Dental Wear: Attrition, Erosion, and Abrasion—A Palaeo-Odontological Approach
Abstract
:1. Introduction
2. Etiology of Dental Wear
3. Enamel Thickness
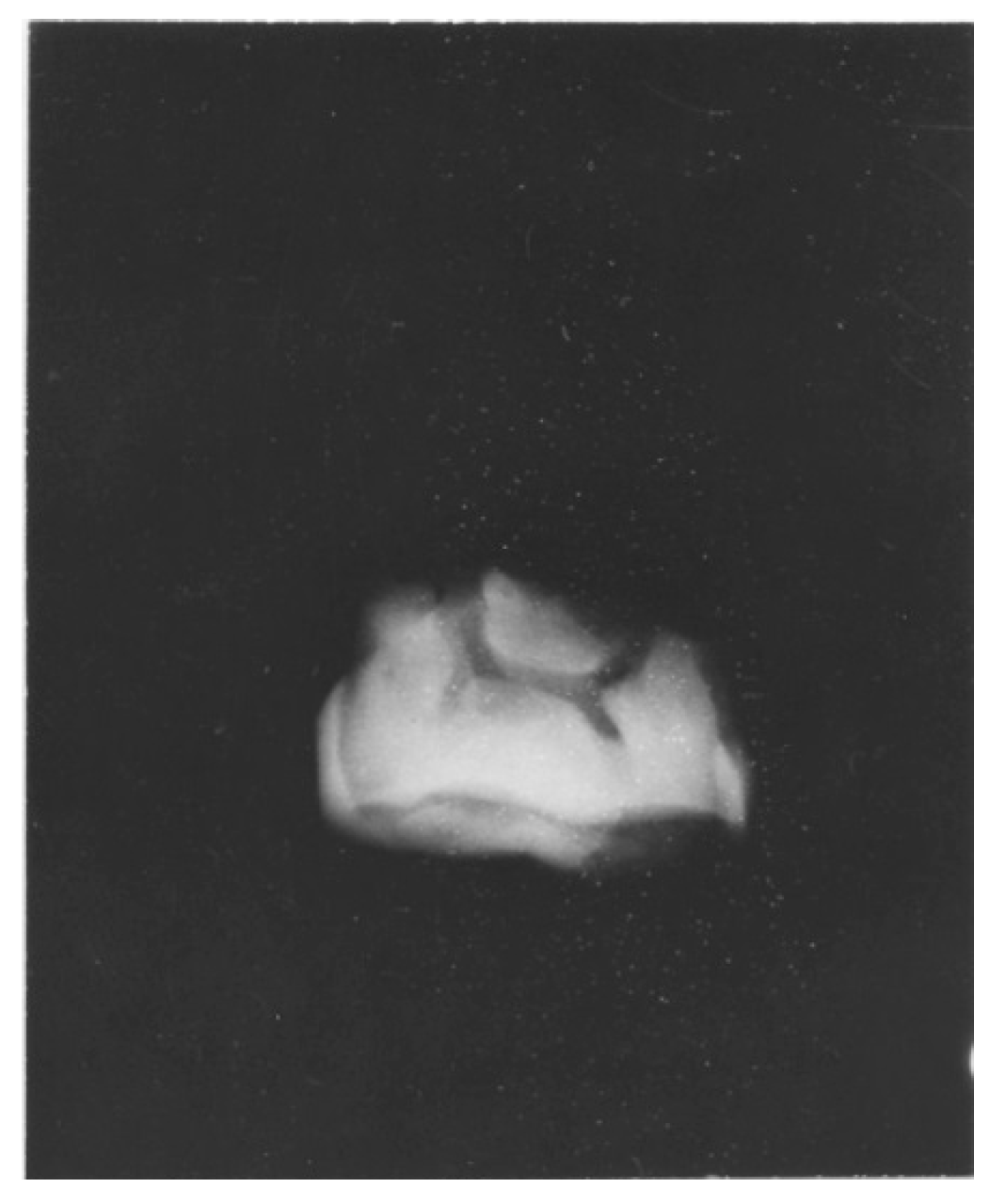
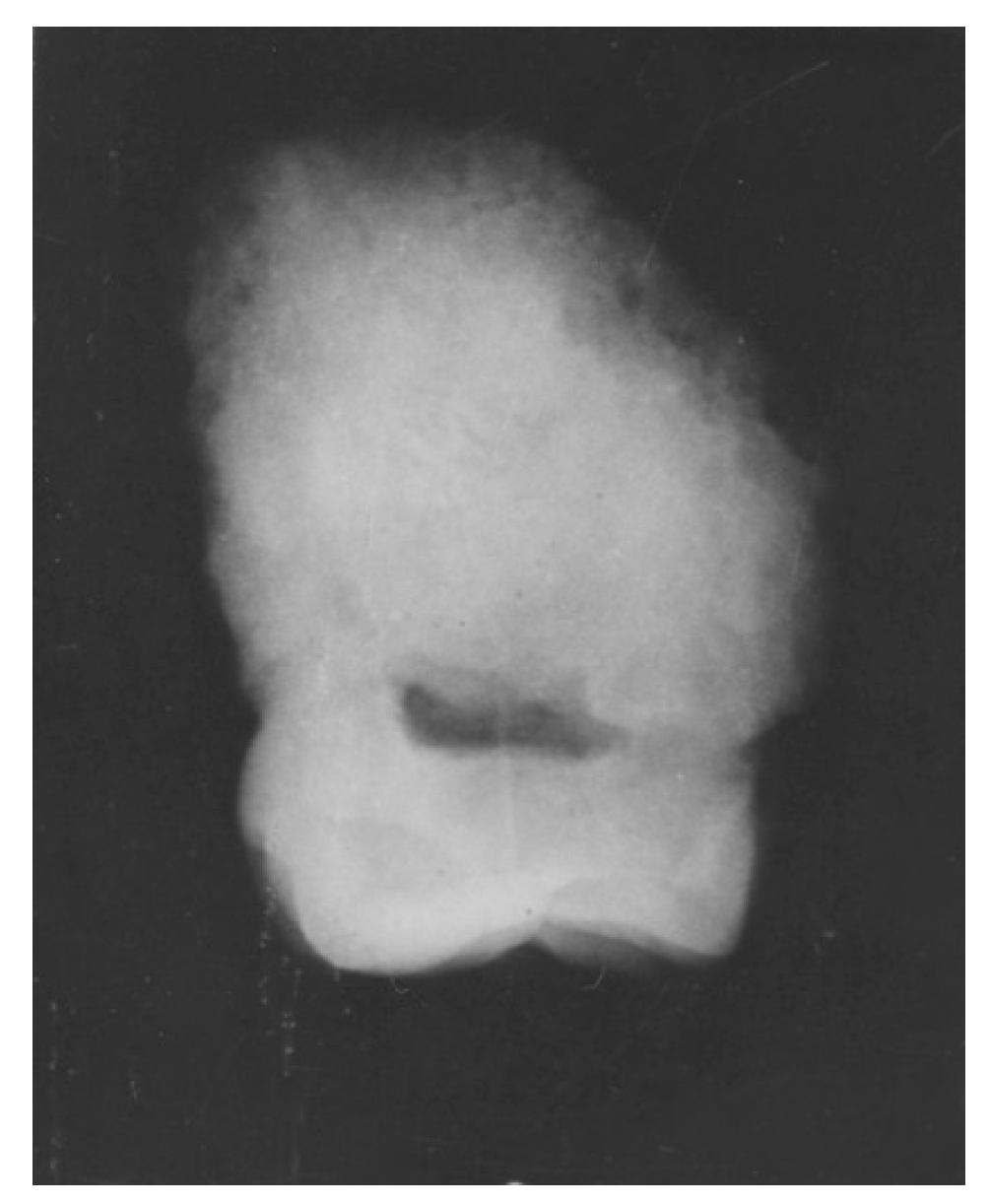
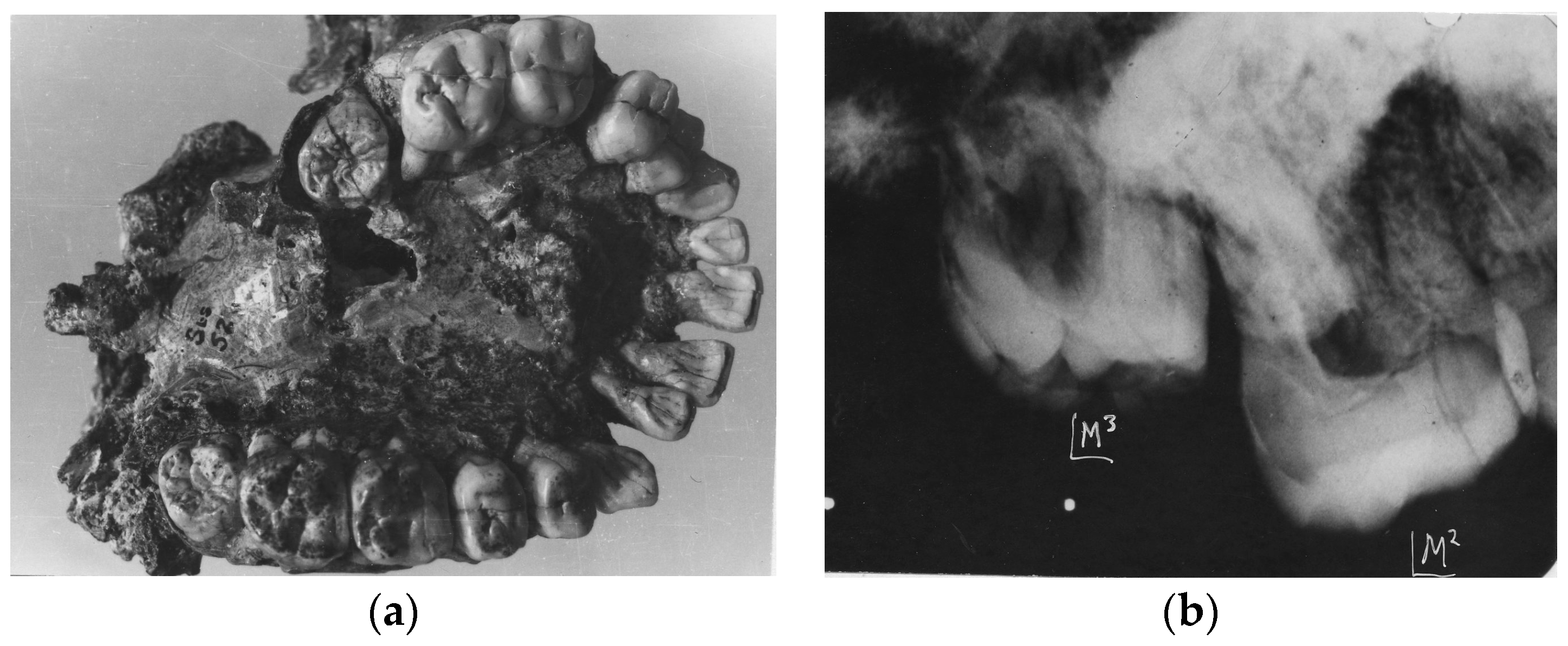
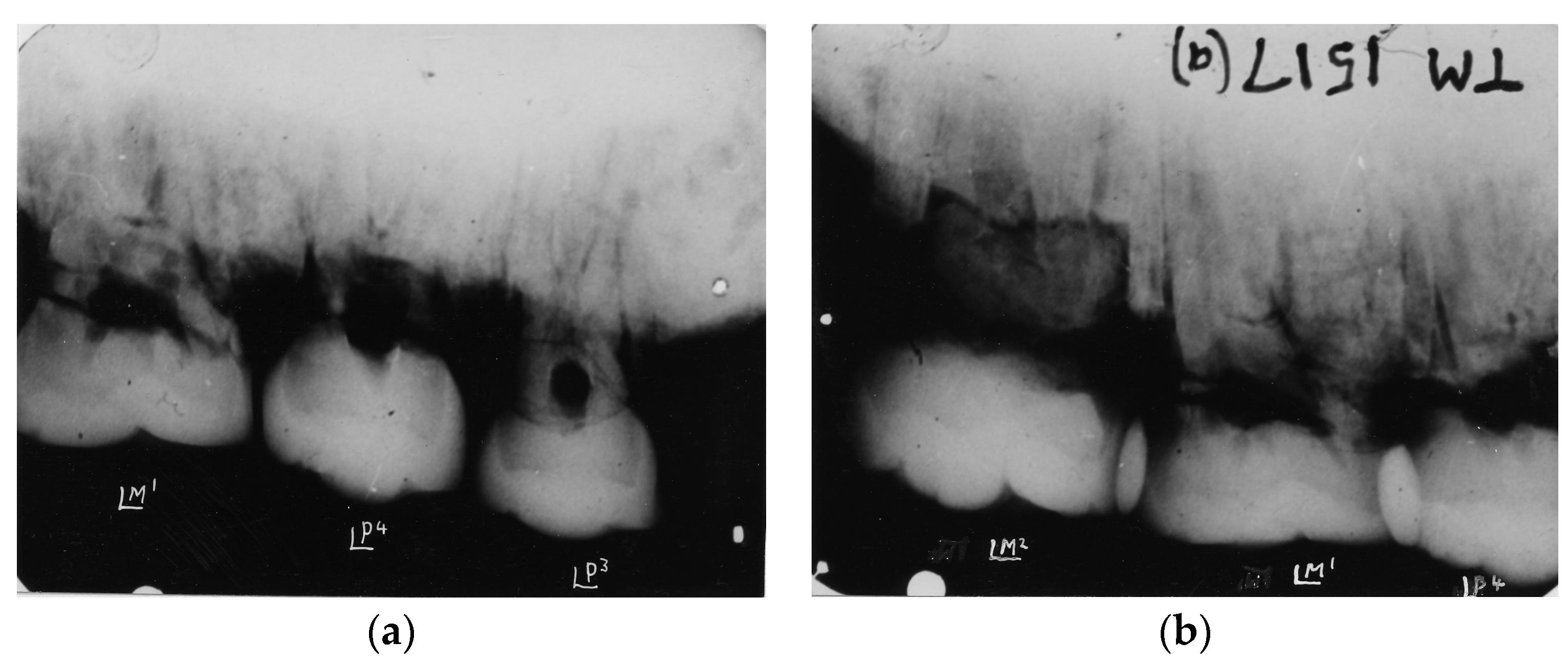
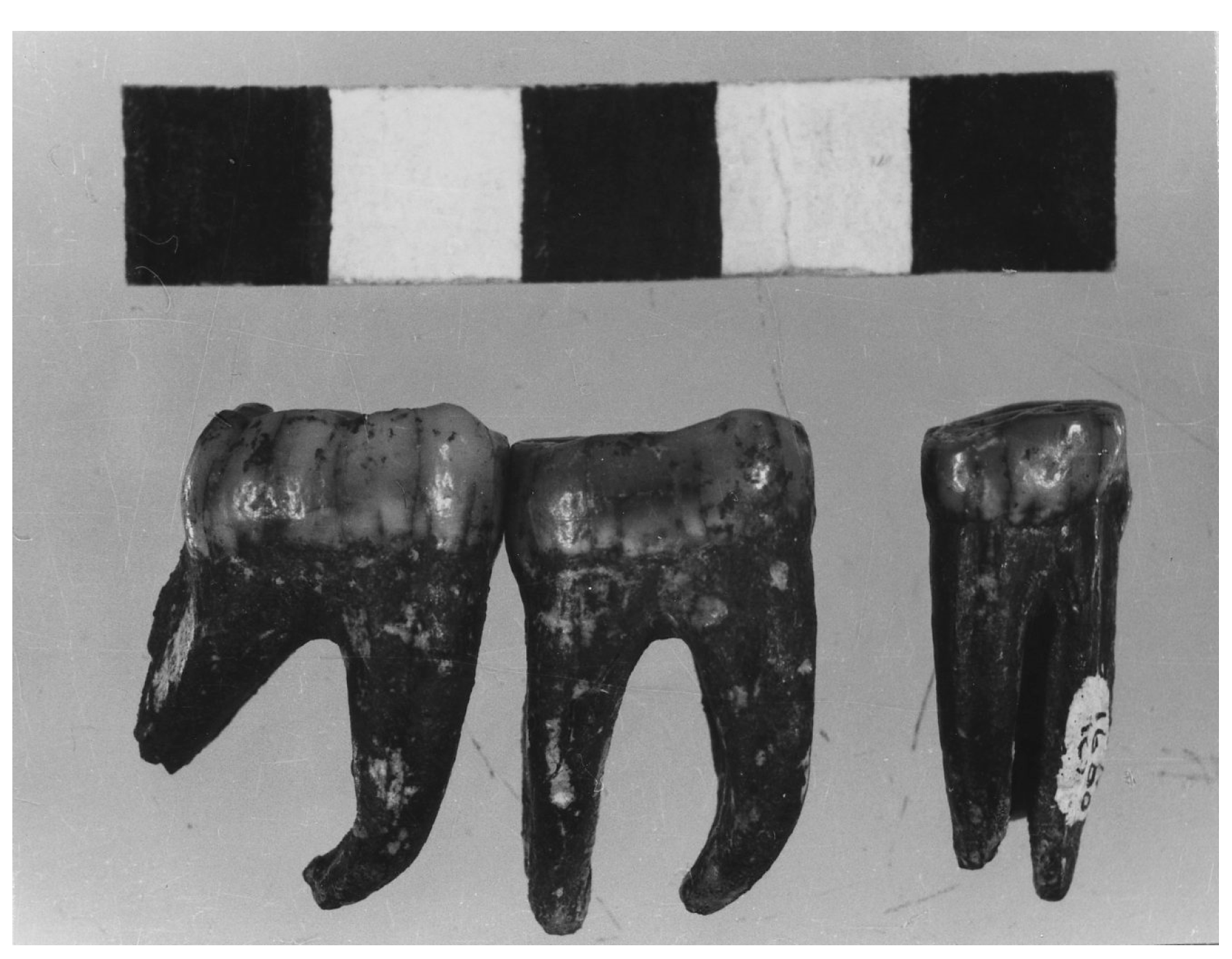
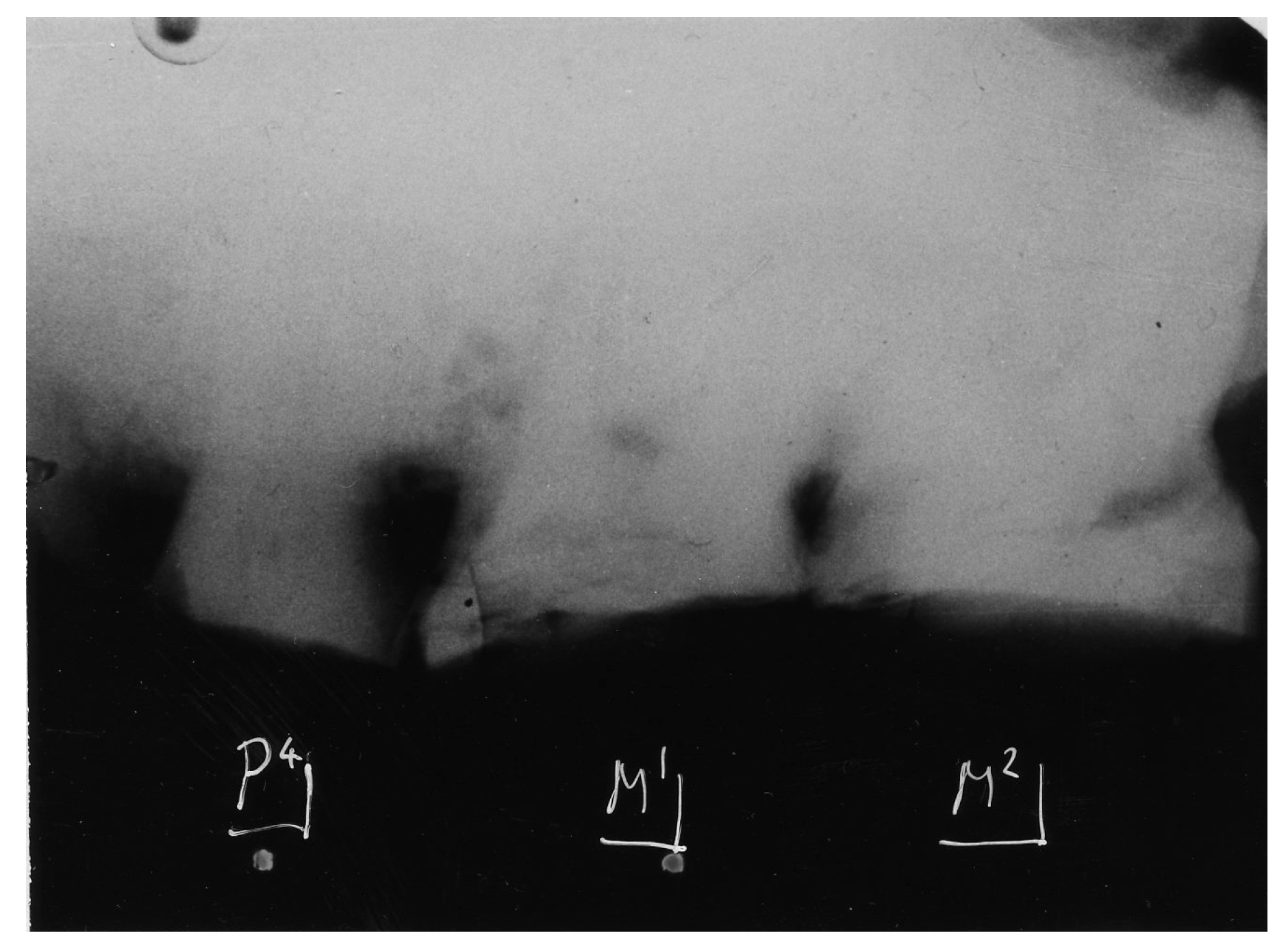
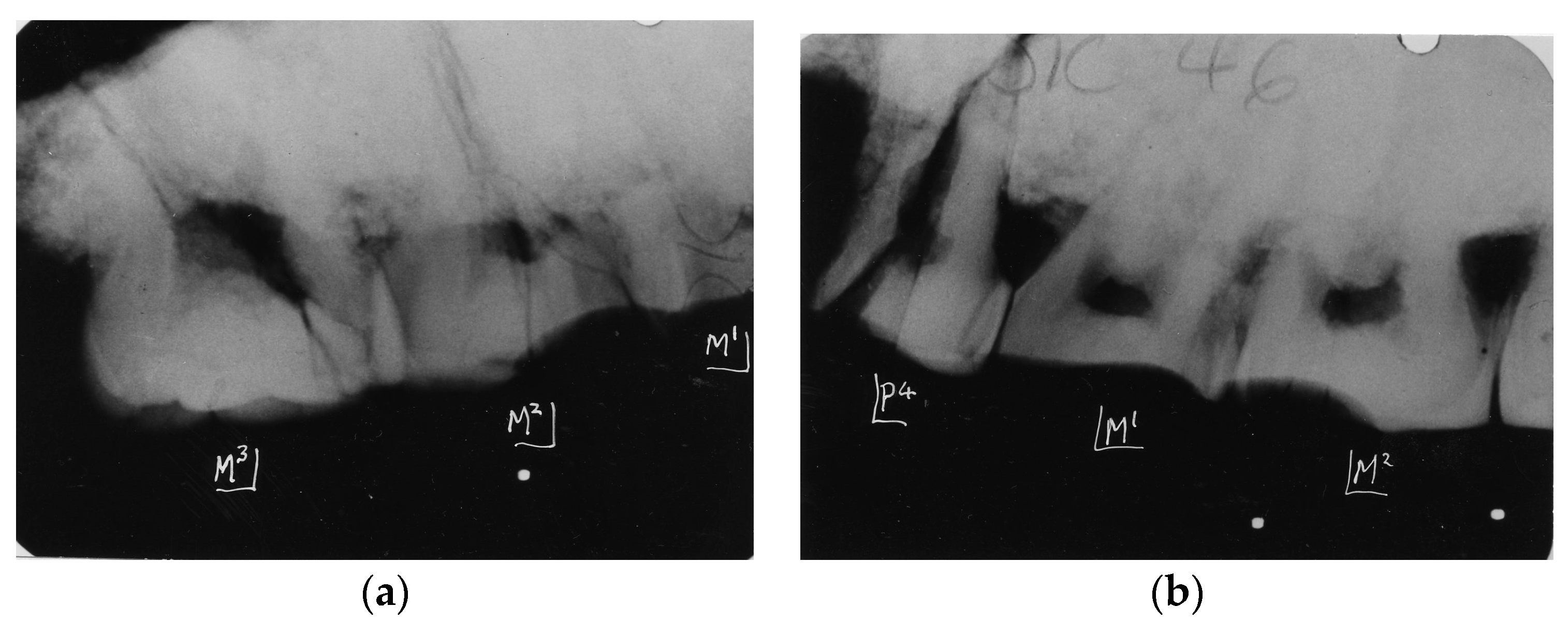
4. Australopithecine and Paranthropus Dentitions
5. Conclusions
Conflicts of Interest
References
- Carvalho, T.S.; Colon, P.; Ganss, C.; Huysmans, M.C.; Lussi, A.; Schlueter, N.; Schmalz, G.; Shellis, P.R.; Tveit, A.B.; Wiegand, A. Consensus report of the European Federation of Conservative Dentistry: Erosive tooth wear-diagnosis and management. Clin. Oral Investig. 2015, 19, 1557–1561. [Google Scholar] [CrossRef] [PubMed]
- Ranjitkar, S.; Turan, A.; Mann, C.; Gully, G.A.; Marsman, M.; Edwards, S.; Kaidonis, J.A.; Hall, C.; Lekkas, D.; Wetselaar, P.; et al. Surface sensitive microwear texture analysis of attrition and erosion. J. Dent. Res. 2017, 96, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Spee, F.G. Die verschiebungsbahn des unterkiefers am schadel. Arch. Anat. Physiol. 1890, 16, 285–294. [Google Scholar]
- Marshall, S.D.; Casperson, M.; Hardinger, R.R.; Franciscus, R.G.; Aquilino, S.A.; Southard, T.E. Development of the curve of Spee. Am. J. Orthod. Dentofac. Orthoped. 2008, 134, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Wang, W.; Wang, X.; Shi, X.; Si, Y.; Zheng, S. Tooth wear: A cross-sectional investigation of the prevalence and risk factors in Beijing, China. BDJ Open 2017, 3, 16012. [Google Scholar] [CrossRef]
- Molnar, S.; Gantt, D.G. Functional implications of primate enamel thickness. Am. J. Phys. Anthrop. 1977, 46, 447. [Google Scholar] [CrossRef] [PubMed]
- Sperber, G.H. Comparative Primate Dental Enamel Thickness: A Radiodontological Study. In Hominid Evolution: Past, Present and Future; Tobias, P.V., Ed.; Alan R. Liss: New York, NY, USA, 1985; pp. 443–454. [Google Scholar]
- Grine, F.E.; Sponheimer, M.; Ungar, P.; Lee-Thorp, J.; Teaford, M.F. Dental microwear and stable isotopes inform the palecology of extinct hominins. Am. J. Phys. Anthrop. 2012, 148, 285–317. [Google Scholar] [CrossRef] [PubMed]
- Lucas, P.W.; Omar, R.; Al-Fadhalah, K.; Almusallam, A.S.; Henry, A.G.; Michael, S.; Thai, L.A.; Watzke, J.; Strait, D.S.; Atkins, A.G. Mechanisms and causes of wear in tooth enamel: implications for hominin diets. J. R. Soc. Interface 2013, 10. [Google Scholar] [CrossRef] [PubMed]
- Grine, F.E. Trophic differences between “gracile” and “robust” australopithecines: A scanning electron microscopic analysis of occlusal events. S. Afr. J. Sci. 1981, 77, 203. [Google Scholar]
- Grine, F.E. Dental evidence for dietary differences in Australopithecus and Paranthropus: a quantitative analysis of permanent molar microwear. J. Hum. Evol. 1986, 15, 783–822. [Google Scholar] [CrossRef]
- Scott, R.S.; Ungar, P.S.; Bergstrom, T.S.; Brown, C.A.; Grine, F.E.; Teaford, M.F.; Walker, A. Dental microwear texture analysis shows within species dietary variability in fossil hominins. Nature 2005, 436, 693–695. [Google Scholar] [CrossRef] [PubMed]
- Sponheimer, M.; Passey, B.H.; de Ruiter, D.J.; Guatelli-Steinberg, D.; Cerling, T.E.; Lee-Thorp, J.A. Isotopic evidence for dietary variability in the early hominin Paranthropus robustus. Science 2006, 314, 980–982. [Google Scholar] [CrossRef] [PubMed]
- Sperber, G.H. Morphology of the Cheek Teeth of Early South African Hominids. Ph.D. Thesis, University of the Witwatersrand, Johannesburg, South Africa, 1974. [Google Scholar]
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sperber, G.H. Dental Wear: Attrition, Erosion, and Abrasion—A Palaeo-Odontological Approach. Dent. J. 2017, 5, 19. https://doi.org/10.3390/dj5020019
Sperber GH. Dental Wear: Attrition, Erosion, and Abrasion—A Palaeo-Odontological Approach. Dentistry Journal. 2017; 5(2):19. https://doi.org/10.3390/dj5020019
Chicago/Turabian StyleSperber, Geoffrey H. 2017. "Dental Wear: Attrition, Erosion, and Abrasion—A Palaeo-Odontological Approach" Dentistry Journal 5, no. 2: 19. https://doi.org/10.3390/dj5020019
APA StyleSperber, G. H. (2017). Dental Wear: Attrition, Erosion, and Abrasion—A Palaeo-Odontological Approach. Dentistry Journal, 5(2), 19. https://doi.org/10.3390/dj5020019



