The Influence of Fermenting Yeast on the Sensory Properties of Graševina Wine
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparing the Must and Fermentation
- -
- the must was left for 3 h to clarify;
- -
- after 3 h the clarified must was decanted into two tanks with a volume of 2630 L;
- -
- each tank was inoculated with the selected yeasts (Sample One and Sample Two).
2.2. Vinification and Fermentation
2.3. Analysis and Sensory Evaluation
2.3.1. Physical–Chemical Analysis
2.3.2. Sensory Evaluation
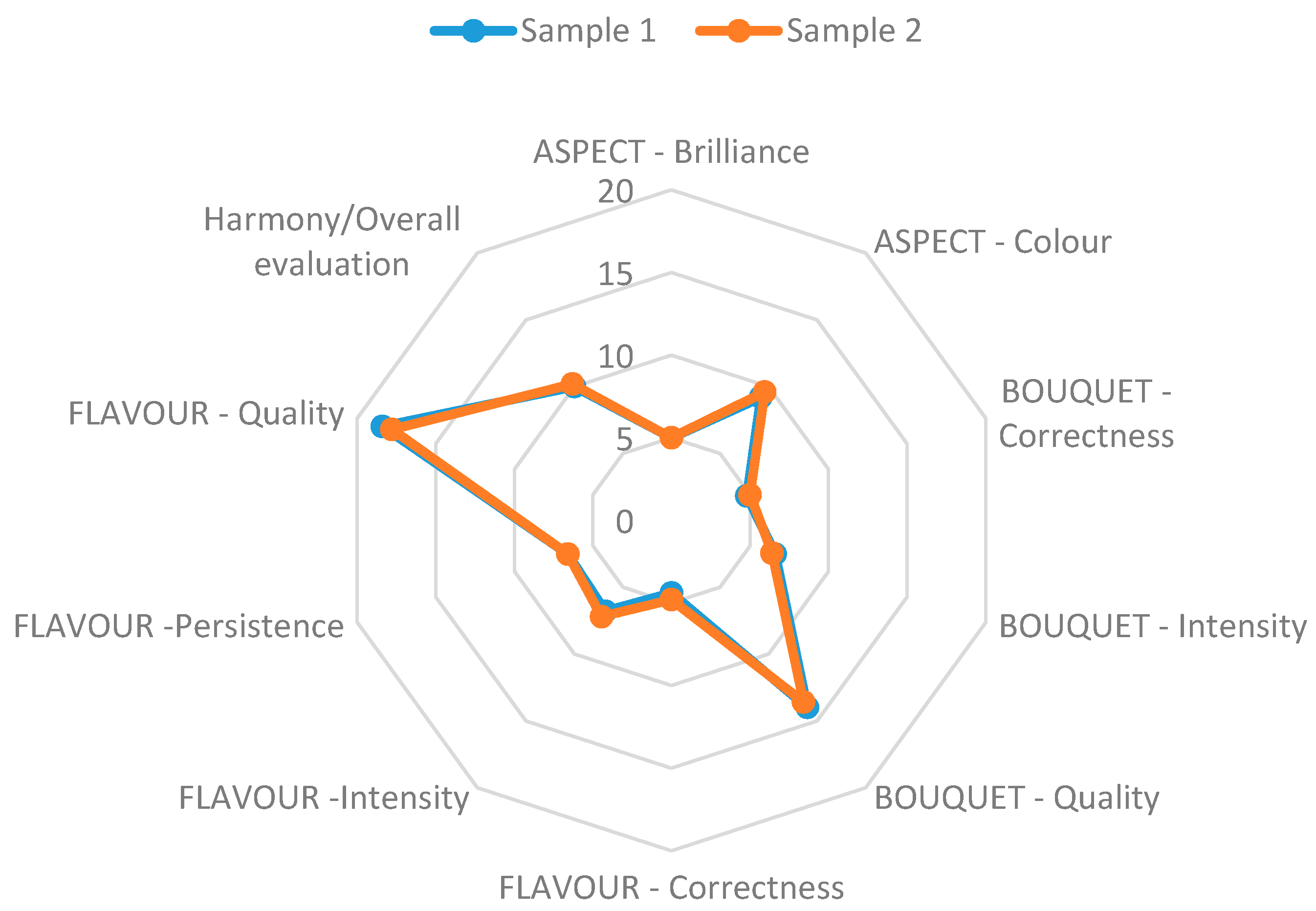
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Padilla, B.; Gil, J.V.; Manzanares, P. Past and future of non-Saccharomyces yeasts: From spoilage microorganisms to biotechnological tools for improving wine aroma complexity. Front. Microbiol. 2016, 7, 411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurtzman, C.P.; Fell, J.W.; Boekhout, T. Definition, classification and nomenclature of the yeasts. In The Yeasts; Elsevier: Amsterdam, The Netherlands, 2011; pp. 3–5. [Google Scholar]
- Romano, P.; Ciani, M.; Fleet, G.H. Yeasts in the Production of Wine; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Di Maro, E.; Ercolini, D.; Coppola, S. Yeast dynamics during spontaneous wine fermentation of the Catalanesca grape. Int. J. Food Microbiol. 2007, 117, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Borren, E.; Tian, B. The Important Contribution of Non-Saccharomyces Yeasts to the Aroma Complexity of Wine: A Review. Foods 2021, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Morata, A.; Loira, I.; Escott, C.; del Fresno, J.M.; Bañuelos, M.A.; Suárez-Lepe, J.A. Applications of Metschnikowia pulcherrima in wine biotechnology. Fermentation 2019, 5, 63. [Google Scholar] [CrossRef] [Green Version]
- Barata, A.; Malfeito-Ferreira, M.; Loureiro, V. The microbial ecology of wine grape berries. Int. J. Food Microbiol. 2012, 153, 243–259. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, M.; Velázquez, R. The yeast Torulaspora delbrueckii: An interesting but difficult-to-use tool for winemaking. Fermentation 2018, 4, 94. [Google Scholar] [CrossRef] [Green Version]
- González-Royo, E.; Pascual, O.; Kontoudakis, N.; Esteruelas, M.; Esteve-Zarzoso, B.; Mas, A.; Canals, J.M.; Zamora, F. Oenological consequences of sequential inoculation with non-Saccharomyces yeasts (Torulaspora delbrueckii or Metschnikowia pulcherrima) and Saccharomyces cerevisiae in base wine for sparkling wine production. Eur. Food Res. Technol. 2015, 240, 999–1012. [Google Scholar] [CrossRef]
- Ramírez, M.; Velázquez, R.; Maqueda, M.; López-Piñeiro, A.; Ribas, J.C. A new wine Torulaspora delbrueckii killer strain with broad antifungal activity and its toxin-encoding double-stranded RNA virus. Front. Microbiol. 2015, 6, 983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Velázquez, R.; Zamora, E.; Álvarez, M.L.; Hernández, L.M.; Ramírez, M. Effects of new Torulaspora delbrueckii killer yeasts on the must fermentation kinetics and aroma compounds of white table wine. Front. Microbiol. 2015, 6, 1222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanl, L.; Sommer, P.; Arneborg, N. The effect of decreasing oxygen feed rates on growth and metabolism of Torulaspora delbrueckii. Appl. Microbiol. Biotechnol. 2005, 67, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Panić, M.; Gunjević, V.; Radošević, K.; Cvjetko Bubalo, M.; Ganić, K.K.; Redovniković, I.R. COSMOtherm as an Effective Tool for Selection of Deep Eutectic Solvents Based Ready-To-Use Extracts from Graševina Grape Pomace. Molecules 2021, 26, 4722. [Google Scholar] [CrossRef] [PubMed]
- Deak, I. Influence of Yeasts of the Genus Saccharomyces and Torulaspora on Chemical and Sensory Properties of Graševina Wine. Bachelor’s Thesis, Polytechnic in Požega, Požega, Slavonia, 18 March 2013. [Google Scholar]
- Ripper Method for SO2. Available online: https://www.enartis.com/wp-content/uploads/2020/04/SO2-Free-by-Ripper.pdf (accessed on 7 October 2021).
- Ebilliometer, Salleron. Available online: https://www.enartis.com/wp-content/uploads/2020/04/Alcohol-Burner-Ebulliometer.pdf (accessed on 7 October 2020).
- Andorra, I.; Berradre, M.; Mas, A.; Esteve-Zarzoso, B.; Guillamon, J.M. Effect of mixed culture fermentations on yeast populations and aroma profile. LWT Food Sci. Technol. 2012, 49, 8–13. [Google Scholar] [CrossRef]
- Azzolini, M.; Tosi, E.; Lorenzini, M.; Finato, F.; Zapparoli, G. Contribution to the aroma of white wines by controlled Torulaspora delbrueckii cultures in association with Saccharomyces cerevisiae. World J. Microbiol. Biotechnol. 2015, 31, 277–293. [Google Scholar] [CrossRef] [PubMed]
- The Use of Sulphur Dioxide in Must and Wine. Available online: http://www.uwe-hofmann.org/The%20use%20of%20Sulphur%20Dioxide.pdf (accessed on 27 October 2021).
- Herraiz, G.; Reglero, M.; Herraiz, P.; Alvarez, M.; Cabezudo, M. The influence of the yeast and type of culture on the volatile composition of wine fermented without sulphur dioxide. Am. J. Enol. Viticult. 1990, 41, 313–318. [Google Scholar]
- Bely, M.; Stoeckle, P.; Masneuf-Pomarède, I.; Dubourdieu, D. Impact of mixed Torulaspora delbrueckii–Saccharomyces cerevisiae culture on high-sugar fermentation. Int. J. Food Microbiol. 2008, 122, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Ciani, M.; Beco, L.; Comitini, F. Fermentation behaviour and metabolic interactions of multistarter wine yeast fermentations. Int. J. Food Microbiol. 2005, 108, 239–245. [Google Scholar] [CrossRef] [PubMed]
| Component | Amount |
|---|---|
| Sugars | 100 °Oe |
| Total acids | 6.3 g/L |
| Free SO2 | 11.52 mg/L |
| pH | 3.30 |
| Time after Inoculation (Days) | Temperature (°C) | Unfermented Sugars (°Oe) | ||
|---|---|---|---|---|
| Sample One | Sample Two | Sample One | Sample Two | |
| 7 | 15.4 | 14.6 | 74 | 70 |
| 8 | 15.1 | 14.9 | 65 | 50 |
| 9 | 15.1 | 14.9 | 55 | 41 |
| 10 | 15.2 | 14.7 | 48 | 37 |
| 13 | 14.1 | 13.5 | 46 | 34 |
| 14 | 16.0 | 12.5 | 37 | 33 |
| 18 | 15.7 | 12.1 | 32 | 33 |
| 21 | 15.7 | 12.5 | 30 | 32 |
| 25 | 15.6 | 12.5 | 30 | 32 |
| Component Results | ||
|---|---|---|
| 45 Days into Fermentation | ||
| Sample One | Sample Two | |
| Free sulphor mg/L | 16.64 a ± 0.35 | 15.36 b ± 0.41 |
| Volatile acids g/L | 0.34 a ± 0.08 | 0.31 a ± 0.09 |
| Total acids g/L | 5.91 a ± 0.13 | 6.05 a ± 0.11 |
| pH | 3.39 a ± 0.12 | 3.32 a ± 0.09 |
| Ethanol % | 14.2 a ± 0.25 | 13.8 a ± 0.31 |
| 160 Days into Fermentation | ||
| Free sulphor mg/L | 19.20 b ± 0.33 | 20.48 a ± 0.40 |
| Volatile acids g/L | 0.37 b ± 0.11 | 0.54 a ± 0.08 |
| Total acids g/L | 5.61 a ± 0.16 | 5.92 a ± 0.14 |
| pH | 3.34 a ± 0.17 | 3.35 a ± 0.15 |
| Ethanol % | 14.2 a ± 0.25 | 13.8 a ± 0.31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deak, I.; Habschied, K.; Mesić, J.; Babić, J.; Kovačević, D.; Nedović, V.; Mastanjević, K. The Influence of Fermenting Yeast on the Sensory Properties of Graševina Wine. Foods 2021, 10, 2752. https://doi.org/10.3390/foods10112752
Deak I, Habschied K, Mesić J, Babić J, Kovačević D, Nedović V, Mastanjević K. The Influence of Fermenting Yeast on the Sensory Properties of Graševina Wine. Foods. 2021; 10(11):2752. https://doi.org/10.3390/foods10112752
Chicago/Turabian StyleDeak, Igor, Kristina Habschied, Josip Mesić, Jurislav Babić, Dragan Kovačević, Viktor Nedović, and Krešimir Mastanjević. 2021. "The Influence of Fermenting Yeast on the Sensory Properties of Graševina Wine" Foods 10, no. 11: 2752. https://doi.org/10.3390/foods10112752
APA StyleDeak, I., Habschied, K., Mesić, J., Babić, J., Kovačević, D., Nedović, V., & Mastanjević, K. (2021). The Influence of Fermenting Yeast on the Sensory Properties of Graševina Wine. Foods, 10(11), 2752. https://doi.org/10.3390/foods10112752









