Enterococci from Raw-Milk Cheeses: Current Knowledge on Safety, Technological, and Probiotic Concerns
Abstract
1. Introduction
2. Safety of Using Enterococcus spp. in Dairy Food
3. Technological Characteristics of Enterococci
3.1. Acidification Activity
3.2. Proteolytic Activity
3.3. Lipolytic Activity
3.4. Production of Aromatic Compounds
4. Probiotic Potential of Enterococcus spp.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Graham, K.; Stack, H.; Rea, R. Safety, beneficial and technological properties of enterococci for use in functional food application-a review. Crit. Rev. Food Sci. Nutr. 2020, 60, 3836–3861. [Google Scholar] [CrossRef] [PubMed]
- Švec, P.; Franz, C.M.A.P. The genus Enterococcus. In Lactic Acid Bacteria: Biodiversity and Taxonomy; Holzapfel, W.H., Wood, B.J.B., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2014; pp. 171–213. [Google Scholar]
- Foulquie´ Moreno, M.R.; Sarantinopoulos, P.; Tsakalidou, E.; De Vuyst, L. The role and application of enterococci in food and health. Int. J. Food Microbiol. 2006, 106, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, F.; Willems, R.J.L.; Gilmore, M.S. Enterococcus diversity, origins in nature, and gut colonization. In Enterococci: From Commensals to Leading Causes of Drug Resistant Infection (Internet); Gilmore, M.S., Clewell, D.B., Ike, Y., Shankar, N., Eds.; Massachusetts Eye and Ear Infirmary: Boston, MA, USA, 2014; pp. 1–59. [Google Scholar]
- Cho, S.; Jackson, J.R.; Frye, J.G. The prevalence and antimicrobial resistance phenotypes of Salmonella, Escherichia coli and Enterococcus sp. in surface water. Lett. Appl. Microbiol. 2020, 71, 3–25. [Google Scholar] [CrossRef]
- Manson, A.L.; Van Tyne, D.; Straub, T.J.; Clock, S.; Crupain, M.; Rangan, U.; Gilmore, M.S.; Earl, A.M. Chicken meat-associated enterococci: Influence of agricultural antibiotic use and connection to the clinic. Appl. Environ. Microbiol. 2019, 85, e01559-19. [Google Scholar] [CrossRef]
- Švec, P.; Vandamme, P.; Bryndová, H.; Holochová, P.; Kosina, M.; Mašlaňová, I.; Sedláček, I. Enterococcus plantarum sp. nov., isolated from plants. Int. J. Syst. Evol. Microbiol. 2012, 62, 1499–1505. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Peixoto, F.; Igrejas, G.; Parelho, C.; Garcia, P.; Carvalho, I.; Sousa, M.; Pereira, J.E.; Rodrigues, A.; Poeta, P.A.C.Q.D. First report on vanA-Enterococcus faecalis recovered from soils subjected to long-term livestock agricultural practices in Azores archipelago. Int. J. Environ. Res. 2018, 12, 39–44. [Google Scholar] [CrossRef]
- Said, L.B.; Klibi, N.; Dziri, R.; Borgo, F.; Boudabous, A.; Slamaa, K.B.; Torres, C. Prevalence, antimicrobial resistance and genetic lineages of Enterococcus spp. from vegetable food, soil and irrigation water in farm environments in Tunisia. J. Sci. Food Agric. 2016, 96, 1627–1633. [Google Scholar] [CrossRef]
- Čolo, J.; Mihajlović, S.; Tolinački, M.; Popović, D.; Kojić, M.; Terzić-Vidojević, A. Characterization of lactic acid bacteria isolated from Bosnian artisanal dry fermented sausage (Sudžuk) during fermentation. Genetika 2015, 47, 819–832. [Google Scholar] [CrossRef]
- Terzić-Vidojević, A.; Veljović, K.; Tolinački, M.; Živković, M.; Lukić, J.; Lozo, J.; Fira, Đ.; Jovčić, B.; Strahinić, I.; Begović, J.; et al. Diversity of non-starter lactic acid bacteria in autochthonous dairy products from Western Balkan Countries—Technological and probiotic properties. Food Res. Int. 2020, 136, 109494. [Google Scholar] [CrossRef] [PubMed]
- Qiao, X.; Du, R.; Wang, Y.; Han, Y.; Zhou, Z. Isolation, characterisation and fermentation optimisation of bacteriocin-producing Enterococcus faecium. Waste Biomass Valorization 2019, 11, 3173–3181. [Google Scholar] [CrossRef]
- Elkenany, R.M.; Elsayed, M.M.; Eltaysh, R.A.; Zakaria, A.I.; El-Baz, A.H. In vitro probiotic characteristics of Enterococcus species isolated from raw cow milk. Int. J. Probiotics Prebiotics 2018, 13, 117–126. [Google Scholar]
- Veljovic, K.; Fira, D.; Terzic-Vidojevic, A.; Abriouel, H.; Galvez, A.; Topisirovic, L. Evaluation of antimicrobial and proteolytic activity of enterococci isolated from fermented products. Eur. Food Res. Technol. 2009, 230, 63–70. [Google Scholar] [CrossRef]
- Popović, N.; Djokić, J.; Brdarić, E.; Dinić, M.; Terzić-Vidojević, A.; Golić, N.; Veljović, K. The influence of heat-killed Enterococcus faecium BGPAS1-3 on the tight junction protein expression and immune function in differentiated Caco-2 cells infected with Listeria monocytogenes ATCC 19111. Front Microbiol. 2019, 10, 412. [Google Scholar] [CrossRef] [PubMed]
- Franz, C.M.A.P.; Stiles, M.E.; Schleifer, K.H.; Holzapfel, W.H. Enterococci in foods–a conundrum for food safety. Int. J. Food Microbiol. 2003, 88, 105–122. [Google Scholar] [CrossRef]
- Giraffa, G. Enterococci from foods. FEMS Microbiol. Rev. 2002, 26, 163–171. [Google Scholar] [CrossRef]
- Câmara, S.P.A.; Dapkevicius, A.; Silva, C.C.G.; Malcata, F.X.; Dapkevicius, M.L.E. Artisanal Pico cheese as reservoir of Enterococcus species possessing virulence and antibiotic resistance properties: Implications for food safety. Food Biotechnol. 2020, 34, 25–41. [Google Scholar] [CrossRef]
- Popović, N. Examination of Probiotic and Immunomodulatory Characteristics of Natural Isolates of Enterococci on In Vitro and In Vivo Models. Ph.D. Thesis, Faculty of Biology, University of Belgrade, Belgrade, Serbia, 2019. [Google Scholar]
- Directive, H. Council Directive 92/46/EEC of 16 June 1992 laying down the health rules for the production and placing on the market of raw milk, heat-treated milk and milk-based products. Off. J. Eur. Comm. 1992, 268, 1–32. [Google Scholar]
- Birollo, G.A.; Reinheimer, J.A.; Vinderola, C.G. Enterococci vs. nonlactic acid microflora as hygiene indicators for sweetened yoghurt. Food Microbiol. 2001, 18, 597–604. [Google Scholar] [CrossRef]
- Giraffa, G. Functionality of enterococci in dairy products. Int. J. Food Microbiol. 2003, 88, 215–222. [Google Scholar] [CrossRef]
- Zaheer, R.; Cook, S.R.; Barbieri, R.; Goji, N.; Cameron, A.; Petkau, A.; Polo, R.O.; Tymensen, L.; Stamm, C.; Song, J.; et al. Surveillance of Enterococcus spp. reveals distinct species and antimicrobial resistance diversity across a One-Health continuum. Sci. Rep. 2020, 10, 3937. [Google Scholar] [CrossRef]
- Veljović, K.; Terzić-Vidojević, A.; Tolinački, M.; Mihajlović, S.; Vukotić, G.; Golić, N.; Kojić, M. Molecular characterization of natural dairy isolates of Enterococcus faecalis and evaluation of their antimicrobial potential. In Enterococcus Faecalis: Molecular Characteristics, Role in Nosocomial Infections and Antimicrobial Effects. (Bacteriology Research Developments); Mack, H.L., Ed.; Nova Science Publishers Inc.: London, UK, 2014; pp. 123–135. [Google Scholar]
- Jeeja, V. Characterization of food and clinical isolates of enterococci. Acta Sci. Microbiol. 2019, 2, 56–61. [Google Scholar]
- Reyes, K.; Bardossy, A.C.; Zervos, M. Vancomycin-resistant enterococci. Epidemiology, infection, prevention and control. Infect. Dis. Clin. N. Am. 2016, 30, 953–965. [Google Scholar] [CrossRef] [PubMed]
- Terkuran, M.; Turhan, E.Ü.; Erginkaya, Z. The risk of vancomycin resistant enterococci infections from food industry. In Health and Safety Aspects of Food Processing Technologies; Malik, A., Erginkaya, Z., Erten, H., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 513–535. [Google Scholar]
- Bonacina, J.; Suarez, N.; Hormigo, R.; Fadda, S.; Lechner, M.; Saavedra, L. A genomic view of food-related and probiotic Enterococcus strains. DNA Res. 2017, 24, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Montealegre, M.C.; Singh, K.V.; Murray, B.E. Gastrointestinal tract colonization dynamics by different Enterococcus faecium clades. J. Infect. Dis. 2016, 213, 1914–1922. [Google Scholar] [CrossRef] [PubMed]
- Ogier, J.-C.; Serror, P. Safety assessment of dairy microorganisms: The Enterococcus genus. Int. J. Food Microbiol. 2008, 126, 291–301. [Google Scholar] [CrossRef]
- Ricci, A.; Allende, A.; Bolton, D.; Chemaly, M.; Davies, R.; Girones, R.; Herman, L.; Koutsoumanis, K.; Lindqvist, R.; Nørrung, B.; et al. Scientific opinion on the update of the list of QPS-recommended biological agents intentionally added to food or feed as notified to European Food Safety Authority (EFSA). Panel on Biological Hazards (BIOHAZ). EFSA J. 2017, 15, e04664. [Google Scholar] [PubMed]
- Hanchi, H.; Mottawea, W.; Sebei, K.; Hammami, R. The genus Enterococcus: Between probiotic potential and safety concerns-an update. Front. Microbiol. 2018, 9, 1791. [Google Scholar] [CrossRef]
- Terzić-Vidojević, A.; Veljović, K.; Begović, J.; Filipić, B.; Popović, D.; Tolinački, M.; Miljković, M.; Kojić, M.; Golić, N. Diversity and antibiotic susceptibility of autochthonous dairy enterococci isolates: Are they safe candidates for autochthonous starter cultures? Front. Microbiol. 2015, 6, 954. [Google Scholar] [CrossRef]
- Veljovic, K.; Terzic-Vidojevic, A.; Vukasinovic, M.; Strahinic, I.; Begovic, J.; Lozo, J.; Topisirovic, L. Preliminary characterization of lactic acid bacteria isolated from Zlatar cheese. J. Appl. Microbiol. 2007, 103, 2142–2152. [Google Scholar] [CrossRef]
- Terzic-Vidojevic, A.; Veljovic, K.; Tolinacki, M.; Nikolic, M.; Ostojic, M.; Topisirovic, L.J. Characterization of lactic acid bacteria isolated from artisanal Zlatar cheeses produced at two different geographical location. Genetika 2009, 41, 117–136. [Google Scholar] [CrossRef]
- Terzic-Vidojevic, A.; Lozo, J.; Topisirovic, L.J. Dominant lactic acid bacteria in artisanal Pirot cheeses of different ripening period. Genetika 2009, 41, 341–352. [Google Scholar] [CrossRef]
- Terzic-Vidojevic, A.; Tolinacki, M.; Nikolic, M.; Veljovic, K.; Jovanovic, S.; Macej, O.; Topisirovic, L. Artisanal Vlasina raw goat’s milk cheeses: Evaluation and selection of autochthonous lactic acid bacteria as starter cultures. Food Technol. Biotechnol. 2013, 51, 554–563. [Google Scholar]
- Terzić-Vidojević, A.; Mihajlović, S.; Uzelac, G.; Golić, N.; Fira, Ð.; Kojić, M.; Topisirović, L. Identification and characterization of lactic acid bacteria isolated from artisanal white brined Golija cows’ milk cheeses. Arch. Biol. Sci. 2014, 66, 179–192. [Google Scholar] [CrossRef]
- Terzic-Vidojevic, A.; Mihajlovic, S.; Uzelac, G.; Veljovic, K.; Tolinacki, M.; Nikolic, M.; Topisirovic, L.; Kojic, M. Characterization of lactic acid bacteria isolated from artisanal Travnik young cheeses, sweet creams and sweet kajmaks over four seasons. Food Microbiol. 2014, 39, 27–38. [Google Scholar] [CrossRef]
- Golić, N.; Čadež, N.; Terzić-Vidojević, A.; Šuranská, H.; Beganović, J.; Lozo, J.; Kos, B.; Šušković, J.; Raspor, P.; Topisirović, L. Evaluation of lactic acid bacteria and yeast diversity in traditional white pickled and fresh soft cheeses from the mountain regions of Serbia and lowland regions of Croatia. Int. J. Food Microbiol. 2013, 166, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Margalho, L.P.; van Schalkwijk, S.; Bachmann, H.; Sant’Ana, A.S. Enterococcus spp. in Brazilian artisanal cheeses: Occurrence and assessment of phenotypic and safety properties of a large set of strains through the use of high throughput tools combined with multivariate statistics. Food Control 2020, 118, 107425. [Google Scholar] [CrossRef]
- Karakas-Sen, A.; Karakas, E. Isolation, identification and technological properties of lactic acid bacteria from raw cow milk. Biosci. J. Uberlândia 2018, 34, 385–399. [Google Scholar] [CrossRef]
- Slyvka, I.M.; Tsisaryk, O.Y.; Dronyk, G.V.; Musiy, L.Y. Strains of lactic acid bacteria isolated from traditional Carpathian cheeses. Regul. Mech. Biosyst. 2018, 9, 62–68. [Google Scholar] [CrossRef]
- Levkov, V.; Mojsova, S.; Nastova, R.; Srbinovska, S.; Gjorgovska, N. Identification and phenotypic characteristics of lactic acid bacteria isolated from some traditional cheeses produced in the Republic of Macedonia. Maced. J. Anim. Sci. 2017, 7, 79–87. [Google Scholar]
- Sofu, A.; Ekinci, F.Y. Bacterial diversity dynamics of traditional Turkish Ezine cheese as evaluated by PCR-DGGE and SSCP analysis. Int. J. Dairy Technol. 2016, 69, 592–600. [Google Scholar] [CrossRef]
- Pogačić, T.; D’Andrea, M.; Kagkli, D.-M.; Corich, V.; Giacomini, A.; Baldan, E.; Čanžek Majhenič, A.; Obermajer, T.; Rogelj, I.; Samaržija, D. Biodiversity of microbial consortia isolated from traditional fresh sheep cheese Karakačanski skakutanac. Mljekarstvo 2011, 61, 208–219. [Google Scholar]
- Muruzović, M.; Mladenović, K.; Žugić-Petrović, T.; Čomić, L. Characterization of lactic acid bacteria isolated from traditionally made Serbian cheese and evaluation of their antagonistic potential against Enterobacteriaceae. J. Food Process. Preserv. 2018, 42, e13577. [Google Scholar] [CrossRef]
- Mrkonjić Fuka, M.; Engel, M.; Skelin, A.; Redžepović, S.; Schloter, M. Bacterial communities associated with the production of artisanal Istrian cheese. Int. J. Food Microbiol. 2010, 142, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Mrkonjić Fuka, M.; Wallisch, S.; Engel, M.; Welzl, G.; Havranek, J.; Schloter, M. Dynamics of bacterial communities during the ripening process of different Croatian cheese types derived from raw ewe’s milk cheeses. PLoS ONE 2013, 8, e80734. [Google Scholar]
- Başar Uymaz, B.; Akçelik, N.; Yüksel, Z. Physicochemical and microbiological characterization of protected designation of origin Ezine cheese: Assessment of nonstarter lactic acid bacterial diversity with antimicrobial activity. Food Sci. Anim. Resour. 2019, 39, 804–819. [Google Scholar] [CrossRef]
- González, L.; Fernández Cuadrillero, A.; Castro, J.M.; Bernardo, A.; Tornadijo, M.E. Selection of lactic acid bacteria isolated from San Simón da Costa cheese (PDO) in order to develop an autochthonous starter culture. Adv. Microbiol. 2015, 5, 748–759. [Google Scholar] [CrossRef][Green Version]
- Kročko, M.; Čanigová, M.; Ducková, V.; Artimová, A.; Bezeková, J.; Poston, J. Antibiotic resistance of Enterococcus species isolated from raw foods of animal origin in south west part of Slovakia. Czech J. Food Sci. 2011, 29, 654–659. [Google Scholar] [CrossRef]
- Jokovic, N.; Nikolic, M.; Begovic, J.; Jovcic, B.; Savic, D.; Topisirovic, L. A survey of the lactic acid bacteria isolated from Serbian artisanal dairy product kajmak. Int. J. Food Microbiol. 2008, 127, 305–311. [Google Scholar] [CrossRef]
- Bojanic Rasovic, M.; Mayrhofer, S.; Ochome, A.A.M.; Ajanovic, E.; Zunabovic, M.; Martinovic, A.; Domig, K.J. Diversity of lactic acid bacteria isolated from traditional Montenegrin dairy products. Genetika 2018, 50, 465–482. [Google Scholar] [CrossRef]
- İspirli, H.; Demirbaş, F.; Dertli, E. Characterization of functional properties of Enterococcus spp. isolated from Turkish white cheese. LWT-Food Sci. Technol. 2017, 75, 358–365. [Google Scholar] [CrossRef]
- Fortina, M.G.; Ricci, G.; Acquati, A.; Zeppa, G.; Gandini, A.; Manachini, P.L. Genetic characterization of some lactic acid bacteria occurring in an artisanal protected denomination origin (PDO) Italian cheese, the Toma piemontese. Food Microbiol. 2003, 20, 397–404. [Google Scholar] [CrossRef]
- Cruciata, M.; Gaglio, R.; Todaro, M.; Settanni, L. Ecology of Vastedda della valle del Belìce cheeses: A review and recent findings to stabilize the traditional production. Food Rev. Int. 2019, 35, 90–103. [Google Scholar] [CrossRef]
- Terzic-Vidojevic, A.; Vukasinovic, M.; Veljovic, K.; Ostojic, M.; Topisirovic, L. Characterization of microflora in homemade semi-hard white Zlatar cheese. Int. J. Food Microbiol. 2007, 114, 36–42. [Google Scholar] [CrossRef]
- Mrkonjic Fuka, M.; Zgomba Maksimovic, A.; Tanuwidjaja, I.; Hulak, N.; Schloter, M. Characterization of enterococcal community isolated from an artisan Istrian raw milk cheese: Biotechnological and safety aspects. Food Technol. Biotechnol. 2017, 55, 368–380. [Google Scholar] [CrossRef]
- Tezel, B.U. Preliminary in-vitro evaluation of the probiotic potential of the bacteriocinogenic strain Enterococcus lactis PMD74 isolated from Ezine Cheese. J. Food Qual. 2019, 4693513. [Google Scholar]
- Morandi, S.; Cremonesi, P.; Povolo, M.; Brasca, B. Enterococcus lactis sp. nov., from Italian raw milk cheeses. Int. J. Syst. Evol. Microbiol. 2012, 62, 1992–1996. [Google Scholar] [CrossRef]
- Jokovic, N.; Vukasinovic, M.; Veljovic, K.; Tolinacki, M.; Topisirovic, L. Characterization of non-starter lactic acid bacteria in traditionally produced homemade Radan cheese during ripening. Arch. Biol. Sci. 2011, 63, 1–10. [Google Scholar] [CrossRef]
- Pogačić, T.; Samaržija, D.; Corich, V.; D’Andrea, M.; Kagkli, D.M.; Giacomini, A.; Rogelj, I. Microbiota of Karakačanski skakutanac, an artisanal fresh sheep cheese studied by culture-independent PCR-ARDRA and PCR-DGGE. Dairy Sci. Technol. 2010, 90, 641–648. [Google Scholar] [CrossRef]
- Guo, L.; Li, T.; Tang, Y.; Yang, L.; Huo, G. Probiotic properties of Enterococcus strains isolated from traditional naturally fermented cream in China. Microb. Biotechnol. 2016, 9, 737–745. [Google Scholar] [CrossRef]
- Medina, R.; Katz, M.; González, S.; Oliver, G. Characterization of the lactic acid bacteria in ewe milk and artisanal cheese from northwest Argentina. J. Food Prot. 2001, 64, 559–563. [Google Scholar] [CrossRef]
- European Parliament. Directive 2000/54/EC of the European Parliament and of the Council of 18 September 2000 on the protection of workers from risks related to exposure to biological agents at work. Off. J. Eur. Comm. 2000, 262, 21–45. [Google Scholar]
- Banwo, K.; Sanni, A.; Tan, H. Technological properties and probiotic potential of Enterococcus faecium strains isolated from cow milk. J. Appl. Microbiol. 2013, 114, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Popović, N.; Dinić, M.; Tolinački, M.; Mihajlović, S.; Terzić-Vidojević, A.; Bojić, S.; Djokić, J.; Golić, N.; Veljović, K. New insight into biofilm formation ability, the presence of virulence genes and probiotic potential of Enterococcus sp. dairy isolates. Front. Microbiol. 2018, 9, 78. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, C.V.; Taddei, A.; Amedei, A. The controversial role of Enterococcus faecalis in colorectal cancer. Therap. Adv. Gastroenterol. 2018, 11, 1–11. [Google Scholar] [CrossRef]
- Maekawa, T.; Fukaya, R.; Takamatsu, S.; Itoyama, S.; Fukuoka, T.; Yamada, M.; Hata, T.; Nagaoka, S.; Kawamoto, K.; Eguchi, H.; et al. Possible involvement of Enterococcus infection in the pathogenesis of chronic pancreatitis and cancer. Biochem. Biophys. Res. Commun. 2018, 506, 962–969. [Google Scholar] [CrossRef]
- Jahan, M.; Zhanel, G.G.; Sparling, R.; Holley, R.A. Horizontal transfer of antibiotic resistance from Enterococcus faecium of fermented meat origin to clinical isolates of E. faecium and Enterococcus faecalis. Int. J. Food Microbiol. 2015, 199, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Lemen, S.W.; Lewalter, K. Antibiotic stewardship and horizontal infection control are more effective than screening, isolation and eradication. Infection 2018, 46, 581–590. [Google Scholar] [CrossRef]
- Alonso, V.P.P.; Queiroz, M.M.; Gualberto, M.L.; Nascimento, M.S. Klebsiella pneumonia carbapenemase (KPC), methicillin-resistant Staphylococcus aureus (MRSA), and vancomycin-resistant Enterococcus spp. (VRE) in the food production chain and biofilm formation on abiotic surfaces. Curr. Opin. Food Sci. 2019, 26, 79–86. [Google Scholar] [CrossRef]
- Faron, M.L.; Ledeboer, N.A.; Buchan, B.W. Resistance mechanisms, epidemiology, and approaches to screening for vancomycin-resistant Enterococcus in the health care setting. J. Clin. Microbiol. 2016, 54, 2436–2447. [Google Scholar] [CrossRef] [PubMed]
- Fiore, E.; Van Tyne, D.; Gilmore, M.S. Pathogenicity of enterococci. Microbiol. Spectr. 2019, 7, GPP3-0053-2018. [Google Scholar] [CrossRef]
- Sartingen, S.; Rozdzinski, E.; Muscholl-Silberhorn, A.; Marre, R. Aggregation substance increases adherence and internalization, but not translocation, of Enterococcus faecalis through different intestinal epithelial cells in vitro. Infect. Immun. 2000, 68, 6044–6047. [Google Scholar] [CrossRef]
- Leavis, H.; Top, J.; Shankar, N.; Borgen, K.; Bonten, M.; van Embden, J.; Willems, R.J.L. A novel putative enterococcal pathogenicity island linked to the esp virulence gene of Enterococcus faecium and associated with epidemicity. J. Bacteriol. 2004, 186, 672–682. [Google Scholar] [CrossRef]
- Koch, S.; Hufnagel, M.; Theilacker, C.; Huebner, J. Enterococcal infections: Host response, therapeutic, and prophylactic possibilities. Vaccine 2004, 22, 822–830. [Google Scholar] [CrossRef]
- Ramos, S.; Silva, V.; Dapkevicius, M.L.E.; Igrejas, G.; Poeta, P. Enterococci, from harmless bacteria to a pathogen. Microorganisms 2020, 8, 1118. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Guidance on the characterisation of microorganisms used as feed additives or as production organisms. Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). EFSA J. 2018, 16, e05206. [Google Scholar]
- Leavis, H.L.; Willems, R.J.; van Wamel, W.J.; Schuren, F.H.; Caspers, M.P.; Bonten, M.J. Insertion sequence-driven diversification creates a globally dispersed emerging multiresistant subspecies of E. faecium. PLoS Pathog. 2007, 3, e7. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Guidance for assessing safety of Enterococcus faecium in animal feed. Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). EFSA J. 2012, 10, 2682. [Google Scholar]
- Kiruthiga, A.; Padmavathy, K.; Shabana, P.; Naveenkumar, V.; Gnanadesikan, S.; Malaiyan, J. Improved detection of esp, hyl, asa1, gelE, cylA virulence genes among clinical isolates of Enterococci. BMC Res. Notes 2020, 13, 170. [Google Scholar] [CrossRef]
- Eaton, T.J.; Gasson, M.J. Molecular screening of Enterococcus virulence determinants and potential for genetic exchange between food and medical isolates. Appl. Environ. Microbiol. 2001, 67, 1628–1635. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.C.; Jonas, D.; Huber, I.; Karygianni, L.; Wölber, J.; Hellwig, E.; Arweiler, N.; Vach, K.; Wittmer, A.; Al-Ahmad, A. Enterococcus faecalis from food, clinical specimens, and oral sites: Prevalence of virulence factors in association with biofilm formation. Front Microbiol. 2016, 6, 1534. [Google Scholar] [CrossRef]
- Ch’ng, J.-H.; Chong, K.K.L.; Lam, L.N.; Wong, J.J.; Kline, K.A. Biofilm-associated infection by enterococci. Nat. Rev. Microbiol. 2019, 17, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Elhadidy, M.; Zahran, E. Biofilm mediates Enterococcus faecalis adhesion, invasion and survival into bovine mammary epithelial cells. Lett. Appl. Microbiol. 2014, 58, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Gobbetti, M.; de Angelis, M.; di Cagno, R.; Mancini, L.; Fox, P.F. Pros and cons for using non-starter lactic acid bacteria (NSLAB) as secondary/adjunct starters for cheese ripening. Trends Food Sci. Technol. 2015, 45, 167–178. [Google Scholar] [CrossRef]
- Villani, F.; Coppola, S. Selection of enterococcal strains for water-buffalo Mozzarella cheese manufacture. Ann. Microbiol. Enzim. 1994, 44, 97–105. [Google Scholar]
- Sarantinopoulos, P.; Kalantzopoulos, G.; Tsakalidou, E. Effect of Enterococcus faecium on microbiological, physicochemical and sensory characteristics of Greek Feta cheese. Int. J. Food Microbiol. 2002, 76, 93–105. [Google Scholar] [CrossRef]
- Casalta, E.; Zennaro, R. Effect of specific starters on microbiological, biochemical and sensory characteristics of Venaco, a Corsican soft cheese. Sci. Alim. 1997, 17, 79–94. [Google Scholar]
- Centeno, J.A.; Menendez, S.; Hermida, M.A.; Rodriguez-Otero, J.L. Effects of the addition of Enterococcus faecalis in Cebreiro cheese manufacture. Int. J. Food Microbiol. 1999, 48, 97–111. [Google Scholar] [CrossRef]
- Gardiner, G.E.; Ross, R.P.; Wallace, J.M.; Scanlan, F.P.; Jagers, P.P.; Fitzgerald, G.F.; Collins, J.K.; Stanton, C. Influence of a probiotic adjunct culture of Enterococcus faecium on the quality of Cheddar cheese. J. Agr. Food Chem. 1999, 47, 4907–4916. [Google Scholar] [CrossRef]
- Hassanzadazar, H.; Ehsani, A.; Mardan, K. Antibacterial activity of Enterococcus faecium derived from Koopeh cheese against Listeria monocytogenes in probiotic ultra-filtrated cheese. Vet. Res. Forum 2014, 5, 169–175. [Google Scholar]
- Yerlikaya, O.; Akbulut, N. Potential use of probiotic Enterococcus faecium and Enterococcus durans strains in Izmir Tulum cheese as adjunct culture. J. Food Sci. Technol. 2019, 56, 2175–2185. [Google Scholar] [CrossRef]
- Pirouzian, H.R.; Hesary, J.; Farajnia, S.; Moghaddam, M.; Ghiassifar, S.; Manafi, M. Inclusion of Enterococcus faecalis and Enterococcus faecium to UF white cheese. Int. J. Nutr. Food Eng. 2010, 4, 405–408. [Google Scholar]
- Andrighetto, C.; Knijff, E.; Lombardi, A.; Torriani, S.; Vancanneyt, M.; Kersters, K.; Swings, J.; Dellaglio, F. Phenotypic and genetic diversity of enterococci isolated from Italian cheeses. J. Dairy. Res. 2001, 68, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Durlu-Ozkaya, F.; Xanthopoulos, V.; Tunail, N.; Litopoulou-Tzanetaki, E. Technologically important properties of lactic acid bacteria isolates from Beyaz cheese made from raw ewes’ milk. J. Appl. Microbiol. 2001, 91, 861–870. [Google Scholar] [CrossRef] [PubMed]
- Sarantinopoulos, P.; Andrighetto, C.; Georgalaki, M.D.; Rea, M.C.; Lombardi, A.; Cogan, T.M.; Kalantzopoulos, G.; Tsakalidou, E. Biochemical properties of enterococci relevant to their technological performance. Int. Dairy J. 2001, 11, 621–647. [Google Scholar] [CrossRef]
- Abeijón, M.C.; Medina, R.B.; Katz, M.B.; González, S.N. Technological properties of Enterococcus faecium isolated from ewe’s milk and cheese with importance for flavour development. Can. J. Microbiol. 2006, 52, 237–245. [Google Scholar] [CrossRef]
- Jamaly, N.; Benjouad, A.; Comunian, R.; Daga, E.; Bouksaim, M. Characterization of Enterococci isolated from Moroccan dairy products. Afr. J. Microbiol. Res. 2010, 4, 1768–1774. [Google Scholar]
- Aspri, M.; Bozoudi, D.; Tsaltas, D.; Hill, C.; Papademas, P. Raw donkey milk as a source of Enterococcus diversity: Assessment of their technological properties and safety characteristics. Food Control 2017, 73, 81–90. [Google Scholar] [CrossRef]
- Beresford, T.P.; Fitzsimons, N.A.; Brennan, N.L.; Cogan, T.M. Recent advances in cheese microbiology. Int. Dairy J. 2001, 11, 259–274. [Google Scholar] [CrossRef]
- Ribeiro, S.C.; Coelho, M.C.; Todorov, S.D.; Franco, B.D.G.M.; Dapkevicius, M.L.E.; Silva, C.C.G. Technological properties of bacteriocin-producing lactic acid bacteria isolated from Pico cheese an artisanal cow’s milk cheese. J. Appl. Microbiol. 2014, 116, 573–585. [Google Scholar] [CrossRef]
- Ziadi, M.; M’hir, S.; Dubois-Dauphin, R.; Chambellon, E.; Yvon, M.; Thonart, P.; Hamdi, M. Analysis of volatile compounds, amino acid catabolism and some technological properties of Enterococcus faecalis strain SLT13 isolated from artisanal Tunisian fermented milk. Br. Microbiol. Res. J. 2016, 14, 17309. [Google Scholar] [CrossRef]
- Turhan, İ.; Öner, Z. Determination of starter culture properties of lactic acid bacteria isolated from cheese. GIDA 2014, 39, 9–15. [Google Scholar]
- de Paula, P.L.M.; de Moraes, M.L.; Schueler, J.; de Souza, N.A.A.; Furlaneto, M.C.; Maia, L.F.; Katsuda, M.S. Enterococcus faecium in artisanal ripening cheese: Technological and safety aspects. Res. Soc. Dev. 2020, 9, e299119452. [Google Scholar] [CrossRef]
- Suzzi, G.; Caruso, M.; Gardini, F.; Lombardi, A.; Vannini, L.; Guerzoni, M.E.; Andrighetto, C.; Lanorte, M.T. A survey of the enterococci isolated from an artisanal Italian goat’s cheese (semicotto caprino). J. Appl. Microbiol. 2000, 89, 267–274. [Google Scholar] [CrossRef]
- Tuncer, Y. Some technological properties of phenotypically identified enterococci strains isolated from Turkish Tulum cheese. Afr. J. Biotechnol. 2009, 8, 7008–7016. [Google Scholar]
- Jaouani, I.; Abbassi, M.S.; Ribeiro, S.C.; Khemiri, M.; Mansouri, R.; Messadi, L.; Silva, C.C.G. Safety and technological properties of bacteriocinogenic enterococci isolates from Tunisia. J. Appl. Microbiol. 2015, 119, 1089–1100. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, M.G.; Guinee, T.P.; O’Callaghan, D.M.; Fox, P.F. Autolysis and proteolysis in different strains of starter bacteria during Cheddar cheese ripening. J. Dairy Res. 1994, 61, 249–262. [Google Scholar] [CrossRef]
- González, L.; Sacristán, N.; Arenas, R.; Fresno, J.M.; Tornadijo, M.E. Enzymatic activity of lactic acid bacteria (with antimicrobial properties) isolated from a traditional Spanish cheese. Food Microbiol. 2010, 27, 592–597. [Google Scholar] [CrossRef]
- Pessione, A.; Lamberti, C.; Cocolin, L.; Campolongo, S.; Grunau, A.; Giubergia, S.; Eberl, L.; Riedel, K.; Pessione, E. Different protein expression profiles in cheese and clinical isolates of Enterococcus faecalis revealed by proteomic analysis. Proteomics 2012, 12, 431–447. [Google Scholar] [CrossRef]
- Gútiez, L.; Gómez-Sala, B.; Recio, I.; del Campo, R.; Cintas, L.M.; Herranz, C.; Hernández, P.E. Enterococcus faecalis strains from food, environmental, and clinical origin produce ACE-inhibitory peptides and other bioactive peptides during growth in bovine skim milk. Int. J. Food Microbiol. 2013, 166, 93–101. [Google Scholar] [CrossRef]
- Chajęcka-Wierzchowska, W.; Zadernowska, A.; Łaniewska-Trokenheim, L. Virulence factors of Enterococcus spp. presented in food. LWT-Food Sci. Technol. 2017, 75, 670–676. [Google Scholar] [CrossRef]
- Galloway-Peña, J.R.; Bourgogne, A.; Qin, X.; Murray, B.E. Diversity of the fsr-gelE region of the Enterococcus faecalis genome but conservation in strains with partial deletions of the fsr operon. Appl. Environ. Microbiol. 2011, 77, 442–451. [Google Scholar] [CrossRef]
- Medeiros, A.W.; Pereira, R.I.; Oliveira, D.V.; Martins, P.D.; D’azevedo, P.A.; Van der Sand, S.; Frazzon, J.; Frazzon, A.P.G. Molecular detection of virulence factors among food and clinical Enterococcus faecalis strains in South Brazil. Braz. J. Microbiol. 2014, 45, 327–332. [Google Scholar] [CrossRef]
- Domann, E.; Hain, T.; Ghai, R.; Billion, A.; Kuenne, C.; Zimmermann, K.; Chakraborty, T. Comparative genomic analysis for the presence of potential enterococcal virulence factors in the probiotic Enterococcus faecalis strain Symbioflor 1. Int. J. Med. Microbiol. 2007, 297, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Worsztynowicz, P.; Olejnik-Schmidt, A.; Bialas, W.; Grajek, W. Identification and partial characterization of proteolytic activity of Enterococcus faecalis relevant to their application in the dairy industry. Acta Biochim. Pol. 2019, 66, 61–69. [Google Scholar] [CrossRef]
- Tulini, F.L.; Hymery, N.; Haertlé, T.; Le Blay, G.; De Martinis, E.C.P. Screening for antimicrobial and proteolytic activities of lactic acid bacteria isolated from cow, buffalo and goat milk and cheeses marketed in the southeast region of Brazil. J. Dairy Res. 2016, 83, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Franz, C.M.; Muscholl-Silberhorn, A.B.; Yousif, N.M.; Vancanneyt, M.; Swings, J.; Holzapfel, W.H. Incidence of virulence factors and antibiotic resistance among enterococci isolated from food. Appl. Environ. Microbiol. 2001, 67, 4385–4389. [Google Scholar] [CrossRef]
- Cariolato, D.; Andrighetto, C.; Lombardi, A. Occurrence of virulence factors and antibiotic resistances in Enterococcus faecalis and Enterococcus faecium collected from dairy and human samples in North Italy. Food Control 2008, 19, 886–892. [Google Scholar] [CrossRef]
- Perin, L.M.; Belvisio, S.; dal Bello, B.; Nero, L.A.; Cocolin, L. Technological properties and biogenic amines production by bacteriocinogenic lactococci and enterococci strains isolated from raw goat’s milk. J. Food Prot. 2017, 80, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Dağdemir, E. Identification of Lactic Acid Bacteria Isolated from Pickled White Cheeses and the Possibilities of Using Some Selected Isolates as Culture. Ph.D. Thesis, Graduate School of Natural and Applied Science, Atatürk University Science and Technology, Erzurum, Turkey, 2006; p. 190. [Google Scholar]
- Cosentino, S.; Pisano, M.B.; Corda, A.; Fadda, M.E.; Piras, C. Genotypic and technological characterization of enterococci isolated from artisanal Fiore Sardo cheese. J. Dairy Res. 2004, 71, 444–450. [Google Scholar] [CrossRef]
- Savitri, M.; Kumar, V.; Kumari, A.; Angmo, K.; Bhalla, T.C. Isolation and characterization of lactic acid bacteria from traditional pickles of Himachal Pradesh, India. J. Food Sci. Technol. 2017, 54, 1945–1952. [Google Scholar]
- Tulini, F.L.; Biscola, V.; Choiset, Y.; Hymery, N.; Le Blay, G.; De Martinis, E.C.P.; Chobert, J.-M.; Haertlé, T. Evaluation of the proteolytic activity of Enterococcus faecalis FT132 and Lactobacillus paracasei FT700, isolated from dairy products in Brazil, using milk proteins as substrates. Eur. Food Res. Technol. 2015, 241, 385–392. [Google Scholar] [CrossRef]
- García-Cano, I.; Rocha-Mendoza, D.; Ortega-Anaya, J.; Wang, K.; Kosmerl, E.; Jiménez-Flores, R. Lactic acid bacteria isolated from dairy products as potential producers of lipolytic, proteolytic and antibacterial proteins. Appl. Microbiol. Biotechnol. 2019, 103, 5243–5257. [Google Scholar] [CrossRef]
- Esteban-Torres, M.; Mancheño, J.M.; de Las Rivas, B.; Muñoz, R. Production and characterization of a tributyrin esterase from Lactobacillus plantarum suitable for cheese lipolysis. J. Dairy Sci. 2014, 97, 6737–6744. [Google Scholar] [CrossRef] [PubMed]
- Dinçer, E.; Kıvanç, M. Lipolytic activity of lactic acid bacteria isolated from Turkish pastırma. Anadolu Univ. J. Sci. Technol. C-Life Sci. Biotechnol. 2018, 7, 12–19. [Google Scholar] [CrossRef]
- Carrasco de Mendoza, M.S.; Scarinci, M.S.; Huerto-Garat, H.E.; Simonetta, A.C. Technological properties of enterococci in lactic starters: Acidifying and lipolytic activities. Microbiol. Aliment. Nutr. 1992, 10, 289–293. [Google Scholar]
- Morandi, S.; Brasca, M.; Andrighetto, C.; Lombardi, A.; Lodi, R. Technological and molecular characterisation of enterococci isolated from north–west Italian dairy products. Int. Dairy J. 2006, 16, 867–875. [Google Scholar] [CrossRef]
- Kilcawley, K.N. Cheese flavour. In Fundamentals of Cheese Science, 2nd ed.; Fox, P.F., Guinee, T.P., Cogan, T.M., Mc Sweeney, P.L.H., Eds.; Springer: Boston, MA, USA, 2017; pp. 443–474. [Google Scholar]
- Smit, G.; Smit, B.A.; Engels, W.J.M. Flavour formation by lactic acid bacteria and biochemical flavour profiling of cheese products. FEMS Microbiol. Rev. 2005, 29, 591–610. [Google Scholar] [CrossRef]
- Blaya, J.; Barzideh, Z.; LaPointe, G. Symposium review: Interaction of starter cultures and nonstarter lactic acid bacteria in the cheese environment. J. Dairy Sci. 2018, 101, 3611–3629. [Google Scholar] [CrossRef]
- Pretorius, N.; Engelbrecht, L.; Du Toit, M. Influence of sugars and pH on the citrate metabolism of different lactic acid bacteria strains in a synthetic wine matrix. J. Appl. Microbiol. 2019, 127, 1490–1500. [Google Scholar] [CrossRef]
- Garabal, J.I.; Rodríguez-Alonso, P.; Centeno, J.A. Characterization of lactic acid bacteria isolated from raw cows’ milk cheeses currently produced in Galicia (NW Spain). LWT-Food Sci. Technol. 2008, 41, 1452–1458. [Google Scholar] [CrossRef]
- Sarantinopoulos, P.; Kalantzopoulos, G.; Tsakalidou, E. Citrate metabolism by Enterococcus faecalis FAIR-E 229. Appl. Environ. Microbiol. 2001, 67, 5482–5487. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas, N.; Arroyo, R.; Calzada, J.; Peirotén, A.; Medina, M.; Rodríguez, J.M.; Fernández, L. Evaluation of technological properties of Enterococcus faecium CECT 8849, a strain isolated from human milk, for the dairy industry. Appl. Microbiol. Biotechnol. 2016, 100, 7665–7677. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Arribas, P.; Seseña, S.; Poveda, J.M.; Chicón, R.; Cabezas, L.; Palop, L. Enterococcus populations in artisanal Manchego cheese: Biodiversity, technological and safety aspects. Food Microbiol. 2011, 28, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Serio, A.; Chaves-López, C.; Paparella, A.; Suzzi, G. Evaluation of metabolic activities of enterococci isolated from Pecorino Abruzzese cheese. Int. Dairy J. 2010, 20, 459–464. [Google Scholar] [CrossRef]
- Baccouri, O.; Boukerb, A.M.; Farhat, L.B.; Zébré, A.; Zimmermann, K.; Domann, E.; Cambronel, M.; Barreau, M.; Maillot, O.; Rincé, I.; et al. Probiotic potential and safety evaluation of Enterococcus faecalis OB14 and OB15, isolated from traditional Tunisian Testouri cheese and Rigouta, using physiological and genomic analysis. Front. Microbiol. 2019, 10, 881. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations; World Health Organization. Probiotics in Food: Health and Nutritional Properties and Guidelines for Evaluation; FAO food and nutrition paper; Food and Agriculture Organization of the United Nations: Rome, Italy; World Health Organization: Rome, Italy, 2006; pp. 2–3. ISBN 978-92-5-105513-7. [Google Scholar]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Nishiyama, K.; Sugiyama, M.; Mukai, T. Adhesion properties of lactic acid bacteria on intestinal mucin. Microorganisms 2016, 4, 34. [Google Scholar] [CrossRef]
- Ness, I.F.; Diep, D.B.; Ike, Y. Enterococcal bacteriocins and antimicrobial proteins that contribute to niche control. In Enterococci: From Commensals to Leading Causes of Drug Resistant Infection (Internet); Gilmore, M.S., Clewell, D.B., Ike, Y., Shankar, N., Eds.; Massachusetts Eye and Ear Infirmary: Boston, MA, USA, 2014; pp. 1–33. [Google Scholar]
- Ramiah, K.; Ten Doeschate, K.; Smith, R.; Dicks, L.M.T. Safety assessment of Lactobacillus plantarum 423 and Enterococcus mundtii ST4SA determined in trials with Wistar rats. Probiotics Antimicrob. Proteins 2009, 1, 15–23. [Google Scholar] [CrossRef]
- Tosoni, N.F.; Perini, H.F.; Terra, M.R.; Katsuda, M.S.; Furlaneto, M.C.; Maia, L.F. Antimicrobial activity of enterocin obtained from Enterococcus durans on Shiga-like toxin-producing Escherichia coli. Ciência Rural 2019, 49, e20190297. [Google Scholar] [CrossRef]
- Pieniz, S.; Andreazza, R.; Anghinoni, T.; Camargo, F.; Brandelli, A. Probiotic potential, antimicrobial and antioxidant activities of Enterococcus durans strain LAB18s. Food Control 2015, 37, 251–256. [Google Scholar] [CrossRef]
- Vimont, A.; Fernandez, B.; Hammami, R.; Ababsa, A.; Daba, H.; Fliss, I. Bacteriocin-producing LCW 44: A high potential probiotic candidate from raw Camel milk. Front. Microbiol. 2017, 8, 865. [Google Scholar] [CrossRef]
- Furlaneto-Maia, L.; Ramalho, R.; Rocha, K.R.; Furlaneto, M.C. Antimicrobial activity of enterocins against Listeria sp. and other food spoilage bacteria. Biotechnol. Lett. 2020, 42, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Živković, M.; Miljković, M.S.; Ruas-Madiedo, P.; Markelić, M.B.; Veljović, K.; Tolinački, M.; Soković, S.; Korać, A.; Golić, N. EPS-SJ exopolisaccharide produced by the strain Lactobacillus paracasei subsp. paracasei BGSJ2-8 is involved in adhesion to epithelial intestinal cells and decrease on E. coli association to Caco-2 cells. Front. Microbiol. 2016, 7, 286. [Google Scholar]
- Hashem, Y.A.; Amin, H.M.; Essam, T.M.; Yassin, A.S.; Aziz, R.K. Biofilm formation in enterococci: Genotype-phenotype correlations and inhibition by vancomycin. Sci. Rep. 2017, 7, 5733. [Google Scholar] [CrossRef]
- Jin, L.; Marquardt, R.; Zhao, X. A strain of Enterococcus faecium (18C23) inhibits adhesion of enterotoxigenic Escherichia coli K88 to porcine small intestine mucus. Appl. Environ. Microbiol. 2000, 66, 4200–4204. [Google Scholar] [CrossRef] [PubMed]
- Luissint, A.-C.; Parkos, C.A.; Nusrat, A. Inflammation and the intestinal barrier: Leukocyte–epithelial cell interactions, cell junction remodeling, and mucosal repair. Gastroenterology 2016, 151, 616–632. [Google Scholar] [CrossRef]
- Drolia, R.; Tenguria, S.; Durkes, A.C.; Turner, J.R.; Bhunia, A.K. Listeria adhesion protein induces intestinal epithelial barrier dysfunction for bacterial translocation. Cell Host Microbe 2018, 23, 470–484. [Google Scholar] [CrossRef]
- Bednorz, C.; Guenther, S.; Oelgeschläger, K.; Kinnemann, B.; Pieper, R.; Hartmann, S.; Tedin, K.; Semmler, T.; Neumann, K.; Schierack, P.; et al. Feeding the probiotic Enterococcus faecium strain NCIMB 10415 to piglets specifically reduces the number of Escherichia coli pathotypes that adhere to the gut mucosa. Appl. Environ. Microbiol. 2013, 79, 7896–7904. [Google Scholar] [CrossRef]
- Vijay, K. Toll-like receptors in immunity and inflammatory diseases: Past, present, and future. Int. Immunopharmacol. 2018, 62, 338. [Google Scholar] [CrossRef]
- D’Orazio, S. Innate and adaptive immune responses during Listeria monocytogenes infection. Microbiol. Spectr. 2019, 7, GPP3-0065-2019. [Google Scholar] [CrossRef]
- Onyiah, J.C.; Colgan, S.P. Cytokine responses and epithelial function in the intestinal mucosa. Cell Mol. Life Sci. 2016, 73, 4203–4212. [Google Scholar] [CrossRef]
- Rochman, Y.; Spolski, R.; Leonard, W.J. New insights into the regulation of T cells by gamma(c) family cytokines. Nat. Rev. Immunol. 2009, 9, 480–490. [Google Scholar] [CrossRef] [PubMed]
- Ihara, S.; Hirata, Y.; Koike, K. TGF-β in inflammatory bowel disease: A key regulator of immune cells, epithelium, and the intestinal microbiota. J. Gastroenterol. 2017, 52, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Rachakonda, G.; Vu, T.; Jin, L.; Samanta, D.; Datta, P.K. Role of TGF-β-induced Claudin-4 expression through c-Jun signaling in non-small cell lung cancer. Cell. Signal. 2016, 28, 1537–1544. [Google Scholar] [CrossRef] [PubMed]
- Sparo, M.; Delpech, G.; Batisttelli, S.; Basualdo, J.A. Immunomodulatory properties of cell wall extract from Enterococcus faecalis CECT7121. Braz. J. Infect. Dis. 2014, 18, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Carasi, P.; Racedo, S.M.; Jacquot, C.; Elie, A.M.; Serradell, M.L.; Urdaci, M.C. Enterococcus durans EP1 a promising anti-inflammatory probiotic able to stimulate sIgA and to increase Faecalibacterium prausnitzii abundance. Front. Immunol. 2017, 8, 88. [Google Scholar] [CrossRef]
- Torres, D.; Barrier, M.; Bihl, F.; Quesniaux, V.J.; Maillet, I.; Akira, S.; Ryffel, B.; Erard, F. Toll-like receptor 2 is required for optimal control of Listeria monocytogenes infection. Infect. Immun. 2004, 72, 2131–2139. [Google Scholar] [CrossRef]
- Avram-Hananel, L.; Stock, J.; Parlesak, A.; Bode, C.; Schwartz, B. E durans strain M4-5 isolated from human colonic flora attenuates intestinal inflammation. Dis. Colon Rectum 2010, 53, 1676–1686. [Google Scholar] [CrossRef] [PubMed]
- Hlivak, P.; Odraska, J.; Ferencik, M.; Ebringer, L.; Jahnova, E.; Mikes, Z. One-year application of probiotic strain Enterococcus faecium M-74 decreases serum cholesterol levels. Bratisl Lek Listy 2005, 106, 67–72. [Google Scholar] [PubMed]
- Yamaguchi, T.; Miura, Y.; Matsumoto, T. Antimicrobial susceptibility of Enterococcus strains used in clinical practice as probiotics. J. Infect. Chemother. 2013, 19, 1109–1115. [Google Scholar] [CrossRef]
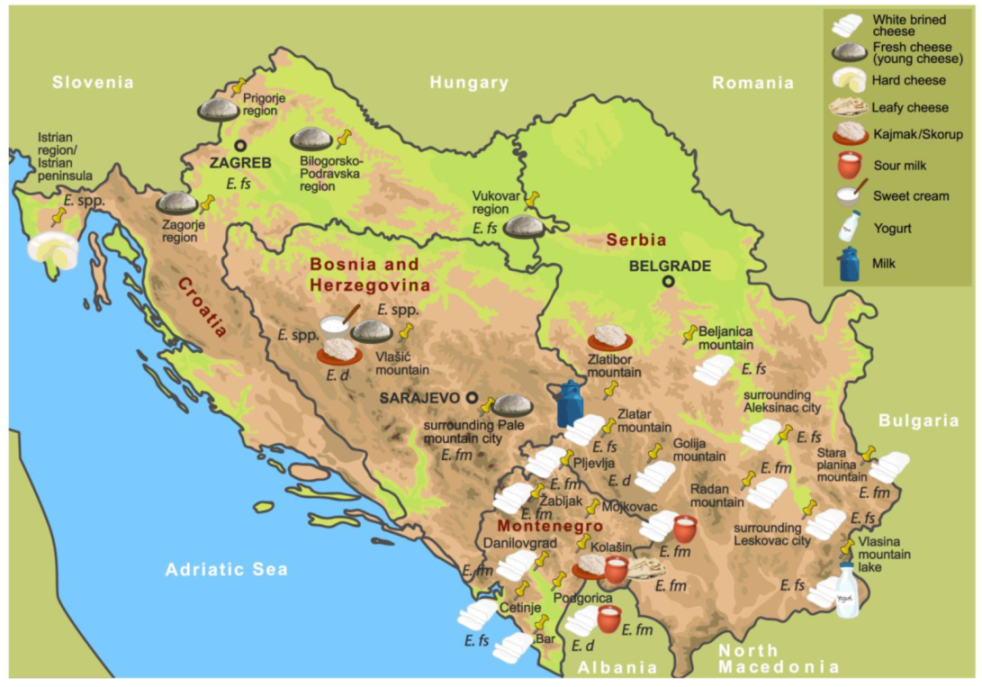
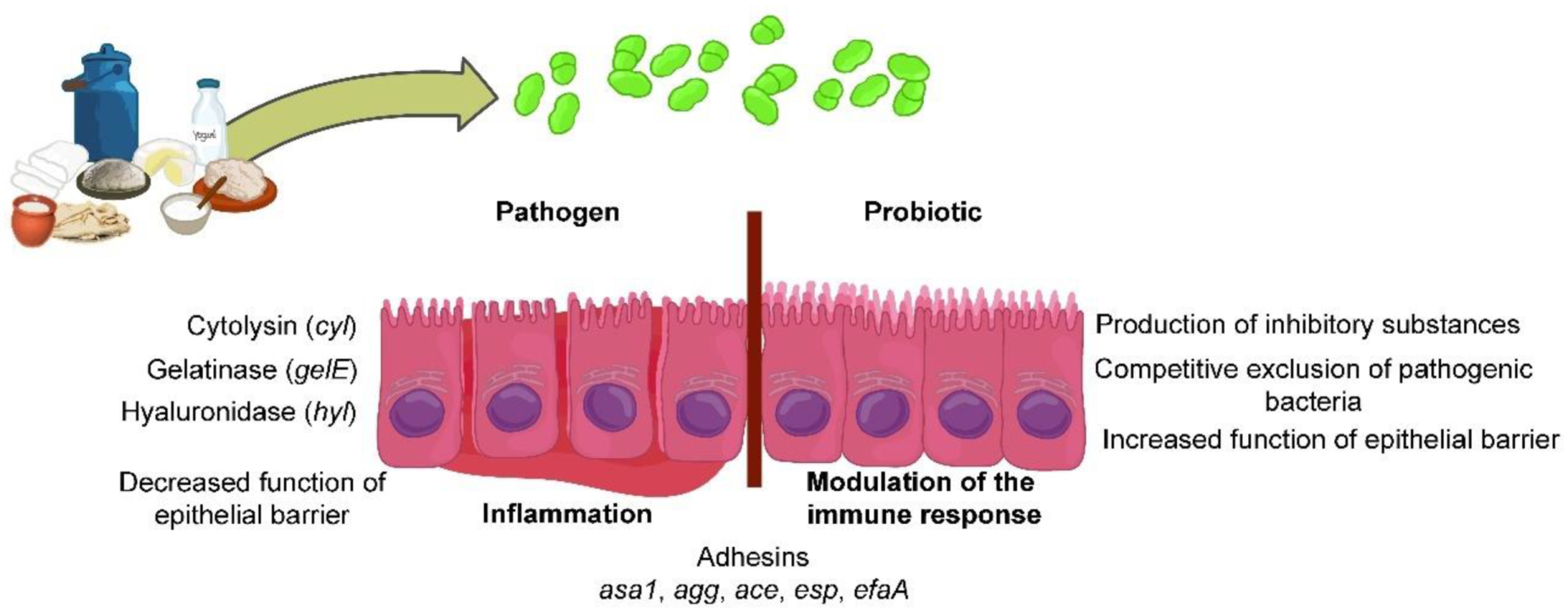
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terzić-Vidojević, A.; Veljović, K.; Popović, N.; Tolinački, M.; Golić, N. Enterococci from Raw-Milk Cheeses: Current Knowledge on Safety, Technological, and Probiotic Concerns. Foods 2021, 10, 2753. https://doi.org/10.3390/foods10112753
Terzić-Vidojević A, Veljović K, Popović N, Tolinački M, Golić N. Enterococci from Raw-Milk Cheeses: Current Knowledge on Safety, Technological, and Probiotic Concerns. Foods. 2021; 10(11):2753. https://doi.org/10.3390/foods10112753
Chicago/Turabian StyleTerzić-Vidojević, Amarela, Katarina Veljović, Nikola Popović, Maja Tolinački, and Nataša Golić. 2021. "Enterococci from Raw-Milk Cheeses: Current Knowledge on Safety, Technological, and Probiotic Concerns" Foods 10, no. 11: 2753. https://doi.org/10.3390/foods10112753
APA StyleTerzić-Vidojević, A., Veljović, K., Popović, N., Tolinački, M., & Golić, N. (2021). Enterococci from Raw-Milk Cheeses: Current Knowledge on Safety, Technological, and Probiotic Concerns. Foods, 10(11), 2753. https://doi.org/10.3390/foods10112753






