S.S.; Muraro, A.; Werfel, T.; Cardona, V.; Dubois, A.E.J.; Halken, S.; Hoffmannsommergruber, K.; Poulsen, L.K.; et al. The Epidemiology of Food Allergy in Europe: A Systematic Review and Meta-Analysis. Allergy 2014, 69, 62–75. [Google Scholar] [CrossRef]
- Panesar, S.S.; Javad, S.; De Silva, D.; Nwaru, B.I.; Hickstein, L.; Muraro, A.; Roberts, G.; Worm, M.; Bilò, M.B.; Cardona, V.; et al. The Epidemiology of Anaphylaxis in Europe: A Systematic Review. Allergy 2013, 68, 1353–1361. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Santamarina, A.; Miranda, J.M.; Mondragon, A.D.C.; Lamas, A.; Cardelle-Cobas, A.; Franco, C.M.; Cepeda, A. Potential Use of Marine Seaweeds as Prebiotics: A Review. Molecules 2020, 25, 1004. [Google Scholar] [CrossRef]
- Lynch, S.V.; Pedersen, O. The Human Intestinal Microbiome in Health and Disease. N. Engl. J. Med. 2016, 375, 2369–2379. [Google Scholar] [CrossRef]
- FAO/WHO Food and Agriculture Organization of the United Nations/World Health Organization. Guidelines for the Evaluation of Probiotics in Food. Report of a Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food; FAO/WHO Food and Agriculture Organization of the United Nations/World Health Organization: Rome, Italy, 2002. [Google Scholar]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics Consensus Statement on the Scope and Appropriate Use of the Term Probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Lopez-Santamarina, A.; Lamas, A.; Mondragón, A.D.C.; Cardelle-Cobas, A.; Regal, P.; Rodriguez-Avila, J.A.; Miranda, J.M.; Franco, C.M.; Cepeda, A. Probiotic Effects against Virus Infections: New Weapons for an Old War. Foods 2021, 10, 130. [Google Scholar] [CrossRef] [PubMed]
- Anatriello, E.; Cunha, M.; Nogueira, J.; Carvalho, J.L.; Sá, A.K.; Miranda, M.; Castro-Faria-Neto, H.; Keller, A.C.; Aimbire, F. Oral Feeding of Lactobacillus Bulgaricus N45.10 Inhibits the Lung Inflammation and Airway Remodeling in Murine Allergic Asthma: Relevance to the Th1/Th2 Cytokines and STAT6/T-bet. Cell. Immunol. 2019, 341. [Google Scholar] [CrossRef]
- Ezendam, J.; De Klerk, A.; Gremmer, E.R.; Van Loveren, H. Effects of Bifidobacterium Animalis Administered during Lactation on Allergic and Autoimmune Responses in Rodents. Clin. Exp. Immunol. 2008, 154, 424–431. [Google Scholar] [CrossRef]
- Plummer, E.L.; Lozinsky, A.C.; Tobin, J.M.; Uebergang, J.B.; Axelrad, C.; Garland, S.M.; Jacobs, S.E.; Tang, M.L.K.; the ProPrems Study Group. Postnatal Probiotics and Allergic Disease in Very Preterm Infants: Sub-Study to the Proprems Randomized Trial. Allergy 2020, 75, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Torow, N.; Hornef, M.W. The Neonatal Window of Opportunity: Setting the Stage for Life-Long Host-Microbial Interaction and Immune Homeostasis. J. Immunol. 2017, 198, 557–563. [Google Scholar] [CrossRef]
- Canani, R.B.; Nocerino, R.; Terrin, G.; Frediani, T.; Lucarelli, S.; Cosenza, L.; Passariello, A.; Leone, L.; Granata, V.; Di Costanzo, M.; et al. Formula Selection for Management of Children with Cow’s Milk Allergy Influences the Rate of Acquisition of Tolerance: A Prospective Multicenter Study. J. Pediatr. 2013, 163, 771–777.e1. [Google Scholar] [CrossRef] [PubMed]
- Kalliomäki, M. The Role of Microbiota in Allergy. Ann Nestlé 2009, 67, 19–26, (English ed.). [Google Scholar] [CrossRef]
- Abrahamsson, T.R.; Jakobsson, H.E.; Andersson, A.F.; Björkstén, B.; Engstrand, L.; Jenmalm, M.C. Low Diversity of the Gut Microbiota in Infants with Atopic Eczema. J. Allergy Clin. Immunol. 2012, 129, 434–440.e2. [Google Scholar] [CrossRef]
- Ismail, I.H.; Oppedisano, F.; Joseph, S.J.; Boyle, R.J.; Licciardi, P.V.; Robins-Browne, R.M.; Tang, M.L. Reduced Gut Microbial Diversity in Early Life Is Associated with Later Development of Eczema but Not Atopy in High-Risk Infants. Pediatr. Allergy Immunol. 2012, 23, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.-Z.; Kondo, S.; Yanagisawa, N.; Takahashi, N.; Odamaki, T.; Iwabuchi, N.; Miyaji, K.; Iwatsuki, K.; Togashi, H.; Enomoto, K. Probiotics in the Treatment of Japanese Cedar Pollinosis: A Double-Blind Placebo-Controlled Trial. Clin. Exp. Allergy 2006, 36, 1425–1435. [Google Scholar] [CrossRef]
- Van Der Aa, L.B.; Van Aalderen, W.M.C.; Heymans, H.S.A.; Smitt, J.H.S.; Nauta, A.J.; Knippels, L.M.J.; Ben Amor, K.; Sprikkelman, A.B. The Synbad Study Group Synbiotics Prevent Asthma-like Symptoms in Infants with Atopic Dermatitis. Allergy 2011, 66, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, A.; West, C.E.; Prescott, S.L.; Jenmalm, M.C. Pre-And Probiotics for Allergy Prevention: Time to Revisit Recommendations? Clin. Exp. Allergy 2016, 46, 1506–1521. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Zhao, F.; Liu, W.; Lv, R.; Khine, W.W.T.; Han, J.; Sun, Z.; Lee, Y.-K.; Zhang, H. Probiotic-Directed Modulation of Gut Microbiota Is Basal Microbiome Dependent. Gut Microbes 2020, 12, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Huo, D.; You, Z.; Peng, Q.; Jiang, S.; Chang, H.; Zhang, J.; Zhang, H. Differential Pattern of Indigenous Microbiome Responses to Probiotic Bifidobacterium Lactis V9 Consumption across Subjects. Food Res. Int. 2020, 136, 109496. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.; Hiel, S.; Neyrinck, A.M.; Le Roy, T.; Pötgens, S.A.; Leyrolle, Q.; Pachikian, B.D.; Gianfrancesco, M.A.; Cani, P.D.; Paquot, N.; et al. Discovery of the Gut Microbial Signature Driving the Efficacy of Prebiotic Intervention in Obese Patients. Gut 2020, 69, 1975–1987. [Google Scholar] [CrossRef]
- Zmora, N.; Zilberman-Schapira, G.; Suez, J.; Mor, U.; Dori-Bachash, M.; Bashiardes, S.; Kotler, E.; Zur, M.; Regev-Lehavi, D.; Brik, R.B.-Z.; et al. Personalized Gut Mucosal Colonization Resistance to Empiric Probiotics Is Associated with Unique Host and Microbiome Features. Cell 2018, 174, 1388–1405.e21. [Google Scholar] [CrossRef] [PubMed]
- Doron, S.; Snydman, D.R. Risk and Safety of Probiotics. Clin. Infect. Dis. 2015, 60, S129–S134. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Luo, X.; Zhang, Q.; He, X.; Zhang, Z.; Wang, X. Bifidobacterium infantis Relieves Allergic Asthma in Mice by Regulating Th1/Th2. Med. Sci. Monit. 2020, 26, e920583-1. [Google Scholar] [CrossRef]
- Del Giudice, M.M.; Indolfi, C.; Capasso, M.; Maiello, N.; Decimo, F.; Ciprandi, G. Bifidobacterium Mixture (B longum BB536, B Infantis M-63, B breve M-16V) Treatment in Children with Seasonal Allergic Rhinitis and Intermittent Asthma. Ital. J. Pediatr. 2017, 43, 25. [Google Scholar] [CrossRef] [PubMed]
- Taniuchi, S.; Hattori, K.; Yamamoto, A.; Sasai, M.; Hatano, Y.; Kojima, T.; Kobayashi, Y.; Iwamoto, H.; Yaeshima, T. Admin-istration of Bifidobacterium to Infants with Atopic Dermatitis: Changes in Fecal Microflora and Clinical Symptoms. J. Appl. Res. 2005, 5, 387–396. [Google Scholar]
- Enomoto, T.; Sowa, M.; Nishimori, K.; Shimazu, S.; Yoshida, A.; Yamada, K.; Furukawa, F.; Nakagawa, T.; Yanagisawa, N.; Iwabuchi, N.; et al. Effects of Bifidobacterial Supplementation to Pregnant Women and Infants in the Prevention of Allergy Development in Infants and on Fecal Microbiota. Allergol. Int. 2014, 63, 575–585. [Google Scholar] [CrossRef]
- Cuello-Garcia, C.A.; Brożek, J.L.; Fiocchi, A.; Pawankar, R.; Yepes-Nuñez, J.J.; Terracciano, L.; Gandhi, S.; Agarwal, A.; Zhang, Y.; Schünemann, H.J. Probiotics for the Prevention of Allergy: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Allergy Clin. Immunol. 2015, 136, 952–961. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.-Q.; Hu, H.-J.; Liu, C.-Y.; Zhang, Q.; Shakya, S.; Li, Z.-Y. Probiotics for Prevention of Atopy and Food Hypersensitivity in Early Childhood. Medicine 2016, 95, e2562. [Google Scholar] [CrossRef]
- Tang, M.L.; Lahtinen, S.J.; Boyle, R.J. Probiotics and Prebiotics: Clinical Effects in Allergic Disease. Curr. Opin. Pediatr. 2010, 22, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; An, J.; Shimada, T.; Liu, S.; Maeyama, K. Oral Administration of Enterococcus Faecalis FK-23 Suppresses Th17 Cell Development and Attenuates Allergic Airway Responses in Mice. Int. J. Mol. Med. 2012, 30, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Won, T.J.; Kim, B.; Lee, Y.; Bang, J.S.; Oh, E.S.; Yoo, J.-S.; Hyung, K.E.; Yoon, J.; Hwang, S.; Park, E.S.; et al. Therapeutic Potential of Lactobacillus Plantarum CJLP133 for House-Dust Mite-Induced Dermatitis in NC/Nga Mice. Cell. Immunol. 2012, 277, 49–57. [Google Scholar] [CrossRef]
- Choi, C.-Y.; Kim, Y.-H.; Oh, S.; Lee, H.; Kim, J.; Park, S.; Kim, H.; Lee, S.; Chun, T. Anti-inflammatory Potential of a Heat-Killed Lactobacillus Strain Isolated from Kimchi on House Dust Mite-Induced Atopic Dermatitis in NC/Nga Mice. J. Appl. Microbiol. 2017, 123, 535–543. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kwon, J.H.; Ahn, S.H.; Lee, S.I.; Han, Y.S.; Choi, Y.O.; Lee, S.Y.; Ahn, K.M.; Ji, G.E. Effect of Probiotic MIX (Bifidobacterium bifidum, Bifidobacterium lactis, Lactobacillus acidophilus) in the Primary Prevention of Eczema: A Double-Blind, Randomized, Placebo-Controlled Trial. Pediatr. Allergy Immunol. 2010, 21, e386–e393. [Google Scholar] [CrossRef] [PubMed]
- Viljanen, M.; Savilahti, E.; Haahtela, T.; Juntunen-Backman, K.; Korpela, R.; Poussa, T.; Tuure, T.; Kuitunen, M. Probiotics in the Treatment of Atopic Eczema/Dermatitis Syndrome in Infants: A Double-Blind Placebo-Controlled Trial. Allergy 2005, 60, 494–500. [Google Scholar] [CrossRef]
- Simpson, M.R.; Dotterud, C.K.; Storrø, O.; Johnsen, R.; Øien, T. Perinatal Probiotic Supplementation in the Prevention of Allergy Related Disease: 6 Year Follow up of a Randomised Controlled Trial. BMC Dermatol. 2015, 15, 1–8. [Google Scholar] [CrossRef]
- Loo, E.X.; Llanora, G.V.; Lu, Q.; Aw, M.M.; Lee, B.W.; Shek, L.P. Supplementation with Probiotics in the First 6 Months of Life Did Not Protect against Eczema and Allergy in At-Risk Asian Infants: A 5-Year Follow-Up. Int. Arch. Allergy Immunol. 2014, 163, 25–28. [Google Scholar] [CrossRef]
- Isolauri, E.; Arvola, T.; Sütas, Y.; Moilanen, E.; Salminen, S. Probiotics in the Management of Atopic Eczema. Clin. Exp. Allergy 2000, 30, 1605–1610. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeldt, V.; Benfeldt, E.; Nielsen, S.D.; Michaelsen, K.F.; Jeppesen, D.L.; Valerius, N.H.; Paerregaard, A. Effect of probiotic Lactobacillus strains in children with atopic dermatitis. J. Allergy Clin. Immunol. 2003, 111, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Weston, S.; Halbert, A.; Richmond, P.; Prescott, S.L. Effects of Probiotics on Atopic Dermatitis: A Randomised Controlled Trial. Arch. Dis. Child. 2005, 90, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Sistek, D.; Kelly, R.; Wickens, K.; Stanley, T.; Fitzharris, P.; Crane, J. Is the Effect of Probiotics on Atopic Dermatitis Confined to Food Sensitized Children? Clin. Exp. Allergy 2006, 36, 629–633. [Google Scholar] [CrossRef]
- Brouwer, M.L.; Wolt-Plompen, S.A.A.; Dubois, A.E.J.; Van Der Heide, S.; Jansen, D.F.; Hoijer, M.A.; Kauffman, H.F.; Duiverman, E.J. No Effects of Probiotics on Atopic Dermatitis in Infancy: A Randomized Placebo-Controlled Trial. Clin. Exp. Allergy 2006, 36, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.L.; Dunstan, J.A.; Prescott, S.L. Probiotic Supplementation for the First 6 Months of Life Fails to Reduce the Risk of Atopic Dermatitis and Increases the Risk of Al-Lergen Sensitization in High-Risk Children: A Randomized Controlled Trial. J. Allergy Clin. Immunol. 2007, 119, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Hol, J.; Van Leer, E.H.; Schuurman, B.E.E.; De Ruiter, L.F.; Samsom, J.N.; Hop, W.; Neijens, H.J.; De Jongste, J.C.; Nieuwenhuis, E.E. The Acquisition of Tolerance toward Cow’s Milk through Probiotic Supplementation: A Randomized, Controlled Trial. J. Allergy Clin. Immunol. 2008, 121, 1448–1454. [Google Scholar] [CrossRef] [PubMed]
- Soh, S.E.; Aw, M.; Gerez, I.; Chong, Y.S.; Rauff, M.; Ng, Y.P.M.; Wong, H.B.; Pai, N.; Lee, B.W.; Shek, L.P.-C. Probiotic Supplementation in the First 6 Months of Life in at Risk Asian Infants—Effects on Eczema and Atopic Sensitization at the Age of 1 Year. Clin. Exp. Allergy 2009, 39, 571–578. [Google Scholar] [CrossRef] [PubMed]
- West, C.E.; Hammarström, M.-L.; Hernell, O. Probiotics during Weaning Reduce the Incidence of Eczema. Pediatr. Allergy Immunol. 2009, 20, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Dotterud, C.K.; Storrø, O.; Johnsen, R.; Øien, T. Probiotics in Pregnant Women to Prevent Allergic Disease: A Randomized, Double-Blind Trial. Br. J. Dermatol. 2010, 163, 616–623. [Google Scholar] [CrossRef]
- Nermes, M.; Kantele, J.M.; Atosuo, T.J.; Salminen, S.; Isolauri, E. Interaction of Orally Administered Lactobacillus Rhamnosus GG with Skin and Gut Microbiota and Humoral Immunity in Infants with Atopic Dermatitis. Clin. Exp. Allergy 2010, 41, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.-J.; Wang, J. Children with Atopic Dermatitis Show Clinical Improvement Afterlactobacillus exposure. Clin. Exp. Allergy 2015, 45, 779–787. [Google Scholar] [CrossRef]
- Cabana, M.D.; Mckean, M.; Caughey, A.B.; Fong, L.; Lynch, S.; Wong, A.; Leong, R.; Boushey, H.A.; Hilton, J.F. Early Probiotic Supplementation for Eczema and Asthma Prevention: A Randomized Controlled Trial. Pediatrics 2017, 140, e20163000. [Google Scholar] [CrossRef] [PubMed]
- Navarro-López, V.; Ramírez-Boscá, A.; Ramón-Vidal, D.; Ruzafa-Costas, B.; Genovés-Martínez, S.; Chenoll-Cuadros, E.; Carrión-Gutiérrez, M.; De La Parte, J.H.; Prieto-Merino, D.; Codoñer-Cortés, F.M. Effect of Oral Administration of a Mixture of Probiotic Strains on SCORAD Index and Use of Topical Steroids in Young Patients with Moderate Atopic Dermatitis. JAMA Dermatol. 2018, 154, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Kukkonen, K.; Savilahti, E.; Haahtela, T.; Juntunen-Backman, K.; Korpela, R.; Poussa, T.; Tuure, T.; Kuitunen, M. Probiotics and Prebiotic Galacto-Oligosaccharides in the Prevention of Allergic Diseases: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Allergy Clin. Immunol. 2007, 119, 192–198. [Google Scholar] [CrossRef]
- Huurre, A.; Laitinen, K.; Rautava, S.; Korkeamäki, M.; Isolauri, E. Impact of Maternal Atopy and Probiotic Supplementation during Pregnancy on Infant Sensitization: A Double-Blind Placebo-Controlled Study. Clin. Exp. Allergy 2008, 38, 1342–1348. [Google Scholar] [CrossRef]
- Wickens, K.; Black, P.N.; Stanley, T.V.; Mitchell, E.; Fitzharris, P.; Tannock, G.W.; Purdie, G.; Crane, J. A Differential Effect of 2 Probiotics in the Prevention of Eczema and Atopy: A Double-Blind, Randomized, Placebo-Controlled Trial. J. Allergy Clin. Immunol. 2008, 122, 788–794. [Google Scholar] [CrossRef]
- Kopp, M.V.; Hennemuth, I.; Heinzmann, A.; Urbanek, R. Randomized, Double-Blind, Placebo-Controlled Trial of Probiotics for Primary Prevention: No Clinical Effects of Lactobacillus GG Supplementation. Pediatrics 2008, 121, e850–e856. [Google Scholar] [CrossRef]
- Niers, L.; Martin, R.; Rijkers, G.; Sengers, F.; Timmerman, H.; Van Uden, N.; Smidt, H.; Kimpen, J.; Hoekstra, M. The Effects of Selected Probiotic Strains on the Development of Eczema (The PandA Study). Allergy 2009, 64, 1349–1358. [Google Scholar] [CrossRef] [PubMed]
- Koyama, T.; Kirjavainen, P.V.; Fisher, C.; Anukam, K.; Summers, K.; Hekmat, S.; Reid, G. Development and Pilot Evaluation of a Novel Probiotic Mixture for the Management of Seasonal Allergic Rhinitis. Can. J. Microbiol. 2010, 56, 730–738. [Google Scholar] [CrossRef] [PubMed]
- Boyle, R.J.; Ismail, I.H.; Kivivuori, S.; Licciardi, P.V.; Robins-Browne, R.M.; Mah, L.-J.; Axelrad, C.; Moore, S.; Donath, S.; Carlin, J.B.; et al. Lactobacillus GG Treatment during Pregnancy for the Prevention of Eczema: A Randomized Controlled Trial. Allergy 2010, 66, 509–516. [Google Scholar] [CrossRef]
- Han, Y.; Kim, B.; Ban, J.; Lee, J.; Kim, B.J.; Choi, B.S.; Hwang, S.; Ahn, K.; Kim, J. A Randomized Trial Oflactobacillus plantarum CJLP133 for the Treatment of Atopic Dermatitis. Pediatr. Allergy Immunol. 2012, 23, 667–673. [Google Scholar] [CrossRef]
- Rautava, S.; Kainonen, E.; Salminen, S.; Isolauri, E. Maternal Probiotic Supplementation during Pregnancy and Breast-Feeding Reduces the Risk of Eczema in the Infant. J. Allergy Clin. Immunol. 2012, 130, 1355–1360. [Google Scholar] [CrossRef] [PubMed]
- Ou, C.-Y.; Kuo, H.-C.; Wang, L.; Hsu, T.-Y.; Chuang, H.; Liu, C.-A.; Chang, J.-C.; Yu, H.-R.; Yang, K.D. Prenatal and Postnatal Probiotics Reduces Maternal but Not Childhood Allergic Diseases: A Randomized, Double-Blind, Placebo-Controlled Trial. Clin. Exp. Allergy 2012, 42, 1386–1396. [Google Scholar] [CrossRef]
- Inoue, Y.; Kambara, T.; Murata, N.; Komori-Yamaguchi, J.; Matsukura, S.; Takahashi, Y.; Ikezawa, Z.; Aihara, M. Effects of Oral Administration of Lactobacillus acidophilus L-92 on the Symptoms and Serum Cytokines of Atopic Dermatitis in Japanese Adults: A Double-Blind, Randomized, Clinical Trial. Int. Arch. Allergy Immunol. 2014, 165, 247–254. [Google Scholar] [CrossRef]
- Rø, A.D.B.; Rø, T.B.; Storrø, O.; Johnsen, R.; Videm, V.; Øien, T.; Simpson, M.R. Reduced Th22 Cell Proportion and Prevention of Atopic Dermatitis in Infants Following Maternal Probiotic Supplementation. Clin. Exp. Allergy 2017, 47, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- Dennis-Wall, J.C.; Culpepper, T.; Nives, C., Jr.; Rowe, C.C.; Burns, A.M.; Rusch, C.T.; Federico, A.; Ukhanova, M.; Waugh, S.; Mai, V.; et al. Probiotics (Lactobacillus gasseri KS-13, Bifidobacterium bifidum G9-1, and Bifidobacterium longum MM-2) Improve Rhinoconjunctivitis-Specific Quality of Life in Individuals with Seasonal Allergies: A Double-Blind, Placebo-Controlled, Randomized Trial. Am. J. Clin. Nutr. 2017, 105, 758–767. [Google Scholar] [CrossRef]
- Prakoeswa, C.; Herwanto, N.; Prameswari, R.; Astari, L.; Sawitri, S.; Hidayati, A.; Indramaya, D.; Kusumowidagdo, E.; Surono, I. Lactobacillus plantarum IS-10506 Supplementation Reduced SCORAD in Children with Atopic Dermatitis. Benef. Microbes 2017, 8, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.-L.; Tang, X.-G.; Qu, F.; Zheng, Y.; Zhang, W.-H.; Diao, Y.-Q. Bifidobacterium May Benefit the Prevention of Necrotizing Enterocolitis in Preterm Infants: A Systematic Review and Meta-Analysis. Int. J. Surg. 2019, 61, 17–25. [Google Scholar] [CrossRef]
- Bertelli, C.; Pillonel, T.; Torregrossa, A.; Prod’Hom, G.; Fischer, C.J.; Greub, G.; Giannoni, E. Bifidobacterium longum Bacteremia in Preterm Infants Receiving Probiotics. Clin. Infect. Dis. 2015, 60, 924–927. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.A. Probiotic Safety—No Guarantees. JAMA Intern. Med. 2018, 178, 1577. [Google Scholar] [CrossRef]
- Vallabhaneni, S.; Walker, T.A.; Lockhart, S.R.; Ng, D.; Chiller, T.; Melchreit, R.; Brandt, M.E.; Smith, R.M. Fatal Gastrointestinal Mucormycosis in a Premature Infant Associated with a Contaminated Dietary Supplement—Connecticut, 2014. MMWR. Morb. Mortal. Wkly. Rep. 2015, 64, 155–156. [Google Scholar]
- Durazzo, A.; Nazhand, A.; Lucarini, M.; Atanasov, A.G.; Souto, E.B.; Novellino, E.; Capasso, R.; Santini, A. An Updated Overview on Nanonutraceuticals: Focus on Nanoprebiotics and Nanoprobiotics. Int. J. Mol. Sci. 2020, 21, 2285. [Google Scholar] [CrossRef]
- Gong, S.; Ji, X.; Su, J.; Wang, Y.; Yan, X.; Wang, G.; Xiao, B.; Dong, H.; Xiang, X.; Liu, S. Yeast Fermentate Prebiotic Ameliorates Allergic Asthma, Associating with Inhibiting Inflammation and Reducing Oxidative Stress Level through Suppressing Autophagy. Mediat. Inflamm. 2021, 2021, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Osborn, A.D.; Sinn, J.K.H. Prebiotics in Infants for Prevention of Allergy. Cochrane Database Syst. Rev. 2013, 28, CD006474. [Google Scholar] [CrossRef]
- Scalabrin, D.; Harris, C.; Johnston, W.H.; Berseth, C.L. Long-Term Safety Assessment in Children Who Received Hydrolyzed Protein Formulas with Lactobacillus Rhamnosus GG: A 5-Year Follow-Up. Eur. J. Nucl. Med. Mol. Imaging 2017, 176, 217–224. [Google Scholar] [CrossRef] [PubMed]
| Type of Study | Probiotic | Dosage and Time of Exposure | Allergic Disease | Main Findings | Reference |
|---|---|---|---|---|---|
| Randomized, double-blind study with 27 infants with atopic disease | Bifidobacterium lactis Bb-12 and Lactobacillus rhamnosus GG (LGG) | Oral administration of 3 × 108 CFU of LGG and 109 CFU of B. lactis for 4 weeks | Atopic eczema | After 2 months, a significant improvement in the skin condition occurred in the probiotic group. The Scoring Atopic Dermatitis (SCORAD) and concentration of soluble CD4+ decreased in the probiotic groups | [80] |
| Double-blind, placebo-controlled, crossover study with 58 children | Lactobacillus rhamnosus 19070-2 and Lactobacillus reuteri DSM 122460 | A dose of 1010 CFU twice daily for 6 weeks | Atopic dermatitis | The duration of eczema decreased during probiotic administration. The treatment response was more pronounced in allergic patients, and the SCORAD score decreased | [81] |
| Randomized, double-blind, placebo-controlled study with 230 infants with suspected cow’s milk allergy (CMA) | LGG, L. rhamnosus LC705, B. breve Bb99 and Propionibacterium freudenreichii ssp shermanii JS | Oral administration of LGG (5 × 109 CFU) or a mixture of LGG (5 × 109 CFU), L. rhamnosus LC705 (5 × 109), B. breve Bb99 (2 × 108), and Propionibacterium freudenreichii spp. shermanii JS (2 × 109) twice daily for 4 weeks. | Atopic dermatitis related to cow’s milk allergy | Treatment with LGG may alleviate symptoms of atopic eczema and/or dermatitis syndrome in IgE-sensitized infants | [77] |
| Randomized, double-blind, placebo-controlled study with 56 children | Lactobacillus fermentum VRI-033 PCC | 1 × 109 CFU twice daily for 8 weeks | Atopic dermatitis | A reduction in the SCORAD index was seen in the probiotic-treated group. At the end of the study, more children treated with this probiotic had milder atopic dermatitis | [82] |
| Randomized, placebo-controlled study with 59 children with AD | L. rhamnosus and B. lactis | A dose of 2 × 1010 CFU daily for 12 weeks | Atopic dermatitis | A combination of L. rhamnosus and B. lactis improved atopic dermatitis only in food-sensitized children | [83] |
| Randomized, double-blind, placebo-controlled study with 50 infants | L. rhamnosus Lrh and LGG | Administering 1, 5, 25 and 125 mL of cow’s milk formula at 30 min intervals (5 × 109 CFU/mL formula) | Atopic dermatitis related to cow’s milk allergy | No clear effects were seen on the SCORAD, sensitization, inflammatory parameters, or cytokine production | [84] |
| Randomized trial with newborns of 231 women with allergies | Lactobacillus acidophilus LAVRI-A1 | 3 × 109 CFU/day for the first 6 months of life | Atopic dermatitis | Atopic dermatitis rates were similar in the probiotic and placebo groups. At 12 months, the rate of sensitization was significantly higher in the probiotic group | [85] |
| Randomized, double-blind, placebo-controlled study with 193 infants diagnosed with cow’s milk allergy (CMA) | Lactobacillus casei CRL431 and Bifidobacterium lactis BB-12 | 107 CFU/g for each of the probiotic bacteria for 6 months | Cow’s milk allergy (CMA) | Supplementation with Lactobacillus and Bifidobacterium in an extensively hydrolyzed formula did not accelerate cow’s milk tolerance in infants with CMA | [86] |
| Double-blind, placebo-controlled, randomized trial with 253 infants | B. longum BL999 and L. rhamnosus LPR | Oral supplementation with 1 × 107 CFU/g/day of B. longum and 2 × 107 CFU/g/day of L. rhamnosus for the first 6 months | Atopy and eczema | No significant effect on the prevention of eczema or allergen sensitization in the first year of life | [87] |
| Double-blind, placebo-controlled, randomized trial with 179 infants | Lactobacillus F19 | 1 × 108 CFU/day for 4–6 months | Eczema | The cumulative incidence of eczema at 13 months was lower in the probiotic group. At 13 months, the INF-y/IL-4 ratio was higher in the probiotic group. No differences in serum concentrations of IgE | [88] |
| Randomized, double-blind trial of children from 415 mothers | LGG, B. animalis ssp. lactis Bb-12 and L. acidophilus La-5 | Milk contained 5 × 1010 CFU/day of L. rhamnosus and Bb-12. 5 × 109 CFU of L. acidophilus for 4 months, from 36 weeks of gestation to 3 months postnatally | Atopic dermatitis and asthma | In the probiotic group, the cumulative incidence of atopic dermatitis was reduced but there was no effect on sensitization | [89] |
| Randomized, double-blind study with 39 infants with AD | LGG | A daily intake of 3.4 × 109 CFU for 3 months | Atopic dermatitis | The proportions of IgA- and IgM-secreting cells decreased in the probiotic group, and the proportions of CD191+ and CD27+ B cells increased | [90] |
| Randomized, placebo-controlled trial with 606 newborns | Escherichia coli DSM 17252 and Enterococcus faecalis DSM 1644 | Oral bacteria lysate containing heat-killed nonpathogenic 1.5–4.5 × 107 bacteria/mL (3 × 0.7 mL/day) | Atopic dermatitis | A significant effect was observed in a subgroup of the probiotic group with single heredity for atopy, which was most pronounced for infants with atopic fathers | [8] |
| Double-blind, placebo-controlled, randomized parallel study with 100 children | Mixture of L. casei, L. rhamnosus, L. plantarum, and B. lactis | Oral administration at 2 × 109 CFU in each strain, twice daily for 6 weeks | Atopic dermatitis | The probiotic mixture did not suppress the growth of other strains, but no differences in clinical improvement were seen between the treated and placebo groups | [41] |
| Double-blind, prospective, randomized, placebo-controlled study with 2020 children | L. paracasei GMNL-133 and/or L. fermentum GM090 | 2 × 109 CFU/day of L. paracasei, L. plantarum, or 4 × 109 CFU of a mixture for 3 months | Atopic dermatitis | Children given either probiotics alone or a mixture of both showed a decrease in the severity of atopic dermatitis scores. Lower test scores were also recorded for people with skin diseases. IgE, TNF-α, and INF-y increased in the probiotic group | [91] |
| Randomized, controlled, double-blind study with 159 newborns | LGG | 1010 CFU/day for the first 6 months of life | Eczema and asthma | The estimated cumulative incidence of eczema and asthma was lower in the probiotic group at 2 years of age | [92] |
| Prospective, double-blind, placebo-controlled, randomized study with 40 children | B. longum BB536, Bifidobacterium infantis M-63, and B. breve M-16 V | Oral supplementation containing B. longum BB536 (3 × 109 CFU), B. infantis M-63 (1 × 109 CFU), and B. breve M-16 V (1 × 109 CFU) as powder in a 3 mg sachet. Administered every day for 8 weeks | Seasonal allergic rhinitis and intermittent asthma | A significant improvement of symptoms and quality of life in the probiotic group | [67] |
| Double-blind, 2-arm, placebo-controlled study with 50 children with AD | B. lactis CECT 8145, B. longum CECT7347, and L. casei CECT 9104 | 109 CFU/day of a mixture of the 3 probiotic strains | Atopic dermatitis | A reduction in IL-4, IL-5, and IL-13 and a decreased activity of Th2 in the probiotic group. The SCORAD index and use of corticosteroids were also reduced in the probiotic group | [93] |
| A multi-center, double-blind, placebo-controlled, randomized trial with 1099 very preterm infants | B. infantis BB-02, Streptococcus thermophilus TH-4, and B. lactis BB-12 | A combination of B. infantis BB-02 (300 × 106 CFU), S. thermophilus TH-4 (350 × 106 CFU), and B. lactis BB-12 (350 × 106). Total: 1 × 109 CFU per 1.5 g in a powder once daily, until discharged from hospital or term-corrected age | Eczema, atopic sensitization, food allergy, and wheezing | There was no difference in eczema incidence between the two groups. Additionally, the incidence of atopic eczema, food allergy, wheezing, and atopic sensitization were similar in both groups | [52] |
| Type of Study | Probiotic | Dosage and Time of Exposure | Allergic Disease | Main Findings | Reference |
|---|---|---|---|---|---|
| Double-blind, randomized, placebo-controlled study with 159 mothers with allergic diseases and their infants | Lactobacillus rhamnosus GG (LGG) | 1 × 1010 CFU daily for 2–4 weeks | Atopic eczema, asthma, and allergic rhinitis | The incidence of eczema in the probiotic group was halved. The concentrations of total immunoglobulin (Ig) E and positive reactions in skin-pick tests were similar in both groups | [55] |
| Randomized, double-blind, placebo-controlled trial with 44 adults | Bifidobacterium longum BB536 | 5 × 1010 CFU twice daily for 13 weeks | Japanese cedar pollinosis (JCPsis) | B. longum reduced severe symptoms and medication and decreased nasal blockage in rhinorrhea, and composite scores. Improvements in all symptoms | [58] |
| Randomized trial with newborns of 231 women with allergy | Lactobacillus acidophilus LAVRI-A1 | 3 × 109 CFU/day for the first 6 months of life | Atopic dermatitis | Atopic dermatitis rates were similar in the probiotic and placebo groups. At 12 months, the rate of sensitization was significantly higher in the probiotic group | [85] |
| Double-blind, randomized, placebo-controlled study with 232 mothers | L. reuteri ATCC 55730 | 1 × 108 CFU/day from gestational week 36 until delivery. Their babies then continued with the same product from birth until 12 months | Eczema | The cumulative incidence of eczema was similar in the two groups. The probiotic group had less IgE-associated, and skin prick test reactivity was also less common | [56] |
| Randomized, double-blind, placebo-controlled study with 2 parallel groups of 1223 pregnant women | LGG, L. rhamnosus LC705, Bifidobacterium breve Bb99, and P. freudenreichii spp. shermanii JS | Oral administration of LGG (5 × 109 CFU), L. rhamnosus LC705 (5 × 109), B. breve Bb99 (2 × 108), and Propionibacterium freudenreichii spp. shermanii JS (2 × 108) twice daily for 2 to 4 weeks before delivery. Their infants received the same probiotics (+ prebiotics) once daily for the first 6 months | Food allergy, eczema, asthma, and allergic rhinitis | Administration of these probiotics significantly prevented eczema and atopic eczema | [94] |
| Placebo-controlled, double-blind study with 171 mother–infant pairs | LGG and B. lactis Bb12 | 1010 CFU/day of each probiotic from the first trimester of pregnancy to the end of exclusive breastfeeding | Atopy (eczema) | The concentration of TGF-b2 was higher in colostrum from dams supplemented with probiotics. This supplementation produced a protective effect against sensitization in infants at a high hereditary risk | [95] |
| Two-center, double-blind, randomized, placebo-controlled trial with 512 pregnant women and 474 infants | L. rhamnosus HN001 and Bifidobacterium animalis spp. lactis HN019 | Two treatment groups: 6 × 109 CFU/d of L. rhamnosus or 9 × 109 CFU/d of B. animalis from 35 weeks of gestation until 6 months in pregnant women and from birth to 2 years in infants | Eczema and atopy | Supplementation with only L. rhamnosus reduced the cumulative prevalence of eczema, but not atopy, by 2 years | [96] |
| Double-blind, placebo-controlled, prospective study with 105 pregnant women | LGG | 5 × 109 CFU twice daily. Started 4–6 weeks before expected delivery until a postnatal period of 6 months | Atopic dermatitis | There was no significant difference between the probiotic group and placebo groups | [97] |
| Randomized, double-blind, placebo-controlled trial with 112 pregnant women | Bifidobacterium bifidum BGN4, B. lactis AD011, and L. acidophilus AD031 | Mixture of probiotics (1.6 × 109 CFU/day of each probiotic) for 4–8 weeks before delivery until 6 months after delivery | Eczema | The prevalence of eczema in the probiotic-added group was lower. The cumulative incidence of eczema during the first 12 months was reduced in the probiotic group. No difference in the serum total IgE level or sensitization to food allergens | [7] |
| Double-blind, randomized, placebo-controlled study with 156 pregnant women | B. bifidum W23 and B. lactis W52 | 1 × 109 CFU/day of each strain during the last 6 weeks of pregnancy and postnatally for 12 months to their offspring | Eczema | The prevalence of eczema during the first 3 months of life was significantly lower in the probiotic group. The cumulative incidence was also lower in the probiotic group | [98] |
| Randomized, double-blind trial of children from 415 mothers | LGG, B. animalis ssp. lactis Bb-12, and L. acidophilus La-5 | Milk contained 5 × 1010 CFU/day of L. rhamnosus and Bb-12. 5 × 109 CFU of L. acidophilus for 4 months, from 36 weeks of gestation to 3 months postnatally | Atopic dermatitis and asthma | In the probiotic group, the cumulative incidence of atopic dermatitis was reduced, but there was no effect on sensitization | [89] |
| Double-blind, placebo-controlled study with 36 subjects with allergic rhinitis | L. rhamnosus GR-1 and Bifidobacterium adolescentis 7007-05 | Added L. rhamnosus at 4% and B. adolescentis at 10% of final milk volume | Rhinitis | Serum IL-10 and IL-12 levels were increased in the probiotic group at the end of the grass pollen season. Additionally, the serum TFG-β levels were higher during the ragweed season | [99] |
| Randomized controlled trial with 250 pregnant women | LGG | 1.8 × 1010 CFU/day from 36 weeks of gestation until delivery | Eczema | Prenatal probiotic treatment was only associated with decreased breast milk soluble CD14 and IgA levels | [100] |
| Randomized, double-blind, placebo-controlled study | Lactobacillus plantarum CJLP133 | 0.5 × 1010 CFU twice a day for 12 weeks | Atopic dermatitis | SCORAD scores were lower with probiotic administration. Additionally, the total eosinophil counts were significantly lower, and the logarithmic IFN-c and IL-4 concentrations were decreased | [101] |
| Parallel, double-blind, placebo-controlled study of 241 mother–infant pairs | L. rhamnosus LPR, B. longum BL999, and Lactobacillus paracasei ST11 | Combination of L. rhamnosus and B. longum or L. paracasei and B. longum. Daily dose for each probiotic (1 × 109 CFU) for 2 months before delivery and during the first 2 months of breastfeeding. The infants were followed for 24 months | Eczema | The risk of eczema was reduced in infants of mothers receiving combinations of probiotics during the first 24 months | [102] |
| Prospective, double-blind, placebo-controlled study with 191 pregnant women | LGG | 1 × 1010 CFU/day from the second trimester of pregnancy | Atopic diseases | No significant effects of prenatal and postnatal probiotic supplementation on sensitization, development of allergic diseases, and maternal IgE levels. The allergic symptoms improved in the probiotic group | [103] |
| Double-blind, parallel-group, placebo-controlled comparison with 49 AD patients | L. acidophilus L-92 | 20.7 mg/day for 8 weeks | Atopic dermatitis | This probiotic contributes to the suppression of Th-2-dominant inflammation and reduces atopic dermatitis symptoms in adults | [104] |
| Randomized controlled study with 415 pregnant women | LGG, B. animalis spp. Lactis Bb-12, and L. acidophilus La-5 | Daily dose of LGG (5 × 1010 CFU), B. animalis spp. Lactis Bb-12 (5 × 1010 CFU), and L. acidophilus La-5 (5 × 109 CFU) from 36 weeks of gestation to 3 months postnatal | Atopic dermatitis | Supplementation with a combination of these probiotics reduced the proportion of Th22 cells. However, the proportion of Tregs, Th1, Th2, and Th17 cells and the Th1/Th2 ratio in the offspring were not affected | [105] |
| Double-blind, placebo-controlled, parallel, randomized clinical study with 173 participants | Lactobacillus gasseri KS-13, B. bifidum G9-1, and B. longum MM-2 | Each capsule (350 mg) contained L. gasseri KS-13 (1.2 billion CFU), B. bifidum G9-1 (0.15 billion), and B. longum MM-2 (0.15 billion) | Rhinoconjunctivitis-specific | An improvement in the rhinoconjunctivitis-specific quality of life during allergy season was seen in the probiotic group | [106] |
| Randomized, double-blind, placebo-controlled study with 22 AD subjects | L. plantarum IS-10506 | 1010 CFU/day for 12 weeks | Atopic dermatitis | Reduction in clinical symptoms in AD children and decreased SCORAD and levels of serum IgE, IL-4, and IL-17. The probiotic acted through the downregulation of the Th2-adaptive immune response | [107] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopez-Santamarina, A.; Gonzalez, E.G.; Lamas, A.; Mondragon, A.d.C.; Regal, P.; Miranda, J.M. Probiotics as a Possible Strategy for the Prevention and Treatment of Allergies. A Narrative Review. Foods 2021, 10, 701. https://doi.org/10.3390/foods10040701
Lopez-Santamarina A, Gonzalez EG, Lamas A, Mondragon AdC, Regal P, Miranda JM. Probiotics as a Possible Strategy for the Prevention and Treatment of Allergies. A Narrative Review. Foods. 2021; 10(4):701. https://doi.org/10.3390/foods10040701
Chicago/Turabian StyleLopez-Santamarina, Aroa, Esther Gonzalez Gonzalez, Alexandre Lamas, Alicia del Carmen Mondragon, Patricia Regal, and Jose Manuel Miranda. 2021. "Probiotics as a Possible Strategy for the Prevention and Treatment of Allergies. A Narrative Review" Foods 10, no. 4: 701. https://doi.org/10.3390/foods10040701
APA StyleLopez-Santamarina, A., Gonzalez, E. G., Lamas, A., Mondragon, A. d. C., Regal, P., & Miranda, J. M. (2021). Probiotics as a Possible Strategy for the Prevention and Treatment of Allergies. A Narrative Review. Foods, 10(4), 701. https://doi.org/10.3390/foods10040701









