Soft Mango Firmness Assessment Based on Rayleigh Waves Generated by a Laser-Induced Plasma Shock Wave Technique
Abstract
1. Introduction
2. Materials and Methods
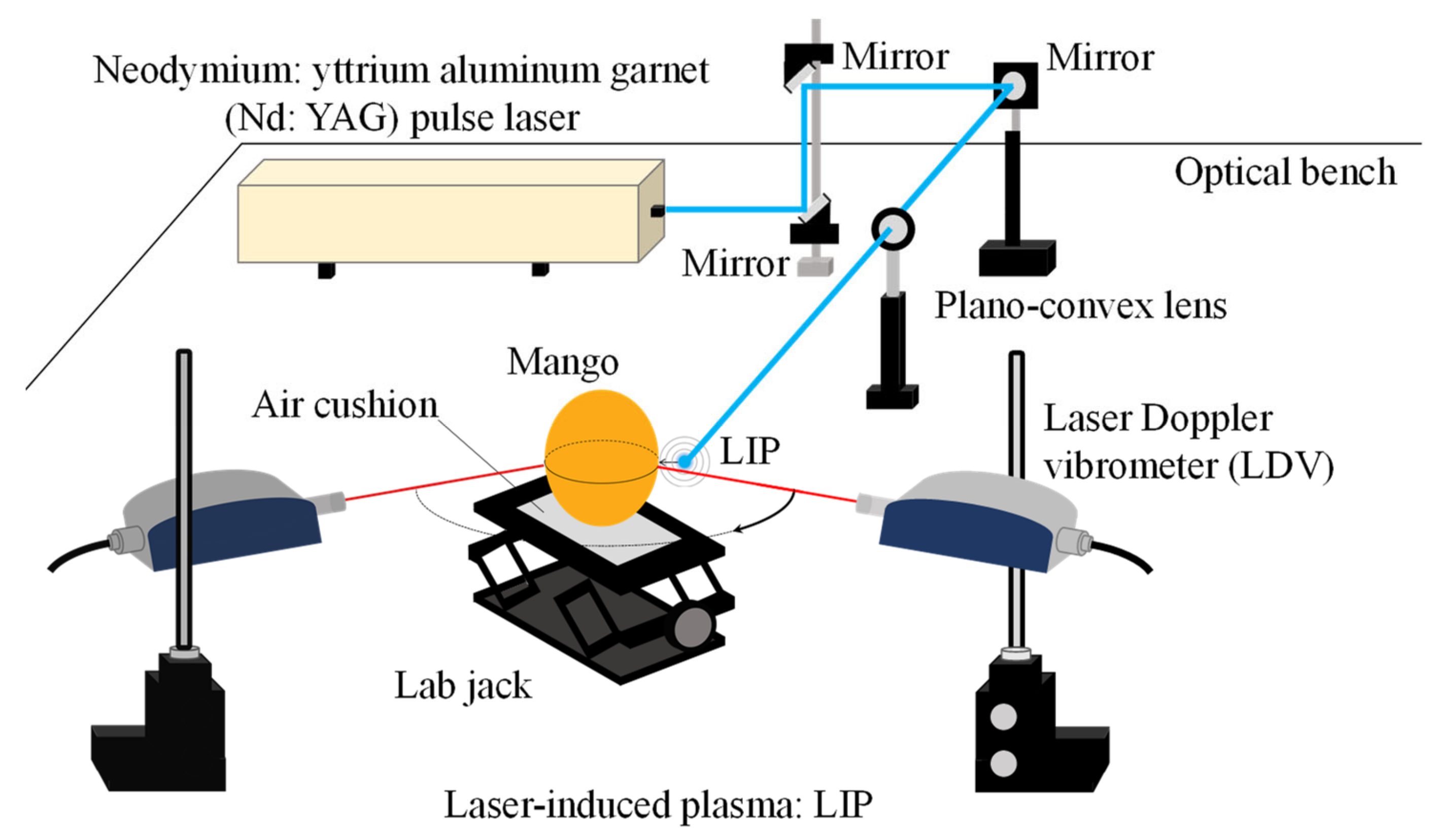
2.1. LIP Shock Wave Excitation Technique
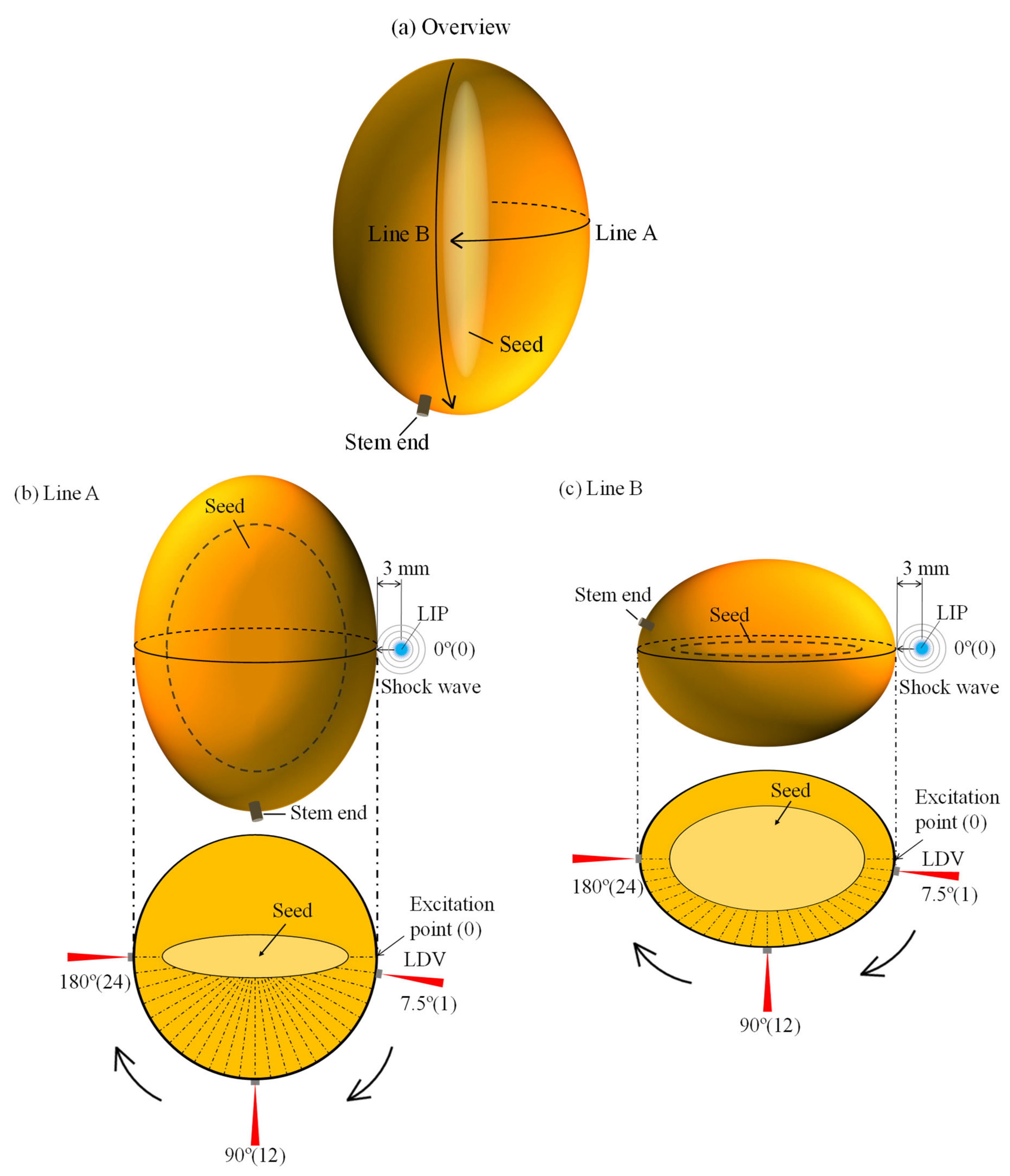
2.2. Storage Time and Measurement Paths of Mangoes
3. Results and Discussion
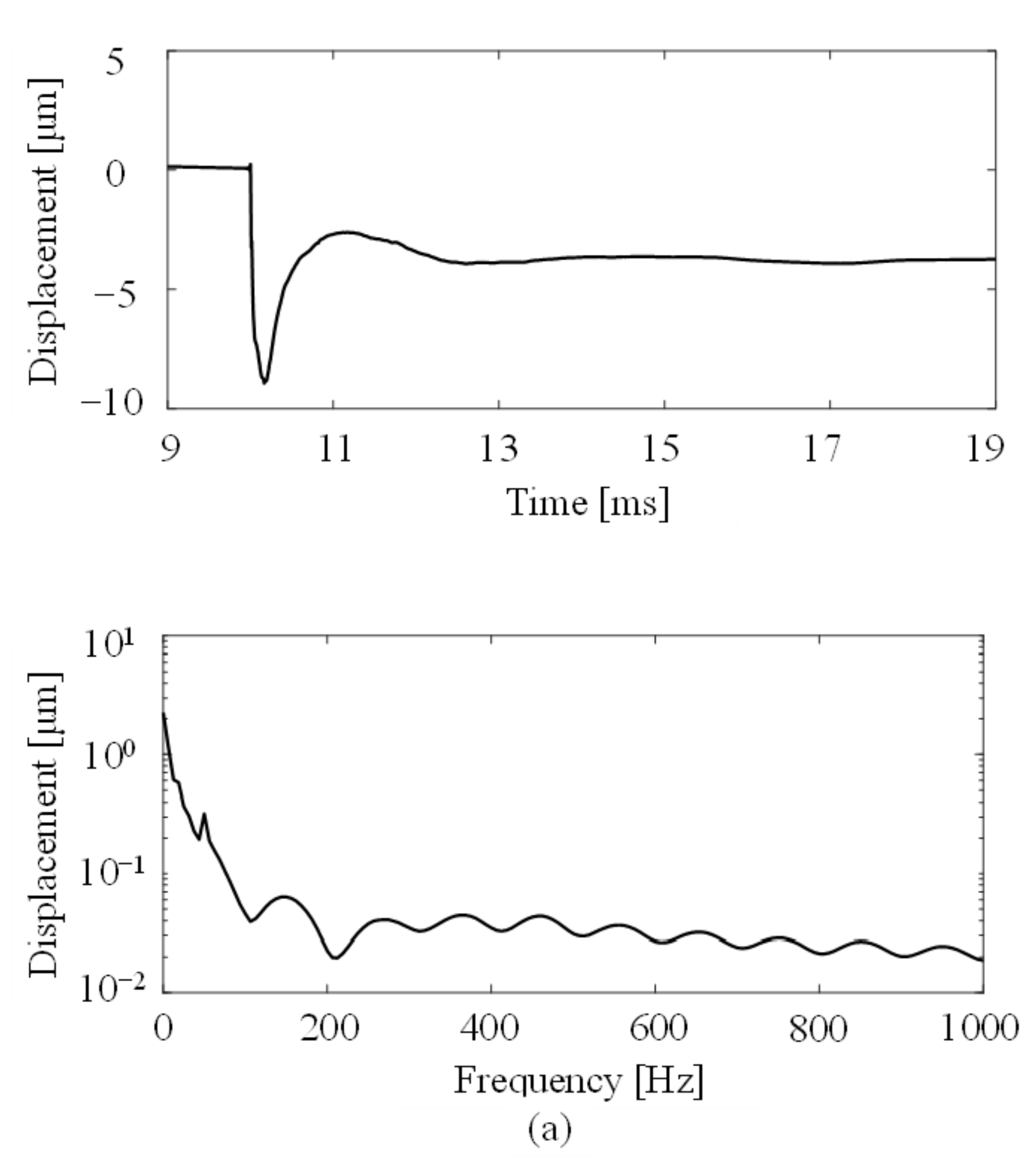
3.1. Measured Time History Responses and Their Power Spectra
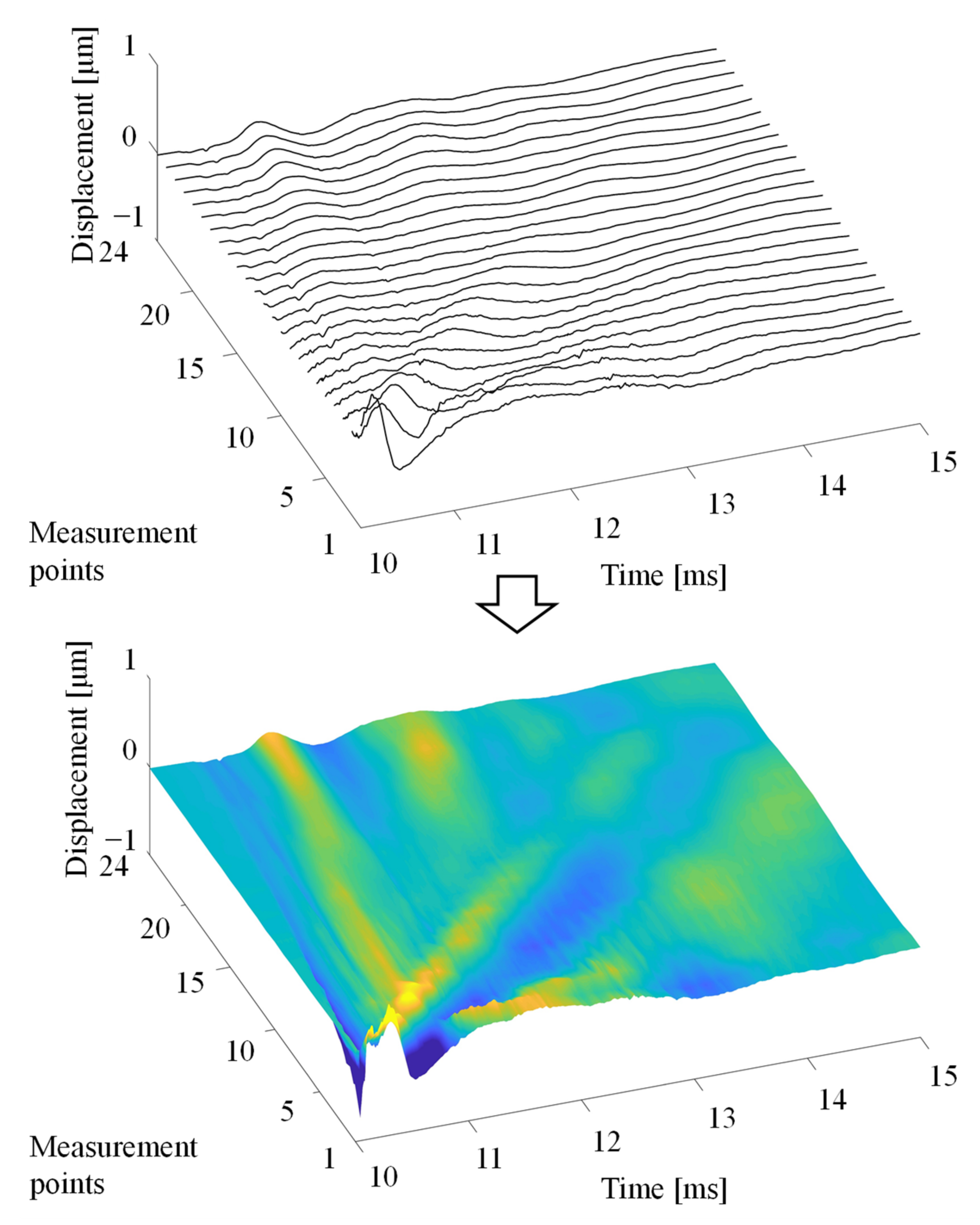
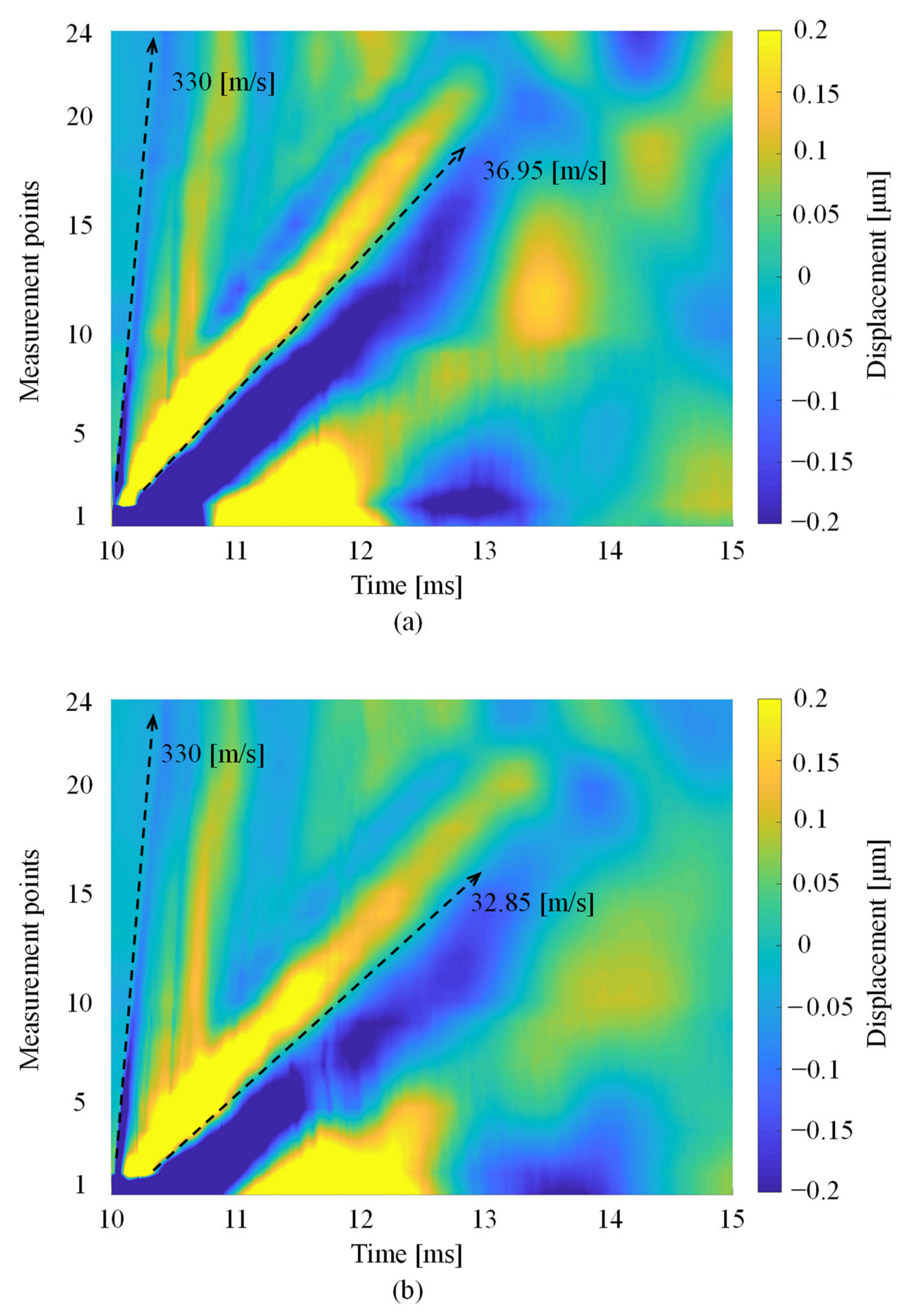
3.2. Detection of the Rayleigh Wave Propagation Velocity
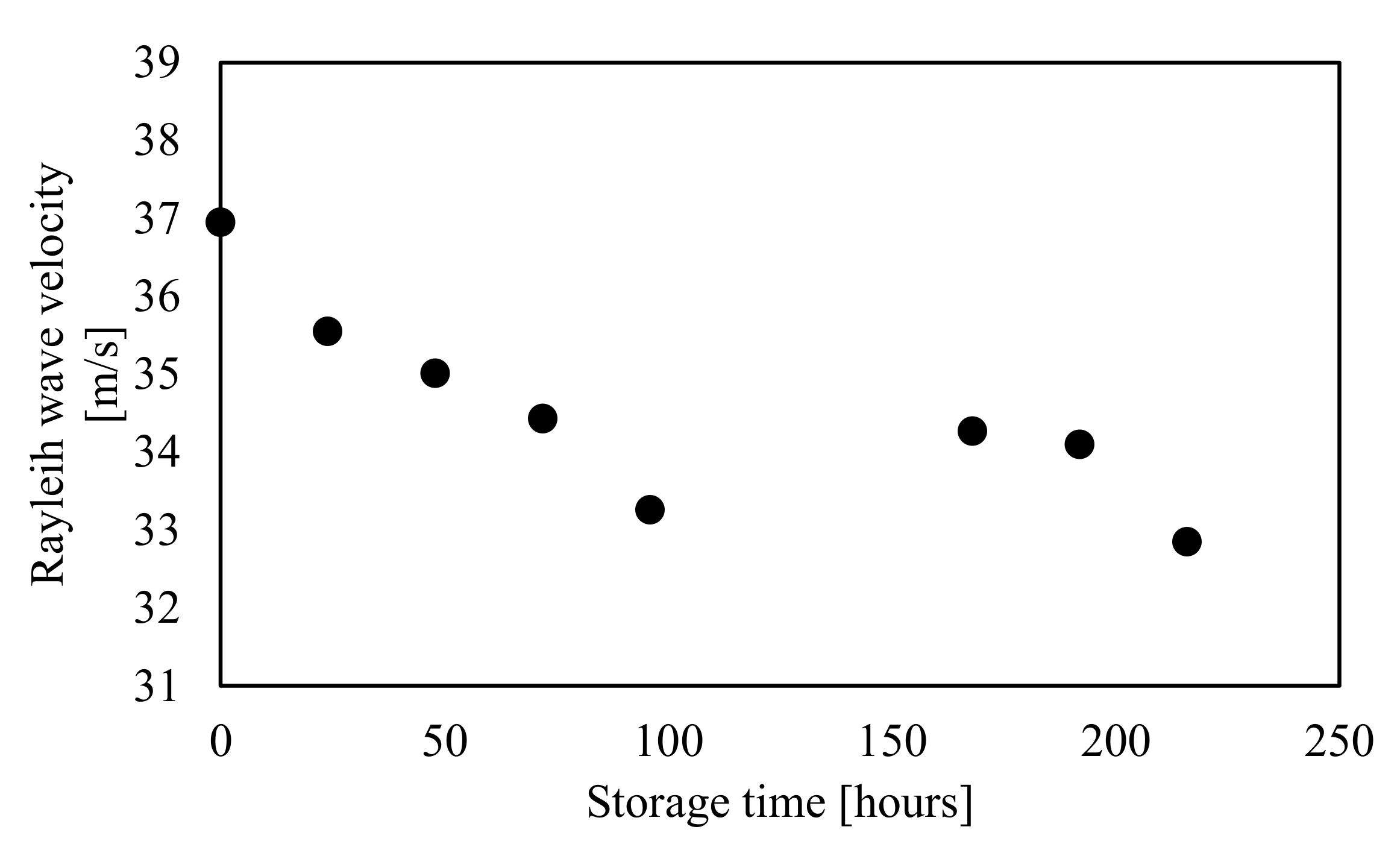
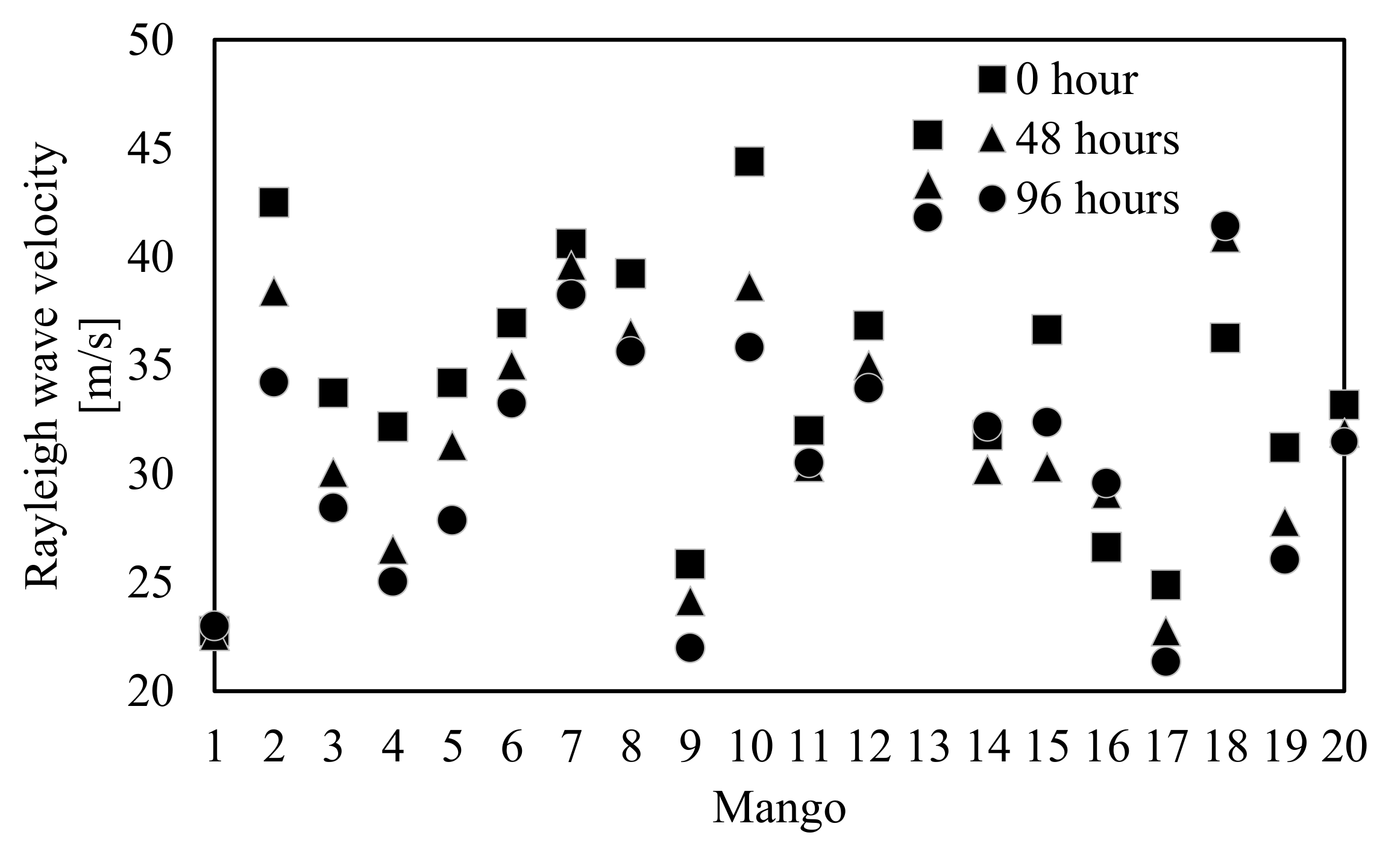
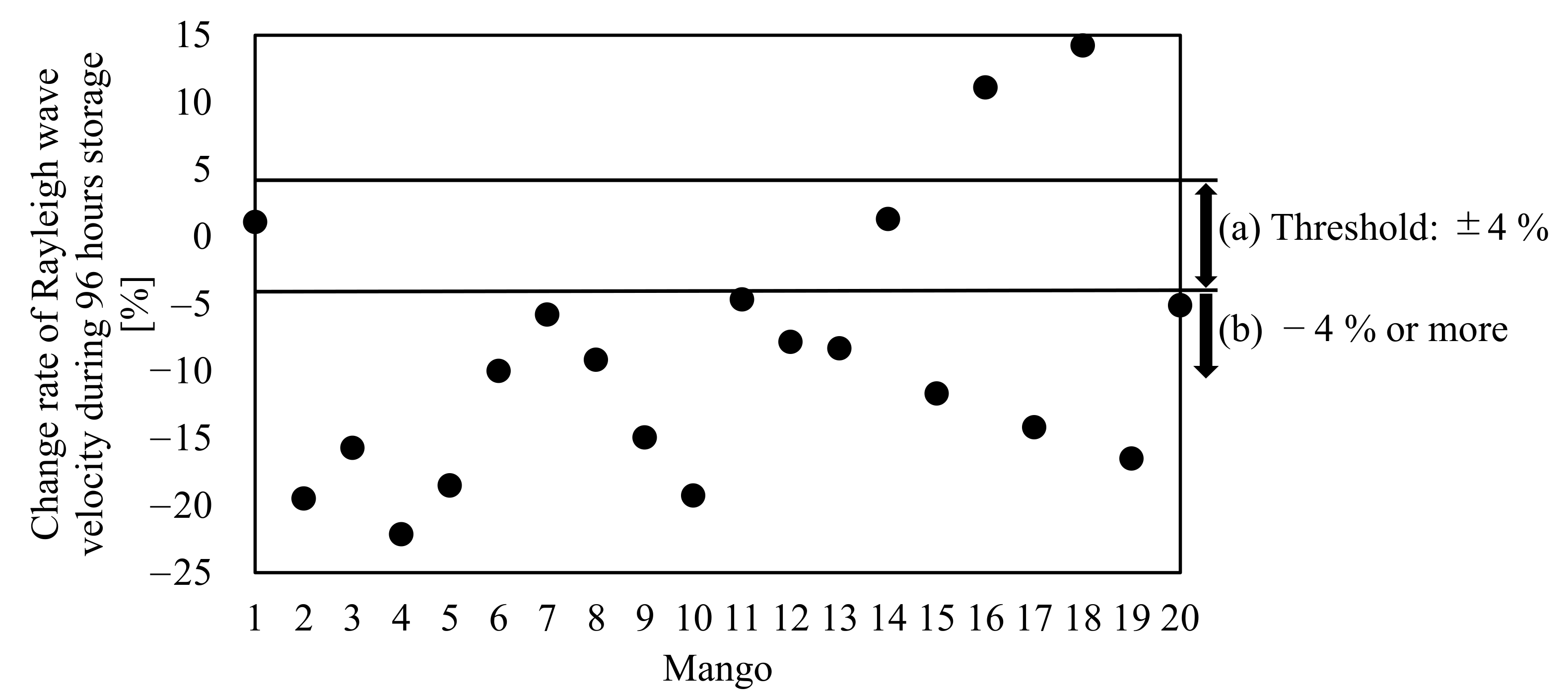
3.3. Shift in the Rayleigh Wave Velocity of Mangoes during Storage
3.4. Effect of a Seed on Measuring the Rayleigh Wave Propagation Velocity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Nordey, T.; Lechaudel, M.; Genard, M.; Joas, J. Factors affecting ethylene and carbon dioxide concentrations during ripening: Incidence on final dry matter, total soluble solids content and acidity of mango fruit. J. Plant Physiol. 2016, 196–197, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Huang, W.; Peng, Y.; Chen, Q.; Ouyang, Q.; Zhao, J. Color compensation and comparison of shortwave near infrared and long wave near infrared spectroscopy for determination of soluble solids content of ‘Fuji’ apple. Postharvest Biol. Technol. 2016, 115, 81–90. [Google Scholar] [CrossRef]
- Fan, S.; Zhang, B.; Li, J.; Huang, W.; Wang, C. Effect of spectrum measurement position variation on the robustness of NRI spectroscopy models for soluble solids content of apple. Biosyst. Eng. 2016, 143, 9–19. [Google Scholar] [CrossRef]
- Zude, M.; Herold, B.; Roger, J.; Bellon-Maurel, V.; Landahl, S. Non-destructive tests on the prediction of apple fruit flesh firmness and soluble solids content on tree and in shelf life. J. Food Eng. 2006, 77, 254–260. [Google Scholar] [CrossRef]
- Peng, Y.; Lu, R. Prediction of apple fruit firmness and soluble solids content using characteristics of multispectral scattering images. J. Food Eng. 2007, 82, 142–152. [Google Scholar] [CrossRef]
- Kawano, S.; Watanabe, H.; Iwamoto, M. Determination of sugar content in intact peaches by near infrared spectroscopy with fiber optics in interactance mode. J. Jpn. Soc. Hort. Sci. 1992, 61, 445–451. [Google Scholar] [CrossRef]
- Ma, T.; Li, X.; Inagaki, T.; Yang, H.; Tsuchikawa, S. Noncontact evaluations of soluble solids content in apples by near-infrared hyperspectral imaging. J. Food Eng. 2018, 224, 53–61. [Google Scholar] [CrossRef]
- Perez-Marin, D.; Sanchez, M.; Paz, P.; Soriano, M.; Guerrero, J.; Garrido-Varo, A. Non-destructive determination of quality parameters in nectarines during on-tree ripening and postharvest storage. Postharvest Biol. Technol. 2009, 52, 180–188. [Google Scholar] [CrossRef]
- Mendoza, F.; Lu, R.; Ariana, D.; Cen, H.; Bailey, B. Integrated spectral and image analysis of hyperspectral scattering data for prediction of apple fruit firmness and soluble solids content. Postharvest Biol. Technol. 2011, 62, 149–160. [Google Scholar] [CrossRef]
- Munera, S.; Amigo, J.M.; Blasco, J.; Cubero, S.; Talens, P.; Aleixos, N. Ripeness monitoring of two cultivars of nectarine using VIS-NIR hyperspectral reflectance imaging. J. Food Eng. 2017, 214, 29–39. [Google Scholar] [CrossRef]
- Rungpichayapichet, P.; Mahayothee, B.; Nagle, M.; Khuwijitjaru, P.; Muller, J. Robust NIRS models for non-destructive prediction of postharvest fruit ripeness and quality in mango. Postharvest Biol. Technol. 2016, 111, 31–40. [Google Scholar] [CrossRef]
- Minas, I.S.; Blanco-Cipollone, F.; Sterle, D. Accurate non-destructive prediction of peach fruit internal quality and physiological maturity with a single scan using near infrared spectroscopy. Food Chem. 2021, 335, 127626. [Google Scholar] [CrossRef] [PubMed]
- Ventura, M.; Jager, A.; Putter, H.; Roelofs, F.P.M.M. Non-destructive determination of soluble solids in apple fruit by near infrared spectroscopy (NIRS). Postharvest Biol. Technol. 1998, 14, 21–27. [Google Scholar] [CrossRef]
- Schmilovitch, Z.; Mizrach, A.; Hoffman, A.; Egozi, H.; Fuchs, Y. Determination of mango physiological indices by near-infrared spectrometry. Postharvest Biol. Technol. 2000, 19, 245–252. [Google Scholar] [CrossRef]
- Saranwong, S.; Sornsrivichai, J.; Kawano, S. Prediction of ripe-stage eating quality of mango fruit from its harvest quality measured nondestructively by near infrared spectroscopy. Postharvest Biol. Technol. 2004, 31, 137–145. [Google Scholar] [CrossRef]
- Liu, H.; An, K.; Su, S.; Yu, Y.; Wu, J.; Xiao, G.; Xu, Y. Aromatic characterization of mangoes (Mangifera indica L.) using solid phase extraction coupled with gas chromatography-mass spectrometry and olfactometry and sensory analysis. Foods. 2020, 9, 75. [Google Scholar] [CrossRef]
- Fernandez-Trujillo, J.P.; Dos-Santos, N.; Martinez-Alcaraz, R.; Bleis, I.L. Non-destructive assessment of aroma volatiles from a climacteric near-isogenic line of melon obtained by headspace stir-bar sorptive extraction. Foods. 2013, 2, 401–414. [Google Scholar] [CrossRef]
- Wei, Z.; Wang, J.; Zhang, W. Detecting internal quality of peanuts during storage using electronic nose responses combined with physicochemical methods. Food Chem. 2015, 177, 89–96. [Google Scholar] [CrossRef]
- Lebrun, M.; Plotto, A.; Goodner, K.; Ducamp, M.; Baldwin, E. Discrimination of mango fruit maturity by volatiles using the electric nose and gas chromatography. Postharvest Biol. Technol. 2008, 48, 122–131. [Google Scholar] [CrossRef]
- Kendra, P.E.; Roda, A.L.; Montgomery, W.S.; Schnell, E.Q.; Niogret, J.; Epsky, N.D.; Heath, R.R. Gas chromatography for detection of citrus infestation by fruit fly larvae (Diptera: Tephritidae). Postharvest Biol. Technol. 2011, 59, 143–149. [Google Scholar] [CrossRef]
- Zhang, B.; Xi, W.; Wei, W.; Shen, J.; Ferguson, I.; Chen, K. Changes in aroma-related volatiles and gene expression during low temperature storage and subsequent shelf-life of peach fruit. Postharvest Biol. Technol. 2011, 60, 7–16. [Google Scholar] [CrossRef]
- Landahl, S.; Terry, L.A. Non-destructive discrimination of avocado fruit ripeness using laser Doppler vibrometry. Biosyst. Eng. 2020, 194, 251–260. [Google Scholar] [CrossRef]
- Fathizadeh, Z.; Aboonajmi, M.; Beygi, S.R.H. Nondestructive firmness prediction of apple fruit using acoustic vibration response. Sci. Hortic. 2020, 262, 109073. [Google Scholar] [CrossRef]
- Kim, K.; Lee, S.; Kim, M.; Cho, B. Determinatino of apple firmness by nondestructive ultrasonic measurement. Postharvest Biol. Technol. 2009, 52, 44–48. [Google Scholar] [CrossRef]
- Vasighi-Shojae, H.; Gholami-Parashkouhi, M.; Mohammadzamani, D.; Soheili, A. Ultrasonic based determination of apple quality as a nondestructive technology. Sens. Bio-Sens. Res. 2018, 21, 22–26. [Google Scholar] [CrossRef]
- Baltazar, A.; Aranda, J.I.; Gonzalez-Aguilar, G. Bayesian classification of ripening stages of tomato fruit using acoustic impact and colorimeter sensor data. Comput. Electron. Agric. 2008, 60, 113–121. [Google Scholar] [CrossRef]
- Schotte, S.; Belie, N.D.; Baerdemaeker, J.D. Acoustic impulse-response technique for evaluation and modelling of firmness of tomato fruit. Postharvest Biol. Technol. 1999, 17, 105–115. [Google Scholar] [CrossRef]
- Shmulevich, I.; Galili, N.; Howarth, M.S. Nondestructive dynamic testing of apples for firmness evaluation. Postharvest Biol. Technol. 2003, 29, 287–299. [Google Scholar] [CrossRef]
- Gomez, A.H.; Wang, J.; Pereira, A.G. Impulse response of pear fruit and its relation to Magness-Taylor firmness during storage. Postharvest Biol. Technol. 2005, 35, 209–215. [Google Scholar] [CrossRef]
- Ketelaere, B.D.; Howarth, M.S.; Crezee, L.; Lammertyn, J.; Viaene, K.; Bulens, I.; Baerdemaeker, J.D. Postharvest firmness changes as measured by acoustic and low-mass impact devices: A comparison of techniques. Postharvest Biol. Technol. 2006, 41, 275–284. [Google Scholar]
- Duprat, F.; Grotte, M.; Pietri, E.; Loonis, D. The Acoustic Impulse Response Method for Measuring the Overall Firmness of Fruit. J. Agric. Eng. Res. 1997, 66, 251–259. [Google Scholar] [CrossRef]
- Cooke, J.R. An Interpretation of the Resonant Behavior of Intact Fruits and Vegetables. ASABE 1972, 15, 1075–1080. [Google Scholar] [CrossRef]
- Hosoya, N.; Mishima, M.; Kajiwara, I.; Maeda, S. Non-destructive firmness assessment of apples using a non-contact laser excitation system based on a laser-induced plasma shock wave. Postharvest Biol. Technol. 2017, 128, 11–17. [Google Scholar] [CrossRef]
- Kajiwara, I.; Akita, R.; Hosoya, N. Damage detection in pipes based on acoustic excitations using laser-induced plasma. Mech. Syst. Sig. Process. 2018, 111, 570–579. [Google Scholar] [CrossRef]
- Hosoya, N.; Yoshinaga, A.; Kanda, A.; Kajiwara, I. Non-contact and non-destructive Lamb wave generation using laser-induced plasma shock wave. Int. J. Mech. Sci. 2018, 140, 486–492. [Google Scholar] [CrossRef]
- Huda, F.; Kajiwara, I.; Hosoya, N. Damage detection in membrane structures using non-contact laser excitation and wavelet transformation. J. Sound Vib. 2014, 333, 3609–3624. [Google Scholar] [CrossRef]
- Hosoya, N.; Nagata, M.; Kajiwara, I. Acoustic testing in a very small space based on a point sound source generated by laser-induced breakdown: Stabilization of plasma formation. J. Sound Vib. 2013, 332, 4572–4583. [Google Scholar] [CrossRef]
- Hosoya, N.; Kajiwara, I.; Inoue, T.; Umenai, K. Non-contact acoustic tests based on nanosecond laser ablation: Generation of a pulse sound source with a small amplitude. J. Sound Vib. 2014, 333, 4254–4264. [Google Scholar] [CrossRef]
- Hosoya, N.; Kajiwara, I.; Umenai, K.; Maeda, S. Dynamic characterizations of underwater structures using noncontact vibration tests based on nanosecond laser ablation in water: Evaluation of passive vibration suppression with damping materials. J. Vib. Control. 2018, 24, 3714–3725. [Google Scholar] [CrossRef]
- Huda, F.; Kajiwara, I.; Hosoya, N.; Kawamura, S. Bolt loosening analysis and diagnosis by non-contact laser excitation vibration tests. Mech. Syst. Sig. Process. 2013, 40, 589–604. [Google Scholar] [CrossRef]
- Kurahashi, S.; Mikami, K.; Kitamura, T.; Hasegawa, N.; Okada, H.; Kondo, S.; Nishikino, M.; Kawachi, T.; Shimada, Y. Demonstration of 25-Hz-inspection speed laser remote sensing for internal concrete defects. J. Appl. Remote Sens. 2018, 12, 015009. [Google Scholar] [CrossRef]
- Hosoya, N.; Umino, R.; Kanda, A.; Kajiwara, I.; Yoshinaga, A. Lamb wave generation using nanosecond laser ablation to detect damage. J. Vib. Control. 2018, 24, 5842–5853. [Google Scholar] [CrossRef]
- Hosoya, N.; Terashima, Y.; Umenai, K.; Maeda, S. High spatial and temporal resolution measurement of mechanical properties in hydrogels by non-contact laser excitation. AIP Adv. 2016, 6, 095223. [Google Scholar]
- Hitchman, S.; Wijk, K.; Davidson, Z. Monitoring attenuation and the elastic properties of an apple with laser ultrasound. Postharvest Biol. Technol. 2016, 121, 71–77. [Google Scholar] [CrossRef]
- Podymova, N.B.; Kalashnikov, I.E.; Bolotova, L.K.; Kobeleva, L.I. Laser-ultrasonic nondestructive evaluation of proposity in particulate reinforced metal-matrix composites. Ultrasonics 2019, 99, 105959. [Google Scholar] [CrossRef] [PubMed]
- Ji, B.; Zhang, Q.; Cao, J.; Li, H.; Zhang, B. Non-contact detection of delamination in stainless steel/carbon steel composites with laser ultrasonic. Optik 2021, 26, 165893. [Google Scholar] [CrossRef]
- Cao-Rial, M.T.; Moreno, C.; Quintela, P. Determination of Young modulus by using Rayleigh waves. Appl. Math. Modell. 2020, 77, 439–455. [Google Scholar] [CrossRef]
- Yue, Z.; Qiu, P.; Yang, R.; Yang, G. Experimental study on a Mach cone and trailing Rayleigh waves in a stress wave chasing running crack problem. Theor. Appl. Fract. Mech. 2019, 104, 102371. [Google Scholar] [CrossRef]
- Na, J.K.; Blackshire, J.L. Interaction of Rayleigh surface waves with a tightly closed fatigue crack. NDT&E Int. 2010, 43, 432–439. [Google Scholar]
- Pfeifer, D.; Kim, J.; Jacobs, L.J. Nonlinear Rayleigh waves to evaluate plasticity damage in X52 pipeline material. Mech. Syst. Sig. Process. 2020, 143, 106794. [Google Scholar] [CrossRef]
- Onodera, Y.; Choi, P.-K. Surface-wave models on soft gels. J. Acoust. Soc. Am. 1998, 104, 3358–3363. [Google Scholar] [CrossRef]
- Ikeda, T.; Choi, P.K.; Ishii, T.; Arai, I.; Osawa, M. Firmness evaluation of watermelon flesh by using surface elastic waves. J. Food Eng. 2015, 160, 28–33. [Google Scholar] [CrossRef]
- Hosoya, N.; Nagata, M.; Kajiwara, I.; Umino, R. Nano-second Laser induced Plasma shock wave in Air for non-contact Vibration Tests. Exp. Mech. 2016, 56, 1305–1311. [Google Scholar] [CrossRef]
- Georgiev, V.B.; Krylov, V.V.; Qin, Q.; Attenborough, K. Generation of flexural waves in plates by laser- initiated airborne shock waves. J. Sound Vib. 2011, 330, 217–228. [Google Scholar] [CrossRef]

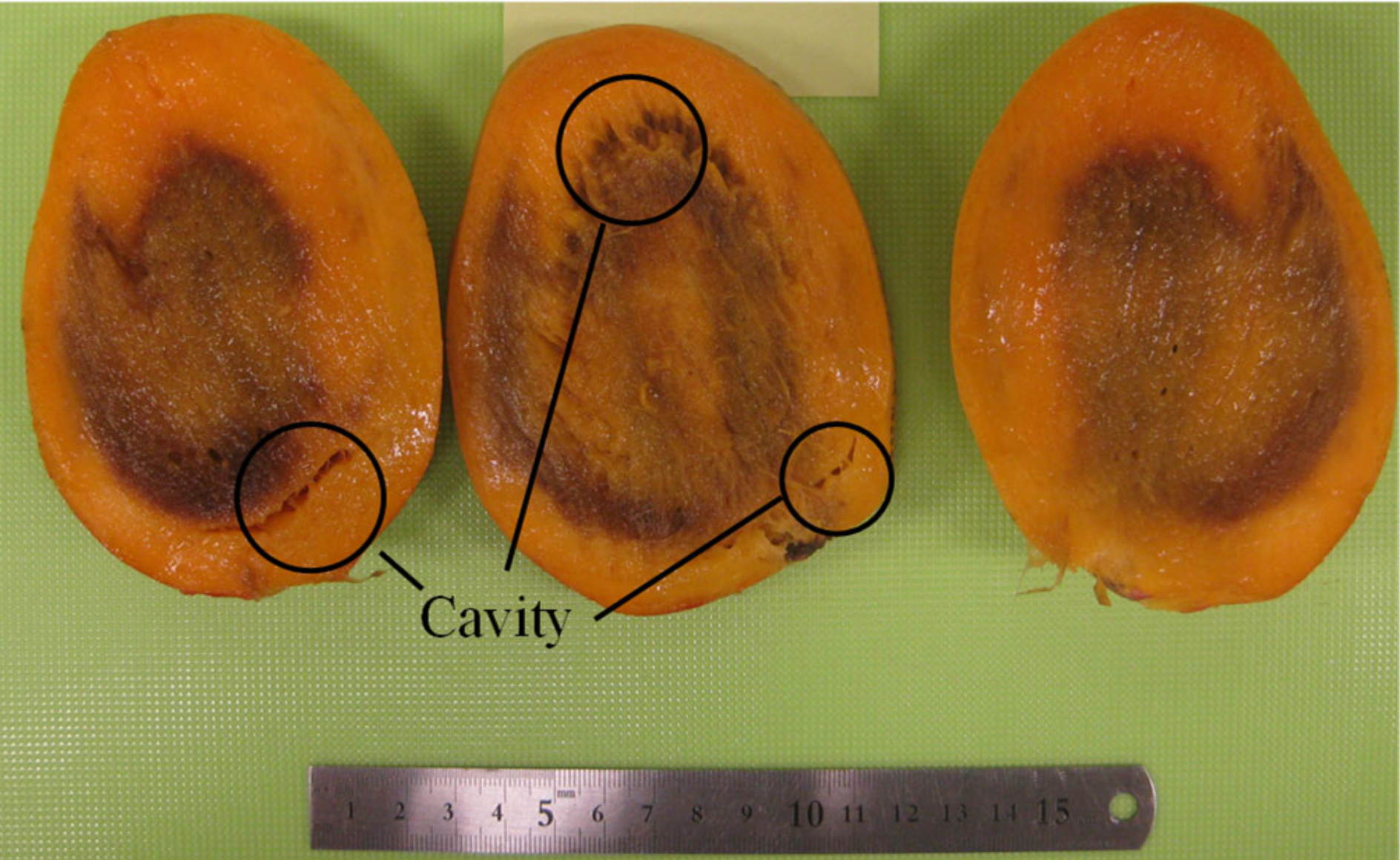
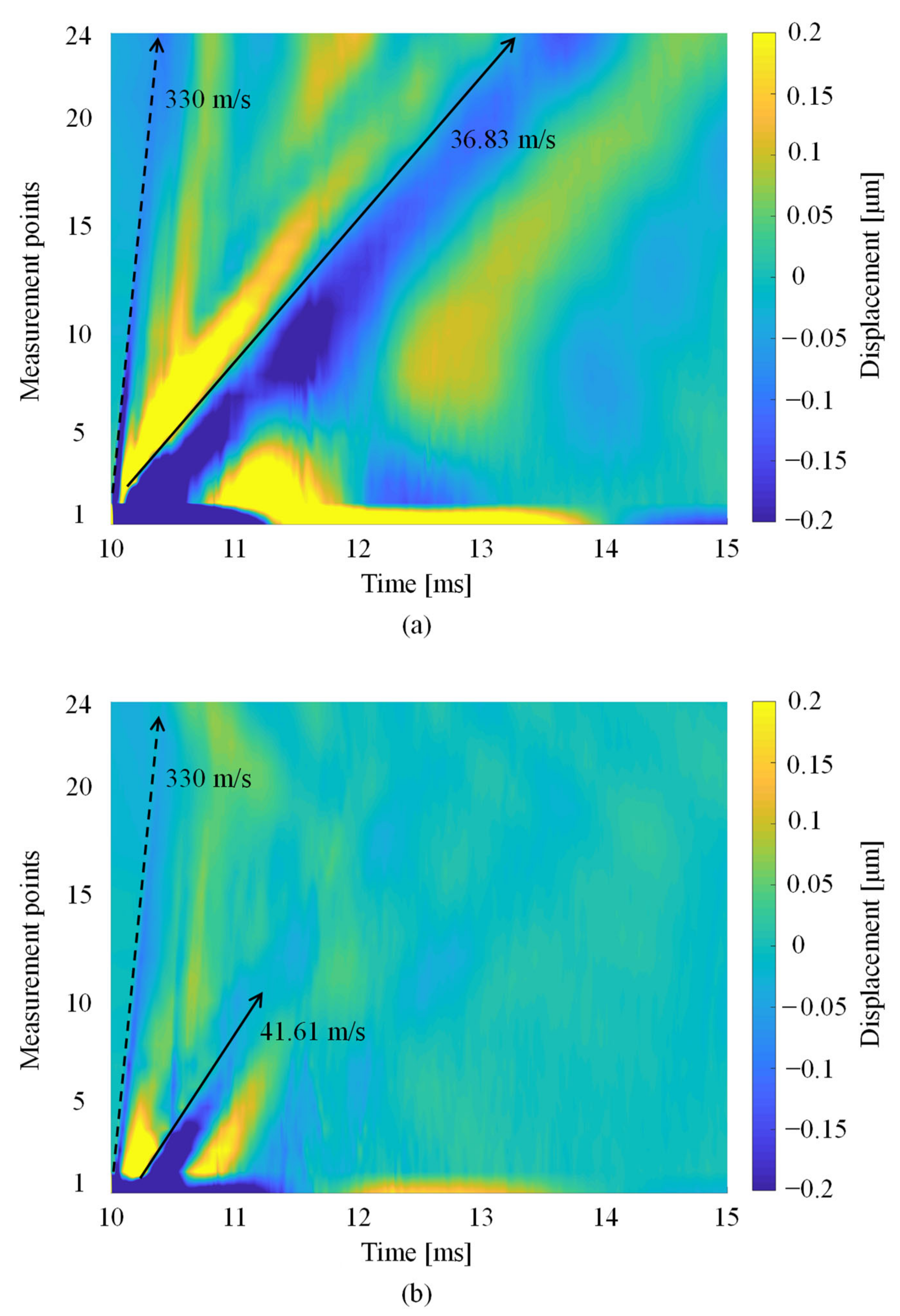
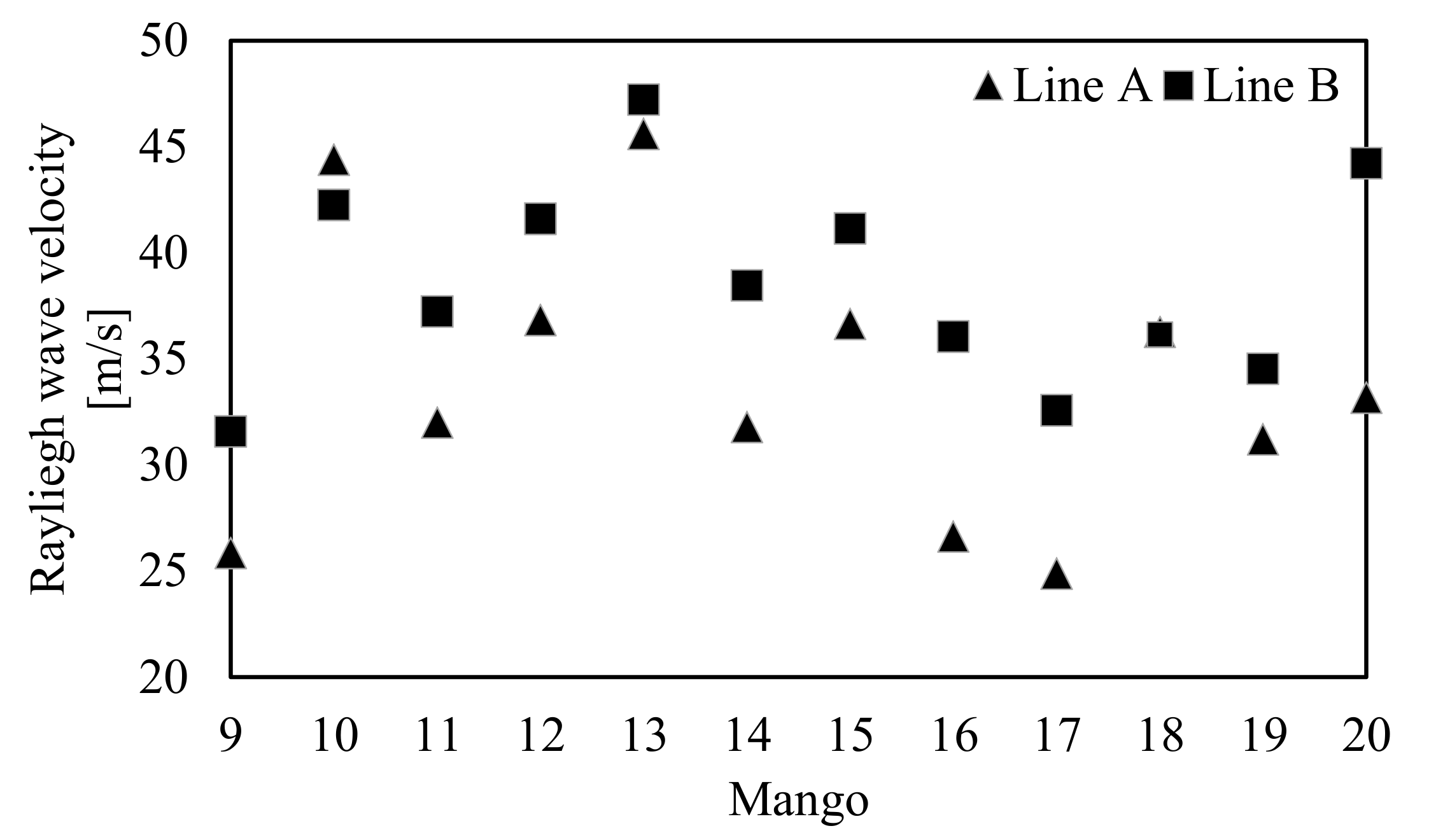
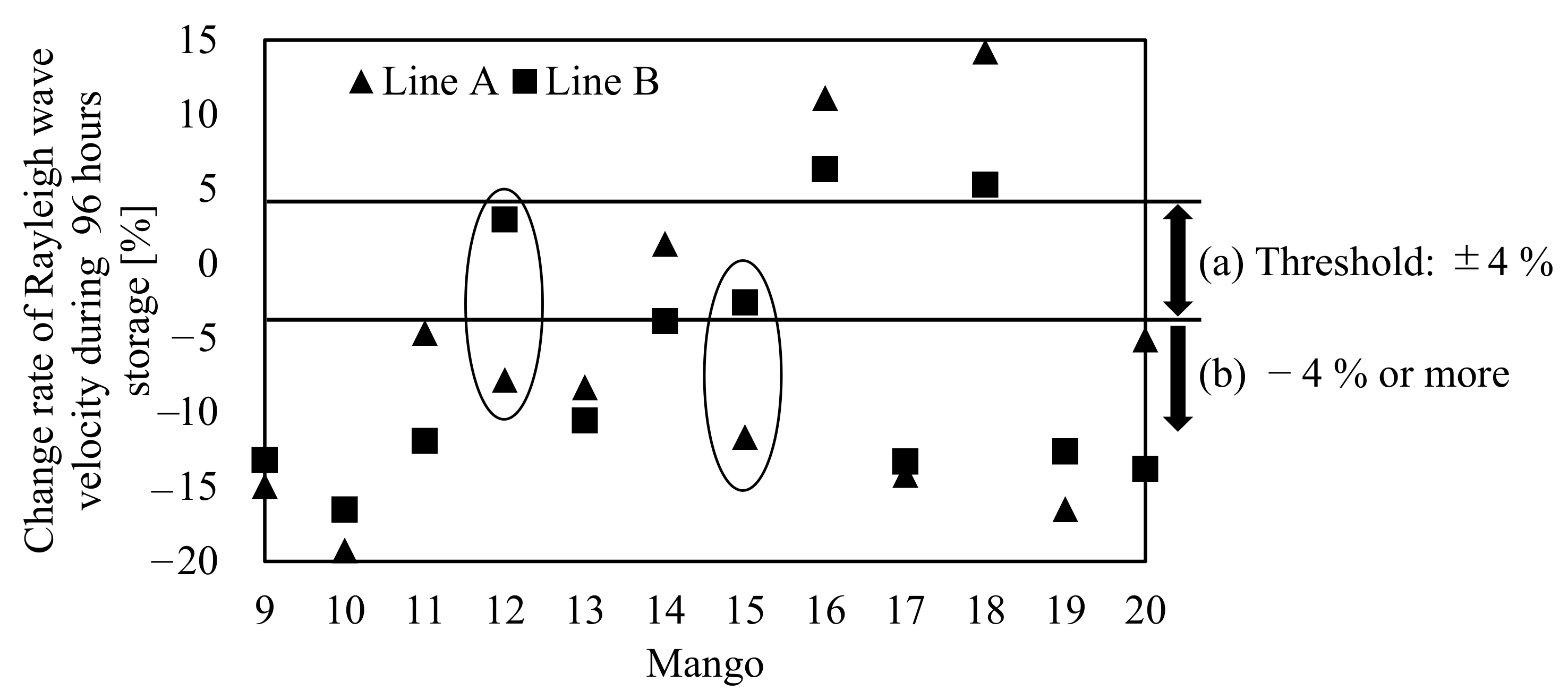
| Sample No. | Mass (g) | Density (kg/m3) | Circumference (m) |
|---|---|---|---|
| Average | 457.0 | 1094 | 0.270 |
| 1 | 531.9 | 978.0 | 0.240 |
| 2 | 510.2 | 1001 | 0.300 |
| 3 | 493.9 | 1026 | 0.280 |
| 4 | 495.4 | 1064 | 0.298 |
| 5 | 493.1 | 990.0 | 0.300 |
| 6 | 496.7 | 1158 | 0.284 |
| 7 | 490.0 | 1174 | 0.284 |
| 8 | 501.8 | 741.0 | 0.277 |
| 9 | 437.6 | 1020 | 0.281 |
| 10 | 435.9 | 815.0 | 0.280 |
| 11 | 371.8 | 1039 | 0.253 |
| 12 | 371.7 | 1039 | 0.242 |
| 13 | 391.4 | 1094 | 0.262 |
| 14 | 401.2 | 1121 | 0.250 |
| 15 | 426.5 | 1192 | 0.240 |
| 16 | 438.9 | 1227 | 0.280 |
| 17 | 465.1 | 1300 | 0.300 |
| 18 | 398.3 | 1113 | 0.284 |
| 19 | 480.6 | 1343 | 0.281 |
| 20 | 519.6 | 1452 | 0.280 |
| Sample No. | Measurement Path | Storage Time (h) |
|---|---|---|
| 1–5, 7, 8 | Line A | 0, 48, 96 |
| 6 | Line A | 0, 24, 48, 72, 96, 168, 192, 216 |
| 9–20 | Lines A and B | 0, 48, 96 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arai, N.; Miyake, M.; Yamamoto, K.; Kajiwara, I.; Hosoya, N. Soft Mango Firmness Assessment Based on Rayleigh Waves Generated by a Laser-Induced Plasma Shock Wave Technique. Foods 2021, 10, 323. https://doi.org/10.3390/foods10020323
Arai N, Miyake M, Yamamoto K, Kajiwara I, Hosoya N. Soft Mango Firmness Assessment Based on Rayleigh Waves Generated by a Laser-Induced Plasma Shock Wave Technique. Foods. 2021; 10(2):323. https://doi.org/10.3390/foods10020323
Chicago/Turabian StyleArai, Nayuta, Masafumi Miyake, Kengo Yamamoto, Itsuro Kajiwara, and Naoki Hosoya. 2021. "Soft Mango Firmness Assessment Based on Rayleigh Waves Generated by a Laser-Induced Plasma Shock Wave Technique" Foods 10, no. 2: 323. https://doi.org/10.3390/foods10020323
APA StyleArai, N., Miyake, M., Yamamoto, K., Kajiwara, I., & Hosoya, N. (2021). Soft Mango Firmness Assessment Based on Rayleigh Waves Generated by a Laser-Induced Plasma Shock Wave Technique. Foods, 10(2), 323. https://doi.org/10.3390/foods10020323







