Presence of Parabens and Bisphenols in Food Commonly Consumed in Spain
Abstract
1. Introduction
2. Material and Methods
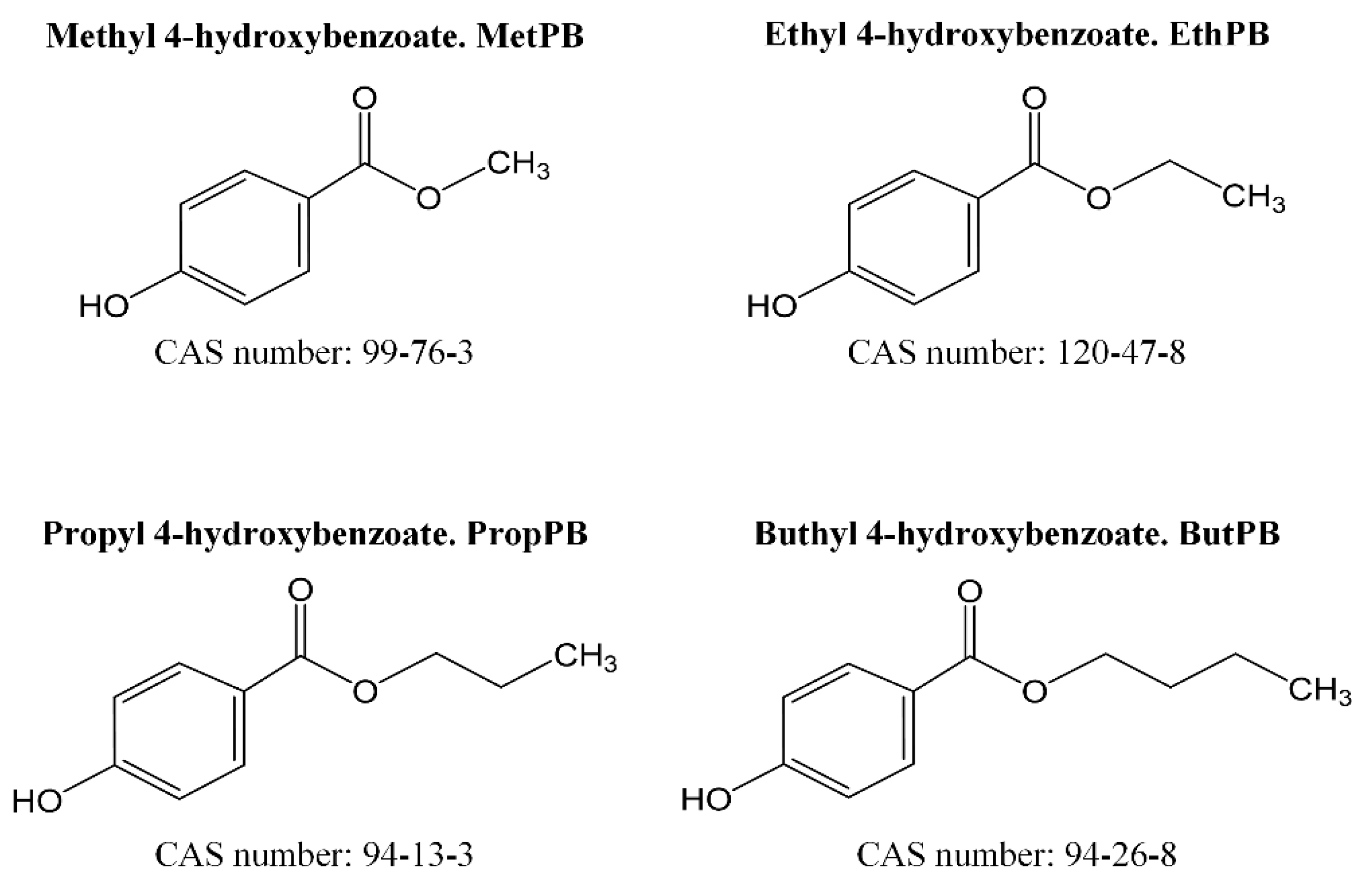
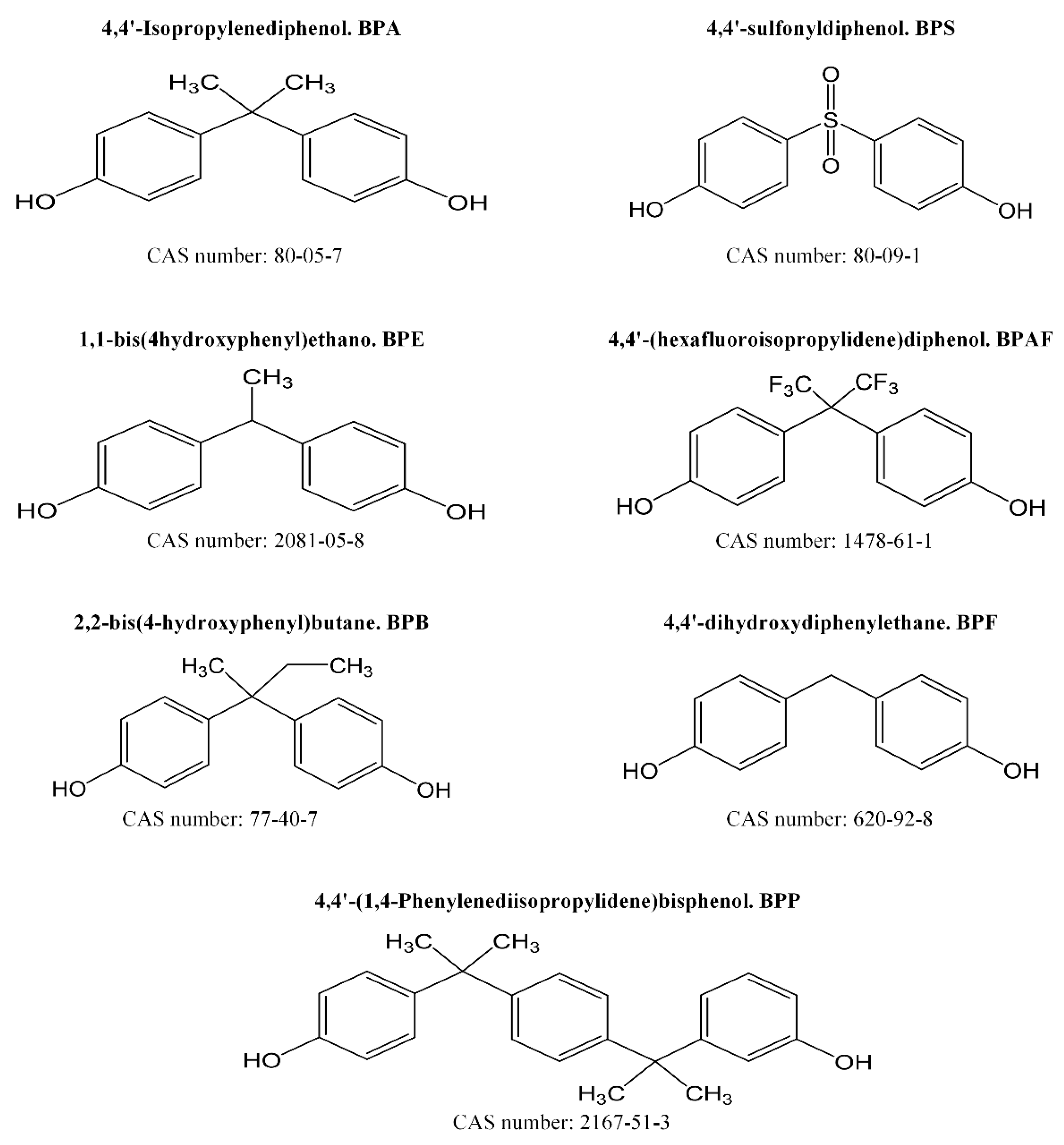
2.1. Chemicals
2.2. Instrumentation and Software
2.3. Food Sampling
2.4. Analytical Methods
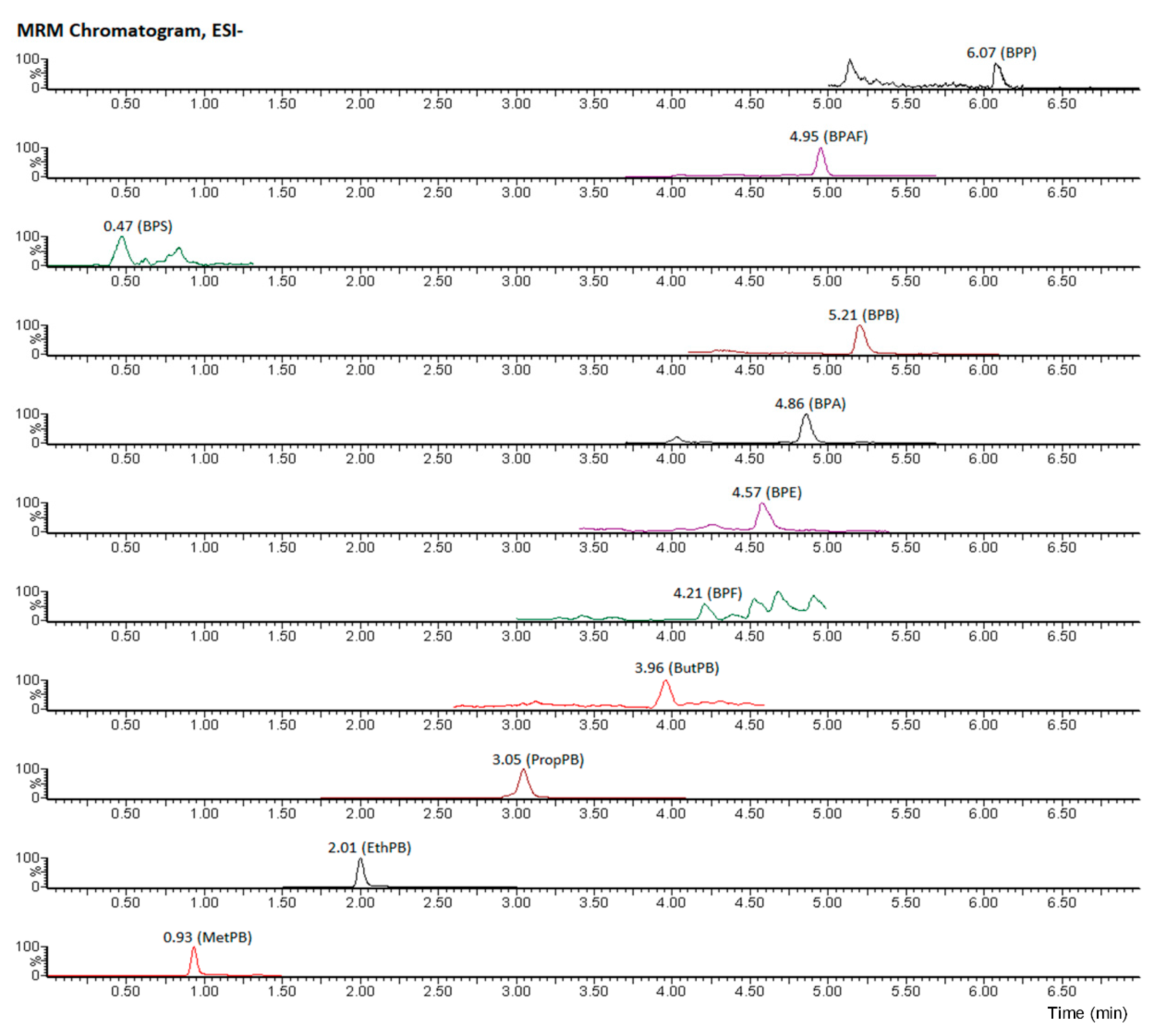
2.5. Ultrahigh Performance Liquid Chromatography—Tandem Mass Spectrometry Analysis
2.6. Method Validation
2.7. Estimation of Dietary Exposure in Spanish Children
2.8. Statistics
3. Results and Discussion
3.1. Parabens in Food Samples
3.2. Bisphenols in Food Samples
3.3. Estimated Dietary Intake in Children
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BPA | Bisphenol A |
| AESAN | Spanish Agency for Food Safety and Nutrition |
| BPA-d16 | Deuterium labelled bisphenol A |
| BPAF | Bisphenol AF |
| BPAP | Bisphenol AP |
| BPB | Bisphenol B |
| BPE | Bisphenol E |
| BPF | Bisphenol F |
| BPM | Bisphenol M |
| BPP | Bisphenol P |
| BPS | Bisphenol S |
| BPZ | Bisphenol Z |
| ButPB | Butyl 4-hydroxybenzoate |
| bw | Body weight |
| C18 | Octadecyl-functionalized silica |
| ECHA | European Chemical Agency |
| EFSA | European Food Safety Authority |
| ESI | Electrospray ionization source |
| EthPB | Ethyl 4-hydroxybenzoate |
| EthPB-d5 | Deuterium labelled ethylparaben |
| LC-MS | Liquid chromatography-mass spectrometry |
| LLE | Liquid-liquid extraction |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| MeOH | Methanol |
| MetPB | Methyl 4-hydroxybenzoate |
| MgSO4 | Magnesium sulphate |
| MPLs | Maximum permitted levels |
| MRM | Multiple-reaction-monitoring |
| NaCl | Sodium chloride |
| PropPB | Propyl 4-hydroxybenzoate |
| PSA | Primary Secondary Amine |
| QuEChERs | Quick, Easy, Cheap, Effective, Rugged, and Safe |
| SCCS | Scientific Committee on Consumer Safety |
| SPE | Solid phase extraction |
| TDI | Tolerable dietary intake |
| UHPLC-MS/MS | Ultra-high performance liquid chromatography-tandem mass spectrometry |
References
- Fransway, A.F.; Fransway, P.J.; Belsito, D.V.; Warshaw, E.M.; Sasseville, D.; Fowler, J.F., Jr.; DeKoven, J.G.; Pratt, M.D.; Maibach, H.I.; Taylor, J.S.; et al. Parabens. Dermatitis 2019, 30, 3–31. [Google Scholar] [CrossRef]
- Liao, C.; Kannan, K. Occurrence of and Dietary Exposure to Parabens in Foodstuffs from the United States. Environ. Sci. Technol. 2013, 47, 3918–3925. [Google Scholar] [CrossRef]
- Liao, C.; Chen, L.; Kannan, K. Occurrence of Parabens in Foodstuffs from China and its Implications for Human Dietary Exposure. Environ. Int. 2013, 57–58, 68–74. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EU) No 1004/2004. of 18 September 2014 amending Annex V to Regulation (EC) No 1223/2009 of the European Parliament and of the Council on cosmetic products. Off. J. Eur. Union 2014, L282, 5–8. [Google Scholar]
- Golden, R.; Gandy, J.; Vollmer, G. A Review of the Endocrine Activity of Parabens and Implications for Potential Risks to Human Health. Crit. Rev. Toxicol. 2005, 35, 435–458. [Google Scholar] [CrossRef]
- Boberg, J.; Taxvig, C.; Christiansen, S.; Hass, U. Possible Endocrine Disrupting Effects of Parabens and their Metabolites. Reprod. Toxicol. 2010, 30, 301–312. [Google Scholar] [CrossRef]
- European Commission. Scientific Committee on Consumer Safety (SCCS). Opinion on Parabens. SCCS/1348/10, Revision 22 March 2011. 36p, Available online: https://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_041.pdf (accessed on 10 December 2020).
- European Food Safety Authority (EFSA). EFSA Advises on the Safety of Paraben Usage in Food. European Union, 29 September 2004; Available online: https://www.efsa.europa.eu/en/news/efsa-advises-safety-paraben-usage-food (accessed on 10 December 2020).
- European Medicines Agency. European Public MRL Assessment Report (EPMAR) Propyl 4-Hydroxybenzoate and Its Sodium Salt (All Food Producing Species). EMA/CVMP/632934/2014. 8 September 2015; 9p, Available online: https://www.ema.europa.eu/en/documents/mrl-report/propyl-4-hydroxybenzoate-its-sodium-salt-all-food-producing-species-european-public-mrl-assessment_en.pdf (accessed on 10 December 2020).
- Nobile, M.; Arioli, F.; Pavlovic, R.; Ceriani, F.; Lin, S.K.; Panseri, S.; Villa, R.; Chiesa, L.M. Presence of emerging contaminants in baby food. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2020, 37, 131–142. [Google Scholar] [CrossRef]
- Pradhan, R.; Hejmady, S.; Taliyan, R.; Khadgawat, R.; Gupta, T.; Kachhawa, G.; Kumar, R.; Singhvi, G.; Dubey, S.K. Simultaneous Estimation of Parabens and Bisphenol a in Ready-to-Eat Foodstuffs by using QbD-Driven High-Pressure Liquid Chromatography Method. Int. J. Environ. Anal. Chem. 2020. [Google Scholar] [CrossRef]
- Huang, Y.Q.; Wong, C.K.; Zheng, J.S.; Bouwman, H.; Barra, R.; Wahlström, B.; Neretin, L.; Wong, M.H. Bisphenol A (BPA) in China: A review of sources, environmental levels, and potential human health impacts. Environ. Int. 2012, 42, 91–99. [Google Scholar] [CrossRef]
- Juan-García, A.; Gallego, C.; Font, G. Toxicidad Del Bisfenol A: Revisión. Rev. Toxicol. 2015, 32, 144–160. [Google Scholar]
- Rochester, J.R. Bisphenol A and Human Health: A Review of the Literature. Reprod. Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef]
- Food and Drug Administration (FDA). Indirect Food Additives: Polymers. Fed. Regist. 2012, 77, 41899–41902.
- Food and Drug Administration (FDA). Indirect Food Additives: Adhesives and Components of Coatings. Fed. Regist. 2013, 78, 41840–41843. [Google Scholar]
- French Republic. Regulation No. 1442/2012 of 24 December 2012 Aiming at Banning the Manufacture, Import, Export and Commercialisation of all Forms of Food Packaging Containing Bisphenol A. Off. J. Fr. Repub. 2012. text 2 of 154. [Google Scholar]
- European Commission. Commission Regulation (EU) 2018/213 of 12 February 2018 on the use of bisphenol A in varnishes and coatings intended to come into contact with food and amending Regulation (EU) No 10/2011 as regards the use of that substance in plastic food contact materials. Off. J. Eur. Union. 2018, L41, 6–12. [Google Scholar]
- Garcia-Córcoles, M.T.; Cipa, M.; Rodriguez-Gomez, R.; Rivas, A.; Olea-Serrano, F.; Vilchez, J.L.; Zafra-Gomez, A. Determination of Bisphenols with Estrogenic Activity in Plastic Packaged Baby Food Samples using Solid-Liquid Extraction and Clean-Up with Dispersive Sorbents Followed by Gas Chromatography Tandem Mass Spectrometry Analysis. Talanta 2018, 178, 441–448. [Google Scholar] [CrossRef]
- Gallo, P.; Di Marco Pisciottano, I.; Esposito, F.; Fasano, E.; Scognamiglio, G.; Mita, G.D.; Cirillo, T. Determination of BPA, BPB, BPF, BADGE and BFDGE in Canned Energy Drinks by Molecularly Imprinted Polymer Cleaning up and UPLC with Fluorescence Detection. Food Chem. 2017, 220, 406–412. [Google Scholar] [CrossRef]
- Liao, C.; Kannan, K. Concentrations and Profiles of Bisphenol A and Other Bisphenol Analogues in Foodstuffs from the United States and their Implications for Human Exposure. J. Agric. Food Chem. 2013, 61, 4655–4662. [Google Scholar] [CrossRef]
- Danzl, E.; Sei, K.; Soda, S.; Ike, M.; Fujita, M. Biodegradation of Bisphenol A, Bisphenol F and Bisphenol S in Seawater. Int. J. Environ. Res. Public Health 2009, 6, 1472–1484. [Google Scholar] [CrossRef]
- Feng, Y.; Yin, J.; Jiao, Z.; Shi, J.; Li, M.; Shao, B. Bisphenol AF may Cause Testosterone Reduction by Directly Affecting Testis Function in Adult Male Rats. Toxicol. Lett. 2012, 211, 201–209. [Google Scholar] [CrossRef]
- Kitamura, S.; Suzuki, T.; Sanoh, S.; Kohta, R.; Jinno, N.; Sugihara, K.; Yoshihara, S.; Fujimoto, N.; Watanabe, H.; Ohta, S. Comparative Study of the Endocrine-Disrupting Activity of Bisphenol A and 19 Related Compounds. Toxicol. Sci. 2005, 84, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.; Barbato, F.; Mita, D.G.; Grumetto, L. Occurrence of Bisphenol A and its analogues in some foodstuff marketed in Europe. Food Chem. Toxicol. 2019, 131, 110575. [Google Scholar] [CrossRef] [PubMed]
- Gallart-Ayala, H.; Moyano, E.; Galceran, M.T. Fast Liquid Chromatography-Tandem Mass Spectrometry for the Analysis of Bisphenol A-Diglycidyl Ether, Bisphenol F-Diglycidyl Ether and their Derivatives in Canned Food and Beverages. J. Chromatogr. A 2011, 1218, 1603–1610. [Google Scholar] [CrossRef] [PubMed]
- Andujar, N.; Galvez-Ontiveros, Y.; Zafra-Gomez, A.; Rodrigo, L.; Jesus Alvarez-Cubero, M.; Aguilera, M.; Monteagudo, C.; Rivas, A. Bisphenol A Analogues in Food and their Hormonal and Obesogenic Effects: A Review. Nutrients 2019, 11, 2136. [Google Scholar] [CrossRef] [PubMed]
- Rivas, A.; Lacroix, M.; Olea-Serrano, F.; Laios, I.; Leclercq, G.; Olea, N. Estrogenic Effect of a Series of Bisphenol Analogues on Gene and Protein Expression in MCF-7 Breast Cancer Cells. J. Steroid Biochem. Mol. Biol. 2002, 82, 45–53. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Scientific Opinion. Scientific Opinion on the risk to public health related to the presence of bisphenol A (BPA) in foodstuffs: Executive summary. EFSA panel on food contact materials, enzymes, flavourings and processing aids (CEF). EFSA J. 2015, 13, 3978. [Google Scholar] [CrossRef]
- Beszterda, M.; Franski, R. Endocrine Disruptor Compounds in Environment: As a Danger for Children Health. Pediatr. Endocrinol. Diabetes Metab. 2018, 24, 88–95. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Cannon, G.; Moubarac, J.; Levy, R.B.; Louzada, M.L.C.; Jaime, P.C. The UN Decade of Nutrition, the NOVA Food Classification and the Trouble with Ultra-Processing. Public Health Nutr. 2018, 21, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Martín-Pozo, L.; Cantarero-Malagón, S.; Hidalgo, F.; Navalón, A.; Zafra-Gómez, A. Determination of endocrine disrupting chemicals in human nails using an alkaline digestion prior to ultra-high performance liquid chromatography–tandem mass spectrometry. Talanta 2020, 208, 120429. [Google Scholar] [CrossRef]
- López-Sobaler, A.M.; Aparicio, A.; Rubio, J.; Marcos, V.; Sanchidrián, R.; Santos, S.; Pérez-Farinós, N.; Dal-Re, M.Á.; Villar-Villalba, C.; Yusta-Boyo, M.J.; et al. Adequacy of usual macronutrient intake and macronutrient distribution in children and adolescents in Spain: A National Dietary Survey on the Child and Adolescent Population, ENALIA 2013–2014. Eur. J. Nutr. 2019, 58, 705–719. [Google Scholar] [CrossRef]
- Perez-Farinos, N.; Lopez-Sobaler, A.M.; Angeles Dal Re, M.; Villar, C.; Labrado, E.; Robledo, T.; Ortega, R.M. The ALADINO Study: A National Study of Prevalence of Overweight and Obesity in Spanish Children in 2011. Biomed Res. Int. 2013, 2013, 163687. [Google Scholar] [CrossRef] [PubMed]
- Soni, M.G.; Carabin, I.G.; Burdock, G.A. Safety Assessment of Esters of P-Hydroxybenzoic Acid (Parabens). Food Chem. Toxicol. 2005, 43, 985–1015. [Google Scholar] [CrossRef]
- Quiros-Alcala, L.; Buckley, J.P.; Boyle, M. Parabens and Measures of Adiposity among Adults and Children from the US General Population: NHANES 2007-2014. Int. J. Hyg. Environ. Health 2018, 221, 652–660. [Google Scholar] [CrossRef]
- Maher, H.M.; Alzoman, N.Z.; Almeshal, M.A.; Alotaibi, H.A.; Alotaibi, N.N.; Al-Showiman, H. Quantitative Screening of Parabens in Ready-to-Eat Foodstuffs Available in the Saudi Market using High Performance Liquid Chromatography with Photodiode Array Detection. Arab. J. Chem. 2020, 13, 2897–2911. [Google Scholar] [CrossRef]
- Alvarez-Muñoz, D.; Rodriguez-Mozaz, S.; Maulvault, A.L.; Tediosi, A.; Fernandez-Tejedor, M.; Van den Heuvel, F.; Kotterman, M.; Marques, A.; Barcelo, D. Occurrence of Pharmaceuticals and Endocrine Disrupting Compounds in Macroalgaes, Bivalves, and Fish from Coastal Areas in Europe. Environ. Res. 2015, 143, 56–64. [Google Scholar] [CrossRef]
- Alvarez-Muñoz, D.; Rodriguez-Mozaz, S.; Jacobs, S.; Serra-Compte, A.; Caceres, N.; Sioen, I.; Verbeke, W.; Barbosa, V.; Ferrari, F.; Fernandez-Tejedor, M.; et al. Pharmaceuticals and Endocrine Disruptors in Raw and Cooked Seafood from European Market: Concentrations and Human Exposure Levels. Environ. Int. 2018, 119, 570–581. [Google Scholar] [CrossRef]
- Azzouz, A.; Palacios Colon, L.; Souhail, B.; Ballesteros, E. A Multi-Residue Method for GC-MS Determination of Selected Endocrine Disrupting Chemicals in Fish and Seafood from European and North African Markets. Environ. Res. 2019, 178, 108727. [Google Scholar] [CrossRef]
- Chiesa, L.M.; Pavlovic, R.; Panseri, S.; Arioli, F. Evaluation of Parabens and their Metabolites in Fish and Fish Products: A Comprehensive Analytical Approach using LC-HRMS. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2018, 35, 2400–2413. [Google Scholar] [CrossRef]
- Villaverde-de-Sáa, E.; Rodil, R.; Quintana, J.B.; Cela, R. Matrix Solid-Phase Dispersion Combined to Liquid Chromatography–tandem Mass Spectrometry for the Determination of Paraben Preservatives in Mollusks. J. Chromatogr. A 2016, 1459, 57–66. [Google Scholar] [CrossRef]
- Jakimska, A.; Huerta, B.; Barganska, Z.; Kot-Wasik, A.; Rodriguez-Mozaz, S.; Barcelo, D. Development of a Liquid Chromatography-Tandem Mass Spectrometry Procedure for Determination of Endocrine Disrupting Compounds in Fish from Mediterranean Rivers. J. Chromatogr. A 2013, 1306, 44–58. [Google Scholar] [CrossRef]
- Pico, Y.; Belenguer, V.; Corcellas, C.; Diaz-Cruz, M.S.; Eljarrat, E.; Farre, M.; Gago-Ferrero, P.; Huerta, B.; Navarro-Ortega, A.; Petrovic, M.; et al. Contaminants of Emerging Concern in Freshwater Fish from Four Spanish Rivers. Sci. Total Environ. 2019, 659, 1186–1198. [Google Scholar] [CrossRef] [PubMed]
- Abril, C.; Martin, J.; Luis Malvar, J.; Luis Santos, J.; Aparicio, I.; Alonso, E. Dispersive Liquid-Liquid Microextraction as a New Clean-Up Procedure for the Determination of Parabens, Perfluorinated Compounds, UV Filters, Biocides, Surfactants, and Plasticizers in Root Vegetables. Anal. Bioanal. Chem. 2018, 410, 5155–5163. [Google Scholar] [CrossRef] [PubMed]
- Žnideršič, L.; Mlakar, A.; Prosen, H. Development of a SPME-GC-MS/MS Method for the Determination of some Contaminants from Food Contact Material in Beverages. Food Chem. Toxicol. 2019, 134, 110829. [Google Scholar] [CrossRef] [PubMed]
- Chung, D.W.; Papadakis, S.E.; Yam, K.L. Release of Propyl Paraben from a Polymer Coating into Water and Food Simulating Solvents for Antimicrobial Packaging Applications. J. Food Process. Preserv. 2001, 25, 71–87. [Google Scholar] [CrossRef]
- Lu, L.; Xiong, W.; Li, X.; Lv, S.; Tang, X.; Chen, M.; Zou, Z.; Lin, Z.; Qiu, B.; Chen, G. Determination of the Migration of Eight Parabens from Antibacterial Plastic Packaging by Liquid Chromatography-Electrospray Ionization-Tandem Mass Spectrometry. Anal. Methods 2014, 6, 2096–2101. [Google Scholar] [CrossRef]
- Herrero, L.; Quintanilla-Lopez, J.E.; Fernandez, M.A.; Gomara, B. Plasticisers and Preservatives in Commercial Milk Products: A Comprehensive Study on Packages used in the Spanish Market. Food Chem. 2021, 338, 128031. [Google Scholar] [CrossRef] [PubMed]
- Pajurek, M.; Pietron, W.; Maszewski, S.; Mikolajczyk, S.; Piskorska-Pliszczynska, J. Poultry eggs as a source of PCDD/Fs, PCBs, PBDEs and PBDD/Fs. Chemosphere 2019, 223, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Hagel, J.M.; Chen, X.; Facchini, P.J. Production of Methylparaben in Escherichia coli. J. Ind. Microbiol. Biotechnol. 2019, 46, 91–99. [Google Scholar] [CrossRef]
- Martínez, J.A. Natural Fungicides Obtained from Plants; IntechOpen Access Publisher: London, UK, 2012; pp. 3–28. [Google Scholar]
- Blažević, I.; Radonic, A.; Mastelie, J.; Zekic, M.; Skocibusic, M.; Maravic, A. Hedge Mustard (Sisymbrium Officinale): Chemical Diversity of Volatiles and their Antimicrobial Activity. Chem. Biodivers. 2010, 7, 2023–2034. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, C.; Wei, S.; Cui, H.; Huang, H. Compounds from the Subterranean Part of Johnsongrass and their Allelopathic Potential. Weed Biol. Manag. 2011, 11, 160–166. [Google Scholar] [CrossRef]
- Huang, H.; Liu, C.; Wei, S.; Wang, J.; Zhang, C. Dynamic Root Exudation of Phenolic Allelochemicals from Johnson Grass (Sorghum Halepense). Weed Biol. Manag. 2015, 15, 133–137. [Google Scholar]
- Hu, K.; Zhao, D.; Wu, G.; Ma, J. Aromatic poly(ether ester)s derived from a naturally occurring building block nipagin and linear aromatic α,ω-diols. RSC Adv. 2017, 7, 32989–33000. [Google Scholar]
- Martinez, M.A.; Rovira, J.; Prasad Sharma, R.; Nadal, M.; Schuhmacher, M.; Kumar, V. Comparing Dietary and Non-Dietary Source Contribution of BPA and DEHP to Prenatal Exposure: A Catalonia (Spain) Case Study. Environ. Res. 2018, 166, 25–34. [Google Scholar]
- Cunha, S.C.; Alves, R.N.; Fernandes, J.O.; Casal, S.; Marques, A. First Approach to Assess the Bioaccessibility of Bisphenol A in Canned Seafood. Food Chem. 2017, 232, 501–507. [Google Scholar] [PubMed]
- Cesen, M.; Lambropoulou, D.; Laimou-Geraniou, M.; Kosjek, T.; Blaznik, U.; Heath, D.; Heath, E. Determination of Bisphenols and Related Compounds in Honey and their Migration from Selected Food Contact Materials. J. Agric. Food Chem. 2016, 64, 8866–8875. [Google Scholar] [PubMed]
- EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids (CEF). Scientific opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs. EFSA J. 2013, 13, 3978. [Google Scholar]
- Mercogliano, R.; Santonicola, S. Investigation on Bisphenol A Levels in Human Milk and Dairy Supply Chain: A Review. Food Chem. Toxicol. 2018, 114, 98–107. [Google Scholar]
- Santonicola, S.; Ferrante, M.C.; di Leo, G.; Murru, N.; Anastasio, A.; Mercogliano, R. Study on Endocrine Disruptors Levels in Raw Milk from Cow’s Farms: Risk Assessment. Ital. J. Food Saf. 2018, 7, 158–161. [Google Scholar]
- González, N.; Cunha, S.C.; Ferreira, R.; Fernandes, J.O.; Marquès, M.; Nadal, M.; Domingo, J.L. Concentrations of Nine Bisphenol Analogues in Food Purchased from Catalonia (Spain): Comparison of Canned and Non-Canned Foodstuffs. Food Chem. Toxicol. 2020, 136, 110992. [Google Scholar]
- Darbre, P.D. Endocrine Disruptors and Obesity. Curr. Obes. Rep. 2017, 6, 18–27. [Google Scholar]
- Rivas, A.; Monteagudo, C.; Heras-Gonzalez, L.; Mariscal-Arcas, M.; Luisa Lorenzo-Tovar, M.; Olea-Serrano, F. Association of Bisphenol A Exposure with Dietary Quality Indices in Spanish Schoolchildren. Food Chem. Toxicol. 2016, 94, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Casajuana, N.; Lacorte, S. New Methodology for the Determination of Phthalate Esters, Bisphenol A, Bisphenol A Diglycidyl Ether, and Nonylphenol in Commercial Whole Milk Samples. J. Agric. Food Chem. 2004, 52, 3702–3707. [Google Scholar]
- Errico, S.; Nicolucci, C.; Migliaccio, M.; Micale, V.; Mita, D.G.; Diano, N. Analysis and Occurrence of some Phenol Endocrine Disruptors in Two Marine Sites of the Northern Coast of Sicily (Italy). Mar. Pollut. Bull. 2017, 120, 68–74. [Google Scholar] [PubMed]
- Ferrer, E.; Santoni, E.; Vittori, S.; Font, G.; Manes, J.; Sagratini, G. Simultaneous Determination of Bisphenol A, Octylphenol, and Nonylphenol by Pressurised Liquid Extraction and Liquid Chromatography-Tandem Mass Spectrometry in Powdered Milk and Infant Formulas. Food Chem. 2011, 126, 360–367. [Google Scholar]
- Gálvez-Ontiveros, Y.; Paez, S.; Monteagudo, C.; Rivas, A. Endocrine Disruptors in Food: Impact on Gut Microbiota and Metabolic Diseases. Nutrients 2020, 12, 1158. [Google Scholar]
- Siracusa, J.S.; Yin, L.; Measel, E.; Liang, S.; Yu, X. Effects of Bisphenol A and its Analogs on Reproductive Health: A Mini Review. Reprod. Toxicol. 2018, 79, 96–123. [Google Scholar]
- Alabi, A.; Caballero-Casero, N.; Rubio, S. Quick and Simple Sample Treatment for Multiresidue Analysis of Bisphenols, Bisphenol Diglycidyl Ethers and their Derivatives in Canned Food Prior to Liquid Chromatography and Fluorescence Detection. J. Chromatogr. A 2014, 1336, 23–33. [Google Scholar]
- Doumas, M.; Rouillon, S.; Venisse, N.; Nadeau, C.; Eugene, P.P.; Farce, A.; Chavatte, P.; Dupuis, A.; Migeot, V.; Carato, P. Chlorinated and Brominated Bisphenol A Derivatives: Synthesis, Characterization and Determination in Water Samples. Chemosphere 2018, 213, 434–442. [Google Scholar]
- Fasano, E.; Cirillo, T.; Esposito, F.; Lacorte, S. Migration of Monomers and Plasticizers from Packed Foods and Heated Microwave Foods using QuEChERS Sample Preparation and Gas Chromatography/Mass Spectrometry. LWT Food Sci. Technol. 2015, 64, 1015–1021. [Google Scholar]
- Cacho, J.I.; Campillo, N.; Vinas, P.; Hernandez-Cordoba, M. Stir Bar Sorptive Extraction Coupled to Gas Chromatography-Mass Spectrometry for the Determination of Bisphenols in Canned Beverages and Filling Liquids of Canned Vegetables. J. Chromatogr. A 2012, 1247, 146–153. [Google Scholar]
- Cirillo, T.; Esposito, F.; Fasano, E.; Scognamiglio, G.; Di Marco Pisciottano, I.; Mita, G.D.; Gallo, P. BPA, BPB, BPF, BADGE and BFDGE in Canned Beers from the Italian Market. Food Addit. Contam. Part B Surveill. 2019, 12, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Grumetto, L.; Gennari, O.; Montesano, D.; Ferracane, R.; Ritieni, A.; Albrizio, S.; Barbato, F. Determination of Five Bisphenols in Commercial Milk Samples by Liquid Chromatography Coupled to Fluorescence Detection. J. Food Prot. 2013, 76, 1590–1596. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, J.; Wenzl, T. Determination of Bisphenols in Beverages by Mixed-Mode Solid-Phase Extraction and Liquid Chromatography Coupled to Tandem Mass Spectrometry. J. Chromatogr. A 2015, 1422, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Barboza, L.G.A.; Cunha, S.C.; Monteiro, C.; Fernandes, J.O.; Guilhermino, L. Bisphenol A and its Analogs in Muscle and Liver of Fish from the North East Atlantic Ocean in Relation to Microplastic Contamination. Exposure and Risk to Human Consumers. J. Hazard. Mater. 2020, 393, 122419. [Google Scholar] [CrossRef]
- Cunha, S.C.; Cunha, C.; Ferreira, A.R.; Fernandes, J.O. Determination of Bisphenol A and Bisphenol B in Canned Seafood Combining QuEChERS Extraction with Dispersive Liquid-Liquid Microextraction Followed by Gas Chromatography-Mass Spectrometry. Anal. Bioanal. Chem. 2012, 404, 2453–2463. [Google Scholar] [CrossRef] [PubMed]
- Fattore, M.; Russo, G.; Barbato, F.; Grumetto, L.; Albrizio, S. Monitoring of Bisphenols in Canned Tuna from Italian Markets. Food Chem. Toxicol. 2015, 83, 68–75. [Google Scholar] [CrossRef]
- Chen, Y.; Shu, L.; Qiu, Z.; Lee, D.Y.; Settle, S.J.; Hee, S.Q.; Telesca, D.; Yang, X.; Allard, P. Exposure to the BPA-Substitute Bisphenol S Causes Unique Alterations of Germline Function. PLoS Genet. 2016, 12, e1006223. [Google Scholar] [CrossRef]
- Vela-Soria, F.; Ballesteros, O.; Zafra-Gómez, A.; Ballesteros, L.; Navalón, A. A Multiclass Method for the Analysis of Endocrine Disrupting Chemicals in Human Urine Samples. Sample Treatment by Dispersive Liquid–liquid Microextraction. Talanta 2014, 129, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Vela-Soria, F.; Rodriguez, I.; Ballesteros, O.; Zafra-Gomez, A.; Ballesteros, L.; Cela, R.; Navalon, A. Simplified Matrix Solid Phase Dispersion Procedure for the Determination of Parabens and Benzophenone-Ultraviolet Filters in Human Placental Tissue Samples. J. Chromatogr. A 2014, 1371, 39–47. [Google Scholar] [CrossRef]
- Xue, J.; Wu, Q.; Sakthivel, S.; Pavithran, P.V.; Vasukutty, J.R.; Kannan, K. Urinary Levels of Endocrine-Disrupting Chemicals, Including Bisphenols, Bisphenol A Diglycidyl Ethers, Benzophenones, Parabens, and Triclosan in Obese and Non-Obese Indian Children. Environ. Res. 2015, 137, 120–128. [Google Scholar] [CrossRef]
- Yang, Y.; Guan, J.; Yin, J.; Shao, B.; Li, H. Urinary Levels of Bisphenol Analogues in Residents Living Near a Manufacturing Plant in South China. Chemosphere 2014, 112, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Kramer, J.P.; Calafat, A.M.; Ye, X. Automated on-Line Column-Switching High Performance Liquid Chromatography Isotope Dilution Tandem Mass Spectrometry Method for the Quantification of Bisphenol A, Bisphenol F, Bisphenol S, and 11 Other Phenols in Urine. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 944, 152–156. [Google Scholar] [CrossRef]
- Deceuninck, Y.; Bichon, E.; Marchand, P.; Boquien, C.; Legrand, A.; Boscher, C.; Antignac, J.P.; Le Bizec, B. Determination of Bisphenol A and Related Substitutes/Analogues in Human Breast Milk using Gas Chromatography-Tandem Mass Spectrometry. Anal. Bioanal. Chem. 2015, 407, 2485–2497. [Google Scholar] [CrossRef] [PubMed]
- Rochester, J.R.; Bolden, A.L. Bisphenol S and F: A Systematic Review and Comparison of the Hormonal Activity of Bisphenol A Substitutes. Environ. Health Perspect. 2015, 123, 643–650. [Google Scholar] [CrossRef] [PubMed]
- AECOSAN. Encuesta Nacional de Alimentación En La Población Infantil Y Adolescente. Resultados Sobre Datos de Consumo. Agencia Española de Consumo, Seguridad Alimentaria Y Nutrición. Ministry of Health, Madrid, Spain. Available online: Http://Www.Aecosan.Msssi.Gob.Es/AECOSAN/Web/Seguridad_alimentaria/Subdetalle/Enalia.Htm (accessed on 10 July 2010).
| tR (min) | Transitions | CV | CE | tR (min) | Transitions | CV | CE | ||
|---|---|---|---|---|---|---|---|---|---|
| BPS | 0.5 | 249.1 → 107.5 b | −4 | −26 | MetPB | 0.9 | 151.0 → 91.4 a | −12 | −18 |
| 249.1 → 155.5 a | −4 | −20 | 151.0 → 135.5 b | −12 | −14 | ||||
| BPF | 4.2 | 199.1 → 76.4 b | −14 | −24 | EthPB | 2 | 165.1→ 91.6 a | −14 | −22 |
| 199.1 → 92.4 a | −14 | −20 | 165.1 → 136.3 b | −14 | −14 | ||||
| BPE | 4.6 | 213.1 → 197.7 | −46 | −18 | PropPB | 3 | 179.1 → 91.5 a | −26 | −22 |
| 179.1 → 114.5 b | −26 | −16 | |||||||
| BPA | 4.9 | 227.2 → 132.9 | −50 | −26 | ButPB | 3.9 | 193.1 → 91.5 a | −18 | −26 |
| 193.1 → 135.9 b | −18 | −16 | |||||||
| BPAF | 5 | 335.2 → 196.7 b | −4 | −36 | BPA-d16 | 4.8 | 241.3 → 141.7 | −24 | −26 |
| 335.2 → 264.9 a | −4 | −22 | |||||||
| BPB | 5.2 | 241.2 → 211.8 b | −10 | −16 | EthPB-d5 | 2 | 170.2 → 91.3 a | −24 | −16 |
| 241.2 → 225.9 a | −10 | −20 | 170.2 → 137.6 b | −24 | −14 | ||||
| BPP | 6.1 | 345.2 → 315.1 b | −18 | −40 | |||||
| 345.2 → 330.1 a | −18 | −24 | |||||||
| Voltage of capilar | 3 kV | Nebulizer gas pressure | 7.0 bar | ||||||
| Source temperature | 150 °C | Cone/desolvation gas | N2 (≥99.995%) | ||||||
| Desolvation temperature | 600 °C | Collision gas | Ar (99.999%) | ||||||
| Cone gas flow | 150 L h−1 | Dwell time | 25 ms | ||||||
| Desolvation gas flow | 500 L h−1 | Inter-scan delay | 3 ms | ||||||
| Collision gas flow | 0.15 mL min−1 | ||||||||
| b | Linearity | LOD | LOQ | Recovery Assay | ||||
|---|---|---|---|---|---|---|---|---|
| g ng−1 | R2 | %Plof | ng g−1 | ng g−1 | Added (ng g−1) | %Rec | %RSD | |
| MetPB | 0.8556 | 0.9535 | 15.2 | 0.1 | 0.4 | 1 | 95 | 10 |
| 100 | 102 | 5.1 | ||||||
| 250 | 96 | 6.3 | ||||||
| EthPB | 0.9615 | 0.9273 | 22.1 | 0.1 | 0.4 | 1 | 106 | 8.2 |
| 100 | 98 | 7.1 | ||||||
| 250 | 103 | 4.8 | ||||||
| PropPB | 0.1064 | 0.9742 | 15.3 | 0.3 | 0.9 | 1 | 94 | 12 |
| 100 | 98 | 6.2 | ||||||
| 250 | 96 | 4.1 | ||||||
| ButPB | 0.2612 | 0.982 | 35.4 | 0.2 | 0.7 | 1 | 96 | 7.2 |
| 100 | 103 | 3.2 | ||||||
| 250 | 98 | 4.6 | ||||||
| BPS | 0.0626 | 0.9665 | 22.1 | 0.3 | 1 | 1 | 91 | 11.1 |
| 100 | 92 | 7.4 | ||||||
| 250 | 103 | 9.4 | ||||||
| BPE | 0.1108 | 0.9945 | 60.2 | 0.3 | 1 | 1 | 93 | 8.2 |
| 100 | 105 | 2.3 | ||||||
| 250 | 97 | 6 | ||||||
| BPF | 0.1996 | 0.9915 | 48.1 | 0.1 | 0.5 | 1 | 103 | 8.6 |
| 100 | 104 | 7.2 | ||||||
| 250 | 94 | 5.6 | ||||||
| BPAF | 0.314 | 0.9851 | 20.3 | 0.1 | 0.4 | 1 | 95 | 8.5 |
| 100 | 103 | 4.1 | ||||||
| 250 | 96 | 10.3 | ||||||
| BPA | 0.0446 | 0.9755 | 12.1 | 0.3 | 0.9 | 1 | 104 | 7.2 |
| 100 | 104 | 4.5 | ||||||
| 250 | 94 | 10.7 | ||||||
| BPB | 0.0989 | 0.9848 | 20.3 | 0.3 | 0.9 | 1 | 104 | 6.2 |
| 100 | 97 | 7 | ||||||
| 250 | 93 | 5.5 | ||||||
| BPP | 0.0019 | 0.9723 | 16.2 | 1 | 4 | 5 | 104 | 8.6 |
| 100 | 92 | 5.2 | ||||||
| 250 | 94 | 4.2 | ||||||
| Parabens | Bisphenols | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MetPB | EthPB | ButPB | PropPB | ∑PBs | BPS | BPE | BPF | BPAF | BPA | BPB | BPP | ∑BPs | |
| Unprocessed or Minimally Processed Foods (n = 32) | |||||||||||||
| Frecuency (%) | 70 | 23.3 | 3.3 | 10 | 73.3 | 46.88 | 6.3 | 0 | 0 | 21.88 | 0 | 0 | 63 |
| Mean (ng g−1) | 106.90 (95.37) | 29.54 (40.06) | 2.5 (1.0) | 34.95 (48.0) | 80.7 (81.56) | 17.27 (13.7) | <LOQ | 0 | 0 | 6 (3.8) | 0 | 0 | 18.35 (15.47) |
| Processed Foods (n = 21) | |||||||||||||
| Frecuency (%) | 42.86 | 23.81 | 9.52 | 4.76 | 52 | 38.1 | 0 | 0 | 0 | 38.1 | 0 | 0 | 67 |
| Mean (ng g−1) | 142.05 (120.2) | 60.28 (56.2) | 55.35 (42.2) | 65.5 (26.2) | 130.7 (87.3) | 39.49 (67.0) | 0 | 0 | 0 | 86.3 (158.6) | 0 | 0 | 35 (119.5) |
| Ultra-Processed Foods (n = 47) | |||||||||||||
| Frecuency (%) | 38.3 | 6.38 | 10.64 | 12.77 | 49 | 6.38 | 4.26 | 0 | 0 | 27.66 | 0 | 0 | 36 |
| Mean (ng g−1) | 41.75 (47.18) | 28.6 (12.0) | 39.87 (60.07) | 3.15 (2.0) | 40.31 (48.46) | 47.48 (73.29) | <LOQ | 0 | 0 | 35.3 (42.4) | 0 | 0 | 38.34 (48.14) |
| All (n = 100) | |||||||||||||
| Frecuency (%) | 49 | 15 | 8.1 | 10.2 | 57.14 | 26.5 | 4.1 | 0 | 0 | 28.6 | 0 | 0 | 52 |
| Mean (ng g−1) | 84.6 (88.5) | 42.19 (44.71) | 39.07 (50.83) | 21.1 (31.5) | 73.86 (76.76) | 28.99 (46.29) | <LOQ | 0 | 0 | 43.28 (91.45) | 0 | 0 | 30.4 (41.9) |
| Sample | Packaging | Parabens | Bisphenols | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MetPB | EthPB | ButPB | PropPB | ∑PBs | BPS | BPE | BPF | BPAF | BPA | BPB | BPP | ∑BPs | ||
| Chicken | Plastic and porex tray | ND | ND | ND | D | ND | ND | ND | ND | 2.1 (1.0) | ND | ND | 2.1 (1.0) | |
| Eggs | Plastic and paperboard | 229.9 (29.2) | ND | ND | ND | 229.9 (29.2) | ND | ND | ND | ND | ND | ND | ND | |
| Whole milk | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Whole milk | Carton | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Whole milk | Carton | ND | ND | ND | ND | ND | D | ND | ND | ND | ND | ND | ||
| Whole milk | Carton | ND | ND | ND | ND | ND | D | ND | ND | ND | ND | ND | ||
| Frozen hake | Plastic and paperboard | ND | ND | ND | ND | ND | ND | ND | ND | D | ND | ND | ||
| Lentils | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | D | ND | ND | ||
| Grape | Plastic | D | ND | ND | ND | D | ND | ND | ND | ND | ND | ND | ||
| Blueberries | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Pineapple | Plastic | D | 5.8 (4.2) | 2.5 (1.0) | 68.9 (4.0) | 77.2 (37.4) | 44.3 (2.8) | ND | ND | ND | 11.3 (4.6) | ND | ND | 55.6 (23.3) |
| Raspberry | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Melon | Plastic | 125.9 | ND | ND | ND | 125.9 | 4.22 (2.13) | ND | ND | ND | 7.86 (4.21) | ND | ND | 12.08 (2.57) |
| Apple | Not packed | 7.4 (3.2) | ND | ND | ND | 7.4 (3.2) | 12.7 (1.6) | ND | ND | ND | 6.0 (9.1) | ND | ND | 18.7 (4.7) |
| Apple | Plastic | 12.1 (9.2) | ND | ND | ND | 12.1 (9.2) | 8.5 (3.3) | ND | ND | ND | 2.9 (2.4) | ND | ND | 11.4 (3.96) |
| Pear | Not packed | 101.6 (84.7) | ND | ND | ND | 101.6 (84.7) | ND | ND | ND | ND | ND | ND | ND | |
| Frozen red fruit mix | Plastic | D | 100.2 (8.2) | ND | ND | 100.2 (8.2) | ND | ND | ND | ND | ND | ND | ND | |
| Frozen mango | Plastic | 39.5 | ND | ND | ND | 39.5 | D | ND | ND | ND | ND | ND | ND | |
| Frozen chopped garlic | Plastic | D | 13.6 (1.3) | ND | ND | 13.6 (1.3) | 1.36 (0.77) | ND | ND | ND | ND | ND | ND | 1.36 (0.77) |
| Frozen chopped onion | Plastic | 231.9 (18.9) | ND | ND | ND | 231.9 (18.9) | ND | ND | ND | ND | ND | ND | ND | |
| Frozen chopped parsley | Plastic | D | ND | ND | ND | 33.3 (13.9) | ND | ND | ND | ND | ND | ND | 33.3 (13.9) | |
| Frozen spinach | Plastic | D | ND | ND | ND | D | ND | ND | ND | ND | ND | ND | ||
| Tomato | Not packed | D | ND | ND | ND | 4.7 (2.2) | ND | ND | ND | ND | ND | ND | 4.7 (2.2) | |
| Tomato | Plastic | D | ND | ND | ND | 25.9 (10.7) | ND | ND | ND | ND | ND | ND | 25.9 (10.7) | |
| Striped carrot | Plastic | D | D | ND | ND | 11.5 (5.3) | ND | ND | ND | ND | ND | ND | 11.5 (5.3) | |
| Carrod | Plastic | D | 6 (3.6) | ND | ND | 6 (3.6) | D | ND | ND | ND | ND | ND | ND | |
| Lettuce | Plastic | D | D | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Pumpkin | Plastic | D | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Mushrooms | Plastic | D | 22.1 (10.4) | ND | 1.0 (0.1) | 23.1 (14.9) | 16.0 (6.9) | ND | ND | ND | ND | ND | ND | 16.0 (6.9) |
| Green pepper | Not packed | D | ND | ND | ND | 27.5 (6.3) | ND | ND | ND | ND | ND | ND | 27.5 (6.3) | |
| Mean | 106.90 (95.37) | 29.5 (40.6) | 2.5 (1.0) | 34.95 (48.0) | 73.17 (79.9) | 17.27 (13.7) | 6 (3.8) | 18.35 (15.47) | ||||||
| Sample | Packaging | Parabens | Bisphenols | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MetPB | EthPB | ButPB | PropPB | ∑PBs | BPS | BPE | BPF | BPAF | BPA | BPB | BPP | ∑BPs | ||
| Cooked ham | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | 6.6 (3.4) | ND | ND | 6.6 (3.4) | |
| Spicy Sausage | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Spicy Sausage | Plastic | D | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Chicken burguer | Plastic | 191.1(118.3) | ND | ND | ND | 191.1 (118.3) | ND | ND | ND | ND | ND | ND | ND | |
| Chicken burguer | Plastic | 281.7 (88.9) | ND | ND | ND | 281.7 (88.9) | ND | ND | ND | ND | ND | ND | ND | |
| Sausage (Chorizo) | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Serrano ham | Plastic | ND | ND | ND | ND | 39.3 (21.3) | ND | ND | ND | 17.3 (14.9) | ND | ND | 56.6 (15.56) | |
| Plain yogurt (sweetened) | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | 29.88 (18.6) | ND | ND | 29.88 (18.6) | |
| Plain yogurt (sweetened) | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | 12.3 (6.0) | ND | ND | 12.3 (6.0) | |
| Guacamole | Plastic | D | ND | ND | ND | D | ND | ND | ND | ND | ND | ND | ||
| Olives | Plastic | 5.2 (1.7) | 29.2 (14.5) | ND | ND | 34.4 (16.97) | 8.5 (5.4) | ND | ND | ND | ND | ND | ND | 8.5 (5.4) |
| Olives | Plastic | D | 13.6 (5.5) | 85.2 (39.5) | 65.5 (26.2) | 164.3 (36.99) | 30.2 (7.7) | ND | ND | ND | ND | ND | ND | 30.2 (7.7) |
| Anchovy stuffed olives | Can | ND | 86.9 (17.5) | ND | ND | 86.9 (17.5) | ND | ND | ND | ND | ND | ND | ND | |
| Semi-cured cheese | Plastic | ND | 24.8 (7.2) | ND | ND | 24.8 (7.2) | ND | ND | ND | ND | D | ND | ND | |
| Semi-cured cheese (slice) | Plastic | D | ND | ND | ND | 5.6 (1.9) | ND | ND | ND | ND | ND | ND | 5.6 (1.9) | |
| Pasta | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Rice (for microwave) | Plastic and paperboard | 90.2 (28.6) | ND | ND | ND | 90.2 (28.6) | 3.3 (1.4) | ND | ND | ND | ND | ND | ND | 3.3 (1.4) |
| Canned tuna in oil | Can | ND | ND | ND | ND | ND | ND | ND | ND | 409.0 (31.0) | ND | ND | 409.0 (31.0) | |
| Canned tuna in oil | Can | D | 146.9 (8.5) | 25.5 (14.9) | ND | 172.4 (85.8) | 187.8 (14.2) | ND | ND | ND | D | ND | ND | 187.8 (14.2) |
| Canned sweet corn | Can | ND | ND | ND | ND | ND | ND | ND | ND | 42.7 (6.2) | ND | ND | 42.7 (6.2) | |
| Cake | Not packed | ND | ND | ND | ND | 1.7 (0.7) | ND | ND | ND | ND | ND | ND | 1.7 (0.7) | |
| Mean | 142.05 (120.2) | 60.28 (56.2) | 55.35 (42.2) | 65.5 (26.2) | 130.7 (87.3) | 39.49 (67.0) | 86.3 (158.6) | 35 (119.5) | ||||||
| Sample | Package Type | Parabens | Bisphenols | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MetPB | EthPB | ButPB | PropPB | ∑PBs | BPS | BPE | BPF | BPAF | BPA | BPB | BPP | ∑BPs | ||
| Sausage (Hot dogs) | Plastic | 6.8 (7.2) | ND | ND | ND | 6.8 (7.2) | ND | ND | ND | ND | ND | ND | ND | |
| Turkey cold cut | Plastic | D | ND | 2.05 (0.06) | ND | 2.05 (0.06) | ND | ND | ND | ND | ND | ND | ND | |
| Sausage (Turkey cold) | Plastic | D | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Mortadella (Bologna) | Plastic | D | ND | 16.5 (11.2) | ND | 16.5 (11.2) | 5.43 (3.34) | ND | ND | ND | ND | ND | ND | 5.43 (3.34) |
| Chocolate milkshake | Carton | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Chocolate milkshake | Carton | D | ND | ND | ND | ND | D | ND | ND | ND | ND | ND | ||
| Chocolate milkshake | Carton | D | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Chocolate milkshake | Carton | ND | ND | ND | ND | ND | D | ND | ND | ND | ND | ND | ||
| Semi-fermented milk | Plastic and foil | 6.1 (3.67) | ND | ND | ND | 6.1 (3.67) | ND | ND | ND | ND | ND | ND | ND | |
| Semi-fermented milk | Plastic and foil | 41.08 (12.55) | ND | ND | ND | 41.08 (12.55) | ND | ND | ND | ND | ND | ND | ND | |
| Semi-fermented milk | Plastic and foil | 88.38 (34.9) | ND | ND | ND | 88.38 (34.9) | ND | ND | ND | ND | ND | ND | ND | |
| Flavoured Yogurt | Plastic | 26.6 (6.1) | ND | ND | ND | 26.6 (6.1) | ND | ND | ND | ND | 60.85 (17.2) | ND | ND | 60.85 (17.2) |
| Flavoured Liquid Yogurt | Plastic | 145.66 (8.86) | ND | ND | ND | 145.66 (8.86) | ND | ND | ND | ND | 115.4 (65.96) | ND | ND | 115.4 (65.96) |
| Flavoured Liquid Yogurt | Plastic | 42.49 (20.46) | ND | ND | ND | 42.49 (20.46) | ND | ND | ND | ND | ND | ND | ND | |
| Spread cheese | Foil and paperboard | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Melted cheese | Plastic | ND | ND | ND | ND | 4.9 (0.9) | ND | ND | ND | D | ND | ND | 4.9 (0.9) | |
| Melted cheese | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | 2 (0.3) | ND | ND | 2 (0.3) | |
| Breadsticks for cheese | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Tuna dumplings | Can | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Battered hake sticks | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Pizza (cooked ham and cheese) | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | 4.3 (1.8) | ND | ND | 4.3 (1.8) | |
| Pizza (4 cheese) | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Pizza (bolognese) | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Ketchup | Plastic | ND | ND | ND | D | ND | ND | ND | ND | ND | ND | ND | ||
| Ketchup | Plastic | ND | ND | ND | D | ND | ND | ND | ND | D | ND | ND | ||
| Tomato sauce | Carton | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Corn snacks | Plastic | ND | ND | ND | 1.4 (0.3) | 1.4 (0.3) | ND | ND | ND | ND | ND | ND | ND | |
| Corn snacks | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Nachos | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | 42.1 (4.2) | ND | ND | 42.1 (4.2) | |
| Chips | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Chips | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Chips | Plastic | D | ND | ND | ND | 132.1 (21.2) | ND | ND | ND | ND | ND | ND | 132.1 (21.2) | |
| Chips (Sour Cream & Onion) | Plastic, foil and paperboard | 7.7 (0.7) | ND | ND | ND | 7.7 (0.7) | ND | ND | ND | ND | 8.8 (10.7) | ND | ND | 8.8 (10.7) |
| Gummy candy | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Gummy candy | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Chocolate doughnuts | Plastic | ND | D | ND | ND | ND | ND | ND | ND | 82 (10.2) | ND | ND | 82 (10.2) | |
| Milk bread | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Croissants | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Croissants | Plastic | D | ND | 4.3 (2.6) | 1.5 (0.8) | 5.8 (1.98) | ND | ND | ND | ND | ND | ND | ND | |
| Chocolate puff pastry | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | 1 | ND | ND | 1 | |
| Cacao-filled roll | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | D | ND | ND | ||
| Muffins | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Muffins | Plastic | D | ND | ND | 4.2 (0.8) | 4.2 (0.8) | ND | ND | ND | ND | ND | ND | ND | |
| Burger bun | Plastic | 10.9 (9.6) | 20.1 (7.7) | ND | ND | 31.0 (6.5) | ND | ND | ND | ND | 1.2 (1.6) | ND | ND | 1.2 (1.6) |
| Sandwich bread | Plastic | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ||
| Milk bread with chocolate chips | Plastic | ND | ND | 145.3 (10.2) | ND | 145.3 (10.2) | ND | ND | ND | ND | D | ND | ND | |
| Puffed rice cake with chocolate | Plastic | D | 37.1 (5.5) | 31.2 (8.3) | 5.5 (3.0) | 73.8 (16.8) | ND | ND | ND | ND | ND | ND | ND | |
| Mean | 41.75 (47.18) | 28.6 (12.0) | 39.87 (60.07) | 3.15 (2.0) | 40.31 (48.46) | 47.48 (73.29) | 35.3 (42.4) | 38.34 (48.14) | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gálvez-Ontiveros, Y.; Moscoso-Ruiz, I.; Rodrigo, L.; Aguilera, M.; Rivas, A.; Zafra-Gómez, A. Presence of Parabens and Bisphenols in Food Commonly Consumed in Spain. Foods 2021, 10, 92. https://doi.org/10.3390/foods10010092
Gálvez-Ontiveros Y, Moscoso-Ruiz I, Rodrigo L, Aguilera M, Rivas A, Zafra-Gómez A. Presence of Parabens and Bisphenols in Food Commonly Consumed in Spain. Foods. 2021; 10(1):92. https://doi.org/10.3390/foods10010092
Chicago/Turabian StyleGálvez-Ontiveros, Yolanda, Inmaculada Moscoso-Ruiz, Lourdes Rodrigo, Margarita Aguilera, Ana Rivas, and Alberto Zafra-Gómez. 2021. "Presence of Parabens and Bisphenols in Food Commonly Consumed in Spain" Foods 10, no. 1: 92. https://doi.org/10.3390/foods10010092
APA StyleGálvez-Ontiveros, Y., Moscoso-Ruiz, I., Rodrigo, L., Aguilera, M., Rivas, A., & Zafra-Gómez, A. (2021). Presence of Parabens and Bisphenols in Food Commonly Consumed in Spain. Foods, 10(1), 92. https://doi.org/10.3390/foods10010092







